Submitted:
01 April 2026
Posted:
03 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Ethical Standards
Study Design and Participants
Laboratory Parameters
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 25(OH)D | 25-hydroxyvitamin D |
| BMI | Body Mass Index |
| CI | Confidence Interval |
| EDTA | Ethylenediaminetetraacetic Acid |
| NHANES | National Health and Nutrition Examination Survey |
| OR | Odds Ratio |
| SIOMMMS | Italian Society for Osteoporosis, Mineral Metabolism and Skeletal Diseases |
References
- Holick, M.F. Resurrection of Vitamin D Deficiency and Rickets. Journal of Clinical Investigation 2006, 116, 2062–2072. [Google Scholar] [CrossRef]
- Adams, J.S.; Rafison, B.; Witzel, S.; Reyes, R.E.; Shieh, A.; Chun, R.; Zavala, K.; Hewison, M.; Liu, P.T. Regulation of the Extrarenal CYP27B1-Hydroxylase. The Journal of Steroid Biochemistry and Molecular Biology 2014, 144, 22–27. [Google Scholar] [CrossRef]
- Tuckey, R.C.; Cheng, C.Y.S.; Slominski, A.T. The Serum Vitamin D Metabolome: What We Know and What Is Still to Discover. The Journal of Steroid Biochemistry and Molecular Biology 2019, 186, 4–21. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Sternberg, M.R.; Lacher, D.A.; Sempos, C.T.; Looker, A.C.; Durazo-Arvizu, R.A.; Yetley, E.A.; Chaudhary-Webb, M.; Maw, K.L.; Pfeiffer, C.M.; et al. The Vitamin D Status of the US Population from 1988 to 2010 Using Standardized Serum Concentrations of 25-Hydroxyvitamin D Shows Recent Modest Increases. The American Journal of Clinical Nutrition 2016, 104, 454–461. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D Deficiency in Europe: Pandemic? The American Journal of Clinical Nutrition 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Lambrinou, C.-P.; Tsoutsoulopoulou, K.; Binou, P.; Karachaliou, A.; Breidenassel, C.; Gonzalez-Gross, M.; Kiely, M.; Cashman, K.D. A Systematic Review of Vitamin D Status in Southern European Countries. Eur J Nutr 2018, 57, 2001–2036. [Google Scholar] [CrossRef]
- Adami, S.; Viapiana, O.; Gatti, D.; Idolazzi, L.; Rossini, M. Relationship between Serum Parathyroid Hormone, Vitamin D Sufficiency, Age, and Calcium Intake. Bone 2008, 42, 267–270. [Google Scholar] [CrossRef]
- Isaia, G.; Giorgino, R.; Rini, G.B.; Bevilacqua, M.; Maugeri, D.; Adami, S. Prevalence of Hypovitaminosis D in Elderly Women in Italy: Clinical Consequences and Risk Factors. Osteoporosis International 2003, 14, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Adami, S.; Bertoldo, F.; Braga, V.; Fracassi, E.; Gatti, D.; Gandolini, G.; Minisola, S.; Battista Rini, G. 25-Hydroxy Vitamin D Levels in Healthy Premenopausal Women: Association with Bone Turnover Markers and Bone Mineral Density. Bone 2009, 45, 423–426. [Google Scholar] [CrossRef]
- Rooney, M.R.; Harnack, L.; Michos, E.D.; Ogilvie, R.P.; Sempos, C.T.; Lutsey, P.L. Trends in Use of High-Dose Vitamin D Supplements Exceeding 1000 or 4000 International Units Daily, 1999-2014. JAMA 2017, 317, 2448. [Google Scholar] [CrossRef] [PubMed]
- Bertoldo, F.; Cianferotti, L.; Di Monaco, M.; Falchetti, A.; Fassio, A.; Gatti, D.; Gennari, L.; Giannini, S.; Girasole, G.; Gonnelli, S.; et al. Definition, Assessment, and Management of Vitamin D Inadequacy: Suggestions, Recommendations, and Warnings from the Italian Society for Osteoporosis, Mineral Metabolism and Bone Diseases (SIOMMMS). Nutrients 2022, 14, 4148. [Google Scholar] [CrossRef]
- Demay, M.B.; Pittas, A.G.; Bikle, D.D.; Diab, D.L.; Kiely, M.E.; Lazaretti-Castro, M.; Lips, P.; Mitchell, D.M.; Murad, M.H.; Powers, S.; et al. Vitamin D for the Prevention of Disease: An Endocrine Society Clinical Practice Guideline. The Journal of Clinical Endocrinology & Metabolism 2024, 109, 1907–1947. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. The Journal of Clinical Endocrinology & Metabolism 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Mithal, A.; Bonjour, J.-P.; Boonen, S.; Burckhardt, P.; Fuleihan, G.E.-H.; Josse, R.G.; Lips, P.; Morales-Torres, J.; Yoshimura, N. IOF Position Statement: Vitamin D Recommendations for Older Adults. Osteoporos Int 2010, 21, 1151–1154. [Google Scholar] [CrossRef] [PubMed]
- Barchetta, I.; Baroni, M.G.; Leonetti, F.; De Bernardinis, M.; Bertoccini, L.; Fontana, M.; Mazzei, E.; Fraioli, A.; Cavallo, M.G. TSH Levels Are Associated with Vitamin D Status and Seasonality in an Adult Population of Euthyroid Adults. Clin Exp Med 2015, 15, 389–396. [Google Scholar] [CrossRef]
- Vignali, E.; Macchia, E.; Cetani, F.; Reggiardo, G.; Cianferotti, L.; Saponaro, F.; Marcocci, C. Development of an Algorithm to Predict Serum Vitamin D Levels Using a Simple Questionnaire Based on Sunlight Exposure. Endocrine 2017, 55, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Li, D.; Tian, Y.; Wang, P. Ambient Air Pollutions Are Associated with Vitamin D Status. IJERPH 2021, 18, 6887. [Google Scholar] [CrossRef]
- Kim, B.; Hwang, J.; Lee, H.; Chae, G.M.; Kim, S.; Kim, H.-S.; Park, B.; Kim, H.-J. Associations between Ambient Air Pollution, Obesity, and Serum Vitamin D Status in the General Population of Korean Adults. BMC Public Health 2022, 22, 1766. [Google Scholar] [CrossRef]
- Golastani, B.; Poursafa, P.; Zarean, M.; Yazdi, M.; Heidari-Beni, M.; Kelishadi, R. Relationship between Air Pollution and Serum Vitamin D Levels: A Systematic Review and Meta-Analysis. Adv Biomed Res 2024, 13, 96. [Google Scholar] [CrossRef]
- Bigi, A.; Ghermandi, G. Trends and Variability of Atmospheric PM2.5 and PM10–2.5 Concentration in the Po Valley, Italy. Atmos. Chem. Phys. 2016, 16, 15777–15788. [Google Scholar] [CrossRef]
- Horàlek, J.; Vlasàkovà, L.; Schreiberovà, M.; Benesovà, N.; Schneider, P.; Kurfurst, P.; Tognet, F.; Schovànkovà, J.; Vlcek, O.; Garcia Vivanco, M.; et al. Air Quality Maps of EEA Members and Cooperating Countries for 2021; European Topic Centre on Human Health and the Environment, 2024. [Google Scholar]
- Bortolus, R.; Filippini, F.; Udali, S.; Rinaldi, M.; Genesini, S.; Gandini, G.; Montagnana, M.; Chiaffarino, F.; Lippi, G.; Pattini, P.; et al. B Vitamin Blood Concentrations and One-Carbon Metabolism Polymorphisms in a Sample of Italian Women and Men Attending a Unit of Transfusion Medicine: A Cross-Sectional Study. Eur J Nutr 2021, 60, 2643–2654. [Google Scholar] [CrossRef]
- Bortolus, R.; Filippini, F.; Chiaffarino, F.; Udali, S.; Rinaldi, M.; Gandini, G.; Montagnana, M.; Lippi, G.; Olivieri, O.; Parazzini, F.; et al. Lifestyle and Dietary Factors, Iron Status and One-Carbon Metabolism Polymorphisms in a Sample of Italian Women and Men Attending a Transfusion Medicine Unit: A Cross-Sectional Study. Br J Nutr 2023, 130, 65–70. [Google Scholar] [CrossRef]
- Oskarsson, V.; Eliasson, M.; Salomaa, V.; Reinikainen, J.; Männistö, S.; Palmieri, L.; Donfrancesco, C.; Sans, S.; Costanzo, S.; De Gaetano, G.; et al. Influence of Geographical Latitude on Vitamin D Status: Cross-Sectional Results from the BiomarCaRE Consortium. Br J Nutr 2022, 128, 2208–2218. [Google Scholar] [CrossRef]
- Hyppönen, E.; Power, C. Hypovitaminosis D in British Adults at Age 45 y: Nationwide Cohort Study of Dietary and Lifestyle Predictors. The American Journal of Clinical Nutrition 2007, 85, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Karacan, M.; Usta, A.; Biçer, S.; Baktir, G.; İpek Gündogan, G.; Sancakli Usta, C.; Akinci, G. Serum Vitamin D Levels in Healthy Urban Population at Reproductive Age: Effects of Age, Gender and Season. Cent Eur J Public Health 2020, 28, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Maggio, D.; Cherubini, A.; Lauretani, F.; Russo, R.C.; Bartali, B.; Pierandrei, M.; Ruggiero, C.; Macchiarulo, M.C.; Giorgino, R.; Minisola, S.; et al. 25(OH)D Serum Levels Decline With Age Earlier in Women Than in Men and Less Efficiently Prevent Compensatory Hyperparathyroidism in Older Adults. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 2005, 60, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Basile, M.; Ciardi, L.; Crespi, I.; Saliva, E.; Bellomo, G.; Vidali, M. ASSESSING SERUM CONCENTRATIONS OF 25-HYDROXY-VITAMIN D IN NORTH-WESTERN ITALY. J Frailty Aging 2013, 1–5. [Google Scholar] [CrossRef]
- Bettica, P.; Bevilacqua, M.; Vago, T.; Norbiato, G. High Prevalence of Hypovitaminosis D among Free-Living Postmenopausal Women Referred to an Osteoporosis Outpatient Clinic in Northern Italy for Initial Screening. Osteoporosis International 1999, 9, 226–229. [Google Scholar] [CrossRef]
- Rossini, M.; Alberti, V.; Flor, L.; Masiero, L.; Giannini, S.; Gatti, D.; Adami, S. Effect of Oral Vitamin D2 Yearly Bolus on Hip Fracture Risk in Elderly Women: A Community Primary Prevention Study. Aging Clin Exp Res 2004, 16, 432–436. [Google Scholar] [CrossRef]
- Samuel, L.; Borrell, L.N. The Effect of Body Mass Index on Adequacy of Serum 25-Hydroxyvitamin D Levels in US Adults: The National Health and Nutrition Examination Survey 2001 to 2006. Annals of Epidemiology 2014, 24, 781–784. [Google Scholar] [CrossRef]
- Pereira-Santos, M.; Costa, P.R.F.; Assis, A.M.O.; Santos, C.A.S.T.; Santos, D.B. Obesity and Vitamin D Deficiency: A Systematic Review and Meta-analysis. Obesity Reviews 2015, 16, 341–349. [Google Scholar] [CrossRef]
- Marzban, M.; Kalantarhormozi, M.; Mahmudpour, M.; Ostovar, A.; Keshmiri, S.; Darabi, A.H.; Khajeian, A.; Bolkheir, A.; Amini, A.; Nabipour, I. Prevalence of Vitamin D Deficiency and Its Associated Risk Factors among Rural Population of the Northern Part of the Persian Gulf. BMC Endocr Disord 2021, 21, 219. [Google Scholar] [CrossRef]
- Saliba, W.; Barnett-Griness, O.; Rennert, G. The Relationship between Obesity and the Increase in Serum 25(OH)D Levels in Response to Vitamin D Supplementation. Osteoporos Int 2013, 24, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Capuano, R.; Marchese, F.; Sica, R.; Capuano, E.; Manilia, M.; Iannone, A.G.; D’Ambrosio, A.; Bisecco, A.; Tedeschi, G.; Gallo, A.; et al. Epidemiologic Data of Vitamin D Deficiency and Its Implication in Cardio-Cerebrovascular Risk in a Southern Italian Population. Journal of Nutrition and Metabolism 2021, 2021, 1–8. [Google Scholar] [CrossRef]
- Gammone, M.; Danese, A.; D’Orazio, N. Prevalence of 25(OH)D Insufficiency and Overweight/Obesity in an Adult Population from the Central Italy. LA CLINICA TERAPEUTICA 2022, 334–341. [Google Scholar] [CrossRef]
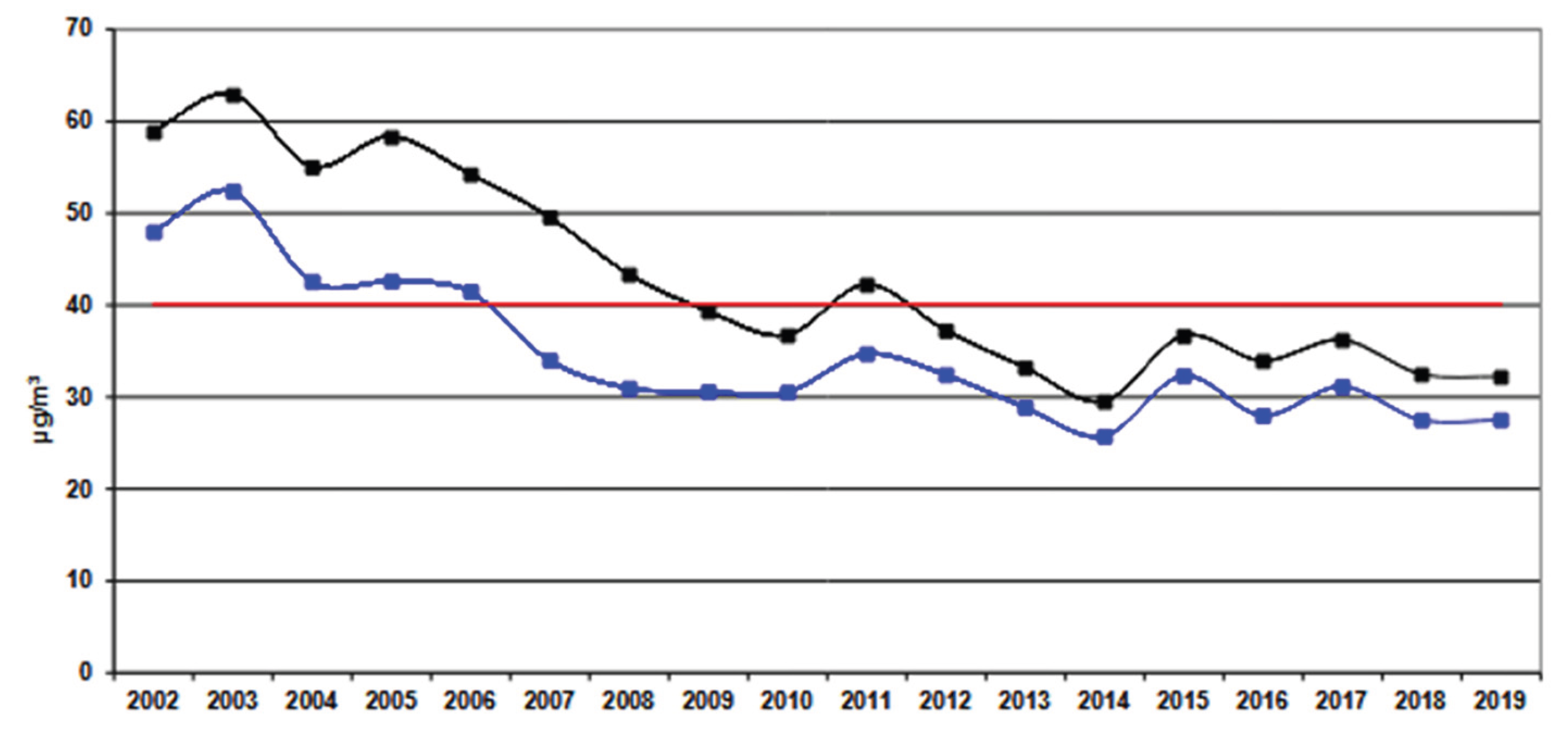
- Agenzia Regionale per la Prevenzione e Protezione Regionale del Veneto Rapporto sullo stato dell’ambiente del Veneto - Anno 2020. 2020. Available online: https://pfas.land/wp-content/uploads/2021/01/rapporto_stato_ambiente_veneto_2020.pdf (accessed on 4th March 2026).
- Saneei, P.; Salehi-Abargouei, A.; Esmaillzadeh, A. Serum 25-hydroxy Vitamin D Levels in Relation to Body Mass Index: A Systematic Review and Meta-analysis. Obesity Reviews 2013, 14, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B.; Konieczynski, E.M.; Ceglia, L. Obesity May Extend the Time Required to Reach a Steady-State 25(OH)D Level after Initiating Vitamin D Supplementation. JBMR Plus 2025, 9, ziaf030. [Google Scholar] [CrossRef]
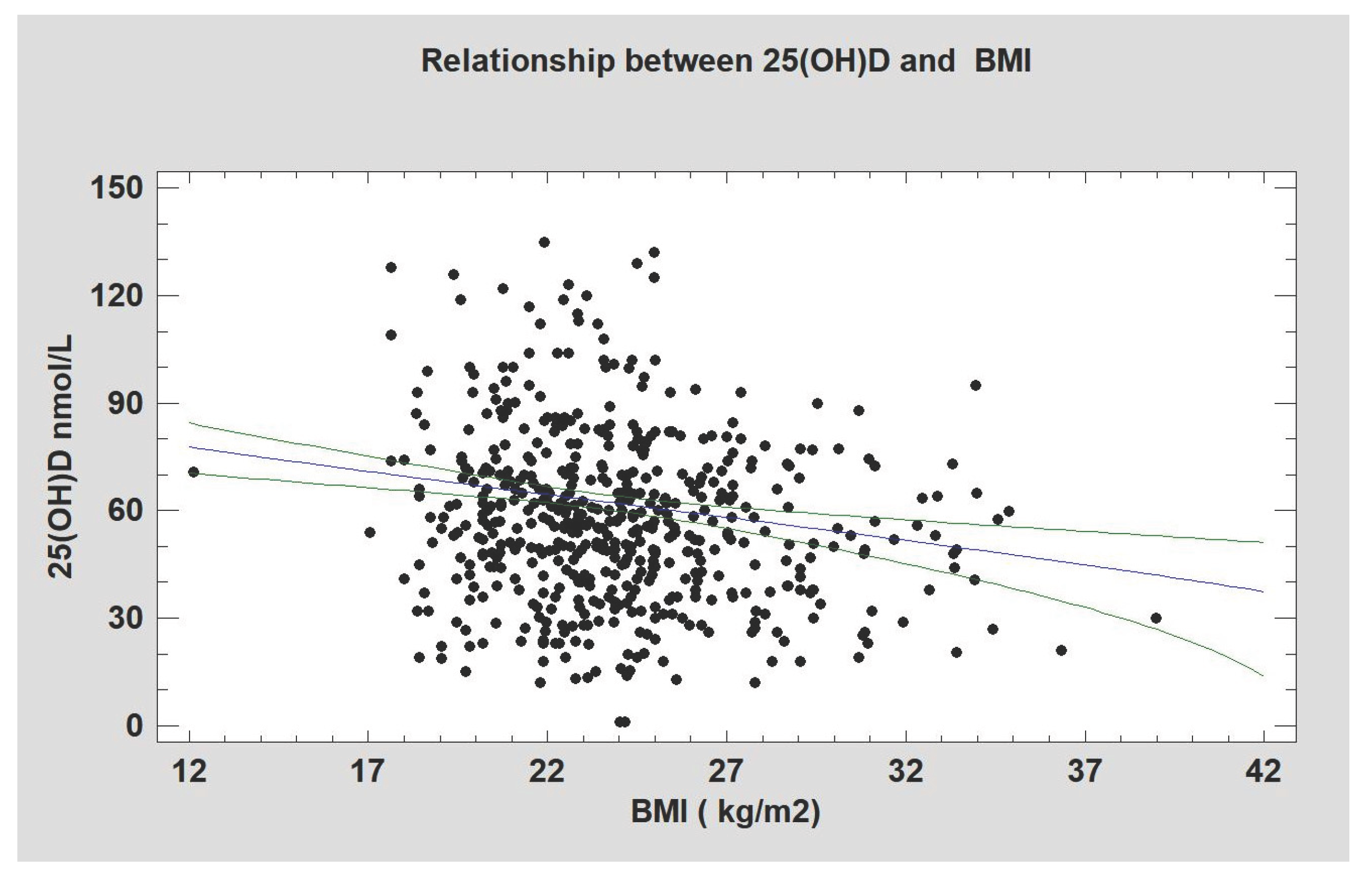
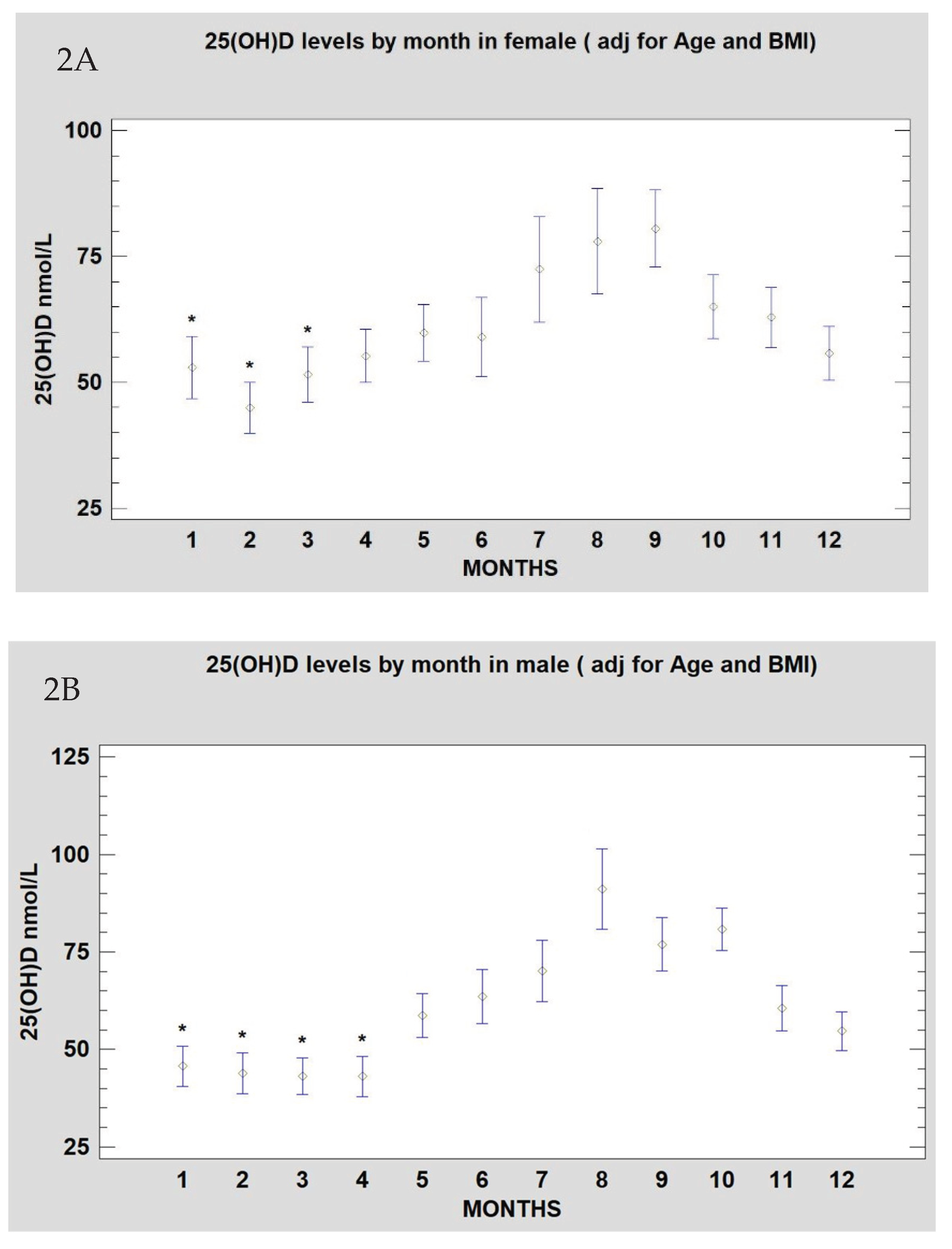
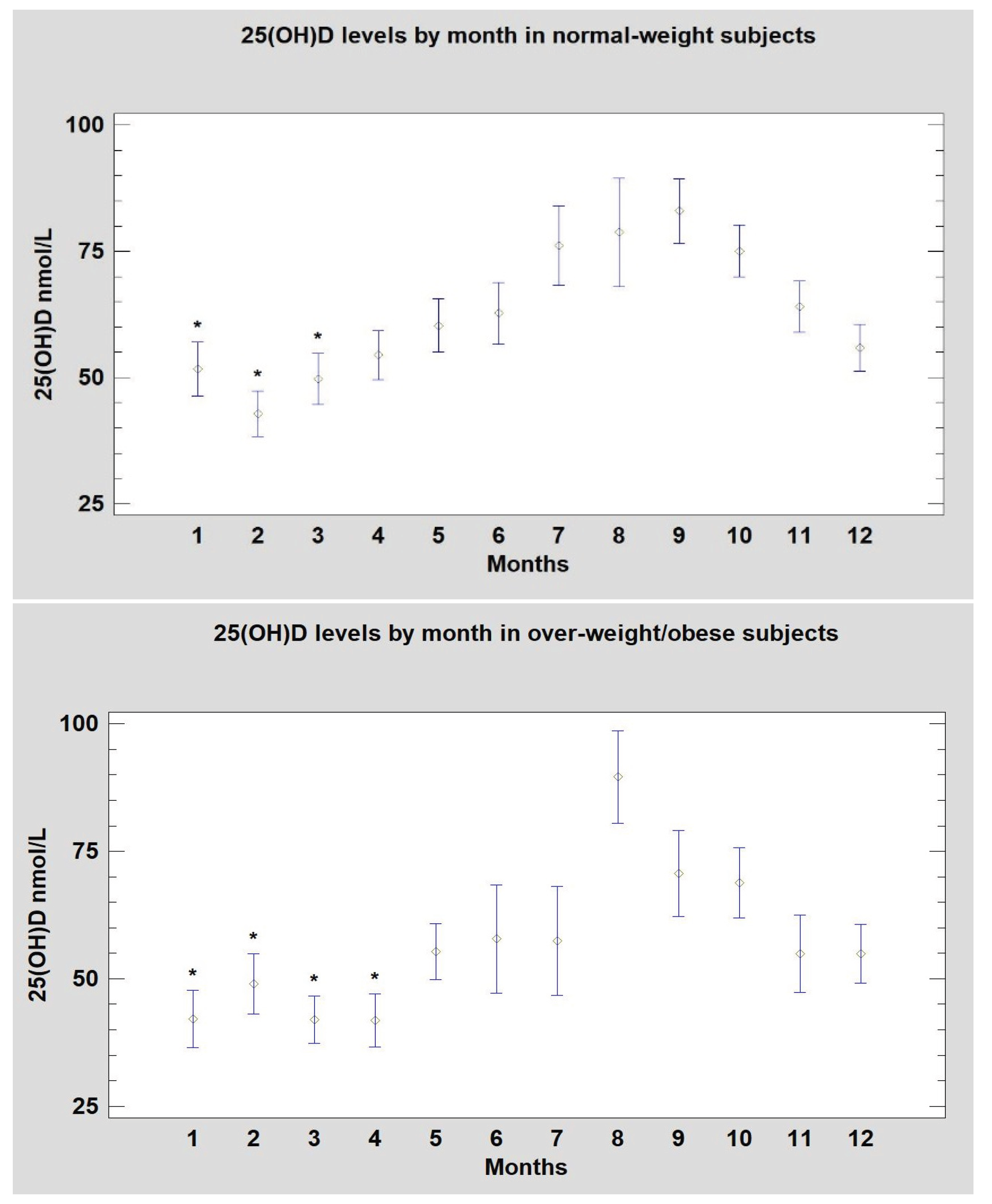
| All | Female | Male | ANOVA p value Between genders |
|
| n | 534 | 266 | 268 | |
| Age | 42.0 (40.9-43.0) | 38.0 (37.0-39.0) | 46.0 (45.0-46.9) | 0.001 |
| BMI | 23.9 (23.6-24.2) | 22.6 (22.3-22.9) | 25.2 (25.0-25.5) | 0.001 |
| 25(OH)D nmol/L | 57.0 (55.0-65.8) | 57.5 (55.5-59.5) | 56.7 (54.5-58.6) | 0.622 |
| 25(OH)D nmol/L # | 56.8 (54.8-60.3) | 56.0 (53.0-58.8) | 58.3 (55.3-61.0) | 0.456 |
| Spring # | 54.5 (50.8-58.3) | 56.8 (51.5-62.3)* | 52.0 (46.5-58.0)a | 0.446 |
| Summer # | 68.8 (63.5-74.0) | 66.8 (58.5-73.0)** | 70.3 (63.5-77.0) b | 0.919 |
| Autumn # | 70.0 (66.3-73.5) | 67.5 (62.0-73.0)** | 72.3 (67.3-77.3)b | 0.190 |
| Winter # | 48.3 (45.5-51.0) | 50.3 (46.5-54.3) | 46.3 (42.5-49.8) | 0.284 |
| 25(OH)D status^ | Chi-square | |||
| Insufficiency (<25 nmol/L) |
7.1% (38) | 6.8% (18) | 9.7% (26) | |
| Deficiency (25-49 nmol/L) |
31.1% (166) | 33.1% (88) | 29.9% (80) | 0.399 |
| Sufficiency (≥ 50 nmol/L) |
61.8% (330) | 60.1% (160) | 60.4% (162) |
| 25(OH)D <50 nmol/L |
|||
|---|---|---|---|
| N TOT | N (%) | OR (95% CI) | |
| Education (y) | |||
| < 8 | 82 | 37 (18.1) | 1+ |
| 9-15 | 325 | 120 (58.8) | 0.9 (0.55-1.62) |
| > 16 | 127 | 47 (23.1) | 0.9 (0.50-1.81) |
| Current smoker | |||
| No | 459 | 173 (84.8) | 1+ |
| Yes | 75 | 31 (15.2) | 1.2 (0.69-1.96) |
| Alcohol drinking | |||
| No | 167 | 60 (29.4) | 1+ |
| Yes | 370 | 144 (70.6) | 0.7 (0.44-1.11) |
| Fruit and Vegetable | |||
| 1 portion/day | 57 | 28 (13.7) | 1+ |
| 2 portions/day | 396 | 154 (75.5) | 1.0 (0.68-1.62) |
| 3 portions/day | 81 | 22 (10.8) | 0.8 (0.44-1.31) |
| Physical activity | |||
| < 2 hours/week | 215 | 101 (49.5) | 1+ |
| 2-5 hours/week | 213 | 67 (32.8) | 0.6 (0.39-0.91) |
| > 5 hours/week | 106 | 36 (17.7) | 0.6 (0.35-1.00) |
| 25(OH)D levels at NADIR | All (65) | FEMALE (32) | MALE (33) |
|---|---|---|---|
| Insufficiency % (n) | 27.7 (18) | 21.9 (7) | 33.3 (11) |
| Deficiency % (n) | 30.8 (20) | 37.5 (12) | 24.3 (8) |
| Sufficiency % (n) | 41.5 (27) | 40.6 (13) | 42.4 (14) |
| Normal weight (370) | Overweight/Obese (164) |
p value | |
|---|---|---|---|
| F/M | 213/157, F 57.6% | 53/111, F 32.3% | |
| Age | 41.0 (40.2-41-9) | 44.1 (42.8-45.4) | 0.002 |
| BMI kg/m2 | 22.2 (22.0-22.3) | 28.0 (27.7-28-2) | 0.001 |
| 25(OH)D nmol/L | 59.3 (57.7-61.0) | 52.0 (49.5-54.5) | 0.002 |
| Insufficiency % (n) | 7.3 (27) | 10.3 (17)a | |
| Deficiency % (n) | 29.2 (108) | 36.6 (60)a | |
| Sufficiency % (n) | 63.5 (235) | 53.0 (87)a | |
| Nadir 25(OH)D (nmol/L) | 42.5 (40.0-44.8) | 41.3 (35.0-47.5) | 0.337 |
| Insufficiency (% at nadir) | 4.2 (3/71) | 10.0 (4/40) b | |
| Deficiency (% at nadir) | 33.8 (24/71) | 47.5 (19/40)b | |
| Sufficiency (% at nadir) | 62.0 (44/71) | 42.5 (17/40)b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




