Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Protocol
2.3. Data Acquisition
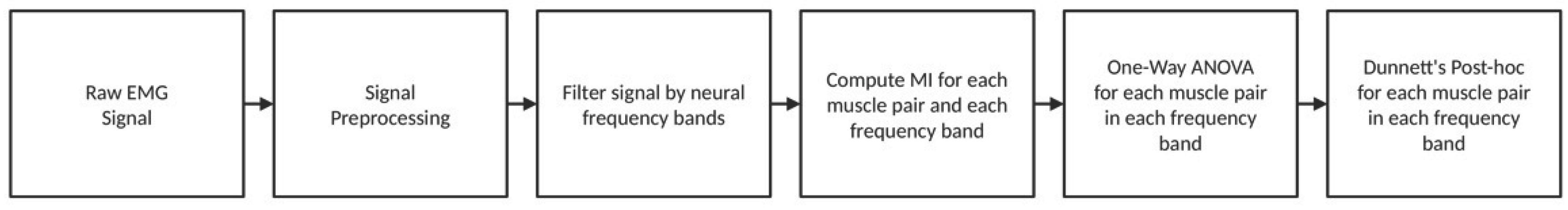
2.4. Data Analysis
2.4.1. Signal Preprocessing
2.4.2. Estimation of MI
2.5. Statistical Analysis
3. Results
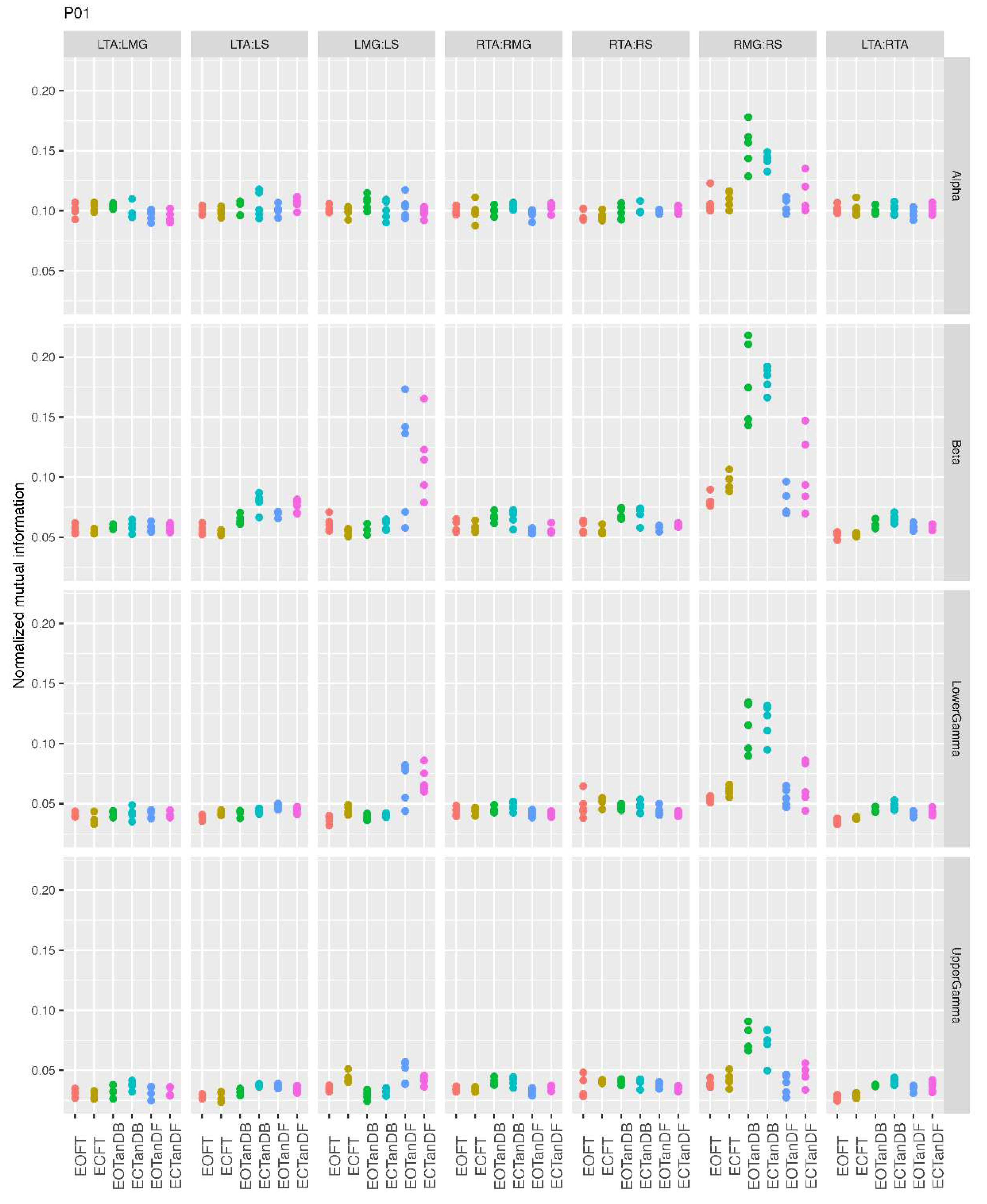
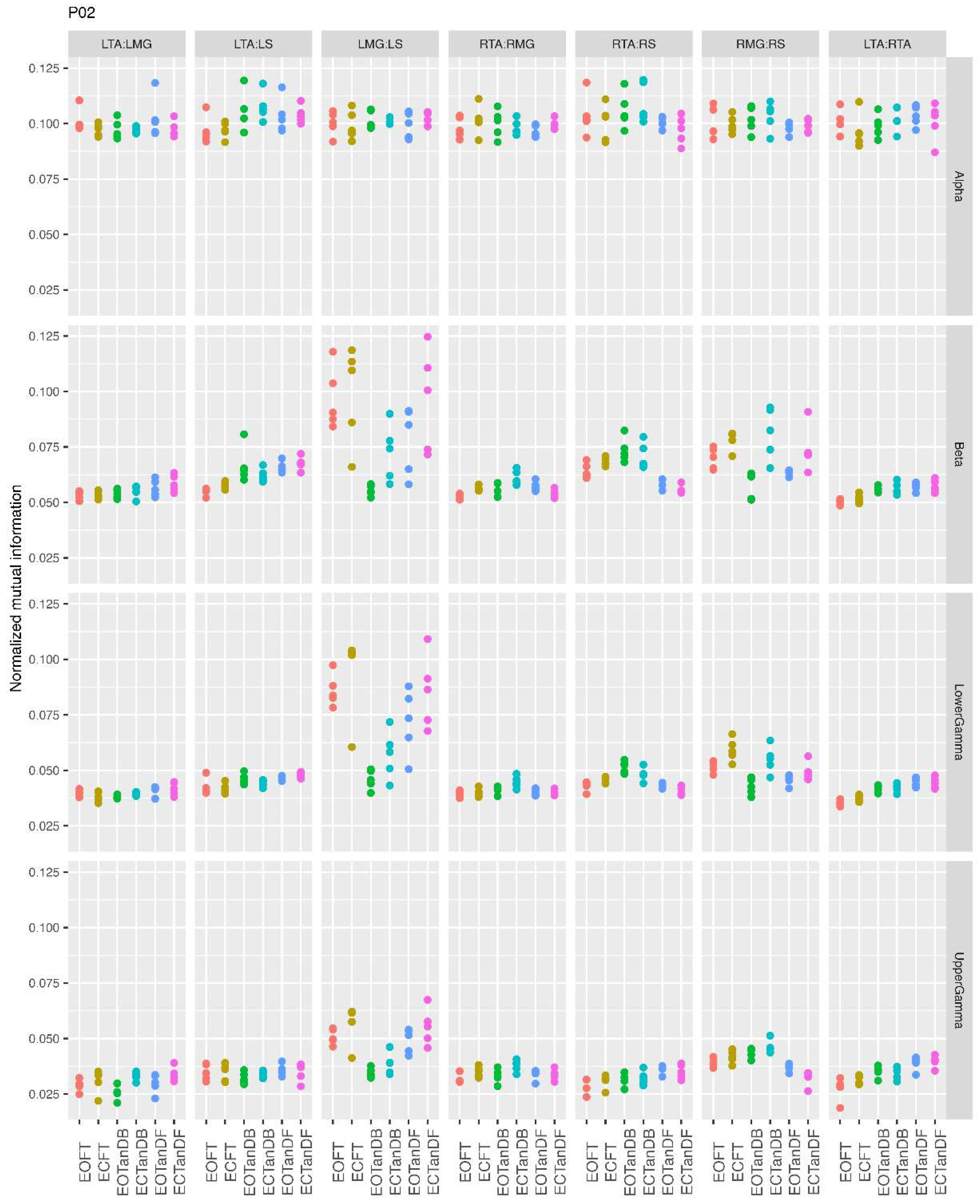
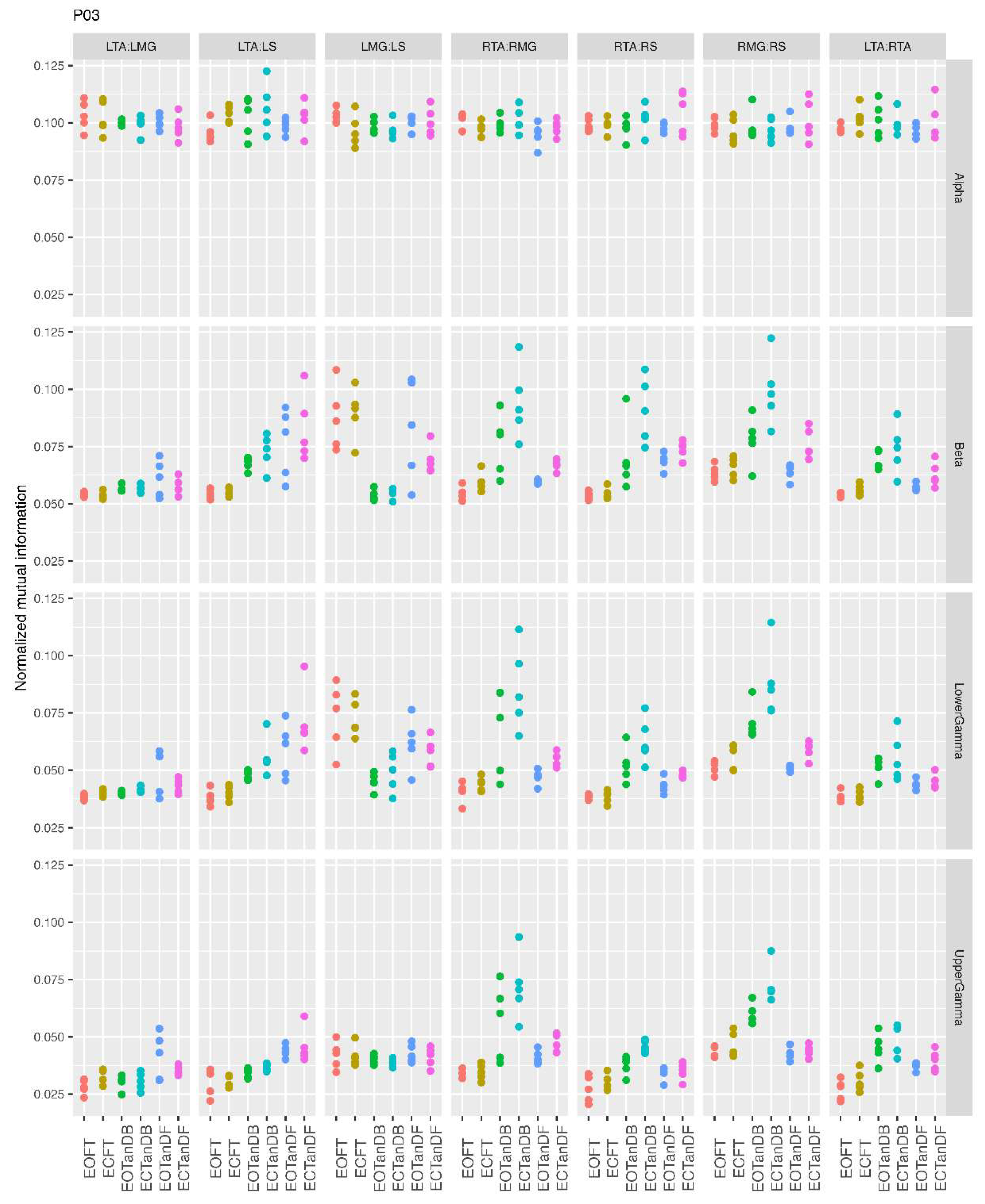
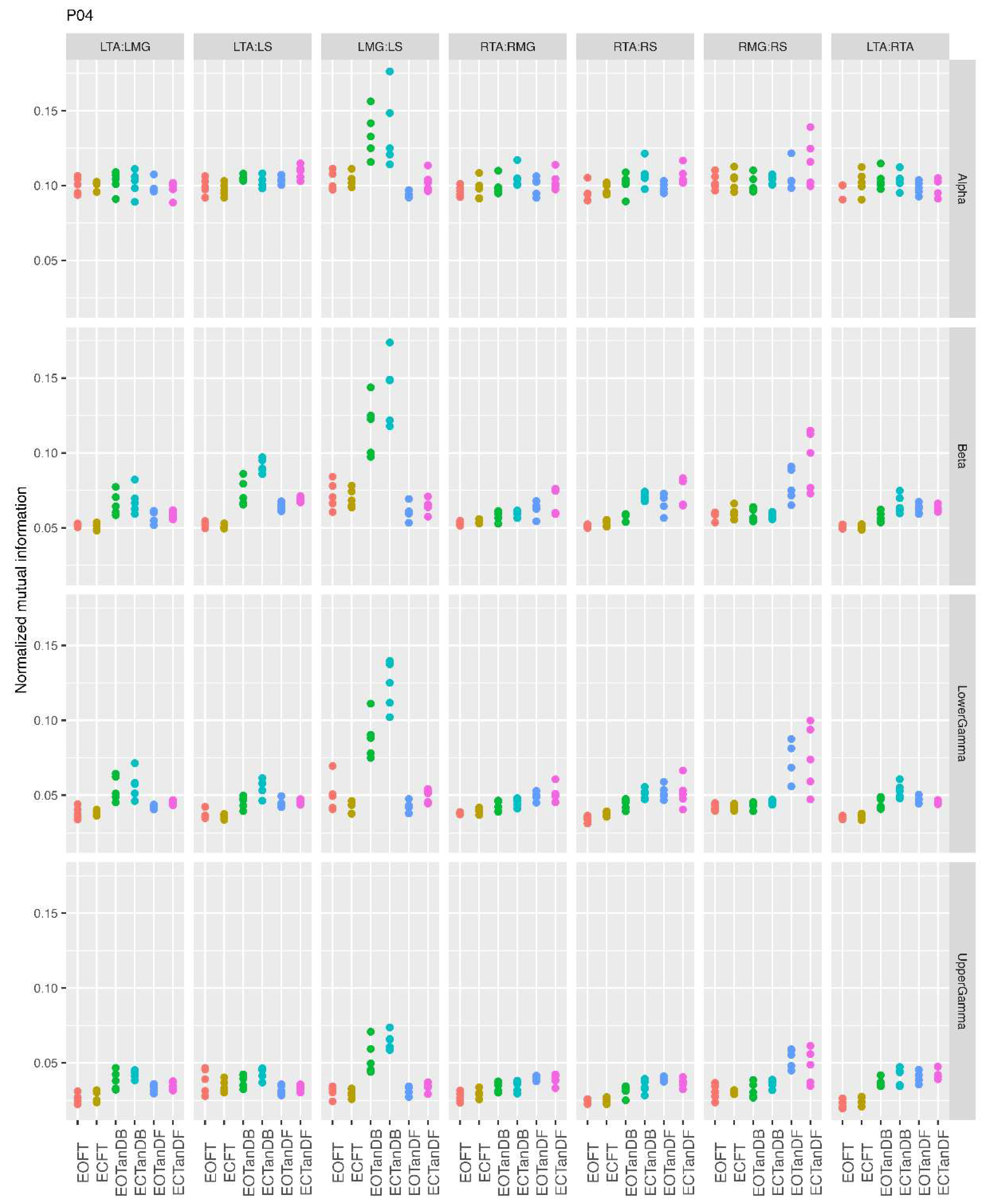
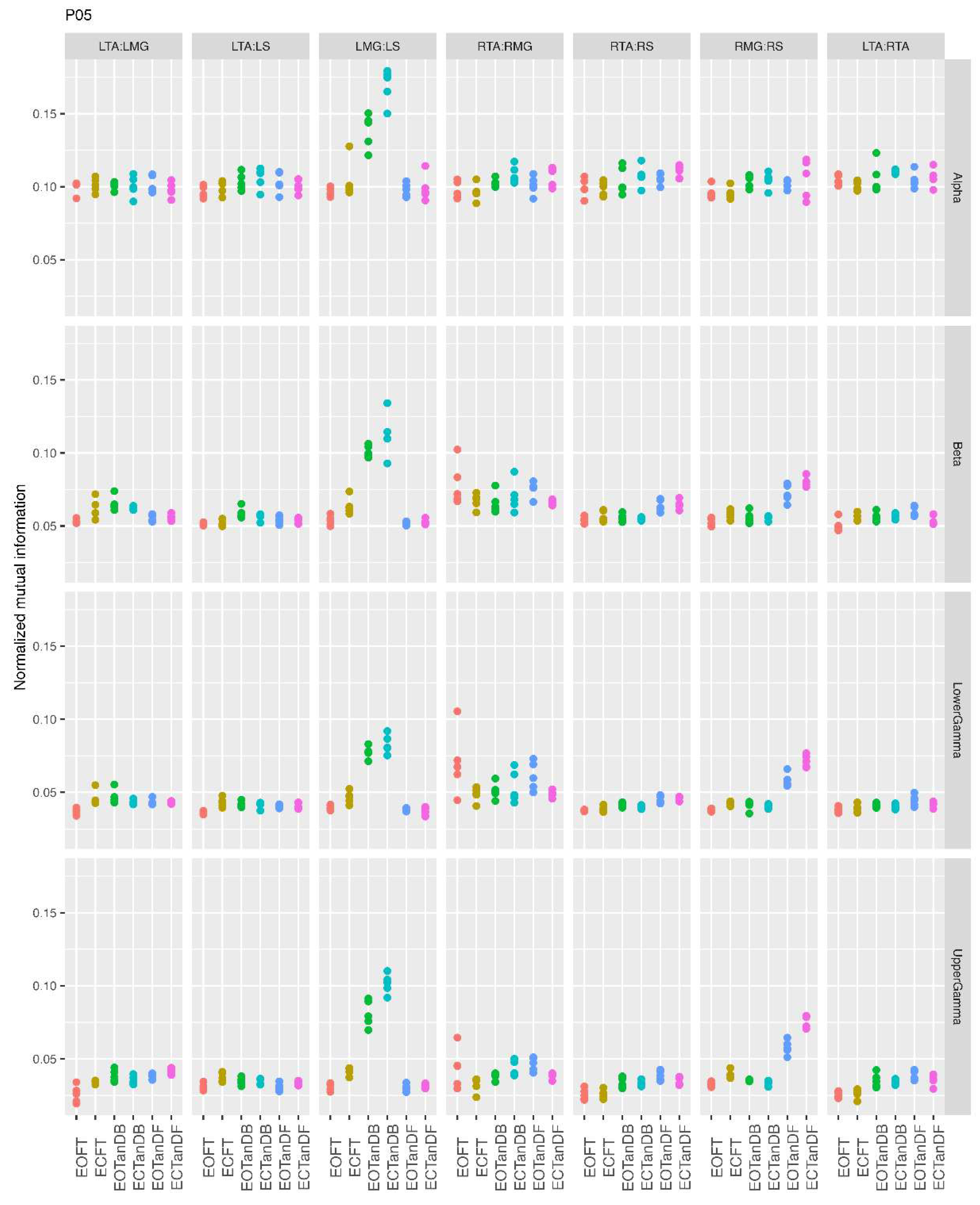
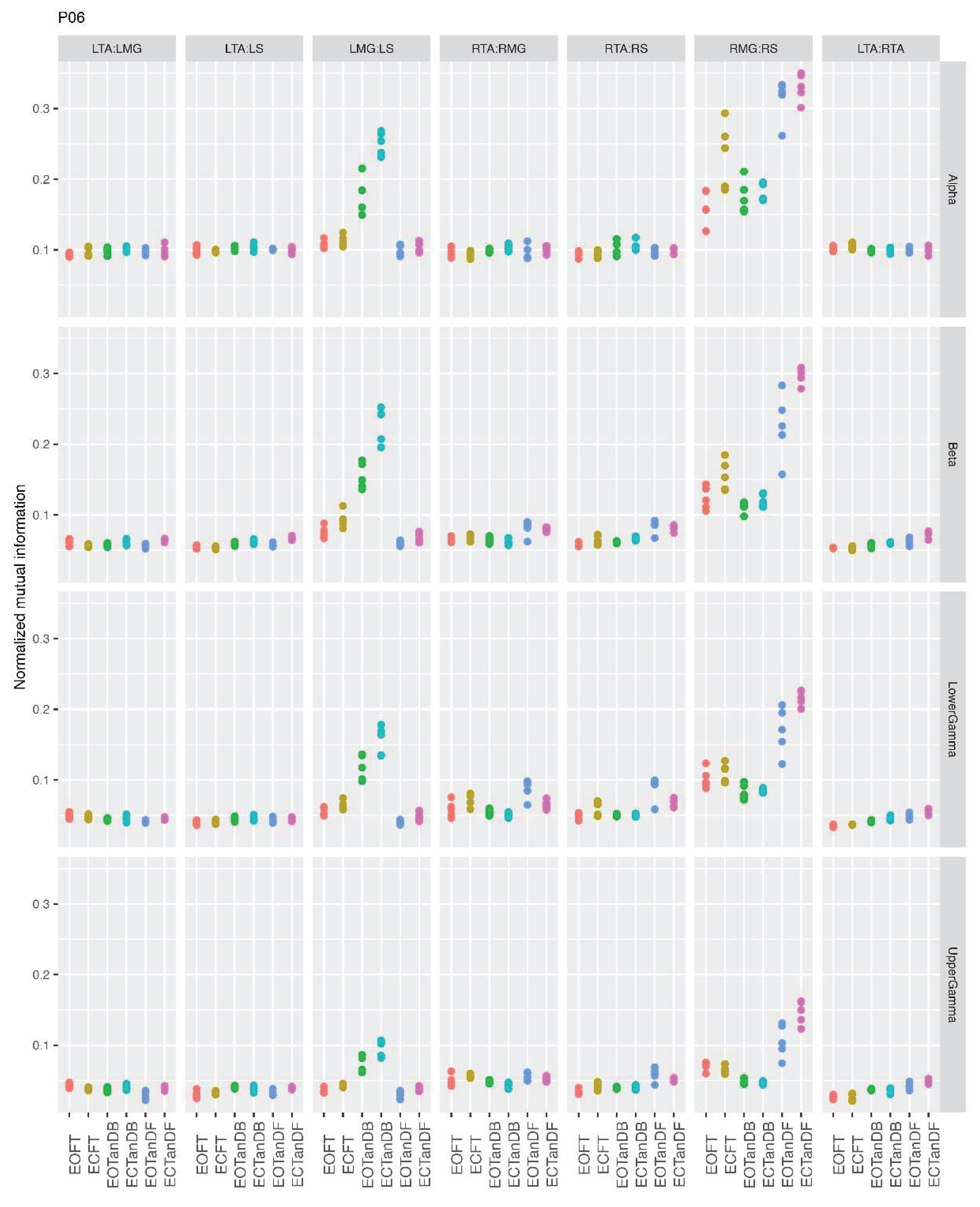
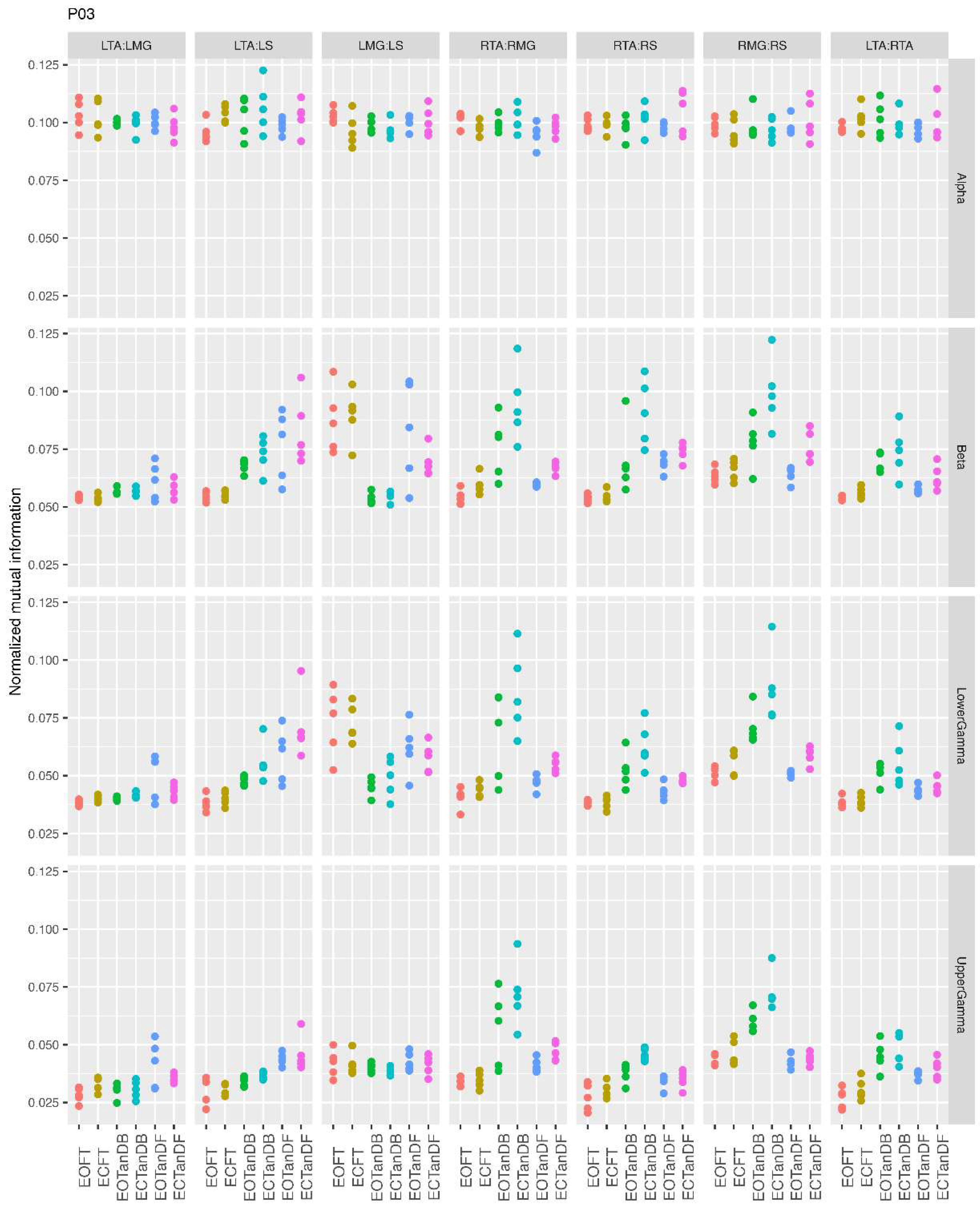
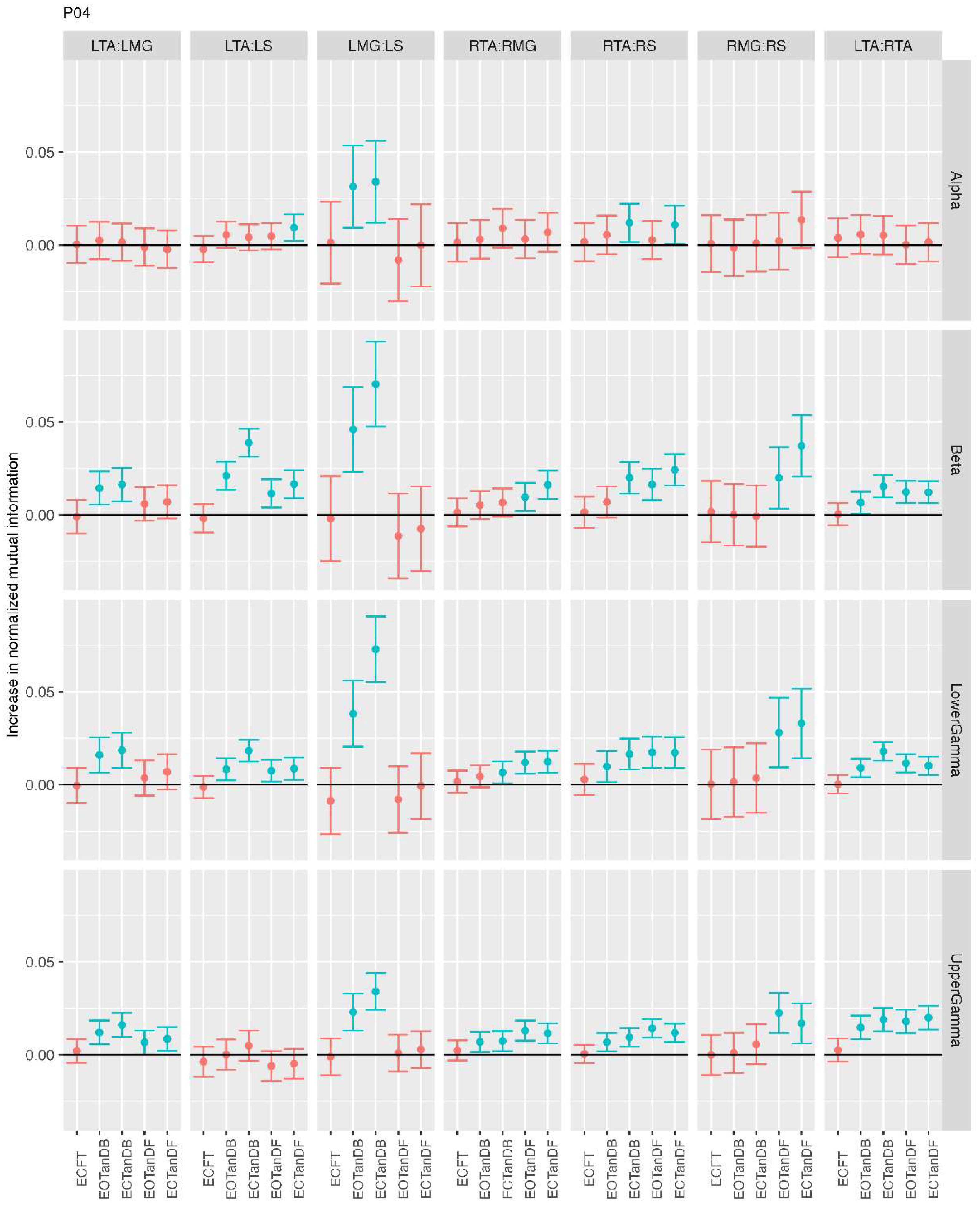
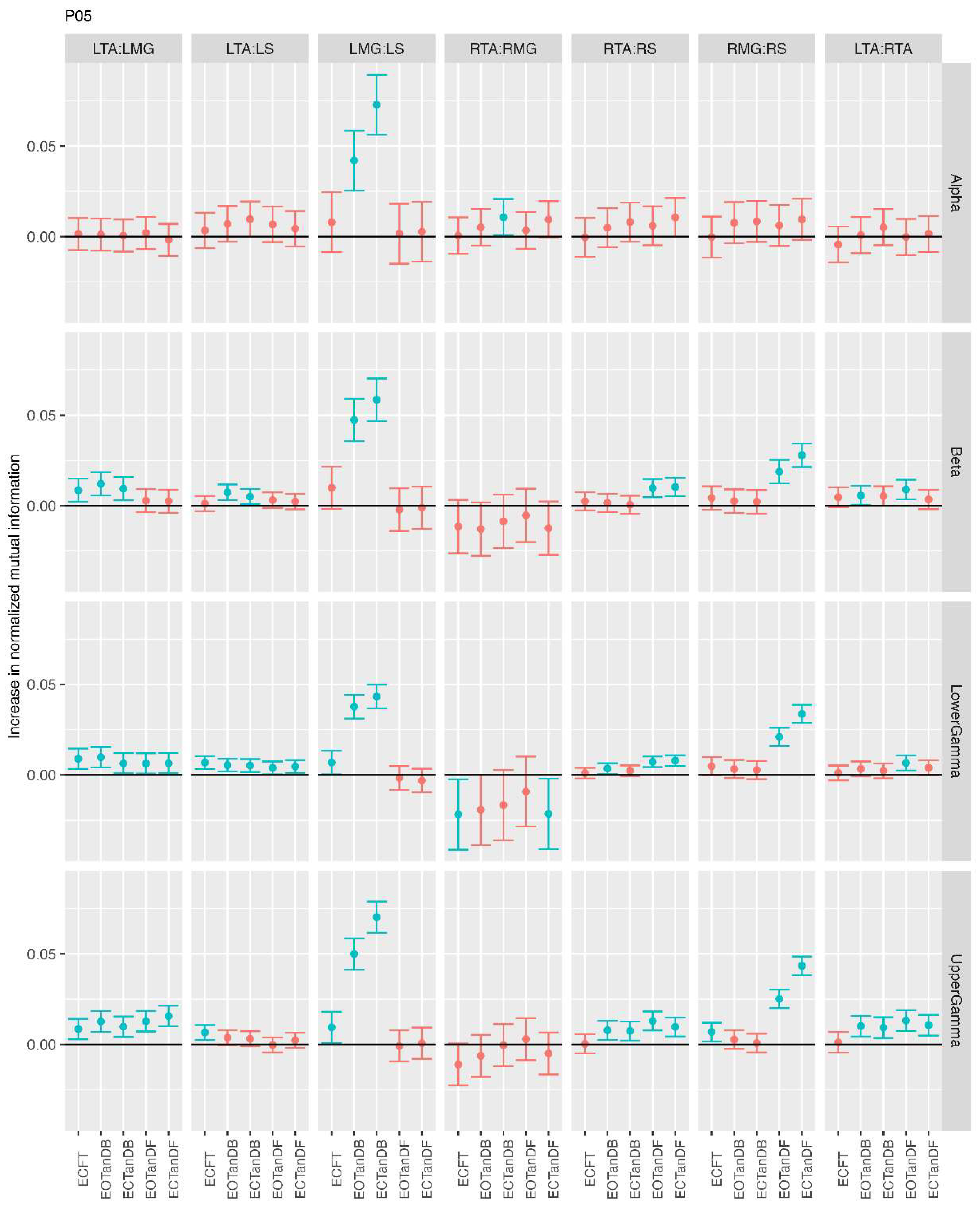
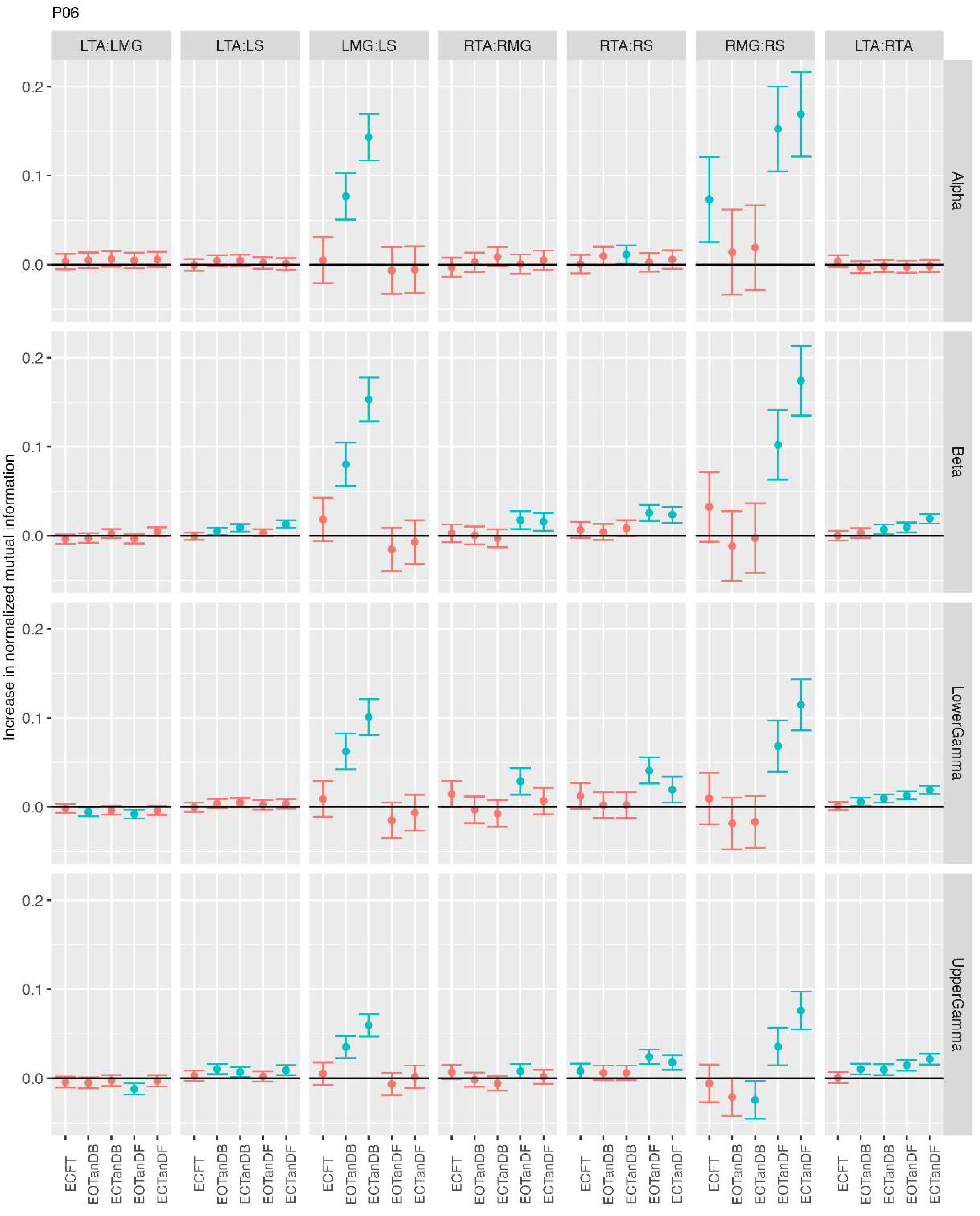
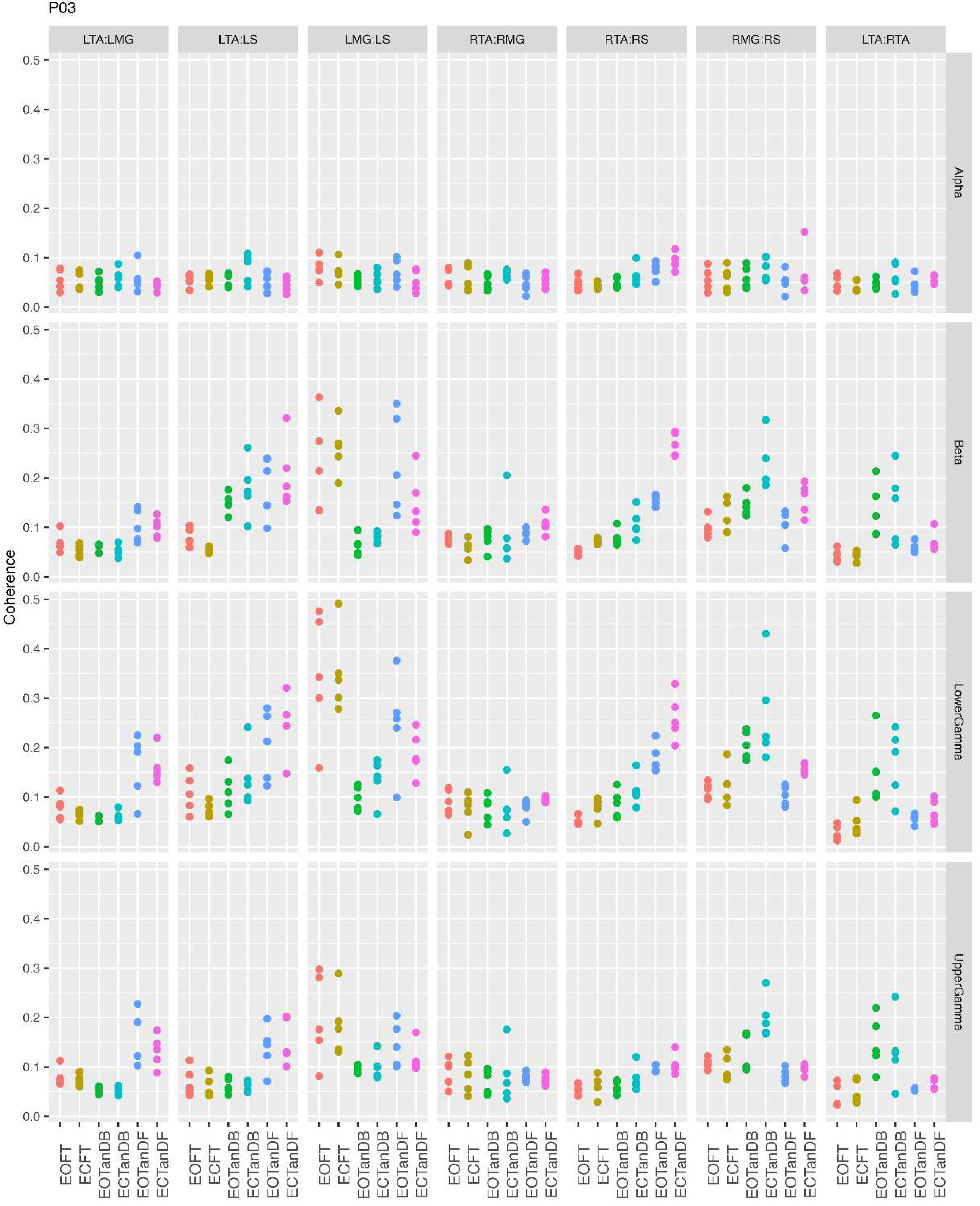
3.1. Overview of MI Across Participants
- Greater normalized MI between muscle pairs in the tandem stance compared to the feet together positions in the beta, lower gamma, and upper gamma frequency bands;
- Consistently greater normalized MI in the LMG:LS and RMG:RS in the beta, lower gamma, and upper gamma frequency bands for tandem stance positions under eyes open and eyes closed conditions;
- Evidence of MI between antagonistic muscle pairs, particularly LTA:LS, RTA:RMG, and RTA:RS; and
- No distinguishable MI patterns in the tandem standing positions with the dominant leg positioned back or forward.
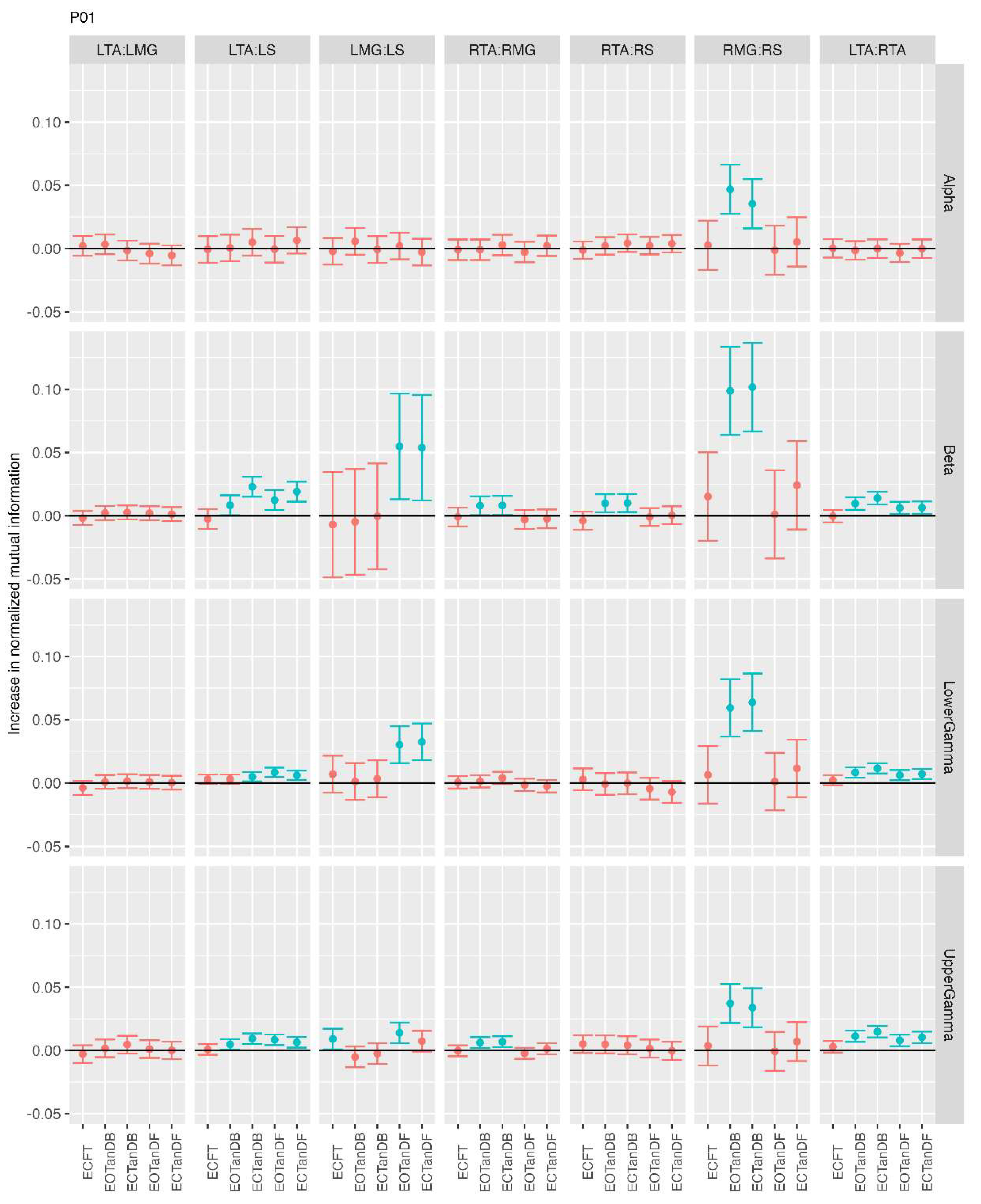
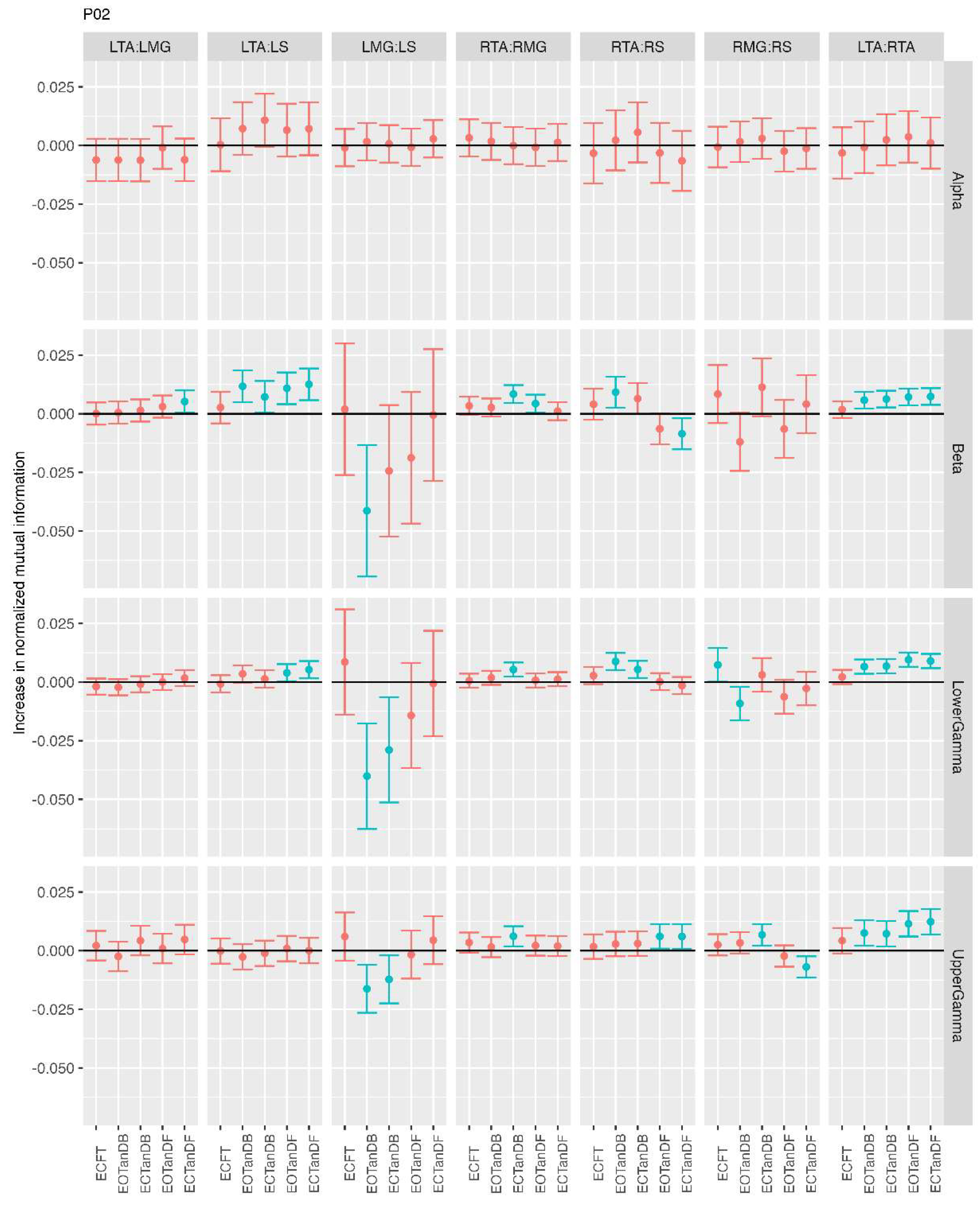
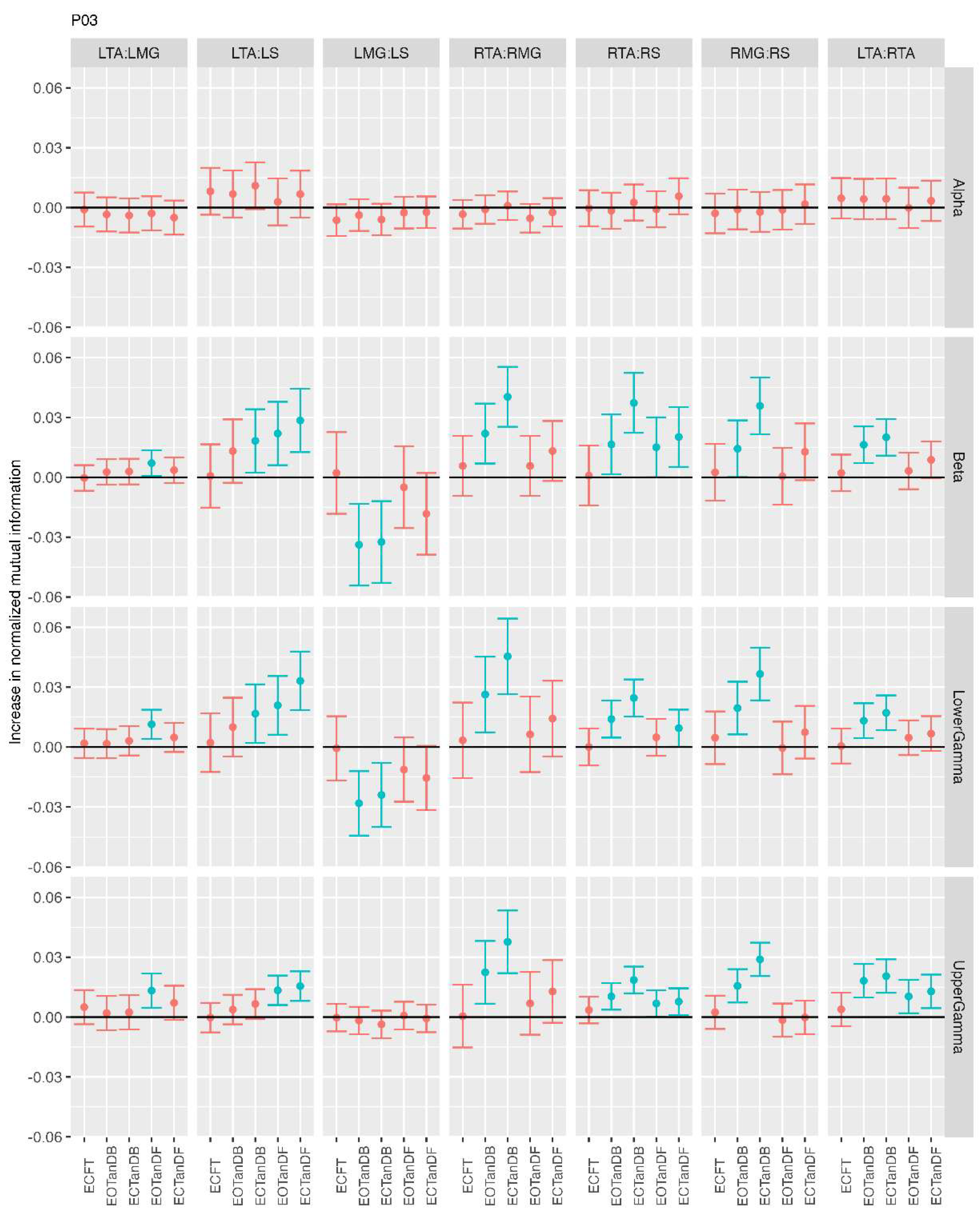
3.2. Comparison of MI Across Conditions
- More significant differences in MI between tandem standing and the baseline standing positions in the beta, lower gamma, and upper gamma frequency bands;
- Greater normalized MI in tandem standing for select muscle pairs, but particularly lower leg synergists, i.e., medial gastrocnemius/soleus;
- Greater normalized MI in the left tibialis anterior/right tibialis anterior in tandem standing, consistently across the beta, lower gamma, and upper gamma frequency bands.
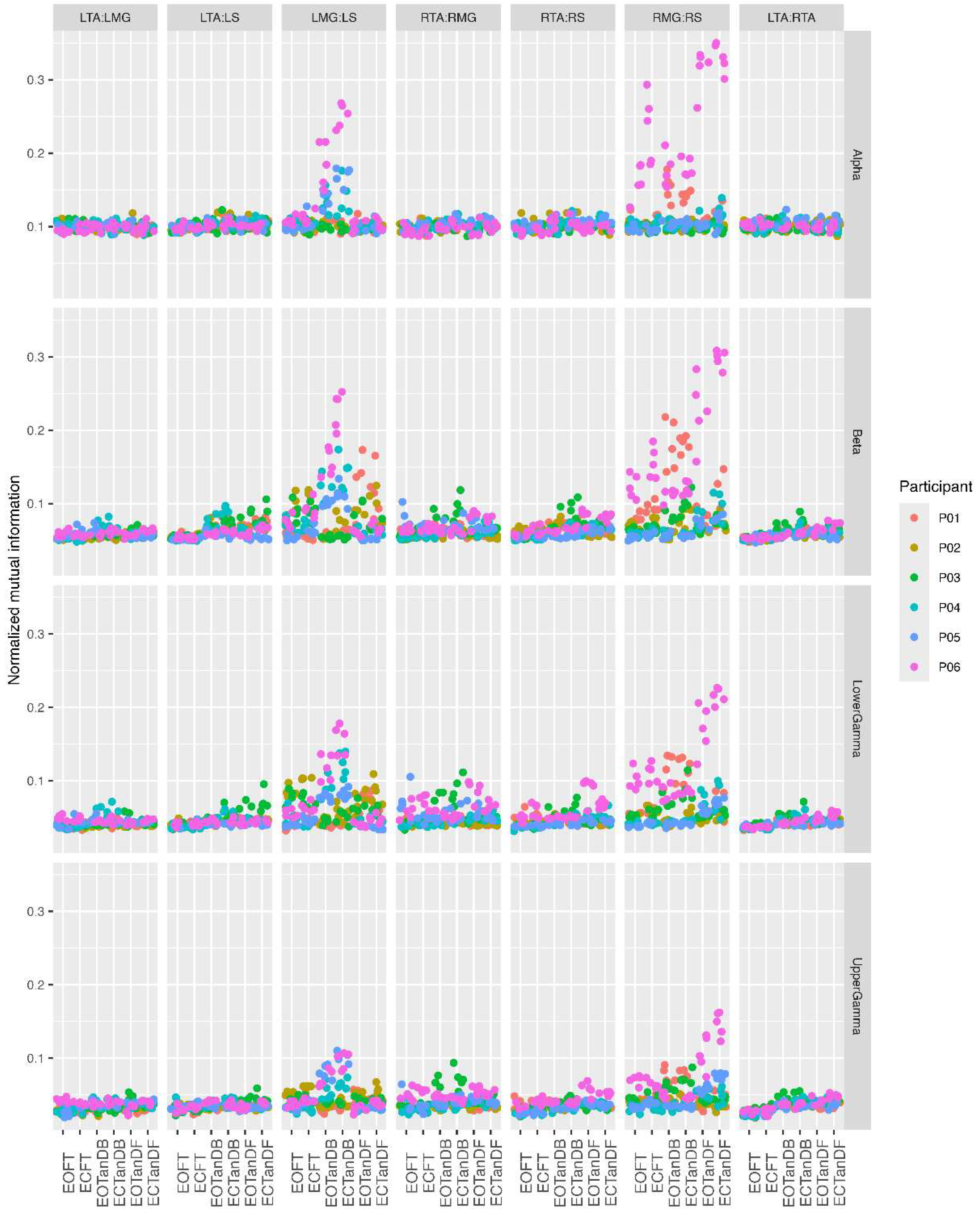
3.3. Inter-Trial and Subject Variability in Normalized MI
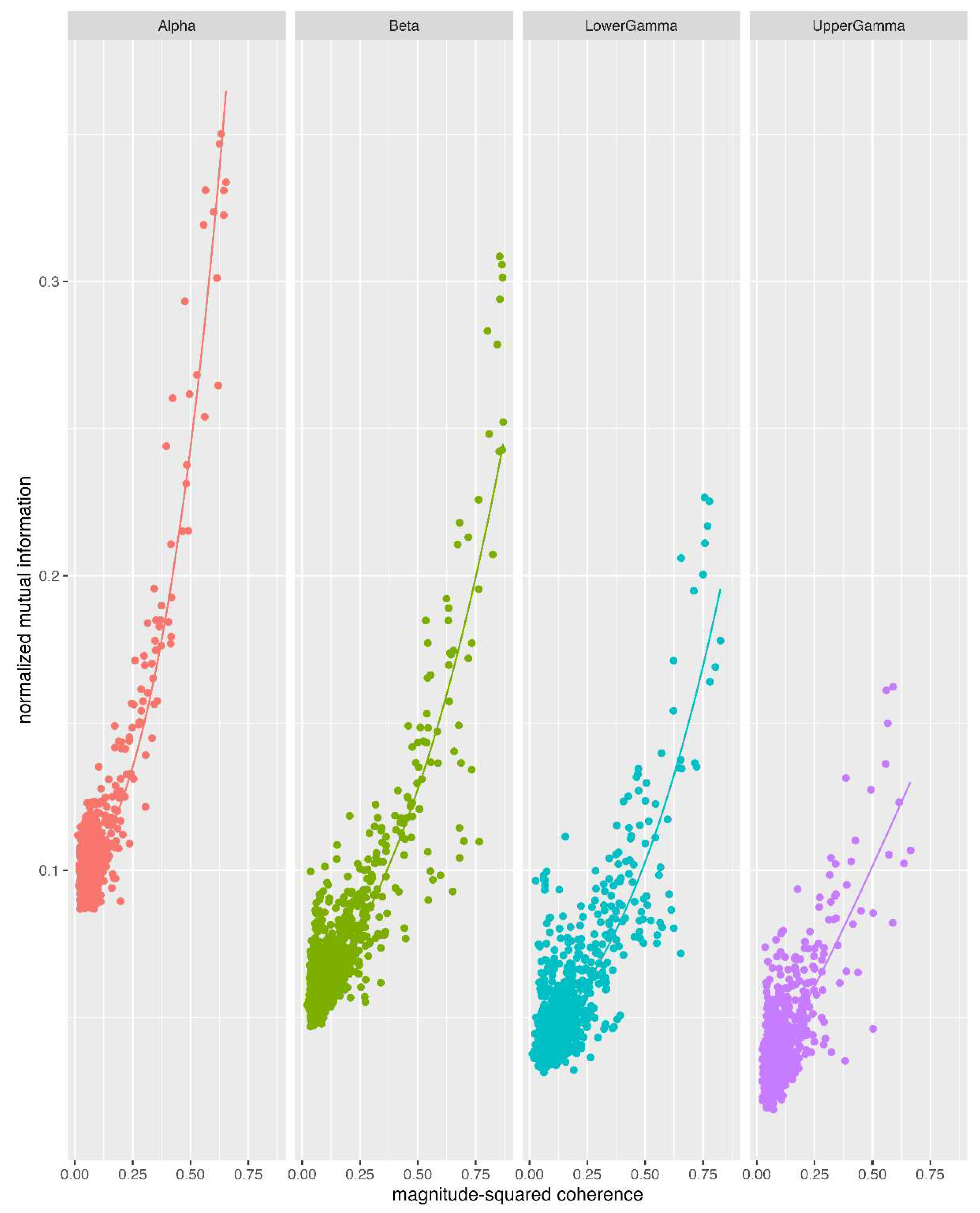
3.4. Relationship Between MI and Magnitude-Sqaured Coherence
4. Discussion
4.1. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
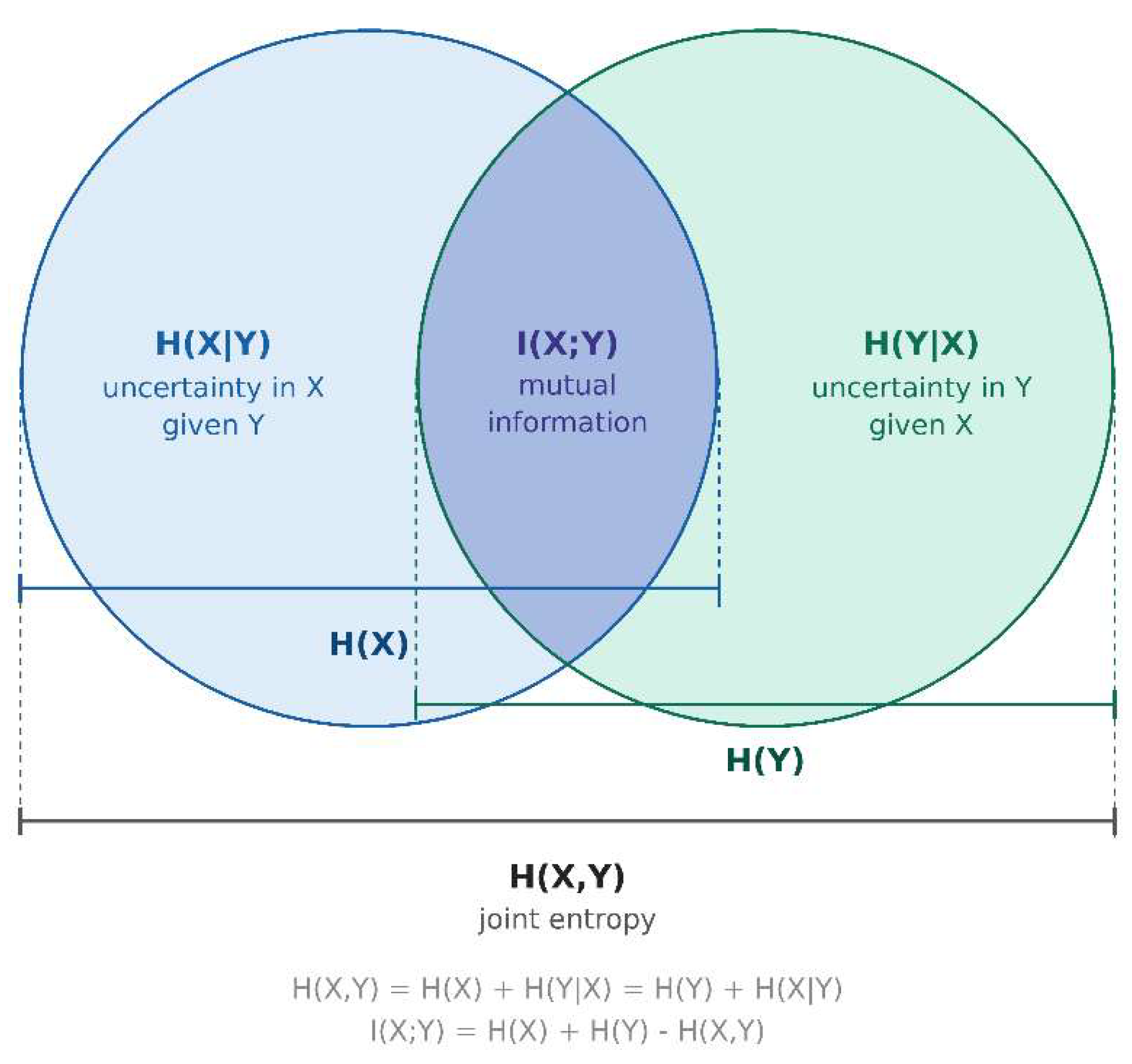
Appendix A. Mutual Information Theory
Appendix B
References
- Roman-Liu, D. Age-related changes in the range and velocity of postural sway. Arch Gerontol Geriatr. 2018, 77, 68–80. [Google Scholar] [CrossRef]
- Lamont, R.M.; Morris, M.E.; Menz, H.B.; McGinley, J.L.; Brauer, S.G. Falls in people with Parkinson’s disease: a prospective comparison of community and home-based falls. Gait Posture 2017, 55, 62–67. [Google Scholar] [CrossRef]
- De Lima, F.; Melo, G.; Fernandes, D.A.; Santos, G.M.; Neto, F.R. Effects of total knee arthroplasty for primary knee osteoarthritis on postural balance: a systematic review. Gait Posture 2021, 89, 139–160. [Google Scholar] [CrossRef]
- Cavanaugh, J.T.; Guskiewicz, K.M.; Giuliani, C.; Marshall, S.; Mercer, V.; Stergiou, N. Detecting altered postural control after cerebral concussion in athletes with normal postural stability. Br J Sports Med. 2005, 39, 805–811. [Google Scholar] [CrossRef]
- Buckley, T.A.; Oldham, J.R.; Caccese, J.B. Postural control deficits identify lingering post-concussion neurological deficits. J Sport Health Sci. 2016, 5, 61–69. [Google Scholar] [CrossRef]
- Weerdesteyn, V.; de Niet, M.; van Duijuhoven, H.J.R.; Geurts, A.C.H. Falls in individuals with stroke. J Rehabil Res Dev. 2008, 45(8), 1195–1214. [Google Scholar] [CrossRef]
- Ambrose, A.F.; Paul, G.; Hausdorff, J.M. Risk factors for falls among older adults: a review of the literature. Maturitas 2013, 75, 51–61. [Google Scholar] [CrossRef]
- Salari, N.; Darvishi, N.; Ahmadipanah, M.; Shohaimi, S.; Mohammadi, M. Global prevalence of falls in older adults: a comprehensive systematic review and meta-analysis. J Orthop Surg Res. 2022, 17, 334. [Google Scholar] [CrossRef] [PubMed]
- Sporns, O.; Chialvo, D.R.; Kaiser, M.; Hilgetag, C.C. Organization, development and function of complex brain networks. Trends Cogn Sci. 2004, 8(9), 418–425. [Google Scholar] [CrossRef] [PubMed]
- Berstein, N. The Coordination and Regulation of Movements, 1st ed.; Pergamon Press Ltd.: Oxford, UK, 1967. [Google Scholar]
- Wang, Z.; Ko, J.H.; Challis, J.H.; Newell, K.M. The degrees of freedom problem in human standing posture: collective and component dynamics. PloS One 2014, 9(1), e85414. [Google Scholar] [CrossRef] [PubMed]
- Bruton, M.; O’Dwyer, N. Synergies in coordination: a comprehensive overview of neural, computational, and behavioral approaches. J Neurophsyiol 2018, 120(6), 2761–2774. [Google Scholar] [CrossRef]
- Winter, D.A.; Prince, F.; Stergiou, P.; Powell, C. Medial-lateral and anterior-posterior responses associated with centre of pressure changes in quiet standing. Neursci Res Comm. 1993, 12(3), 141–148. [Google Scholar]
- Winter, D. Human balance and posture control during standing and walking. Gait Posture 1995, 3(4), 193–214. [Google Scholar] [CrossRef]
- Winter, D.A.; Prince, G.; Frank, J.S.; Powell, C.; Zabjek, K.F. Unified theory regarding A/P and M/L balance in quiet stance. J Neurophysiol. 1996, 75(6), 2334–2343. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.A.; Patla, A.E.; Prince, F.; Ishac, M.; Krystyna, G.-P. Stiffness control of balance in quiet standing. J Neurophysiol. 1998, 80, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.A.; Patla, A.E.; Ishac, M.; Gage, W.H. Motor mechanisms of balance during quiet standing. J Electromyogr Kinesiol. 2003, 13, 49–56. [Google Scholar] [CrossRef]
- Warnica, M.J.; Weaver, T.B.; Prentice, S.D.; Laing, A.C. The influence of ankle muscle activation on postural sway during quiet stance. Gait Posture 2014, 39(4), 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, V.; Goodman, S.; Zatsiorsky, V.; Latash, M.L. Muscle synergies during shifts of the center of pressure by standing persons: identification of muscle modes. Biol Cybern. 2003, 89, 152–161. [Google Scholar] [CrossRef]
- Krishnamoorthy, V.; Latash, M.L.; Scholz, J.P.; Zatsiorsky, V.M. Muscle synergies during shifts of the center of pressure by standing persons. Exp Brain Res. 2003, 152(3), 281–292. [Google Scholar] [CrossRef]
- Torres-Oviedo, G.; Ting, L.H. Muscle synergies characterizing human postural responses. J Neurophysiol. 2007, 98, 2144–2156. [Google Scholar] [CrossRef]
- Boonstra, T.W.; Danna-Dos-Santos, A.; Xie, H.; Roerdink, M.; Stins, J.F.; Breakspear, M. Muscle networks: connectivity analysis of EMG activity during postural control. Sci Rep. 2016, 5, 17830. [Google Scholar] [CrossRef]
- Tanabe, H.; Fujii, K.; Kouzaki, M. Intermittent muscle activity in the feedback loop of postural control system during natural quiet standing. Sci Rep. 2017, 7(1), 10631. [Google Scholar] [CrossRef]
- Ting, L.H.; McKay, J.L. Neuromechanics of muscle synergies for posture and movement. Curr Opin Neurobiol. 2007, 17(6), 622–628. [Google Scholar] [CrossRef]
- Grosse, P.; Cassidy, M.J.; Brown, P. EEG-EMG, MEG-EMG and EMG-EMG frequency analysis: physiological principles and clinical applications. Clin Neurophysiol. 2002, 113(10), 1523–1531. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, T.W. The potential of corticomuscular and intermuscular coherence for research on human motor control. Front Hum Neurosci. 2013, 7–2013. [Google Scholar] [CrossRef] [PubMed]
- Farina, D.; Negro, F.; Dideriksen, J.L. The effective neural drive to muscles is the common synaptic input to motor neurons. J Physiol. 2014, 592 Pt 16, 3427–3441. [Google Scholar] [CrossRef]
- Farina, D.; Merletti, R.; Enoka, R.M. The extraction of neural strategies from the surface EMG: an update. J Appl Physiol. 2014, 117(11), 1215–1230. [Google Scholar] [CrossRef] [PubMed]
- Hug, F.; Del Vecchio, A.; Avrillon, S.; Farina, D.; Tucker, K. Muscles from the same muscle group do not necessarily share common drive: evidence from the human triceps surae. J Appl Physiol. 2021, 130, 342–354. [Google Scholar] [CrossRef]
- Kenville, R.; Maudrich, T.; Vidaurre, C.; Maudrich, D.; Villringer, A.; Ragert, P.; et al. Intermuscular coherence between homologous muscles during dynamic and static movement periods of bipedal squatting. J Neurophsiol. 2020, 124, 1045–1055. [Google Scholar] [CrossRef]
- Tanabe, H.; Fujii, K; Kouzaki, M. Joint coordination and muscle activities of ballet dancers during tiptoe standing. Motor Control 2017, 21, 72–89. [Google Scholar] [CrossRef]
- Formaggio, E.; Masiero, S.; Volpe, D.; Demertzis, E.; Gallo, L.; Del Felice, A. Lack of inter-muscular coherence as axial muscles in Pisa Syndrome. Neurol Sci. 2019, 40, 1465–1468. [Google Scholar] [CrossRef]
- Mochizuki, G.; Semmler, J.G.; Ivanova, T.D.; Garland, S.J. Low-frequency common modulation of soleus motor unit discharge is enhanced during postural control in humans. Exp Brain Res. 2006, 175(4), 584–595. [Google Scholar] [CrossRef]
- Mochizuki, G.; Ivanova, T.D.; Garland, S.J. Factors affecting the common modulation of bilateral motor unit discharge in human soleus muscles. J Neurophysiol. 2007, 97, 3917–3925. [Google Scholar] [CrossRef]
- Noé, F.; García-Massó, X.; Paillard, T. Inter-joint coordination of posture on a seesaw device. J Electromyogr Kinesiol. 2017, 34, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Nandi, T.; Hortobagyi, T.; van Keeken, H.G.; Salem, G.J.; Lamoth, C.J.C. Standing task difficulty related increase in agonist-agonist and agonist-antagonist common inputs are driven by corticospinal and subcortical inputs respectively. Sci Rep. 2019, 9(1), 2439. [Google Scholar] [CrossRef] [PubMed]
- Ojha, A.; Alderink, G.; Rhodes, S. Coherence between electromyographic (EMG) signals of anterior tibialis, soleus, and gastrocnemius during standing balance tasks. Front Hum Neurosci. 2023, 17–2023. [Google Scholar] [CrossRef]
- Tsiouri, C.; Amiridis, I.G.; Kannas, T.; Varvariotis, N.; Sahinis, C.; Hazitaki, V.; Enoka, R.M. EMG coherence of foot and ankle muscles increases with a postural challenge in men. Gait Posture 2024, 113, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Alderink, G.; McCrumb, D.; Zeitler, D.; Rhodes, S. Analysis of connectivity in electromyography signals to examine neural correlations in the activation of lower leg muscles for postural stability: A pilot study. Bioengineering 2025, 12, 84. [Google Scholar] [CrossRef]
- Saffer, M.; Kiemel, T.; Jeka, J. Coherence analysis of muscle activity during quiet stance. Exp Brain Res. 2008, 185(2), 215–226. [Google Scholar] [CrossRef]
- Danna-Dos-Santos, A.; Boonstra, T.W.; Degani, A.M.; Cardoso, V.S.; Magalhaes, T.; Mochizuki, L.; Leonard, C.T. Multi-muscle control during bipedal stance: an EMG-EMG analysis approach. Exp Brain Res. 2014, 232(1), 75–87. [Google Scholar] [CrossRef]
- Kraskov, A.; Stögbauer, H.; Grassberger, P. Estimating mutual information. Phys Rev E 2004, 69, 066138. [Google Scholar] [CrossRef]
- Farmer, S.F.; Halliday, D.M.; Conway, B.A.; Stephens, J.A.; Rosenburg, J.R. A review of recent applications of cross-correlation methodologies to human motor unit recording. J Neurosi Methods 1997, 74, 175–187. [Google Scholar] [CrossRef]
- Pereda, E.; Quiroga, R.Q.; Bhattacharya, J. Nonlinear multivariate analysis of neurophysiological signals. Prog Neurobiol. 2005, 77, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.-H.; Lin, P.; Hallet, M. Linear and nonlinear information flow based on time-delayed mutual information method and its application to corticomuscular interaction. Clin Neurophysiol. 2010, 121, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Shannon, C.E. The mathematical theory of communication. Bell Syst Tech J. 1948, 27, 379-423, 623-656. [Google Scholar] [CrossRef]
- Cover, T.M.; Thomas, J.A. Elements of Information Theory, 2nd ed.; John Wiley & Sons Inc.: Hoboken, New Jersey, USA, 2006. [Google Scholar]
- Stone, J.V. Information Theory, A Tutorial Introduction, 1st ed.; Sebtel Press, 2015. [Google Scholar]
- Delgado-Bonal, A.; Marshak, A. Approximate entropy and sample entropy: A comprehensive tutorial. Entropy 2019, 21, 541. [Google Scholar] [CrossRef]
- Chen, C.-C.; Hsieh, J.-C.; Wu, Y.-Z.; Lee, P.O.; Chen, S.-S.; Niddam, D.M.; Yeh, T.-C.; Wu, Y.-T. Mutual-information-based approach for neural connectivity during self-paced finger lifting task. Hum Brain Mapp. 2008, 29, 265–280. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.-H.; Lin, P.; Hallet, M. Linear and nonlinear information flow based on time-delayed mutual information method and its application to corticomuscular interaction. Clin Neurophysiol. 2010, 121, 392–401. [Google Scholar] [CrossRef]
- Sun, W.; Liang, J.; Yang, Y.; Wu, Y.; Yan, T.; Song, R. Investigating age-related changes in the coordination of agonist and antagonist muscles using fuzzy entropy and mutual information. Entropy 2016, 18, 229. [Google Scholar] [CrossRef]
- Ó’Reilly, D.; Delis, I. A network information theoretic framework to characterize muscle synergies in space and time. J Neural Eng. 2022, 19, 016031. [Google Scholar] [CrossRef]
- O’Reilly, D.; Delis, I. Dissecting muscle synergies in the task space. eLife 2023, 13, RP87651. [Google Scholar] [CrossRef]
- O’Keeffe, R.; Shirazi, S.Y.; Bilaloglu, S.; Jahed, S.; Bighamian, R.; Raghavan, P.; Atashzar, S.F. Nonlinear functional muscle network based on information theory tracks sensorimotor integration post stroke. Sci Rep. 2022, 12, 130029. [Google Scholar] [CrossRef]
- Kraskov, A. Synchronization and interdependence measures and their application to the electroencephalogram of epilepsy patients and clustering of data. Dissertation PhD thesis, University of Wuppertal, North Rhine-Westphalia, Germany, 2004. Available online: https://www.fz-juelich,de/nic-series/NIC-Series-e.html.
- Cantú, H.; Nantel, J.; Millàn, M.; Paquette, C.; Cõte, J.N. Abnormal muscle activity and variability before, during, and after the occurrence of freezing in Parkinson’s disease. Fron Neurol. 2019, 10, 951. [Google Scholar] [CrossRef] [PubMed]
- Ince, R.A.A.; Giordano, B.L.; Kayser, C.; Rousselet, G.A.; Gross, J.; Schyns, P.G. A statistical framework for neuroimaging data analysis based on mutual information estimated via a Gaussian copula. Hum Brain Mapp. 2017, 38, 1541–1573. [Google Scholar] [CrossRef]
- Wang, Z.; Hallac, R.R.; Conroy, K.C.; White, S.P.; Kane, A.A.; Collinsworth, A.L.; Sweeney, J.A.; Mosconi, M.W. Postural orientation and equilibrium processes associated with increased postural sway in autism spectrum disorder (ASD). J Neurodev Dis. 2016, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Bojanek, E.K.; Wang, Z.; White, S.P.; Mosconi, M.W. Postural control processes during standing and step initiation in autism spectrum disorder. J Neurodev Dis. 2020, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, T.W.; Faes, L.; Kerkman, J.N.; Marinazzo, D. Information decomposition of multichannel EMG to map interactions in the distributed motor system. NeuroImage 2019, 202, 116093. [Google Scholar] [CrossRef]
- Faes, L; Porta, A.; Nollo, G.; Javorka, M. Information decomposition in multivariate systems: definitions, implementation and application to cardiovascular networks. Entropy 2017, 19, 5. [Google Scholar] [CrossRef]
- O’Reilly, D.; Shaw, W.; Hilt, P.; de Castro Aguiar, R.; Astill, S.L.; Delis, I. Quantifying the diverse contributions of hierarchical muscle interactions to motor function. IScience 2025, 28, 111613. [Google Scholar] [CrossRef]
- Perotto, A.O. Anatomical guide for the electromyographer, 3rd ed.; Charles C. Thomas: Springfield, IL, USA, 2011; pp. 154–167. [Google Scholar]
- Hermens, H.J.; Freriks, B.; Disselhorst-Klug, C.; Rau, G. Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol. 2000, 10(5), 361–74. [Google Scholar] [CrossRef]
- Villaverde, A.F.; Ross, J.; Morán, F.; Banga, J.R. MIDER: network inference with mutual information distance and entropy reduction. PLos ONE 2014, 9(5), e96732. [Google Scholar] [CrossRef]
- Cellucci, C.J.; Albano, A.M.; Rapp, P.E. Statistical validation of mutual information calculations: comparison of alternative numerical algorithms. Phys Rev E 2005, 71, 66208. [Google Scholar] [CrossRef]
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. 2024. Available online: https://www.R-project.org (accessed on 10 June 2024).
- Posit Team (2024), RStudio: Integrated Development Environment for R.Posit Software, PBC, Boston, MA. Available online: https://www.posit.co/ (accessed on 10 June 2024).
- Latash, M.L.; Scholz, J.P.; Schöner, G. Motor control strategies revealed in the structure of motor variability. Exerc Sport Sci Rev. 2002, 30(1), 26–31. [Google Scholar] [CrossRef]
- Latash, M.L. The bliss (not the problem) of motor abundance (not redundancy). Exp Brain Res. 2012, 217, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Latash, M.L. Motor synergies and the equilibrium-point hypothesis. Mot Contr. 2020, 14, 294–2010. [Google Scholar] [CrossRef]
- Proferta, V.L.; Turvey, M.T. Berstein’s levels of movement construction: A contemporary perspective. Hum Mov Sci. 2018, 57, 111–133. [Google Scholar] [CrossRef] [PubMed]
- Scholz, J.P.; Schöner, G. The uncontrolled manifold concept: Identifying control variables for a functional task. Exp Brain Res. 1999, 126, 289–306. [Google Scholar] [CrossRef]
- Boonstra, T.W.; Breakspear, M. Neural mechanisms of intermuscular coherence: Implications for the rectification of surface electromyography. J Neurophysiol. 2011, 107, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Semmler, J.G. Motor unit synchronization and neuromuscular performance. Exer Sport Sci Rev. 2002, 30, 8–14. [Google Scholar] [CrossRef]
- Danna-Dos-Santos, A.; Degani, A.M.; Boonstra, T.W.; Mochizuki, L.; Harney, A.M.; Schmeckpeper, M.M.; Tabor, L.C.; Leonard, C.T. The influence of visual information on multi-muscle control during quiet stance: a spectral analysis approach. Exp Brain Res. 2015, 233(2), 657–669. [Google Scholar] [CrossRef]
- De Luca, C.J.; Erim, Z. Common drive in motor units of a synergistic muscle pair. J Neurophysiol. 2002, 87(4), 2200–2204. [Google Scholar] [CrossRef]
- Farmer, S.G. Rhythmicity, synchronization and binding in human and primate motor systems. J Physiol. 1998, 509 Pt 1, 3–14. [Google Scholar] [CrossRef]
- Bizzi, E.; Cheung, V.C.K. The neural origin of muscle synergies. Front Comput Neurosci. 2013, 7–2013. [Google Scholar] [CrossRef]
- Laine, C.M.; Valero-Cuevas, F.J. Intermuscular coherence reflects functional coordination. J Neurophysiol 2017, 118, 1775–1783. [Google Scholar] [CrossRef]
- Kerkman, J.N.; Daffertshofer, A.; Gollo, L.L.; Breakshpear, M.; Boonstra, T.W. Network structure of the human musculoskeletal system shapes neural interactions on multiple time scales. Sci Adv. 2018, 4(6), eaat0497. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, T.W.; Roerdink, M.; Daffertshofer, A.; van Vugt, B.; van Werven, G.; Beek, P.J. Low-alcohol doses reduce common 10- to 15-Hz input to bilateral leg muscles during quiet standing. J Neurophysiol. 2008, 100(4), 2158–2164. [Google Scholar] [CrossRef]
- Obata, H.; Abe, M.O.; Masani, K.; Nakazawa, K. Modulation between bilateral legs and within unilateral muscle synergists of postural muscle activity changes with development and aging. Exp Brain Res. 2014, 232(1), 1–11. [Google Scholar] [CrossRef] [PubMed]
- Degani, A.M.; Leonard, C.T.; Danna-Dos-Santos, A. The use of intermuscular coherence analysis as a novel approach to detect age-related changes in postural synergy. Neurosci Lett. 2017, 656, 108–113. [Google Scholar] [CrossRef]
- Degani, A.M.; Leonard, C.T.; Danna-Dos-Santos, A. The effects of aging on the distribution and strength of correlated neural inputs to postural muscles during unperturbed bipedal stance. Exp Brain Res. 2020, 238, 1537–1553. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Saito, K.; Ishida, K.; Tanabe, S.; Nojima, I. Age-related changes in the ability to modulate common input to bilateral and unilateral plantar flexors during forward postural lean. Front Hum Neurosci. 2018, 12, 254. [Google Scholar] [CrossRef]
- Walker, S.; Piitulainen, H.; Manlangit, T.; Avela, J.; Baker, S.N. Older adults show elevated intermuscular coherence in eyes-open standing by only young adults increase coherence in response to closing the eyes. Exp Physiol. 2020, 105, 1000–1011. [Google Scholar] [CrossRef]
- Yamanake, E.; Horiuchi, Y.; Nojima, I. EMG-EMG coherence during voluntary control of human standing tasks: a systematic scoping review. Front Neurosci. 2023, 17, 1145751. [Google Scholar] [CrossRef]
- García-Massó, X.; Pellicer-Chenoll, M.; Gonzalez, L.M.; Toca-Herrera, J.L. The difficulty of the postural control task affects multi-muscle control during quiet standing. Exp Brain Res. 2016, 234(7), 1977–1986. [Google Scholar] [CrossRef]
- Watanabe, T.; Saito, K.; Ishida, K.; Tanabe, S.; Nojima, I. Fatigue-induced decline in low-frequency common input to bilateral and unilateral plantar flexors during quiet standing. Neurosci Lett. 2018c, 686, 193–197. [Google Scholar] [CrossRef]
- Glass, S.M.; Wildman, L.; Brummitt, C.; Ratchford, K.; Westbrook, G.M.; Aron, A. Effects of global postural alignment on posture-stabilizing synergy and intermuscular coherence in bipedal standing. Exp Brain Res. 2022, 240, 841–851. [Google Scholar] [CrossRef]
- Nandi, T.; Fisher, B.E.; Hortobágyi, T.; Salem, G.J. Increasing mediolateral standing sway is associated with increasing corticospinal excitability, and decreasing M1 inhibition and facilitation. Gait Posture 2018, 60, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Torres-Oviedo, G.; Ting, L.H. Subject-specific muscle synergies in human balance control are consistent across different biomechanical contexts. J Neurophysiol. 2010, 13, 9259. [Google Scholar] [CrossRef]
- Montesinos, L.; Castaldo, R.; Pecchia, L. On the use of approximate entropy and sample entropy with centre of pressure time series. J Neuroeng Rehabil. 2018, 15, 116. [Google Scholar] [CrossRef] [PubMed]
- Pincus, S.M.; Gladstone, I.M.; Ehrenkranz, R.A. A regularity statistic for medical data analysis. J Clin Monit. 1991, 7(4), 335–45. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am J Physiol Heart Circ Physiol. 2000, 278(6), H2039–49. [Google Scholar] [CrossRef] [PubMed]
- Tipton, N.; Alderink, G.; Rhodes, S. Approximate entropy and the velocity of center of pressure to determine postural stability: A pilot study. Appl Sci. 2023, 13, 9259. [Google Scholar] [CrossRef]
- Wesley, J.; Rhodes, S.; Zeitler, D.W.; Alderink, G. Approximate and sample entropy of the center of pressure during unperturbed tandem standing: Effect of altering the tolerance window. Appl Sci. 2025, 15(2), 576. [Google Scholar] [CrossRef]
- Morasso, P.; Cherif, A.; Zenzeri, J. Quiet standing: the single inverted pendulum model is not so bad after all. PLoS One 2019, 14(3), e0213870. [Google Scholar] [CrossRef] [PubMed]
- Gunther, M.; Grimmer, S.; Siebert, T.; Blickhan, R. All joints contribute to quiet stance: a mechanical analysis. J Biomech. 2009, 42, 2739–2746. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Nomura, T.; Casadio, M.; Morasso, P. Intermittent control with ankle, hip, and mixed strategies during quiet standing: a theoretical proposal based on a double inverted pendulum model. J Theor Biol. 2012, 310, 55–79. [Google Scholar] [CrossRef]
- Sasagawa, S.; Shinya, M.; Nakazawa, K. Interjoint dynamic interaction during constrained human quiet standing by induced acceleration analysis. J Neurophysiol. 2014, 111, 313–322. [Google Scholar] [CrossRef]
| Wave | Frequency (Hz) | Origin | Task Manifestation |
| Delta | 0.5 – 4 | Unknown | Isometric contraction, slow movements |
| Theta | 4 – 8 | Unknown | Isometric contraction, slow movements |
| Alpha | 8 – 13 | Unknown | Isometric contraction, slow movements |
| Beta | 13 – 30 | Motor cortex | Submaximal voluntary contraction |
| Lower Gamma | 30 – 60 | Motor cortex | Voluntary contraction, slow movements |
| Upper Gamma | 60 – 100 | Brainstem | Eye movement (60 – 90 Hz), respiration |
| P | Gender | Foot Dominance | Age | Height (cm) | BM (kg) |
| P01 | F | Right | 26 | 170.4 | 64.5 |
| P02 | F | Right | 21 | 162.6 | 64.7 |
| P03 | M | Left | 23 | 174.7 | 63.9 |
| P04 | M | Left | 28 | 190 | 98.2 |
| P05 | F | Right | 25 | 163 | 70.1 |
| P06 | F | Right | 25 | 164 | 63.2 |
| Balance Condition | Description |
| EOFT | Eyes Open, Feet Together |
| ECFT | Eyes Closed, Feet Together |
| EOTanDF | EO, Feet Tandem, Dominant Foot Forward |
| ECTanDF | EC, Feet Tandem, Dominant Foot Forward |
| EOTanDB | EO, Feet Tandem, Dominant Foot Back |
| ECTanDB | EC, Feet Tandem, Dominant Foot Back |
| Bands | Range (Hz) |
| Delta | 0 - 4 |
| Theta | 4 – 8 |
| Apha | 8 – 13 |
| Beta | 13 – 30 |
| Lower Gamma | 30 – 60 |
| Upper Gamma | 60 - 100 |
| Left Unilateral | Right Unilateral | Bilateral Homologous |
| LTA:LMG | RTA:RMG | LTA:RTA |
| LTA:LS | RTA:RS | LMG:RMG |
| LMG:LS | RMG:RS | LS:RS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




