Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Methods
2.1. Materials and Test Sample
2.2. Data Acquisition Methods
2.2.1. Weather and Environmental Data
2.2.2. Coating Performance Data
2.3. Data Analytics
3. Results and Discussion
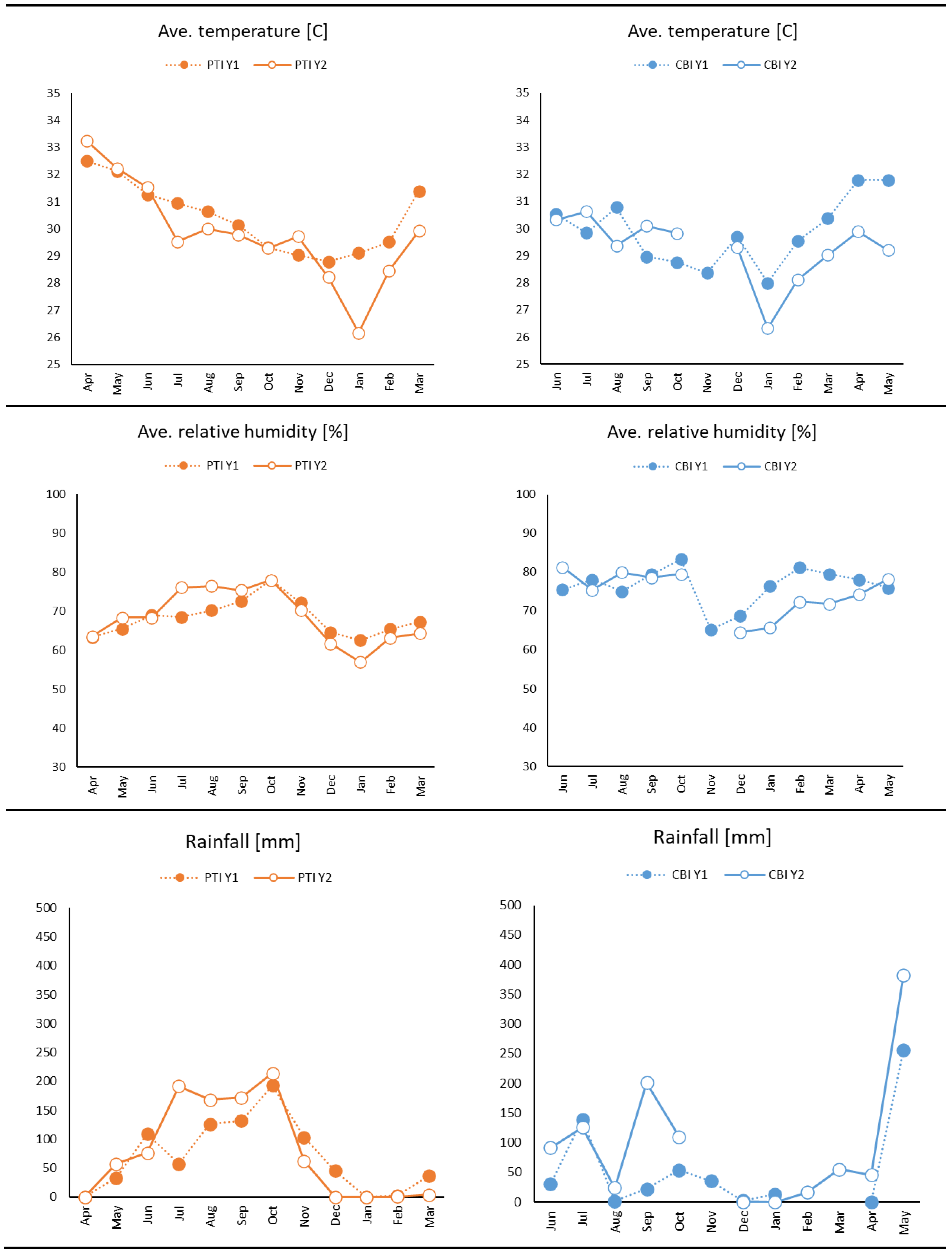
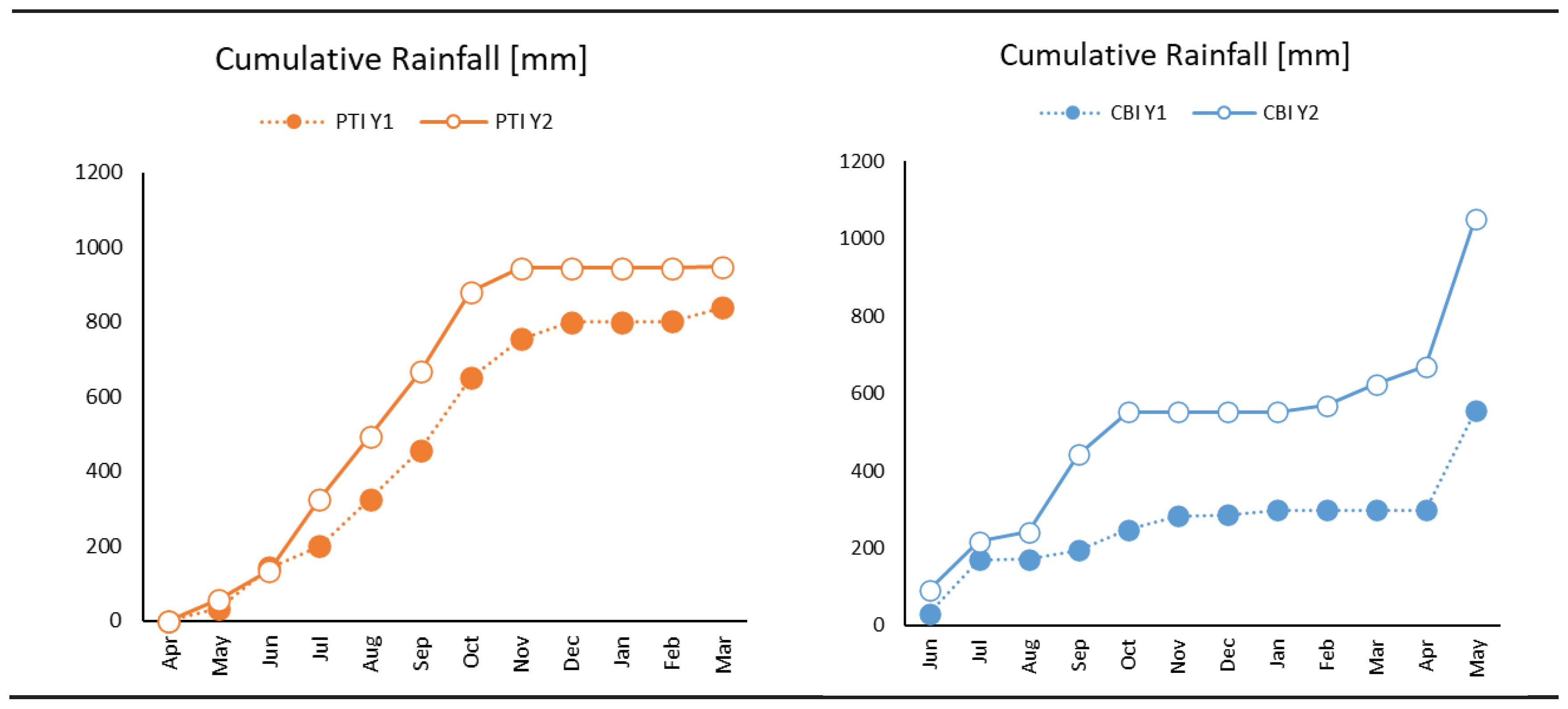
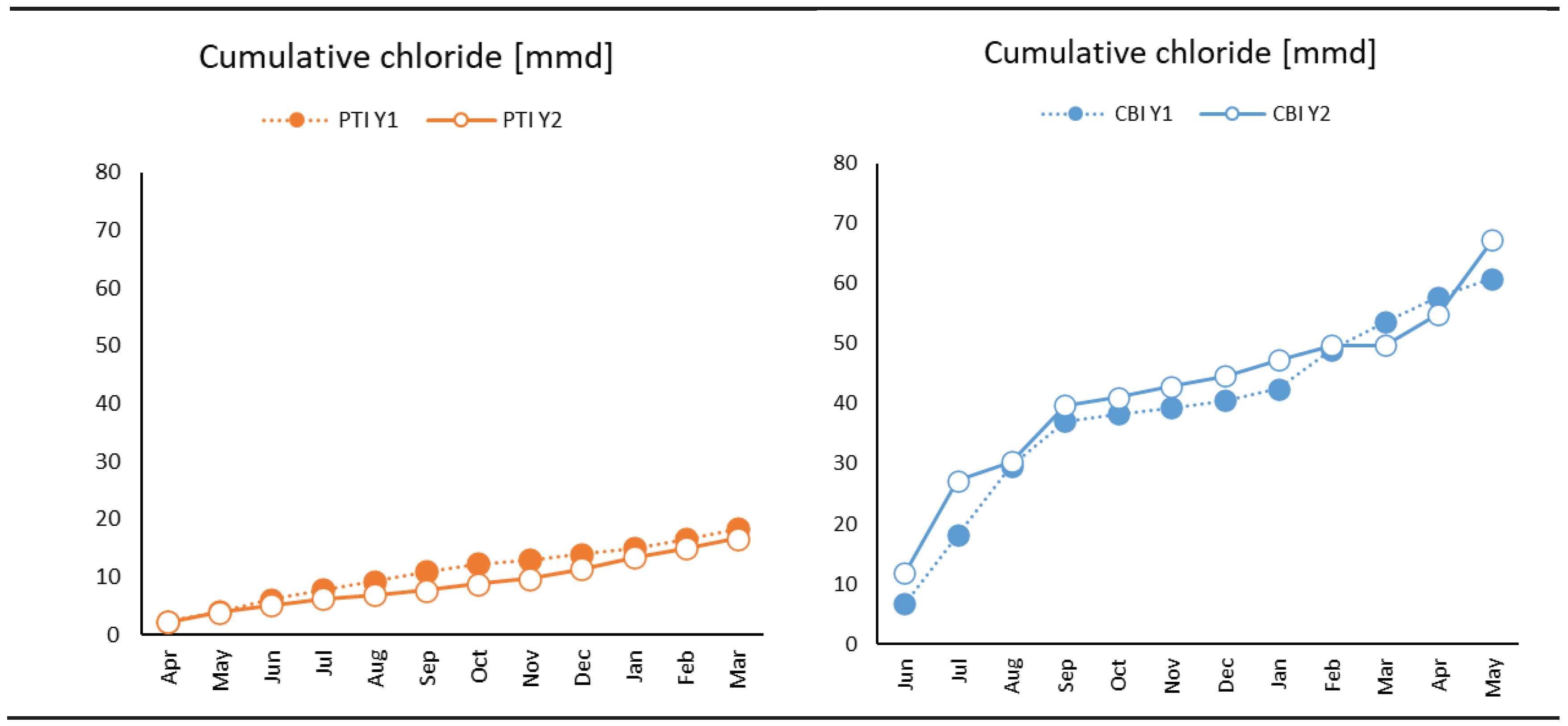
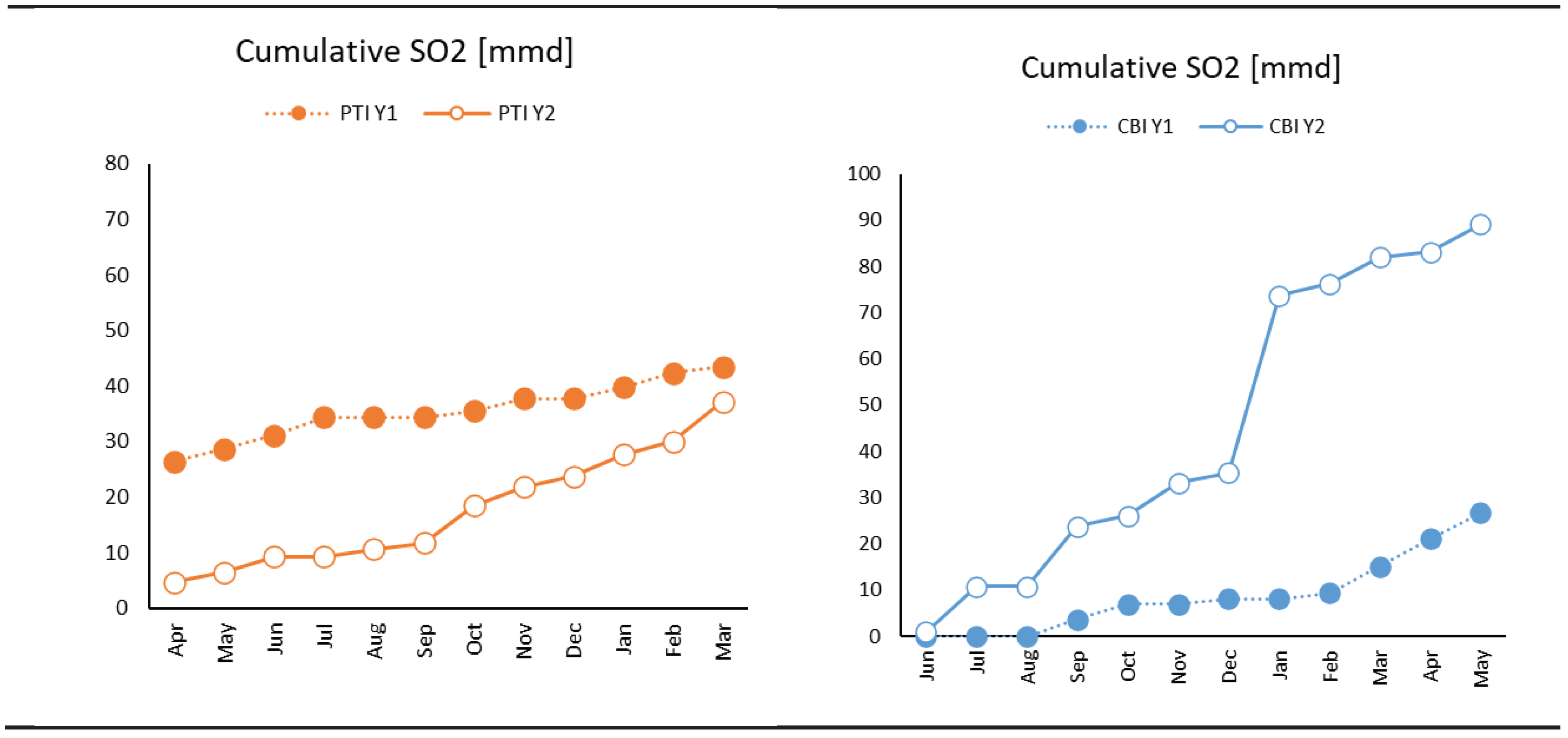
3.1. Weather and Environmental Results
3.2. Coating Performance Results
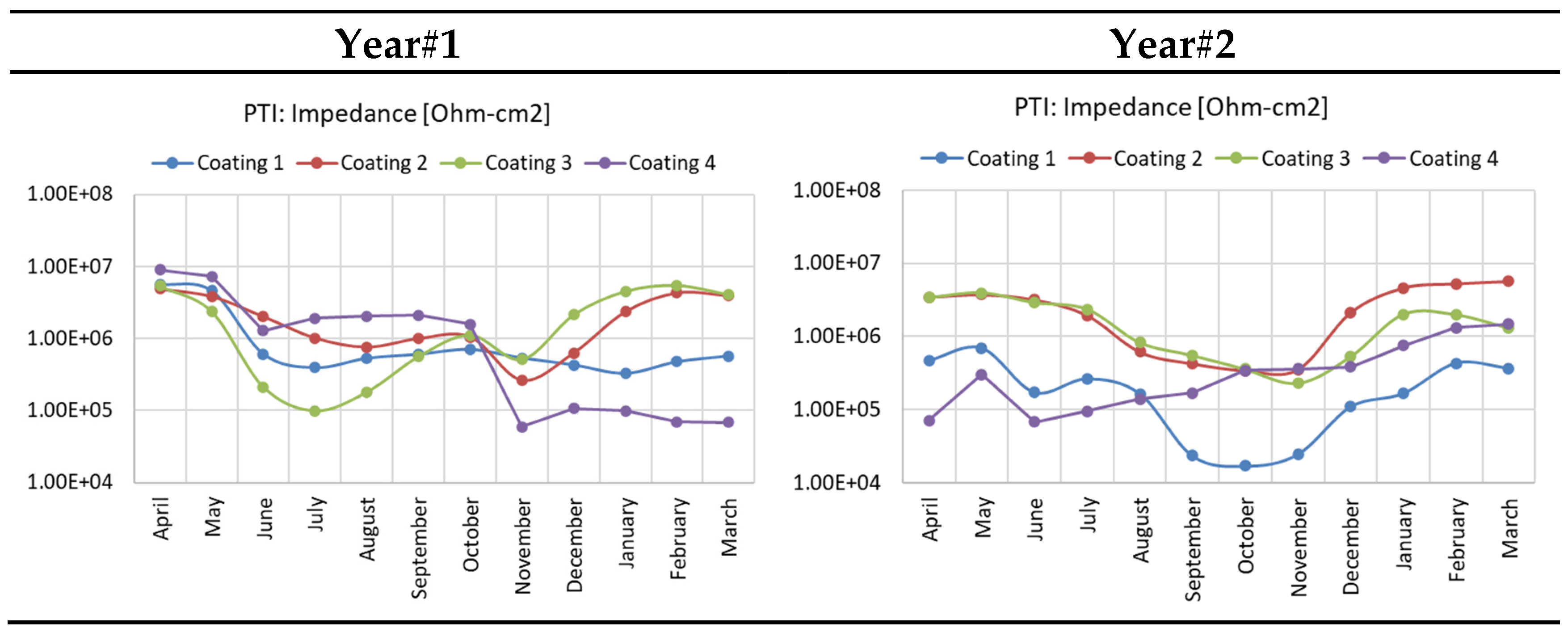
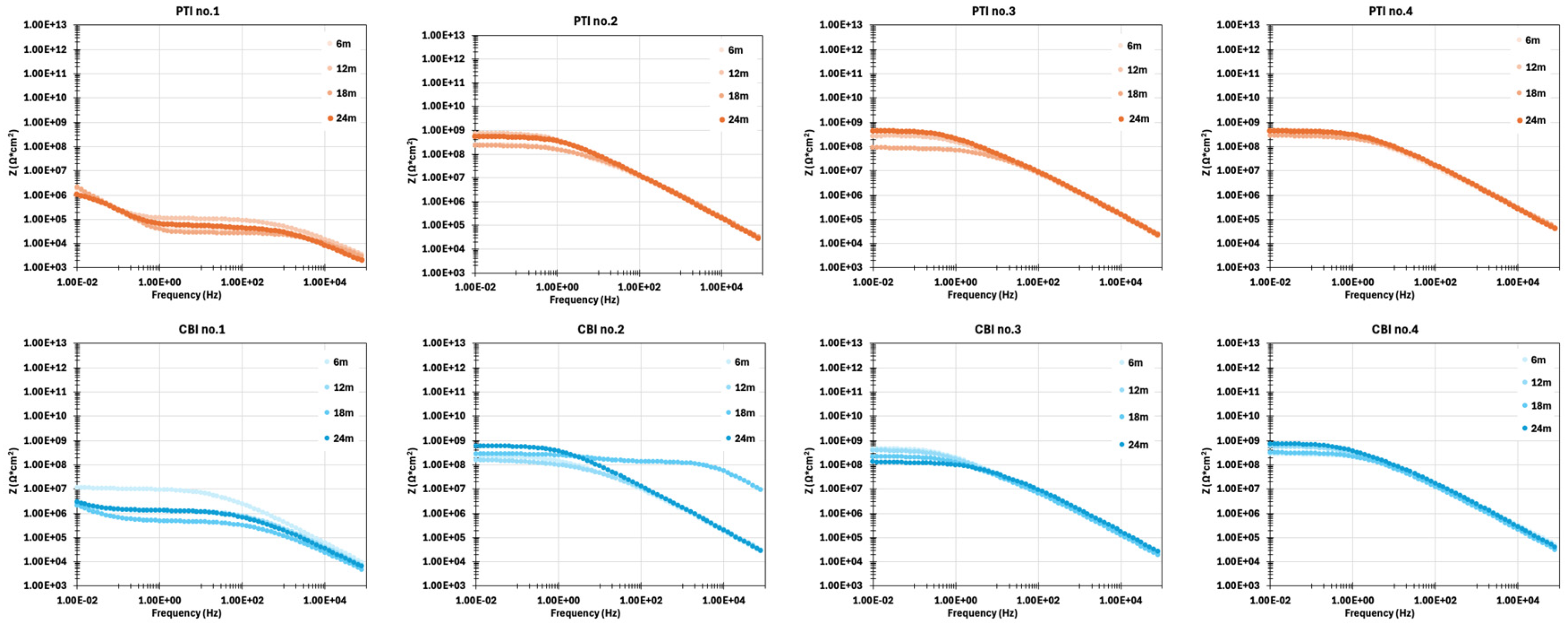
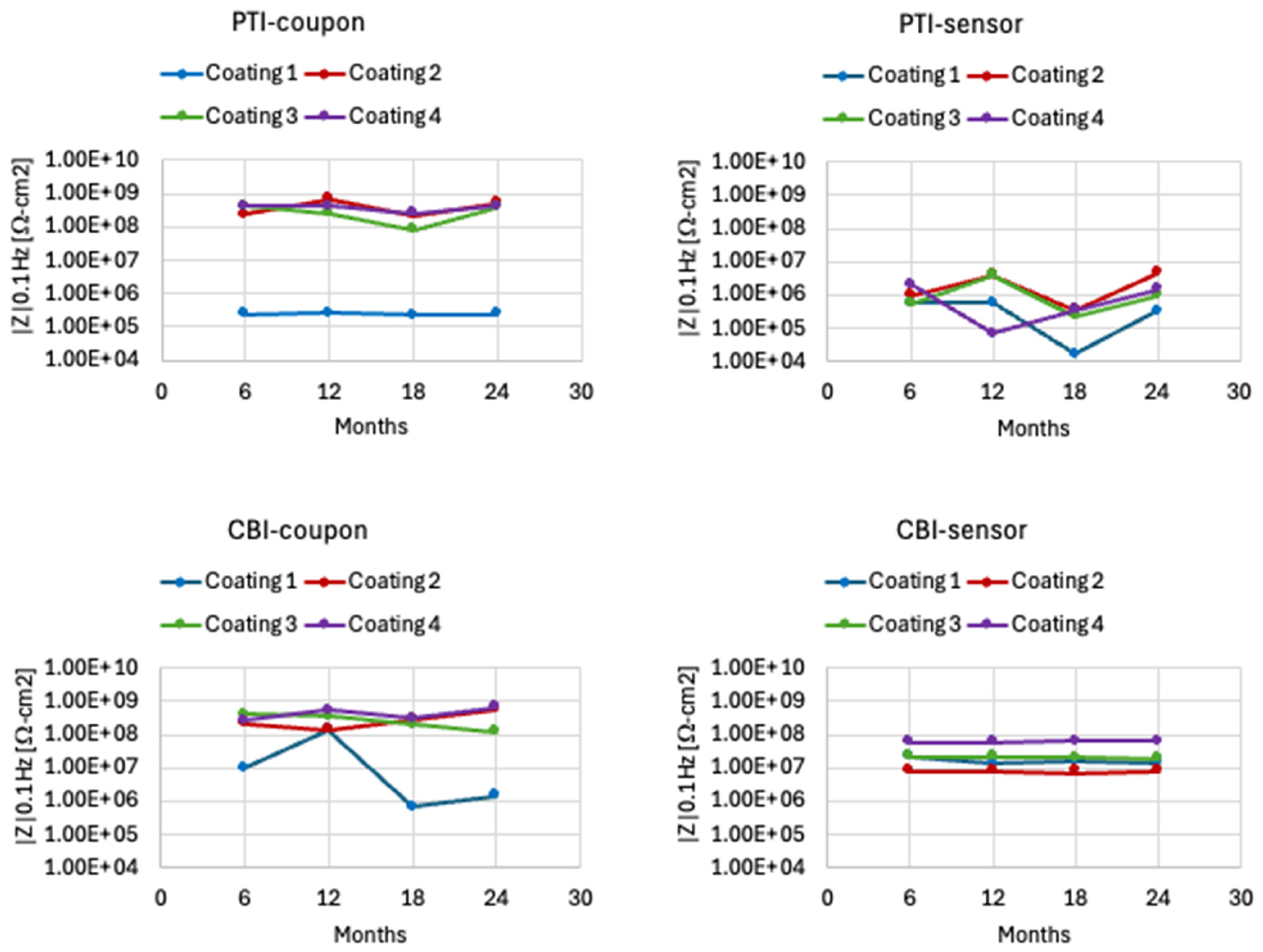
3.3. EIS Measurement on Coupon Samples
3.4. Machine Learning Model
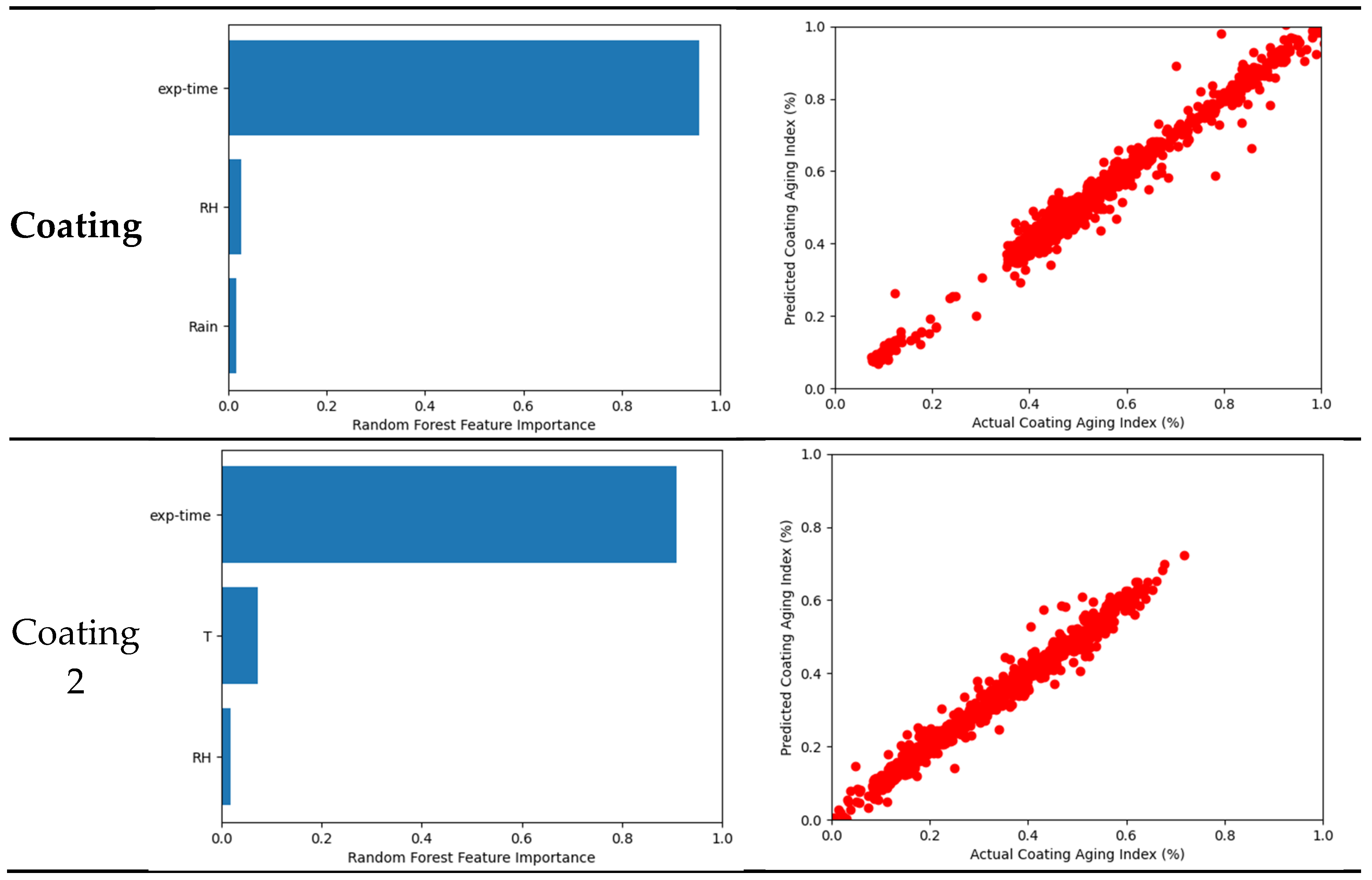
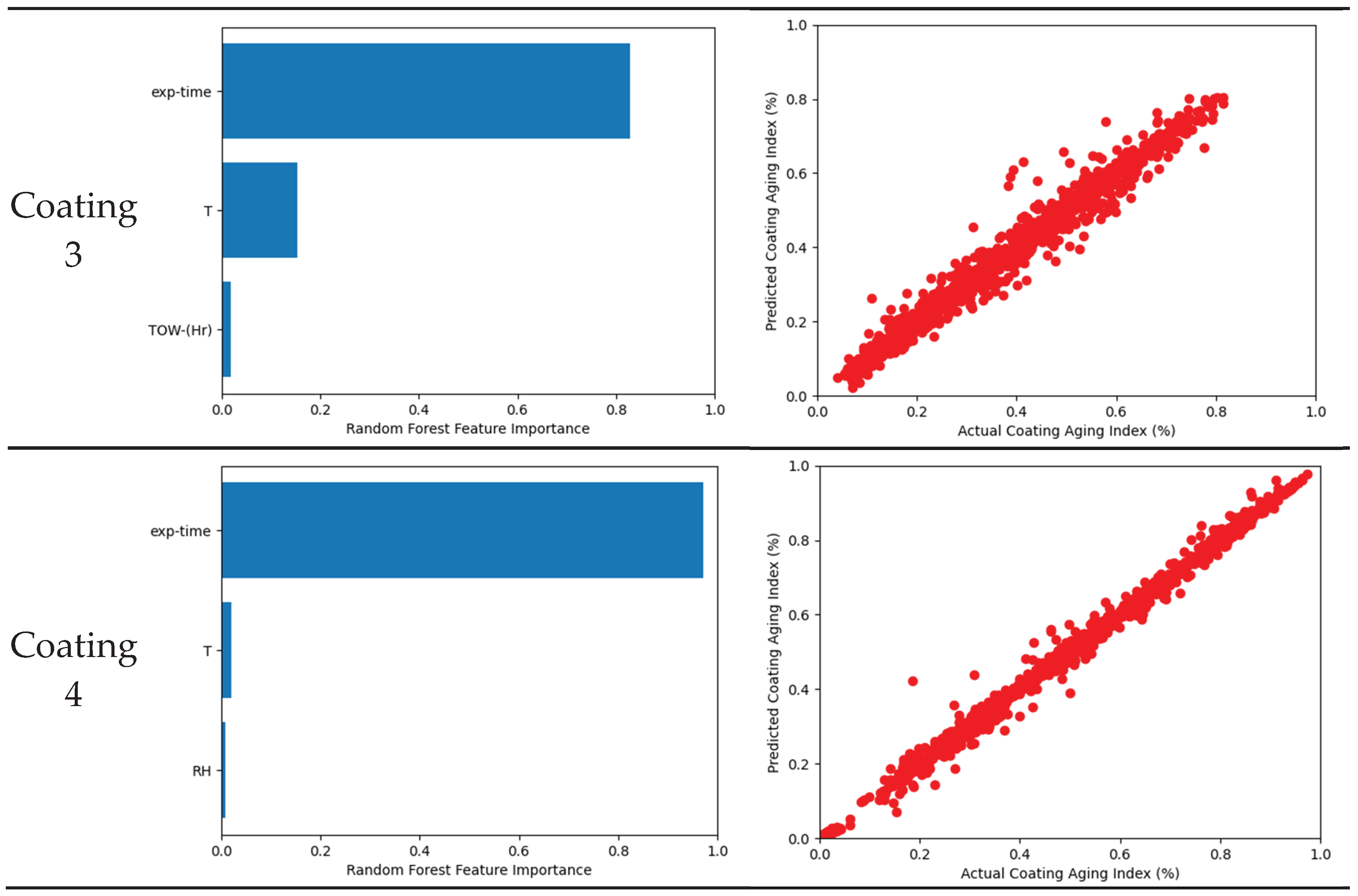
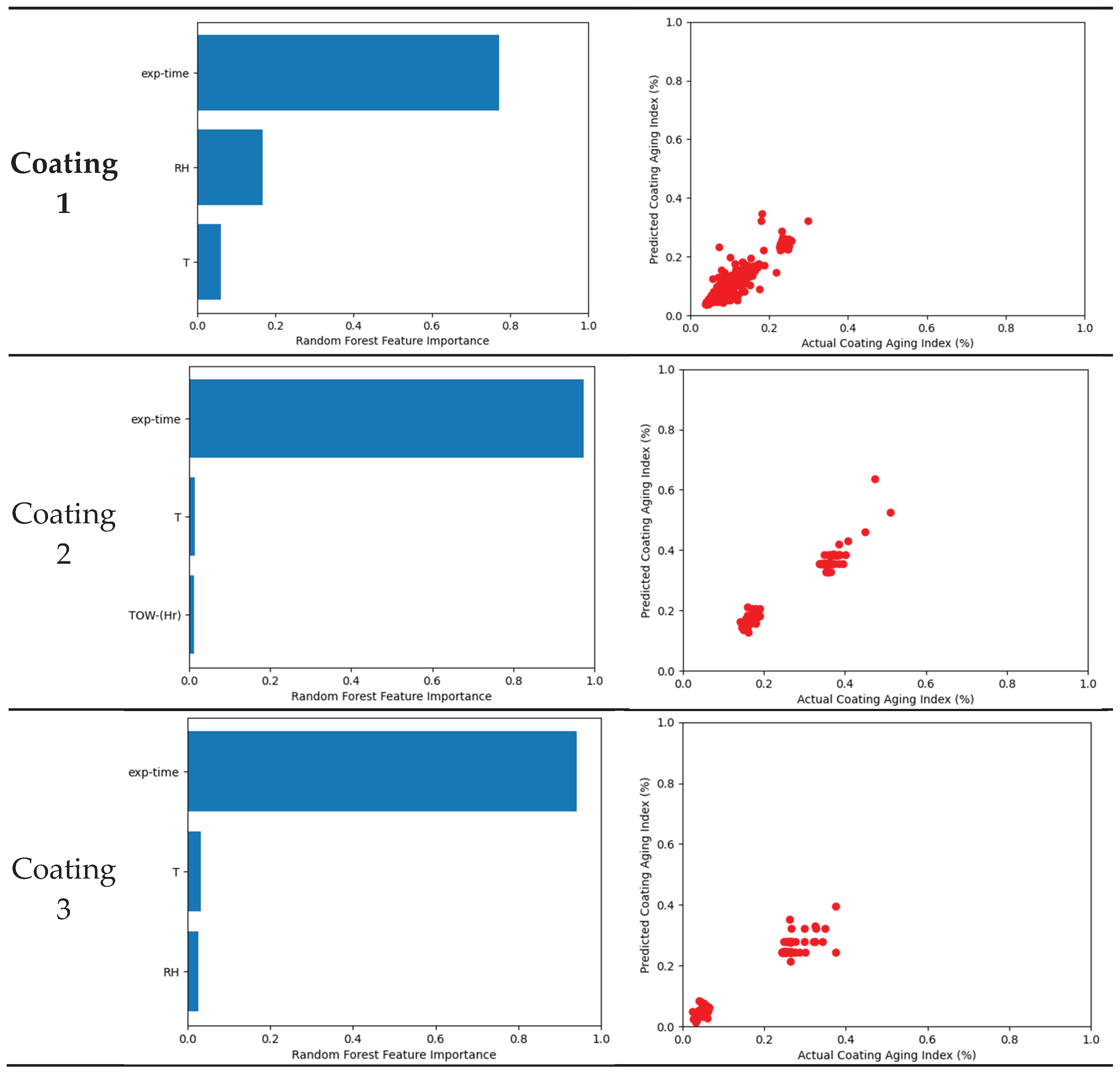
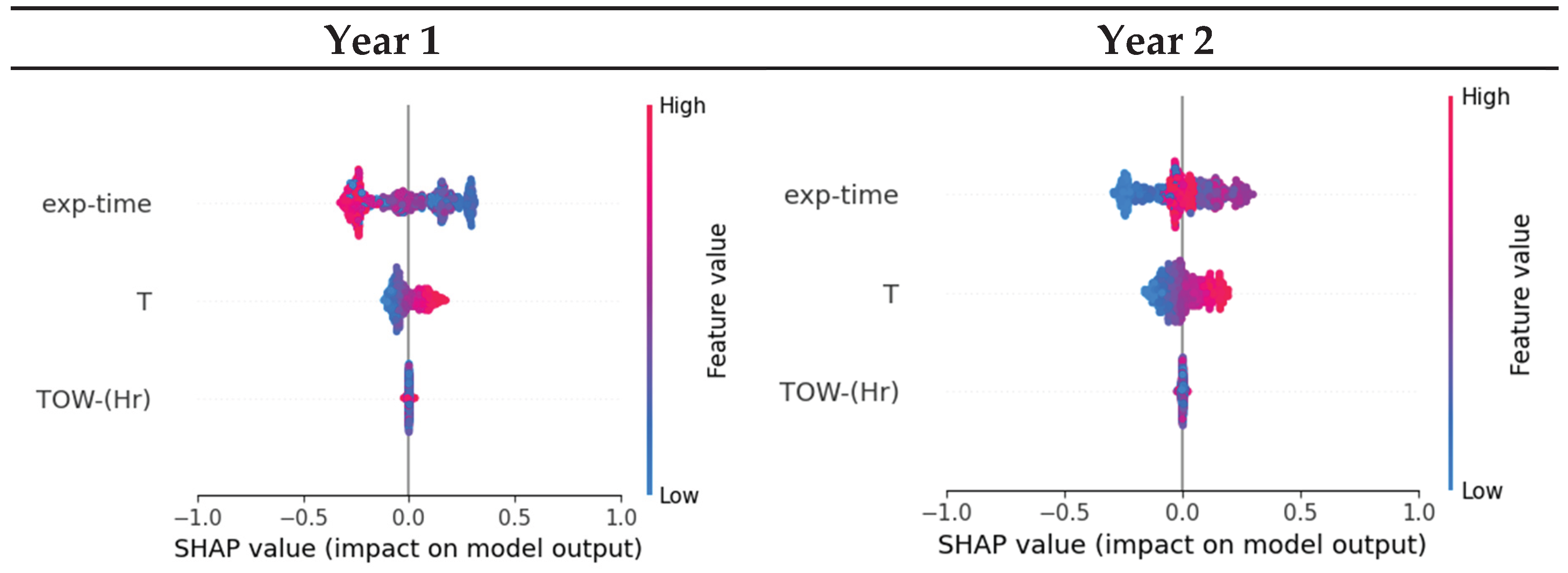
3.5. Coating System
4. Conclusions
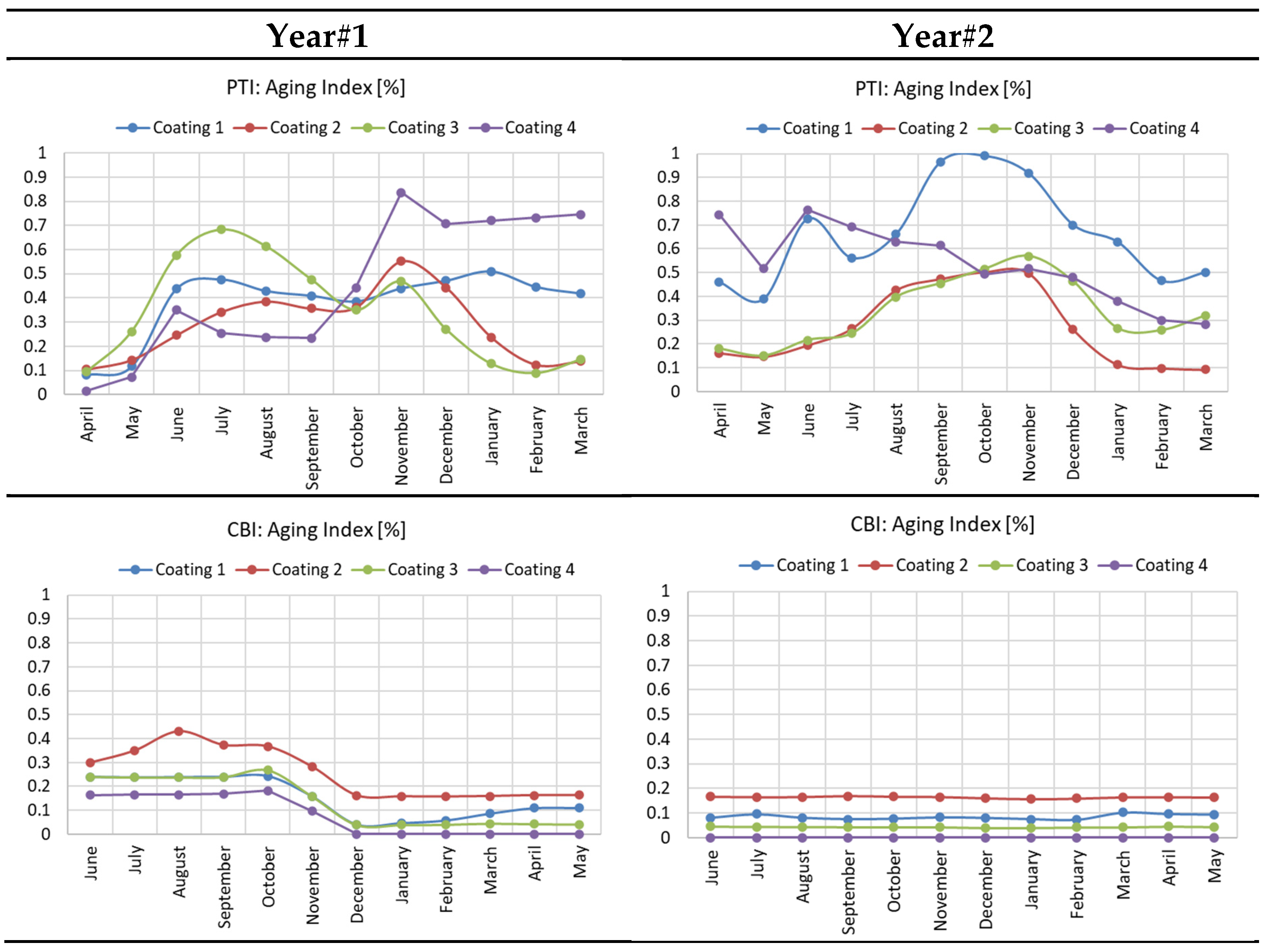
- Impedance data from EIS sensor indicated the coating resistance and can be converted to Coating aging index to imply coating degradation.
- Coating degradation was more rapid at PTI station as observed by increasing Coating aging index during wet season, whereas coating degradation at CBI remained negligible.
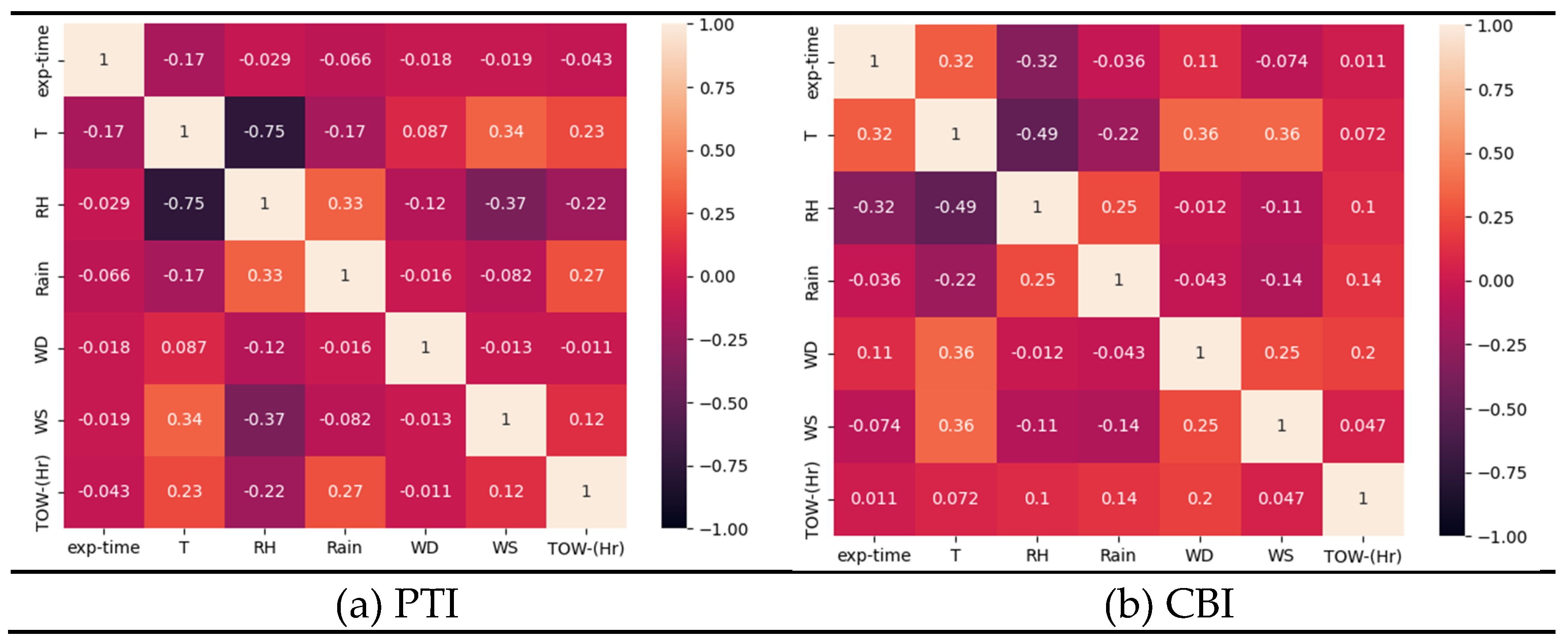
- Tested coating degradation was influenced by various parameters. The most influential factor is the exposure time because the change in Coating aging index was slow compared to the instantaneous change in weather characteristic. Weak influences were generally temperature and moisture such as relative humidity, rain, or time of wetness (TOW).
- Coating 1 (epoxy) showed the lowest impedance due to UV-induced degradation, the topcoats of polyurethane, aluminum-pigmented polyester, and acrylic layers progressively improved durability by enhancing UV protection, barrier properties, and mechanical performance.
Acknowledgments
References
- S. Sun, Q. Zheng, D. Li, J. Wen, Long-term atmospheric corrosion behaviour of aluminium alloys 2024 and 7075 in urban, coastal and industrial environments, Corrosion Science. 51 (2009) 719–727. [CrossRef]
- Z. Cui, F. Ge, X. Li, M. Zhu, K. Xiao, C. Dong, X. Wang, Mechanistic studies of atmospheric corrosion behavior of Al and Al-based alloys in a tropical marine environment, Journal of Wuhan University of Technology-Mater. Sci. Ed. 32 (2017) 633–639. [CrossRef]
- Z. Cui, X. Li, H. Zhang, K. Xiao, C. Dong, Z. Liu, L. Wang, Atmospheric Corrosion Behavior of 2A12 Aluminum Alloy in a Tropical Marine Environment, Advances in Materials Science and Engineering. 2015 (2015) 1–17. [CrossRef]
- L. Mariaca, C. Menchaca, E. Sarmiento, O. Sarmiento, J. Ramirez, J. Uruchurtu, Atmospheric Corrosion Dose/Response Functions from Statistical Data Analysis for Different Sites of Mexico, Innovations in Corrosion and Materials Science (Formerly Recent Patents on Corrosion Science). 4 (2014) 11–20. [CrossRef]
- S. Syed, ATMOSPHERIC CORROSION OF MATERIALS, 11 (2006) 25.
- F. Corvo, T. Perez, J. Reyes-Trujeque, L. Dzib, J. González-Sánchez, A. Castañeda, Atmospheric corrosion in tropical humid climates, 37661 (2009) 0–0.
- F.J. Hernández, J.J. Santana, R.M. Souto, S. González, J. Morales, Characterization of the Atmospheric Corrosion of Aluminum in Archipelagic Subtropical Environments, Int. J. Electrochem. Sci. 6 (2011) 15.
- Belén Chico, Daniel de la Fuente, Iván Díaz, Joaquín Simancas, Manuel Morcillo, Annual Atmospheric Corrosion of Carbon Steel Worldwide. An Integration of ISOCORRAG, ICP/UNECE and MICAT Databases, Materials. 10 (2017) 601. [CrossRef]
- I.S. Cole, D.A. Paterson, W.D. Ganther, A. Neufeld, B. Hinton, G. McAdam, M. McGeachie, R. Jeffery, L. Chotimongkol, C. Bhamornsut, N.V. Hue, S. Purwadaria, Holistic model for atmospheric corrosion: Part 3—Effect of natural and man made landforms on deposition of marine salts in Australia and south-east Asia, Corrosion Engineering, Science and Technology. 38 (2003) 267–274. [CrossRef]
- C.Y. Chao, L.F. Lin, D.D. Macdonald, A Point Defect Model for Anodic Passive Films: I . Film Growth Kinetics, J. Electrochem. Soc. 128 (1981) 1187. [CrossRef]
- L.F. Lin, C.Y. Chao, D.D. Macdonald, A Point Defect Model for Anodic Passive Films: II . Chemical Breakdown and Pit Initiation, J. Electrochem. Soc. 128 (1981) 1194. [CrossRef]
- Z. Cui, X. Li, H. Zhang, K. Xiao, C. Dong, Z. Liu, L. Wang, Atmospheric Corrosion Behavior of 2A12 Aluminum Alloy in a Tropical Marine Environment, Adv. Mater. Sci. Eng. (2015) 163025. [CrossRef]
- Fujii, K., et al., Atmospheric Corrosivity Estimation at Electrical Control Unit Room by Multichannel Quartz Crystal Microbalance Corrosion Sensors. MATERIALS TRANSACTIONS, 2012. 53(2): p. 412-416. [CrossRef]
- Zakipour, S. and C. Leygraf, Quartz crystal microbalance applied to studies of atmospheric corrosion of metals. British Corrosion Journal, 1992. 27(4): p. 295-298. [CrossRef]
- Benounis, M. and N. Jaffrezic-Renault, Elaboration of an optical fibre corrosion sensor for aircraft applications. Sensors and Actuators B: Chemical, 2004. 100(1): p. 1-8. [CrossRef]
- Geng, R., Modern acoustic emission technique and its application in aviation industry. Ultrasonics, 2006. 44: p. e1025-e1029. [CrossRef]
- Bellenger, F., H. Mazille, and H. Idrissi, Use of acoustic emission technique for the early detection of aluminum alloys exfoliation corrosion. NDT & E International, 2002. 35(6): p. 385-392. [CrossRef]
- Yasri, M., et al., Fundamental basis of electromagnetic wave propagation in a zinc microstrip lines during its corrosion. Sensors and Actuators B: Chemical, 2016. 223: p. 352-358. [CrossRef]
- Yasri, M., et al., Monitoring uniform and localised corrosion by a radiofrequency sensing method. Sensors and Actuators B: Chemical, 2018. 257: p. 988-992. [CrossRef]
- Choudhary, S., A. Garg, and K. Mondal, Relation between open circuit potential and polarization resistance with rust and corrosion monitoring of mild steel. Journal of Materials Engineering and Performance, 2016. 25(7): p. 2969-2976. [CrossRef]
- Motoda, S.-i., et al., ACM (Atmospheric Corrosion Monitor) type corrosion sensor to evaluate corrosivity of marine atmosphere. Zairyo-to-Kankyo, 1994. 43(10): p. 550-556. [CrossRef]
- Motoda, S.-i., et al., Corrosive factors of a Marine atmosphere analyzed by ACM aensor for 1 year. Zairyo-to-Kankyo, 1995. 44(4): p. 218-225. [CrossRef]
- Pongsaksawad, W., et al., Corrosion assessment of carbon steel in Thailand by atmospheric corrosion monitoring (ACM) sensors. Journal of Metals, Materials and Minerals, 2017. 20(2).
- Mizuno, D., et al., Corrosion monitoring and materials selection for automotive environments by using Atmospheric Corrosion Monitor (ACM) sensor. Corrosion Science, 2014. 83: p. 217-225. [CrossRef]
- Ma, X., et al., Monitoring marine atmospheric corrosion by electrochemical impedance spectroscopy under various relative humidities. International Journal of Electrochemical Science, 2015. 10(12): p. 10402-10421. [CrossRef]
- Xia, D.-H., et al., Detection of atmospheric corrosion of aluminum alloys by electrochemical probes: Theoretical analysis and experimental tests. Journal of The Electrochemical Society, 2019. 166(12): p. B1000–B1009.
- Diler, E., et al., Real-time corrosion monitoring of aluminium alloys under chloride-contaminated atmospheric conditions. Materials and Corrosion, 2021. 72(8): p. 1377-1387. [CrossRef]
- Cai, J.P. and S.B. Lyon, A mechanistic study of initial atmospheric corrosion kinetics using electrical resistance sensors. Corrosion Science, 2005. 47(12): p. 2956-2973. [CrossRef]
- Steen, N.V.d., et al., Comparing modeled and experimental accelerated corrosion tests on steel. Journal of The Electrochemical Society, 2017. 164(9): p. C554. [CrossRef]
- Komary, M., et al. Low-cost technologies used in corrosion monitoring. Sensors, 2023. 23. [CrossRef]
- Adhikari, S., Unocic, K.A., Zhai, Y., Frankel, G.S., Zimmerman, J., Fristad, W., 2011. Hexafluorozirconic acid based surface pretreatments: Characterization and performance assessment. Electrochimica Acta, ADVANCES IN CORROSION SCIENCE FOR LIFETIME PREDICTION AND SUSTAINABILITY Selection of papers from the 8th ISE Spring Meeting 2-5 May 2010, Columbus, OH, USA 56, 1912–1924. [CrossRef]
- Wang, B., Cheng, X., Wang, L., Li, Z., Liu, C., Zhang, D., and Li, X. Big data mining of corrosion for weathering steel in marine atmospheric environments: Discovery and mechanism of critical temperature influencing corrosion resistance. Journal of Materials Science and Technology, 2025. 256(6): p. 221-235. [CrossRef]
- Awaja, F. and P. J. Pigram, Surface Molecular Characterisation of Different Epoxy Resin Composites Subjected to UV Accelerated Degradation Using XPS and ToF-SIMS. Polymer Degradation and Stability, 2009. 94 (4): p. 651–658. [CrossRef]
- [34] Liang, Y.; Liu, F.-C.; Nie, M.; Zhao, S.; Lin, J.; Han, E.-H. Influence of Nano-Al Concentrates on the Corrosion Resistance of Epoxy Coatings. Journal of Materials Science & Technology, 2013. 29 (4), p. 353–358. [CrossRef]
- [35] Liu, X.; Zhang, Y.; Jiang, Y.; Li, M.; Bai, J.; Zhou, X. Effect of Acrylic Resin on the Protection Performance of Epoxy Coating for Magnesium Alloy. Coatings, 2024. 14 (5), p. 577. [CrossRef]
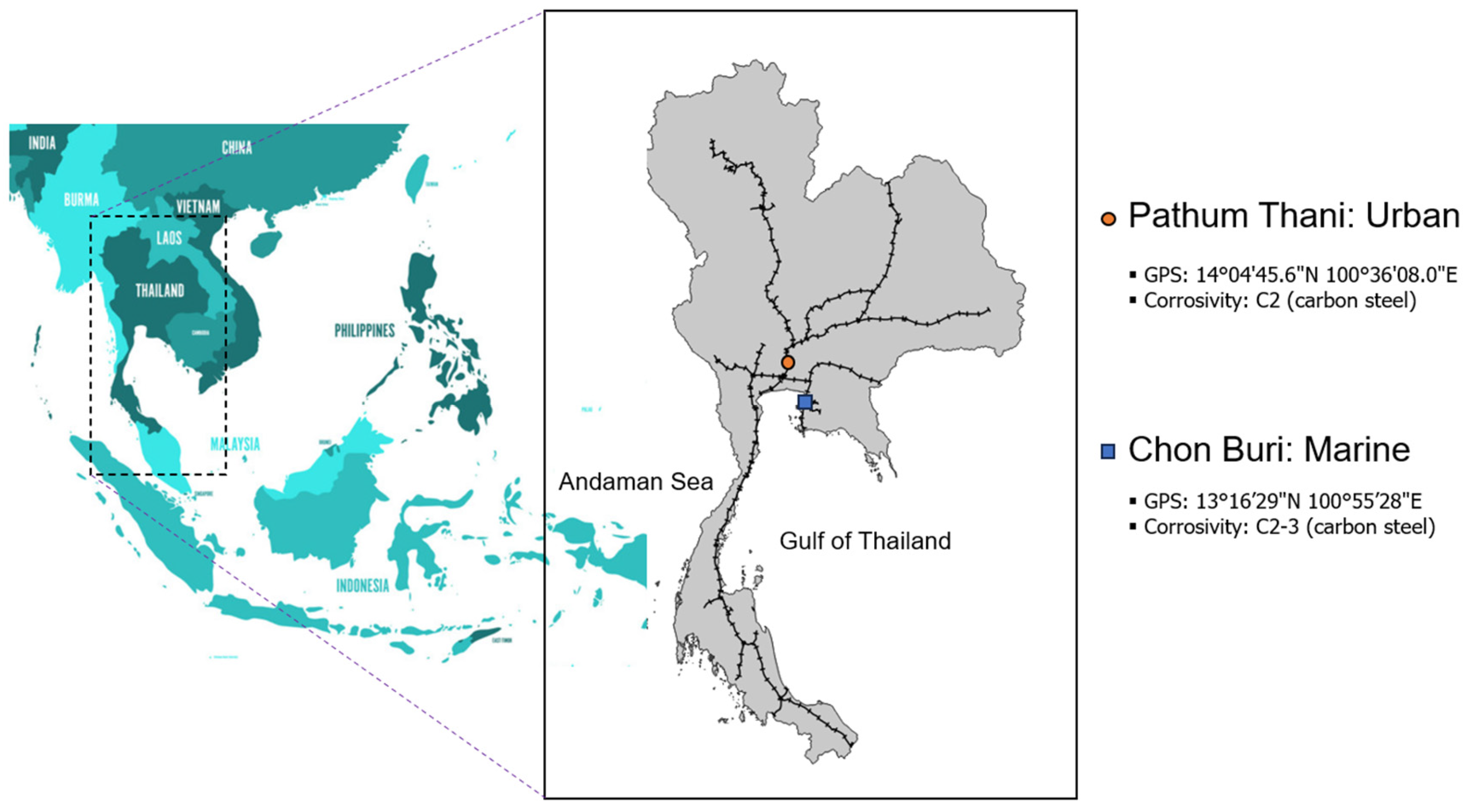

| No. | ID | GPS | Corrosivity (carbon steel) |
Distance from the sea (km) |
|---|---|---|---|---|
| 1. | Pathum Thani (PTI) | 14°04′45.6”N 100°36′08.0”E | C2 | 64 |
| 2. | Chon Buri (CBI) | 13°16′29”N 100°55′28”E | C2-C3 | 0.4 |
| Station | PTI | CBI | |||||
|---|---|---|---|---|---|---|---|
| Coating | 1 | 2 | 3 | 4 | 1 | 2 | 3 |
| MAE | 0.0142 | 0.0117 | 0.0176 | 0.0099 | 0.0138 | 0.0046 | 0.0052 |
| MSE | 0.0006 | 0.0004 | 0.0008 | 0.0003 | 0.0006 | 0.0001 | 0.0002 |
| R2 | 0.9872 | 0.9857 | 0.9777 | 0.9949 | 0.8878 | 0.9773 | 0.9775 |
| Cross validation R2 | 0.9524 | 0.9727 | 0.9638 | 0.9779 | 0.8374 | 0.9568 | 0.8670 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




