Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
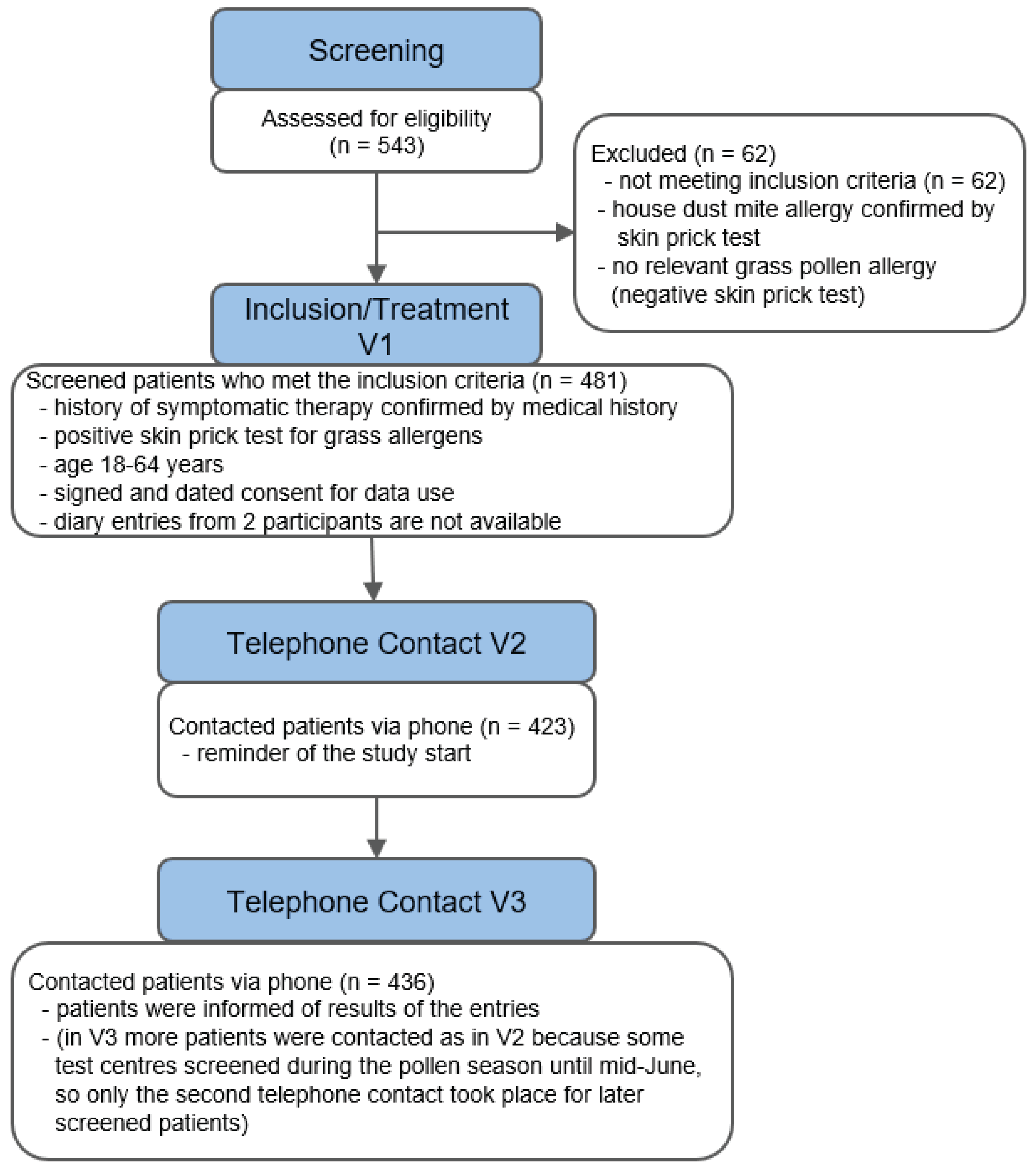
2.1. Study Design and Endpoints
2.2. Setting and Subjects
- Male, female, or diverse individuals interested in allergen-specific immunotherapy, with a history of at least 2 previous seasons in which symptomatic therapy was required confirmed by medical history and a positive SPT for grass allergens.
- Age 18–64 years with access to a smartphone or tablet computer.
- Provision of a signed and dated informed consent form for data use.
- Clinically relevant house dust mite allergy confirmed by SPT.
- Lack of access to a smartphone or tablet.
2.3. Clinical Endpoints
- 0 = no symptoms
- 1 = mild symptoms (present but minimally noticeable; easily tolerable)
- 2 = moderate symptoms (clearly perceptible; bothersome but tolerable)
- 3 = severe symptoms (difficult to tolerate; interferes with daily activities and/or sleep)
- 0 points: no medication
- 1 point: non-sedating oral antihistamines
- 1.5 points: nasal corticosteroids
- 0.5 points: antihistamine eye drops
2.4. Data Sources and Management
- Baseline patient characteristics: year of birth, age, gender, history of grass pollen allergy symptoms over at least the previous two years, and anti-allergic medication use over the same period.
- Diagnostic data: skin prick test results recorded on the observation form during the screening visit.
- Daily monitoring data: CSMS, VAS ratings for general allergy symptoms, nasal and ocular complaints, and asthma symptoms; daily asthma symptom scores; and medication intake, all recorded via the CCC STUDY Diary app.
2.5. Statistical Methods
2.6. Ethical Supervision
3. Results
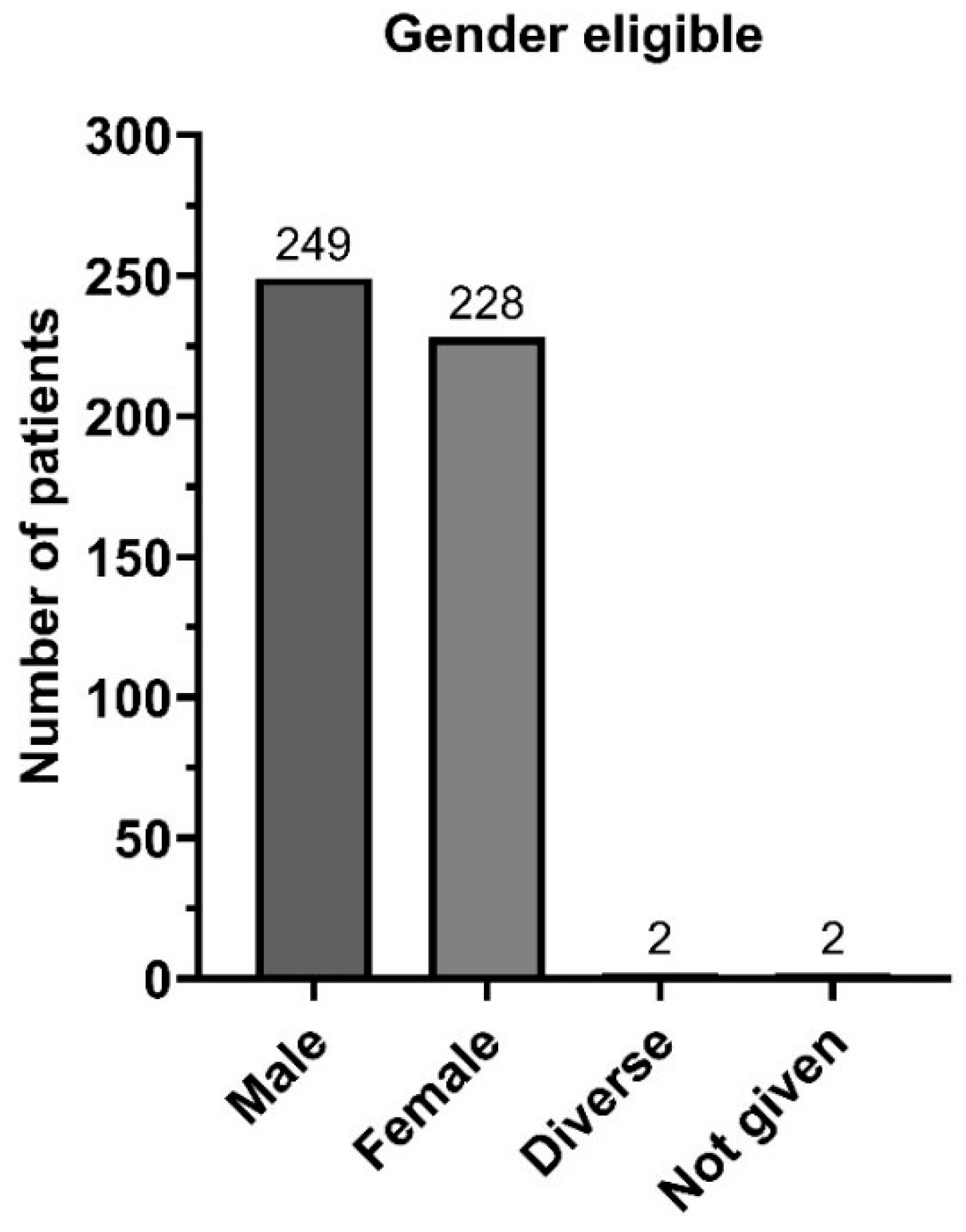
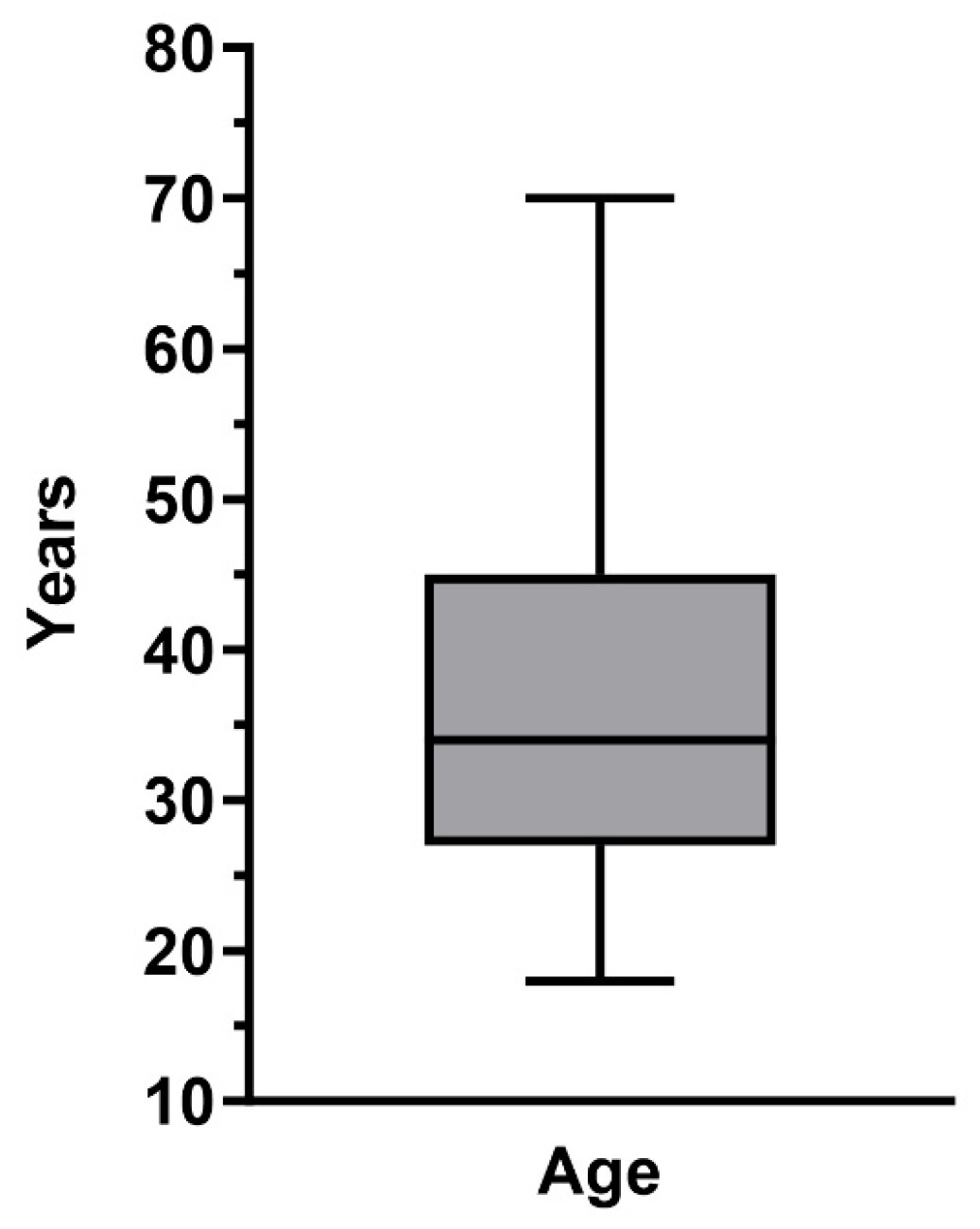
3.1. Baseline Data
3.2. Outcome Data
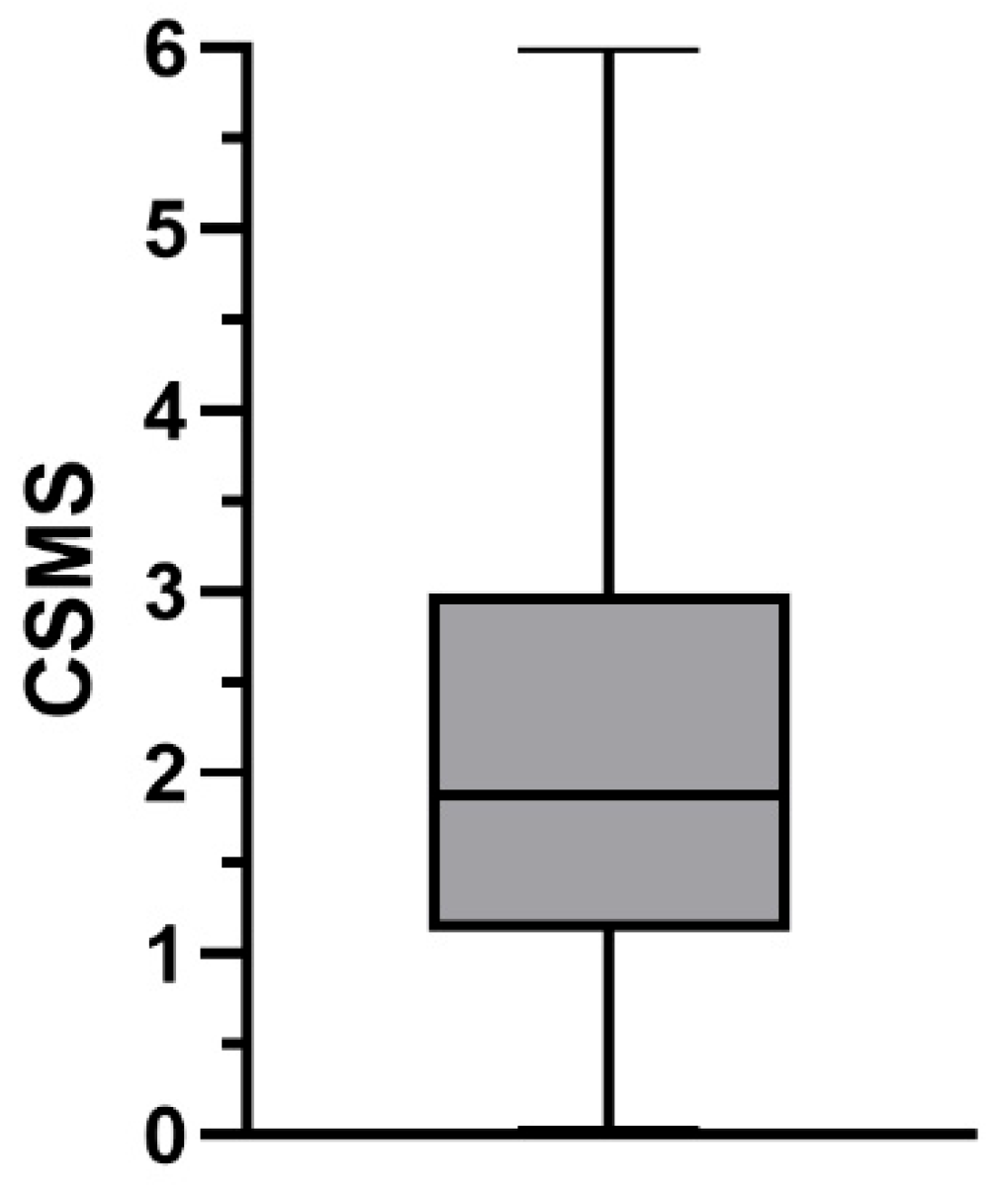
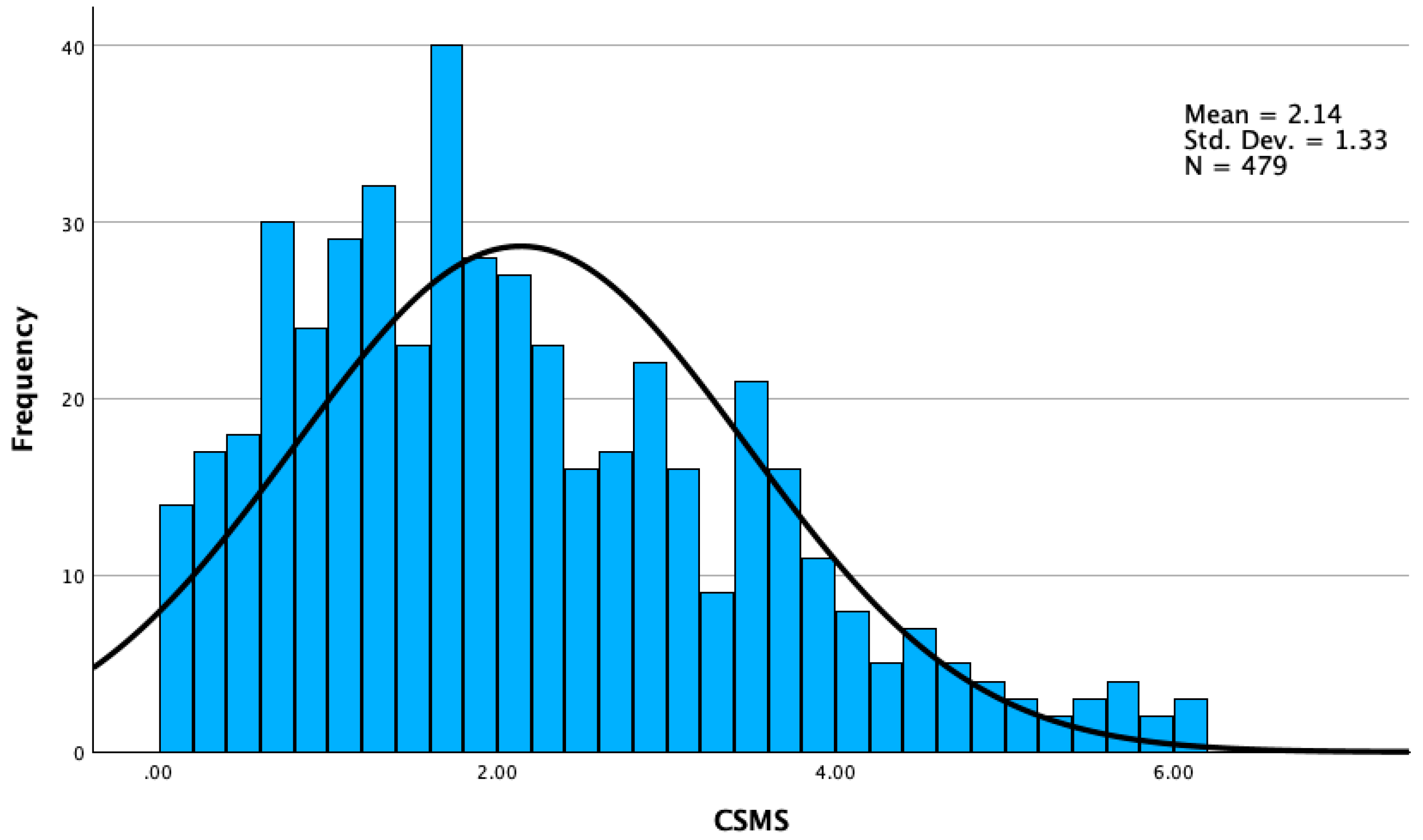
3.2.1. Additive CSMS
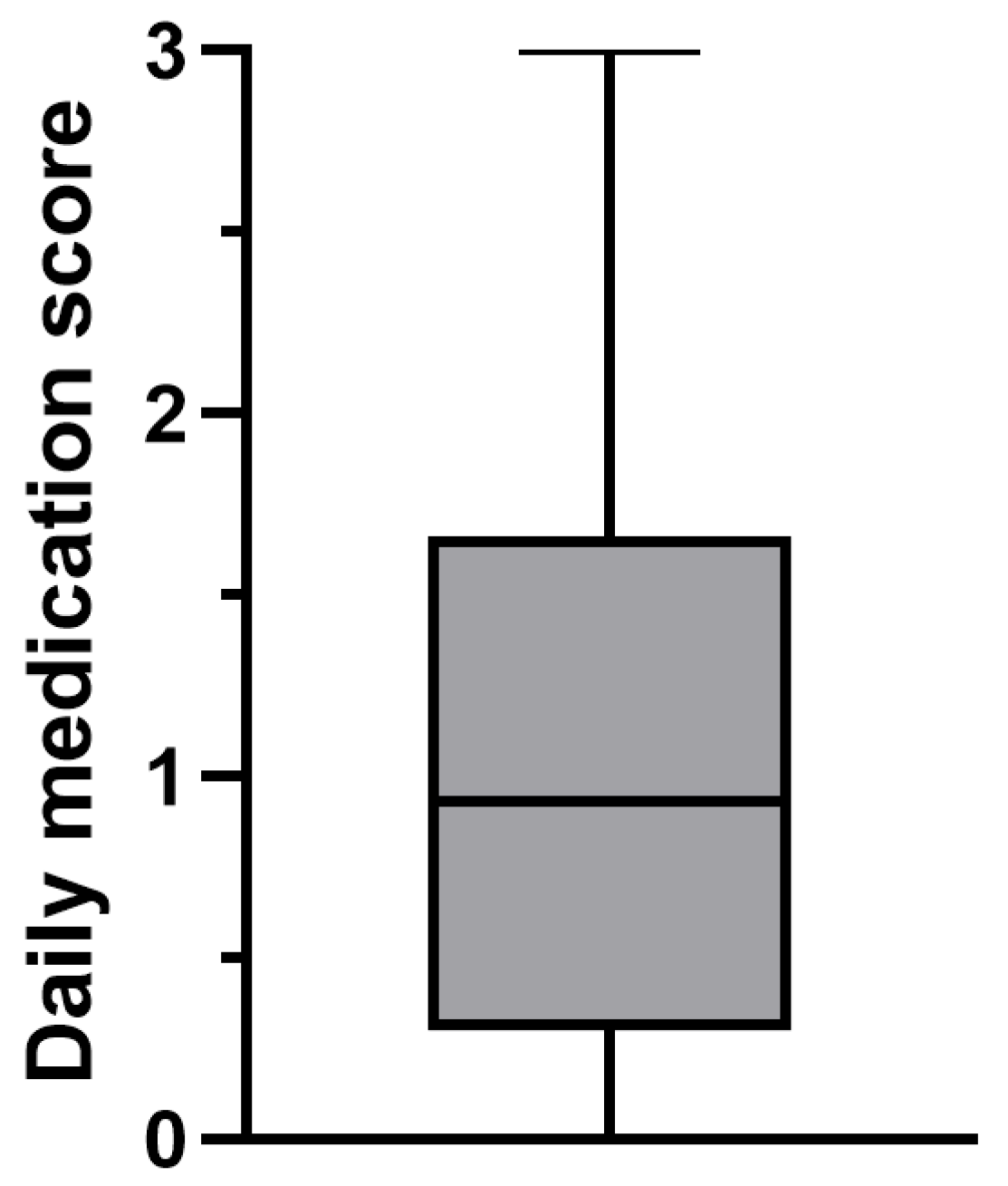
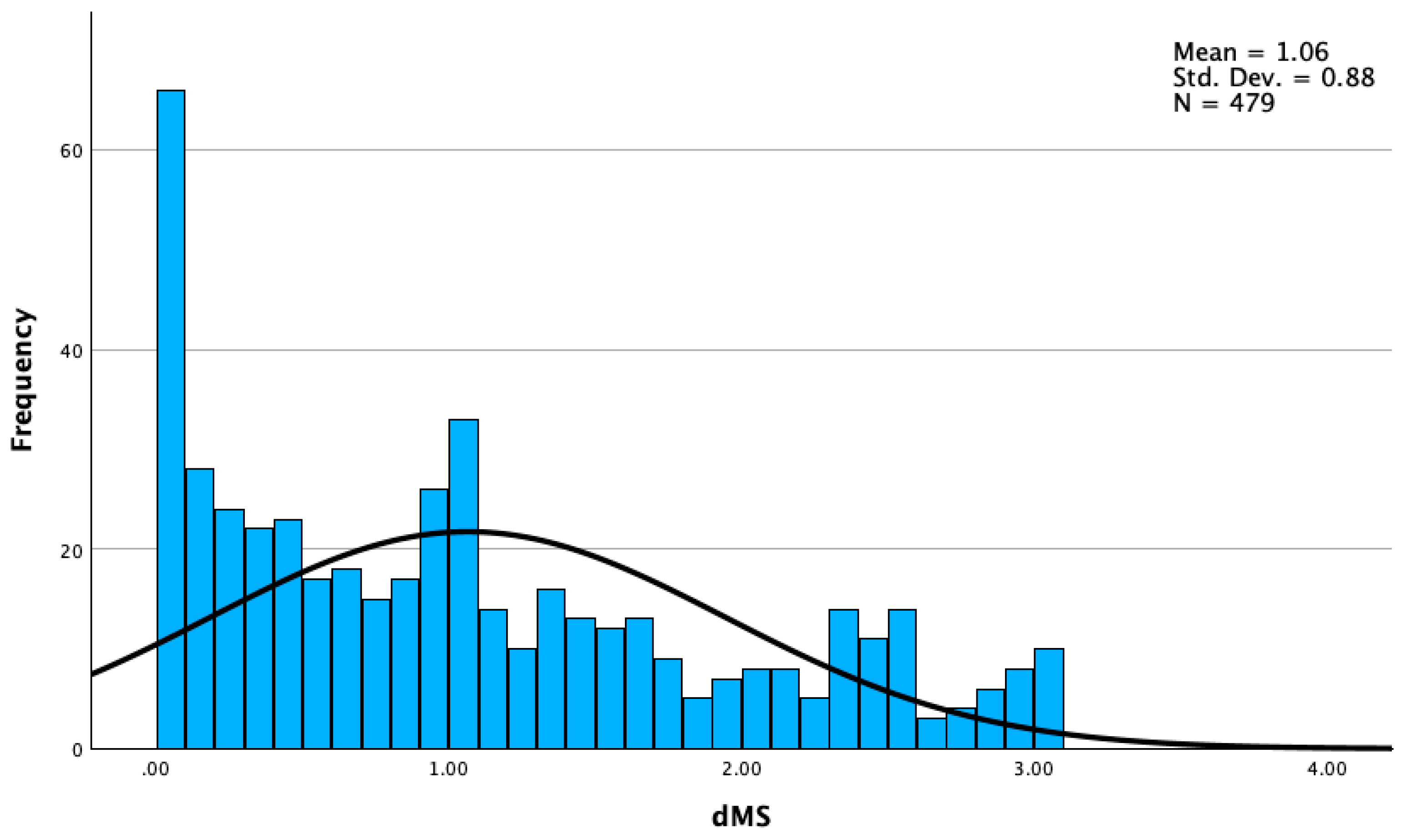
3.2.2. dMS
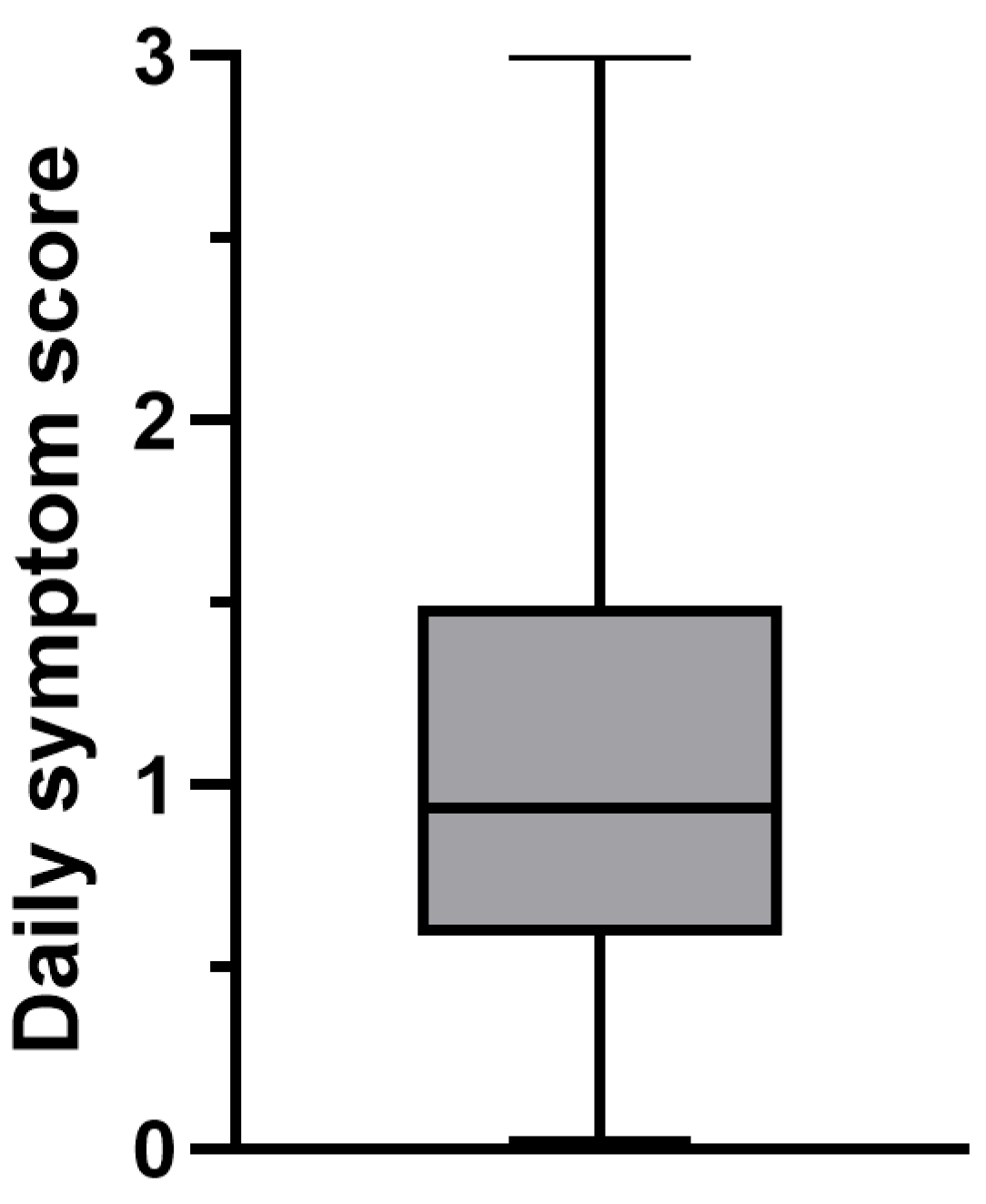
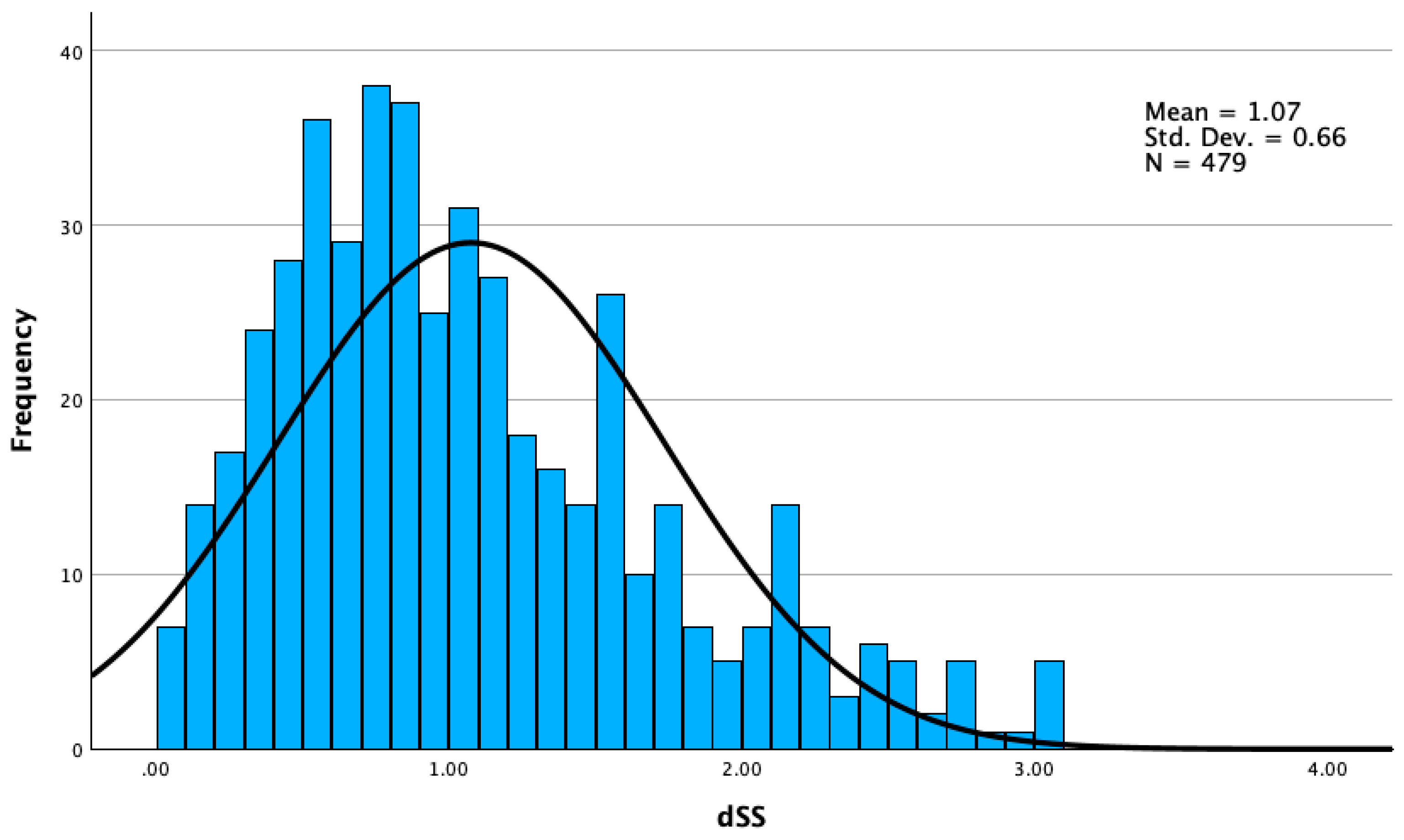
3.2.3. dSS
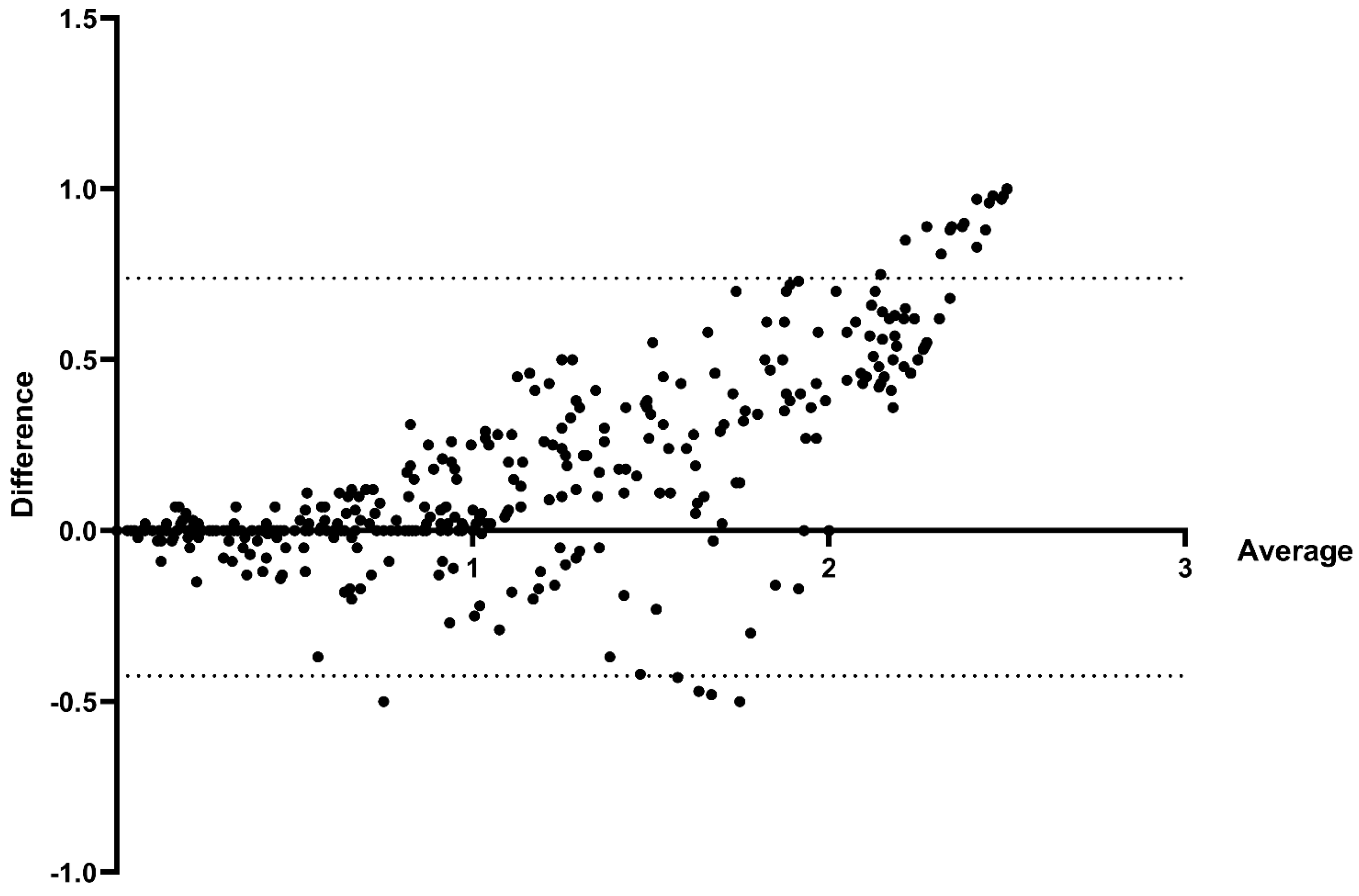
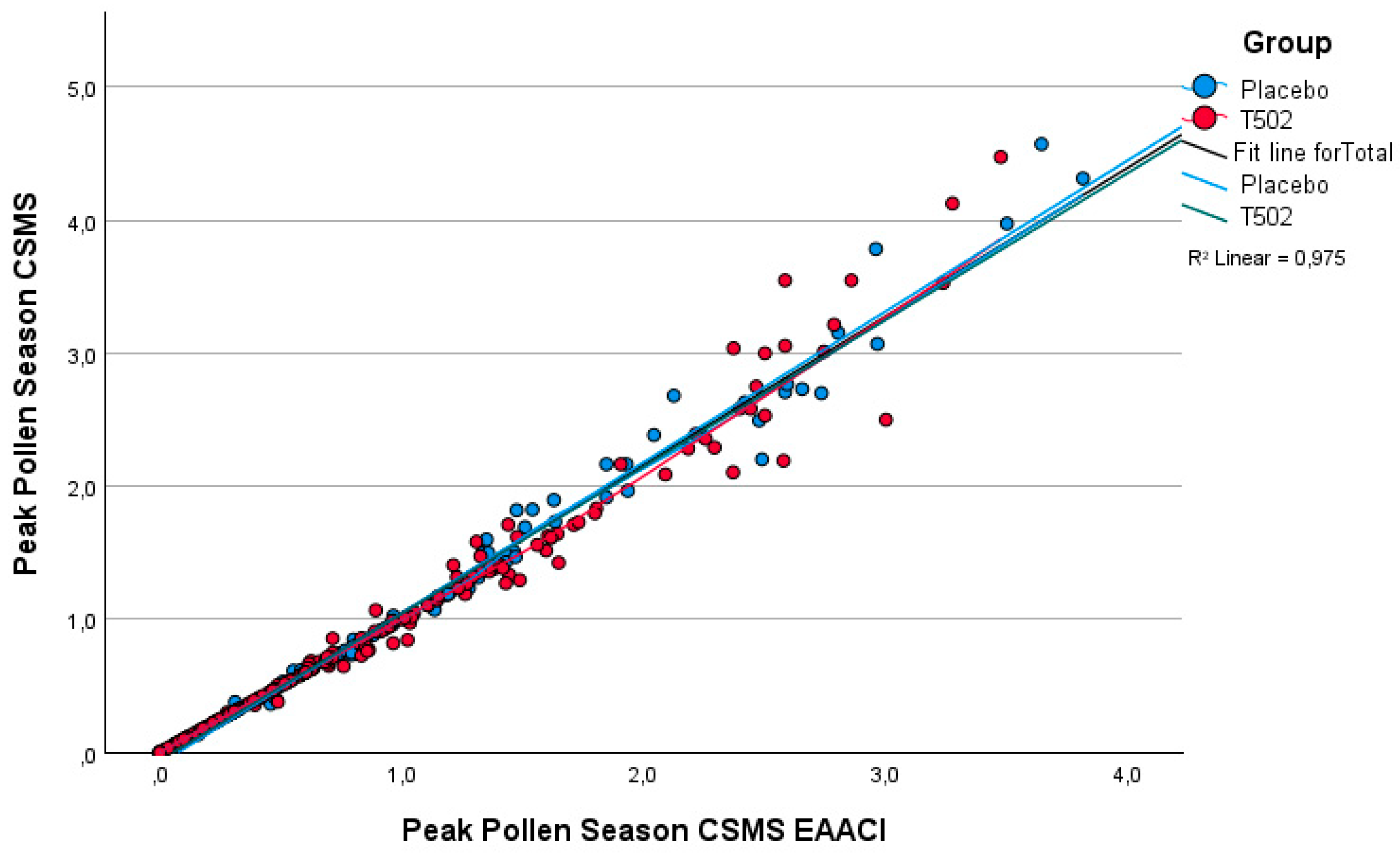
3.3. Comparison of the Two Methods of CSMS Collection (Additive vs. Classical)
- 1
- point – Oral and/or topical (eyes or nose) nonsedative H1 antihistamines H1A
- 2
- points – Intranasal corticosteroids (INS) with/without H1A
- 3
- points – Oral corticosteroids with/without INS, with/without H1A
3.4. Other analyses
3.4.1. Severity of Allergy Symptoms (Recorded by Visual Analogue Scale, VAS)
3.4.1. Asthma
4. Discussion
4.1. Limitations
4.2. Interpretation
4.3. Generalisability
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AIT | Allergen Immunotherapy |
| AMG | German Medicines Act (Arzneimittelgesetz) |
| CCC | ClinCompetence Cologne GmbH |
| CRO | Clinical Research Organisation |
| CSMS | Combined Symptom Medication Score |
| dSS | Daily Symptom Score |
| dMS | Daily Medication Score |
| DRKS | German Clinical Trials Register |
| EAACI | European Association of Allergy and Clinical Immunology |
| eCRF | Electronic Case Report Form |
| eDiary | Electronic patient Diary |
| EDC system | Electronic Data Capture System |
| IgE | Immunoglobulin E |
| IQR | Interquartile Range |
| NIS | Non-Interventional Study |
| p | (p-value) value for significance |
| P25 | Percentile 25 |
| P75 | Percentile 75 |
| SPT | Skin Prick Test |
| VAS | Visual Analogue Scale |
References
- Bachert, C.; Hoogeveen, H.; ten Have, R.; Yu, D.; Worm, M.; Pfaar, O.; Jutel, M.; Distler, A.; Bozek, A.; Opstelten, D.-J.; Narkus, A.; van Ree, R. Subcutaneous Allergen Immunotherapy in Adults Allergic to House Dust Mites: A Phase 3 Randomized Controlled Trial. Allergy 2025, 80, 3391–3400. [Google Scholar] [CrossRef] [PubMed]
- Pfaar, O.; Demoly, P.; van Wijk, R.G.; Bonini, S.; Bousquet, J.; Canonica, G.W.; Calderon, M.A. Recommendations for the standardization of clinical outcomes used in allergen immunotherapy trials for allergic rhinoconjunctivitis: An EAACI Position Paper. Allergy 2014, 69, 854–867. [Google Scholar] [CrossRef] [PubMed]
- Mosges, R.; Zeyen, C.; Raskopf, E.; Acikel, C.; Sahin, H.; Allekotte, S.; Cuevas, M.; Shamji, M.H.; Subiza, J.L.; Casanovas, M. A randomized, double-blind, placebo-controlled trial with mannan-conjugated birch pollen allergoids. Allergy 2023, 79, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- Hox, V.; Lourijsen, E.; Jordens, A.; et al. Benefits and harms of systemic steroids for short- and long-term use in rhinitis and rhinosinusitis: an EAACI position paper. Clin Transl Allergy 2020, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J; Heinzerling, L; Bachert, C; et al. Practical guide to skin prick tests in allergy to aeroallergens. Allergy 2012, 67(1), 18–24. [Google Scholar] [CrossRef] [PubMed]
- Mösges, R; Raskopf, E; Klimek, L; et al. Short-course subcutaneous treatment with birch pollen allergoids greatly improves symptom and medication scores in birch allergy. Allergy 2025, 80(3), 817–826. [Google Scholar] [CrossRef] [PubMed]
- Landesberger, V.; Huß, J.; Grenzebach, K.; et al. Association of grass pollen concentration and physical symptoms as well as impairments in day-to-day life in pollen allergy patients. Sci Rep 2025, 15, 18685. [Google Scholar] [CrossRef] [PubMed]
- Luyten, A; Bürgler, A; Glick, S; et al. Ambient pollen exposure and pollen allergy symptom severity in the EPOCHAL study. Allergy 2024, 79, 1908–1920. [Google Scholar] [CrossRef] [PubMed]
- Bastl, Maximilian; et al. Variability of grass pollen allergy symptoms throughout the season: comparing symptom data profiles from the Patient's Hayfever Diary from 2014 to 2016 in Vienna (Austria). World Allergy Organization Journal 2021, 14.3, 100518. [Google Scholar] [CrossRef] [PubMed]
- Durham, SR; Nelson, HS; Nolte, H; Bernstein, DI; Creticos, PS; Li, Z; Andersen, JS. The magnitude of efficacy measurements in grass allergy immunotherapy trials is highly dependent on pollen exposure. Allergy 2014, 69, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Novak, N; Worm, M; Staubach, P; Jutel, M; Sager, A; Pfaar, O. Subcutaneous birch pollen allergen immunotherapy with a depigmented polymerised extract shows only sustained and long-term efficacy in a subgroup of monosensitised adults and adolescents with allergic rhinitis. Clin Transl Allergy 2022, e12185. [Google Scholar] [CrossRef] [PubMed]
- Zielen, S.; Bernstein, J.A.; Sturm, G.J.; Jutel, M.; Pfaar, O.; the RESONATE Investigator Group; Shamji, M.H.; Mösges, R.; Berger, M.; Berger, U.E.; DuBuske, L.; Layhadi, J.A.; Klimek, L.; Ollert, M.; Skinner, M.A.; Kramer, M.F.; de Kam, P.-J. Six Injections of Modified Adjuvanted PQ Grass Is Effective and Well-Tolerated in a Pivotal Phase III Trial. Allergy 2025, 80, 1982–1994. [Google Scholar] [CrossRef] [PubMed]
- de Kam, PJ; Zielen, S; Bernstein, JA; et al. Short-course subcutaneous treatment with PQ Grass strongly improves symptom and medication scores in grass allergy. Allergy 2023, 78, 2756–2766. [Google Scholar] [CrossRef] [PubMed]
- Malling, H.-J.; Montagut, A.; Melac, M.; Patriarca, G.; Panzner, P.; Seberova, E.; Didier, A. Efficacy and safety of 5-grass pollen sublingual immunotherapy tablets in patients with different clinical profiles of allergic rhinoconjunctivitis. Clinical & Experimental Allergy 2009, 39, 387–393. [Google Scholar] [CrossRef]
- Murphy, K.; Gawchik, S.; Bernstein, D.; et al. A phase 3 trial assessing the efficacy and safety of grass allergy immunotherapy tablet in subjects with grass pollen-induced allergic rhinitis with or without conjunctivitis, with or without asthma. J Negat Results BioMed 2013, 12, 10. [Google Scholar] [CrossRef] [PubMed]
- Datz, N.; Arens, A.; Kordonouri, O. Die Hyposensibilisierung bleibt nur selten als Therapieoption übrig. hautnah dermatologie 2015, 31, 38–44. [Google Scholar] [CrossRef]
- Bousquet, J; Sousa-Pinto, B; Anto, JM; et al. Concurrent validity, cut-offs and ability to change of patient-reported outcome measures for rhinitis and asthma in MASK-air®. Clin Transl Allergy 2024, e12390. [Google Scholar] [CrossRef]
- Sousa-Pinto, B; Azevedo, LF; Jutel, M; et al. Development and validation of combined symptom-medication scores for allergic rhinitis. Allergy 2022, 77(7), 2147–2162. [Google Scholar] [CrossRef]
- Kvedariene, V; Biliute, G; Didziokaite, G; et al. Mobile health app for monitoring allergic rhinitis and asthma in real life in Lithuanian MASK-air users. Clin Transl Allergy 2022, 12(9), e12192. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Pinto, B; Schunemann, HJ; Sa-Sousa, A; et al. Consistent trajectories of rhinitis control and treatment in 16,177 weeks: the MASK-air(R) longitudinal study. Allergy 2023, 78(4), 968–983. [Google Scholar] [CrossRef] [PubMed]
| Timepoint | Visit | Assessments |
| May 2025 | Screening Visit | Introduction at the study centre with information, consent, allergen related medical history and previous anti-allergic medication use, skin prick test and, if suitable, activation of the CCC STUDY Diary app. |
| June 2025 | Telephone contact 1 | Beginning of June to start entries and documentation in the app from 1 June 2025 to 30 June 2025. |
| August 2025 | Telephone contact | Telephone consultation, discussion of the results from the app. |
| dSS | dMS | CSMS | |
| Count | 479 | 479 | 479 |
| Valid N | 479 | 479 | 479 |
| Mean | 1.07 | 1.06 | 2,14 |
| Standard Deviation | 0.66 | 0.88 | 1.33 |
| Maximum | 3.00 | 3.00 | 6.00 |
| Median | 0.94 | 0.93 | 1.88 |
| Minimum | 0.02 | 0,00 | 0,02 |
| Percentile 25 | 0.59 | 0.30 | 1.13 |
| Percentile 75 | 1.49 | 1.66 | 2.99 |
| 95,0% Lower CL for Mean | 1.02 | 0.99 | 2.02 |
| 95,0% Upper CL for Mean | 1.13 | 1.14 | 2.26 |
| dMS | CSMS | |||
| Additive | Weighted | Additive | Weighted | |
| Total N | 479 | 479 | 479 | 479 |
| Valid N | 479 | 479 | 479 | 479 |
| Mean | 1.06 | 0.91 | 2.14 | 1.98 |
| Standard Deviation | 0.88 | 0.66 | 1.33 | 1.12 |
| Standard Error of Mean | 0.04 | 0.03 | 0.06 | 0.05 |
| Minimum | 0.00 | 0.00 | 0.02 | 0.02 |
| Percentile 25 | 0.30 | 0.32 | 1.13 | 1.12 |
| Median | 0.93 | 0.88 | 1.88 | 1.86 |
| Percentile 75 | 1.66 | 1.47 | 2.99 | 2.73 |
| Maximum | 3.00 | 2.00 | 6.00 | 5.00 |
| p* | <0.001 | <0.001 | ||
| Spearmans rho | 0.983 | 0.990 | ||
| p** | <0.001 | <0.001 | ||
| Coefficients | |||||
| Model | Unstandardized Coefficients | Standardized Coefficients | t | Sig. | |
| B | Std. Error | Beta | |||
| (Constant) | -0.109 | 0.018 | -6.169 | <0.001 | |
| dMS_weighted_mean | 1.293 | 0.016 | 0.966 | 82.079 | <0.001 |
| a. Dependent Variable: dMS_additive_mean | |||||
| (Constant) | -0.186 | 0.021 | -8.917 | <0.001 | |
| CSMS_weighted_mean | 1.173 | 0.009 | 0.986 | 127.924 | <0.001 |
| a. Dependent Variable: CSMS_additive_mean | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




