Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Synthesis of Thiol–Functionalized PVA (PVA–SH)
2.3. Preparation of UV–Curable L(-)−Borneol−Functionalized Polyurethane Acrylate Prepolymers (LB−PUAs)
2.4. Fabrication of UV−Curable Hydrogels (UV−LBs)
2.5. Analytical Methods
2.5.1. Fourier–Transform Infrared (FT–IR) Spectroscopic Analysis
2.5.2. TGA (Thermogravimetric Analysis) Experiment
2.5.3. Swelling Performance
2.5.4. DMA Analysis
2.5.5. In Vitro Antibacterial Property Study
2.5.6. Bacterium and Fungi Morphology Study
2.5.7. In Vitro Cytotoxicity Assay
2.5.8. Packaging Investigation of Fresh−Cut Banana and Cherry Tomato
2.5.9. Statistical Analysis
3. Results and Discussion
3.1. FT–IR Analysis
3.2. Thermal Stability Investigation of UV−LBs
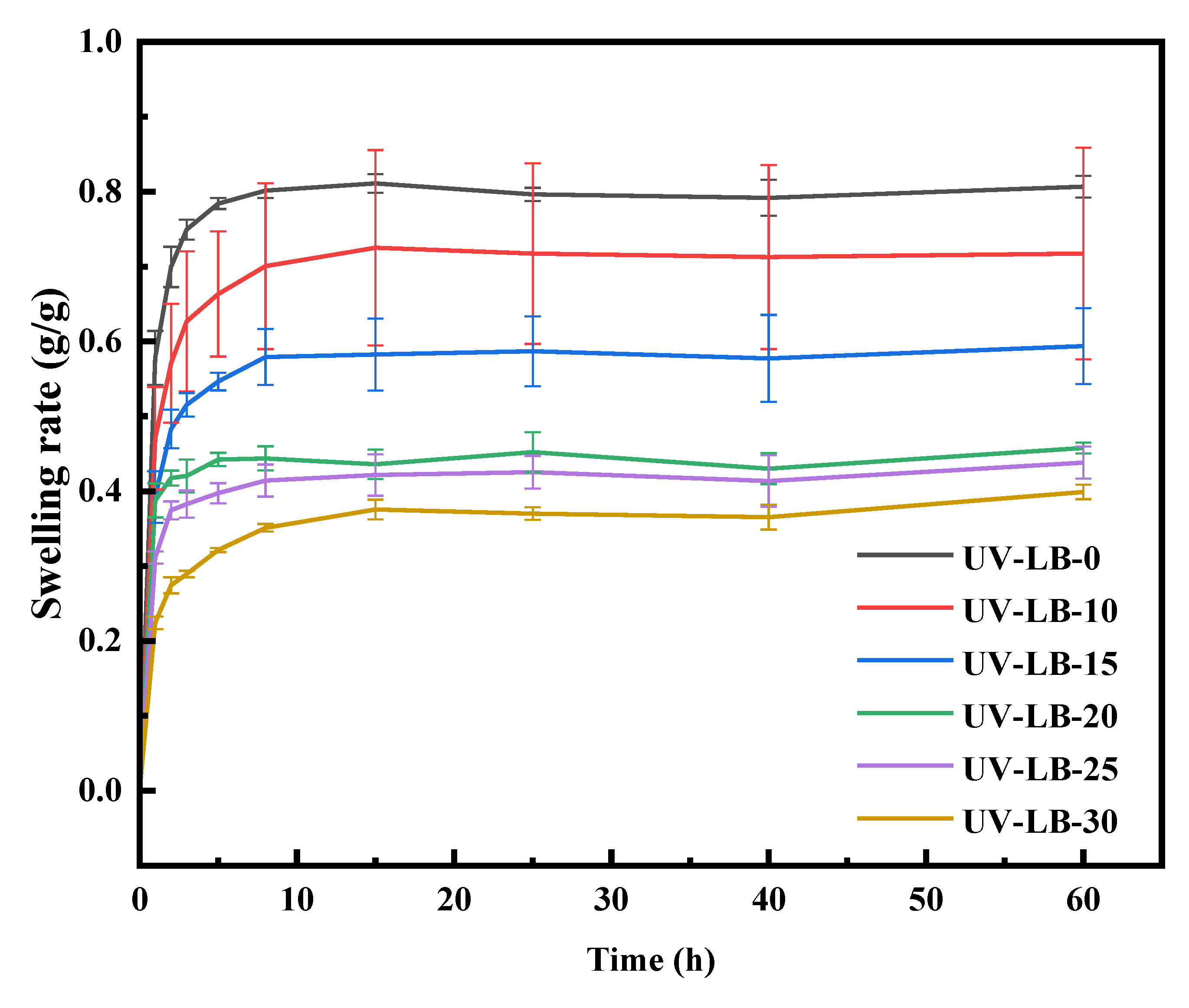
3.3. Swelling Behavior of UV−LBs
3.4. DMA Analysis of UV−LBs
3.5. Antibacterial Property of UV−LBs
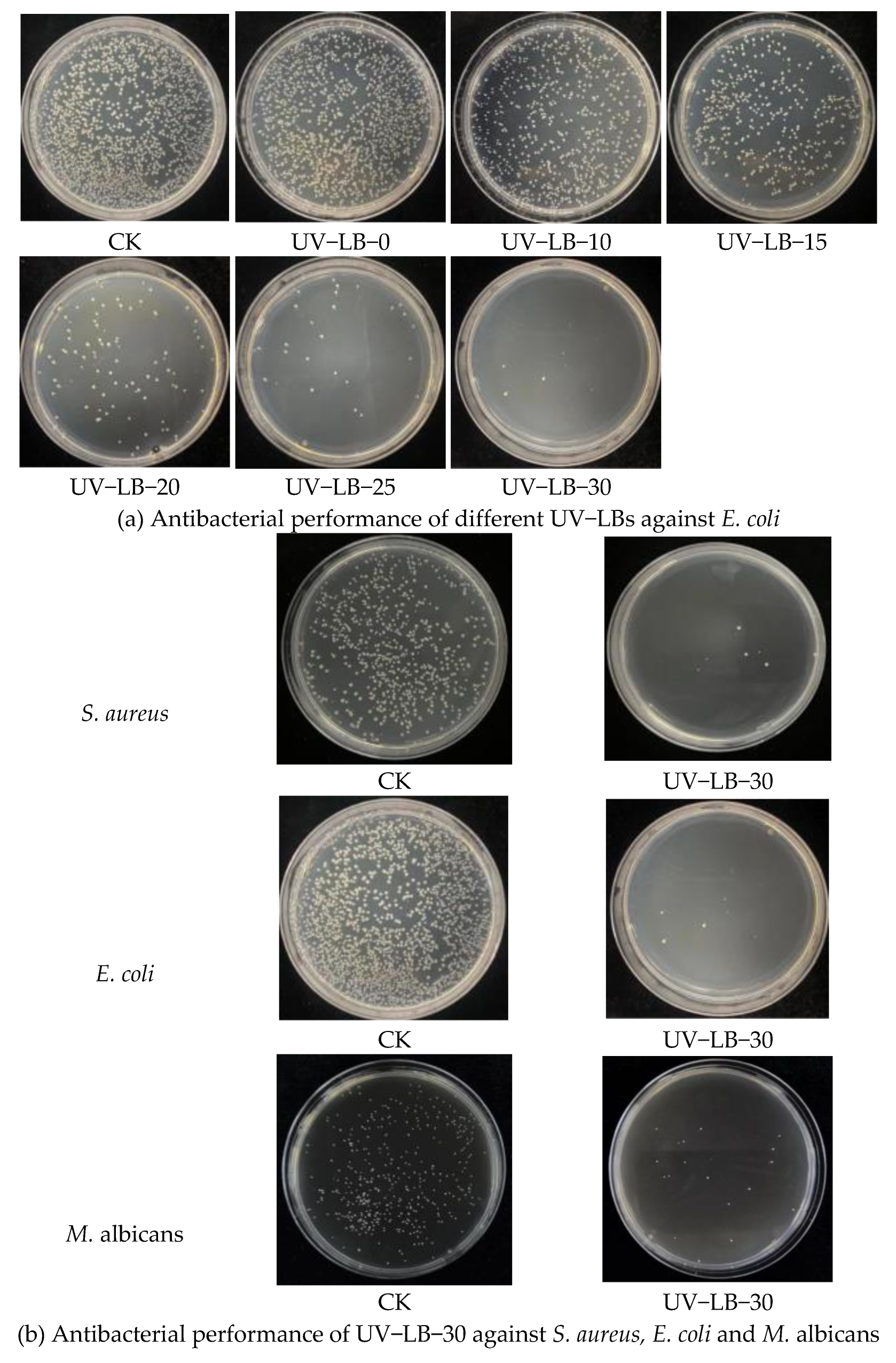
3.5.1. Antibacterial Property Evaluated with the Coated Plates and Colony Count Method
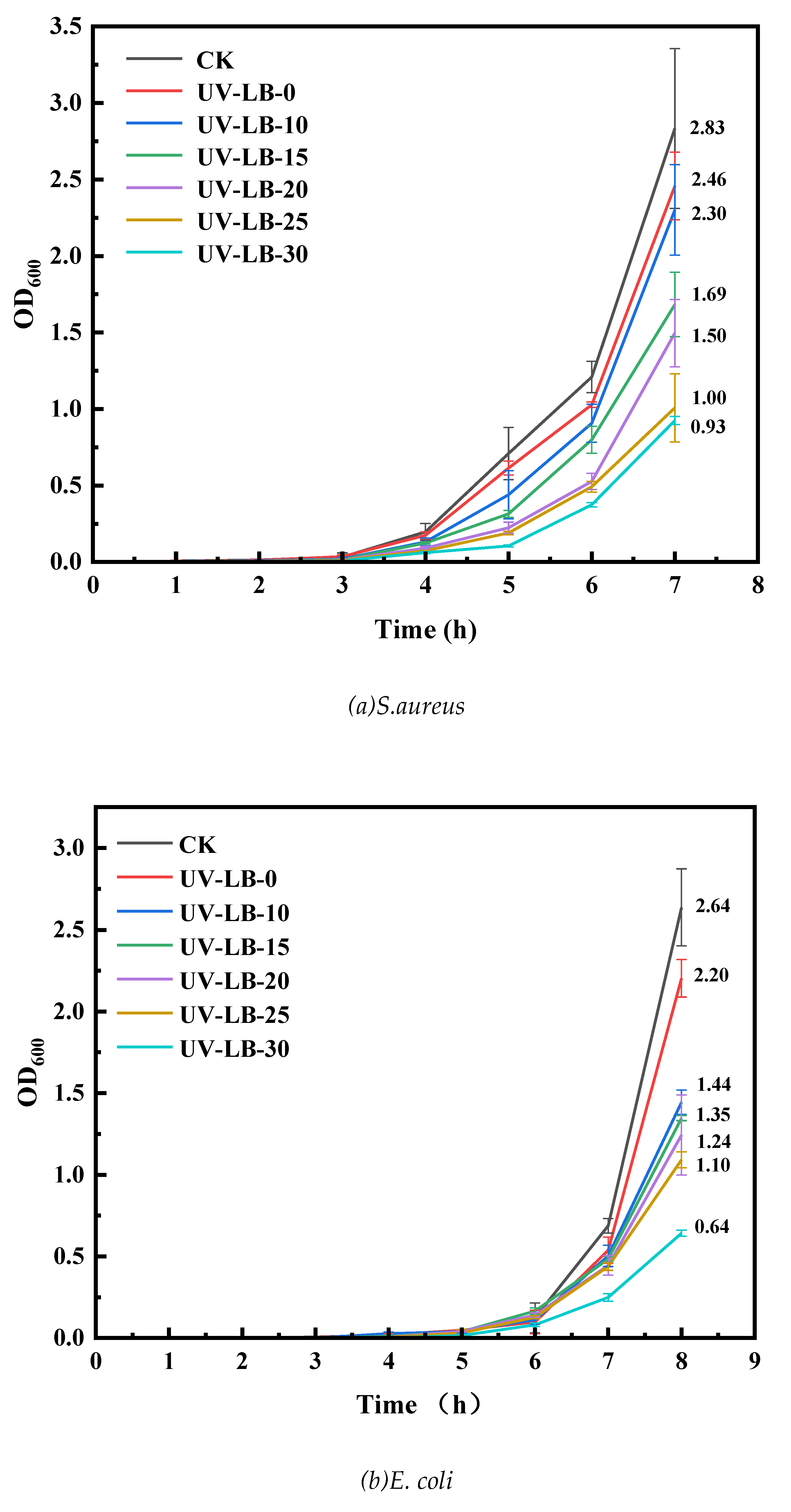
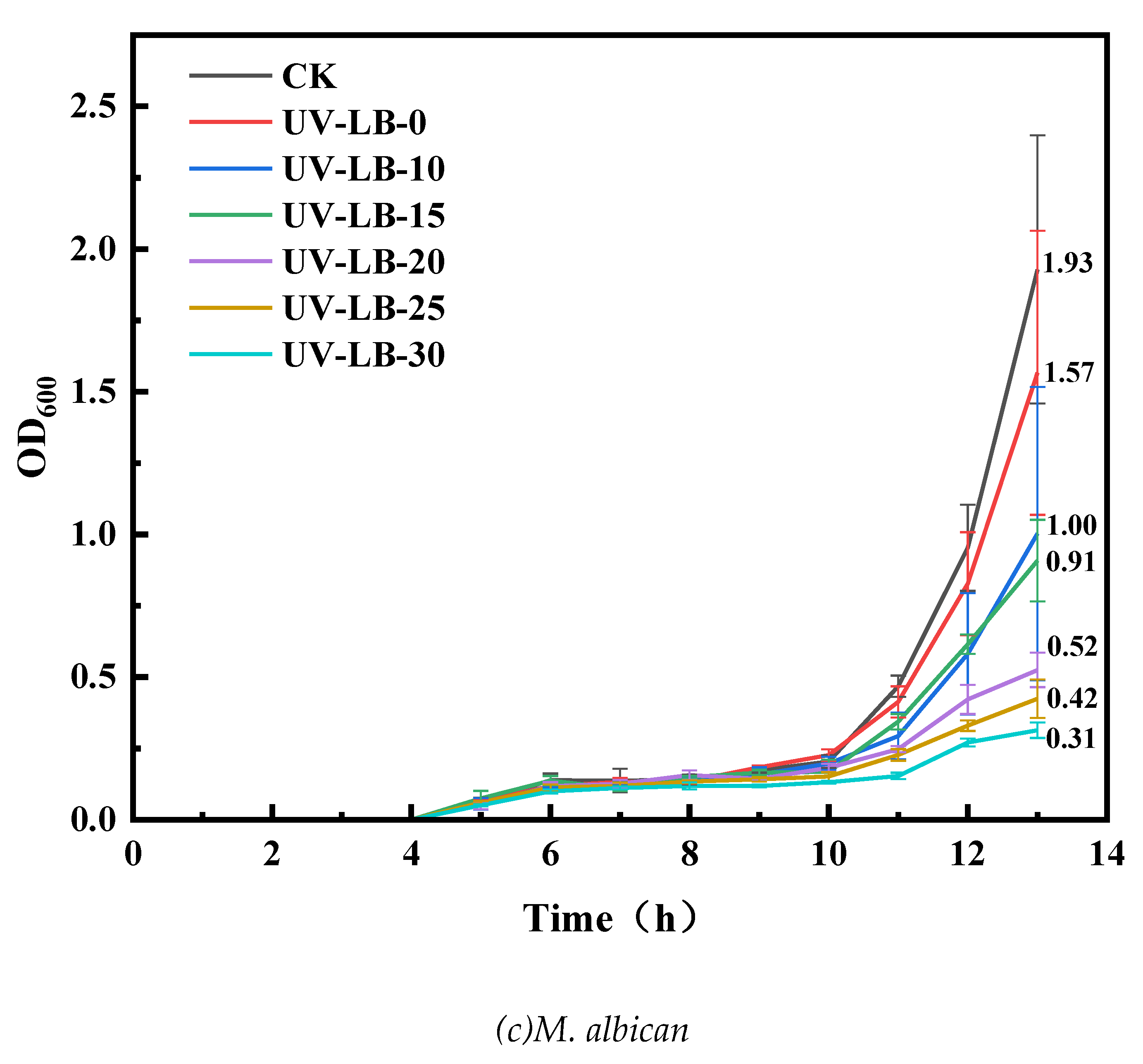
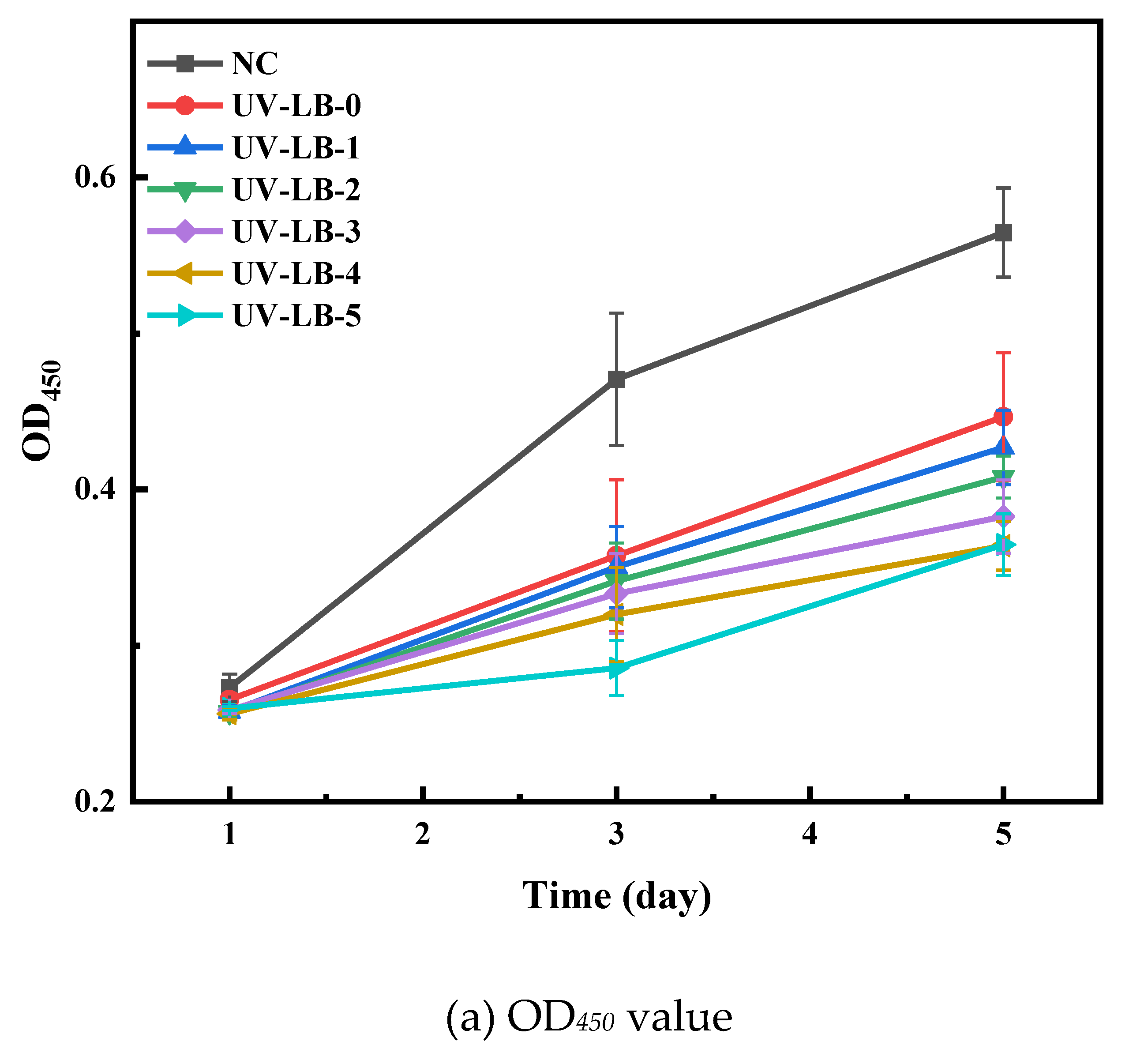
3.5.2. OD600 Evaluation of UV−LBs Using Co−Culturing Method
3.5.3. Bacterium and Fungi Morphology Study
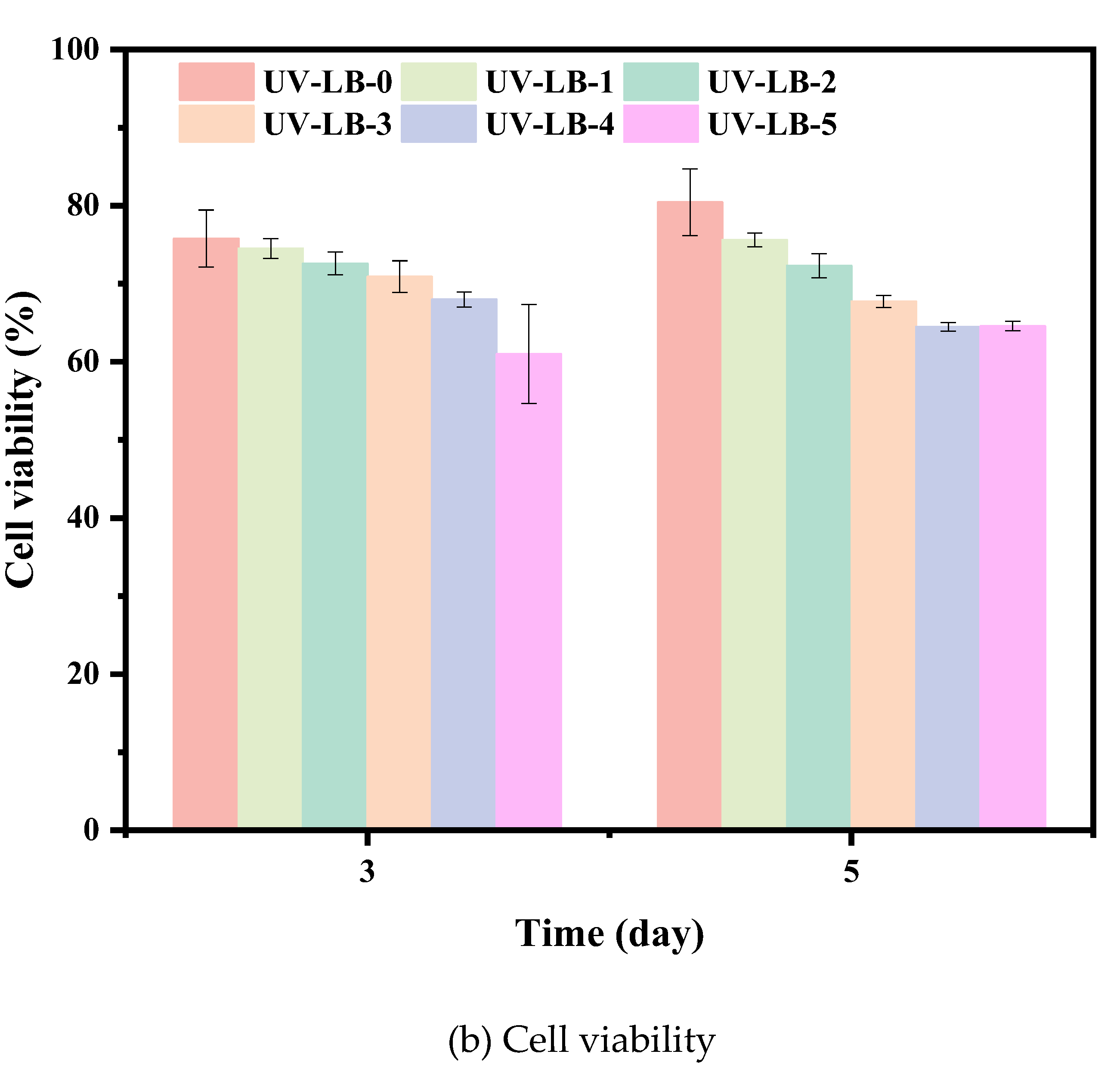
3.6. Cytotoxicity Assay
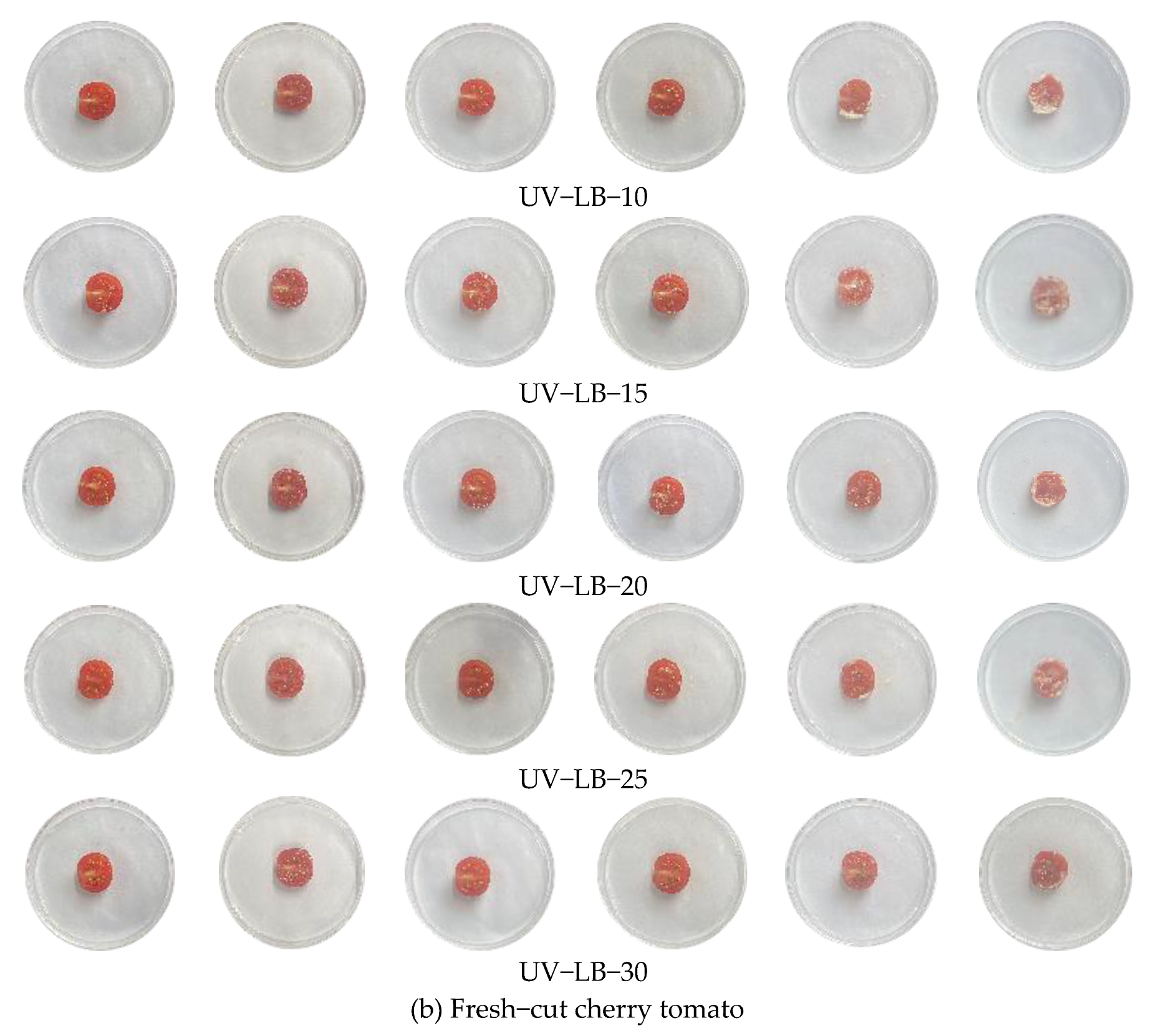
3.7. Packaging Experiment of Resh−Cut Banana and Cherry Tomato
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Han, P.; Sun, J. Y.; Mao, S.; Li, F. Y.; Yan, X. X.; Zhang, T. H.; Lu, C. W. Multifunctional pectin−based films containing schiff base triggered by pH microenvironment for freshness monitoring and preservation of fresh−cut papayas. Food Hydrocolloids 2024, 157, 110417. [Google Scholar] [CrossRef]
- Pan, X. L.; Duan, Y.; Liu, S.; Wang, Y.; Li, Q.; Jiang, F. C.; Li, Y. X.; Huang, Z.; Su, L. J.; Li, X. B.; Liu, M. Y.; Zhou, X.; Tang, H. C. All−in−one: Harnessing multifunctional natural polysaccharide spray hydrogel loaded with polyphenol−metal nanoparticles for fruit preservation. Food Chemistry 2025, 470, 142638. [Google Scholar] [CrossRef]
- Dwibedi, V.; Kaur, G.; George, N.; Rana, P.; Ge, Y. H.; Sun, T. Research progress in the preservation and packaging of fruits and vegetables: From traditional methods to innovative technologies. Food Packaging and Shelf Life 2024, 46, 101385. [Google Scholar] [CrossRef]
- Ganesh, K. S.; Sridhar, A.; Vishali, S. Utilization of fruit and vegetable waste to produce value−added products: Conventional utilization and emerging opportunities A review. Chemosphere 2022, 287, 132221. [Google Scholar] [CrossRef] [PubMed]
- Han, C. Q.; Wang, W. X.; Suo, B. X.; Zhang, J.; Ma, Q. Y.; Sun, J. F. Sodium nitroprusside affects energy metabolism, gamma−aminobutyric acid branching, and circRNA regulation of fresh−cut purple potatoes. Postharvest Biology and Technology 2025, 227, 113599. [Google Scholar] [CrossRef]
- Qiao, L. P.; Hou, X. R.; Li, X. K.; Hu, N. J.; Yang, X.; Wang, Y. S.; Li, X. H.; Lu, L. F.; Liu, X. Glutamate induction of whole potatoes alleviated the browning of fresh cuts: Jasmonate signalling may play a key role. Food Chemistry 2025, 482, 144138. [Google Scholar] [CrossRef] [PubMed]
- Liu, G. C.; Liu, R.; Fu, C. Y.; Hu, X.; Li, K.; Yan, L. K.; Yang, P.; Zhao, J. Protein/polysaccharide composite nanocoating based on amyloid−like aggregation for fresh−cut fruits preservation. Chemical Engineering Journal 2025, 519, 165048. [Google Scholar] [CrossRef]
- Liu, L. L.; Zhang, Y. Y.; Jin, L. X.; Abdollahi, M.; Zhao, G. L.; Venkatachalam, K.; Ban, Z. J. Controlled release and stability enhancement of cinnamon essential oil in glutathione−modified soy protein particles: Its antimicrobial application for fresh−cut cantaloupe. Food Research International 2025, 211, 116523. [Google Scholar] [CrossRef]
- Treviño−Garza, M. Z.; García, S.; Heredia, N.; Alanís−Guzmán, M. G.; Arévalo−Niño, K. Layer−by−layer edible coatings based on mucilages, pullulan and chitosan and its effect on quality and preservation of fresh−cut pineapple (Ananas comosus). Postharvest Biology and Technology 2017, 128, 63–75. [Google Scholar] [CrossRef]
- Alessio, A.; Eugenia, G.; Casales, F. G.; Gimenez, M. J.; Alessandra, G.; Gugino, I. M.; Daniela, P.; Giuseppe, S. Extending shelf−life of fresh−cut apples using manna from ash tree (Fraxinus angustifolia) as natural antioxidant agent in comparision with calcium ascorbate. Postharvest Biology and Technology 2024, 214, 112986. [Google Scholar]
- Wang, D.; Li, D.; Xu, Y. Q.; Li, L.; Belwal, T.; Zhang, X. C.; Luo, Z. S. Elevated CO2 alleviates browning development by modulating metabolisms of membrane lipids, proline, and GABA in fresh−cut Asian pear fruit. Scientia Horticulturae 2021, 281, 109932. [Google Scholar] [CrossRef]
- Sharma, R.; Nath, P. C.; Das, P.; Rustagi, S.; Sharma, M.; Sridhar, N.; Hazarika, T. K.; Rana, P.; Nayak, P. K.; Sridhar, K. Essential oil−nanoemulsion based edible coating: Innovative sustainable preservation method for fresh/fresh−cut fruits and vegetables. Food Chemistry 2024, 460, 140545. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.−Y.; Chen, T.−Y.; Chuang, W.−T.; Hsu, S.−H. Self−assembled chitosan−boronic acid hydrogel as dynamic crosslinker to produce 3D−printable glucose−sensitive hydrogel. Carbohydrate Polymers 2025, 363, 123737. [Google Scholar] [CrossRef] [PubMed]
- Wang, X. J.; Zhu, H. Y.; Yang, Y. N.; Lai, G. Q.; Yang, X. F. UV−curable choline chloride and bromophenol red covalent functionalized chitosan antibacterial and pH−sensitive hydrogels. Food Hydrocolloids 2024, 154, 110103. [Google Scholar] [CrossRef]
- Wang, X. J.; Yuan, J. Z.; Sun, N. N.; Jiang, Y. H.; Yu, Y. C.; Lai, G. Q.; Yang, X. F. UV−Curable antibacterial and pH−sensitive eugenol functionalized chitosan−polyurethane hydrogels for shelf−life extension of chicken. Food Control 2025, 168, 110918. [Google Scholar] [CrossRef]
- Mao, L.; Wang, C. Y.; Dong, Z. Y.; Yao, J.; Dong, F.; Dai, X. L. Fabrication of polylactic acid bilayer composite films using polyvinyl alcohol based coatings containing functionalized carbon dots and layered clay for active food packaging. Industrial Crops and Products 2025, 225, 120460. [Google Scholar] [CrossRef]
- Wu, J. J.; Zhang, Y.; Zhang, F.Y.; Mi, S.; Yu, W. L.; Sang, Y. X.; Wang, X. H. Preparation of chitosan/polyvinyl alcohol antibacterial indicator composite film loaded with AgNPs and purple sweet potato anthocyanins and its application in strawberry preservation. Food Chemistry 2025, 463, 141442. [Google Scholar] [CrossRef]
- Sheng, W. Y.; Yang, L.; Yang, Y. C.; Wang, C. Z.; Jiang, G. Y.; Tian, Y. Q. Photo−responsive Cu−tannic acid nanoparticle−mediated antibacterial film for efficient preservation of strawberries. Food Chemistry 2025, 464, 141711. [Google Scholar] [CrossRef]
- Zhu, Y. D.; Pang, X. H.; Zhang, W. L.; Zhang, C.; Zhang, B. L.; Fu, J. M.; Zhao, H. F.; Han, W. J. Green synthesis of silver nanoparticles using persimmon polysaccharides for enhanced polysaccharide−based film performance. Food Research International 2025, 209, 116252. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, Z.; Yin, J.; Shen, L. Y.; Zhu, L. L.; Redshaw, C.; Zhang, Q. L. Block cationic copolymer/quaternary ammonium chitosan−based composite antibacterial hydrogel dressings with NIR photothermal effects for bacteria−infected wound healing. International Journal of Biological Macromolecules 2025, 288, 138716. [Google Scholar] [CrossRef]
- Cai, Z. H.; Wang, L. Y.; Zhang, Q. Q.; Yang, W. Y.; Zhang, C.; Wang, H.; Xiao, H. M. Eco−friendly coating engineered with antimicrobial lipopeptides maintains freshness and induces genes expression in anthocyanin biosynthesis of blueberry. International Journal of Biological Macromolecules 2025, 306, 141590. [Google Scholar] [CrossRef] [PubMed]
- Li, C. H.; Zhao, Y. X.; Zhang, A. J.; Xu, Y. C.; Wang, H. Y. Preparation, characterization, and antibacterial properties of a soybean protein isolate/gelatin composite film containing rosemary−modified bentonite and application of fresh lemon slices. International Journal of Biological Macromolecules 2025, 308, 142516. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.T. S.; Reddy, J. P.; Vanajakshi, V.; Dasalkar, A. H.; Yannam, S. K.; Hebbar, U. H.; Singh, S. A. Development and characterization of carrageenan−based antibacterial films incorporated with natural melanin pigment from niger seed hulls (Guizotia abyssinica) and their efficacy to enhance the shelf−life of strawberries. Food Control 2025, 174, 111235. [Google Scholar] [CrossRef]
- Lu, Z. W.; Mu, J. B.; Guan, C. W.; Sui, T. S.; Liu, C. Z.; Guo, Z. C.; Liao, S. M. Green and recyclable photocatalytic hydrogel film with antibacterial and ethylene scavenging properties for fruit preservation. Food Chemistry 2025, 475, 143266. [Google Scholar] [CrossRef]
- Yang, H. J.; Li, L. P.; Li, C.; Xu, Z. H.; Tao, Y. H.; Lu, J.; Xia, X. D.; Tan, M. Q.; Du, J.; Wang, H. S. Multifunctional and antimicrobial carboxymethyl cellulose−based active hydrogel film for fruits packaging and preservation. Food Bioscience 2024, 59, 104005. [Google Scholar] [CrossRef]
- Yu, K. J.; Zhang, S. Y.; Yang, L. N.; Liu, H.; Li, X. P.; Xu, Y. X.; Li, J. R. Strong, tough, antibacterial, antioxidant, biodegradable multi−functional intelligent hydrogel film for real−time detection and maintenance of salmon freshness. Food Research International 2025, 201, 115594. [Google Scholar] [CrossRef]
- Gong, W.; Yang, T. Q.; He, W. Y.; Li, Y. X.; Hu, J. N. On−demand removable hydrogel film derived from gallic acid−phycocyanin and polyvinyl alcohol for fruit preservation. Food Chemistry 2025, 463, 141404. [Google Scholar] [CrossRef]
- Wu, Y. B.; Gu, Z. M.; Chen, T. T.; Zu, D. T.; Gan, Y. H.; Chen, H. L.; Yang, J. N.; Yu, X.; Cai, H. H.; Sun, P. H.; Ning, J. Y.; Zhou, H. B.; Zheng, J. X. Effect of different crosslinking agents on carboxymethyl chitosan−glycyrrhizic acid hydrogel: Characterization and biological activities comparison. International Journal of Biological Macromolecules 2025, 298, 139977. [Google Scholar] [CrossRef]
- Qian, Y.; Zheng, Y.; Jin, J.; Wu, X.; Xu, K.; Dai, M.; Niu, Q.; Zheng, H.; He, X.; Shen, J. Immunoregulation in diabetic wound repair with a photoenhanced glycyrrhizic acid hydrogel scaffold. Advanced Materials 2022, 34(29), 2200521. [Google Scholar] [CrossRef]
- Laquerbe, S.; Sayed, J. E.; Lorthioir, C.; Meyer, C.; Narita, T.; Ducouret, G.; Perrin, P.; Sanson, N. Supramolecular crosslinked hydrogels: similarities and differences with chemically crosslinked hydrogels. Macromolecules 2023, 56(18), 7406–7418. [Google Scholar] [CrossRef]
- Mehta, P.; Sharma, M.; Devi, M. Hydrogels: an overview of its classifications, properties, and applications. Journal of the Mechanical Behavior of Biomedical Materials 2023, 147, 106145. [Google Scholar] [CrossRef]
- Wang, D. B.; Zhu, C. Q.; Yang, Q. F.; Xu, Y. Q.; Zhang, D. Q.; Wang, D. Y.; Liu, F.; Hou, C. L. Stretchable, controlled release of active substances, and biodegradable chitosan−polyvinyl alcohol hydrogel film for antibacterial and chilled meat preservation. Food Chemistry 2025, 477, 143608. [Google Scholar] [CrossRef] [PubMed]
- Sanchez−Cid, P.; Alonso−Gonzalez, M.; Jimenez−Rosado, M.; Benhnia, M. R. E.; RuizMateos, E.; Ostos, F. J.; Romero, A.; Perez−Puyana, V. M. Effect of different crosslinking agents on hybrid chitosan/collagen hydrogels for potential tissue engineering applications. International Journal of Biological Macromolecules 2024, 263, 129858. [Google Scholar] [CrossRef] [PubMed]
- Dodda, J. M.; Azar, M. G.; Sadiku, R. Crosslinking trends in multicomponent hydrogels for biomedical applications. Macromolecular Bioscience 2021, 21(12), 2100232. [Google Scholar] [CrossRef] [PubMed]
- Lechuga−Islas, V. D.; Gillissen, E.; Bourguignon, M.; Grignard, B.; Detrembleur, C. Foam−to−adhesive recycling of self−blown non−isocyanate polyurethane foams facilitated by integration of disulfide exchangeable bonds and moisture. Chemical Engineering Journal 2025, 516, 163998. [Google Scholar] [CrossRef]
- Wang, W.; Coenye, T.; Su, J. Y.; Qiu, S.−X. Biofilm inhibition and eradication activity of citral and borneol against foodborne bacteria. Industrial Crops and Products 2025, 223, 120199. [Google Scholar] [CrossRef]
- Zhou, Z. L.; Chen, R. X.; Li, P. Z.; Fan, P. H.; Ma, L.; Cai, X. Y.; Hou, Y. C.; Li, B. B.; Su, J. Y. Natural borneol improves cellular uptake of curcumin to enhance its photodynamic bactericidal activity against Escherichia coli ATCC 8739. Food Microbiology 2025, 127, 104686. [Google Scholar] [CrossRef]
- Zhao, Z. J.; Fan, X.; Li, X. Y.; Qiu, Y. W.; Yi, Y. F.; Wei, Y. P.; Wang, Y. All−natural injectable antibacterial hydrogel enabled by chitosan and borneol. Biomacromolecules 2024, 25, 134−142. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, H. C.; Yan, C. F.; Wu, J.; Wang, Y. T.; Jiang, M. H.; Wang, Y. N. Yunnan Baiyao−enhanced cellulose nanofiber composite hydrogel wearable patch for transdermal drug delivery and anti−freezing applications. International Journal of Biological Macromolecules 2025, 315, 144684. [Google Scholar] [CrossRef]
- Deng, Z. X.; Guo, Y.; Wang, X. F.; Song, J. J.; Yang, G.; Shen, L. T.; Wang, Y. H.; Zhao, X.; Guo, B. L.; Wang, W. Multiple crosslinked, self−healing, and shape−adaptable hydrogel laden with pain−relieving chitosan@borneol nanoparticles for infected burn wound healing. Theranostics 2025, 15(4), 1439−1455. [Google Scholar] [CrossRef]
- Liu, Z. Z.; Liao, H. B.; Li, H. L.; Zou, Z. R. Fabrication and characterization of cinnamomum camphora chvar. Borneol essential oil microcapsules decorated by β−cyclodextrin with ultrasound−assisted complexation method. Arabian Journal for Science and Engineering 2025, 50(24), 20599−20612. [Google Scholar] [CrossRef]
- Abbasi, Z.; Esfandiari, Z.; Rostamabadi, H. Postbiotic−loaded κ−carrageenan hydrogels double cross−linked with carboxymethyl cellulose and calcium ions. Food Hydrocolloids 2025, 168, 111551. [Google Scholar] [CrossRef]
- Yang, Z. M.; Liu, Y.; Wang, E. Z.; Yin, W.; Wang, Y. J.; Guo, Y. C.; Zhang, W. T.; Qi, H. Insights into the highly selective and efficient adsorption of Pb2+ by fish skin collagen−enabled sodium alginate−based composite gel spheres: adsorption and interference mechanisms. Food Hydrocolloids 2026, 170, 111700. [Google Scholar] [CrossRef]
- Feng, X.; Li, M.; Li, S. H.; Lin, M. T.; Nie, Y.; Yao, N.; Deng, T. X.; Yang, X. H.; Ding, H. Y.; Xu, L. N. Fabrication and properties of recyclable tung oil–based polymer coatings based on dual cross–linked dynamic covalent polymer networks. Progress in Organic Coatings 2024, 192, 108521. [Google Scholar] [CrossRef]
- Kim, G. Y.; Sung, S. J.; Kim, M. P.; Kim, S. C.; Lee, S. H.; Park, Y. I.; Noh, S. M.; Cheong, I. W.; Kim, J. C. Reversible polymer networks based on the dynamic hindered urea bond for scratch healing in automotive clearcoats. Applied Surface Science 2020, 505, 144546. [Google Scholar] [CrossRef]
- Liu, W. Z.; Li, L.; Liu, S. N.; Liu, B.; Wu, Z. Y.; Deng, J. R. Novel robust ion−specific responsive photonic hydrogel elastomer. Journal of Materials Chemistry C 2024, 7(29), 8946−8953. [Google Scholar] [CrossRef]



| LB−PUAs | Molar ratio of L(-)−borneol to HPA | L(-)−borneol /mol |
HPA /mol |
PEG 600 /mol | TMP /mol |
IPDI /mol |
| LB−PUA−0 | 0:100 | 0 | 0.0390 |
0.01 |
0.002 |
0.0325 |
| LB−PUA−10 | 10:90 | 0.0039 | 0.0351 | |||
| LB−PUA−15 | 15:85 | 0.0059 | 0.0332 | |||
| LB−PUA−20 | 20:80 | 0.0078 | 0.0312 | |||
| LB−PUA−25 | 25:75 | 0.0098 | 0.0293 | |||
| LB−PUA−30 | 30:70 | 0.0117 | 0.0273 |
| UV−LBs | The categories and amount of LB−PUAs | The amount of PVA−SH |
| UV−LB−0 | 1 g LB−PUA−0 | 1.1925 g |
| UV−LB−10 | 1 g LB−PUA−10 | 1.0679 g |
| UV−LB−15 | 1 g LB−PUA−15 | 1.0060 g |
| UV−LB−20 | 1 g LB−PUA−20 | 0.9445 g |
| UV−LB−25 | 1 g LB−PUA−25 | 0.8833 g |
| UV−LB−30 | 1 g LB−PUA−30 | 0.8232 g |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




