Submitted:
31 March 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Animals
RNA Analysis
Statistical Analysis
Results
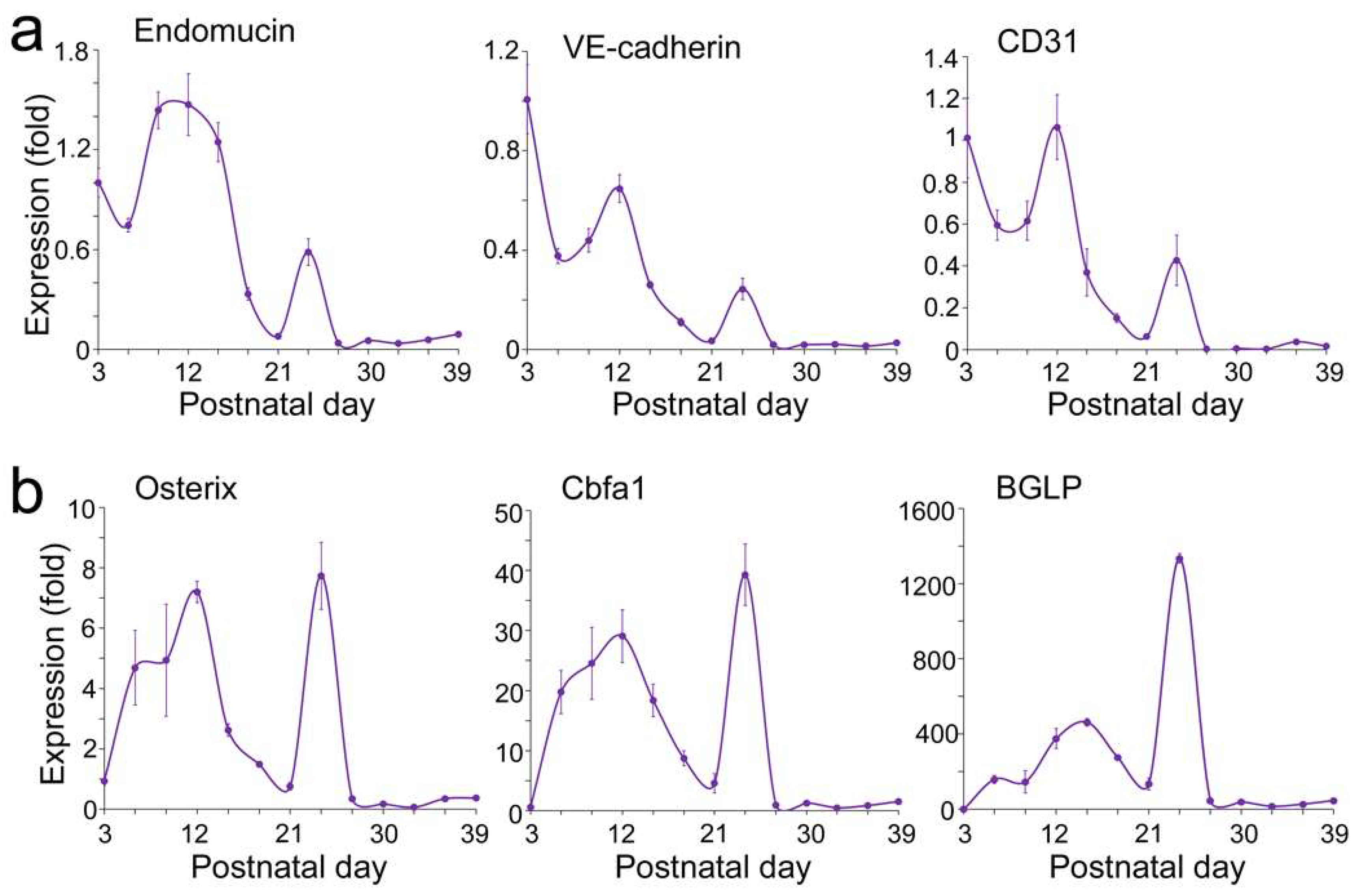
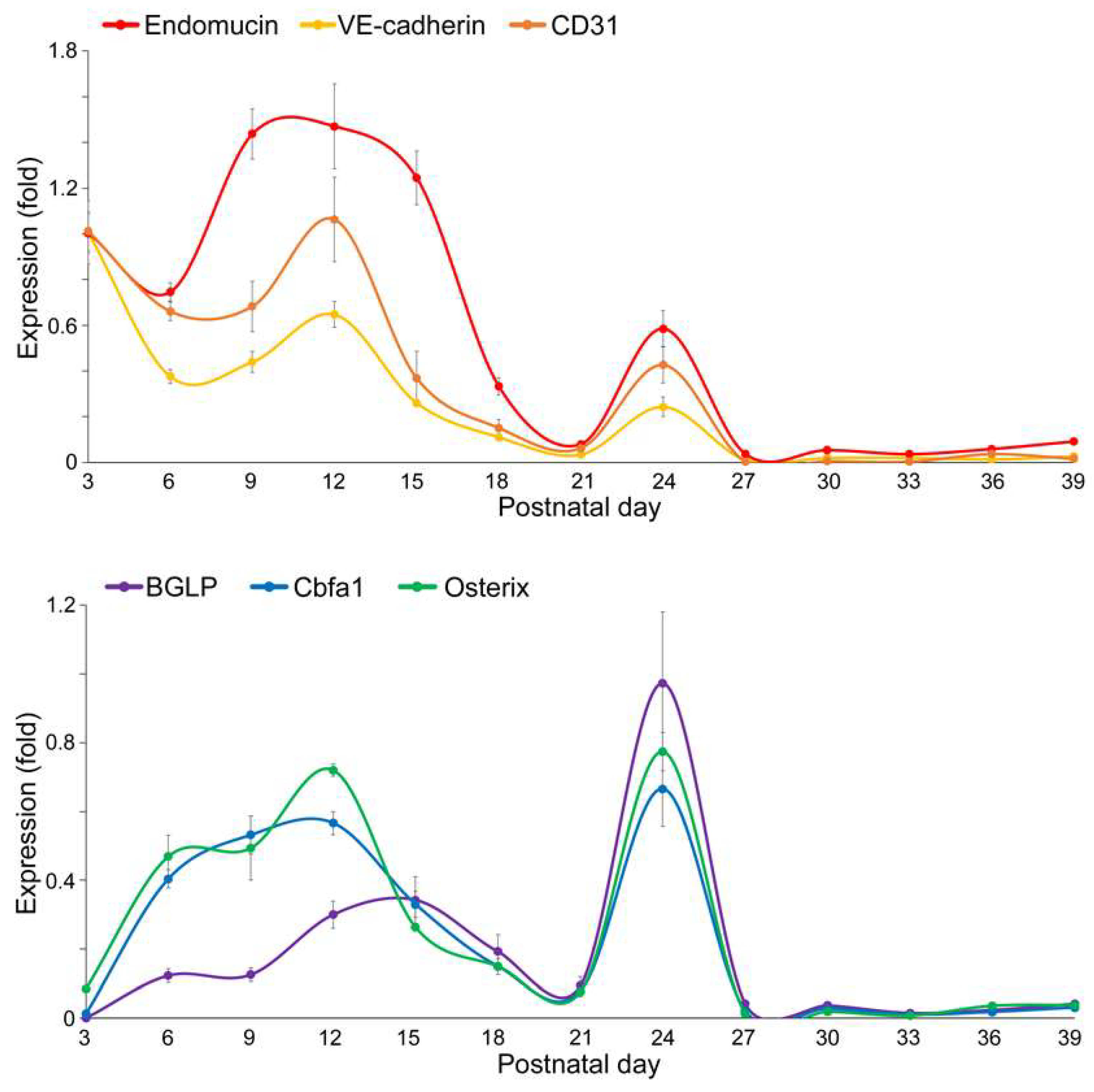
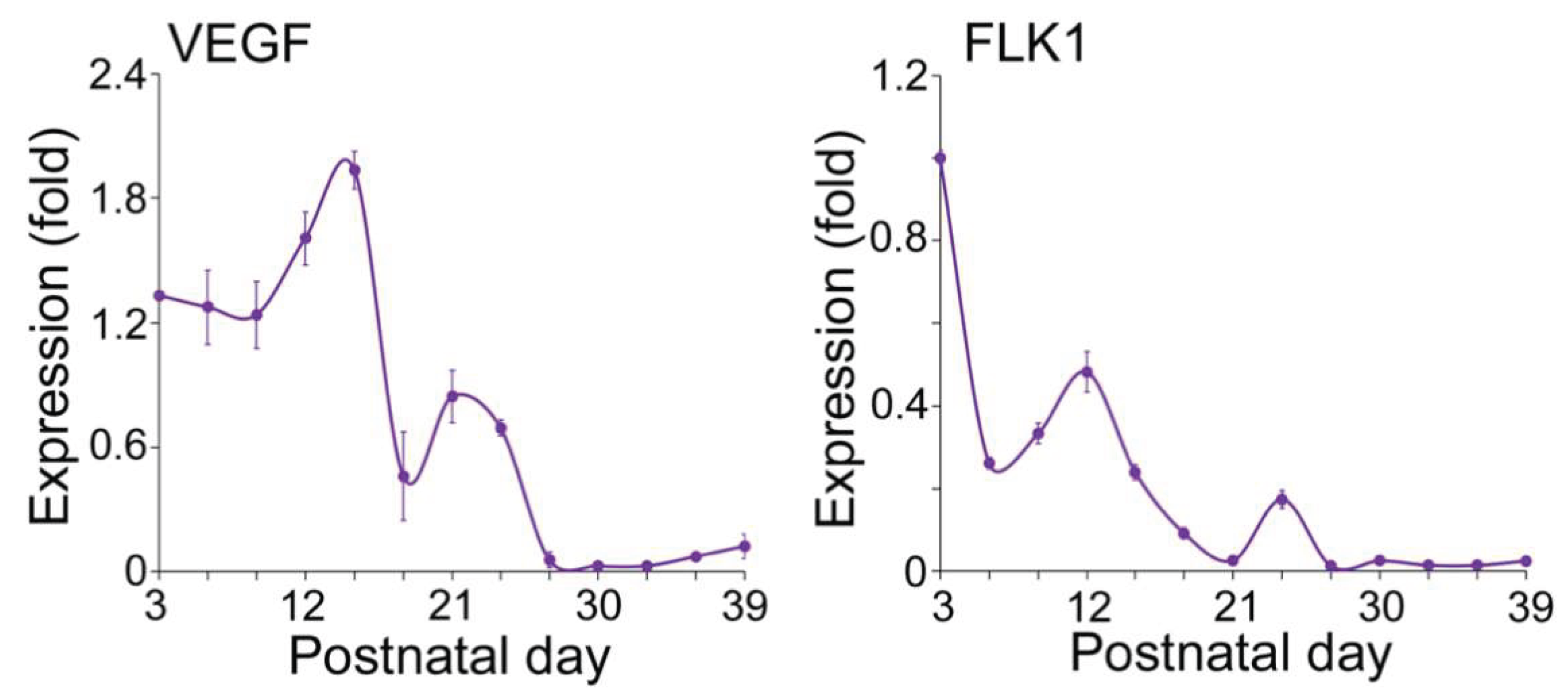
Expression of Endothelial and Osteogenic Markers During Early Development of Long Bone
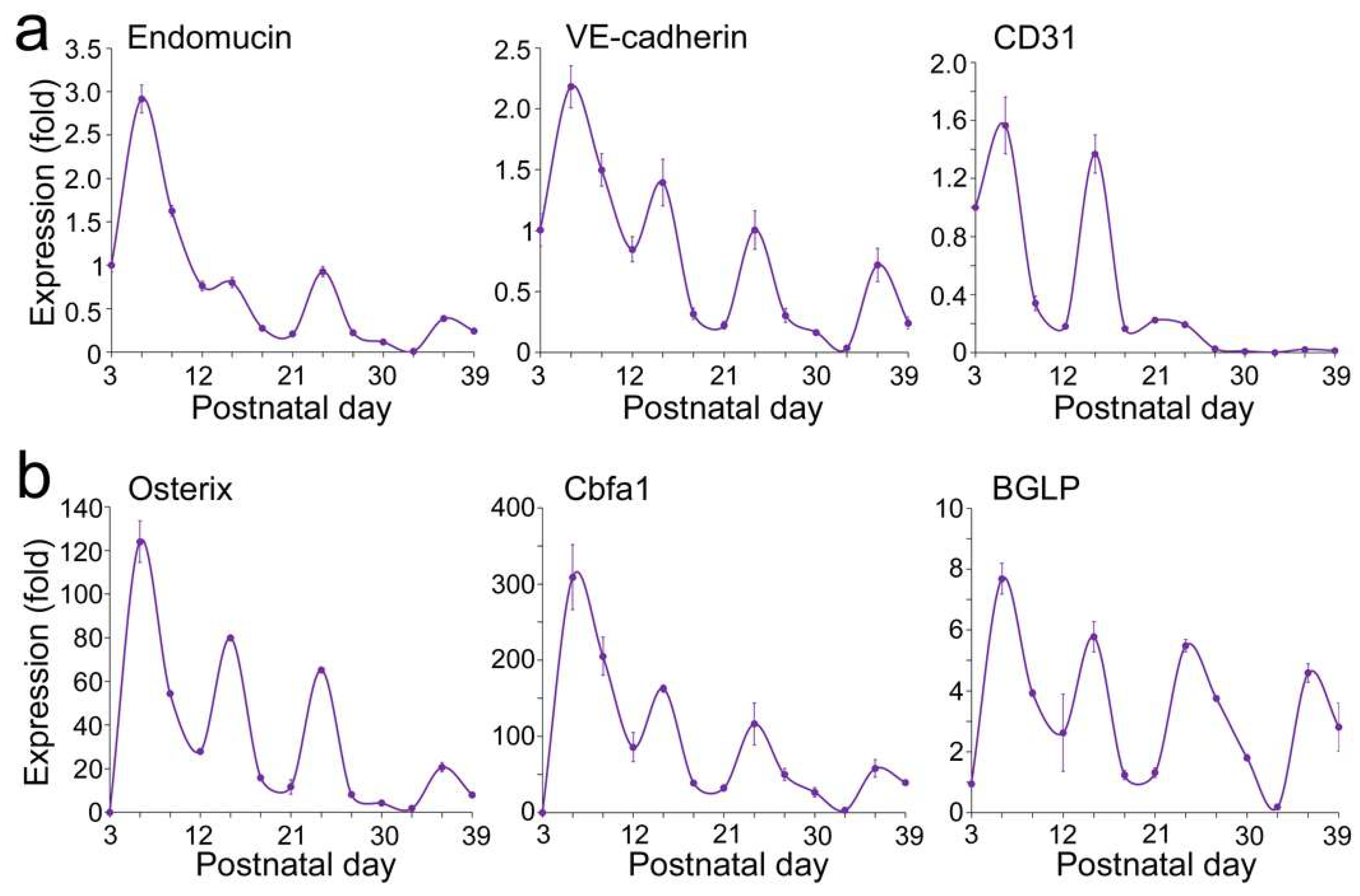
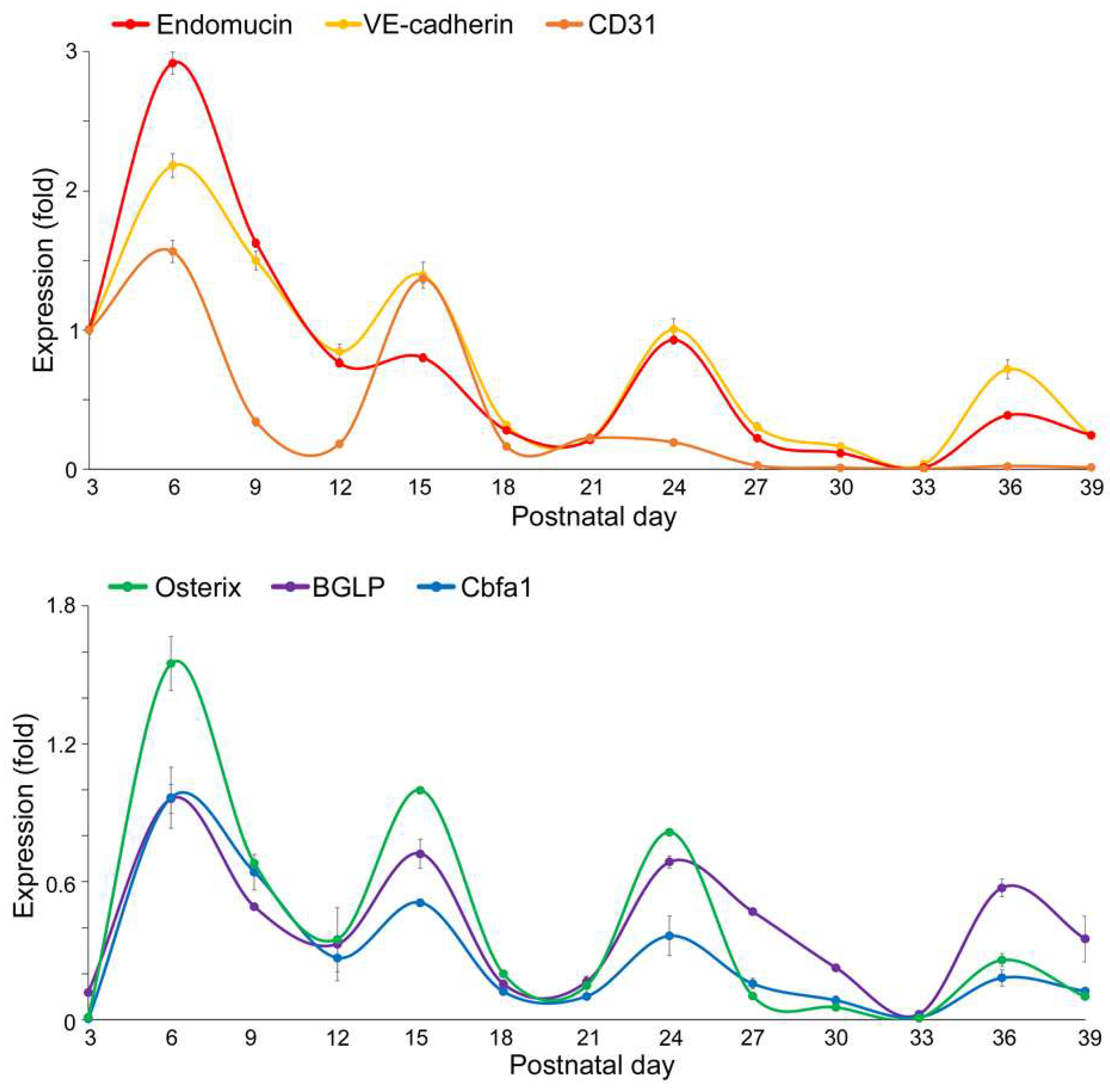
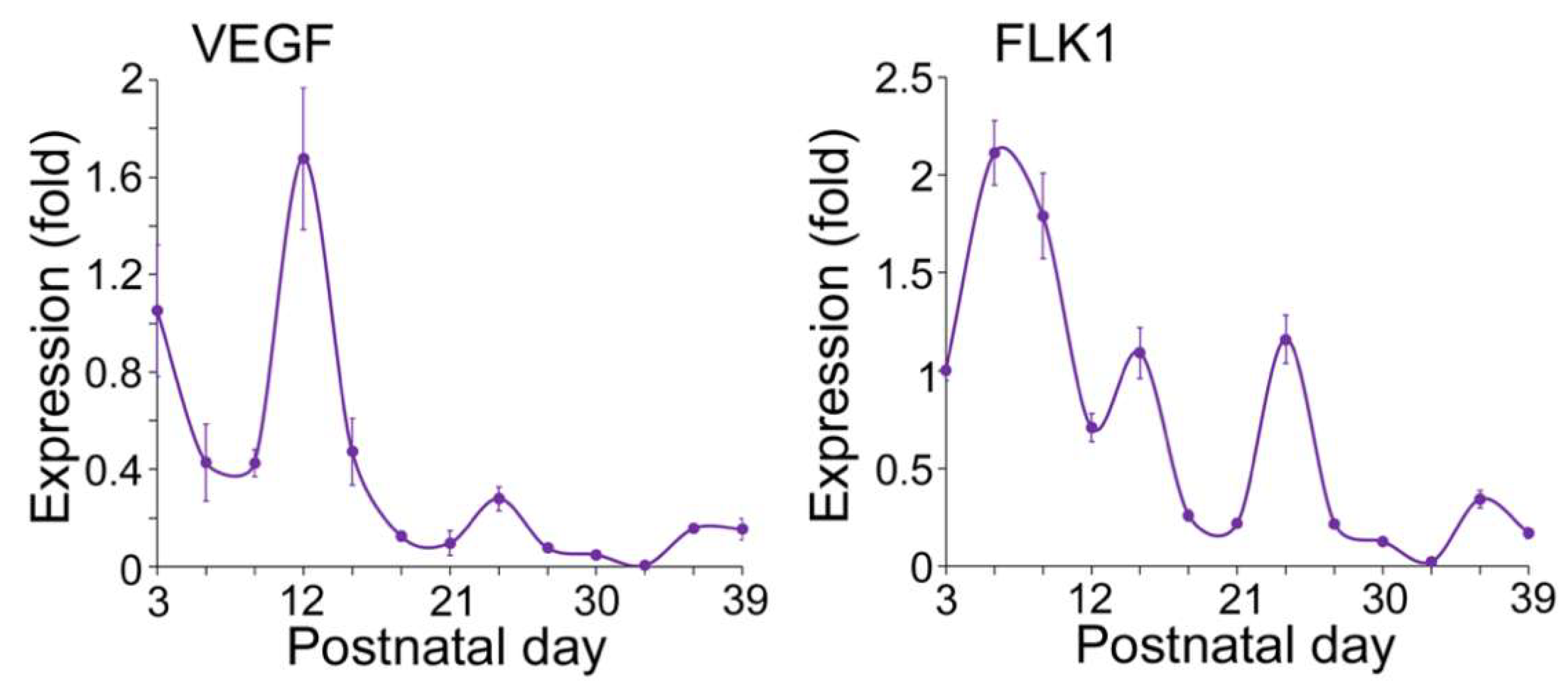
Expression of Endothelial and Osteogenic Markers During Early Development of Flat Bone
VEGFA–FLK1 Signaling in Endothelial–Osteogenic Crosstalk During Long and Flat Bone Formation
Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Rafii, S.; Butler, J.M.; Ding, B.S. Angiocrine functions of organ-specific endothelial cells. Nature 2016, 529, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Cines, D.B.; Pollak, E.S.; Buck, C.A.; Loscalzo, J.; Zimmerman, G.A.; McEver, R.P.; Pober, J.S.; Wick, T.M.; Konkle, B.A.; Schwartz, B.S.; et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 1998, 91, 3527–3561. [Google Scholar] [PubMed]
- Potente, M.; Gerhardt, H.; Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 2011, 146, 873–887. [Google Scholar] [CrossRef] [PubMed]
- Watson, E.C.; Adams, R.H. Biology of Bone: The Vasculature of the Skeletal System. Cold Spring Harb Perspect Med 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Pannarale, L.; Morini, S.; D'Ubaldo, E.; Gaudio, E.; Marinozzi, G. SEM corrosion-casts study of the microcirculation of the flat bones in the rat. Anat Rec 1997, 247, 462–471. [Google Scholar] [CrossRef]
- Kusumbe, A.P.; Ramasamy, S.K.; Adams, R.H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 2014, 507, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Long, F. Building strong bones: molecular regulation of the osteoblast lineage. Nat Rev Mol Cell Biol 2011, 13, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wu, X.; Zhao, Y.; Zhang, D.; Zhang, L.; Cai, X.; Ji, J.; Jing, Z.; Bostrom, K.I.; Yao, Y. Arterial-Lymphatic-Like Endothelial Cells Appear in Hereditary Hemorrhagic Telangiectasia 2 and Contribute to Vascular Leakage and Arteriovenous Malformations. Circulation 2025, 151, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wu, X.; Yang, Y.; Zhang, L.; Cai, X.; Chen, S.; Vera, A.; Ji, J.; Bostrom, K.I.; Yao, Y. Inhibition of endothelial histone deacetylase 2 shifts endothelial-mesenchymal transitions in cerebral arteriovenous malformation models. J Clin Invest 2024, 134. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, H.M. Developmental regulation of the growth plate. Nature 2003, 423, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Gordon, E.; Claesson-Welsh, L. Mechanisms and regulation of endothelial VEGF receptor signalling. Nat Rev Mol Cell Biol 2016, 17, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Murata, Y.; Liu, Y.; Nicolae, C.; Olsen, B.R.; Berendsen, A.D. Vegfa regulates perichondrial vascularity and osteoblast differentiation in bone development. Development 2015, 142, 1984–1991. [Google Scholar] [CrossRef] [PubMed]
- Guihard, P.J.; Guo, Y.; Wu, X.; Zhang, L.; Yao, J.; Jumabay, M.; Yao, Y.; Garfinkel, A.; Bostrom, K.I. Shaping Waves of Bone Morphogenetic Protein Inhibition During Vascular Growth. Circ Res 2020, 127, 1288–1305. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Garfinkel, A. A common pathway to cancer: Oncogenic mutations abolish p53 oscillations. Prog Biophys Mol Biol 2022, 174, 28–40. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



