Submitted:
31 March 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Location, Communities and Historical Setting
Data Collection
Meteorological Variables
Infectious Diseases Report and Wolbachia Program Reporting
Socio-Demographic and Economic Variables
Statistical Analysis
3. Results
Meteorological and Socio-Demographic Variable
Infectious Agents
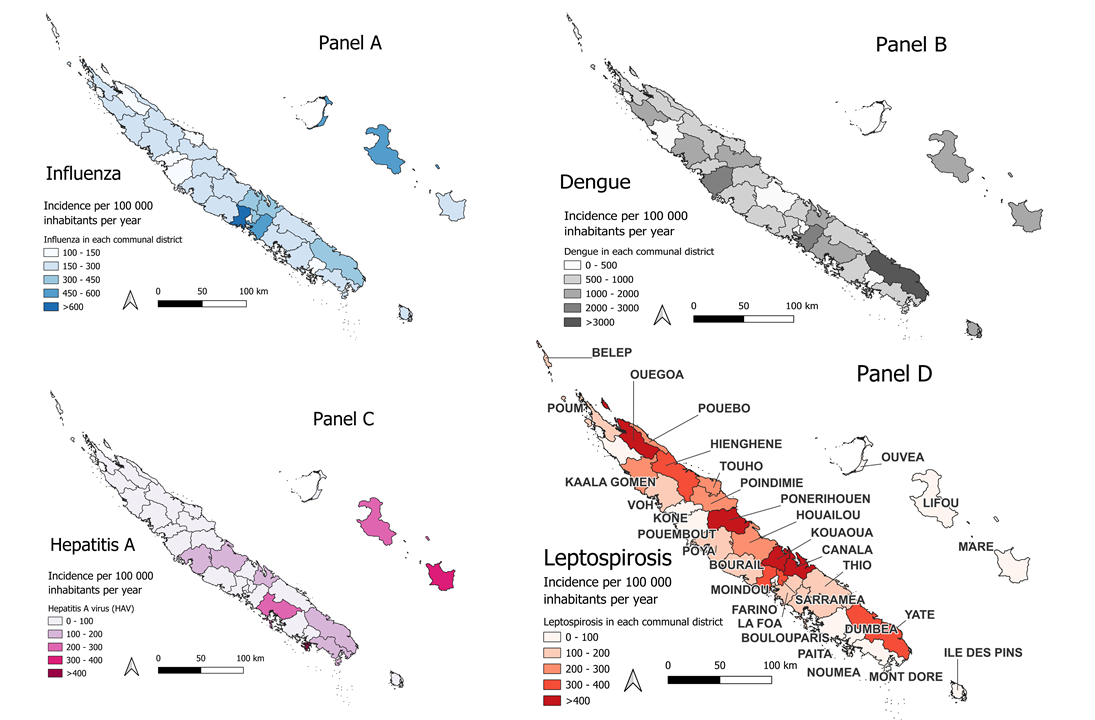
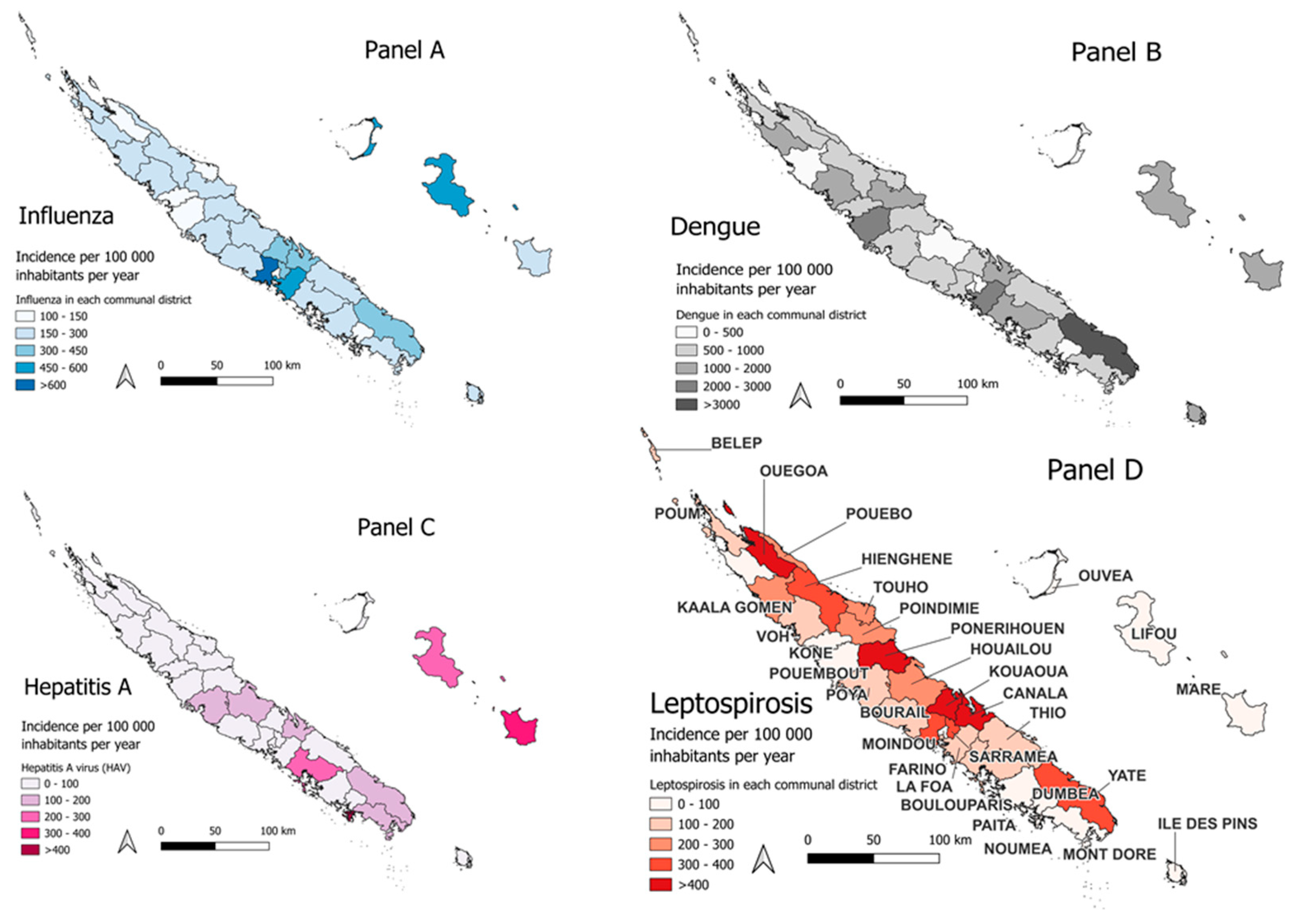
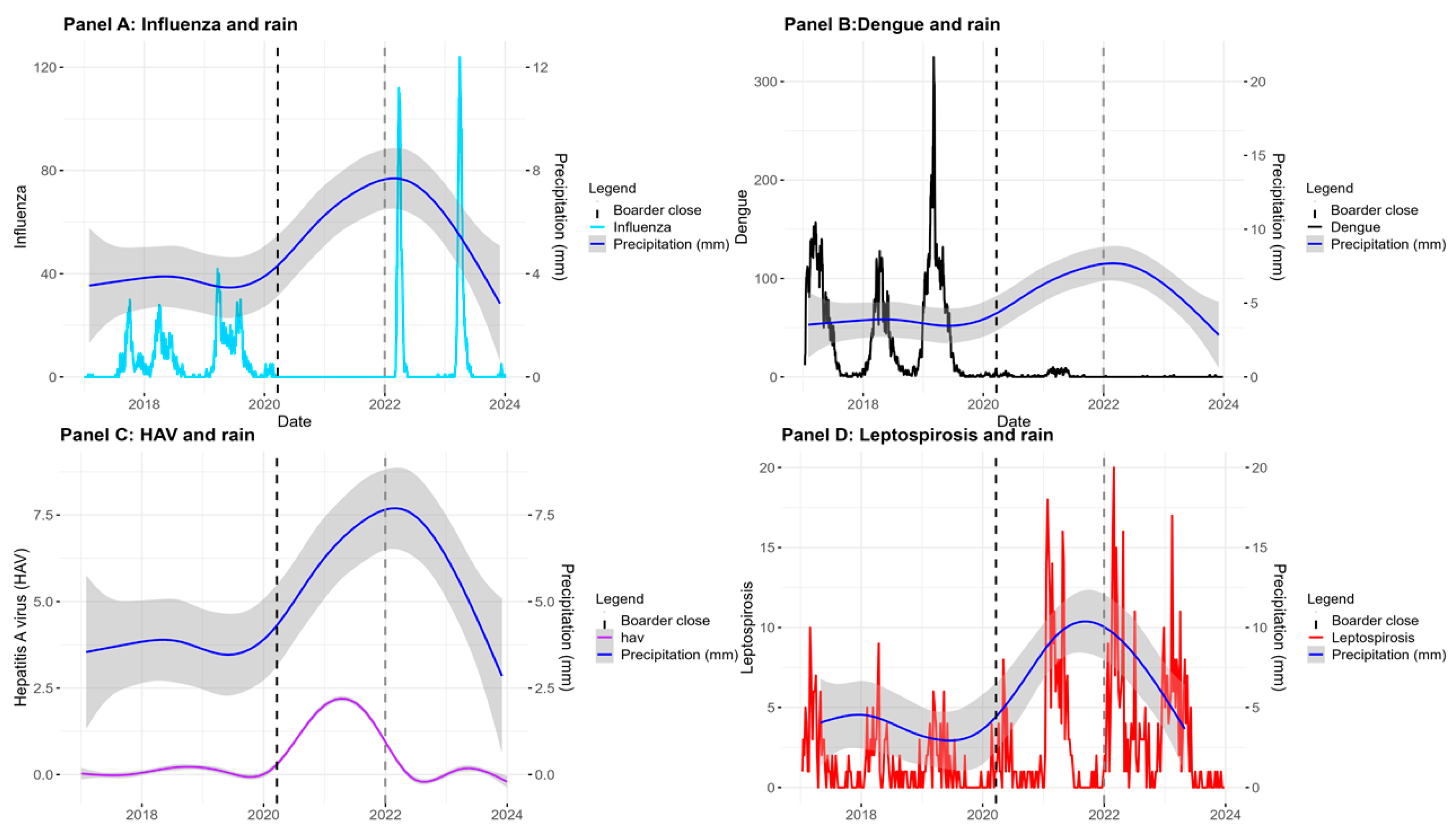
Influenza
Dengue
Hepatitis A Virus (HAV)
Leptospirosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Moury, P.-H.; Tromhae, M.; Cazorla, C.; Série, M.; Flahault, A.; Couadau, E.; Fleury, C.; Mangeas, M.; De Greslan, T. Colonial Transition as a Major Mediator of Global Health Transition: Lessons from the 2024 New Caledonia Crisis. J. Glob. Health 2025, 15, 03004. [Google Scholar] [CrossRef] [PubMed]
- Kerbaj, J.; Cazorla, C.; De Greslan, T.; Serie, M.; Gourinat, A.-C.; Marot, B. COVID-19: The New Caledonia Experience. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020, ciaa600. [Google Scholar] [CrossRef]
- Moury, P.-H.; Ochida, N.; Motiejunaite, J.; Collart, V.; Série, M.; Gervolino, S.; Mangeas, M.; Bouvier, J.-B.; Couadau, E.; Mebazaa, A.; et al. Impact of Lockdown on Cardiovascular Disease Hospitalizations in a Zero-COVID-19 Country. Public Health 2023, 217, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Moury, P.-H.; Gourinat, A.-C.; Riou, O.; Laumond, S.; Dupont-Rouzeyrol, M.; Cazorla, C.; Mangeas, M. Successful COVID-19 Elimination after an Alpha Variant Outbreak in a “Safe Travel Zone.”. Travel Med. Infect. Dis. 2021, 44, 102202. [Google Scholar] [CrossRef]
- Ochida, N.; Dupont-Rouzeyrol, M.; Moury, P.-H.; Demaneuf, T.; Gourinat, A.-C.; Mabon, S.; Jouan, M.; Cauchemez, S.; Mangeas, M. Evaluating the Strategies to Control SARS-CoV-2 Delta Variant Spread in New Caledonia, a Zero-COVID Country until September 2021. IJID Reg. 2023, 8, 64–70. [Google Scholar] [CrossRef]
- Murphy, B.F.; Power, S.B.; McGree, S. The Varied Impacts of El Niño–Southern Oscillation on Pacific Island Climates. J. Clim. 2014, 27, 4015–4036. [Google Scholar] [CrossRef]
- Moron, V.; Barbero, R.; Robertson, A.W. Subseasonal-to-Interannual Variability of Rainfall over New Caledonia (SW Pacific). Clim. Dyn. 2016, 46, 2449–2468. [Google Scholar] [CrossRef]
- Moron, V.; Barbero, R.; Robertson, A.W. Subseasonal-to-Interannual Variability of Rainfall over New Caledonia (SW Pacific). Clim. Dyn. 2016, 46, 2449–2468. [Google Scholar] [CrossRef]
- Inizan, C.; Tarantola, A.; O’Connor, O.; Mangeas, M.; Pocquet, N.; Forfait, C.; Descloux, E.; Gourinat, A.-C.; Pfannstiel, A.; Klement-Frutos, E.; et al. Dengue in New Caledonia: Knowledge and Gaps. Trop. Med. Infect. Dis. 2019, 4, 95. [Google Scholar] [CrossRef]
- Ochida, N.; Mangeas, M.; Dupont-Rouzeyrol, M.; Dutheil, C.; Forfait, C.; Peltier, A.; Descloux, E.; Menkes, C. Modeling Present and Future Climate Risk of Dengue Outbreak, a Case Study in New Caledonia. Environ. Health 2022, 21, 20. [Google Scholar] [CrossRef]
- Western Pacific Region Global Influenza Surveillance and Response System Epidemiological and Virological Characteristics of Influenza in the Western Pacific Region of the World Health Organization, 2006-2010. PloS One 2012, 7, e37568. [CrossRef]
- Koff, R.S. Hepatitis A. The Lancet 1998, 351, 1643–1649. [Google Scholar] [CrossRef]
- Goarant, C.; Laumond-Barny, S.; Perez, J.; Vernel-Pauillac, F.; Chanteau, S.; Guigon, A. Outbreak of Leptospirosis in New Caledonia: Diagnosis Issues and Burden of Disease. Trop. Med. Int. Health TM IH 2009, 14, 926–929. [Google Scholar] [CrossRef] [PubMed]
- Douchet, L.; Menkes, C.; Herbreteau, V.; Larrieu, J.; Bador, M.; Goarant, C.; Mangeas, M. Climate-Driven Models of Leptospirosis Dynamics in Tropical Islands from Three Oceanic Basins. PLoS Negl. Trop. Dis. 2024, 18, e0011717. [Google Scholar] [CrossRef]
- Findlater, A.; Bogoch, I.I. Human Mobility and the Global Spread of Infectious Diseases: A Focus on Air Travel. Trends Parasitol. 2018, 34, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Mora, C.; McKenzie, T.; Gaw, I.M.; Dean, J.M.; von Hammerstein, H.; Knudson, T.A.; Setter, R.O.; Smith, C.Z.; Webster, K.M.; Patz, J.A.; et al. Over Half of Known Human Pathogenic Diseases Can Be Aggravated by Climate Change. Nat. Clim. Change 2022, 12, 869–875. [Google Scholar] [CrossRef]
- Barbero, R.; Moron, V. Seasonal to Decadal Modulation of the Impact of El Niño–Southern Oscillation on New Caledonia (SW Pacific) Rainfall (1950–2010). J. Geophys. Res. Atmospheres 2011, 116. [Google Scholar] [CrossRef]
- Kerbaj, J.; Cazorla, C.; De Greslan, T.; Serie, M.; Gourinat, A.-C.; Marot, B. COVID-19: The New Caledonia Experience. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2020, ciaa600. [Google Scholar] [CrossRef]
- Zhang, T.; Hoell, A.; Perlwitz, J.; Eischeid, J.; Murray, D.; Hoerling, M.; Hamill, T.M. Towards Probabilistic Multivariate ENSO Monitoring. Geophys. Res. Lett. 2019, 46, 10532–10540. [Google Scholar] [CrossRef]
- Utarini, A.; Indriani, C.; Ahmad, R.A.; Tantowijoyo, W.; Arguni, E.; Ansari, M.R.; Supriyati, E.; Wardana, D.S.; Meitika, Y.; Ernesia, I.; et al. Efficacy of Wolbachia-Infected Mosquito Deployments for the Control of Dengue. N. Engl. J. Med. 2021, 384, 2177–2186. [Google Scholar] [CrossRef]
- Kurz, C.F. Tweedie Distributions for Fitting Semicontinuous Health Care Utilization Cost Data. BMC Med. Res. Methodol. 2017, 17, 171. [Google Scholar] [CrossRef]
- Ma, R.; Yan, G.; Hasan, M.T. Tweedie Family of Generalized Linear Models with Distribution-Free Random Effects for Skewed Longitudinal Data. Stat. Med. 2018, 37, 3519–3532. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, C.; Moirano, G.; Alcayna, T.; Rollock, L.; Van Meerbeeck, C.J.; Mahon, R.; Trotman, A.; Boodram, L.-L.; Browne, T.; Best, S.; et al. Compound and Cascading Effects of Climatic Extremes on Dengue Outbreak Risk in the Caribbean: An Impact-Based Modelling Framework with Long-Lag and Short-Lag Interactions. Lancet Planet. Health 2025, 9, 101279. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, D.; Baroux, N.; Grangeon, J.-P.; Ko, A.I.; Goarant, C. El Niño Southern Oscillation and Leptospirosis Outbreaks in New Caledonia. PLoS Negl. Trop. Dis. 2014, 8, e2798. [Google Scholar] [CrossRef]
- Li, N.; Feng, Y.; Vrancken, B.; Chen, Y.; Dong, L.; Yang, Q.; Kraemer, M.U.G.; Pybus, O.G.; Zhang, H.; Brady, O.J.; et al. Assessing the Impact of COVID-19 Border Restrictions on Dengue Transmission in Yunnan Province, China: An Observational Epidemiological and Phylogenetic Analysis. Lancet Reg. Health - West. Pac. 2021, 14, 100259. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, G.K.; Taylor, J.; Kok, J.; Dwyer, D.E.; Dilcher, M.; Hua, H.; Levy, A.; Smith, D.; Minney-Smith, C.A.; Wood, T.; et al. Circulation of Influenza and Other Respiratory Viruses during the COVID-19 Pandemic in Australia and New Zealand, 2020–2021. West. Pac. Surveill. Response J. WPSAR 2023, 14, 1–9. [Google Scholar] [CrossRef]
- Taylor-Salmon, E.; Hill, V.; Paul, L.M.; Koch, R.T.; Breban, M.I.; Chaguza, C.; Sodeinde, A.; Warren, J.L.; Bunch, S.; Cano, N.; et al. Travel Surveillance Uncovers Dengue Virus Dynamics and Introductions in the Caribbean. Nat. Commun. 2024, 15, 3508. [Google Scholar] [CrossRef]
- Huang, Q.S.; Turner, N.; Wood, T.; Anglemyer, A.; McIntyre, P.; Aminisani, N.; Dowell, T.; Trenholme, A.; Byrnes, C.; Balm, M.; et al. Impact of the COVID-19 Related Border Restrictions on Influenza and Other Common Respiratory Viral Infections in New Zealand. Influenza Other Respir. Viruses 2024, 18, e13247. [Google Scholar] [CrossRef]
- Zellweger, R.M.; Cano, J.; Mangeas, M.; Taglioni, F.; Mercier, A.; Despinoy, M.; Menkès, C.E.; Dupont-Rouzeyrol, M.; Nikolay, B.; Teurlai, M. Socioeconomic and Environmental Determinants of Dengue Transmission in an Urban Setting: An Ecological Study in Nouméa, New Caledonia. PLoS Negl. Trop. Dis. 2017, 11, e0005471. [Google Scholar] [CrossRef]
- Teurlai, M.; Menkès, C.E.; Cavarero, V.; Degallier, N.; Descloux, E.; Grangeon, J.-P.; Guillaumot, L.; Libourel, T.; Lucio, P.S.; Mathieu-Daudé, F.; et al. Socio-Economic and Climate Factors Associated with Dengue Fever Spatial Heterogeneity: A Worked Example in New Caledonia. PLoS Negl. Trop. Dis. 2015, 9, e0004211. [Google Scholar] [CrossRef]
- Chen, Y.; Li, N.; Lourenço, J.; Wang, L.; Cazelles, B.; Dong, L.; Li, B.; Liu, Y.; Jit, M.; Bosse, N.I.; et al. Measuring the Effects of COVID-19-Related Disruption on Dengue Transmission in Southeast Asia and Latin America: A Statistical Modelling Study. Lancet Infect. Dis. 2022, 22, 657–667. [Google Scholar] [CrossRef]
- Inizan, C.; Minier, M.; Prot, M.; O’Connor, O.; Forfait, C.; Laumond, S.; Marois, I.; Biron, A.; Gourinat, A.-C.; Goujart, M.-A.; et al. Viral Evolution Sustains a Dengue Outbreak of Enhanced Severity. Emerg. Microbes Infect. 2021, 10, 536. [Google Scholar] [CrossRef]
- Rzymski, P.; Zarębska-Michaluk, D.; Genowska, A.; Tyszko, P.; Strukcinskiene, B.; Flisiak, R. Trends of Hepatitis A Virus Infection in Poland: Assessing the Potential Impact of the COVID-19 Pandemic and War in Ukraine. Viruses 2024, 16, 469. [Google Scholar] [CrossRef] [PubMed]
- Murakoshi, K.; Mori, H.; Prasertbun, R.; Valenti, S.; Krokva, D.; Remez, D.; Mahittikorn, A.; Hadano, Y.; Naito, T. Hepatitis A Epidemics in Japan, France, and Thailand from 2007 to 2021, Highlighting a Post-COVID-19 Decline. Sci. Rep. 2025, 15, 7228. [Google Scholar] [CrossRef] [PubMed]
- Berlioz-Arthaud, A.; Barny, S.; Yvon, J.F.; Roque-Afonso, A.M.; Dussaix, E. Laboratory based hepatitis A surveillance in New Caledonia: from an endemic to an epidemic pattern (1986-2007). Bull. Soc. Pathol. Exot. 1990 2008, 101, 336–342. [Google Scholar] [CrossRef]
- Fischer, G.E.; Thompson, N.; Chaves, S.S.; Bower, W.; Goldstein, S.; Armstrong, G.; Williams, I.; Bialek, S. The Epidemiology of Hepatitis A Virus Infections in Four Pacific Island Nations, 1995–2008. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 906–910. [Google Scholar] [CrossRef]
- Bierque, E.; Thibeaux, R.; Girault, D.; Soupé-Gilbert, M.-E.; Goarant, C. A Systematic Review of Leptospira in Water and Soil Environments. PloS One 2020, 15, e0227055. [Google Scholar] [CrossRef]
- Thibeaux, R.; Genthon, P.; Govan, R.; Selmaoui-Folcher, N.; Tramier, C.; Kainiu, M.; Soupé-Gilbert, M.-E.; Wijesuriya, K.; Goarant, C. Rainfall-Driven Resuspension of Pathogenic Leptospira in a Leptospirosis Hotspot. Sci. Total. Environ. 2024, 911, 168700. [Google Scholar] [CrossRef]
- Govan, R.; Scherrer, R.; Fougeron, B.; Laporte-Magoni, C.; Thibeaux, R.; Genthon, P.; Fournier-Viger, P.; Goarant, C.; Selmaoui-Folcher, N. Spatio-Temporal Risk Prediction of Leptospirosis: A Machine-Learning-Based Approach. PLoS Negl. Trop. Dis. 2025, 19, e0012755. [Google Scholar] [CrossRef] [PubMed]
- Moury, P.-H. Colonial Transitions in Health. Ethics Med. Public Health 2026, 34, 101217. [Google Scholar] [CrossRef]
- Cai, W.; Lengaigne, M.; Borlace, S.; Collins, M.; Cowan, T.; McPhaden, M.J.; Timmermann, A.; Power, S.; Brown, J.; Menkes, C.; et al. More Extreme Swings of the South Pacific Convergence Zone Due to Greenhouse Warming. Nature 2012, 488, 365–369. [Google Scholar] [CrossRef]
- Cai, W.; Wang, G.; Santoso, A.; McPhaden, M.J.; Wu, L.; Jin, F.-F.; Timmermann, A.; Collins, M.; Vecchi, G.; Lengaigne, M.; et al. Increased Frequency of Extreme La Niña Events under Greenhouse Warming. Nat. Clim. Change 2015, 5, 132–137. [Google Scholar] [CrossRef]
- Haines, A.; Lam, H.C.Y. El Niño and Health in an Era of Unprecedented Climate Change. The Lancet 2023. [Google Scholar] [CrossRef] [PubMed]
- Baroux, N.; Maire, L.; Cadic, L.; Lemaitre, A.-F.; Borceux, P.; Glasman, B. Riots in New Caledonia: Impact of Constrained Management on Peritoneal Dialysis Patients. Bull. Dial. À Domic. 2024, 7, 89–99. [Google Scholar] [CrossRef]
- Geng, T.; Jia, F.; Cai, W.; Wu, L.; Gan, B.; Jing, Z.; Li, S.; McPhaden, M.J. Increased Occurrences of Consecutive La Niña Events under Global Warming. Nature 2023, 619, 774–781. [Google Scholar] [CrossRef]
| Period | Influenza | Dengue | Hepatitis viral A | Leptospirosis | ||||
|---|---|---|---|---|---|---|---|---|
| Border Status Period | Number of cases | Incidence for 100 000 habitants/year | Number of cases | Incidence for 100 000 habitants/year | Number of cases | Incidence for 100 000 habitants/year | Number of cases | Incidence for 100 000 habitants |
| Before (1176 days) | 1,072 | 123 | 7,213 | 829 | 14 | 2 | 242 | 28 |
| Closed (649 days) | 0 | 0 | 153 | 32 | 144 | 30 | 256 | 53 |
| After (729 days) | 950 | 176 | 11 | 2 | 7 | 2 | 380 | 71 |
| Variable | Coefficient | Standard error | Odds Ratio 95%CI | p-value | |
|---|---|---|---|---|---|
| Influenza before border closure | |||||
| Mean daily precipitation over each commune (mm/d) | -0.4 | 0.1 | 0.7 (0.6 - 0.8) | <0.001 | |
| Influenza after border closure | |||||
| Mean daily precipitation over each commune (mm/d) | 0.2 | 0.2 | 1.2 (0.8 - 1.8) | 0.301 | |
| Dengue before border closure | |||||
| MEI index | -0.3 | 0.1 | 0.78 (0.6 – 1) | 0.049 | |
| Hepatitis A | |||||
| MEI index | 0.6 | 0.3 | 1.8 (1 - 3.3) | 0.042 | |
| Percentage of inhabitants with a BEPC or lower | -0.1 | 0.0 | 0.9 (0.9 - 1) | 0.052 | |
| Leptospirosis | |||||
| Mean daily precipitation over each commune (mm/d) averaged from weekly precipitation | |||||
| Precipitation week 1 | -0.04 | 0.05 | 0.96 (0.9-1.1) | 0.46 | |
| Precipitation week 2 | 0.11 | 0.05 | 1.12 (1-1.23) | 0.017 | |
| Precipitation week 3 | -0.11 | 0.05 | 0.90 (0.8-1) | 0.025 | |
| Precipitation week 4 | 0.05 | 0.02 | 1.05 (1-1.1) | 0.037 | |
| Precipitation week 5 | 0.11 | 0.02 | 1.12 (1.1-1.2) | <0.001 | |
| Percentage of inhabitants who walks as major transport mean | 0.3 | 0.2 | 1.33 (1 – 1.9) | 0.08 | |
| Percentage of inhabitants with water supply | -0.4 | 0.1 | 0.7 (0.5 - 0.9) | 0.015 | |
| Percentage of inhabitants with a BEPC or lower | 0.04 | 0.01 | 1.04 (1.02 – 1.1) | 0.008 | |
| Note: BEPC: first grade diploma obtained at 14-15years old. Odds ratios were calculated based on the exponent of the coefficient. CI: Confidence Interval at 95%; MEI: Multivariate El Niño/Southern Oscillation (ENSO) index | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




