Submitted:
01 April 2026
Posted:
02 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Methods
2.2.1. O2•− Quantification
2.2.2. OS Markers Quantification
2.3. Statistical Analysis
3. Results
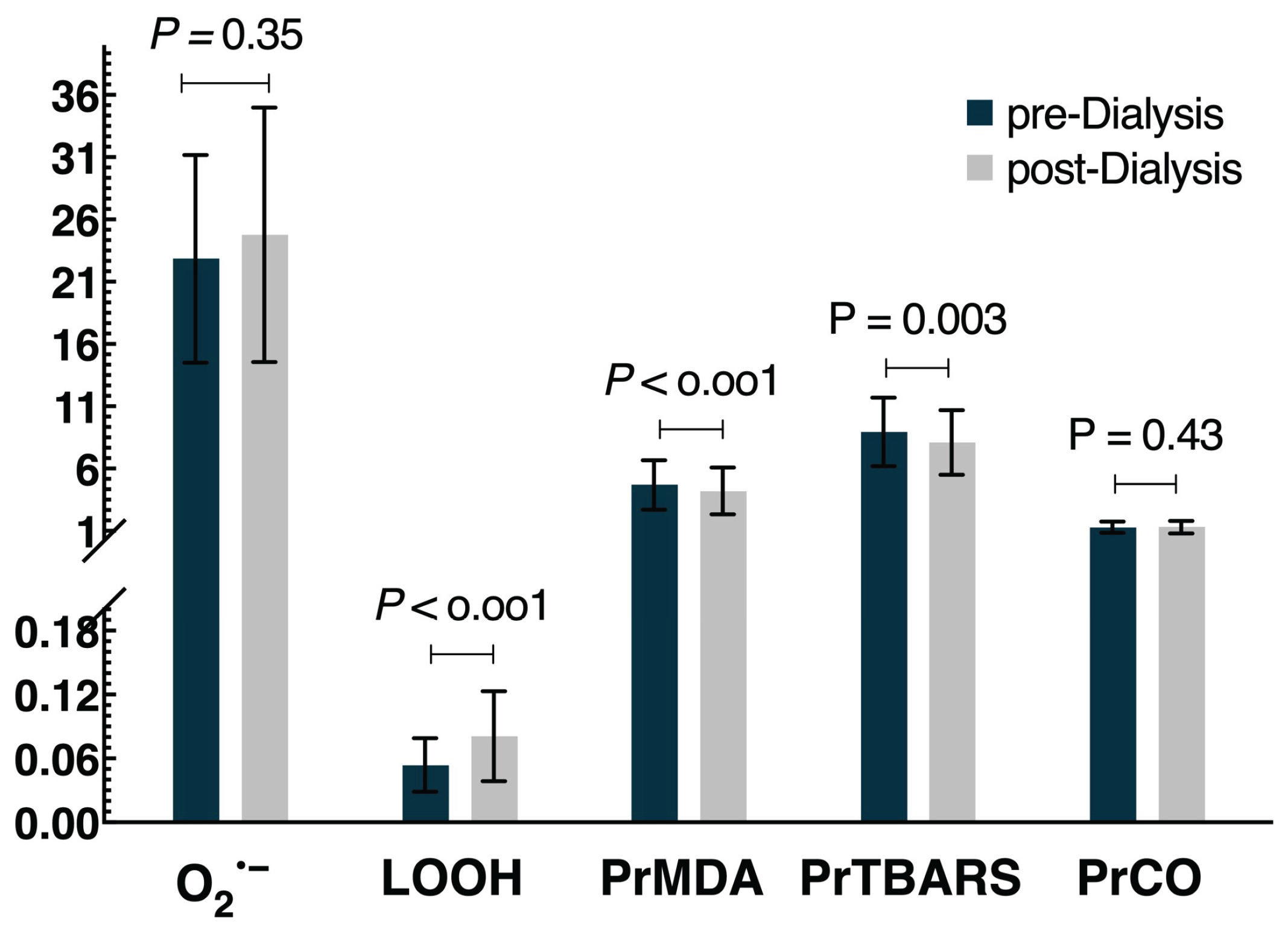
3.1. OS Biomarkers Levels Before and After a Single Dialysis Session
3.2. Impact of Dialysis Modalities on OS Biomarkers
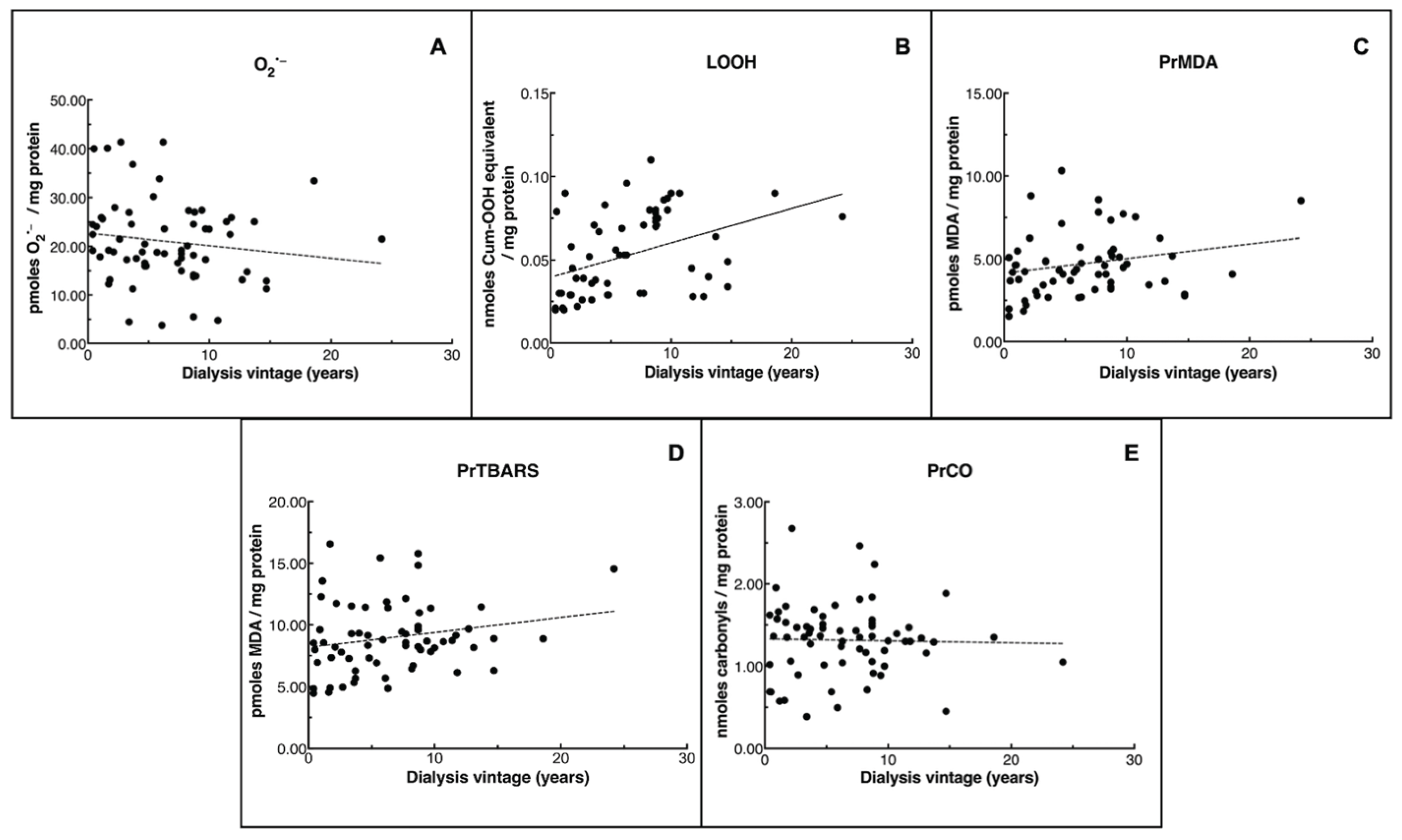
3.3. Associations Between OS Biomarkers and Dialysis Vintage
3.4. Additional Analyses
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sies, H.; Jones, D. Oxidative Stress. In Encyclopedia of Stress; Elsevier, 2007; pp. 45–48. ISBN 978-0-12-373947-6. [Google Scholar]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative Stress and Antioxidant Defense. World Allergy Organization Journal 2012, 5, 9–19. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Medicine and Cellular Longevity 2014, 2014, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Indo, H.P.; Yen, H.-C.; Nakanishi, I.; Matsumoto, K.; Tamura, M.; Nagano, Y.; Matsui, H.; Gusev, O.; Cornette, R.; Okuda, T.; et al. A Mitochondrial Superoxide Theory for Oxidative Stress Diseases and Aging. J. Clin. Biochem. Nutr. 2015, 56, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Dalle-Donne, I.; Giustarini, D.; Colombo, R.; Rossi, R.; Milzani, A. Protein Carbonylation in Human Diseases. Trends in Molecular Medicine 2003, 9, 169–176. [Google Scholar] [CrossRef]
- Papadea, P.; Skipitari, M.; Kalaitzopoulou, E.; Varemmenou, A.; Spiliopoulou, M.; Papasotiriou, M.; Papachristou, E.; Goumenos, D.; Onoufriou, A.; Rosmaraki, E.; et al. Methods on LDL Particle Isolation, Characterization, and Component Fractionation for the Development of Novel Specific Oxidized LDL Status Markers for Atherosclerotic Disease Risk Assessment. Front Med (Lausanne) 2022, 9, 1078492. [Google Scholar] [CrossRef]
- Valaitienė, J.; Laučytė-Cibulskienė, A. Oxidative Stress and Its Biomarkers in Cardiovascular Diseases. Artery Res 2024, 30, 18. [Google Scholar] [CrossRef]
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; Yazbi, A.E.; Pintus, G.; Eid, A.H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front Biosci (Landmark Ed) 2022, 27, 105. [Google Scholar] [CrossRef]
- Poulianiti, K.P.; Kaltsatou, A.; Mitrou, G.I.; Jamurtas, A.Z.; Koutedakis, Y.; Maridaki, M.; Stefanidis, I.; Sakkas, G.K.; Karatzaferi, C. Systemic Redox Imbalance in Chronic Kidney Disease: A Systematic Review. Oxidative Medicine and Cellular Longevity 2016, 2016, 8598253. [Google Scholar] [CrossRef]
- Antic, S.; Draginic, N.; Nikolic, T.; Jeremic, N.; Petrovic, D. Oxidative Stress in Hemodialysis Patients: Pathophysiological Mechanisms, Clinical Consequences and Basic Principles of Treatment. Experimental and Applied Biomedical Research (EABR) 2024, 25, 199–206. [Google Scholar] [CrossRef]
- Rysz, J.; Franczyk, B.; Ławiński, J.; Gluba-Brzózka, A. Oxidative Stress in ESRD Patients on Dialysis and the Risk of Cardiovascular Diseases. Antioxidants (Basel) 2020, 9, 1079. [Google Scholar] [CrossRef]
- Roumeliotis, S.; Liakopoulos, V.; Dounousi, E.; Mark, P.B. Oxidative Stress in End-Stage Renal Disease: Pathophysiology and Potential Interventions. Oxidative Medicine and Cellular Longevity 2023, 2023, 1–4. [Google Scholar] [CrossRef]
- Liakopoulos, V.; Roumeliotis, S.; Zarogiannis, S.; Eleftheriadis, T.; Mertens, P.R. Oxidative Stress in Hemodialysis: Causative Mechanisms, Clinical Implications, and Possible Therapeutic Interventions. Seminars in Dialysis 2019, 32, 58–71. [Google Scholar] [CrossRef]
- Ling, X.C.; Kuo, K.-L. Oxidative Stress in Chronic Kidney Disease. Ren Replace Ther 2018, 4, 53. [Google Scholar] [CrossRef]
- Canaud, B.; Cristol, J.-P.; Morena, M.; Leray-Moragues, H.; Bosc, J.-Y.; Vaussenat, F. Imbalance of Oxidants and Antioxidants in Haemodialysis Patients. Blood Purif 1999, 17, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-F.; Chang, C.-L.; Liou, S.-Y. Increase in Resting Levels of Superoxide Anion in the Whole Blood of Uremic Patients on Chronic Hemodialysis. Blood Purif 1998, 16, 290–300. [Google Scholar] [CrossRef]
- Liochev, S.I.; Fridovich, I. Lucigenin as Mediator of Superoxide Production: Revisited. Free Radical Biology and Medicine 1998, 25, 926–928. [Google Scholar] [CrossRef]
- Tarpey, M.M.; White, C.R.; Suarez, E.; Richardson, G.; Radi, R.; Freeman, B.A. Chemiluminescent Detection of Oxidants in Vascular Tissue. Lucigenin but Not Coelenterazine Enhances Superoxide Formation. Circ Res 1999, 84, 1203–1211. [Google Scholar] [CrossRef]
- González Rico, M.; Puchades, M.J.; García Ramón, R.; Saez, G.; Tormos, M.C.; Miguel, A. [Effect of oxidative stress in patients with chronic renal failure]. Nefrologia 2006, 26, 218–225. [Google Scholar] [PubMed]
- Usberti, M.; Gerardi, G.M.; Gazzotti, R.M.; Benedini, S.; Archetti, S.; Sugherini, L.; Valentini, M.; Tira, P.; Bufano, G.; Albertini, A.; et al. Oxidative Stress and Cardiovascular Disease in Dialyzed Patients. Nephron 2002, 91, 25–33. [Google Scholar] [CrossRef]
- Yeh, E.-L.; Chen, C.-H.; Huang, S.-C.; Huang, Y.-C. Higher Glutathione Demand May Be Necessary for Assisting Haemodialysis Patients to Cope with Increased Oxidative Stress. Nephrology (Carlton) 2020, 25, 90–95. [Google Scholar] [CrossRef]
- Marques, M.B.; Lima, T.F.O.; Guaratti, M.; Costa, M.C.; Saheb, J.L.; Brunetti, I.L.; Assis, R.P.; Baviera, A.M. Assessment of the Antioxidant Status and Markers of Oxidative Stress in Patients with Kidney Failure: Effects of a Hemodialysis Session. An Acad Bras Cienc 2024, 96, e20240297. [Google Scholar] [CrossRef]
- Varan, H.I.; Dursun, B.; Dursun, E.; Ozben, T.; Suleymanlar, G. Acute Effects of Hemodialysis on Oxidative Stress Parameters in Chronic Uremic Patients: Comparison of Two Dialysis Membranes. Int J Nephrol Renovasc Dis 2010, 3, 39–45. [Google Scholar] [CrossRef]
- Agarwal, R.; Vasavada, N.; Sachs, N.G.; Chase, S. Oxidative Stress and Renal Injury with Intravenous Iron in Patients with Chronic Kidney Disease. Kidney International 2004, 65, 2279–2289. [Google Scholar] [CrossRef]
- Garcia-Fernandez, N.; Echeverria, A.; Sanchez-Ibarrola, A.; Páramo, J.A.; Coma-Canella, I. Randomized Clinical Trial on Acute Effects of i.v. Iron Sucrose during Haemodialysis. Nephrology 2010, 15, 178–183. [Google Scholar] [CrossRef]
- Nguyen-Khoa, T.; Massy, Z.A.; De Bandt, J.P.; Kebede, M.; Salama, L.; Lambrey, G.; Witko-Sarsat, V.; Drüeke, T.B.; Lacour, B.; Thévenin, M. Oxidative Stress and Haemodialysis: Role of Inflammation and Duration of Dialysis Treatment. Nephrology Dialysis Transplantation 2001, 16, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Swarnalatha, G.; Ram, R.; Neela, P.; Naidu, M.U.R.; Dakshina Murty, K.V. Oxidative Stress in Hemodialysis Patients Receiving Intravenous Iron Therapy and the Role of N-Acetylcysteine in Preventing Oxidative Stress. Saudi J Kidney Dis Transpl 2010, 21, 852–858. [Google Scholar]
- Trimarchi, H.; Mongitore, M.R.; Baglioni, P.; Forrester, M.; Freixas, E. a. R.; Schropp, M.; Pereyra, H.; Alonso, M. N-Acetylcysteine Reduces Malondialdehyde Levels in Chronic Hemodialysis Patients--a Pilot Study. Clin Nephrol 2003, 59, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Dursun, E.; Ozben, T.; Süleymanlar, G.; Dursun, B.; Yakupoglu, G. Effect of Hemodialysis on the Oxidative Stress and Antioxidants. Clin Chem Lab Med 2002, 40, 1009–1013. [Google Scholar] [CrossRef]
- Sangeetha Lakshmi, B.; Harini Devi, N.; Suchitra, M.M.; Srinivasa Rao, P.V.L.N.; Siva Kumar, V. Changes in the Inflammatory and Oxidative Stress Markers during a Single Hemodialysis Session in Patients with Chronic Kidney Disease. Ren Fail 2018, 40, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Navarro-García, J.A.; Rodríguez-Sánchez, E.; Aceves-Ripoll, J.; Abarca-Zabalía, J.; Susmozas-Sánchez, A.; González Lafuente, L.; Bada-Bosch, T.; Hernández, E.; Mérida-Herrero, E.; Praga, M.; et al. Oxidative Status before and after Renal Replacement Therapy: Differences between Conventional High Flux Hemodialysis and on-Line Hemodiafiltration. Nutrients 2019, 11, 2809. [Google Scholar] [CrossRef]
- Hsu, S.-P.; Chiang, C.-K.; Yang, S.-Y.; Chien, C.-T. N-Acetylcysteine for the Management of Anemia and Oxidative Stress in Hemodialysis Patients. Nephron Clin Pract 2010, 116, c207–c216. [Google Scholar] [CrossRef]
- Zisimopoulos, D.N.; Kalaitzopoulou, E.; Skipitari, M.; Papadea, P.; Panagopoulos, N.T.; Salahas, G.; Georgiou, C.D. Detection of Superoxide Radical in All Biological Systems by Thin Layer Chromatography. Arch Biochem Biophys 2022, 716, 109110. [Google Scholar] [CrossRef]
- Grintzalis, K.; Zisimopoulos, D.; Grune, T.; Weber, D.; Georgiou, C.D. Method for the Simultaneous Determination of Free/Protein Malondialdehyde and Lipid/Protein Hydroperoxides. Free Radical Biology and Medicine 2013, 59, 27–35. [Google Scholar] [CrossRef]
- Georgiou, C.D.; Zisimopoulos, D.; Argyropoulou, V.; Kalaitzopoulou, E.; Salachas, G.; Grune, T. Protein and Cell Wall Polysaccharide Carbonyl Determination by a Neutral pH 2,4-Dinitrophenylhydrazine-Based Photometric Assay. Redox Biology 2018, 17, 128–142. [Google Scholar] [CrossRef]
- Zhang, N.; Liao, H.; Lin, Z.; Tang, Q. Insights into the Role of Glutathione Peroxidase 3 in Non-Neoplastic Diseases. Biomolecules 2024, 14, 689. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, A.; Akazaki, S.; Nagano, Y.; Ueda, A.; Chang-il Lee, M.; Aoyagi, K.; Oowada, S.; Sato, K. Hemodialysis Raises Oxidative Stress through Carbon-Centered Radicals despite Improved Biocompatibility. J. Clin. Biochem. Nutr. 2021, 69, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Schettler, V. Oxidative Stress during Dialysis: Effect on Free Radical Scavenging Enzyme (FRSE) Activities and Glutathione (GSH) Concentration in Granulocytes. Nephrology Dialysis Transplantation 1998, 13, 2588–2593. [Google Scholar] [CrossRef]
- Esterbauer, H.; Schaur, R.J.; Zollner, H. Chemistry and Biochemistry of 4-Hydroxynonenal, Malonaldehyde and Related Aldehydes. Free Radical Biology and Medicine 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Ward, R.A.; Ouseph, R.; Mcleish, K.R. Effects of High-Flux Hemodialysis on Oxidant Stress. Kidney International 2003, 63, 353–359. [Google Scholar] [CrossRef]
- Calo, L.A.; Naso, A.; Carraro, G.; Wratten, M.L.; Pagnin, E.; Bertipaglia, L.; Rebeschini, M.; Davis, P.A.; Piccoli, A.; Cascone, C. Effect of Haemodiafiltration with Online Regeneration of Ultrafiltrate on Oxidative Stress in Dialysis Patients. Nephrology Dialysis Transplantation 2007, 22, 1413–1419. [Google Scholar] [CrossRef] [PubMed]
- González-Diez, B.; Cavia, M.; Torres, G.; Abaigar, P.; Muñiz, P. Effect of a Hemodiafiltration Session with On-Line Regeneration of the Ultrafiltrate on Oxidative Stress. Blood Purif 2008, 26, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Liakopoulos, V.; Roumeliotis, S.; Gorny, X.; Eleftheriadis, T.; Mertens, P.R. Oxidative Stress in Patients Undergoing Peritoneal Dialysis: A Current Review of the Literature. Oxidative Medicine and Cellular Longevity 2017, 2017, 3494867. [Google Scholar] [CrossRef] [PubMed]
- Fujii, J.; Homma, T.; Osaki, T. Superoxide Radicals in the Execution of Cell Death. Antioxidants 2022, 11, 501. [Google Scholar] [CrossRef] [PubMed]
| HD patients | ||
|---|---|---|
| Age (years) Mean (± SD) | 25 to 86 62.4 (± 14.6) |
|
| Dialysis vintage (years) Mean (± SD) | 0.1 to 24.6 6.9 (± 5.5) |
|
| N | ||
| Gender | ||
| Male | 48 | |
| Female | 20 | |
| Medical status | ||
| Type of dialysis | Low-flux HD | 30 |
| High-flux HD | 17 | |
| Pre-dilution HDF | 21 | |
| Medical conditions | ||
| Hypertension | 32 | |
| Coronary artery disease (CAD) |
14 | |
| Cardiovascular disease (CVD) |
31 | |
| Peripheral artery disease (PAD) |
21 | |
| Diabetes | 24 | |
| Medication | ||
| Statin | 22 | |
| Alfacalcidol | 8 | |
| Paricalcitol | 9 | |
| Angiotensin-converting enzyme inhibitors (ACEi) |
3 | |
| Angiotensin receptor blockers (ARBs) |
9 | |
| Calcium channel blockers (CaChBl) |
19 | |
| B blockers | 38 | |
| Levocarnitine | 12 | |
| Cholecalciferol | 17 | |
| Oxidative stress markers | Modality of dialysis |
Pre-dialysis values | P value |
|---|---|---|---|
| O2•− | Low-flux HD | 22.03 (±9.16) | 0.929 |
| High-flux HD | 23.456 (±6.8) | ||
| Pre-dilution HDF | 23.09 (±11.22) | ||
| LOOH | Low-flux HD | 0.045 (±0.027) | 0.067 |
| High-flux HD | 0.067 (±0.023) | ||
| Pre-dilution HDF | 0.059 (±0.022) | ||
| PrMDA | Low-flux HD | 3.474 (±1.21) | 0.458 |
| High-flux HD | 4.04 (±0.99) | ||
| Pre-dilution HDF | 3.6 (±0.88) | ||
| PrTBARS | Low-flux HD | 7.643 (±2.41) | 0.725 |
| High-flux HD | 8.144 (±1.33) | ||
| Pre-dilution HDF | 7.547 (±2.48) | ||
| PrCO | Low-flux HD | 1.134 (±0.52) | 0.850 |
| High-flux HD | 1.143 (±0.26) | ||
| Pre-dilution HDF | 1.07 (±0.33) |
| Oxidative stress markers | Modality of dialysis |
Δ-value (pre – post dialysis) |
P value |
|---|---|---|---|
| O2•− | Low-flux HD | -4.59 (±2.2) | 0.185 |
| High-flux HD | -2.98(±3.3) | ||
| Pre-dilution HDF | -4.06 (±2.7) | ||
| LOOH | Low-flux HD | -0.045 (±0.031) | 0.016 |
| High-flux HD | -0.008 (±0.035) | ||
| Pre-dilution HDF | -0.018 (±0.029) | ||
| PrMDA | Low-flux HD | 0.53 (±0.66) | 0.910 |
| High-flux HD | 0.68 (±0.73) | ||
| Pre-dilution HDF | 0.59 (±0.99) | ||
| PrTBARS | Low-flux HD | 0.95 (±1.54) | 0.570 |
| High-flux HD | 1.71 (±1.92) | ||
| Pre-dilution HDF | 1.06 (±1.9) | ||
| PrCO | Low-flux HD | 0.027 (±0.29) | 0.701 |
| High-flux HD | 0.054 (±0.17) | ||
| Pre-dilution HDF | 0.024 (±0.27) |
| OS markers vs Age | Spearman’s rho coefficient (r) | P value |
|---|---|---|
| O2•− | -0.006 | 0.973 |
| LOOH | -0.121 | 0.483 |
| PrMDA | 0.303 | 0.16 |
| PrTBARS | 0.187 | 0.127 |
| PrCO | 0.204 | 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




