Submitted:
31 March 2026
Posted:
01 April 2026
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods Section Transcribed from Part of the Protocol Used with COVID-19 Patients in the Mexican Case [Reference [14]]
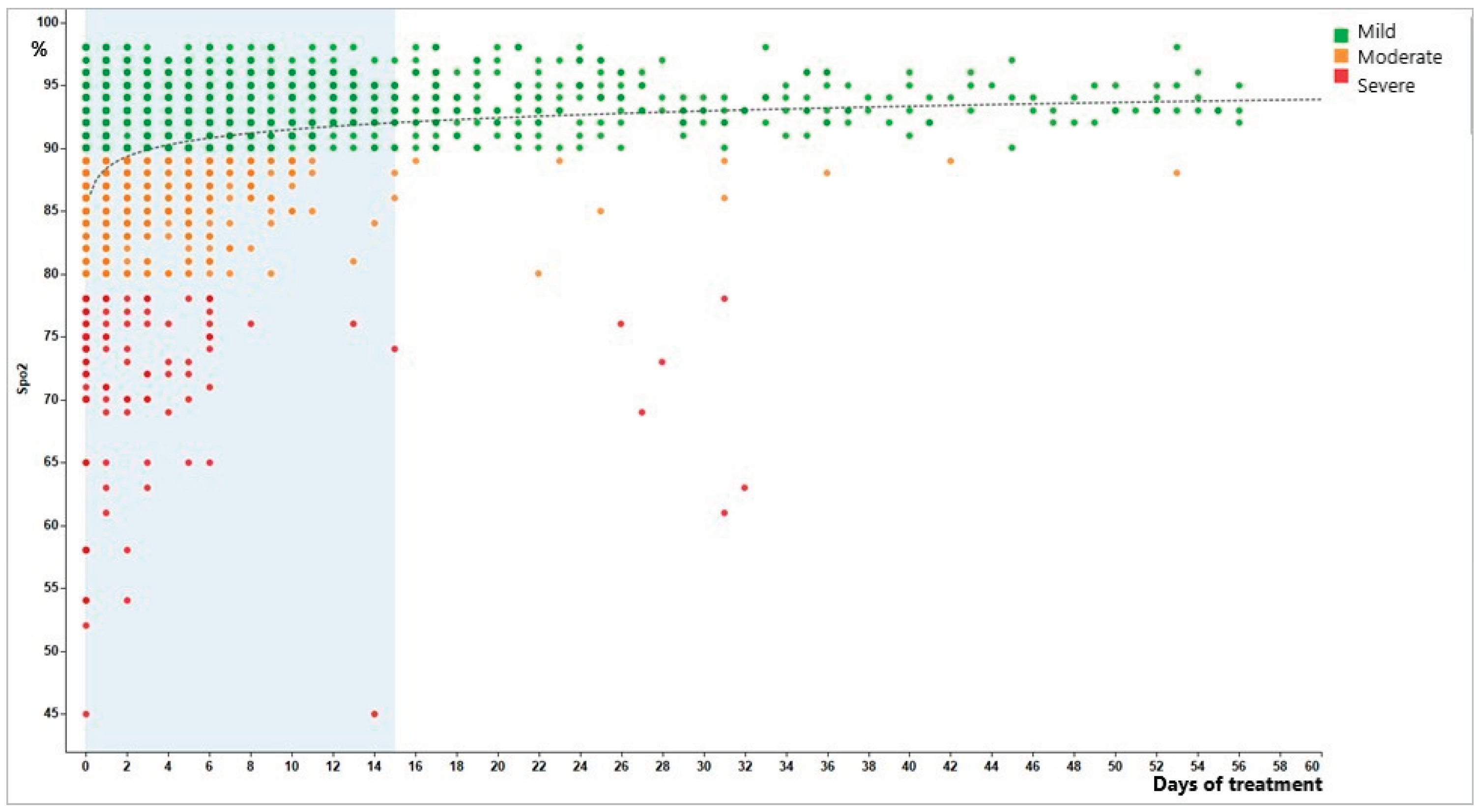
3. Results Transcription, Concerning the Observed Oxygen Saturation in Patients of the Mexican Case [Reference [14]]
4. Discussion Aimed to Give Better Understanding of the Oxygen Saturation Observed in Patients of the Mexican Case
5. Conclusions
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CDS | Chlorine Dioxide in Solution |
| HBOT | Hyper Baric Oxygen Treatment |
References
- Helou, M.; Mahdi, A.; Abou Fayad, A.; Sleiman A.; Matar, G. M.; Zoghbi S.; Madani T.; Rola Husni. Antimicrobial effects of chlorine dioxide in a hospital setting. Sci Rep. 2013, 13, 22866. [CrossRef]
- US-EPA. U.S. Environmental Protection Agency. Toxicological review of chlorine dioxide and chlorite 2000. https://cfpub.epa.gov/ncea/iris/iris_documents/documents/toxreviews/0496tr.pdf.
- ATSDR-Agency for Toxic Substances and Disease Registry. 2004, Evaluation of the toxicology of chemical mixtures commonly found at hazardous waste sites. Draft. Atlanta, GA.
- Pichert A, Arnhold J. Interaction of the chlorite-based drug WF10 and chlorite with hemoglobin, methemoglobin and ferryl hemoglobin. Arch. of Bioch. and Biophys. 2015, 585: 82-89.
- Condori Macuri, R.M.; Alzamora-Gonzales, L.; Colona-Vallejos, E.H.; Cruz Riquelme, R.T.; Pecho Chávez, L.I.; Cisneros Gutierrez, J.O.; Montejo Anlas, V.A. Synergistic Anticancer Activity of Fucoidan from Lessonia trabeculata Combined with Chemotherapeutic Agents in 4T1 Breast Spheroids. Mar. Drugs 2025; 23: 451. [CrossRef]
- Rattan, S.I.S. Hormesis in aging. Ageing Res. Rev. 2008; 7(1): 63-78. [CrossRef]
- Callisperis, P.; Franco-Paredes, C.; Liester, M.B. Necrotizing Fasciitis treatment with chlorine dioxide: A case report in patient. Cureus 2025, 17(7): e88800. [CrossRef]
- Callisperis, P.; Pineda Aquino, R.; Raj Kota, S.; Vargas, M.; Rodriguez, N.; Liester, M. Chlorine dioxide treatment for diabetic foot ulcers: Three case studies. Int. J Medicine and Medical Sci. 2024, Vol. 16(2), pp. 44-49 Article Number: 407DA8672766. [CrossRef]
- Rubio-Casillas, A.; Campras-Madrid, P. Farmacocinética y farmacodinamia del dióxido de cloro e-CUCBA 2021; 8(16):21-35. [CrossRef]
- Schwartz, L. Chlorine dioxide as a possible adjunct to metabolic treatment. J Cancer Treat. and Diag. 2017, 1:6–10. [CrossRef]
- Yildiz, S.; Bilir, C.; Eskiler, G.; Bilir, F. The Anticancer Potential of Chlorine Dioxide in Small-Cell Lung Cancer Cells. Cureus 2022, 14(10): e29989. [CrossRef]
- Aparicio-Alonso, M.; Torres-Solórzano, V. Case Report: Compassionate application of chlorine dioxide-based solution in a patient with metastatic prostate cancer. Salud, Ciencia y Tecnología 2024, 4:699. [CrossRef]
- Martinez, E.A. . A New Perspective for Prevention and to Cure COVID-19 Patients: Encouraging Medical Teams to Contact Healed People Treated with Chlorine Dioxide in Solution (CDS). Integr. J. of Med. Sci. 2020, 7,229. [CrossRef]
- Aparicio-Alonso M.; Domínguez-Sánchez C.A.; Banuet-Martínez M. Chlorine Dioxide as an Alternative Treatment for COVID-19. J. Infect Dis Ther. 2021, 9,5:477. https://www.omicsonline.org/open-access/chlorine-dioxide-as-an-alternative-treatment-for-covid19.pdf. ISSN: 2332-0877.
- David, S.R.; Sawal, N.S.; Bin Hamzah, M.N.; Rajabalaya, R. The blood blues: A review on methemoglobinemia. J Pharmacol Pharmacother. 2018;9:1-5.
- Schaffner, I.; Hofbauer, S.; Krutzler, M.; Pirker, K.F.; Furtmüller, P.G.; Obinger, C. Mechanism of chlorite degradation to chloride and dioxygen by the enzyme chlorite dismutase,. Arch. of Bioch. and Biophy. 2015; 574:18-26, ISSN 0003-9861. [CrossRef]
- Elstner EF. Heme activated oxydations using the chlorine-oxygen complex “TCDO” (Oxyferin)-an overview. J. of Biosc. 1988; 43 (11-12): 893-902.
- da Veiga Moreira, J.; Nleme, N.; Schwartz, L.; Leclerc-Desaulniers, K.; Carmona, E.; Mes-Masson, A-M.; Jolicoeur, M. Methylene Blue Metabolic Therapy Restrains In Vivo Ovarian Tumor Growth. Cancers 2024, 16: 355. [CrossRef]
- Cañeque, T.; Baron, L.; Müller, S.; Baron, L.; Müller, S.; Carmona, A.; Colombeau, L.; Versini, A.; Solier, S.; Gaillet, C.; Sindikubwabo, F.; Sampaio, J.L.; Sabatier, M.; Mishima, E.; Picard-Bernes, A.; Syx, L.; Servant ,N.; Lombard, B.; Loew ,D.; Zheng, J.; Proneth, B.; Thoidingjam, L.K.; Grimaud8, L.; Fraser, C.S.; Szylo, K.J.; Der Kazarian, E.; Bonnet, C.; Charafe-Jauffret, E.; Ginestier, C.; Santofimia-Castaño, P.; Estaras, M.; Dusetti, N.; Iovanna, J.L.; Sa Cunha, A.; Pittau, G.; Hammel, P.; Tzanis, D.; Bonvalot, S.; Watson, S.; Gandon, V; Upadhyay, A.; Pratt, D.A.; Porto Freitas, F.; Friedmann Angeli, J.P.; Stockwell, B.R.; Conrad, M.; Ubellacker, J.M.; Rodriguez, R. Activation of lysosomal iron triggers ferroptosis in cancer. Nature 2025; 642: 492–500. [CrossRef]
- Cai, T. Hyperbaric oxygen therapy as an adjunt treatment for glioma and brain metastasis: a literature review. Med Gas Res. 2025;15(3):420-426.
- Hinz, J.; Kuhne, F.W.; Stahl, K.W. Local Tetrachlorodecaoxide treatment to improve oxygen supply to non-healing wounds. The Lancet 1984, 324 (8403) 630.
- Dang, S. Efficacy of Hyperbaric Oxygen Therapy on Diabetic Foot Ulcer Healing. The PA Department J. of Med. Sci. 2025. https://digitalcommons.gardner-webb.edu/pa-department-journal-of-medical-science/14.
| COVID-19 Severity | ||||||
|---|---|---|---|---|---|---|
| Mild | Moderate | Severe | ||||
| SpO2 (%) | ≥ 95 | 90-94 | <90 | |||
| n | % | n | % | n | % | |
| Patients | 776 | 68.31 | 109 | 9.59 | 251 | 22.09% |
| Sex | ||||||
| Male | 351 | 45.23 | 49 | 44.95 | 151 | 60.16 |
| Female | 375 | 48.32 | 60 | 55.05 | 90 | 35.86 |
| Other | 50 | 6.44 | 0 | 0.00 | 10 | 3.98 |
| Age | ||||||
| 0-9 | 29 | 3.74 | 0 | 0.00 | 1 | 0.40 |
| 10-19 | 48 | 6.18 | 5 | 4.59 | 0 | 0.00 |
| 20-29 | 38 | 4.90 | 6 | 5.50 | 6 | 2.39 |
| 30-39 | 49 | 6.31 | 7 | 6.42 | 9 | 3.58 |
| 40-49 | 80 | 10.31 | 18 | 16.51 | 13 | 5.18 |
| 50-59 | 64 | 8.25 | 19 | 17.43 | 42 | 16.73 |
| 60-69 | 41 | 5.28 | 10 | 9.17 | 23 | 9.16 |
| >70 | 31 | 3.99 | 12 | 11.01 | 33 | 13.15 |
| No info | 396 | 51.03 | 32 | 29.36 | 124 | 42.63 |
| Days of symptoms Duration of treatment ClO2 dose (mg/kg) ClO2 per day (ml) Total ClO2 (ml) |
2.52-3.33a | 7.89-12.21bc | 6.73-9.95bc | |||
| 14.86-15.69a | 17.19-21.95b | 14.41-17.73c | ||||
| 0.87-0.94a | 1.16-1.33b | 1.98-2.18c | ||||
| 20.43-21.93a | 27.17-30.97b | 46.33-50.89c | ||||
| 309.83-337.38a | 518.77-619.19b | 733.67-828.79b | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




