Submitted:
01 April 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population and Specimen Collection
2.2. DNA Extraction and Quantification
2.3. Detection and Genotyping of HPV
2.4. Radiotherapy
2.5. Statystical Analysis
3. Results
3.1. Patients Characteristic
3.2. Association Between HPV Status and Clinical and Morphological Features
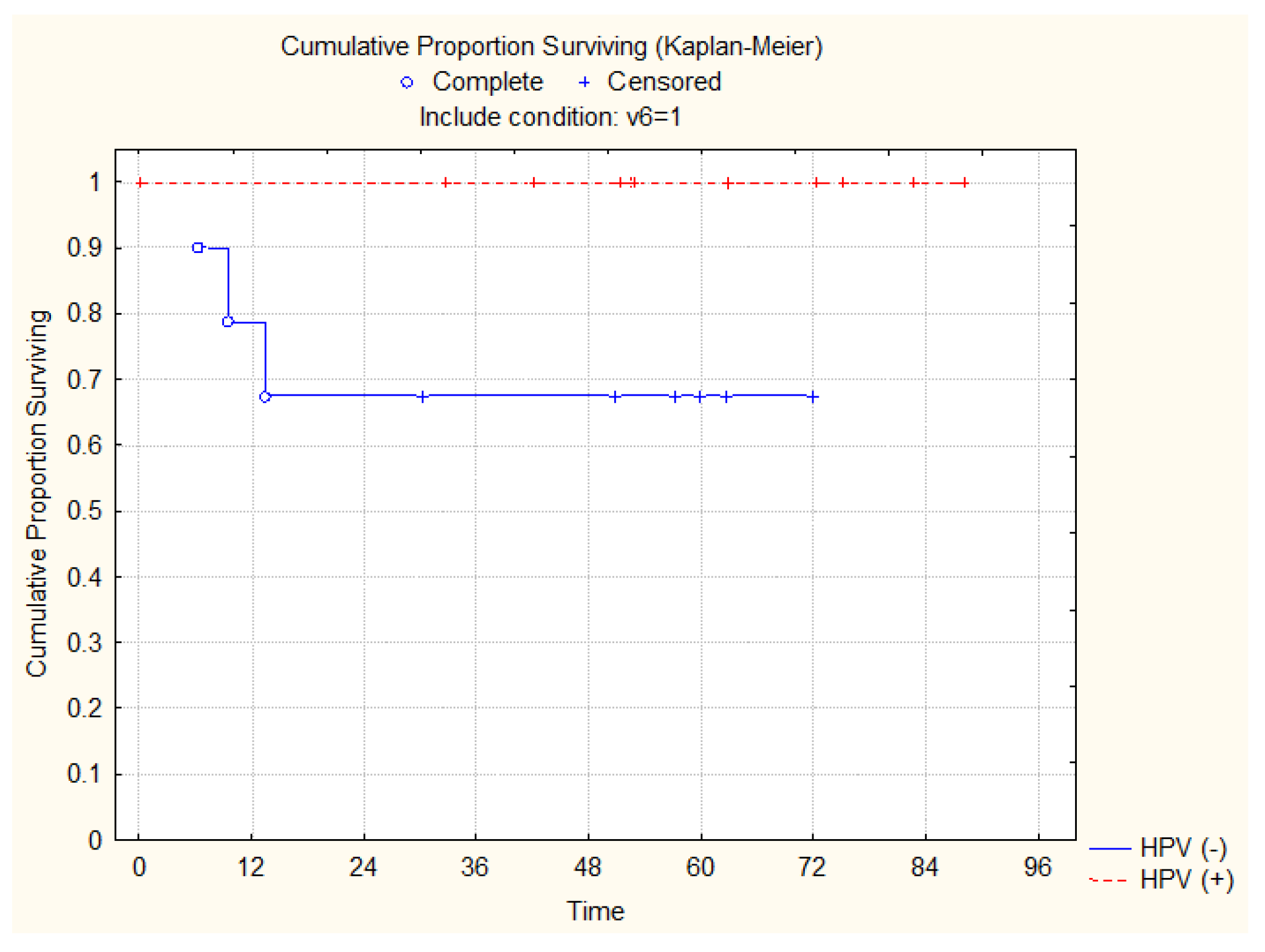
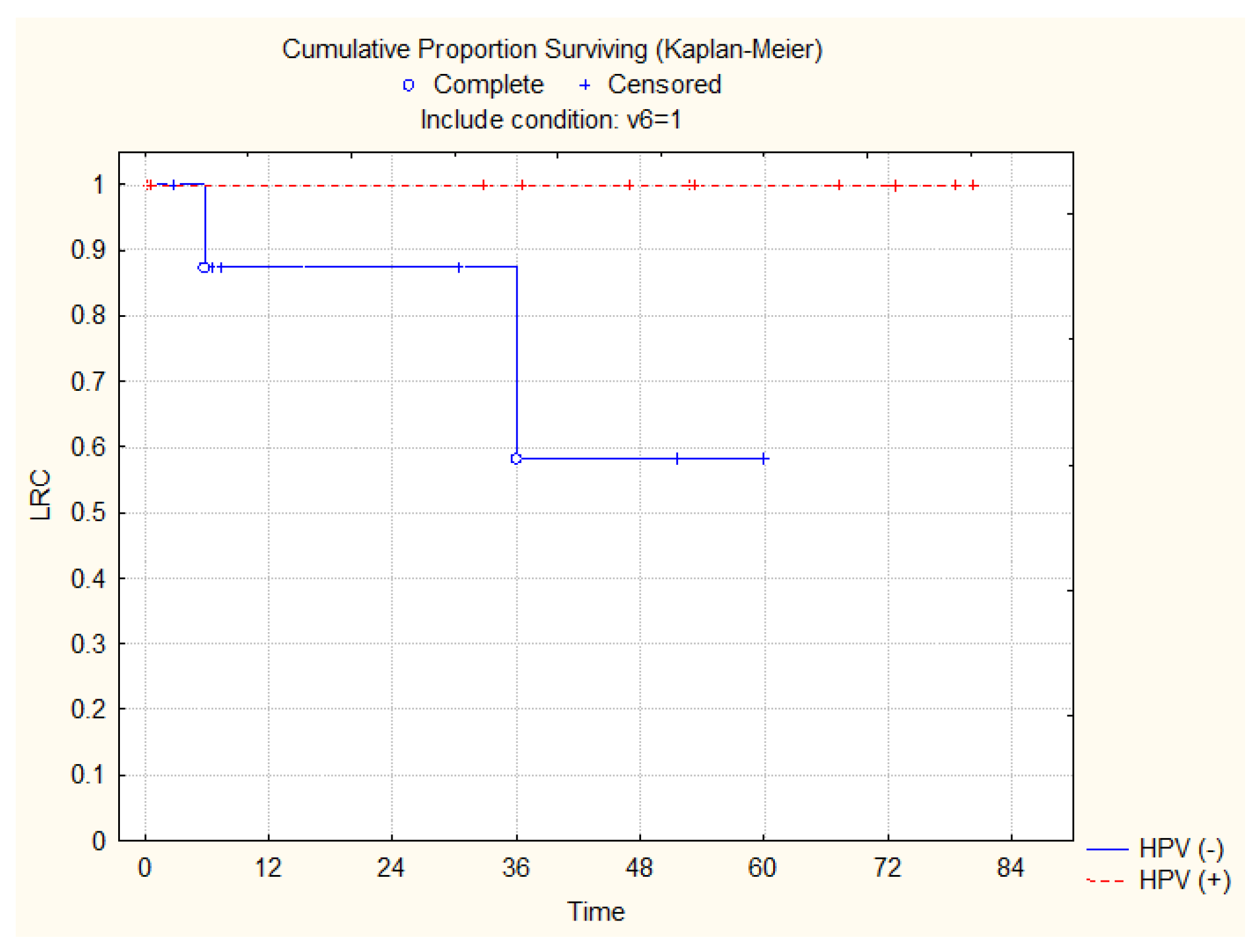
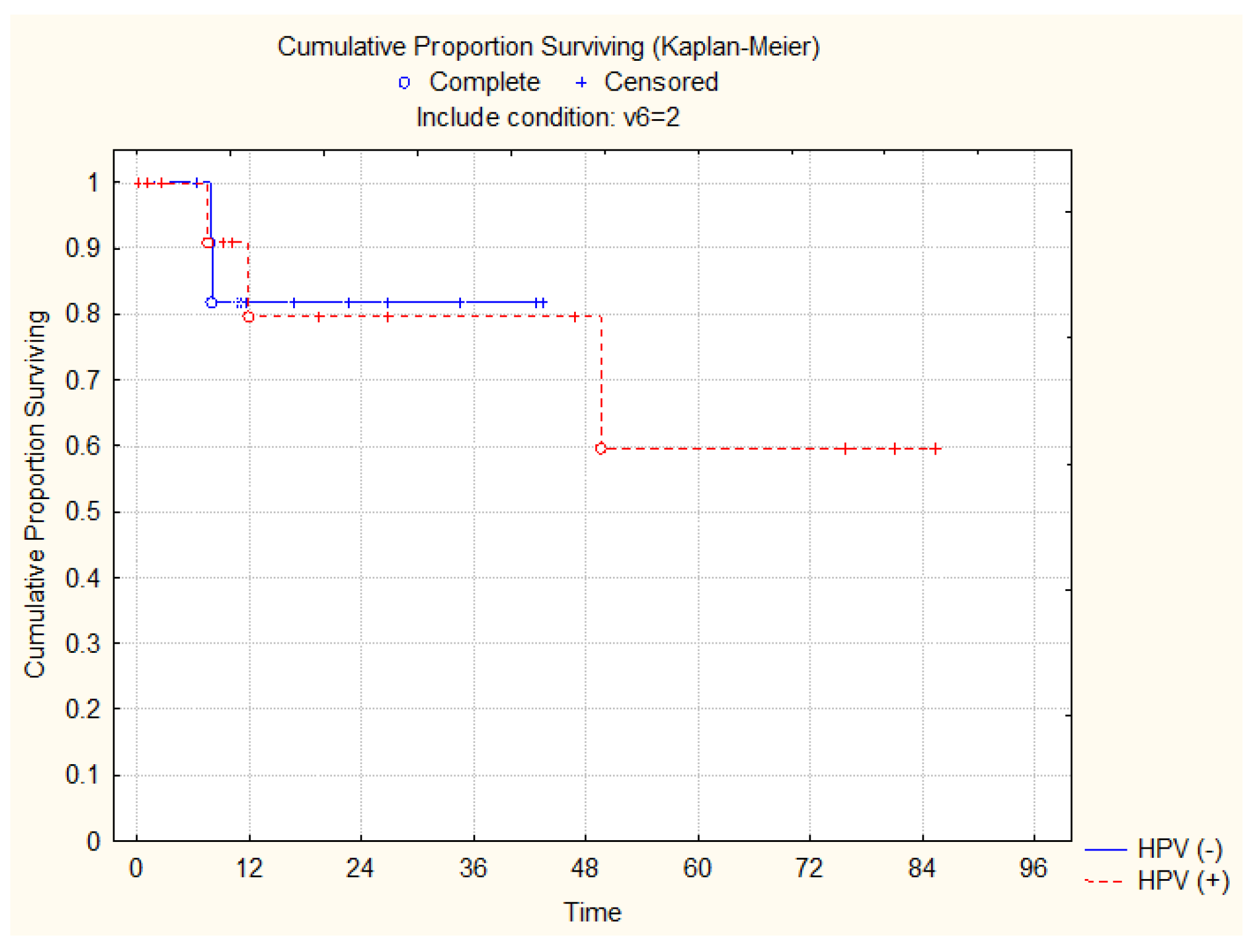
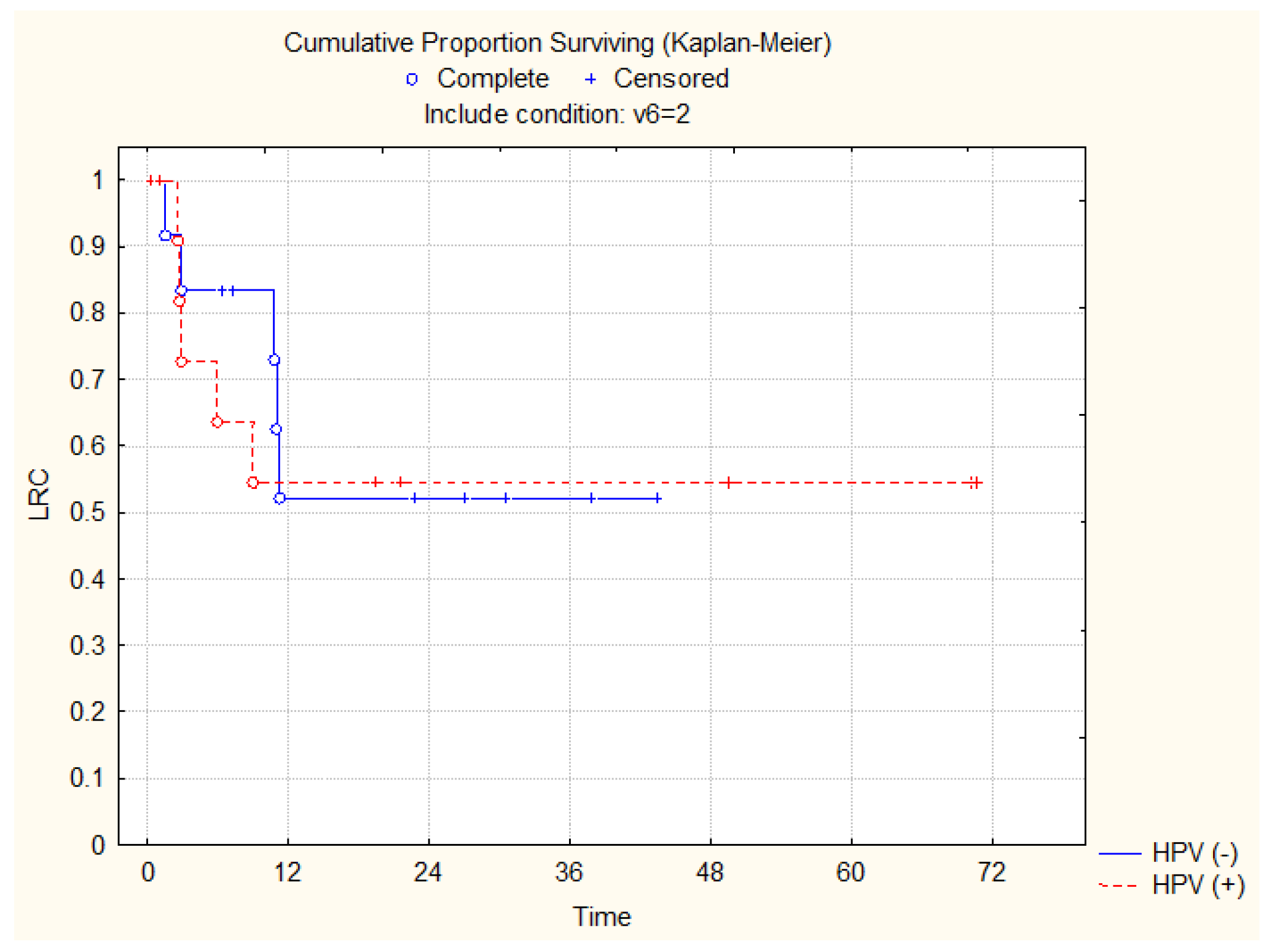
3.3. Overall Survival and Locoregional Control Rates
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HPV | Human Papilloma Virus |
| cT stage | clinical Tumor stage |
| cN stage | clinical Nodal stage |
| G | Grading |
| OS | Overall Survival |
| LRC | Locoregional Control |
| ECE | Extracapsular invasion |
| UICC | Union for International Cancer Control |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray.; F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021, 71, 209- 249. [CrossRef]
- Algudaibi, L.Y.; AlMeaigel, S.; AlQahtani, N.; Shaheen, N.A.; Aboalela, A. Oral and oropharyngeal cancer: Knowledge, attitude and practices among medical and dental practitioners. Cancer Rep (Hoboken). 2021, 4(4):e1349. [CrossRef]
- Moro, JDS.; Maroneze, MC.; Ardenghi, T.M.; Barin, L.M.; Danesi, C.C. Oral and oropharyngeal cancer: epidemiology and survival analysis. Einstein (Sao Paulo). 2018, 16(2):eAO4248. [CrossRef]
- Chi, A.C.; Day, T.A.; Neville, B.W. Oral cavity and oropharyngeal squamous cell carcinoma-an update. CA: A Cancer Journal for Clinicians. 2015, 65, 401-421. [CrossRef]
- You, E.L.; Henry, M.; Zeitouni, A.G. Human papillomavirus-associated oropharyngeal cancer: review of current evidence and management. Curr Oncol. 2019, 26(2), 119-123. [CrossRef]
- Sher, D.J.; Yan, J.; Day, A.; Sumer, B.D.; Pham, N.L.; Khan, S.; Zhu, H. Comparative effectiveness of primary radiotherapy versus surgery in elderly patients with locally advanced oropharyngeal squamous cell carcinoma. Oral Oncol. 2019, 88, 18-26. [CrossRef]
- Denittis, A.S.; Machtay, M.; Rosenthal, D.I.; Sanfilippo, N.J.; Lee, J.H.; Goldfeder, S.; Chalian, A.A.; Weinstein, G.S.; Weber, R.S. Advanced oropharyngeal carcinoma treated with surgery and radiotherapy: oncologic outcome and functional assessment. Am J Otolaryngol. 2001, 22(5), 329-35. [CrossRef]
- Pedro, C.; Mira, B.; Silva, P.; Netto, E.; Pocinho, R.; Mota, A.; Labareda, M.; Magalhães, M.; Esteves, S.; Santos, F. Surgery vs. primary radiotherapy in early-stage oropharyngeal cancer. Clin Transl Radiat Oncol. 2017, 28(9), 18-22. [CrossRef]
- Zhou, P.; Chen, DL.; Lian, C.L.; Wu, S.G.; Zhang, S.Y. The effect of human papillomavirus status on prognosis and local treatment strategies of T1-2N0 oropharyngeal squamous cell cancer. Front Public Health. 2022, 25, 10. [CrossRef]
- Li, H.; Zhang, X.; Chen, W.; Zhang, Q.; Li, Q.; Chen, S.; Yang, Z.; Su, X.; Yan, S.; Yang, A.; Song, M. Analysis of T1-T2 stage oropharyngeal squamous cell carcinoma treated with transoral robotic surgery. Laryngoscope Investig Otolaryngol. 2023, 8(1),103-112. [CrossRef]
- Kim, T.W.; Youm, H.Y.; Byun, H.; Son, Y.I.; Baek, C.H. Treatment Outcomes and Quality of Life in Oropharyngeal Cancer after Surgery-based versus Radiation-based Treatment. Clin Exp Otorhinolaryngol. 2010, 3(3), 153-60. [CrossRef]
- Patel, S.H.; Hinni, M.L.; Hayden, R.E.; Wong, W.W.; Dueck, A.C.; Zarka, M.A.; Curtis, K.K.; Halyard, M.Y. Transoral laser microsurgery followed by radiation therapy for oropharyngeal tumors: the Mayo Clinic Arizona experience. Head Neck. 2014, 36(2), 220-5. [CrossRef]
- Moore, E.J.; Olsen, S.M.; Laborde, R.R.; García, J.J.; Walsh, F.J.; Price, D.L.; Janus, J.R.; Kasperbauer, J.L.; Olsen, K.D. Long-term functional and oncologic results of transoral robotic surgery for oropharyngeal squamous cell carcinoma. Mayo Clin Proc. 2012, 87(3), 219-25. [CrossRef]
- Yuan, Y.; Wang, L.; Li, Q.X.; Zhang, J.Y.; Xu, Z.X.; Guo, C.B. Retrospective study of survival in human papillomavirus-negative oropharyngeal squamous cell carcinoma treated with primary surgery and associated prognostic factors. Onco Targets Ther. 2018, 27(11), 2355-2362. [CrossRef]
- Kelly, J.R.; Park, H.S.; An, Y.; Contessa, J.N.; Yarbrough, W.G.; Burtness, B.A.; Decker, R.; Husain, Z. Comparison of Survival Outcomes Among Human Papillomavirus-Negative cT1-2 N1-2b Patients With Oropharyngeal Squamous Cell Cancer Treated With Upfront Surgery vs Definitive Chemoradiation Therapy: An Observational Study. JAMA Oncol. 2017, 3(8), 1107-1111. [CrossRef]
- Parhar, H.S.; Weinstein, G.S.; O’Malley, B.W.Jr.; Shimunov, D.; Rassekh, C.H.; Chalian, A.A.; Newman, J.G.; Basu, D.; Cannady, S.B.; Rajasekaran, K.; Lin, A.; Lukens, J.N.; Swisher-McClure, S.; Cohen, R.B.; Bauml, J.M.; Aggrawal, C.; Brody, R.M. Oncologic outcomes of transoral robotic surgery for HPV-negative oropharyngeal carcinomas. Head Neck. 2021, 43(10), 2923-2934. [CrossRef]
- Spiotto, M.T.; Jefferson, G.; Wenig, B.; Markiewicz, M.; Weichselbaum, R.R.; Koshy, M. Differences in Survival With Surgery and Postoperative Radiotherapy Compared With Definitive Chemoradiotherapy for Oral Cavity Cancer: A National Cancer Database Analysis. JAMA Otolaryngol Head Neck Surg. 2017, 143(7), 691-699. [CrossRef]
- Bera, RN.; Tripathi R. Survival Outcomes and Factors Affecting Survival in Resectable Locally Advanced Oral Squamous Cell Carcinoma. Indian J Otolaryngol Head Neck Surg. 2023, 75(2), 607-616. [CrossRef]
- Fan, K.H.; Kang, C.J.; Lin, C.Y.; Ng, S.H.; Wang, H.M.; Hsieh, C.H.; Yeh, C.H.; Lin, C.H.; Tsao, C.K.; Huang, S.F.; Fang, K.H.; Wang, Y.C.; Chang, J.T.; Liao, C.T.; Lee, L.Y. Quantitative Measurement of Perineural Invasion for Prognosis Analysis of Oral Cavity Cancer Treated by Radical Surgery With or Without Adjuvant Therapy. Technol Cancer Res Treat. 2023, 22. [CrossRef]
- Hosni, A.; Huang, S.H.; Chiu, K.; Xu, W.; Su, J.; Bayley, A.; Bratman, S.V.; Cho, J.; Giuliani, M.; Kim, J.; O’Sullivan, B.; Ringash, J.; Waldron, J.; Spreafico, A.; Yu, E.; de Almeida, J.R.; Monteiro, E.; Chepeha, D.B.; Irish, J.C.; Goldstein, D.P.; Hope, A. Predictors of Early Recurrence Prior to Planned Postoperative Radiation Therapy for Oral Cavity Squamous Cell Carcinoma and Outcomes Following Salvage Intensified Radiation Therapy. Int J Radiat Oncol Biol Phys. 2019, 103(2), 363-373. [CrossRef]
- Shim, S.J.; Cha, J.; Koom, W.S.; Kim, G.E.; Lee, C.G.; Choi, E.C.; Keum, K.C. Clinical outcomes for T1-2N0-1 oral tongue cancer patients underwent surgery with and without postoperative radiotherapy. Radiat Oncol. 2010, 27(5), 43. [CrossRef]
- Hoffmann, M.; Saleh-Ebrahimi, L.; Zwicker, F.; Haering, P.; Schwahofer, A.; Debus, J.; Huber, PE.; Roeder, F. Long term results of postoperative Intensity-Modulated Radiation Therapy (IMRT) in the treatment of Squamous Cell Carcinoma (SCC) located in the oropharynx or oral cavity. Radiat Oncol. 2015, 4(10), 251. [CrossRef]
- Mione, C.; Casile, M.; Moreau, J.; Miroir, J.; Molnar, I.; Chautard, E.; Bernadach, M.; Kossai, M.; Saroul, N.; Martin, F.; Pham-Dang, N.; Lapeyre, M.; Biau, J. Outcomes among oropharyngeal and oral cavity cancer patients treated with postoperative volumetric modulated arctherapy. Front Oncol. 2023, 31(13), 1272856. [CrossRef]
- Daly, M.E.; Le, Q.T.; Kozak, M.M.; Maxim, P.G.; Murphy, J.D.; Hsu, A.; Loo, B.W.Jr.; Kaplan, M.J.; Fischbein, N.J.; Chang, D.T. Intensity-modulated radiotherapy for oral cavity squamous cell carcinoma: patterns of failure and predictors of local control. Int J Radiat Oncol Biol Phys. 2011, 80(5), 1412-22. [CrossRef]
- Collan, J.; Lundberg, M.; Vaalavirta, L.; Bäck, L.; Kajanti, M.; Mäkitie. A.; Tenhunen, M.; Saarilahti, K. Patterns of relapse following surgery and postoperative intensity modulated radiotherapy for oral and oropharyngeal cancer. Acta Oncol. 2011, 50(7), 1119-1125. [CrossRef]
- Quinlan-Davidson, S.R.; Mohamed, A.S.R.; Myers, J.N.; Gunn, G.B.; Johnson, F.M.; Skinner, H.; Beadle, B.M.; Gillenwater, A.M.; Phan, J.; Frank, S.J.; William, W.N.; Wong, A.J.; Lai, S.Y.; Fuller, C.D.; Morrison, W.H.; Rosenthal, D.I.; Garden, A.S. Outcomes of oral cavity cancer patients treated with surgery followed by postoperative intensity modulated radiation therapy. Oral Oncol. 2017, 72, 90-97. [CrossRef]
- Tangthongkum, M.; Phisalmongkhon, S.; Leelasawatsuk, P.; Supanimitjaroenporn, P.; Kirtsreesakul, V.; Tantipisit, J. Impact of human papillomavirus status on survival in patients with oral cancer. Laryngoscope Investig Otolaryngol. 2024, 9(3), e1294. [CrossRef]
- Wang, F.; Zhang, H.; Xue, Y.; Wen, J.; Zhou, J.; Yang, X.; Wei, J. A systematic investigation of the association between HPV and the clinicopathological parameters and prognosis of oral and oropharyngeal squamous cell carcinomas. Cancer Med, 2017 6(5), 910-917. [CrossRef]
- Doll, C.; Steffen, C.; Beck-Broichsitter, B.; Richter, M.; Neumann, K.; Pohrt, A.; Lohneis, P.; Lehmann, A.; Heiland, M.; Stromberger, C.; Coordes, A.; Johrens, K.; Raguse, J.D. The Prognostic Significance of p16 and its Role as a Surrogate Marker for Human Papilloma Virus in Oral Squamous Cell Carcinoma: An Analysis of 281 Cases. Anticancer Res. 2022, 42(5), 2405-2413. [CrossRef]
- Nauta, I.H.; Heideman, D.A.M.; Brink, A.; van der Steen, B.; Bloemena, E.; Koljenović, S.; Baatenburg, de Jong R.J.; Leemans, C.R.; Brakenhoff, R.H. The unveiled reality of human papillomavirus as risk factor for oral cavity squamous cell carcinoma. Int J Cancer. 2021, 149(2), 420-430. [CrossRef]
- Strzelczyk, J.K.; Biernacki, K.; Gaździcka, J.; Chełmecka, E.; Miśkiewicz-Orczyk, K.; Zięba, N.; Strzelczyk, J.; Misiołek, M. The Prevalence of High- and Low-Risk Types of HPV in Patients with Squamous Cell Carcinoma of the Head and Neck, Patients with Chronic Tonsillitis, and Healthy Individuals Living in Poland. Diagnostics 2021, 11, 2180. [CrossRef]
- Sobin, L.H.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours. 7th ed.Oxford: Wiley-Blackwell; 2009.
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017, 67(2), 93-99. [CrossRef]
- Lee, L.A.; Huang, C.G.; Liao, C.T.; Lee, L.Y.; Hsueh, C.; Chen, T.C.; Lin, C.Y.; Fan, K.H.; Wang, H.M.; Huang, S.F.; Chen, I.H.; Kang, C.J.; Ng, S.H.; Yang, S.L.; Tsao, K.C.; Chang, Y.L.; Yen, T.C. Human papillomavirus-16 infection in advanced oral cavity cancer patients is related to an increased risk of distant metastases and poor survival. PLoS One. 2012, 7(7), e40767. [CrossRef]
- Lee, L.A.; Huang, C.G.; Tsao, K.C.; Liao, C.T.; Kang, C.J.; Chang, K.P.; Huang, S.F.; Chen, I.H.; Fang, T.J.; Li, H.Y.; Yang, S.L.; Lee, L.Y.; Hsueh, C.; Chen, T.C.; Lin, C.Y.; Fan, K.H.; Wang, H.M.; Ng, S.H.; Chang, Y.L.; Lai, C.H.; Shih, S.R.; Yen, T.C. Increasing rates of low-risk human papillomavirus infections in patients with oral cavity squamous cell carcinoma: association with clinical outcomes. J Clin Virol. 2013, 57(4), 331-7. [CrossRef]
- Pytynia, K.B.; Dahlstrom, K.R.; Sturgis, E.M. Epidemiology of HPV-associated oropharyngeal cancer. Oral Oncol. 2014, 50(5), 380-386. [CrossRef]
- Woods, R.Sr.; O’Regan, E.M.; Kennedy, S.; Martin, C.; O’Leary, J.J.; Timon, C. Role of human papillomavirus in oropharyngeal squamous cell carcinoma: A review. World J Clin Cases. 2014, 12(6), 172-93. [CrossRef]
- Martinez, I.; Wang, J.; Hobson, K.F.; Ferris, R.L.; Khan, S.A. Identification of differentially expressed genes in HPV-positive and HPV-negative oropharyngeal squamous cell carcinomas. Eur J Cancer. 2007, 43(2), 415-432. [CrossRef]
- Gillison, M.L.; Chaturvedi, A.K.; Anderson, W.F.; Fakhry, C. Epidemiology of Human Papillomavirus-Positive Head and Neck Squamous Cell Carcinoma. J Clin Oncol. 2015, 33(29), 3235-3242. [CrossRef]
- Saito, Y.; Hayashi, R.; Iida, Y.; Mizumachi, T.; Fujii, T.; Matsumoto, F.; Beppu, T.; Yoshida, M.; Shinomiya, H.; Kamiyama, R.; Kitano, M.; Yokoshima, K.; Fujimoto, Y.; Hama, T.; Yamashita, T.; Okami, K.; Miura, K.; Fujisawa, T.; Sano, D.; Kato, H.; Minami, S.; Sugasawa, M.; Masuda, M.; Ota, I.; Iwae, S.; Kawata, R.; Monden, N.; Imai, T.; Asakage, T.; Okada, M.; Yoshikawa, T.; Tanioka, K.; Kitayama, M.; Doi, M.; Fujii, S.; Fujii, M.; Oridate, N.; Nakamizo, M.; Yoshimoto, S.; Homma, A.; Nibu, KI.; Yane. K. Optimization of therapeutic strategy for p16-positive oropharyngeal squamous cell carcinoma: Multi-institutional observational study based on the national Head and Neck Cancer Registry of Japan. Cancer. 2020, 126(18), 4177-4187. [CrossRef]
- Hughes, RT.; Levine, B.J.; May, N.; Shenker, R.F.; Yang, J.H.; Lanier, C.M.; Frizzell, B.A.; Greven, K.M.; Waltonen, J.D. Survival and Swallowing Function after Primary Radiotherapy versus Transoral Robotic Surgery for Human Papillomavirus-Associated Oropharyngeal Squamous Cell Carcinoma. ORL J Otorhinolaryngol Relat Spec. 2023, 85(5), 284-293. [CrossRef]
- Kelly, J.R.; Park, H.S.; An, Y.; Yarbrough, W.G.; Contessa, J.N.; Decker, R.; Mehra, S.; Judson, B.L.; Burtness, B.; Husain, Z. Upfront surgery versus definitive chemoradiotherapy in patients with human Papillomavirus-associated oropharyngeal squamous cell cancer. Oral Oncol. 2018, 79, 64-70. [CrossRef]
- Jackson, R.S.; Chen. S.; Last, A.; Khan, A.; Kallogjeri, D.; Van Abel, K.M.; Moore, E.J.; Pipkorn, P. Multi-institutional analysis of outcomes following transoral surgery for HPV-positive oropharyngeal squamous cell carcinoma in elderly patients. Head Neck. 2019, 41(11), 3933-3939. [CrossRef]
- Mehanna, H.; Rapozo, D.; von Zeidler, S.V.; Harrington, K.J.; Winter, S.C.; Hartley, A.; Nankivell, P.; Schache, A.G.; Sloan, P.; Odell, E.W.; Thavaraj, S.; Hunter, K.D.; Shah, K.A.; Thomas, G.J.; Long, A.; Amel-Kashipaz, R.; Brown, R.M.; Conn, B.; Hall, G.L.; Matthews, P.; Weir, J.; Yeo, Y.; Pring M.; West, C.M.L.; McCaul, J.; Golusinski, P.; Sitch, A.; Spruce, R.; Batis, N.; Bryant, JL.; Brooks, J.M.; Jones, T.M.; Buffa, F.; Haider, S.; Robinson, M. Developing and Validating a Multivariable Prognostic-Predictive Classifier for Treatment Escalation of Oropharyngeal Squamous Cell Carcinoma: The PREDICTR-OPC Study. Clin Cancer Res. 2024, 30(2), 356-367. [CrossRef]
- Kim, D.Y.; Wu, H.G.; Kim, J.H.; Lee, J.H.; Ahn, S.H.; Chung, E.J.; Eom, K.Y.; Jung, Y.H.; Jeong, W.J.; Kwon, T.K.; Kim, S.; Wee, C.W. Radiotherapy versus Surgery in Early-Stage HPV-Positive Oropharyngeal Cancer. Cancer Res Treat. 2022, 54(2), 406-416. [CrossRef]
- Wright, C.M.; Baron, J.; Lee, D.Y.; Kim, M.; Barsky, A.R.; Teo, B.K.; Lukens, J.N.; Swisher-McClure, S.; Lin, A. Dosimetric Results for Adjuvant Proton Radiation Therapy of HPV-Associated Oropharynx Cancer. Int J Part Ther. 2021, 8(4), 47-54. [CrossRef]
- Shenker, R.F.; May, N.H.; Waltonen, J.D.; Yang, J.P.; O’Neill, S.S.; Frizzell, B.A.; Greven, K.M.; Hughes, R.T. Comparing Outcomes for Patients with Human Papillomavirus (HPV) Type 16 versus Other High-Risk HPV Types in Oropharyngeal Squamous Cell Carcinoma. Head Neck Pathol. 2021, 15(3), 866-874. [CrossRef]
- Bloebaum, M.; Poort, L.; Böckmann, R.; Kessler, P. Survival after curative surgical treatment for primary oral squamous cell carcinoma. J Craniomaxillofac Surg. 2014, 42(8), 1572-6. [CrossRef]
- Kawecki, A. Nowotwory. Journal of Oncology. 2016, 66(1): 70–74doi: 10.5603/NJO.2016.0010.
- Lewis, J.S.; Thorstad, W.L.; Chernock, R.D.; Haughey, B.H.; Yip, J.H.; Zhang, Q.M.S.; El-Mofty, S.K. p16 Positive Oropharyngeal Squamous Cell Carcinoma:An Entity With a Favorable Prognosis Regardless of Tumor HPV Status. Am J of Surg Pathol. 2010, 34(8), 1088-1096. [CrossRef]
- Weinberger, P.M.; Yu, Z.; Haffty, B.G.; Kowalski, D.; Harigopal, M.; Sasaki, C.; Rimm, D.L.; Psyrri, A. Prognostic significance of p16 protein levels in oropharyngeal squamous cell cancer. Clin Cancer Res. 2004, 10(17), 5684-5691. [CrossRef]
- Reimers, N.; Kasper, H.U.; Weissenborn, S.J.; Stützer, H.; Preuss, S.F.; Hoffmann, T.K.; Speel, E.J.M.; Dienes, H.P.; Pfister, H.J.; Guntinas-Lichius, O. Combined analysis of HPV-DNA, p16 and EGFR expression to predict prognosis in oropharyngeal cancer. Int J Cancer. 2007, 120(8), 1731–1738. [CrossRef]
- Gormley, M.; Creaney, G.; Schache, A.; Ingarfield, K.; Conway, D.I. Reviewing the epidemiology of head and neck cancer: Definitions, trends and risk factors. Br Dent J. 2022, 233, 780–786.
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and neck squamous cell carcinoma. Nat Rev Dis. Primers 2020, 6, 92.
- Shlomo, E.M.; Koyfman, A.; Weiss, J. Personalizing Postoperative Treatment of Head and Neck Cancers. Am Soc Clin Oncol Educ Book, 2018, 515-522. doi: 10.1200/EDBK_20108.
- Fakhry, C.; Westra, W.H.; Cmelak, A.; Ridge, J.A.; Pinto, H.; Forastiere, A., Gillison, M.L. Improved Survival of Patients With Human Papillomavirus–Positive Head and Neck Squamous Cell Carcinoma in a Prospective Clinical Trial. JNCI: Journal of the National Cancer Institute. 2008, 100(4), 261–269. [CrossRef]
- Johnson, D.E.; Burtness, B.C.; Lemans, R.; Liu, V.W.L.; Bauman, J.E.; Grandis, J.R. Head and neck squamous cell carcinoma. Nature Reviews Disease Primers. 6: 2020: 92. [CrossRef]
| Type of adjuvant treatment | N (%) |
| None | 18 (35%) |
| Radiochemotherapy | 6 (12%) |
| Radiotherapy | 24 (47%) |
| Palliative radiotherapy | 2 (4%) |
| Brachytherapy | 1 (2%) |
| Feature | N (%) |
| Location | |
| Oral cavity | 28 (55%) |
| Oropharynx | 23 (45%) |
| cT stage | |
| T1 | 11 (22%) |
| T2 | 23 (45%) |
| T3 | 16 (31%) |
| T4 | 1 (2%) |
| cN stage | |
| N0 | 25 (49%) |
| N1 | 2 (4%) |
| N2a | 3 (6) |
| N2b | 12 (24%) |
| N2c | 6 (12) |
| N3 | 3 (6) |
| Grading | |
| G1 | 9 (18%) |
| G2 | 24 (47%) |
| G3 | 18 (35%) |
| HPV status | |
| HPV-positive | 27 (53%) |
| HPV-negative p16 p16+ p16 - |
24 (47%) 21 (41%) 30 (59%) |
| Feature | HPV-positive | HPV-negative | Chi2; p |
| Location | |||
| Oral cavity | 14 (52%) | 14 (58%) | Chi2=0.216; p=0.642 |
| Oropharynx | 13 (48%) | 10 (42%) | |
| Grading | |||
| G1 | 8 (30%) | 1 (4%) | Chi2=7.01; p=0.069 |
| G2 | 9 (33%) | 15 (62.5%) | |
| G3 | 10 (37%) | 8 (33.5%) | |
| cT stage | |||
| T1 | 6 (22%) | 5 (21%) | Chi2=0.96; p=0.810 |
| T2 | 12 (44%) | 11 (46%) | |
| T3 | 8 (30%) | 8 (33%) | |
| T4 | 1 (4%) | 0 | |
| cN stage | |||
| N0 | 15 (56%) | 10 (42%) | Chi2=2.16; p=0.826 |
| N1 | 1 (4%) | 1 (4%) | |
| N2a | 2 (7%) | 1 (4%) | |
| N2b | 6 (22%) | 6 (25%) | |
| N2c | 2 (7%) | 4 (17%) | |
| N3 | 1 (4%) | 2 (8%) | |
| Adjuvant treatment | |||
| No | 13 (48%) | 5 (21%) | Chi2=4.15; p=0.041 |
| Yes | 14 (52%) | 19 (79%) | |
| Factor | Univariate Cox proportional hazard analysis (Wald statistics) | p-value | Kaplan-Meier comparison log-rank test | p-value |
| Gender M vs F |
0.31 | 0.57 | 0.54 | 0.588 |
| Tumor location Oropharynx vs oral cavity |
0.90 | 0.34 | 0.96 | 0.339 |
| T-stage T1-2 vs T3-4 |
0.39 | 0.53 | 0.65 | 0.515 |
| N-stage N0-2a vs N2b+ |
1.06 | 0.30 | 1.06 | 0.285 |
| HPV status Negative vs positive |
1.16 | 0.28 | 1.09 | 0.273 |
| Adjuvant treatment No vs yes |
0.47 | 0.49 | 0.65 | 0.516 |
| Factor | Univariate Cox proportional hazard analysis (Wald statistics) | p-value | Kaplan-Meier comparison log-rank test | p-value |
| Gender M vs F |
0.04 | 0.85 | 0.19 | 0.848 |
| T-stage |
0.14 | 0.71 | 0.38 | 0.706 |
| N-stage N0-2a vs N2b+ |
0.11 | 0.74 | 0.33 | 0.739 |
| HPV status Negative vs positive |
0.00 | 0.96 | 0.04 | 0.965 |
| Adjuvant treatment No vs yes |
0.00 | 0.99 | 0.00 | 0.99 |
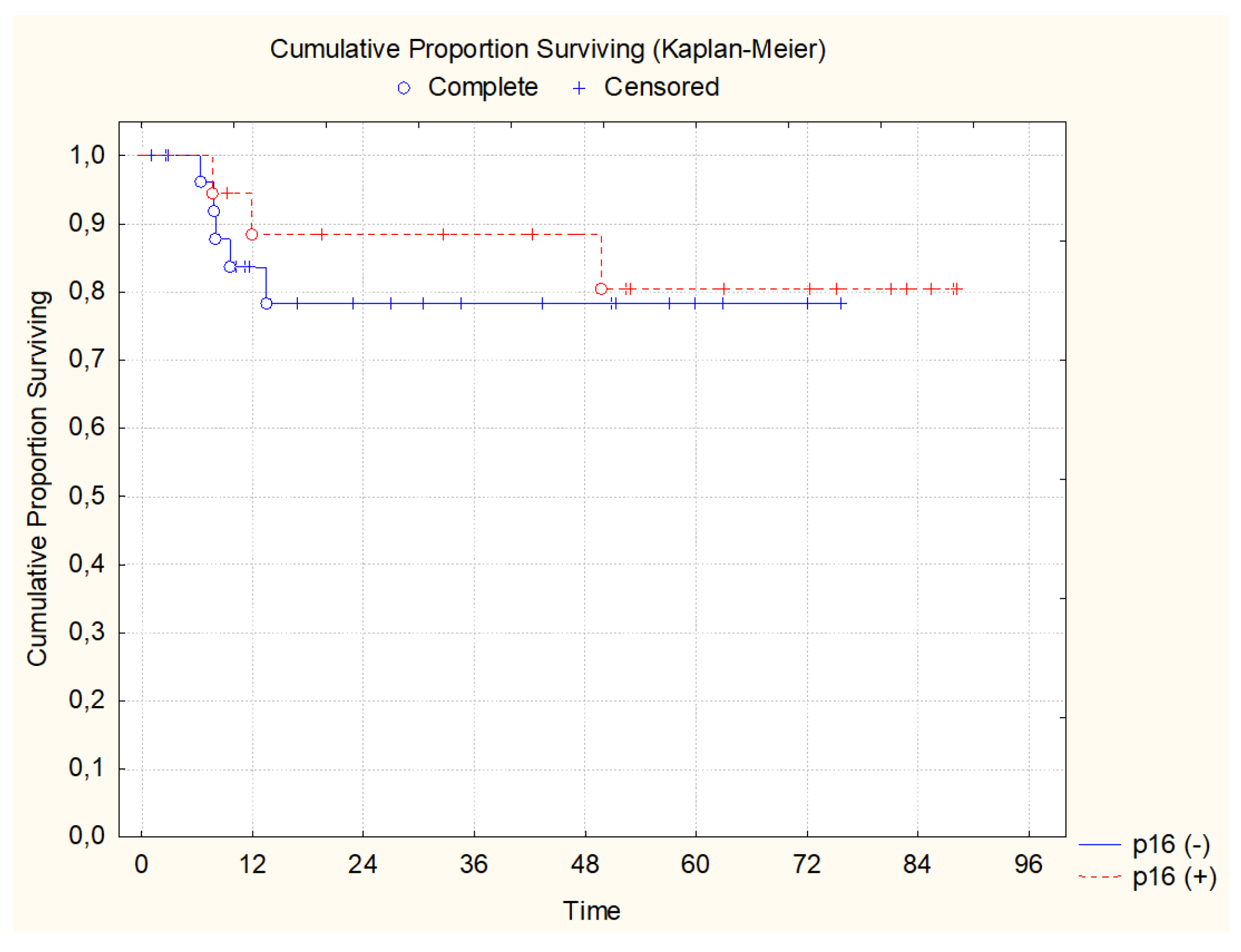
| Factor | Univariate Cox proportional hazard analysis (Wald statistics) | p-value | Kaplan-Meier comparison log-rank test | p-value |
| Gender M vs F |
Not estimated* | 0.72 | 0.469 | |
| T-stage T1-2 vs T3-4 |
Not estimated* | 1.37 | 0.169 | |
| N-stage N0-2a vs N2b+ |
Not estimated* | 1.68 | 0.092 | |
| HPV status Negative vs positive |
Not estimated* | 2.00 | 0.045 | |
| Adjuvant treatment No vs yes |
0.52 | 0.48 | 0.64 | 0.522 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




