Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Methods
2.1. Study Design
2.2. Study Population
2.3. Variables and Data Sources
2.4. Definitions and Endpoints
2.5. Classification of Trial-Eligible vs. Real-World Patients
2.6. Quality Indicators and Guidelines Adherence
3. Statistical Analysis
4. Results
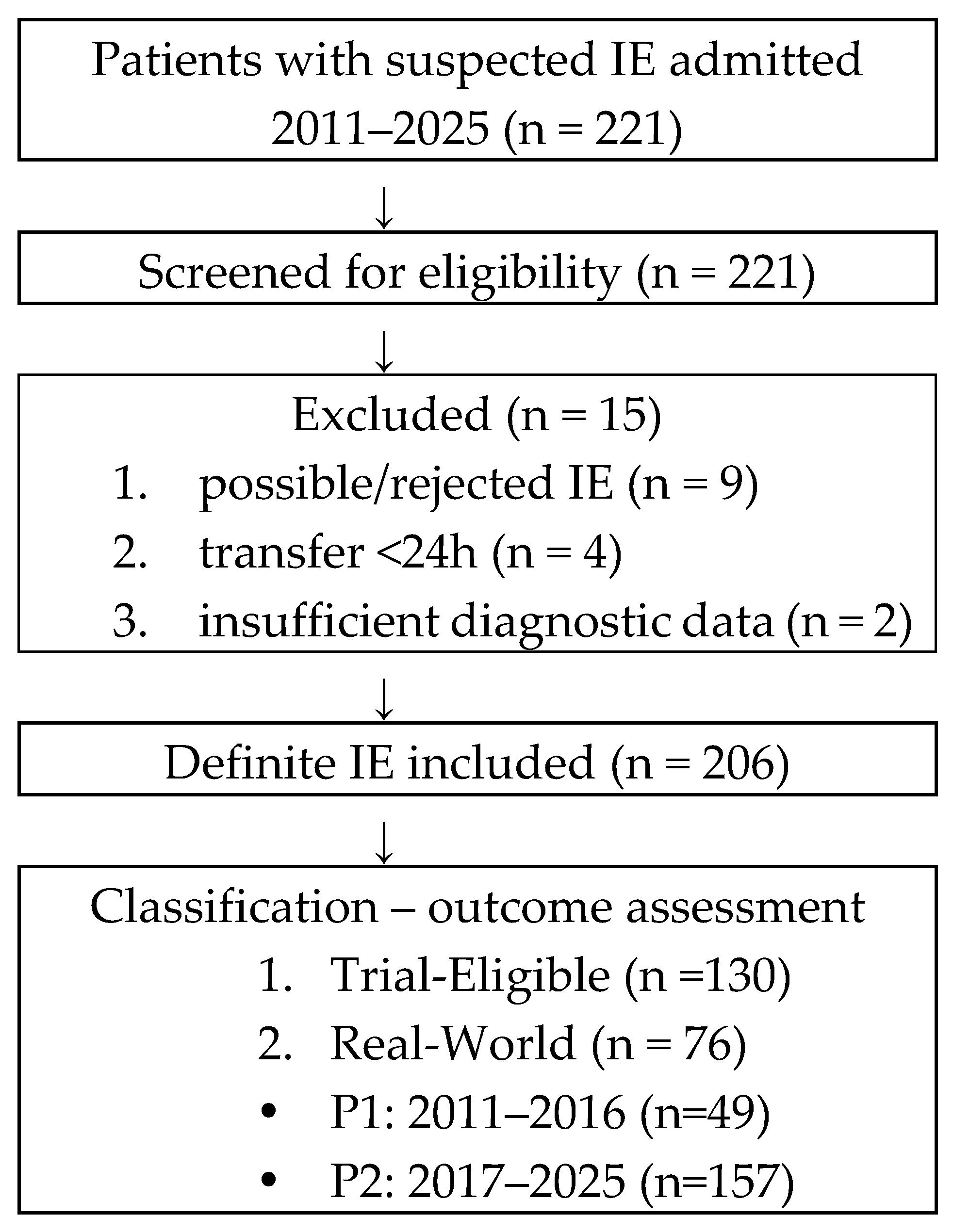
4.1. Study Population
4.2. Baseline Characteristics According to Trial Eligibility
4.3. Temporal Trends Between Study Periods
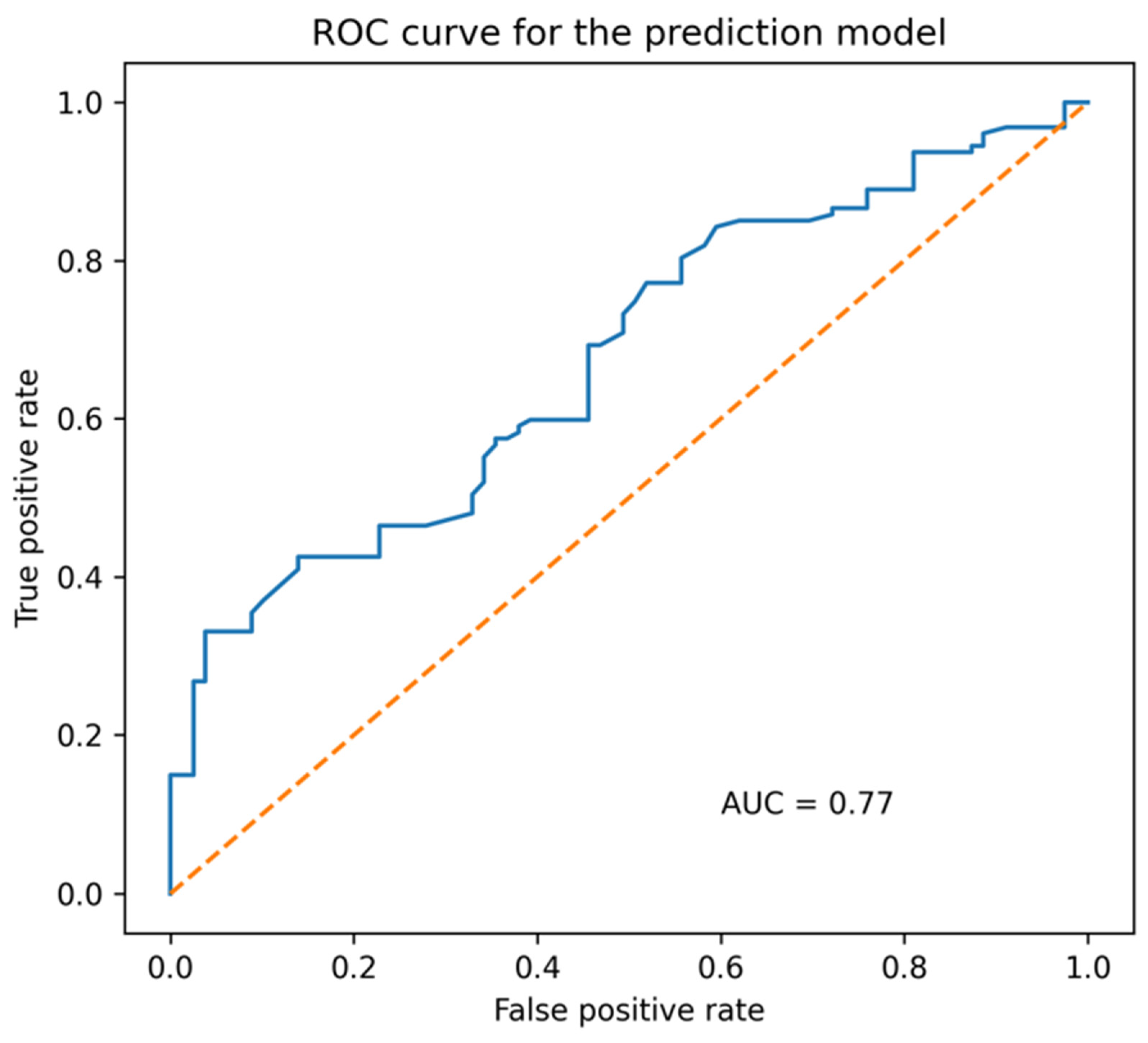
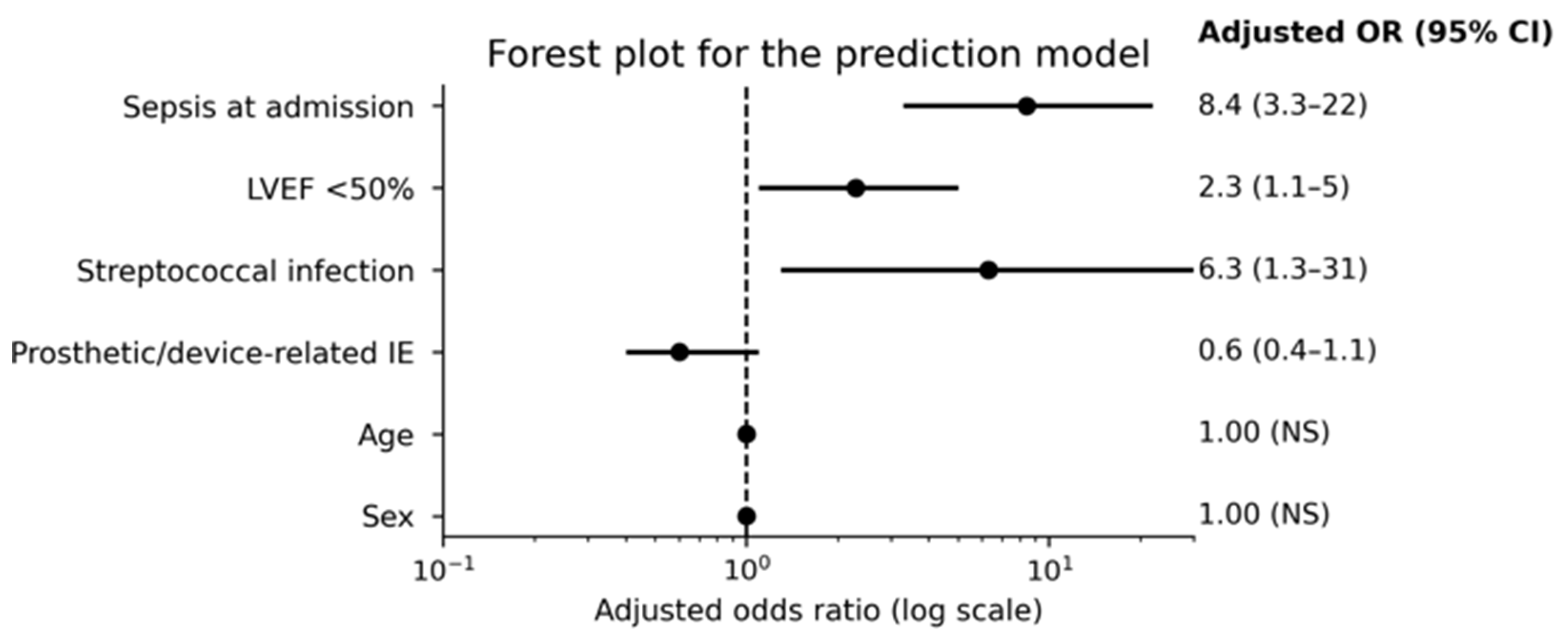
4.4. Prediction of the Composite Endpoint
4.5. Quality Indicators and Outcome Linkage
5. Discussion
6. Study Strengths and Limitations
7. Future Directions
8. Conclusions
- RW IE patients differ substantially from RCT populations.
- Guideline adherence improved markedly over time.
- Complication burden increased due to greater severity.
- Mortality remained stable despite higher clinical complexity.
- Early severity markers predict in-hospital outcomes.
- Structured care pathways support consistent management.
- ESC quality indicator adherence is achievable in regional hospitals.
- Regional registries complement tertiary sources of evidence.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Delgado, V.; Ajmone Marsan, N.; De Waha, S.; Bonaros, N.; Brida, M.; Burri, H.; Caselli, S.; Doenst, T.; Ederhy, S.; Erba, P.A.; et al. 2023 ESC Guidelines for the Management of Endocarditis. European Heart Journal 2023, 44, 3948–4042. [Google Scholar] [CrossRef]
- Ambrosioni, J.; Hernández-Meneses, M.; Durante-Mangoni, E.; Tattevin, P.; Olaison, L.; Freiberger, T.; Hurley, J.; Hannan, M.M.; Chu, V.; Hoen, B.; et al. Correction: Epidemiological Changes and Improvement in Outcomes of Infective Endocarditis in Europe in the Twenty-First Century: An International Collaboration on Endocarditis (ICE) Prospective Cohort Study (2000–2012). Infect. Dis. Ther. 2023, 12, 2819–2821. [Google Scholar] [CrossRef]
- Habib, G.; Lancellotti, P.; Antunes, M.J.; Bongiorni, M.G.; Casalta, J.-P.; Del Zotti, F.; Dulgheru, R.; El Khoury, G.; Erba, P.A.; Iung, B.; et al. 2015 ESC Guidelines for the Management of Infective Endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC)Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J 2015, 36, 3075–3128. [Google Scholar] [CrossRef]
- McDonald, E.G.; Aggrey, G.; Aslan, A.T.; Casias, M.; Cortes-Penfield, N.; Dong, M.Q.; Egbert, S.; Footer, B.; Isler, B.; King, M.; et al. Guidelines for Diagnosis and Management of Infective Endocarditis in Adults: A WikiGuidelines Group Consensus Statement. JAMA Netw Open 2023, 6, e2326366. [Google Scholar] [CrossRef]
- Pericàs, J.M.; Ambrosioni, J.; Muñoz, P.; De Alarcón, A.; Kestler, M.; Mari-Hualde, A.; Moreno, A.; Goenaga, M.Á; Fariñas, M.C.; Rodríguez-Álvarez, R.; et al. Prevalence of Colorectal Neoplasms Among Patients With Enterococcus Faecalis Endocarditis in the GAMES Cohort (2008–2017). Mayo Clinic Proceedings 2021, 96, 132–146. [Google Scholar] [CrossRef]
- Cimmino, G.; Bottino, R.; Formisano, T.; Orlandi, M.; Molinari, D.; Sperlongano, S.; Castaldo, P.; D’Elia, S.; Carbone, A.; Palladino, A.; et al. Current Views on Infective Endocarditis: Changing Epidemiology, Improving Diagnostic Tools and Centering the Patient for Up-to-Date Management. Life 2023, 13, 377. [Google Scholar] [CrossRef]
- Ha, K.E.; Kim, W.D.; Ko, K.-Y.; Kim, H.; Gwak, S.-Y.; Kim, K.; Lee, H.-J.; Shim, C.Y.; Hong, G.-R.; Ha, J.-W.; et al. Trends in Epidemiology, Clinical Characteristics, and Outcomes of Infective Endocarditis: A 16-Year Nationwide Cohort Study in Korea. J. Korean Med. Sci. 2025, 40, e183. [Google Scholar] [CrossRef] [PubMed]
- Peijster, A.J.L.; Heinen, F.J.; Douiyeb, S.; Van Der Stoel, M.D.; Selder, J.L.; Watson-de Lepper, A.G.W.; Houthuizen, P.; Meijs, M.F.L.; Kampschreur, L.M.; Schalla, S.; et al. ENDOCOR: A Nationwide Consortium of Endocarditis Teams—Initiating a Registry for Infective Endocarditis within the Netherlands Heart Registration. Neth Heart J 2025, 33, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Iversen, K.; Ihlemann, N.; Gill, S.U.; Madsen, T.; Elming, H.; Jensen, K.T.; Bruun, N.E.; Høfsten, D.E.; Fursted, K.; Christensen, J.J.; et al. Partial Oral versus Intravenous Antibiotic Treatment of Endocarditis. N Engl. J. Med. 2019, 380, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Freling, S.; Wald-Dickler, N.; Banerjee, J.; Canamar, C.P.; Tangpraphaphorn, S.; Bruce, D.; Davar, K.; Dominguez, F.; Norwitz, D.; Krishnamurthi, G.; et al. Real-World Application of Oral Therapy for Infective Endocarditis: A Multicenter, Retrospective, Cohort Study. Clin. Infect. Dis. 2023, 77, 672–679. [Google Scholar] [CrossRef]
- Rallet, B.; Pouy, R.; Coutureau, C.; Blot, M.; Bani-Sadr, F.; Sixt, T.; Moutel, M.; Piroth, L.; Hentzien, M. Should We Extend the Use of Oral Antibiotics in Infective Endocarditis? The ENDO-ORAL Study. Clin. Infect. Dis. 2025, ciaf452. [Google Scholar] [CrossRef]
- Moss, R.; Munt, B. Injection Drug Use and Right Sided Endocarditis. Heart 2003, 89, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Scheggi, V.; Menale, S.; Tonietti, B.; Giovacchini, J.; Del Pace, S.; Zoppetti, N.; Alterini, B.; Stefàno, P.L.; Marchionni, N. Infective Endocarditis in Octogenarians. A Retrospective Study in a Single, High-Volume Surgical Centre. BMC Geriatr 2023, 23, 659. [Google Scholar] [CrossRef] [PubMed]
- Loghin, I.I.; Surdu, A.E.; Rusu, Șerban A.; Cecan, I.; Dorobăț, V.D.; Mihăescu, A.A.; Dorobăţ, C.M. Etiological Aspects of Infectious Endocarditis in a Tertiary Hospital in Northeastern Romania. Medicina 2025, 61, 95. [Google Scholar] [CrossRef] [PubMed]
- “Grigore T. Popa” University of Medicine and Pharmacy Iasi, Romania; Buburuz, A.-M. Infective Endocarditis Etiologic Profile – Still a Classic Distribution of Microorganisms? MSJ 2022, 126, 25–32. [CrossRef]
- Nanu, A.-A.; Lazăr, D. Ștefan; Popescu, C.P.; Lazăr, M.-I.; Nica, M.; Florescu, S.A. Characteristics of Infective Endocarditis in Intravenous Drug Users vs. Non-Users: A Retrospective Study Conducted in Bucharest, Romania. Medicina 2025, 61, 1785. [Google Scholar] [CrossRef]
- Anton, C.-I.; Buzilă, C.A.; Stanciu, S.M.; Bucurică, S.; Anghel, D.; Ștefan, A.T.; Ștefan, I.; Streinu-Cercel, A. Prosthetic Valve Endocarditis: A Retrospective Cohort Study Conducted at “Dr. Carol Davila” Central Military Emergency University Hospital in Bucharest. Microorganisms 2024, 12, 1442. [Google Scholar] [CrossRef]
- Babeș, E.E.; Lucuța, D.A.; Petcheși, C.D.; Zaha, A.A.; Ilyes, C.; Jurca, A.D.; Vesa, C.M.; Zaha, D.C.; Babeș, V.V. Clinical Features and Outcome of Infective Endocarditis in a University Hospital in Romania. Medicina 2021, 57, 158. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for Reporting Observational Studies. The Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Li, J.S.; Sexton, D.J.; Mick, N.; Nettles, R.; Fowler, V.G.; Ryan, T.; Bashore, T.; Corey, G.R. Proposed Modifications to the Duke Criteria for the Diagnosis of Infective Endocarditis. Clin. Infect. Dis. 2000, 30, 633–638. [Google Scholar] [CrossRef]
- Waksman, R.; Pahuja, M.; Van Diepen, S.; Proudfoot, A.G.; Morrow, D.; Spitzer, E.; Nichol, G.; Weisfeldt, M.L.; Moscucci, M.; Lawler, P.R.; et al. Standardized Definitions for Cardiogenic Shock Research and Mechanical Circulatory Support Devices: Scientific Expert Panel From the Shock Academic Research Consortium (SHARC). Circulation 2023, 148, 1113–1126. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801. [Google Scholar] [CrossRef]
- Baddour, L.M.; Wilson, W.R.; Bayer, A.S.; Fowler, V.G.; Tleyjeh, I.M.; Rybak, M.J.; Barsic, B.; Lockhart, P.B.; Gewitz, M.H.; Levison, M.E.; et al. Infective Endocarditis in Adults: Diagnosis, Antimicrobial Therapy, and Management of Complications: A Scientific Statement for Healthcare Professionals From the American Heart Association. Circulation 2015, 132, 1435–1486. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023; Available online: https://www.R-project.org/ (accessed on 11 February 2026).
- Jordal, S.; Kommedal, Ø; Haaverstad, R.; Saeed, S.; Davidsen, E.S.; Salminen, P.-R.; Hufthammer, K.O.; Kittang, B.R. Epidemiological and Microbial Trends of Infective Endocarditis in Western Norway: A 7-Year Prospective Observational Study. BMC Infect Dis 2024, 24, 702. [Google Scholar] [CrossRef] [PubMed]
- Cresti, A.; Chiavarelli, M.; Scalese, M.; Nencioni, C.; Valentini, S.; Guerrini, F.; D’Aiello, I.; Picchi, A.; De Sensi, F.; Habib, G. Epidemiological and Mortality Trends in Infective Endocarditis, a 17-Year Population-Based Prospective Study. Cardiovasc. Diagn. Ther. 2017, 7, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Khardori, N. Changing Paradigms in Epidemiology, Microbiology, and Outcomes of Infective Endocarditis. Infect. Dis. Clin. Pract. 2024, 32, 1–3. [Google Scholar] [CrossRef]
- Camp, J.; Nüßle, K.; Deyhle, N.; Mathé, P.; Hornuss, D.; Grundmann, S.; Asmussen, A.; Zientara, A.; Bothe, W.; Schmidt-Hellerau, K.; et al. Partial Oral Treatment of Infective Endocarditis in Real-World Settings – an in-Depth Analysis of the Prospectively Evaluated German DERIVE Cohort. CMI Communications 2024, 1, 105032. [Google Scholar] [CrossRef]
- Pries-Heje, M.M.; Hjulmand, J.G.; Lenz, I.T.; Hasselbalch, R.B.; Povlsen, J.A.; Ihlemann, N.; Køber, N.; Tofterup, M.L.; Østergaard, L.; Dalsgaard, M.; et al. Clinical Implementation of Partial Oral Treatment in Infective Endocarditis: The Danish POETry Study. European Heart Journal 2023, 44, 5095–5106. [Google Scholar] [CrossRef]
- Habib, G.; Lancellotti, P.; Erba, P.-A.; Sadeghpour, A.; Meshaal, M.; Sambola, A.; Furnaz, S.; Citro, R.; Ternacle, J.; Donal, E.; et al. The ESC-EORP EURO-ENDO (European Infective Endocarditis) Registry. Eur. Heart J. - Qual. Care Clin. Outcomes 2019, 5, 202–207. [Google Scholar] [CrossRef]
- Dobreva-Yatseva, B.; Nikolov, F.; Raycheva, R.; Tokmakova, M. Infective Endocarditis—Predictors of In-Hospital Mortality, 17 Years, Single-Center Experience in Bulgaria. Microorganisms 2024, 12, 1919. [Google Scholar] [CrossRef]
- Philip, M.; Dumonceau, R.G.; Citro, R.; Cosyns, B.; Donal, E.; Erba, P.A.; Gouriet, F.; Iung, B.; Kong, W.K.F.; Lancellotti, P.; et al. Clinical Characteristics, Management and Outcomes of Enterococcal Infective Endocarditis: An Ancillary Study from the ESC-EORP EURO-ENDO Registry. Eur. Heart J. - Qual. Care Clin. Outcomes 2025, qcaf145. [Google Scholar] [CrossRef]
- Becher, P.M.; Goßling, A.; Fluschnik, N.; Schrage, B.; Seiffert, M.; Schofer, N.; et al. Temporal trends in incidence, patient characteristics, microbiology and in-hospital mortality in patients with infective endocarditis: a contemporary analysis of 86,469 cases between 2007 and 2019. Clin. Res. Cardiol. 2024, 113, 205–215. [Google Scholar] [CrossRef]
- Murdoch, D.R. Clinical Presentation, Etiology, and Outcome of Infective Endocarditis in the 21st Century: The International Collaboration on Endocarditis–Prospective Cohort Study. Arch Intern Med 2009, 169, 463. [Google Scholar] [CrossRef]
- Wang, A.; Gaca, J.G.; Chu, V.H. Management Considerations in Infective Endocarditis: A Review. JAMA 2018, 320, 72. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.-H.; Kim, Y.-J.; Kim, S.-H.; Sun, B.J.; Kim, D.-H.; Yun, S.-C.; Song, J.-M.; Choo, S.J.; Chung, C.-H.; Song, J.-K.; et al. Early Surgery versus Conventional Treatment for Infective Endocarditis. N Engl. J. Med. 2012, 366, 2466–2473. [Google Scholar] [CrossRef] [PubMed]
- Fowler, V.G.; Miro, J.M.; Hoen, B.; Cabell, C.H.; Abrutyn, E.; Rubinstein, E.; Corey, G.R.; Spelman, D.; Bradley, S.F.; Barsic, B.; et al. Staphylococcus Aureus Endocarditis: A Consequence of Medical Progress. JAMA 2005, 293, 3012. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Felix, B.M.; Barca, L.V.; Garcia-Esquinas, E.; Correa-Pérez, A.; Fernández-Hidalgo, N.; Muriel, A.; Lopez-Alcalde, J.; Álvarez-Diaz, N.; Pijoan, J.I.; Ribera, A.; et al. Prognostic Models for Mortality after Cardiac Surgery in Patients with Infective Endocarditis: A Systematic Review and Aggregation of Prediction Models. Clin. Microbiol. Infect. 2021, 27, 1422–1430. [Google Scholar] [CrossRef]
- Varela, L.; López-Menéndez, J.; Redondo, A.; Fajardo, E.R.; Miguelena, J.; Centella, T.; Martín, M.; Muñoz, R.; Navas, E.; Moya, J.L.; et al. Mortality Risk Prediction in Infective Endocarditis Surgery: Reliability Analysis of Specific Scores†. Eur. J. Cardio-Thorac. Surg. 2018, 53, 1049–1054. [Google Scholar] [CrossRef]
- Wang, Q.; Fu, B.; Hu, P.; Liao, X.; Guo, W.; Yu, D.; Wang, Z.; Wei, X. Clinical Evaluation of Sepsis-1 and Sepsis-3 in Infective Endocarditis. Int. J. Cardiol. 2023, 393, 131365. [Google Scholar] [CrossRef]
| Variable | Overall (n=206) | Trial-eligible (n=130) | Real World (n=76) | p-value (95% CI) |
|---|---|---|---|---|
| Demographics | ||||
| Age, years (median) | 63.0 ± 14.8 | 59.9 ± 15.4 | 68.3 ± 12.1 | < 0.001 (-12.2 to -4.6) |
| Male sex, n (%) | 145 (70.4%) | 102 (78.5%) | 43 (56.6%) | 0.002. (3.5% to 39.3%) |
| Comorbidities | ||||
| Hypertension, n (%) | 125 (60.7%) | 79 (60.8%) | 46 (60.5%) | 1.000 |
| Diabetes mellitus, n (%) | 37 (18.0%) | 24 (18.5%) | 13 (17.1%) | 0.955 |
| CKD (≥stage 3), n (%) | 47 (22.8%) | 21 (18.5%) | 26 (34.2%) | 0.01 (28.6% to 4.9%) |
| COPD, n (%) | 17 (8.3%) | 8 (6.2%) | 9 (11.8%) | 0.242 |
| IVDU, n (%) | 2 (1.0%) | 0 (0.0%) | 2 (2.6%) | 0.135 |
| Chronic HF, n (%) | 139 (67.5%) | 86 (66.2%) | 53 (69.7%) | 0.707 |
| Liver cirrhosis, n (%) | 5 (2.42%) | 2 (2.6%) | 3 (2.3%) | 1.000 |
| Malignancy, n (%) | 33 (16.0%) | 0 (0.0%) | 33 (43.4%) | 0.0001 (54.6% to 30.0%) |
| Infection location | ||||
| Left-sided IE, n (%) | 197 (95.6%) | 125 (96.2%) | 72 (94.7%) | 0.728 |
| Right-sided IE, n (%) | 9 (4.4%) | 5 (3.8%) | 4 (5.3%) | 0.718 |
| Prosthetic IE, n (%) | 81 (%) | 54 (41.5%) | 27 (35.5%) | 0.394 |
| CIED-related IE, n (%) | 32 (15.5%) | 12 (9.2%) | 20 (26.3%) | 0.001 (2.3 to 31.8) |
| TAVI-associated IE | 8 (3.8%) | 0 (0.0%) | 8 (10.5%) | <0.001 (2.6 to 19.4) |
| Microbiology | ||||
| S. aureus, n (%) | 29 (14.1%) | 22 (16.9%) | 7 (9.2%) | 0.184 |
| Enterococcus spp., n (%) | 26 (12.6%) | 10 (7.7%) | 16 (21.1%) | 0.010 (27.3% to 0.2%) |
| Viridans streptococci, n (%) | 20 (9.7%) | 18 (13.8%) | 2 (2.6%) | 0.009 (-20.1 to 0.2) |
| Culture-negative IE, n (%) | 74 (35.9%) | 45 (34.6%) | 29 (38.1%) | 0.480 |
| Echocardiography | ||||
| Vegetation present, n (%) | 150 (72.8%) | 93 (71.5%) | 57 (75.0%) | 0.590 |
| Vegetation >10 mm, n (%) | 95 (46.1%) | 60 (46.2%) | 35 (46.1%) | 0.989 |
| Perivalvular abscess, n (%) | 46 (22.3%) | 36 (27.7%) | 10 (13.2%) | 0.02 (1.8% to 28.6%) |
| Severe valvular regurgitation, n (%) | 79 (38.3) | 53 (40.8%) | 26 (34.2%) | 0.350 |
| LV EF <50%, n (%) | 93 (45.1%) | 61 (46.9%) | 32 (42.1%) | 0.599 |
| Clinical presentation / severity | ||||
| Acute HF on admission, n (%) | 25 (12.1%) | 12 (9.2%) | 13 (17.1%) | 0.147 |
| Sepsis at admission, n (%) | 52 (25.2%) | 23 (17.7%) | 29 (38.2%) | 0.002 (-37.3% to -2.9%) |
| Septic shock, n (%) | 17 (8.3%) | 2 (1.5%) | 15 (19.7%) | 0.0001 (−29.6% to −6.9%) |
| Cardiogenic shock, n (%) | 32 (15.5%) | 15 (11.5%) | 17 (22.4%) | 0.061 (25.8% to 3.7%) |
| Outcomes | ||||
| Composite endpoint, n (%)* | 126 (61.6%) | 75 (57.6%) | 51 (67.1%) | 0.218 |
| In-hospital mortality, n (%) | 67 (32.5%) | 37 (28.5%) | 30 (39.5%) | 0.141 |
| Renal replacement therapy (acute) | 8 (3.8%) | 6 (4.6%) | 2 (2.6%) | 0.713 |
| Any embolic event, n (%) ** | 54 (26.2%) | 27 (20.8%) | 27 (35.5%) | 0.02 (-2.8 to 32.1) |
| Length of stay, days | 13,05 ± 10,6 | 12.6 ± 11.1 | 13.5 ± 10.2 | 0.331 |
| Section / Variable | P1 (2011–2016) |
P2 (2017–2025) |
p-value 95% CI |
|---|---|---|---|
| Cohort composition | 49 | 157 | |
| Trial-eligible (TE) | 40 (81.6%) | 90 (57.3%) | 0.002 (-40.5 to -3.8) |
| Real-world (RW) | 9 (18.4%) | 67 (42.7%) | 0.002 (3.8 to 40.5) |
| Demographics | |||
| Age, years (mean) | 62.0 ± 13.3 | 63.4 ± 15.3 | 0.563 |
| Male sex, n % | 39 (79.6%) | 106 (67.5%) | 0.151 |
| Comorbidities | |||
| Hypertension | 34 (69.4%) | 91 (58.0%) | 0.153 |
| Diabetes mellitus | 7 (14.3%) | 30 (19.1%) | 0.443 |
| Coronary artery disease | 11 (22.4%) | 43 (27.4%) | 0.492 |
| Chronic HF, n % | 43 (87.8%) | 96 (61.1%) | <0.001 (-38.5 to -14.7) |
| Atrial fibrillation | 14 (28.6%) | 59 (37.6%) | 0.250 |
| CKD (≥stage 3), n (%) | 8 (16.3%) | 39 (24.8%) | 0.01 (12.6% to 28.9%) |
| Liver cirrhosis, n (%) | 1(2.1%) | 4(2.5%) | 0.983 |
| Malignancy, n % | 4 (8.2%) | 29 (18.5%) | 0.086 (-6.0 to 22.0) |
| IVDU | 0 (0.0%) | 2 (1.2) | 0.275 |
| Infection location | |||
| Prosthetic valve IE, n % | 21 (42.8%) | 60 (38.2) | 0.384 |
| CIED-related IE, n % | 1 (2.0%) | 31 (19.7%) | 0.003 (3.6 to 26.3) |
| TAVI-associated IE, n % | 0 (0.0%) | 8 (5.1%) | 0.202 |
| Microbiology | |||
| S. aureus, n (%) | 6 (12.2%) | 23 (14.6%) | 0.262 |
| Enterococcus spp., n (%) | 4 (8.1%) | 22 (14.01%) | 0.275 |
| Viridans streptococci, n (%) | 2 (4.08) | 18 (11.4%) | 0.126 |
| Culture-negative IE, n % | 32 (65.3%) | 42 (26.7%) | 0.001 (-43.1 to -12.4) |
| Echocardiography | |||
| TEE performed, n % | 9 (18.3%) | 81 (51.5%) | 0.01 (-1.1 to 38.9) |
| Vegetation >10 mm, n (%) | 42 (85.7%) | 53 (33.8%) | <0.001 (-66.1 to -31.9) |
| Perivalvular abscess, n % | 4/49 (8.2%) | 42 (26.8%) | 0.006 (8.3 to 28.9) |
| Severe regurgitation, n % | 25 (51.0%) | 54 (34.4%) | 0.05 (-32.5 to -0.8) |
| LVEF <50% | 30 (61.2%) | 63 (40.1%) | 0.010 (-40.8 to 0.7) |
| Clinical presentation / severity | |||
| Acute HF on admission, n (%) | 7 (14.2%) | 18 (11.4%) | 0.317. |
| Sepsis at admission, n (%) | 9 (18.3%) | 43 (27.3%) | 0.07 (15.8% to 37.7%) |
| Septic shock, n (%) | 2 (4.08%) | 15 (9.5%) | 0.02 (3.0 to 14.2) |
| Cardiogenic shock, n (%) | 3 (6.1%) | 29 (18.4%) | 0.01 (2.8% to 28.7%) |
| Outcomes | |||
| Composite endpoint, n (%)* | 21 (42.9%) | 105 (66.8%) | 0.002 (3.1 to 44.3) |
| In-hospital mortality, n % | 17 (34.7%) | 50 (31.8%) | 0.844 (-18.0 to 12.3) |
| Renal replacement therapy (acute), n % | 4 (8.2%) | 4 (2.5%) | 0.09 (-18.2 to 3.1) |
| Any embolic event, n % ** | 4 (8.2%) | 50 (31.8%) | <0.001 (5.9 to 36.3) |
| Length of stay, days | 8.2 ± 6.0 | 14.4 ± 11.5 | 0.002 (3.6 to 8.6) |
| Variable | Univariable OR | 95% CI | p-value |
|---|---|---|---|
| Sepsis at admission | 9.1 | 3.8–21.5 | <0.001 |
| Streptococcal infection | 5.8 | 1.4–24.0 | 0.014 |
| Cardiogenic shock | 3.9 | 1.8–8.4 | <0.001 |
| Acute heart failure | 2.7 | 1.4–5.0 | 0.002 |
| LVEF <50% | 2.6 | 1.3–5.2 | 0.006 |
| Perivalvular abscess | 2.2 | 1.0–4.9 | 0.048 |
| Staphylococcus aureus infection | 1.8 | 0.9–3.6 | 0.080 |
| Enterococcal infection | 1.4 | 0.7–2.9 | 0.320 |
| Age (per 10 years) | 1.02 | 0.99–1.04 | 0.160 |
| Male sex | 0.93 | 0.55–1.60 | 0.800 |
| Prosthetic/device-related IE | 0.70 | 0.40–1.20 | 0.190 |
| Predictor | Adjusted OR (95% CI) | p-value |
|---|---|---|
| Sepsis at admission | ≈8.4 (3.3–21.8) | <0.001 |
| LVEF <50% | ≈2.3 (1.1–5.0) | 0.032 |
| Streptococcal infection | ≈6.3 (1.3–30.6) | 0.024 |
| Prosthetic/device-related IE | ≈0.6 (0.4–1.1) | 0.080 |
| Age | ≈1.0 | NS |
| Sex | ≈1.0 | NS |
| Variable | P1 n (%) |
P2 n (%) |
OR (95% CI) P2 vs P1 |
p | TE n (%) |
RW n (%) |
OR (95% CI) TE vs RW |
p |
|---|---|---|---|---|---|---|---|---|
| Overall patients (206), n | 49 | 157 | - | - | 130 | 76 | - | - |
| QI score ≥ 3 | 9 (18.3%) | 40 (25.4%) |
1.63 (0.62–4.27) |
0.32 | 35 (26.9%) |
14 (18.4%) |
2.23 (0.96–5.19) |
0.05 |
| Adequate blood cultures, n % * |
17 (34.7%) | 115 (73.2%) | 5.14 (2.47–10.7) |
<0.001 | 81 (62.3%) | 51 (67.1%) |
0.80 (0.44–1.46) |
0.588 |
| TEE performed when indicated, n % | 9 (18.3%) | 81 (51.5%) |
4.72 (2.10–10.6) |
0.001 | 52 (40.0%) |
38 (50.0%) |
0.67 (0.37–1.21) |
0.18 |
| Appropriate antimicrobial therapy, n % ** | 16 (32.7%) | 116 (73.9%) |
5.86 (2.85–12.0) |
<0.001 | 81 (62.3%) |
51 (67.1%) |
0.80 (0.44–1.46) |
0.588 |
| Surgery when indicated n % |
6 (12.2%) | 48 (30.5%) |
3.18 (1.2–8.2) |
0.01 | 35 (26.9%) |
19 (25.0%) | 1.10 (0.56–2.15) |
0.890 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




