Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanism of Microglial Activation in Epileptogenesis
2.1. Pattern Recognition Receptors: Toll-Like Receptor 4 (TLR4)
2.2. Purinergic Signaling: ATP Release and P2X7 Receptors
2.3. Proinflammatory Cytokines: IL-1β, TNF-α, and IL-6
3. Microglia-Targeted Therapeutic Strategies
3.1. Anti-Inflammatory Pharmacotherapy
3.2. Microglial Modulation Therapy
3.3. Cannabinoid Therapy
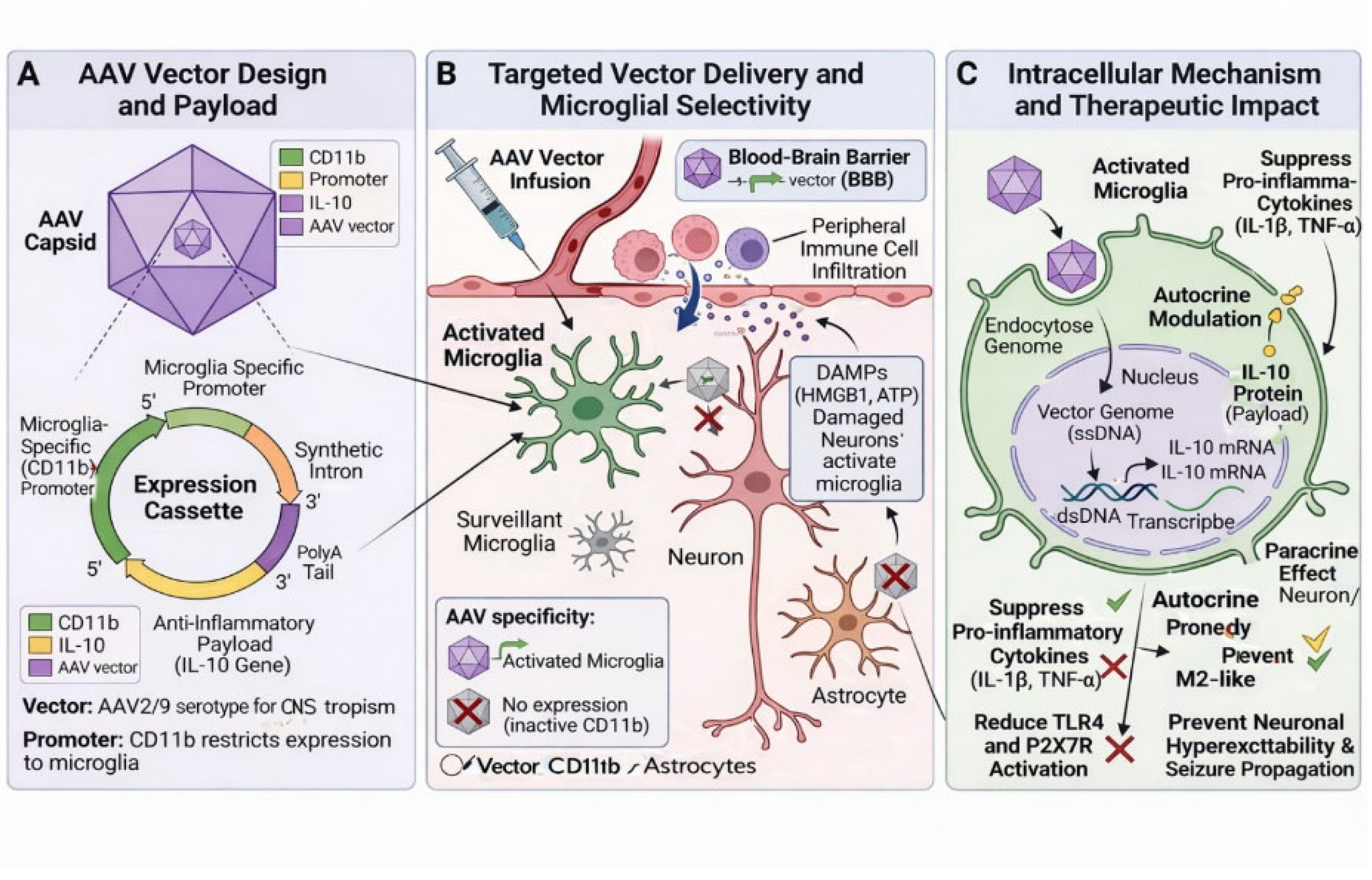
3.4. Gene Therapy
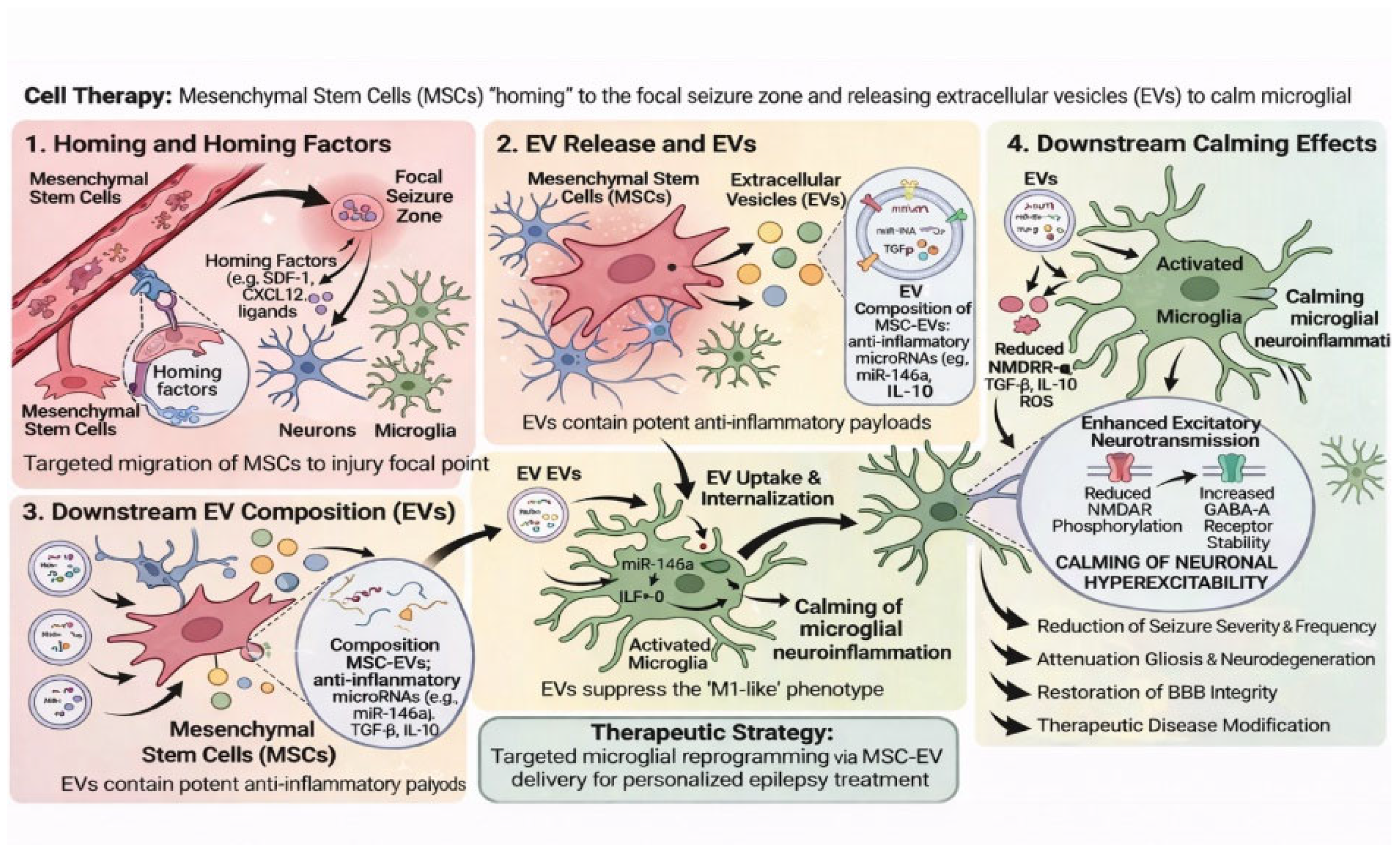
3.5. Stem Cell-Based Therapy
4. Therapeutic Agents Targeting Microglia
4.1. Anakinra
4.2. Minocycline
4.3. Cannabidiol (CBD)
4.4. Glucocorticoids
4.5. Celecoxib
4.6. JNJ-47965567 and Brilliant Blue G
4.7. Glycyrrhizin and Anti-HMGB1 Therapy
4.8. CSF1R Inhibitors: PLX3397 and PLX5622
5. Challenges and Limitations
6. Conclusion
References
- Feigin, Valery L; et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18(May), 459–80. [Google Scholar] [CrossRef]
- Beghi, Ettore; et al. Global, regional, and national burden of epilepsy, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18(April), 357–75. [Google Scholar] [CrossRef]
- Valery, L; Feigin, A. Articles Global, regional, and national burden of epilepsy, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Public Heal. 2025, 10(February), 203–27. [Google Scholar]
- Kwan, P; Arzimanoglou, A; Berg, AT; Brodie, MJ; Perucca, E; Wiebe, S; et al. Definition of drug resistant epilepsy: Consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 2010, 51(6), 1069–77. [Google Scholar] [CrossRef]
- KWAN, P; BRODIE, MJ. EARLY IDENTIFICATION OF REFRACTORY EPILEPSY. N Engl J Med. 2000, 342(5), 314–9. [Google Scholar] [CrossRef]
- Matus, JSM; Suárez, SO; Escalante, RG. Factors not considered in the study of drug- resistant epilepsy: Psychiatric comorbidities, age, and gender. Epilepsia Open. 2022, 7, 81–93. [Google Scholar]
- Rheims, S; Sperling, MR; Ryvlin, P. resistant epilepsy and mortality — Why and when do neuromodulation and epilepsy surgery reduce overall mortality. Epilepsia 2022, 63, 3020–36. [Google Scholar] [CrossRef]
- Khateb, M; Bosak, N; Herskovitz, M. The Effect of Anti-seizure Medications on the Propagation of Epileptic Activity: A Review. Front Neurol. 2021, 12(May), 1–12. [Google Scholar] [CrossRef]
- Perucca, P; Gilliam, FG. Adverse eff ects of antiepileptic drugs. Lancet Neurol. 2012, 11(September), 792–802. [Google Scholar] [CrossRef]
- Vezzani, A; French, J; Bartfai, T; Baram, TZ. The role of inflammation in epilepsy. Nat Publ Gr [Internet] 2010, 7(1), 31–40. [Google Scholar] [CrossRef]
- Salter, MW; Stevens, B. Microglia emerge as central players in brain disease. Nat Med [Internet] 2017, 23(9), 1018–27. [Google Scholar] [CrossRef]
- Eyo, UB; Murugan, M; L jun, Wu. Microglia—Neuron Communication in Epilepsy. Glia 2016, 00(00), 1–14. [Google Scholar]
- Devinsky, O; Vezzani, A; Najjar, S; De Lanerolle, NC; Rogawski, MA. Glia and epilepsy: excitability and inflammation. Trends Neurosci [Internet] 2013, 36(3), 174–84. [Google Scholar] [CrossRef] [PubMed]
- Ravizza, T; Terrone, G; Salamone, A; Frigerio, F; Balosso, S; Antoine, DJ; et al. High Mobility Group Box 1 is a novel pathogenic factor and a mechanistic biomarker for epilepsy. Brain Behav Immun [Internet] 2018, 72, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Maroso, M; Balosso, S; Ravizza, T; Liu, J; Aronica, E; Iyer, AM; et al. Toll-like receptor 4 and high-mobility group box-1 are involved in ictogenesis and can be targeted to reduce seizures. Nat Med [Internet] 2010, 16(4), 413–9. [Google Scholar] [CrossRef] [PubMed]
- White, HS; Galanopoulou, AS; Barker-haliski, ML; Wolfgang, L. Neuroinflammation in epileptogenesis: Insights and translational perspectives from new models of epilepsy. Epilepsia 2017, 58(3), 39–47. [Google Scholar] [CrossRef]
- Balosso, S; Liu, J; Bianchi, ME; Vezzani, A. Disulfide-Containing High Mobility Group Box-1 Promotes N -Methyl- d -Aspartate Receptor Function and Excitotoxicity by Activating Toll-Like Receptor 4-Dependent Signaling in Hippocampal Neurons. Antioxid Redox Signal. 2014, 21(12), 1726–40. [Google Scholar]
- Engel, T. Purinergic signaling-induced neuroinflammation and status epilepticus. Expert Rev Neurother [Internet] 2016, 16(7), 735–7. [Google Scholar] [CrossRef]
- Di, Virgilio F; Ben, DD; Sarti, AC; Giuliani, AL; Falzoni, S. Review The P2X7 Receptor in Infection and Inflammation. Immun Rev [Internet] 2017, 47(1), 15–31. [Google Scholar] [CrossRef]
- Di, Virgilio F; Sarti, AC; Coutinho-silva, R. Purinergic signaling, DAMPs, and inflammation. Am J Physiol Cell Physiol. 2026, 318(26), 832–5. [Google Scholar]
- Rijkers, K; Majoie, HJ; Hoogland, G; Kenis, G; Baets, M; De, *!!! REPLACE !!!*; Vles, JS. The role of interleukin-1 in seizures and epilepsy: A critical review. Exp Neurol [Internet] 2009, 216(2), 258–71. [Google Scholar] [CrossRef] [PubMed]
- Viviani, B; Bartesaghi, S; Gardoni, F; Vezzani, A; Behrens, MM; Bartfai, T; et al. Interleukin-1B Enhances NMDA Receptor-Mediated Intracellular Calcium Increase through Activation of the Src Family of Kinases. J Neurosci. 2003, 23(25), 8692–700. [Google Scholar] [CrossRef] [PubMed]
- Vezzani, A; Viviani, B. Neuropharmacology Neuromodulatory properties of inflammatory cytokines and their impact on neuronal excitability. Neuropharmacology [Internet] 2015, 96, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S; Arisi, GM; Mims, K; Hollingsworth, G; Neil, KO; Shapiro, LA. Neuroinflammatory mechanisms of post- traumatic epilepsy. J Neuroinflammation 2020, 17(193), 1–11. [Google Scholar] [CrossRef]
- Stellwagen, D; Malenka, RC. Synaptic scaling mediated by glial TNF- a. Nature 2006, 440, 1–6. [Google Scholar] [CrossRef]
- Erta, M; Quintana, A; Hidalgo, J. Interleukin-6, a Major Cytokine in the Central Nervous System. Int J Biol Sci. 2012, 8(9), 1254–66. [Google Scholar] [CrossRef]
- Bialer, M; Johannessen, SI; Levy, RH; Perucca, E; Tomson, T; White, HS. Progress report on new antiepileptic drugs: A summary of the Eleventh Eilat Conference (EILAT XI). Epilepsy Res [Internet] 2013, 103(1), 2–30. [Google Scholar] [CrossRef]
- Aronica, E; Bauer, S; Bozzi, Y; Caleo, M; Dingledine, R; Louboutin, J; pierre; et al. Neuroinflammatory targets and treatments for epilepsy validated in experimental models. Epilepsia 2017, 58(3), 27–38. [Google Scholar] [CrossRef]
- Tikka, T; Keina, R. A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window. PNAS 1999, 96(23), 13496–13500. [Google Scholar]
- Zarzuelo, A; Gálvez, J. What is behind the non-antibiotic properties of minocycline ? Pharmacol Res [Internet] 2013, 67(1), 18–30. [Google Scholar] [CrossRef]
- Marchi, N; Granata, T; Janigro, D. Inflammatory pathways of seizure disorders. Trends Neurosci [Internet] 2014, 37(2), 55–65. [Google Scholar] [CrossRef] [PubMed]
- Rojas, A; Chen, D; Ganesh, T; Varvel, NH; Dingledine, R. The COX-2 / prostanoid signaling cascades in seizure disorders. Expert Opin Ther Targets 2020, 23(1), 1–13. [Google Scholar]
- Bien, CG; Holtkamp, M. Autoimmune Epilepsy “: Encephalitis With Autoantibodies for Epileptologists. Epilepsy Curr. 2017, 17(3), 134–41. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, A; Minghetti, L. Regulation of Glial Cell Functions by PPAR- γ Natural and Synthetic Agonists. PPAR Res. 2008, 2008, 1–10. [Google Scholar] [CrossRef]
- Elmore, MRP; Najafi, AR; Koike, MA; Nazih, N; Spangenberg, EE; Rice, RA; et al. CSF1 receptor signaling is necessary for microglia viability, which unmasks a cell that rapidly repopulates the microglia- depleted adult brain. Neuron 2015, 82(2), 380–97. [Google Scholar] [CrossRef]
- Devinsky, O; Cilio, MR; Cross, H; Fernandez-Ruiz, J; French, J; Hill, C; et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 2016, 55(6), 791–802. [Google Scholar] [CrossRef]
- Gaston, TE; Friedman, D. Pharmacology of cannabinoids in the treatment of epilepsy. Epilepsy Behav [Internet] 2017, 70, 313–8. [Google Scholar] [CrossRef]
- Löscher, W; Potschka, H; Sisodiya, SM; Vezzani, A. Drug Resistance in Epilepsy: Clinical Impact, Potential Mechanisms, and New Innovative Treatment Options. Pharmacol Rev [Internet] 2020, 72(3), 606–38. [Google Scholar] [CrossRef]
- Snowball, A; Chabrol, E; Wykes, RC; Shekh-ahmad, T; Cornford, JH; Lieb, A; et al. Epilepsy Gene Therapy Using an Engineered Potassium Channel. J Neurosci. 2019, 39(16), 3159–69. [Google Scholar]
- Kazantsev, AG; Thompson, LM. Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nat Rev. 2008, 7, 854–68. [Google Scholar]
- Hattiangady, B; Kuruba, R; Shuai, B; Grier, R; Shetty, AK. Hippocampal Neural Stem Cell Grafting after Status Epilepticus Alleviates Chronic Epilepsy and Abnormal Plasticity, and Maintains Better Memory and Mood Function. Aging Dis. 2020, 11(6), 1374–94. [Google Scholar] [CrossRef]
- Uccelli, A; Moretta, L; Pistoia, V. Mesenchymal stem cells in health and disease. Nat Rev. 2008, 8(September), 726–36. [Google Scholar] [CrossRef]
- Takahashi, K; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell. 2006, 2(126), 663–76. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, CA; Simon, A; Meer, JWM; Van, Der. Treating inflammation by blocking interleukin—1 in a broad spectrum of diseases. Nat Rev [Internet] 2012, 11(August), 633–52. [Google Scholar] [CrossRef] [PubMed]
- Kenney-Jung, DL; Kahoud, RJ; Vezzani, A; Lafrance-corey, RG; Ho M lan; Muskardin, TW. Super-refractory status epilepticus and febrile infection-related epilepsy syndrome treated with anakinra. Ann Neurol. 2017, 80(6), 939–45. [Google Scholar] [CrossRef]
- Plane, JM; Shen, Y; Pleasure, DE; Deng, W. Prospects for Minocycline Neuroprotection. Arch Neurol. 2015, 67(12), 1442–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, N; Mi, X; Gao, B; Gu, J; Wang, W; Zhang, Y; et al. MINOCYCLINE INHIBITS BRAIN INFLAMMATION AND ATTENUATES SPONTANEOUS RECURRENT SEIZURES FOLLOWING PILOCARPINE-INDUCED STATUS EPILEPTICUS. Neuroscience 2015, 287, 144–56. [Google Scholar] [CrossRef]
- Abraham, J; Foxa, PD; Condello, C; Bartolinia, A; Koh, S. Minocycline attenuates microglia activation and blocks the long- term epileptogenic effects of early-life seizures. Neurobiol Dis. 2013, 94(3), 242–57. [Google Scholar]
- Devinsky, O; Cross, JH; Laux, L; Marsh, E; Miller, I; Nabbout, R; et al. Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome. N Engl J Med. 2017, 376(21), 2011–20. [Google Scholar] [CrossRef]
- Thiele, EA; Marsh, ED; French, JA; Mazurkiewicz-beldzinska, M; Benbadis, SR; Joshi, C; et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome ( GWPCARE4 ): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018, 391, 1085–96. [Google Scholar] [CrossRef]
- Rhen, T; Cidlowski, JA. Antiinflammatory Action of Glucocorticoids — New Mechanisms for Old Drugs. N Engl J Med. 2005, 353(16), 1711–23. [Google Scholar] [CrossRef] [PubMed]
- Barnes, PJ. Anti-inflammatory actions of glucocorticoids: molecular mechanisms. Clin Sci. 1998, 572, 557–72. [Google Scholar] [CrossRef] [PubMed]
- Abbott, NJ; Patabendige, AAK; Dolman, DEM; Yusof, SR; Begley, DJ. Structure and function of the blood—brain barrier. Neurobiol Dis [Internet] 2010, 37(1), 13–25. [Google Scholar] [CrossRef] [PubMed]
- Vezzani, A; Friedman, A; Dingledine, RJ. The role of in fl ammation in epileptogenesis. Neuropharmacology 2013, 69, 16–24. [Google Scholar] [CrossRef]
- Rojas, A; Jiang, J; Ganesh, T; Yang, M; soon; Lelutiu, N. Cyclooxygenase-2 in epilepsy. Epilepsia 2014, 55(1), 17–25. [Google Scholar] [CrossRef]
- Alsaegh, H; Eweis, H; Kamal, F; Alrafiah, A. Celecoxib Decrease Seizures Susceptibility in a Rat Model of Inflammation by Inhibiting HMGB1 Translocation. Pharmaceuticals [Internet] 2021, 14(4), 1–19. [Google Scholar] [CrossRef]
- Jimenez-pacheco, A; Mesuret, G; Sanz-rodriguez, A; Tanaka, K; Mooney, C; Conroy, R; et al. Increased neocortical expression of the P2X7 receptor after status epilepticus and anticonvulsant effect of P2X7 receptor antagonist A-438079. Epilepsia [Internet] 2013, 54(9), 1551–61. [Google Scholar] [CrossRef]
- Sperlagh, B; Illes, P. P2X7 receptor: an emerging target in central nervous system diseases. Trends Pharmacol Sci. 2014, 35(10), 537–47. [Google Scholar] [CrossRef]
- Mollica, L; Marchis, F; De; Spitaleri, A; Dallacosta, C; Pennacchini, D; Zamai, M; et al. Glycyrrhizin Binds to High-Mobility Group Box 1 Protein and Inhibits Its Cytokine Activities. Chem Biol. 2007, 14(April), 431–41. [Google Scholar] [CrossRef]
- Iori, V; Maroso, M; Rizzi, M; Iyer, AM; Vertemara, R; Carli, M; et al. Receptor for Advanced Glycation Endproducts is upregulated in temporal lobe epilepsy and contributes to experimental seizures. Neurobiol Dis [Internet] 2013, 58, 102–14. [Google Scholar] [CrossRef]
- Vezzani, A; Balosso, S; Ravizza, T. Neuroinflammatory pathways as treatment targets and biomarkers in epilepsy Annamaria. Nat Rev Neurol [Internet]. 2019, pp. 1–14. [CrossRef]
- Srivastava, PK; Van, Eyll J; Godard, P; Mazzuferi, M; Delahaye-duriez, A; Van, Steenwinckel J; et al. A systems-level framework for drug discovery identifies Csf1R as an anti-epileptic drug target Prashant. Nat Commun [Internet]. 2018, pp. 1–15. [CrossRef]
- Paolicelli, RC; Paolicelli, RC; Bolasco, G; Pagani, F; Maggi, L; Scianni, M; et al. Synaptic Pruning by Microglia Is Necessary for Normal Brain Development. Science (80- ) 2011, 333, 1456–8. [Google Scholar] [CrossRef]
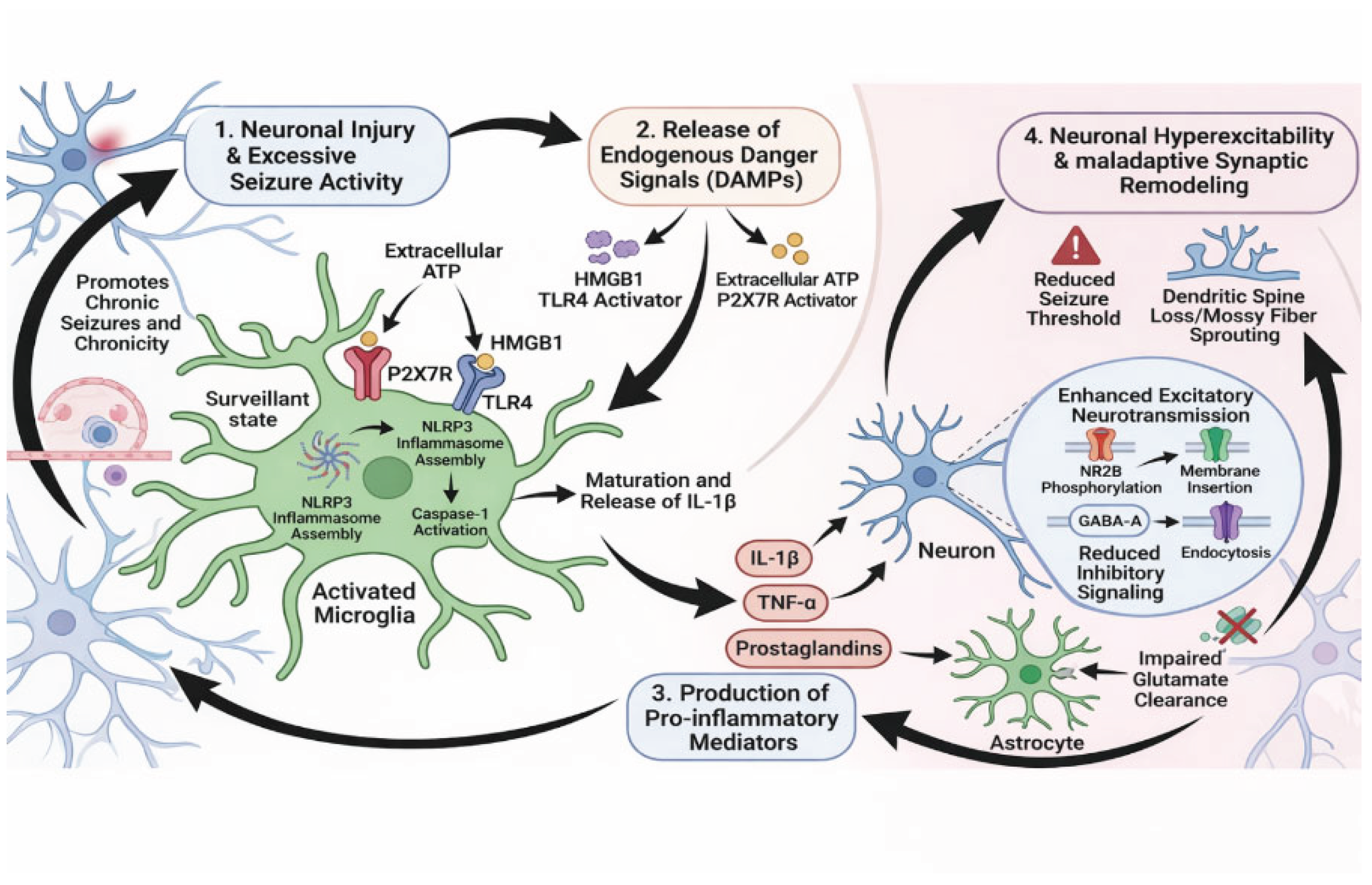
| Pathway | Primary Mediators | Effects on Neuronal Network | Impact on Drug Resistance |
|
Pattern Recognition |
HMGB1, TLR4 | Enhances phosphorylation of NMDA receptors; increases calcium channel permeability; disrupts BBB integrity. | Upregulates P-glycoprotein (Pgp) efflux transporters that pumps AEDs out of brain. |
| Purinergic Signaling | ATP, P2X7 Receptor | Triggers NLRP3 inflammasome; releases glutamate. | Sustains a pro-inflammatory microenvironment. |
| Cytokine Signaling | IL-1β, TNF-α, IL-6 | Enhances NMDA current; internalizes GABA-A receptors. | Alters drug target sensitivity; promotes chronic gliosis. |
| Strategy | Example Agents | Mechanism of Action | Clinical Status |
|
Anti-Inflammatory Pharmacotherapy: Cytokine Inhibition Purinergic Antagonism Caspase-1 inhibition HMGB1/TLR4 Axis inhibition Anti-inflammatory antibiotics Broad-spectrum anti-inflammatory agents Immunomodulatory therapies |
Anakinra P2X7R Antagonists like BBG VX-765 Glycyrrhizin (HMGB1 inhibition), TAK-242 (TLR4 inhibition). Minocycline Glucocorticoids Immunoglobulins |
Blocks IL-1R1 signaling Blocks NLRP3 inflammasome assembly Prevents the maturation of IL-1β Prevents microglial activation Modulate neuroinflammation Direct anti-inflammatory effects Attenuates pathogenic immune response |
Clinical use in FIRES/NORSE. Preclinical (High potential). Preclinical (High potential). Preclinical (High potential). Preclinical (High potential). Clinical use in Epileptic spasm/Autoimmune epilepsies. Clinical use in FIRES |
| Microglial Modulation | PPAR-γ agonists; CSF1R inhibitors like PLX3397 | Shifts M1 to M2 phenotype; Microglial depletion. | Preclinical; Pilot human studies. |
| Cannabinoid Therapy | Cannabidiol (CBD) | CB2 engagement; reduces intracellular calcium | FDA-approved for specific syndromes like Dravet. |
| Regenerative Medicine | MSCs, iPSCs | Paracrine release of IL-10 and TGF-β. | Early-phase clinical trials. |
| Gene therapy | Viral vectors | Promotes a neuroprotective microenvironment | Early-phase clinical trials. |
| Agent | Molecular Target | Primary Mechanism | Effect on Epileptogenesis | Primary Benefit | Key Limitation |
| Anakinra | IL-1R1 | Blocks IL-1β binding; stops NF-κB/MAPK activation. | Prevents IL-1β induced synaptic changes (NMDAR/ GABA-A). | Potent in acute inflammatory crises (e.g., FIRES). | Short half-life; risk of systemic infections. |
| Minocycline | Multi (iNOS, NF-κB) | Suppresses microglial M1 polarization and ROS. | Attenuates hippocampal neuronal loss and mossy fiber sprouting. | Crosses BBB easily; well-established safety profile. | Primarily effective when given early; limited human data. |
| Cannabidiol | CB2, TRPV1, NLRP3 | Modulates calcium and inhibits adenosine reuptake. |
Pushes microglia toward neuroprotective M2 phenotype. | Dual action: Anticonvulsant + Anti-inflammatory. | Complex drug-drug interactions (CYP450). |
| Glucocorticoids | GC Receptor | Broad genomic/non-genomic immune suppression. | Restores BBB integrity; reduces chronic gliosis. | High efficacy in autoimmune/infantile syndromes. | Serious long-term side effects (metabolic/bone). |
| Celecoxib | COX-2 | Inhibits Prostaglandin E2 (PGE2) synthesis. | Reduces prostaglandin-mediated hyperexcitability. | Synergistic effect when used as an adjunct. | Limited clinical evidence for primary epilepsy control. |
| JNJ-47965567 | P2X7 Receptor | Prevents ATP-driven NLRP3 inflammasome assembly. | Long-lasting reduction in spontaneous seizure frequency. | Targets a very upstream “sensor” of seizure stress. | Mostly preclinical; potential for off-target CNS effects. |
| Glycyrrhizin | HMGB1 | Binds HMGB1 to prevent TLR4 receptor interaction. | Blocks the initial trigger of the inflammatory cascade. | Low toxicity; targets a validated human biomarker. | Potential mineralocorticoid-like side effects (hypertension). |
| PLX5622 | CSF1R | Depletes microglial population by blocking survival signals. | Disrupts the feedback loop between glia and neurons. | Powerful research tool for total inflammatory “reset.” | Complete depletion may impair cognitive/homeostatic repair. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




