Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
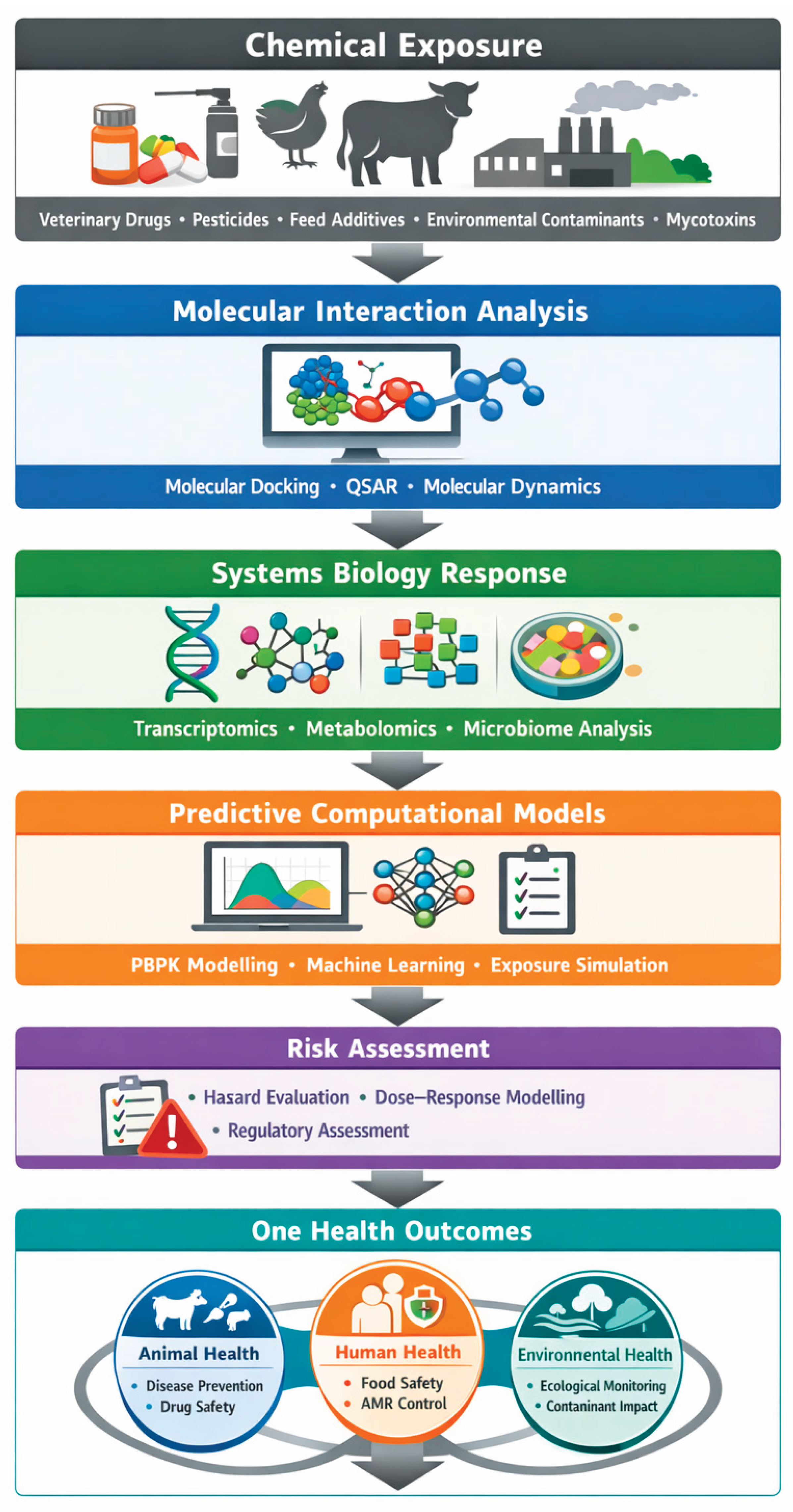
1. Introduction
2. Computational Strategies in Veterinary Biochemistry and Toxicology
2.1. Vetinformatics and Systems Integration
2.2. Predictive and Computational Toxicology
2.3. Computational Pharmacology and Drug–Toxin Interaction Modelling
2.4. Data-Driven Toxicogenomics
2.5. Environmental and Food Chain Modelling
3. Translational Applications within the One Health Framework
4. Computational Toxicology and Food Contaminant Assessment
5. Conclusion
References
- Lammie, S. L.; Hughes, J. M. Antimicrobial Resistance, Food Safety, and One Health: The Need for Convergence. Annual Review of Food Science and Technology 2016, 7, 287–312. [Google Scholar] [CrossRef]
- Bagaria, A. AMR-MoEGA: Antimicrobial Resistance Prediction using Mixture of Experts and Genetic Algorithms. 2025. Available online: http://arxiv.org/abs/2511.12223.
- Rhouma, M.; Archambault, M.; Butaye, P. Antimicrobial Use and Resistance in Animals from a One Health Perspective. Veterinary Sciences 2023, 10(5), 10–12. [Google Scholar] [CrossRef]
- Scarpa, F.; Casu, M. Genomics and Bioinformatics in One Health: Transdisciplinary Approaches for Health Promotion and Disease Prevention. International Journal of Environmental Research and Public Health 2024, 21(10). [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.; Begum, F.; Alfaifi, M. Y.; Elbehairi, S. E. I.; Siddique, A.; Shaw, P. Exploring Antimicrobial Potency, ADMET, and Optimal Drug Target of a Non-ribosomal Peptide Sevadicin from Bacillus pumilus, through In Vitro Assay and Molecular Dynamics Simulation. Probiotics and Antimicrobial Proteins 2025, 17(6), 4237–4253. [Google Scholar] [CrossRef] [PubMed]
- Kim, J. M.; Pathak, R. K. Editorial: Vetinformatics: an insight for decoding livestock systems through in silico biology. Frontiers in Veterinary Science 2023, 10(11), 4–6. [Google Scholar] [CrossRef] [PubMed]
- Vlasiou, M. C. Vet informatics and the future of drug discovery in veterinary medicine. Frontiers in Veterinary Science 2024, 11(4). [Google Scholar] [CrossRef]
- Anahtar, M. N.; Yang, J. H.; Kanjilal, S. Applications of Machine Learning to the Problem of Antimicrobial Resistance: an Emerging Model for Translational Research. Journal of Clinical Microbiology 2021, 59(7). [Google Scholar] [CrossRef]
- Su, M.; Satola, S. W.; Read, T. D. Genome-based prediction of bacterial antibiotic resistance. Journal of Clinical Microbiology 2019, 57(3). [Google Scholar] [CrossRef]
- Raies, A. B.; Bajic, V. B. In silico toxicology: computational methods for the prediction of chemical toxicity. Wiley Interdisciplinary Reviews: Computational Molecular Science 2016, 6(April), 147–172. [Google Scholar] [CrossRef]
- de Sousa, D. S.; Gomes, A. O. C. V.; Roberto, C. H. A.; Belarmino, A. B.; da Silva Mendes, F. R.; Marinho, M. M.; de Lima-Neto, P.; Marinho, G. S. Ligand and structure-based toxicological assessment of (thio)semicarbazones on cholinesterases. Journal of Computer-Aided Molecular Design 2026, 40(1). [Google Scholar] [CrossRef]
- Toropova, A. P.; Toropov, A. A.; Colombo, E.; Viganò, E. L.; Lombardo, A.; Roncaglioni, A.; Benfenati, E. Simulation of Fish Acute Toxicity of Pharmaceuticals Using Simplified Molecular Input Line Entry System (SMILES) Notation as a Representation of Molecular Structure. International Journal of Molecular Sciences 2025, 26(19). [Google Scholar] [CrossRef]
- Thomas, R. S.; Bahadori, T.; Buckley, T. J.; Cowden, J.; Deisenroth, C.; Dionisio, K. L.; Frithsen, J. B.; Grulke, C. M.; Gwinn, M. R.; Harrill, J. A.; Higuchi, M.; Houck, K. A.; Hughes, M. F.; Sidney Hunter, E.; Isaacs, K. K.; Judson, R. S.; Knudsen, T. B.; Lambert, J. C.; Linnenbrink, M.; Williams, A. J. The next generation blueprint of computational toxicology at the U.S. Environmental protection agency. Toxicological Sciences 2019, 169(2), 317–332. [Google Scholar] [CrossRef] [PubMed]
- Rusyn, I.; Greene, N. The impact of novel assessment methodologies in toxicology on green chemistry and chemical alternatives. Toxicological Sciences 2018, 161(2), 276–284. [Google Scholar] [CrossRef] [PubMed]
- Omiecinski, C. J.; Vanden Heuvel, J. P.; Perdew, G. H.; Peters, J. M. Xenobiotic metabolism, disposition, and regulation by receptors: From biochemical phenomenon to predictors of major toxicities. Toxicological Sciences 2011, 120. [Google Scholar] [CrossRef]
- Su, Z.; Zhang, L.; Chen, K.; Zhi, L.; Huan, X.; Chen, F.; Wang, X.; Wang, J. Artificial intelligence-driven environmental toxicology: Emerging strategies for microplastics risk assessment. Gondwana Research 2026, 153, 170–183. [Google Scholar] [CrossRef]
- Parekh, V. A.; Amina, M.; Islam, M. L.; Patil, P. C.; Ali, M. A.; Wabaidur, S. M.; Islam, M. A. Identification of phosphodiesterase 10 A modulators for neurodegenerative and psychiatric disorders: Combination of physics-based virtual screening and machine learning approaches. Computational Biology and Chemistry 2026, 121, 108875. [Google Scholar] [CrossRef]
- Roveri, V.; Correia, A. T.; dos Santos, P. G.; Ferreira Povoas, M. N.; Toma, W.; Seabra Pereira, C. D.; Guimarães, L. L. AI-assisted QSAR framework for ecological risk assessment of pharmaceuticals: integrating experimental, mechanistic, and deep learning evidence. Computational Toxicology 2026, 37. [Google Scholar] [CrossRef]
- Fahim, A. M. Structure-based drug design; Computational strategies in drug discovery; Antihypertensive agents; Antiviral drugs; Molecular docking; QSAR; Pharmacological insights. Computational Biology and Chemistry 2026, 120(P1), 108663. [Google Scholar] [CrossRef]
- Tompkins, L. A. R.; Khoei, A.; Kapeljushnik, O.; Dumond, J.; Kashuba, A. D. M.; Tropsha, A.; Hubal, R.; Cottrell, M. L. HIV Pharmacology Data Repository: Setting the New Information-Sharing Standard for Clinical and Preclinical Pharmacokinetic Studies. Clinical Pharmacology and Therapeutics 2025, 118(6), 1340–1349. [Google Scholar] [CrossRef]
- Chen, J.; Lin, A.; Jiang, A.; Qi, C.; Liu, Z.; Cheng, Q.; Yuan, S.; Luo, P. Computational frameworks transform antagonism to synergy in optimizing combination therapies. Npj Digital Medicine 2025, 8(1). [Google Scholar] [CrossRef]
- Wang, L.; Yu, X. Research hotspots and evolution trends of food safety risk assessment techniques and methods. EFood 2024, 5(6), 1–13. [Google Scholar] [CrossRef]
- Wang, J.; Li, J. The digital evolution in toxicology: pioneering computational education for emerging challenges. BMC Medical Education 2024, 24(1). [Google Scholar] [CrossRef]
- Deng, X.; Liu, Y.; Zhu, X.; Zhu, H.; Huang, C.; Hong, Y.; Wu, D.; Han, Y. Momordicae Semen: A Review of Phytochemistry, Pharmacology, Toxicology, Herbal Processing, Clinical Applications, and Q-Markers Prediction. Drug Design, Development and Therapy 2025, 19(November), 10303–10341. [Google Scholar] [CrossRef] [PubMed]
- Salian-Mehta, S.; Poling, J.; Birkebak, J.; Rensing, S.; Carosino, C.; Santos, R.; West, W.; Adams, K.; Orsted, K.; Fillman-Holliday, D.; Burns, M. Non-Human Primate Husbandry and Impact on Non-Human Primates Health: Outcomes From an IQ DruSafe/3RS Industrial Benchmark Survey. International Journal of Toxicology 2023, 42(2), 111–121. [Google Scholar] [CrossRef]
- Gao, H.; Chen, M. Dynamics of integrated multi-trophic aquaculture ( IMTA): A modeling approach incorporating zooplankton and scavenger. 620 2026. [Google Scholar] [CrossRef] [PubMed]
- Sachan, R. S. K.; Kauts, S.; Dholaria, M.; Sengupta, A.; Praneeth, Y.; Devgon, I.; Rana, A.; Kaur, M.; Karnwal, A.; Mahmoud, A. E. D. Recent Advances on Impact, Hazard, and Microbial Bioremediation of Microplastics in Marine Ecosystems: Challenges and Artificial Intelligence Way Forward. Water, Air, and Soil Pollution 2026, 237(5), 1–26. [Google Scholar] [CrossRef]
- Meng, X.; Nag, R. Human health risk assessment of nanoparticles through food consumption — occurrence, exposure, and toxicological implications. Science of the Total Environment 2026, 1013, 181340. [Google Scholar] [CrossRef]
- Ioachimescu, O. C.; Shaker, R. Translational science and related disciplines. Journal of Investigative Medicine 2025, 73(1), 3–26. [Google Scholar] [CrossRef]
- Khalid, M. H.; Farooq, F.; Aslam, B.; Saria, M. Probiotic-Derived Bacteriocins for Veterinary Biofilm Control: Mechanisms, Evidence, and One Health Translation. In Probiotics and Antimicrobial Proteins; 2025. [Google Scholar] [CrossRef]
- Aguirre, A. A.; Beasley, V. R.; Augspurger, T.; Benson, W. H.; Whaley, J.; Basu, N. One health—Transdisciplinary opportunities for SETAC leadership in integrating and improving the health of people, animals, and the environment. Environmental Toxicology and Chemistry 2016, 35(10), 2383–2391. [Google Scholar] [CrossRef]
- Anadón, A. Perspectives in veterinary pharmacology and toxicology. Frontiers in Veterinary Science 2016, 3(SEP), 1–12. [Google Scholar] [CrossRef]
- Choffnes, E. R.; Relman, D. A.; Olsen, L.; Hutton, R. Improving Food Safety Through a One Health Approach. In Improving Food Safety Through a One Health Approach; 2012. [Google Scholar] [CrossRef]
- Deblais, L.; Kathayat, D.; Helmy, Y. A.; Closs, G.; Rajashekara, G. Translating “big data”: Better understanding of host-pathogen interactions to control bacterial foodborne pathogens in poultry. Animal Health Research Reviews 2020, 21(1), 15–35. [Google Scholar] [CrossRef]
- Wu, D. D.; Olson, D. L. Computational simulation and risk analysis: An introduction of state of the art research. Mathematical and Computer Modelling 2013, 58(9–10), 1581–1587. [Google Scholar] [CrossRef]
- Kim, E. S. Antimicrobial stewardship programs in sepsis treatment: principles, impact, and future directions. Korean Journal of Internal Medicine 2026, 41(1), 60–73. [Google Scholar] [CrossRef] [PubMed]
- Almuzaini, A. M.; Aljohani, A. S. M.; Alajaji, A. I.; Elbehiry, A.; Abalkhail, A.; Almujaidel, A.; Aljarallah, S. N.; Sherif, H. R.; Marzouk, E.; Draz, A. A. Seroprevalence of brucellosis in camels and humans in the Al-Qassim region of Saudi Arabia and its implications for public health. AMB Express 2025, 15(1). [Google Scholar] [CrossRef] [PubMed]
- Dadar, M.; Foster, J. T. Camel brucellosis: a narrative review of epidemiology and control strategies. Veterinary Research Communications 2026, 50(1). [Google Scholar] [CrossRef]
- Leist, M.; Hartung, T.; Nicotera, P. Visions on Toxicity Testing in the 21st Century : Reflections on a Strategy Document of the US National Research Council; 2009. [Google Scholar]
- Gabrielsson, J.; Andersson, K.; Tobin, G.; Ingvast-Larsson, C.; Jirstrand, M. Maxsim2-Real-time interactive simulations for computer-assisted teaching of pharmacokinetics and pharmacodynamics. Computer Methods and Programs in Biomedicine 2014, 113(3), 815–829. [Google Scholar] [CrossRef]
- Wang, X.; Zheng, Y.; Ma, Y.; Du, L.; Chu, F.; Gu, H.; Dahlgren, R. A.; Li, Y.; Wang, H. Lipid metabolism disorder induced by up-regulation of miR-125b and miR-144 following β-diketone antibiotic exposure to F0-zebrafish (Danio rerio). Ecotoxicology and Environmental Safety 2018, 164(August), 243–252. [Google Scholar] [CrossRef]
- Fairman, K.; Li, M.; Kabadi, S. V.; Lumen, A. Physiologically based pharmacokinetic modeling: A promising tool for translational research and regulatory toxicology. Current Opinion in Toxicology 2020, 17–22. [Google Scholar] [CrossRef]
- Khan, R.; Khan, S. Integrative computational modeling framework linking mycotoxin contamination, microbial hazards, and antimicrobial resistance risk in dairy systems. BMC Microbiology 2026, Volume 26, 30. [Google Scholar] [CrossRef]
- Novakovic, J.; Milosavljevic, I.; Stepanova, M.; Ramenskaya, G.; Jeremic, N. Safe Meat, Smart Science : Biotechnology ’ s Role in Antibiotic Residue Removal 2025, 1–24.
- Obinwanne, C.; Ebhohimhen, S.; Chidi, B.; Chen, X.; Jiang, H.; Wu, Y. Machine learning-based predictive modeling of foodborne pathogens and antimicrobial resistance in food microbiomes using omics techniques: A systematic review. Food Research International 2025, 221(P1), 117255. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Xu, Z.; Lei, H. Investigation of an Immunoassay with Broad Speci fi city to Quinolone Drugs by Genetic Algorithm with Linear Assignment of Hypermolecular Alignment of Data Sets and Advanced Quantitative Structure − Activity Relationship Analysis 2016. [CrossRef]
- Rüdel, H.; Böhmer, W.; Müller, M.; Fliedner, A.; Ricking, M.; Teubner, D.; Schröter-kermani, C. Chemo sphere Retrospective study of triclosan and methyl-triclosan residues in fish and suspended particulate matter: Results from the German Environmental Specimen Bank. Chemosphere 2013, 91(11), 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Goh, L.; Kao, T.; Pan, Y.; Chang, C.; Lu, K. International Journal of Food Microbiology Growth modeling of uropathogenic Escherichia coli in raw and cooked beef as a function of storage temperature for shelf-life predictions. International Journal of Food Microbiology 2025, 442(June), 111359. [Google Scholar] [CrossRef]
- Silva, B. S.; Pia, A. K. R.; Furtado, M. M.; Freire, L.; Lemos, W. J. F.; Sant, A. S. International Journal of Food Microbiology Growth kinetics modeling of Pseudomonas aeruginosa in natural mineral water. International Journal of Food Microbiology 2026, 445, 111495. [Google Scholar] [CrossRef]
- Bhattacharya, R.; Bose, D.; Siddique, K. R.; Rodriguez, R. V.; Ray, A. Artificial intelligence for sustainable solutions in combating antimicrobial resistance through data driven health innovations. Discover Public Health 2026, Volume 23(10), 1–17. [Google Scholar] [CrossRef]
- Umeshkumar, K. U.; Karwasra, R. Zoonoticus: A machine learning model for genomic prediction of zoonotic bacterial strains using virulence, resistance, and mobile genetic elements. Computational Biology and Chemistry 2026, 120(P1), 108760. [Google Scholar] [CrossRef]
| Method | Application | Example |
|---|---|---|
| QSAR | Predict chemical toxicity | Feed additives |
| PBPK | Tissue residue modelling | Veterinary drugs |
| Molecular docking | Drug–target interactions | Transporter inhibition |
| Toxicogenomics | Biomarker discovery | Hepatotoxicity |
| AI / ML | AMR prediction | Antibiotic resistance |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




