Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Patient Selection
2.3. Treatment and Target Delineation
2.4. Molecular and Clinical Variables
2.5. Endpoints
2.6. Pattern of Failure Assessment
2.7. Statistical Analysis
3. Results
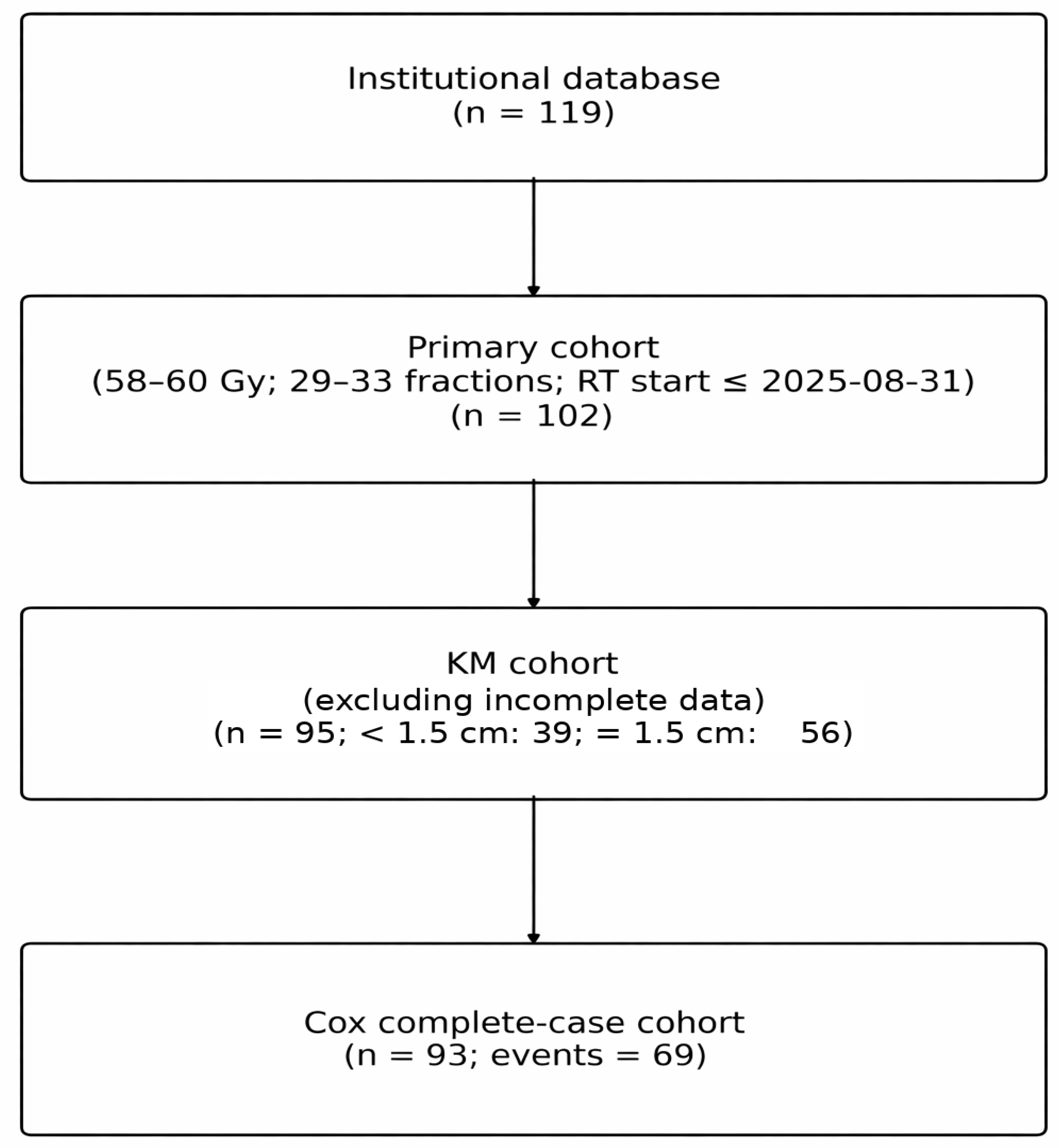
3.1. Patient Selection and Baseline Characteristics
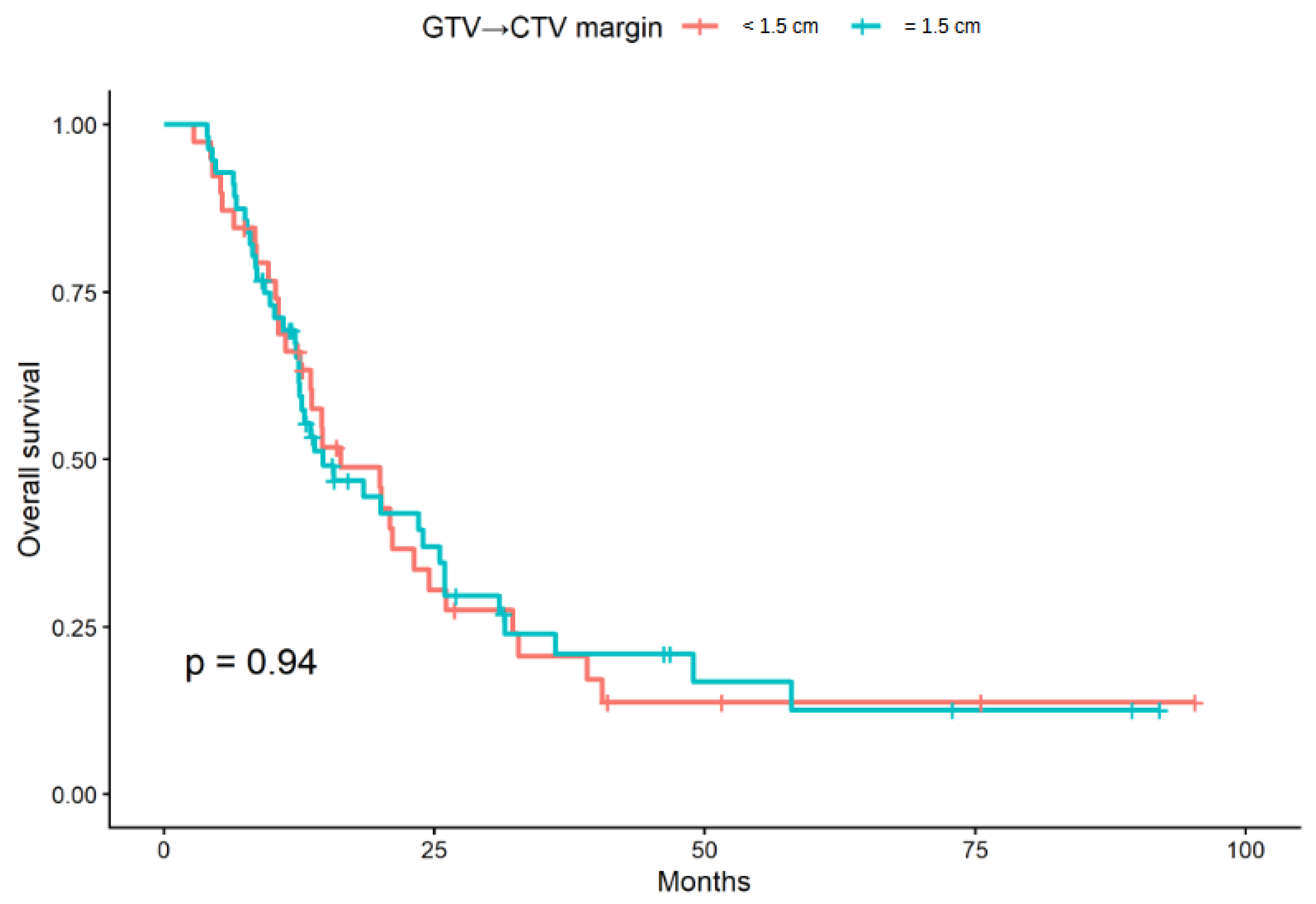
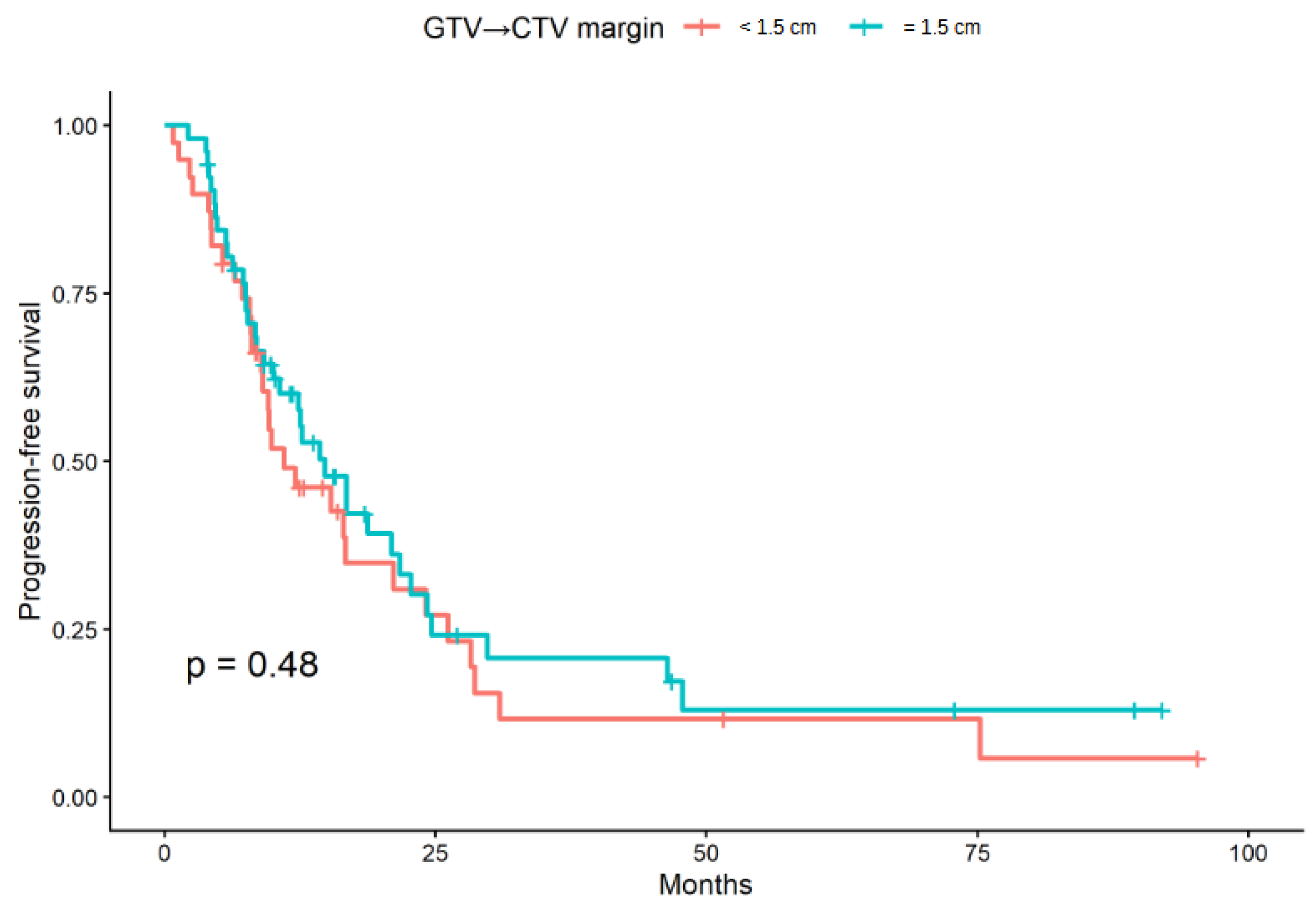
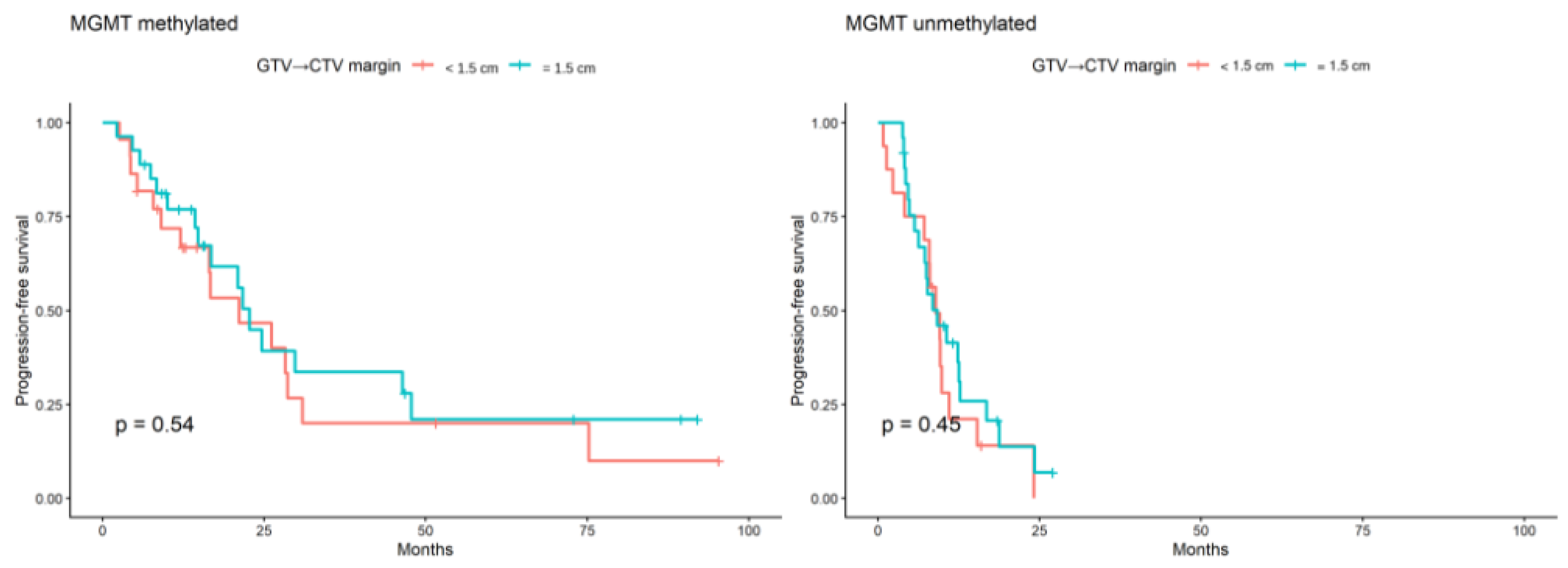
3.2. Overall Survival and Progression-Free-Survival According to CTV Margin
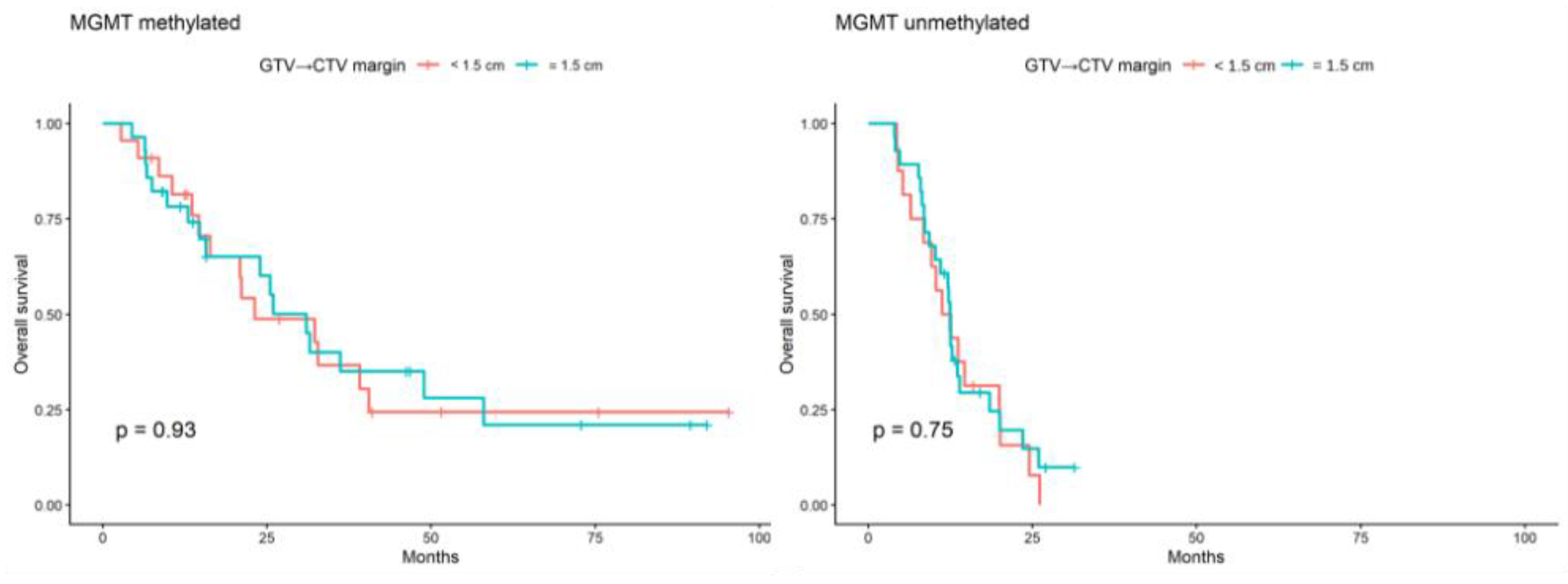
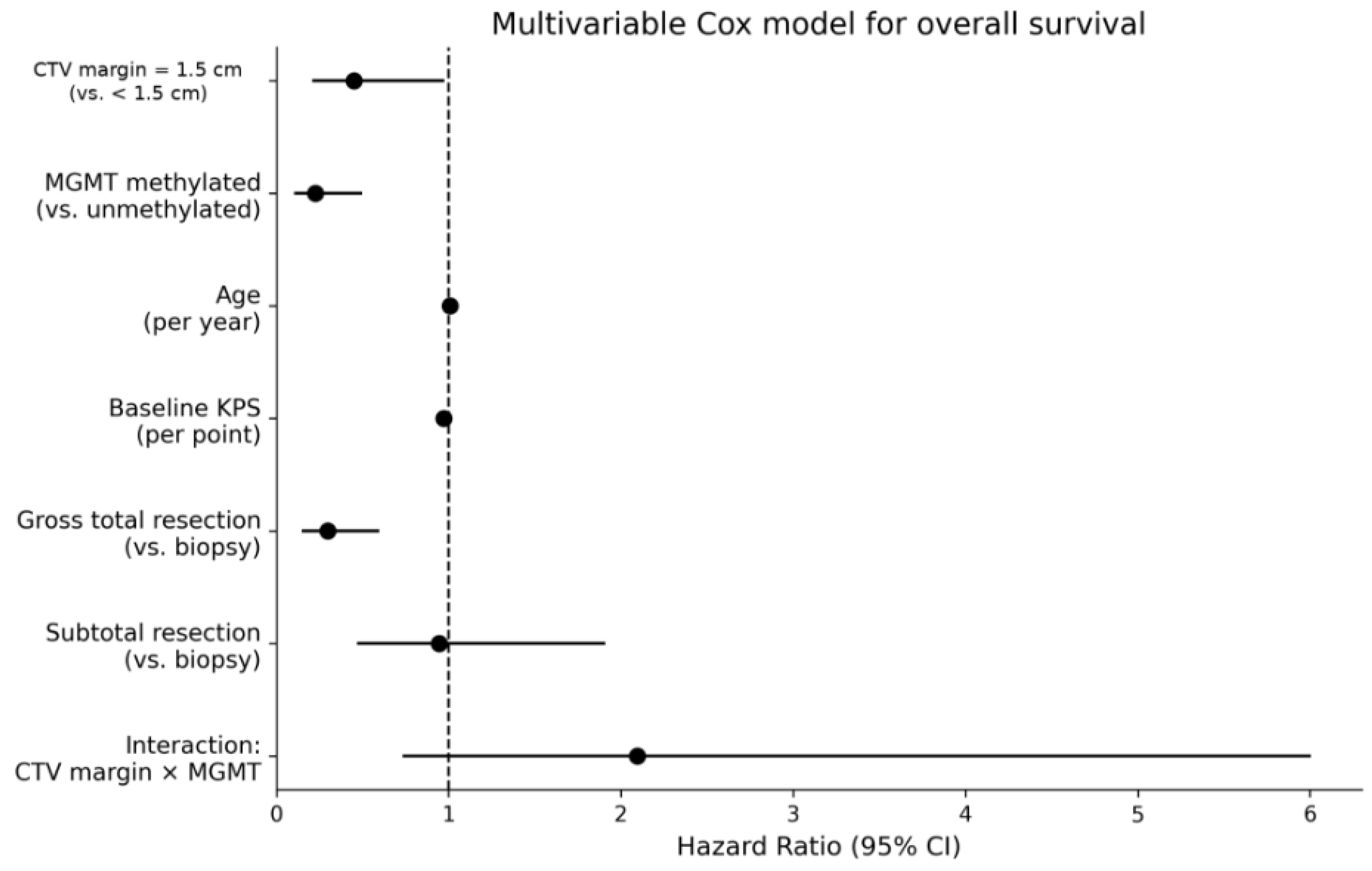
3.3. Multivariable Analysis
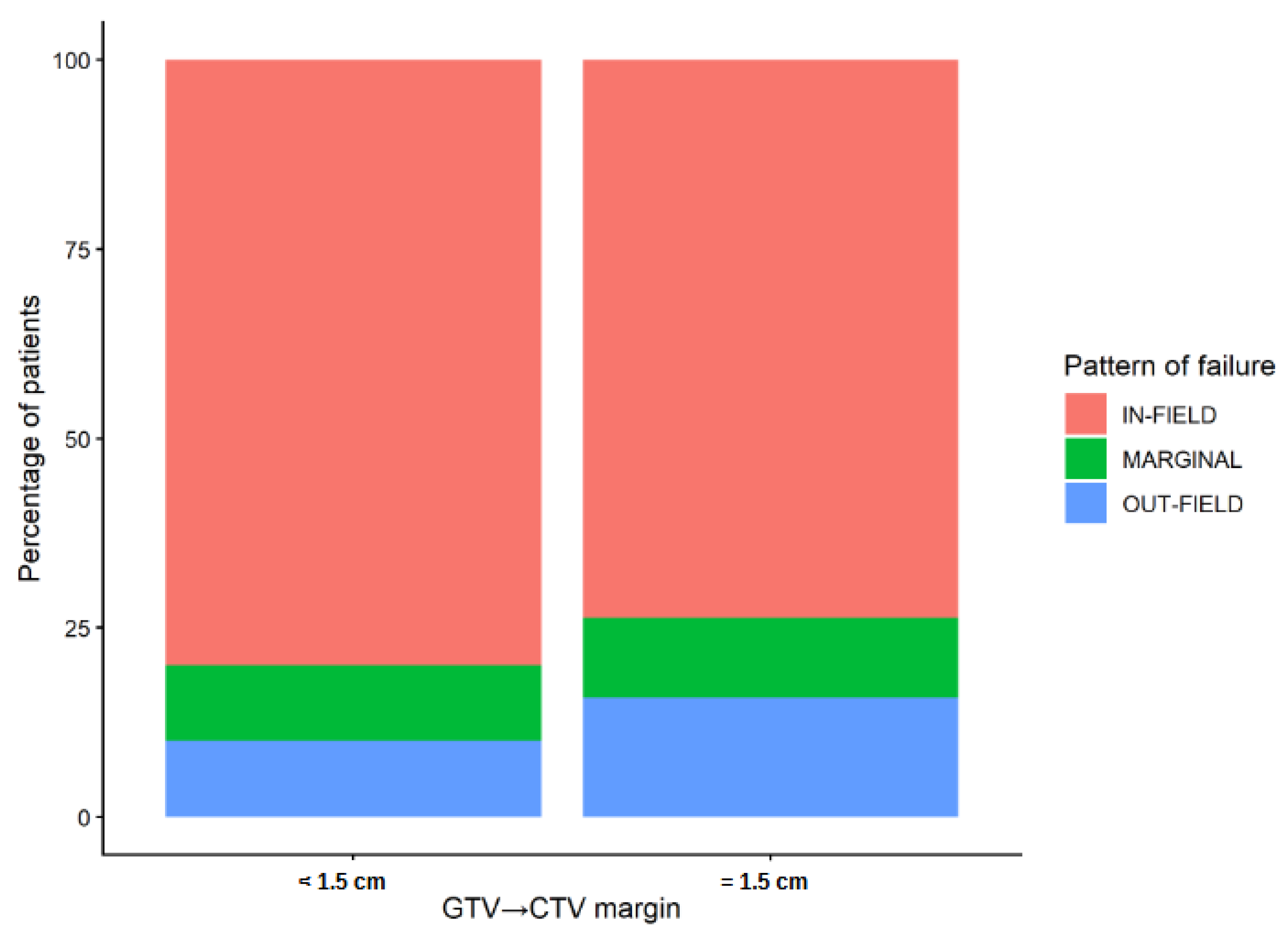
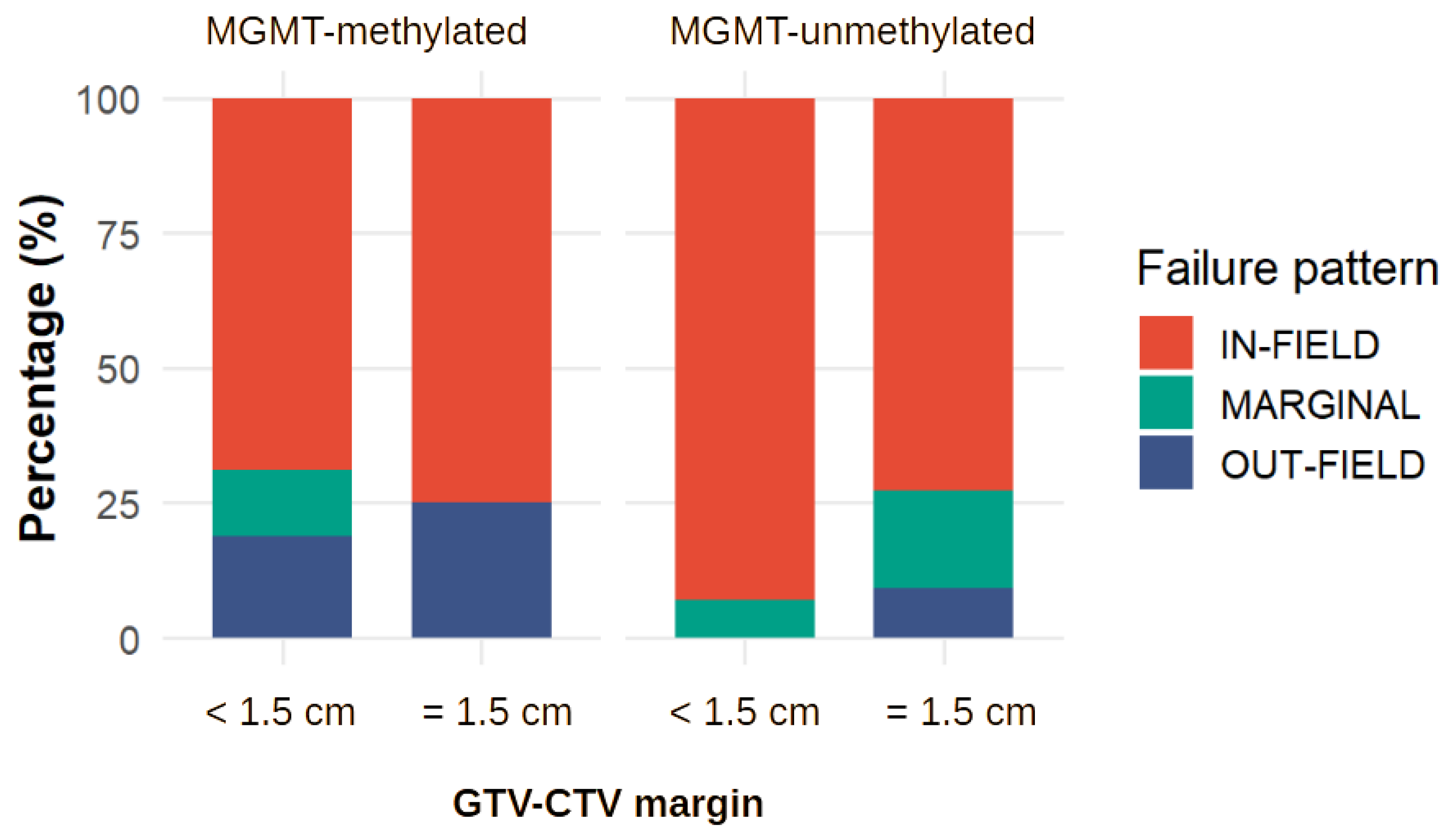
3.4. Pattern of Failure Analysis
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987-996. [CrossRef]
- Hegi, M.E.; Diserens, A.-C.; Gorlia, T.; Hamou, M.-F.; de Tribolet, N.; Weller, M.; Kros, J.M.; Hainfellner, J.A.; Mason, W.; Mariani, L.; et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 2005, 352, 997-1003. [CrossRef]
- Niyazi, M.; Andratschke, N.; Bendszus, M.; Chalmers, A.J.; Erridge, S.C.; Galldiks, N.; Lagerwaard, F.J.; Navarria, P.; Munck Af Rosenschöld, P.; Ricardi, U.; et al. ESTRO-EANO guideline on target delineation and radiotherapy details for glioblastoma. Radiother. Oncol. 2023, 184, 109663. [CrossRef]
- McDonald, M.W.; Shu, H.-K.G.; Curran, W.J., Jr.; Crocker, I.R. Pattern of failure after limited margin radiotherapy and temozolomide for glioblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 130-136. [CrossRef]
- Paulsson, A.K.; McMullen, K.P.; Peiffer, A.M.; Hinson, W.H.; Kearns, W.T.; Johnson, A.J.; Lesser, G.J.; Ellis, T.L.; Tatter, S.B.; Debinski, W.; et al. Limited margins using modern radiotherapy techniques does not increase marginal failure rate of glioblastoma. Am. J. Clin. Oncol. 2014, 37, 177-181. [CrossRef]
- Gebhardt, B.J.; Dobelbower, M.C.; Ennis, W.H.; Bag, A.K.; Markert, J.M.; Fiveash, J.B. Patterns of failure for glioblastoma multiforme following limited-margin radiation and concurrent temozolomide. Radiat. Oncol. 2014, 9, 130. [CrossRef]
- Minniti, G.; Tini, P.; Giraffa, M.; Capone, L.; Raza, G.; Russo, I.; Cinelli, E.; Gentile, P.; Bozzao, A.; Paolini, S.; et al. Feasibility of clinical target volume reduction for glioblastoma treated with standard chemoradiation based on patterns of failure analysis. Radiother. Oncol. 2023, 181, 109435. [CrossRef]
- Tini, P.; Donnini, F.; Marampon, F.; Vannini, M.; Carfagno, T.; Pastina, P.; Rubino, G.; Chibbaro, S.; Cerase, A.; Bagnacci, G.; et al. Biomolecular predictors of recurrence patterns and survival in IDH-wild-type glioblastoma: A retrospective analysis of patients treated with radiotherapy and temozolomide. Brain Sci. 2025, 15, 713. [CrossRef]
- Brandes AA, Tosoni A, Franceschi E, Sotti G, Frezza G, Amistà P, Morandi L, Spagnolli F, Ermani M. Recurrence pattern after temozolomide concomitant with and adjuvant to radiotherapy in newly diagnosed patients with glioblastoma: correlation With MGMT promoter methylation status. J Clin Oncol. 2009 Mar 10;27(8):1275-9. [CrossRef]
- Wen, P.Y.; Macdonald, D.R.; Reardon, D.A.; Cloughesy, T.F.; Sorensen, A.G.; Galanis, E.; DeGroot, J.; Wick, W.; Gilbert, M.R.; Lassman, A.B.; et al. Updated response assessment criteria for high-grade gliomas: Response assessment in neuro-oncology working group. J. Clin. Oncol. 2010, 28, 1963-1972. [CrossRef]
- Leao, D.J.; Craig, P.G.; Godoy, L.F.; Leite, C.D.C.; Policeni, B. Response Assessment in Neuro-Oncology criteria for gliomas: Practical approach using conventional and advanced techniques. AJNR Am. J. Neuroradiol. 2020, 41, 10-20. [CrossRef]
| Variable | CTV margin <1.5 cm (n = 37) | CTV margin = 1.5 cm (n = 56) | p-value | |
| Age, years (mean ± SD) | 59.24 (10.61) | 62.13 (9.91) | 0.184 | |
| Baseline KPS (mean ± SD) | 88.65 (9.76) | 90.00 (8.94) | 0.493 | |
| MGMT promoter methylation status, n (%) | Unmethylated | 16 (43.2) | 28 (50.0) | 0.670 |
| Methylated | 21 (56.8) | 28 (50.0) | ||
| Extent of resection, n (%) | Biopsy | 7 (18.9) | 8 (14.3) | 0.612 |
| Gross total resection | 19 (51.4) | 26 (46.4) | ||
| Subtotal resection | 11 (29.7) | 22 (39.3) |
| Covariate | Hazard Ratio (95% CI) | p-value |
| CTV margin = 1.5 cm (vs. <1.5 cm), within the MGMT-unmethylated reference group | 0.45 (0.208-0.975) | 0.0429 |
| MGMT methylated (vs. unmethylated) | 0.226 (0.103-0.498) | 0.000225 |
| Age, years | 1.007 (0.981-1.034) | 0.583 |
| Baseline KPS, per point | 0.972 (0.944-1) | 0.0514 |
| Gross total resection (vs. biopsy) | 0.299 (0.149-0.598) | 0.000641 |
| Subtotal resection (vs. biopsy) | 0.946 (0.469-1.911) | 0.878 |
| Interaction: CTV margin × MGMT methylation | 2.096 (0.732-6.005) | 0.168 |
| Derived effect: CTV margin = 1.5 cm in MGMT-methylated subgroup | 0.944 (0.449-1.985) | 0.879 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




