Submitted:
30 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
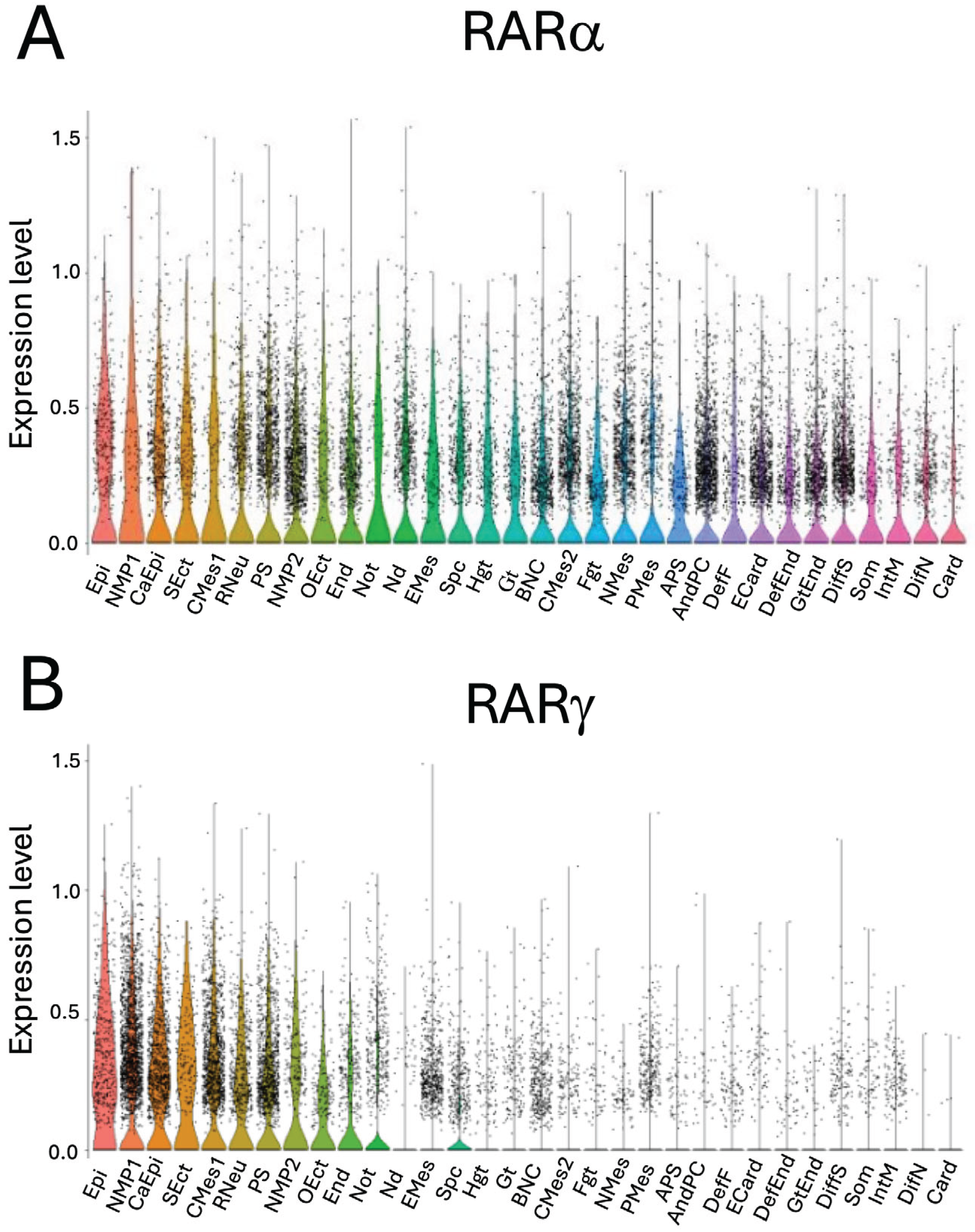
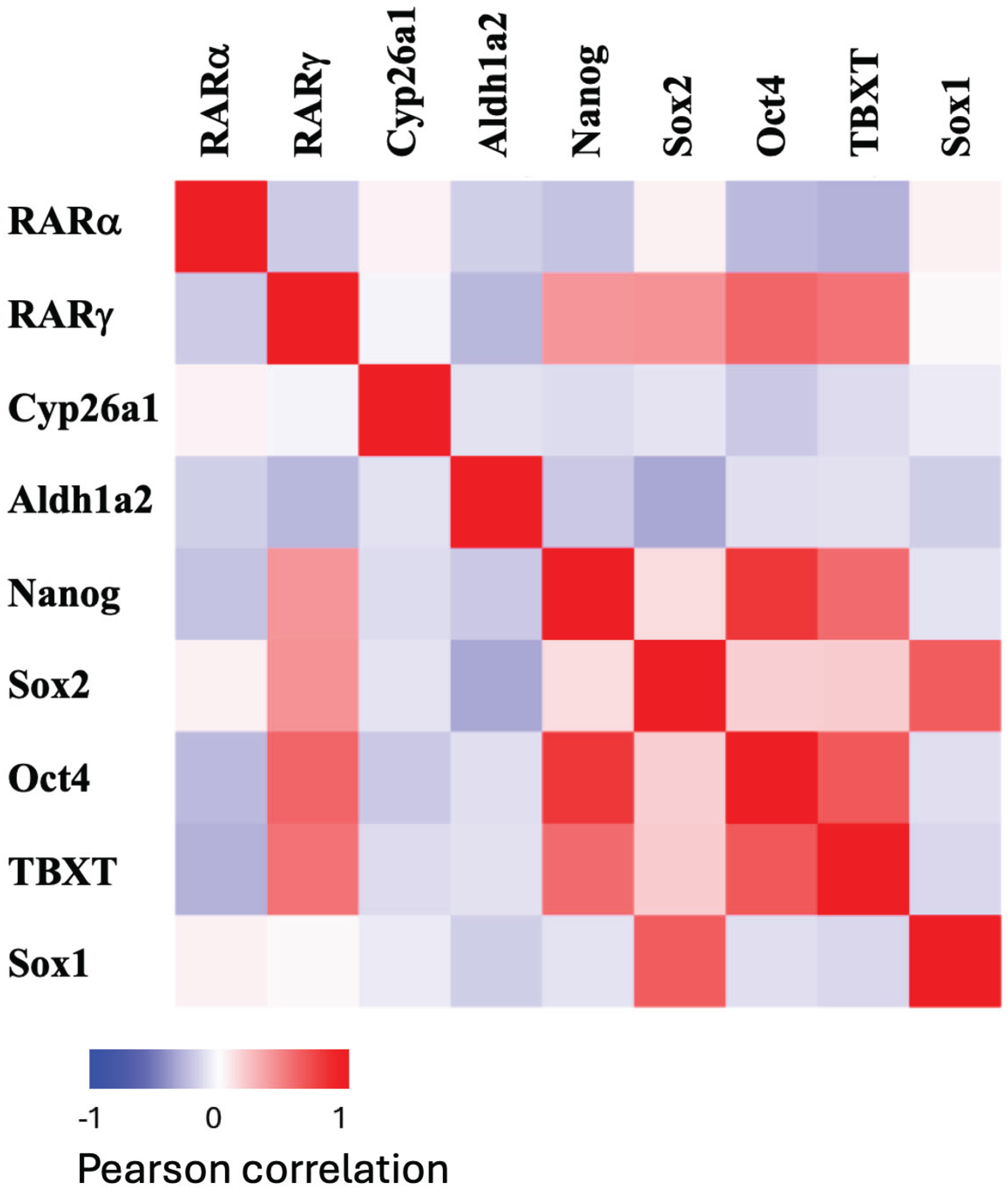
2.1. The Expression of RARα and RARγ During Gastruloid Development
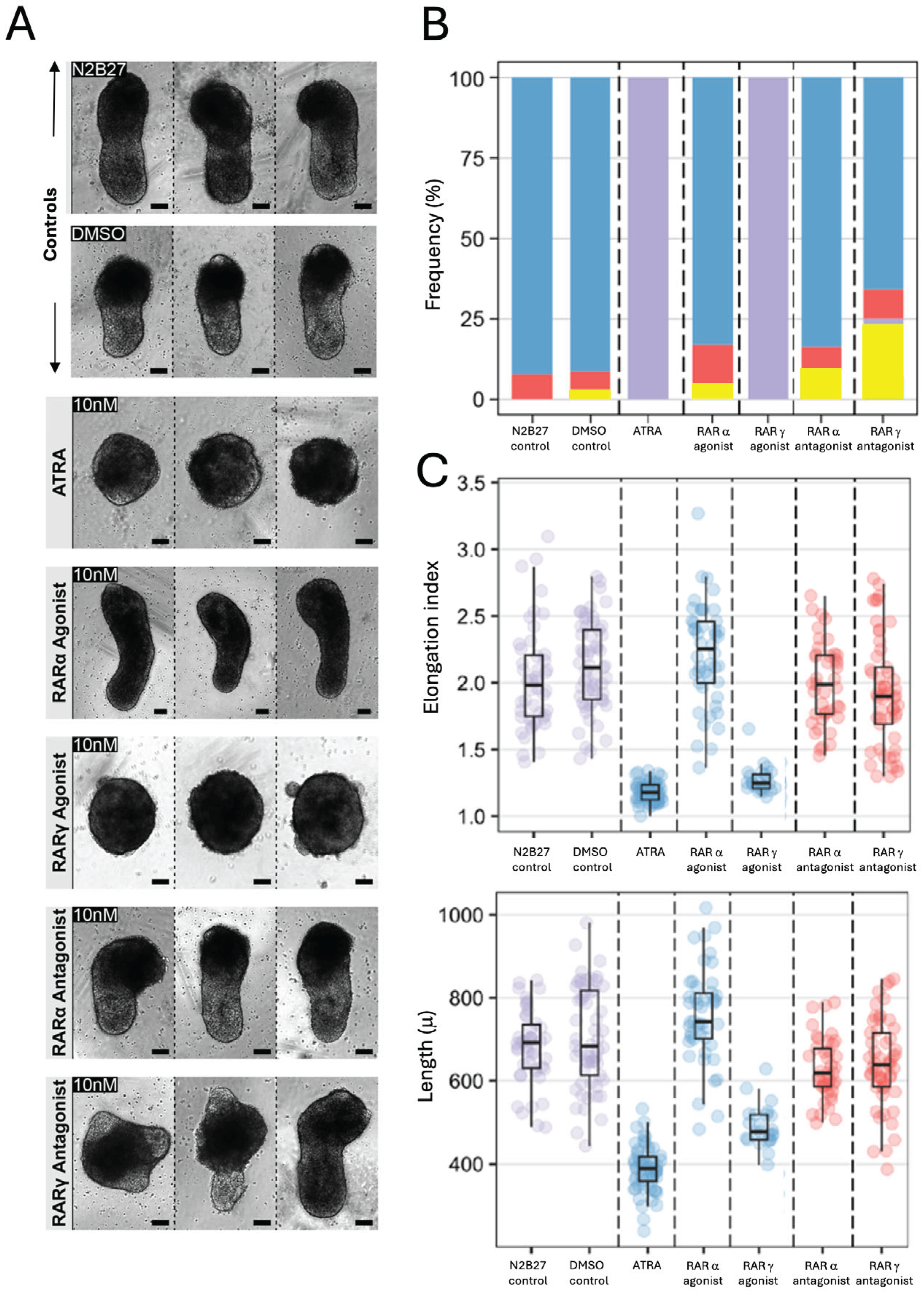
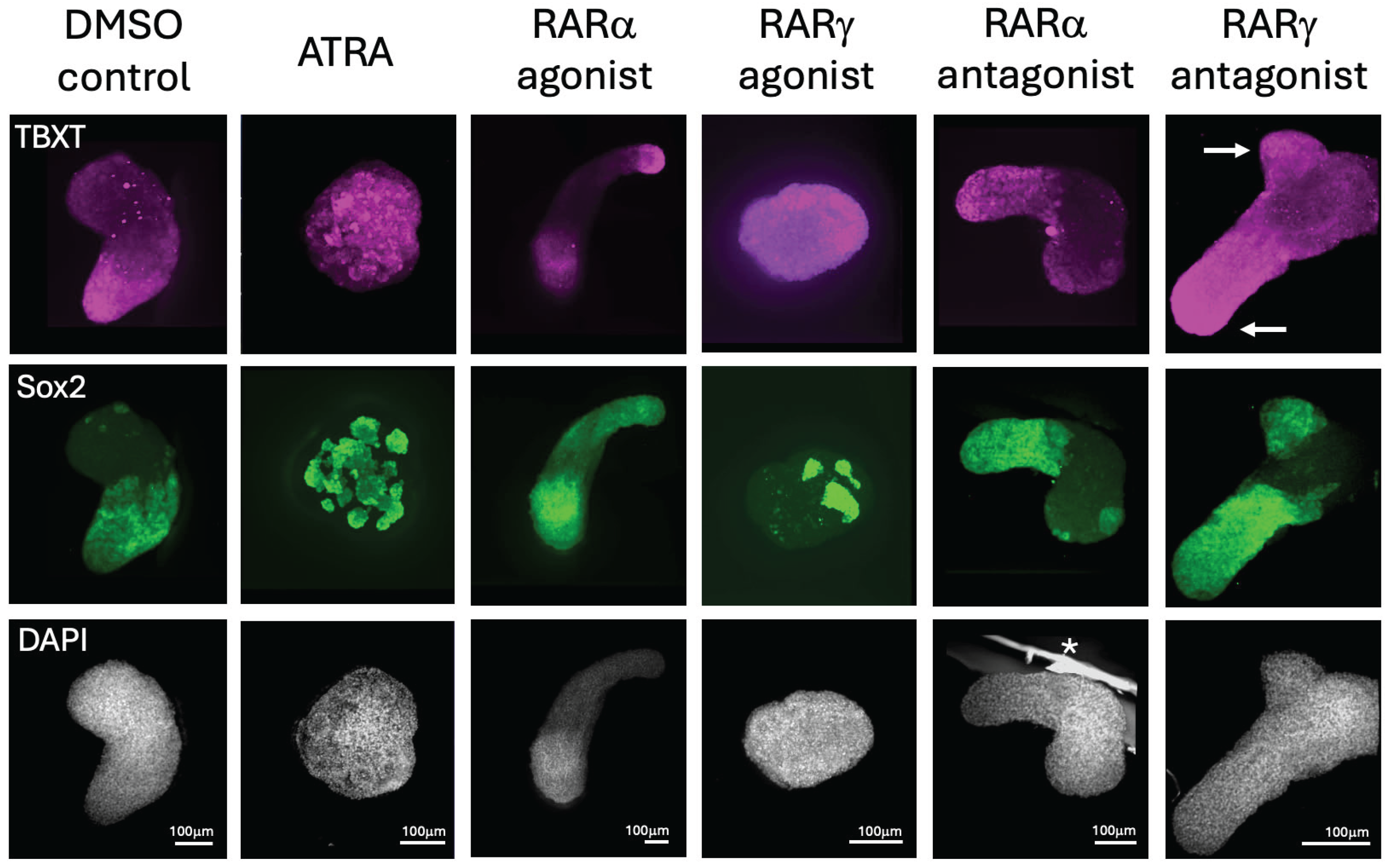
2.2. The Effects of ATRA and RARa and RARγ Specific Agonists and Antagonists on Gastruloid Development
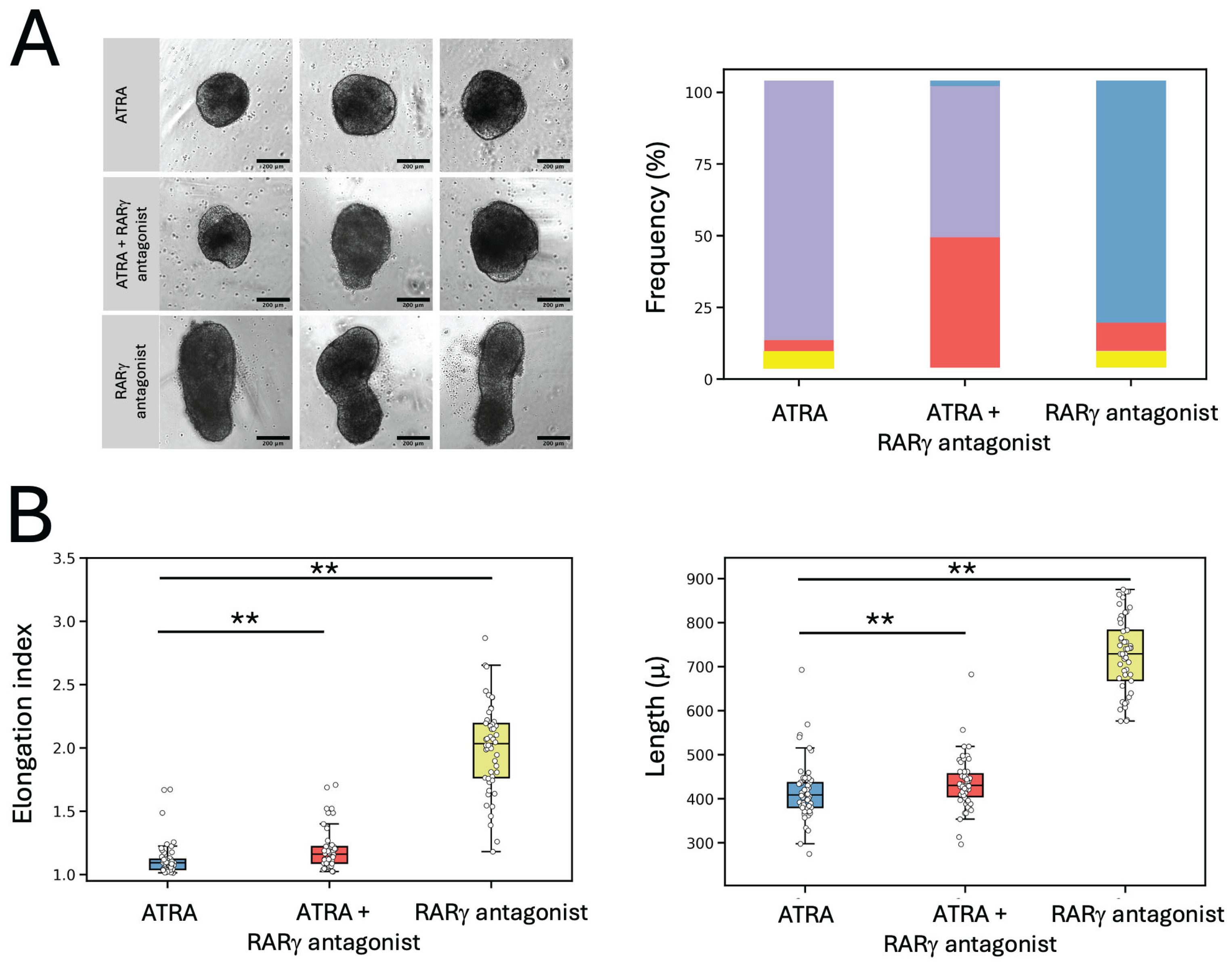
2.3. The Inhibitory Effects of ATRA on Gastruloid Axial Elongation Were Partially Abrogated by RARγ Specific Antagonism
3. Discussion
4. Materials and Methods
4.1. Single Cell RNA-Seq Analysis
4.2. Retinoids
4.3. Routine ES cell culture
4.4. Generation of gastruloids and treatments with ATRA and synthetic retinoids
4.4. Immunostaining
4.5. Microscopy and Image Analysis
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Duester, G. Retinoic acid synthesis and signaling during early organogenesis. Cell 2008, 134, 921–931. [Google Scholar] [CrossRef]
- Rhinn, M; Dollé, P. Retinoic acid signalling during development. Development 2012, 139, 843–58. [Google Scholar] [CrossRef]
- Berenbguer, M; Duester, G. Retinoic acid, RARs and early development. J Mol Endocrinol. 2022, 69, 59–67. [Google Scholar] [CrossRef]
- Rossant, J.; Zirngibl, R.; Cado, D.; Shago, M.; Giguère, V. Expression of a retinoic acid response element-hsplacZ transgene defines specific domains of transcriptional activity during mouse embryogenesis. Genes Dev. 1991, 5, 1333–1344. [Google Scholar] [CrossRef]
- Duester, G. Early retinoic acid signaling organizes the body axis and defines domains for the forelimb and eye. Curr Top Dev Biol. 2025, 161, 1–32. [Google Scholar]
- Sandell, L.L.; Sanderson, B.W.; Moiseyev, G.; Johnson, T.; Mushegian, A.; Young, K.; Rey, J-P.; Ma, J-X.; Staehling-Hampton, K.; Trainor, P.A. RDH10 is essential for synthesis of embryonic retinoic acid and is required for limb, craniofacial, and organ development. Genes Dev. 2007, 21, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Clagett-Dame, M.; Knutson, D. Vitamin A in reproduction and development. Nutrients 2011, 3, 385–428. [Google Scholar] [CrossRef] [PubMed]
- Kam, R.K.; Shi, W.; Chan, S.O.; Chen, Y.; Xu, G.; Lau, C.B.; Fung, K.P.; Chan, W.Y.; Zhao, H. Dhrs3 protein attenuates retinoic acid signaling and is required for early embryonic patterning. J Biol Chem. 2013, 288, 31477–87. [Google Scholar] [CrossRef] [PubMed]
- Roeske, TC; Scharff, C; Olson, CR; Nshdejan, A; Mello, CV. Long-distance retinoid signaling in the zebra finch brain. PLoS One. 9 2014, e111722. [Google Scholar] [CrossRef]
- Hashimoto, AS; Yu, J; Williams, C; Gaudenz, K; Varshosaz, P; Zhao, R; Pilli, N; Liu, T; Russell, J; Tooze, RS; Twigg, SRF; Banka, S; Sweeney, E; McGowan, SJ; Knight, SJL; Taylor, JC; Froukh, TJ; Palafoll, MIV; Martínez-Gil, N; Costa-Roger, M; Villarreal-Molina, MT; Lieberman Hernandez, E; Abou Jamra, R; Gattermann, F; Koch-Hogrebe, M; Wieczorek, D; Trainor, PA; Moise, AR; Wilkie, AOM; Kane, MA. Identification and characterization of short-chain dehydrogenase/reductase 3 (DHRS3) deficiency, a retinoic acid embryopathy of humans. Genet Med Open. 2025, 3, 103427. [Google Scholar] [CrossRef]
- Uehara, M.; Yashiro, K.; Takaoka, K.; Yamamoto, M.; Hamada, H. Removal of maternal retinoic acid by embryonic CYP26 is required for correct Nodal expression during early embryonic patterning. Genes Dev. 2009, 23, 1689–98. [Google Scholar] [CrossRef]
- Fujii, H.; Sato, T.; Kaneko, S.; Gotoh, O.; Fujii-Kuriyama, Y.; Osawa, K.; Kato, S.; Hamada, H. Metabolic inactivation of retinoic acid by a novel P450 differentially expressed in developing mouse embryos. EMBO J. 1997, 16, 4163–73. [Google Scholar] [CrossRef]
- Sakai, Y.; Meno, C.; Fujii, H.; Nishino, J.; Shiratori, H.; Saijoh, Y.; Rossant, J.; Hamada, H. The retinoic acid-inactivating enzyme CYP26 is essential for establishing an uneven distribution of retinoic acid along the anterio-posterior axis within the mouse embryo. Genes Dev 2001, 15, 213–25. [Google Scholar] [CrossRef]
- Ribes, V; Fraulob, V; Petkovich, M; Dollé, P. The oxidizing enzyme CYP26a1 tightly regulates the availability of retinoic acid in the gastrulating mouse embryo to ensure proper head development and vasculogenesis. Dev Dyn 2007, 236, 644–53. [Google Scholar] [CrossRef]
- Shaker, M.R.; Lee, J.H.; Park, S.H.; Kim, J.Y.; Son, G.H.; Son, J.W.; Park, B.H.; Rhyu, I.J.; Kim, H.; Sun, W. Anteroposterior Wnt-RA gradient defines adhesion and migration properties of neural progenitors in developing spinal cord. Stem Cell Reports 2020, 15, 898–911. [Google Scholar] [CrossRef]
- Yamanaka, Y.; Hamidi, S.; Yoshioka-Kobayashi, K.; Munira, S.; Sunadome, K.; Zhang, Y.; Kurokawa, Y.; Ericsson, R.; Mieda, A.; Thompson, J.L.; Kerwin, J.; Lisgo, S.; Yamamoto, T.; Moris, N.; Martinez-Arias, A.; Tsujimura, T.; Alev, C. Reconstituting human somitogenesis in vitro. Nature 2023, 614, 509–520. [Google Scholar] [CrossRef]
- Chambon, P. A decade of molecular biology of retinoic acid receptors. FASEB J 1996, 10, 940–54. [Google Scholar] [CrossRef] [PubMed]
- Wai, H.A.; Kawakami, K.; Wada, H.; Müller, F.; Vernallis, A.B.; Brown, G.; Johnson, W.E. The development and growth of tissues derived from cranial neural crest and primitive mesoderm is dependent on the ligation status of retinoic acid receptor gamma: evidence that retinoic acid receptor gamma functions to maintain stem/progenitor cells in the absence of retinoic acid. Stem Cells Dev. 2015, 24, 507–19. [Google Scholar]
- Brown, G. Retinoic acid receptor regulation of decision-making for cell differentiation. Front Cell Dev Biol. 2023, 11, 1182204. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Jia, X.; Yu, L. The building blocks of embryo models: embryonic and extraembryonic stem cells. Cell Discov. 2025, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.A.; Girgin, M.; Alonso-Crisostomo, L.; Trivedi, V.; Baillie-Johnson, P.; Glodowski, C.R.; Hayward, P.C.; Collignon, J.; Gustavsen, C.; Serup, P.; Steventon, B.; Lutolf, M.P.; Arias, A.M. Anteroposterior polarity and elongation in the absence of extra-embryonic tissues and of spatially localised signalling in gastruloids: mammalian embryonic organoids. Development 2017, 144, 3894–3906. [Google Scholar]
- Siggia, E.D.; Warmflash, A. Modeling mammalian gastrulation with embryonic stem cells. Curr Top Dev Biol 2018, 129, 1–23. [Google Scholar] [PubMed]
- Suppinger, S.; Zinner, M.; Aizarani, N.; Lukonin, I.; Ortiz, R.; Azzi, C.; Stadler, M.B.; Vianello, S.; Palla, G.; Kohler, H.; Mayran, A.; Lutolf, M.P.; Liberali, P. Multimodal characterization of murine gastruloid development. Cell Stem Cell 2023, 30, 867–884. [Google Scholar] [CrossRef] [PubMed]
- Mantziou, V.; Baillie-Benson, P.; Jaklin, M.; Kustermann, S.; Arias, A.M.; Moris, N. In vitro teratogenicity testing using a 3D, embryo-like gastruloid system. Reprod Toxicol. 2021, 105, 72–90. [Google Scholar] [CrossRef]
- Hamazaki, N.; Yang, W.; Kubo, C.A.; Qiu, C.; Martin, B.K.; Garge, R.K.; Regalado, S.G.; Nichols, E.K.; Pendyala, S.; Bradley, N.; Fowler, D.M.; Lee, C.; Daza, R.M.; Srivatsan, S.; Shendure, J. Retinoic acid induces human gastruloids with posterior embryo-like structures. Nat Cell Biol. 2024, 26, 1790–1803. [Google Scholar] [CrossRef] [PubMed]
- De Leenheer, A.P.; Lambert, W.E.; Claeys, I. All-trans-retinoic acid: measurement of reference values in human serum by high performance liquid chromatography. J Lipid Res. 1982, 23, 1362–1367. [Google Scholar] [CrossRef]
- Eckhoff, C.; Nau, H. Identification and quantitation of all-trans- and 13-cis-retinoic acid and 13-cis-4-oxo-retinoic acid in human plasma. J Lipid Res. 1990, 31, 1445–1454. [Google Scholar] [CrossRef]
- Brown, G. Deregulation of all-trans retinoic acid signaling and development in cancer. Int. J. Mol. Sci. 2023, 24, 12089. [Google Scholar] [CrossRef]
- Li, Y.; Mao, X.; Zhou, X.; Su, Y.; Zhou, X.; Shi, K.; Zhao, S. An optimized method for neuronal differentiation of embryonic stem cells in vitro. Neurosci Methods 2020, 15, 330:108486. [Google Scholar] [CrossRef]
- Iturbide, A.; Ruiz Tejada Segura, M.L.; Noll, C.; Schorpp, K.; Rothenaigner, I.; Ruiz-Morales, E.R.; Lubatti, G.; Agami, A.; Hadian, K.; Scialdone, A.; Torres-Padilla, M.E. Retinoic acid signaling is critical during the totipotency window in early mammalian development. Nat Struct Mol Biol. 2021, 28, 521–532. [Google Scholar] [CrossRef]
- Oeda, S.; Hayashi, Y.; Chan, T.; Takasato, M.; Aihara, Y.; Okabayashi, K.; Ohnuma, K.; Asashima, M. Induction of intermediate mesoderm by retinoic acid receptor signaling from differentiating mouse embryonic stem cells. Int J Dev Biol. 2013, 57, 383–9. [Google Scholar] [CrossRef]
- Hughes, P.J.; Zhao, Y.; Chandraratna, R.A.; Brown, G. Retinoid-mediated stimulation of steroid sulfatase activity in myeloid leukemic cell lines requires RAR alpha and RXR and involves the phosphoinositide 3-kinase and ERK-MAP kinase pathways. J of Cell Biochem. 2006, 97, 327–350. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.; Broguiere, N.; Miyamoto, M.; Boni, A.; Guiet, R.; Girgin, M.; Kelly, R.G.; Kwon, C.; Lutolf, M.P. Capturing cardiogenesis in gastruloids. Cell Stem Cell 2021, 28, 230–240. [Google Scholar] [CrossRef]
- Braccioli, L.; van den Brand, T.; Alonso Saiz, N.; Fountas, C.; Celie, P.H.N.; Kazokaitė-Adomaitienė, J.; de Wit, E. Identifying cross-lineage dependencies of cell-type-specific regulators in mouse gastruloids. Dev Cell. 2025, 60, 2007–2022. [Google Scholar] [CrossRef] [PubMed]
- Fiuza, U.M.; Bonavia, S.; Pascual-Mas, P.; Torregrosa-Cortés, G.; Casaní-Galdón, P.; Robertson, G.; Dias, A.; Martinez Arias, A. Morphogenetic constraints in the development of gastruloids: Implications for mouse gastrulation. Cells Dev. 2025, 183, 204043. [Google Scholar] [CrossRef]
- Yang, J.; Wang, W.; Ooi, J.; Campos, L.S.; Lu, L.; Liu, P. Signalling Through Retinoic Acid Receptors is Required for reprogramming of both mouse embryonic fibroblast cells and epiblast stem cells to induced pluripotent stem cells. Stem Cells. 2015, 33, 1390–404. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yang, J.; Liu, H.; Lu, D.; Chen, X.; Zenonos, Z.; Campos, L.S.; Rad, R.; Guo, G.; Zhang, S.; Bradley, A.; Liu, P. Rapid and efficient reprogramming of somatic cells to induced pluripotent stem cells by retinoic acid receptor gamma and liver receptor homolog 1. Proc Natl Acad Sci U S A. 2011, 108, 18283–8. [Google Scholar] [CrossRef]
- Taei, A.; Kiani, T.; Taghizadeh, Z.; Moradi, S.; Samadian, A.; Mollamohammadi, S.; Sharifi-Zarchi, A.; Guenther, S.; Akhlaghpour, A.; Asgari Abibeiglou, B.; Najar-Asl, M.; Karamzadeh, R.; Khalooghi, K.; Braun, T.; Hassani, S.N.; Baharvand, H. Temporal activation of LRH-1 and RAR-γ in human pluripotent stem cells induces a functional naïve-like state. EMBO Rep. 2020, 21, e47533. [Google Scholar] [CrossRef]
- Shimono, K.; Tung, W.E.; Macolino, C.; Chi, A.H.; Didizian, J.H.; Mundy, C.; Chandraratna, R.A.; Mishina, Y.; Enomoto-Iwamoto, M.; Pacifici, M.; Iwamoto, M. Potent inhibition of heterotopic ossification by nuclear retinoic acid receptor-γ agonists. Nat Med. 2011, 17, 454–60. [Google Scholar] [CrossRef]
- Beederman, M.; Lamplot, J.D.; Nan, G.; Wang, J.; Liu, X.; Yin, L.; Li, R.; Shui, W.; Zhang, H.; Kim, S.H.; Zhang, W.; Zhang, J.; Kong, Y.; Denduluri, S.; Rogers, M.R.; Pratt, A.; Haydon, R.C.; Luu, H.H.; Angeles, J.; Shi, L.L.; He, T.C. BMP signaling in mesenchymal stem cell differentiation and bone formation. J Biomed Sci Eng. 2013, 6, 32–52. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, J.; Mu, W. BMP-2 promotes fracture healing by facilitating osteoblast differentiation and bone defect osteogenesis. Am J Transl Res. 2023, 15, 6751–6759. [Google Scholar]
- Pendaries, V.; Verrecchia, F.; Michel, S.; Mauviel, A. Retinoic acid receptors interfere with the TGF-beta/Smad signaling pathway in a ligand-specific manner. Oncogene 2003, 22, 8212–20. [Google Scholar] [CrossRef] [PubMed]
- Brown, G. The influences of RARγ on the behavior of normal and cancer stem cells. Int J Mol Sci. 2026, 27, 1291. [Google Scholar] [CrossRef]
- Qin, K.; Yu, M.; Fan, J.; Wang, H.; Zhao, P.; Zhao, G.; Zeng, W.; Chen, C.; Wang, Y.; Wang, A.; Schwartz, Z.; Hong, J.; Song, L.; Wagstaff, W.; Haydon, R.C.; Luu, H.H.; Ho, S.H.; Strelzow, J.; Reid, R.R.; He, T.C.; Shi, L.L. Canonical and noncanonical Wnt signaling: Multilayered mediators, signaling mechanisms and major signaling crosstalk. Genes Dis. 2023, 11, 103–134. [Google Scholar] [CrossRef] [PubMed]
- Osei-Sarfo, K.; Gudas, L.J. Retinoic acid suppresses the canonical Wnt signaling pathway in embryonic stem cells and activates the noncanonical Wnt signaling pathway. Stem Cells 2014, 32, 2061–2071. [Google Scholar] [CrossRef]
- Mullen, A.C.; Wrana, J.L. TGF-β family signaling in embryonic and somatic stem-cell renewal and differentiation. Cold Spring Harb Perspect Biol. 2017, 9, a022186. [Google Scholar] [CrossRef]
- Huang, G.L.; Luo, Q.; Rui, G.; Zhang, W.; Zhang, Q.Y.; Chen, Q.X.; Shen, D.Y. Oncogenic activity of retinoic acid receptor γ is exhibited through activation of the Akt/NF-κB and Wnt/β-catenin pathways in cholangiocarcinoma. Mol Cell Biol. 2013, 33, 3416–25. [Google Scholar] [CrossRef]
- Huang, G.L.; Song, W.; Zhou, P.; Fu, Q.R.; Lin, C.L.; Chen, Q.X.; Shen, D.Y. Oncogenic retinoic acid receptor γ knockdown reverses multi-drug resistance of human colorectal cancer via Wnt/β-catenin pathway. Cell Cycle 2017, 16, 685–692. [Google Scholar] [CrossRef]
- Mendoza-Parra, M.A.; Walia, M.; Sankar, M.; Gronemeyer, H. Dissecting the retinoid-induced differentiation of F9 embryonal stem cells by integrative genomics. Mol Syst Biol. 2011, 11, 538. [Google Scholar] [CrossRef]
- Hoxie, H.R.; Tang, X.H.; Gudas, L.J. Multiple modes of transcriptional regulation by the nuclear hormone receptor RARγ in human squamous cell carcinoma. J Biol Chem. 2026, 302, 110965. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Hao, S.; Andersen-Nissen, E.; Mauck, W.M., 3rd; Zheng, S.; Butler, A.; Lee, M.J.; Wilk, A.J.; Darby, C.; Zager, M.; Hoffman, P.; Stoeckius, M.; Papalexi, E.; Mimitou, E.P.; Jain, J.; Srivastava, A.; Stuart, T.; Fleming, L.M.; Yeung, B.; Rogers, A.J.; McElrath, J.M.; Blish, C.A.; Gottardo, R.; Smibert, P.; Satija, R. Integrated analysis of multimodal single-cell data. Cell 2021, 184, 3573–3587. [Google Scholar] [CrossRef] [PubMed]
- Morabito, S.; Reese, F.; Rahimzadeh, N.; Miyoshi, E.; Swarup, V. hdWGCNA identifies co-expression networks in high-dimensional transcriptomics data. Cell Rep Methods 2023, 3, 100498. [Google Scholar] [CrossRef] [PubMed]
- Girgin, M. U.; Broguiere, N.; Mattolini, L.; Lutolf, M. P. Gastruloids generated without exogenous Wnt activation develop anterior neural tissues. 2021. Stem Cell Reports 2021, 16, 1143–1155. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



