Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Overall Workflow
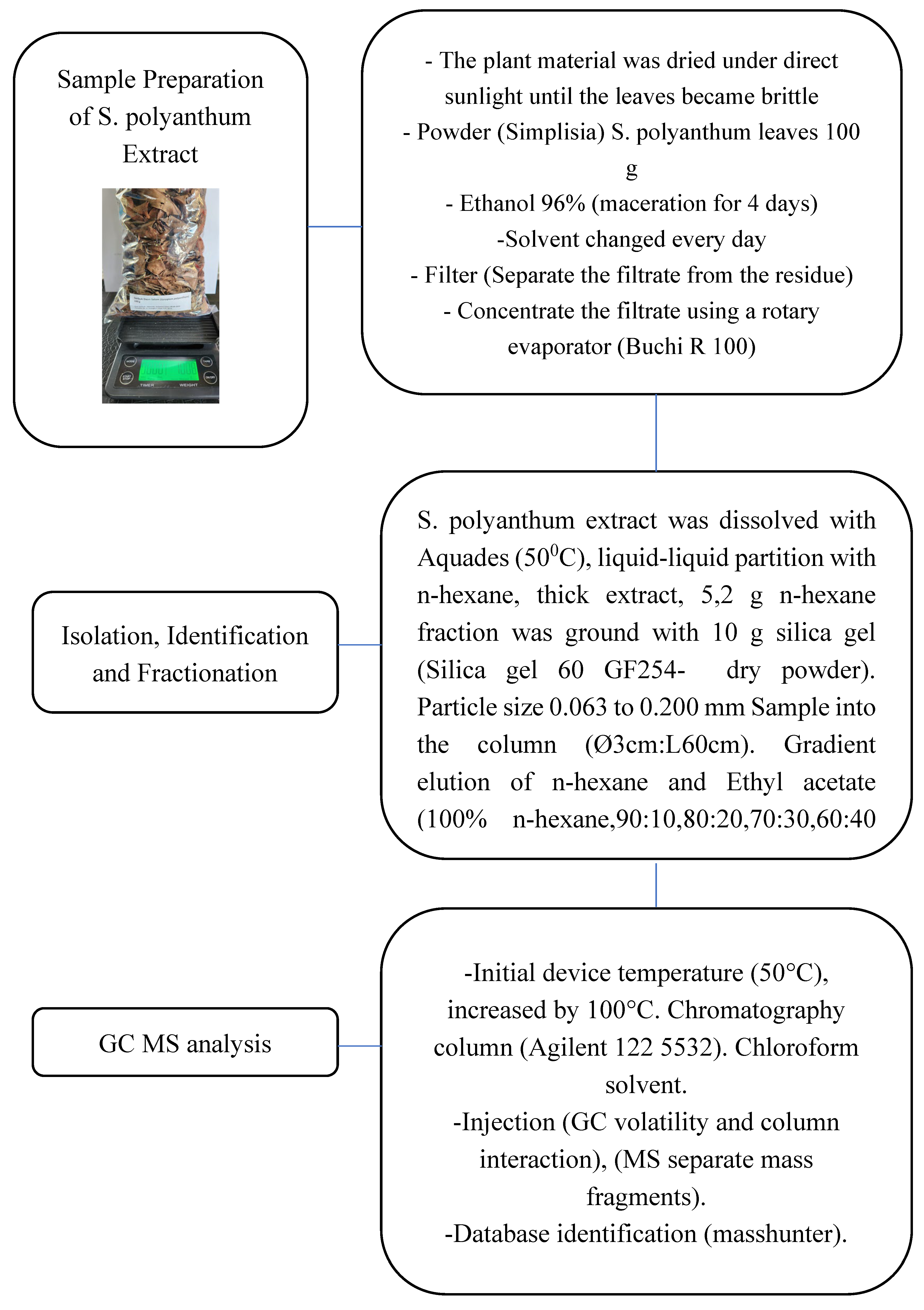
2.2. Materials, Reagents, and Instruments
| Category | Item | Grade or specification | Notes |
|---|---|---|---|
| Plant material | Syzygium polyanthum leaves | Fresh leaves, field collected | Collected in Paniki Bawah, Mapanget, Manado |
| Solvents | Ethanol | 96% technical grade | Used for maceration |
| Solvents | n Hexane | Technical grade | Used for liquid liquid partitioning |
| Solvents | Ethyl acetate | Technical grade | Used for liquid liquid partitioning |
| Solvents | Distilled water | Laboratory grade | Used to dissolve crude extract before partitioning |
| Solvent for GC MS | Chloroform | Laboratory solvent | Used to dissolve samples prior to injection |
| Stationary phase | Silica gel 60 GF254 | Particle size 0.063 to 0.200 mm | Used for dry loading and chromatographic handling |
| TLC plates | Silica gel plates | Commercial plates | Used to monitor fractions when applicable |
| Instrument | GC MS | Shimadzu QP 5050A | Used for compound profiling |
| Instrument | Rotary evaporator | Buchi R 100 | Used to concentrate extracts and fractions |
| Instrument | Oven | Memmert | Used for drying steps as needed |
| Instrument | Analytical balance | Ohaus | Used for accurate weighing |
2.3. Plant Material Collection and Sample Preparation
2.4. Ethanolic Extraction by Maceration
2.5. Liquid Liquid Partitioning and Preparation of the n Hexane Fraction
2.6. Sample Preparation for Chromatographic Handling Prior to GC MS
2.7. GC MS Analysis and Compound Identification
3. Results and Discussion
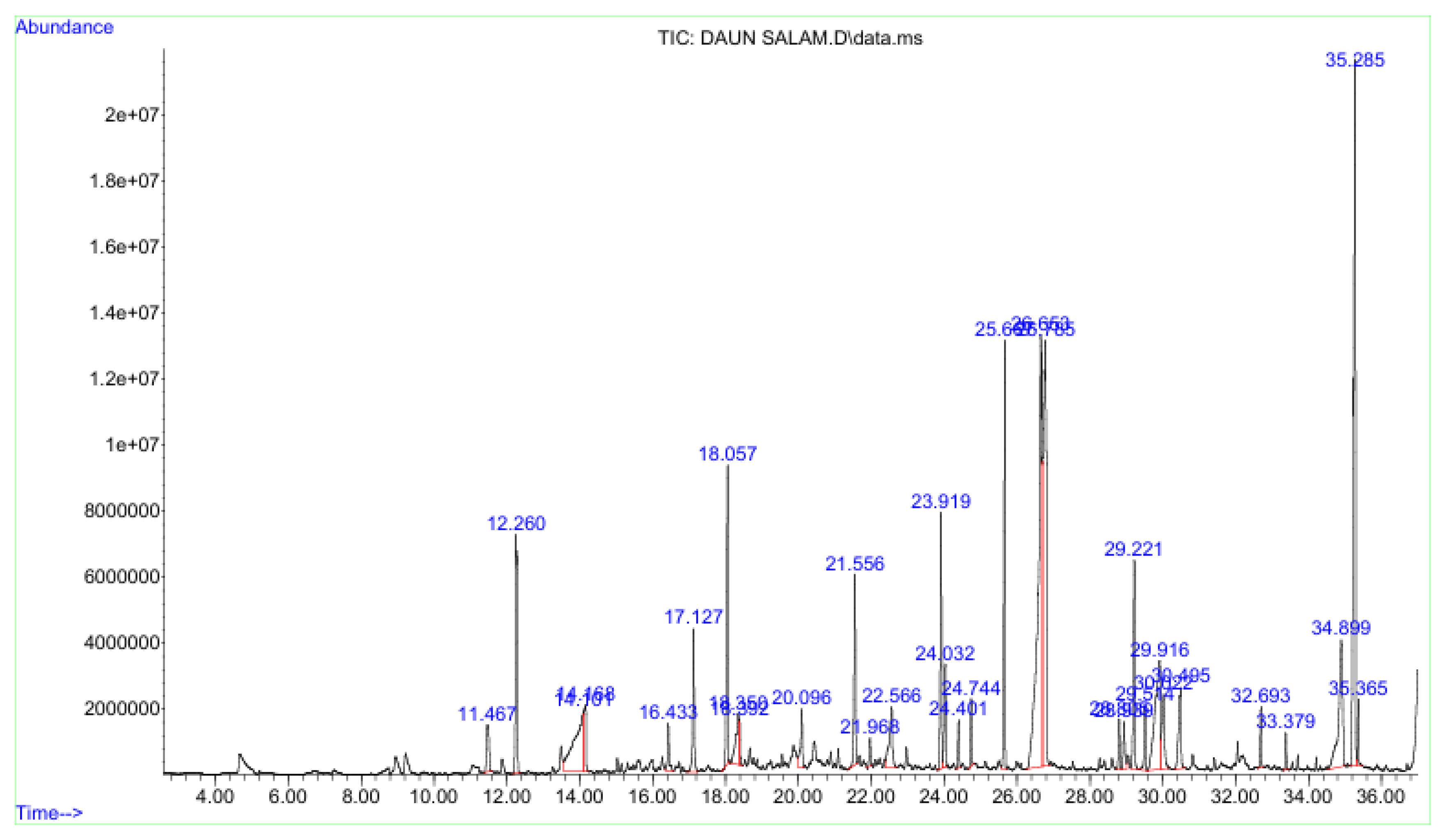
3.1. GC–MS Chromatographic Profile of the n-Hexane Fraction
3.2. Tentative Identification of Volatile and Semi-Volatile Constituents
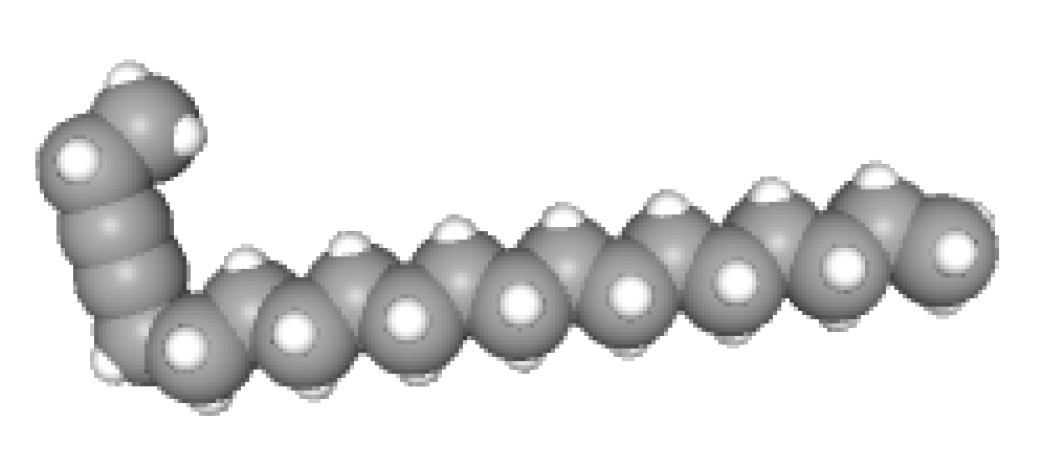
Tridecanedial
3.3. Chemical-Class Interpretation of the Identified Profile
3.4. Discussion of Key Constituents and Bioactivity Relevance
3.5. Data-Quality Considerations and Confidence of Identification
3.6. Positioning Relative to Recent Syzygium polyanthum Literature
4. Conclusion
Acknowledgments
References
- Ahuayo, M. A. M.; Berasain, M. D. M.; Luckie, R. A. M.; Orozco, D. G. Geographical and temporal variability in the extraction efficiency of bioactive metabolites from Cosmos bipinnatus. Applied Food Research 2025, 5(2), 101098. [Google Scholar] [CrossRef]
- Amponsah, I. K.; Owusu, F. Y.; Sarkodie, J. A.; Nkrumah, D.; Kontoh, E. Q.; Akotey, A. N. A. O.; Baah, K. A. Application of the Herbal Marker Ranking System (Herb MaRS) affords chemical markers for the standardization of medicinal plants used for male vitality herbal products. Phytomedicine Plus 2025, 5(4), 100864. [Google Scholar] [CrossRef]
- Anuar, T. A. F. T.; Ismail, A.; Mohamed Suffian, I. F.; Abdul Hamid, A. A.; Arzmi, M. H.; Omar, M. N. LCMS dataset on compounds in Syzygium polyanthum (Wight) Walp. leaves variant from the East coast of Peninsular Malaysia. Data in Brief 2021, 39, 107485. [Google Scholar] [CrossRef]
- Ayyal Salman, H.; Yaakop, A. S.; Al-Rimawi, F.; Ahmad Makhtar, A. M.; Mousa, M.; Semreen, M. H.; Alharbi, N. S. Ephedra alte extracts’ GC-MS profiles and antimicrobial activity against multidrug-resistant pathogens (MRSA). Heliyon 2024, 10(5), e27051. [Google Scholar] [CrossRef] [PubMed]
- Benkhaira, N.; Zouine, N.; Fadil, M.; Ibnsouda Koraichi, S.; El Hachlafi, N.; Jeddi, M.; Lachkar, M.; Fikri-Benbrahim, K. Application of mixture design for the optimum antibacterial action of chemically-analyzed essential oils and investigation of the antiadhesion ability of their optimal mixtures on 3D printing material. Bioprinting 2023, 34, e00299. [Google Scholar] [CrossRef]
- Cain, C. N.; Synovec, R. E. Enhancing gas chromatography-mass spectrometry resolution and pure analyte discovery using intra-chromatogram elution profile matching. Talanta 2024, 278, 126453. [Google Scholar] [CrossRef]
- Chen, E.; Ma, Z.; Geng, X.; Li, Q.; Zheng, F.; Sun, J.; Sun, B. Comprehensive two-dimensional gas chromatography technique and its applications in fermented food aroma analysis: A review. Food Bioscience 2024, 62, 105473. [Google Scholar] [CrossRef]
- de Araújo, A. N. V.; de Souza, E. L.; Nascimento, D. dos S.; Alves, J. M.; Brito Sampaio, K.; da Silva, S. R. F.; de Brito Alves, J. L.; de Albuquerque, T. M. R. Revisiting the nutritional and functional value and health-promoting potential of Syzygium species. Journal of Functional Foods 2024, 118, 106265. [Google Scholar] [CrossRef]
- El Hachlafi, N.; Benkhaira, N.; Al-Mijalli, S. H.; Mrabti, H. N.; Abdnim, R.; Abdallah, E. M.; Jeddi, M.; Bnouham, M.; Lee, L.-H.; Ardianto, C.; Ming, L. C.; Bouyahya, A.; Fikri-Benbrahim, K. Phytochemical analysis and evaluation of antimicrobial, antioxidant, and antidiabetic activities of essential oils from Moroccan medicinal plants: Mentha suaveolens, Lavandula stoechas, and Ammi visnaga. Biomedicine & Pharmacotherapy 2023, 164, 114937. [Google Scholar] [CrossRef]
- Ferretti, M.; Spiezia, G.; Scalzo, M. Lo; Todeschini, V.; Lingua, G.; Conterosito, E.; Guerrieri, E.; Gianotti, V. Accurate and robust method for plant volatilome analysis by GC-MS. Talanta Open 2025, 12, 100578. [Google Scholar] [CrossRef]
- Hasby, H.; Astuti, L.; Rizali, A. Bioactivities of bay leaf (Syzygium polyanthum) fumigant tablets againts Araecerus fasciculatus (De Geer) (Coleoptera: Anthribidae). Jurnal Entomologi Indonesia 2024, 21, 118–129. [Google Scholar] [CrossRef]
- Huynh, T. L.; Manh, T. D.; Phuong Nguyen, L. T.; Vu, D. T.; Huu Nguyen, K. D.; Duong Ngo, K. L. Corrosion mitigation of carbon steel in sulfuric and hydrochloric acid solutions by Syzygium polyanthum (Wight) Walp. leaf extract. Results in Surfaces and Interfaces 2024, 17, 100318. [Google Scholar] [CrossRef]
- Iskandi, S.; Fauziah, F.; Oktavia, S. Review: Antibacterial Activity of Syzygium polyanthum. International Journal of Pharmaceutical Sciences and Medicine 2021, 6, 182–186. [Google Scholar] [CrossRef]
- Julizan, N.; Ishmayana, S.; Zainuddin, A.; Hùng, P.; Dikdik, K. Potential of Syzygnium polyanthum as Natural Food Preservative: A Review. Foods 2023, 12, 2275. [Google Scholar] [CrossRef]
- Kebede, B. H.; Them, R. L.; Mengistu, B. A. Effects of extraction solvents on phenolic contents, in vitro antioxidant, and antimicrobial activities of roselle (Hibiscus sabdariffa L.) Calyx. Food Chemistry Advances 2025, 9, 101119. [Google Scholar] [CrossRef]
- Khan, S.; Rukayadi, Y.; Jaafar, A. H.; Ahmad, N. H. Antibacterial potential of silver nanoparticles (SP-AgNPs) synthesized from Syzygium polyanthum (Wight) Walp. against selected foodborne pathogens. Heliyon 2023, 9(12), e22771. [Google Scholar] [CrossRef]
- Laldinsangi, C. The therapeutic potential of Houttuynia cordata: A current review. Heliyon 2022, 8(8), e10386. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Huang, X.; Li, C.; Kong, B.; Cao, C.; Sun, F.; Zhang, H.; Liu, Q.; Shen, L. Effect of different natural antioxidants on the quality promotion of pork chip snacks during storage as revealed by lipid profiles and volatile flavor compounds. Food Chemistry 2025, 478, 143716. [Google Scholar] [CrossRef]
- Meng, X.-W.; Wang, Q.-Q.; Zhang, N.; Guo, F.-M.; Li, J.-R.; Zhu, Q.; Liu, R.-H.; Zhu, W.-F. Anti-inflammatory mechanisms of flavonoids in Pueraria lobata: Immune cell regulation and molecular mechanisms. Journal of Functional Foods 2025, 131, 106954. [Google Scholar] [CrossRef]
- Najib, A.; Olli, A.; Puspitasari, Y. Optimasi Ekstrak Daun Salam (Syzygium Polyanthum) Menggunakan Metode Konvensional dan Green Extraction Serta Profil Kimia dan Potensi Antioksidannya. Jurnal Mandala Pharmacon Indonesia 2025, 11, 55–65. [Google Scholar] [CrossRef]
- Nguyen, K. D. H.; Manh, T. D.; Nguyen, L. T. P.; Vu, D. T.; Duong Ngo, K. L. Syzygium polyanthum (Wight) Walp. leaf extract as a sustainable corrosion inhibitor for carbon steel in hydrochloric acidic environment. Journal of Industrial and Engineering Chemistry 2025, 143, 468–487. [Google Scholar] [CrossRef]
- Oladele, O. T.; Oladele, J. O.; Ajayi, E. I. O.; Alabi, K. E.; Oyeleke, O. M.; Atolagbe, O. S.; Olowookere, B. D.; Bamigboye, M. O. Bioactive composition and protective properties of Talium triangulare in dextran sodium sulphate-induced ulcerative colitis in rats. Pharmacological Research - Modern Chinese Medicine 2024, 10, 100344. [Google Scholar] [CrossRef]
- Onyedikachi, U. B.; Nkwocha, C. C.; Ejiofor, E.; Nnanna, C. C. Investigation of chemical constituents, antioxidant, anti-inflammatory and nutritional properties of oil of Persea americana (Avocado) seeds. Food Chemistry Advances 2024, 5, 100770. [Google Scholar] [CrossRef]
- Parvez, S.; Karole, A.; Mudavath, S. L. Fabrication, physicochemical characterization and In vitro anticancer activity of nerolidol encapsulated solid lipid nanoparticles in human colorectal cell line. Colloids and Surfaces B: Biointerfaces 2022, 215, 112520. [Google Scholar] [CrossRef]
- Pisoschi, A. M.; Pop, A.; Iordache, F.; Stanca, L.; Predoi, G.; Serban, A. I. Oxidative stress mitigation by antioxidants - An overview on their chemistry and influences on health status. European Journal of Medicinal Chemistry 2021, 209, 112891. [Google Scholar] [CrossRef] [PubMed]
- Saadellaoui, W.; Kahlaoui, S.; Hcini, K.; Haddada, A.; Sleimi, N.; Ascrizzi, R.; Flamini, G.; Harzallah-Skhiri, F.; Stambouli-Essassi, S. Profiles of the Headspace Volatile Organic and Essential Oil Compounds from the Tunisian Cardaria draba (L.) Desv. and Its Leaf and Stem Epidermal Micromorphology. Phyton-International Journal of Experimental Botany 2024, 93(4), 725–744. [Google Scholar] [CrossRef]
- Shen, T.; Chen, L.; Liu, Y.; Shi, S.; Liu, Z.; Cai, K.; Liao, C.; Wang, C. Decanoic acid modification enhances the antibacterial activity of PMAP-23RI-Dec. European Journal of Pharmaceutical Sciences 2021, 157, 105609. [Google Scholar] [CrossRef]
- Srisittiratkul, P.; Limsuvan, S.; Pattanapholkornsakul, S.; Akarasereenont, P. Optimizing herbal drug use through multivariable standardization and precision approaches. Phytomedicine Plus 2025, 5(4), 100907. [Google Scholar] [CrossRef]
- Sunarsono, H.; Abral, H.; Mahardika, M.; Railis, R. M.; Sapuan, S. M.; Azka, M. A.; Rahmad; Arrafi, M. R. Enhancing mechanical properties of durian (Durio ziberthinus murr.) seed starch biopolymer filled with Indonesian bay leaf (Syzygium polyanthum) extract. Biomass and Bioenergy 2025, 202, 108159. [Google Scholar] [CrossRef]
- Sunarsono, H.; Abral, H.; Pratoto, A.; Gulo, E. F.; Arrafi, M. R.; Sibarani, E. I.; Railis, R. M.; Mahardika, M.; Rushdan, A. I.; Wahit, M. U.; Handayani, D.; Sandrawati, N.; Rahmadiawan, D.; Sugiarti, E.; Muslimin, A. N.; Shi, S.-C. Indonesian bay leaf (Syzygium polyanthum Wight) extract as a natural additive for UV and red light-blocking polyvinyl alcohol films with enhanced mechanical properties. Case Studies in Chemical and Environmental Engineering 2025, 11, 101237. [Google Scholar] [CrossRef]
- Syabana, M. A.; Yuliana, N. D.; Batubara, I.; Fardiaz, D. α-glucosidase inhibitors from Syzygium polyanthum (Wight) Walp leaves as revealed by metabolomics and in silico approaches. Journal of Ethnopharmacology 2022, 282, 114618. [Google Scholar] [CrossRef]
- Vignesh, A.; Amal, T. C.; Sarvalingam, A.; Vasanth, K. A review on the influence of nutraceuticals and functional foods on health. Food Chemistry Advances 2024, 5, 100749. [Google Scholar] [CrossRef]
- Wei, P.; Luo, Q.; Hou, Y.; Zhao, F.; Li, F.; Meng, Q. Houttuynia Cordata Thunb.: A comprehensive review of traditional applications, phytochemistry, pharmacology and safety. Phytomedicine 2024, 123, 155195. [Google Scholar] [CrossRef]
- Widyawati, T.; Nor Adlin, Y.; Bello, I.; Abdullah, M.; Ahmad, M. Bioactivity-Guided Fractionation and Identification of Antidiabetic Compound of Syzygium polyanthum (Wight.)’s Leaf Extract in Streptozotocin-Induced Diabetic Rat Model. Molecules 2022, 27, 6814. [Google Scholar] [CrossRef]
- Xi, B.-N.; Zhang, J.-J.; Xu, X.; Li, C.; Shu, Y.; Zhang, Y.; Shi, X.; Shen, Y. Characterization and metabolism pathway of volatile compounds in walnut oil obtained from various ripening stages via HS-GC-IMS and HS-SPME-GC–MS. Food Chemistry 2024, 435, 137547. [Google Scholar] [CrossRef]
- Xiang, X.; Kwame, A. W.; Qing, Y.; Li, S.; Wang, M.; Ren, J. Natural antioxidants inhibit oxidative stress-induced changes in the morphology and motility of cells. Food Bioscience 2023, 52, 102442. [Google Scholar] [CrossRef]
- Yenduri, S.; P, R.; N. P., K. Recent advances in analytical methods and instrumentation for natural antioxidants analysis. Talanta Open 2026, 13, 100602. [Google Scholar] [CrossRef]
- Yildiz, A. Y.; Öztekin, S.; Anaya, K. Effects of plant-derived antioxidants to the oxidative stability of edible oils under thermal and storage conditions: Benefits, challenges and sustainable solutions. Food Chemistry 2025, 479, 143752. [Google Scholar] [CrossRef] [PubMed]
| No. | Retention time (min) | Tentative compound identification |
|---|---|---|
| 1 | 11.467 | 2-Undecanone |
| 2 | 12.260 | Decanoic acid, methyl ester |
| 3 | 14.101 | n-Decanoic acid |
| 4 | 16.433 | 2-Tridecanone |
| 5 | 17.127 | Dodecanoic acid, methyl ester |
| 6 | 18.057 | 1,6,10-Dodecatrien-3-ol, 3,7,11-trimethyl (nerolidol) |
| 7 | 18.35 | Dodecanoic acid |
| 8 | 20.096 | Heptadecanoic acid, 3-oxo-, methyl ester |
| 9 | 20.096 | Octanoic acid, 3-oxo-, methyl ester |
| 10 | 20.096 | Propanoic acid, methyl(tetramethylene)silyl ester |
| 11 | 21.968 | Cyclohexene, 3-pentyl |
| 12 | 21.968 | 1-Eicosyne |
| 13 | 21.968 | 3-Nonen-2-one |
| 14 | 22.566 | Tetradecanoic acid |
| 15 | 23.919 | Bicyclo [3.1.1]heptane, 2,6,6-trimethyl-, (1α,2β,5α) |
| 16 | 23.919 | Bicyclo [3.1.1]heptane, 2,6,6-trimethyl-, [1R-(1α,2β,5α)] |
| 17 | 23.919 | Tridecanedial |
| 18 | 24.032 | 2-Pentadecanone, 6,10,14-trimethyl (hexahydrofarnesyl acetone, phytone) |
| 19 | 24.401 | Cyclohexanol, 1-ethynyl- |
| 20 | 24.401 | Cyclohexene, 1-(2-methylpropyl)- |
| Chemical class | Count (out of 20) | Examples from Table 2 |
|---|---|---|
| Fatty acid methyl esters | 4 | Decanoic acid, methyl ester; Dodecanoic acid, methyl ester |
| Fatty acids | 3 | n-Decanoic acid; Dodecanoic acid; Tetradecanoic acid |
| Ketones | 3 | 2-Undecanone; 2-Tridecanone; 3-Nonen-2-one |
| Alcohols and terpenoid alcohols | 2 | Nerolidol; Cyclohexanol, 1-ethynyl- |
| Alkene hydrocarbons | 2 | Cyclohexene, 3-pentyl; Cyclohexene, 1-(2-methylpropyl)- |
| Terpene-like hydrocarbons | 2 | Bicyclo [3.1.1]heptane derivatives |
| Aldehydes | 1 | Tridecanedial |
| Alkyne hydrocarbons | 1 | 1-Eicosyne |
| Silylated derivative (possible artifact or derivatization-related hit) | 1 | Propanoic acid, methyl(tetramethylene)silyl ester |
| Other | 1 | Assigned compound at the same retention-time cluster |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




