Submitted:
31 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Cell Culture
Preparation of Radiopharmaceuticals
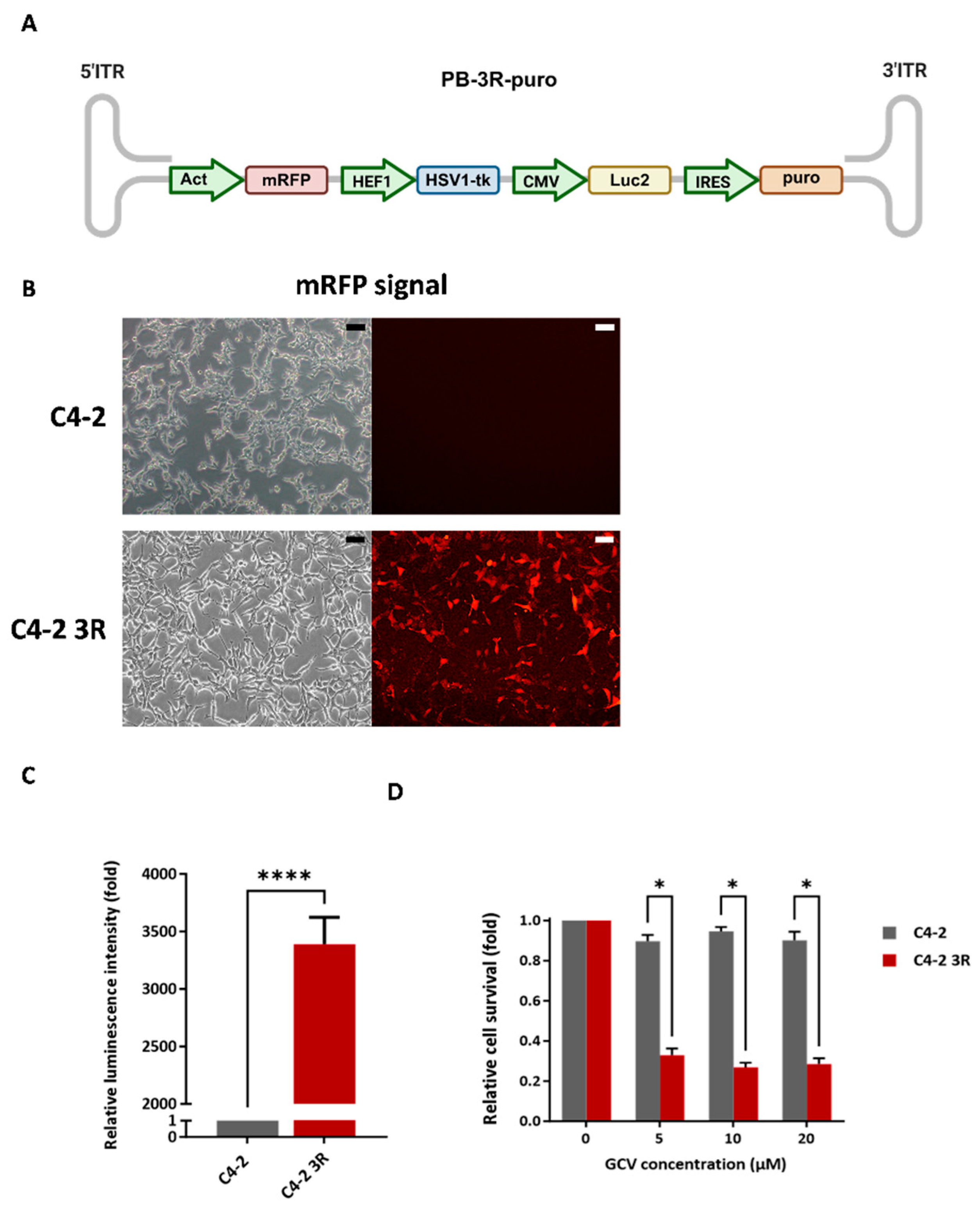
Transfection of PiggyBac Transposon Constructs and Cell Sorting
Luciferase Reporter Gene Assay
Cell Viability Assay
X-Rays Source
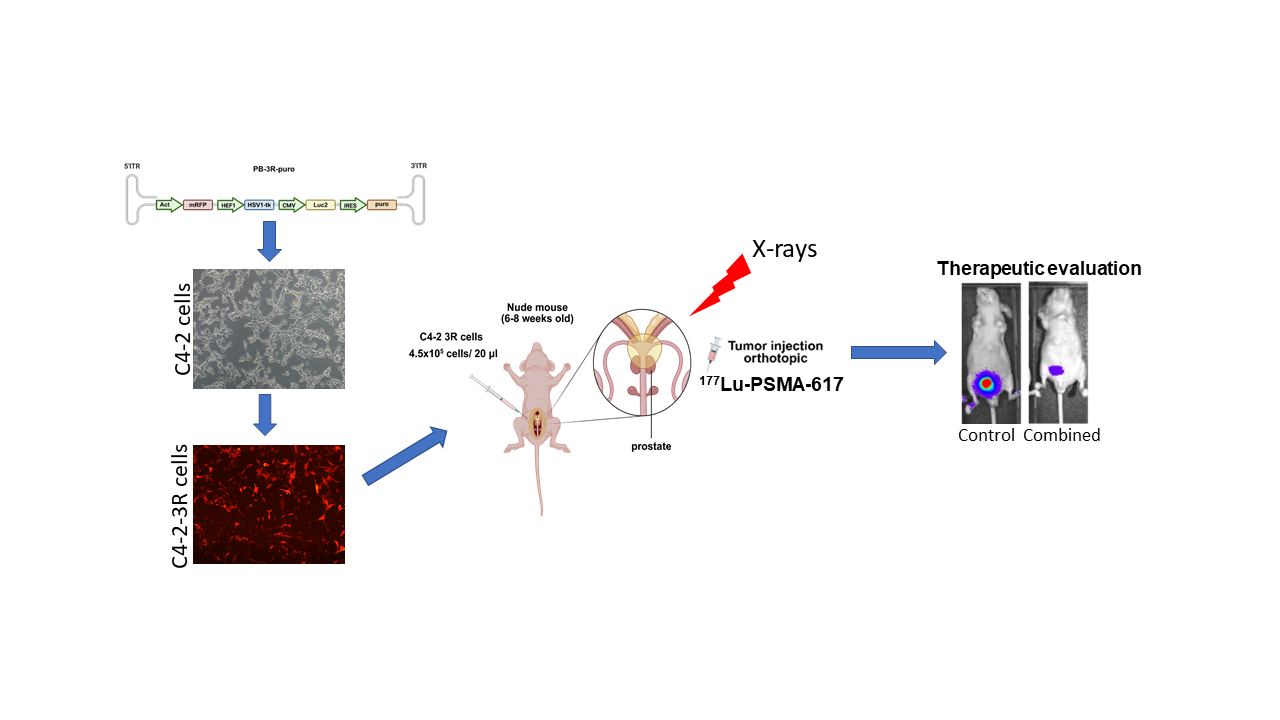
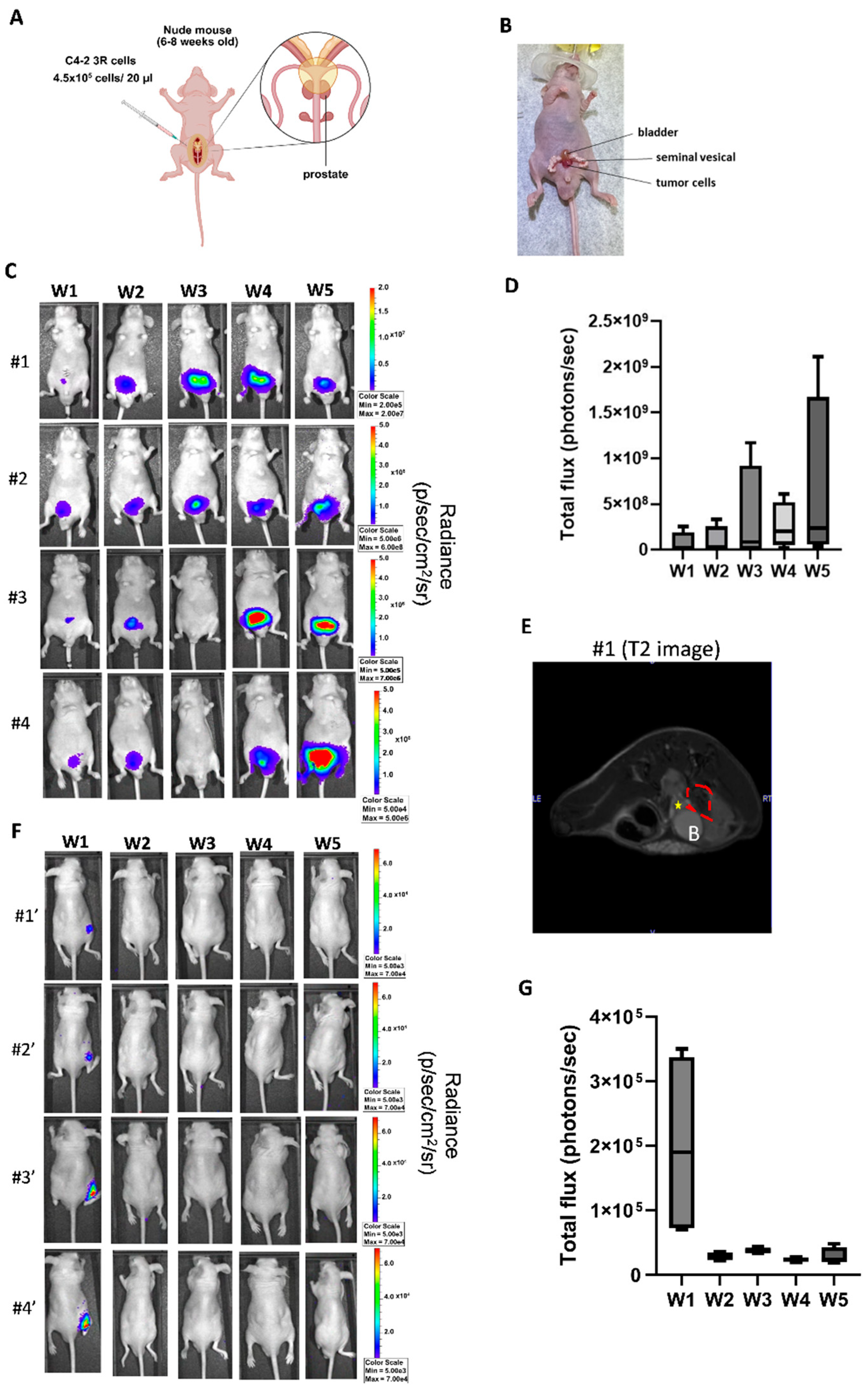
Establishment of Tumor Models in Small Animals
Bioluminescence Imaging
Micro-Magnetic Resonance Imaging (MRI) for Small Animals
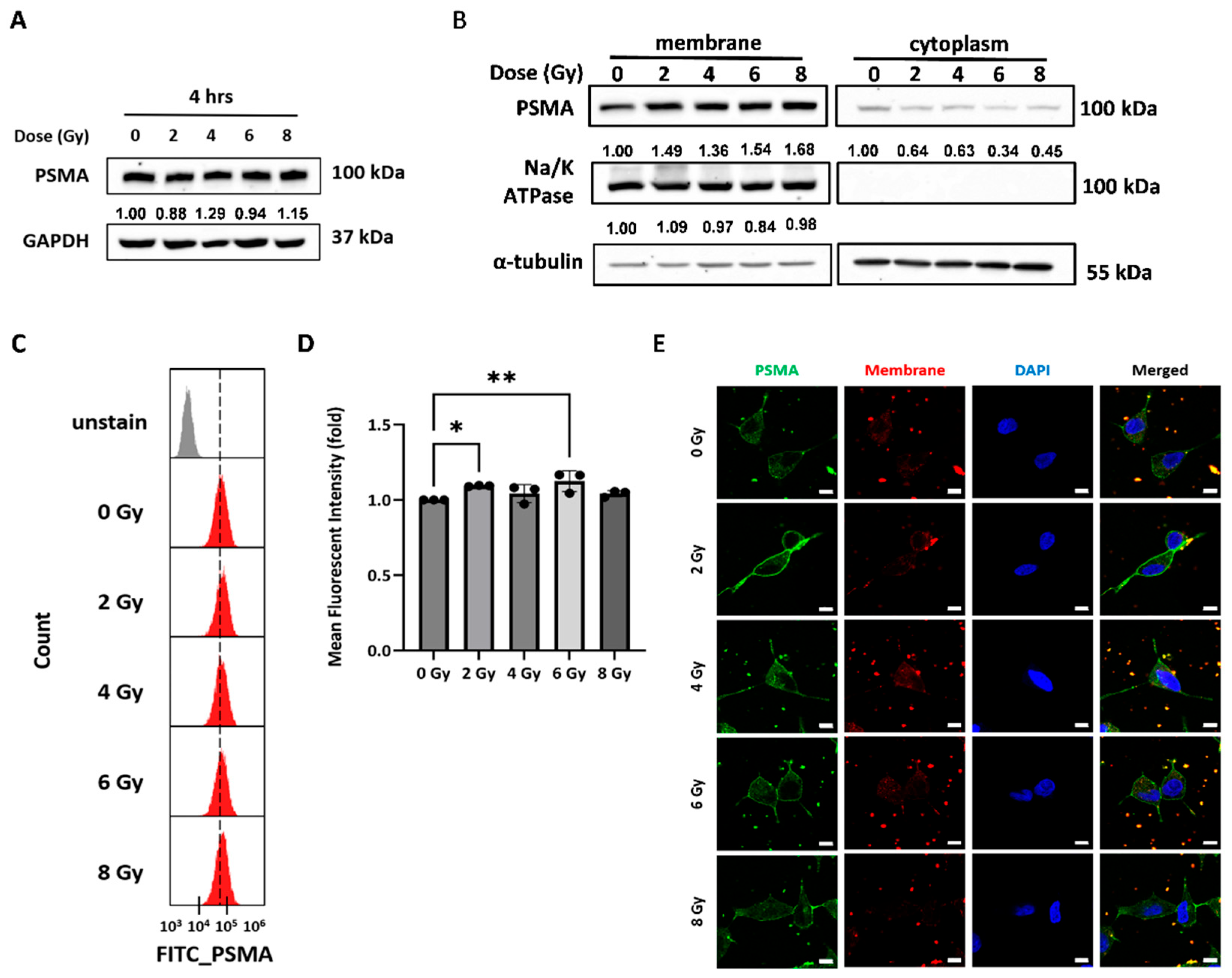
Preparation of Membrane Proteins
Western Blot Analysis
Flow Cytometric Analysis of PSMA
Immunofluorescence Microscopy
Radiation Exposure and Radioligand Therapy on Orthotopic Tumors
Positron Emission Tomography/Computed Tomography (PET/CT) for Small Animals
Hematoxylin and Eosin (H&E) Staining
Statistical Analysis
Results
Establishment and Validation of PiggyBac Transposon System Mediated Delivery of Triple Reporter Genes into C4-2 Cells
RGI of Tumor Formation in Orthotopic Model and Subcutaneous Xenograft Model Using C4-2 3R Cells
Assessment of PSMA Expression After X-Rays Irradiation in C4-2 Cells
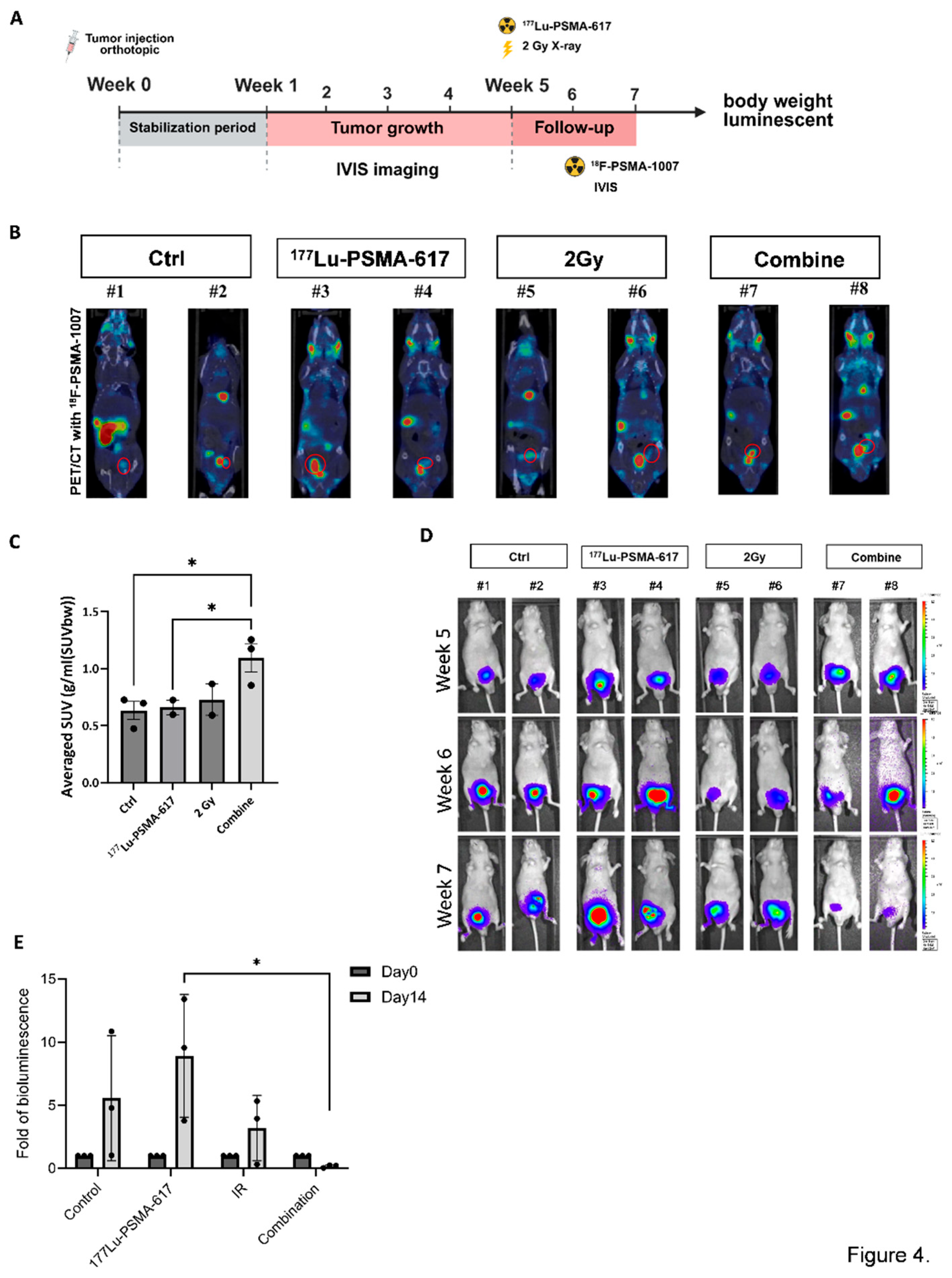
Potential of C4-2 3R Cells on Evaluation of Combined X-Ray EBRT and 177Lu-PSMA-617 Radioligand Therapy In Vivo
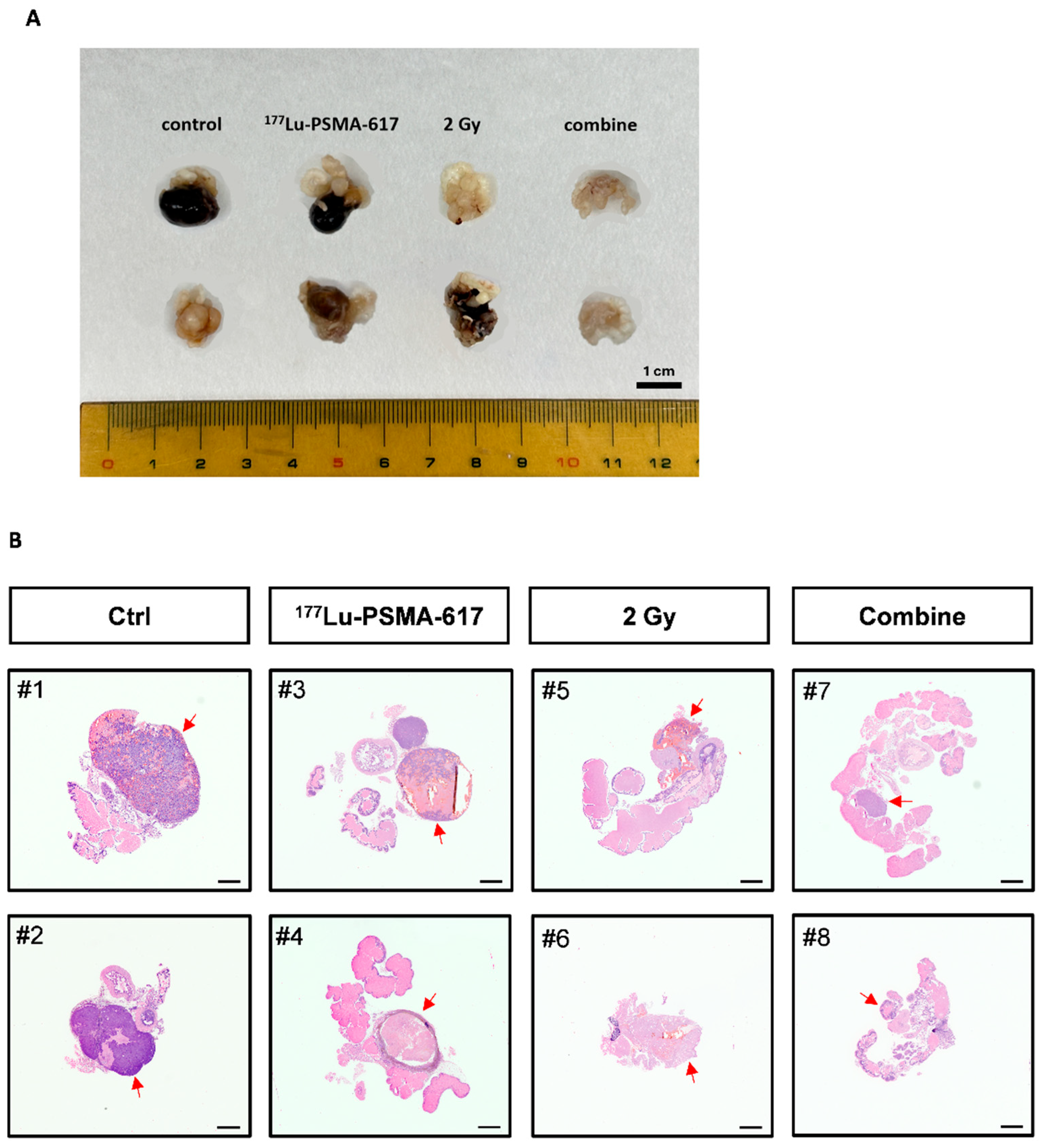
Dissection of C4-2 3R Cells Formed Orthotopic Tumor Tissue with Different Treatments
Discussion
Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Rebello, R.J.; Oing, C.; Knudsen, K.E.; Loeb, S.; Johnson, D.C.; Reiter, R.E.; Gillessen, S.; Van der Kwast, T.; Bristow, R.G. Prostate cancer. Nat Rev Dis Primers 2021, 7, 9. [CrossRef]
- Horoszewicz, J.S.; Leong, S.S.; Kawinski, E.; Karr, J.P.; Rosenthal, H.; Chu, T.M.; Mirand, E.A.; Murphy, G.P. LNCaP model of human prostatic carcinoma. Cancer Res 1983, 43, 1809-1818. [PubMed]
- Shaikh, F.A.; Kurtys, E.; Kubassova, O.; Roettger, D. Reporter gene imaging and its role in imaging-based drug development. Drug Discov Today 2020, 25, 582-592. [CrossRef]
- Nayerossadat, N.; Maedeh, T.; Ali, P.A. Viral and nonviral delivery systems for gene delivery. Adv Biomed Res 2012, 1, 27. [CrossRef]
- Taghdiri, M.; Mussolino, C. Viral and Non-Viral Systems to Deliver Gene Therapeutics to Clinical Targets. Int J Mol Sci 2024, 25. [CrossRef]
- Najafi, S.; Rahimpour, A.; Ahmadieh, H.; Rezaei Kanavi, M.; Maleki Tehrani, M.; Suri, F.; Ranjbari, J. The effect of enhancers on the lentiviral transduction efficiency in the human RPE cells: Insights for advancing retinal gene therapies. Biochem Biophys Rep 2025, 42, 102010. [CrossRef]
- Lin, P.; Correa, D.; Lin, Y.; Caplan, A.I. Polybrene inhibits human mesenchymal stem cell proliferation during lentiviral transduction. PLoS One 2011, 6, e23891. [CrossRef]
- Mellott, A.J.; Forrest, M.L.; Detamore, M.S. Physical non-viral gene delivery methods for tissue engineering. Ann Biomed Eng 2013, 41, 446-468. [CrossRef] [PubMed]
- Wang, K.; Huang, Q.; Qiu, F.; Sui, M. Non-viral Delivery Systems for the Application in p53 Cancer Gene Therapy. Curr Med Chem 2015, 22, 4118-4136. [CrossRef]
- Aronovich, E.L.; Hyland, K.A.; Hall, B.C.; Bell, J.B.; Olson, E.R.; Rusten, M.U.; Hunter, D.W.; Ellinwood, N.M.; McIvor, R.S.; Hackett, P.B. Prolonged Expression of Secreted Enzymes in Dogs After Liver-Directed Delivery of Sleeping Beauty Transposons: Implications for Non-Viral Gene Therapy of Systemic Disease. Hum Gene Ther 2017, 28, 551-564. [CrossRef] [PubMed]
- Claeys Bouuaert, C.; Chalmers, R.M. Gene therapy vectors: the prospects and potentials of the cut-and-paste transposons. Genetica 2010, 138, 473-484. [CrossRef]
- Feschotte, C. The piggyBac transposon holds promise for human gene therapy. Proc Natl Acad Sci U S A 2006, 103, 14981-14982. [CrossRef]
- Wilson, M.H.; Coates, C.J.; George, A.L., Jr. PiggyBac transposon-mediated gene transfer in human cells. Mol Ther 2007, 15, 139-145. [CrossRef]
- Chen, Y.L.; Wang, S.Y.; Liu, R.S.; Wang, H.E.; Chen, J.C.; Chiou, S.H.; Chang, C.A.; Lin, L.T.; Tan, D.T.; Lee, Y.J. Remnant living cells that escape cell loss in late-stage tumors exhibit cancer stem cell-like characteristics. Cell Death Dis 2012, 3, e399. [CrossRef] [PubMed]
- Lin, M.Y.; Wang, C.Y.; Chan, Y.H.; Su, S.P.; Chiang, H.K.; Yang, M.H.; Lee, Y.J. The Emergence of Tumor-Initiating Cells in an Advanced Hypopharyngeal Tumor Model Exhibits Enhanced Angiogenesis and Nuclear Factor Erythroid 2-Related Factor 2-Associated Antioxidant Effects. Antioxid Redox Signal 2024, 41, 505-521. [CrossRef]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med 2021, 385, 1091-1103. [CrossRef]
- Algin, E.; Okudan, B.; Acikgoz, Y.; Sayan, H.; Bal, O.; Seven, B. Impact of 68Ga-PSMA PET/CT on Survival and Management in Prostate Cancer. Curr Med Imaging 2024, 20, e15734056276494. [CrossRef] [PubMed]
- Teunissen, F.R.; Oprea-Lager, D.E.; Peters, S.M.B.; Smeenk, R.J.; Heskamp, S.; Bussink, J. Current Developments in Combining External-Beam Radiotherapy and (177)Lu-Labeled PSMA Ligands for Prostate Cancer Treatment. J Nucl Med 2025. [CrossRef] [PubMed]
- Arbuznikova, D.; Klotsotyra, A.; Uhlmann, L.; Domogalla, L.C.; Steinacker, N.; Mix, M.; Niedermann, G.; Spohn, S.K.B.; Freitag, M.T.; Grosu, A.L.; et al. Exploring the role of combined external beam radiotherapy and targeted radioligand therapy with [(177)Lu]Lu-PSMA-617 for prostate cancer - from bench to bedside. Theranostics 2024, 14, 2560-2572. [CrossRef]
- Kleinendorst, S.C.; Oosterwijk, E.; Bussink, J.; Westdorp, H.; Konijnenberg, M.W.; Heskamp, S. Combining Targeted Radionuclide Therapy and Immune Checkpoint Inhibition for Cancer Treatment. Clin Cancer Res 2022, 28, 3652-3657. [CrossRef]
- Ruigrok, E.A.M.; van Vliet, N.; Dalm, S.U.; de Blois, E.; van Gent, D.C.; Haeck, J.; de Ridder, C.; Stuurman, D.; Konijnenberg, M.W.; van Weerden, W.M.; et al. Extensive preclinical evaluation of lutetium-177-labeled PSMA-specific tracers for prostate cancer radionuclide therapy. Eur J Nucl Med Mol Imaging 2021, 48, 1339-1350. [CrossRef]
- Meyer, C.; Stuparu, A.; Lueckerath, K.; Calais, J.; Czernin, J.; Slavik, R.; Dahlbom, M. Tandem Isotope Therapy with (225)Ac- and (177)Lu-PSMA-617 in a Murine Model of Prostate Cancer. J Nucl Med 2023, 64, 1772-1778. [CrossRef]
- Kristiansson, A.; Orbom, A.; Ahlstedt, J.; Karlsson, H.; Zedan, W.; Gram, M.; Akerstrom, B.; Strand, S.E.; Altai, M.; Strand, J.; et al. (177)Lu-PSMA-617 Therapy in Mice, with or without the Antioxidant alpha(1)-Microglobulin (A1M), Including Kidney Damage Assessment Using (99m)Tc-MAG3 Imaging. Biomolecules 2021, 11. [CrossRef]
- Chen, Y.-L.; Wang, Y.-C.; Hou, K.-Y.; Lin, M.-Y.; Lin, Y.-C.; Chuang, H.-Y.; Lee, Y.-J. Use of piggyBac Transposon System Constructed Murine Breast Cancer Model for Reporter Gene Imaging and Characterization of Metastatic Tumor Cells. Journal of Medical and Biological Engineering 2022, 42, 341-350. [CrossRef]
- Kang, Y.; Zhang, X.Y.; Jiang, W.; Wu, C.Q.; Chen, C.M.; Gu, J.R.; Zheng, Y.F.; Xu, C.J. The piggyBac transposon is an integrating non-viral gene transfer vector that enhances the efficiency of GDEPT. Cell Biol Int 2009, 33, 509-515. [CrossRef] [PubMed]
- Pavese, J.; Ogden, I.M.; Bergan, R.C. An orthotopic murine model of human prostate cancer metastasis. J Vis Exp 2013, e50873. [CrossRef]
- Xie, B.X.; Zhang, H.; Yu, L.; Wang, J.; Pang, B.; Wu, R.Q.; Qian, X.L.; Li, S.H.; Shi, Q.G.; Wang, L.L.; et al. The radiation response of androgen-refractory prostate cancer cell line C4-2 derived from androgen-sensitive cell line LNCaP. Asian J Androl 2010, 12, 405-414. [CrossRef] [PubMed]
- Lim, D.J.; Liu, X.L.; Sutkowski, D.M.; Braun, E.J.; Lee, C.; Kozlowski, J.M. Growth of an androgen-sensitive human prostate cancer cell line, LNCaP, in nude mice. Prostate 1993, 22, 109-118. [CrossRef]
- Jennbacken, K.; Gustavsson, H.; Tesan, T.; Horn, M.; Vallbo, C.; Welen, K.; Damber, J.E. The prostatic environment suppresses growth of androgen-independent prostate cancer xenografts: an effect influenced by testosterone. Prostate 2009, 69, 1164-1175. [CrossRef]
- Saar, M.; Korbel, C.; Linxweiler, J.; Jung, V.; Kamradt, J.; Hasenfus, A.; Stockle, M.; Unteregger, G.; Menger, M.D. Orthotopic tumorgrafts in nude mice: A new method to study human prostate cancer. Prostate 2015, 75, 1526-1537. [CrossRef]
- Liu, W.; Zhu, Y.; Ye, L.; Zhu, Y.; Wang, Y. Establishment of an orthotopic prostate cancer xenograft mouse model using microscope-guided orthotopic injection of LNCaP cells into the dorsal lobe of the mouse prostate. BMC Cancer 2022, 22, 173. [CrossRef]
- Baba, D.; Çoban, S.; Çalışkan, A.; Senoglu, Y.; Kayıkçı, M.; Tekin, A. Optimizing Prostate Cancer Diagnosis: A Prospective, Randomized Comparison of 12-core vs. 20-core Biopsy for Detection Accuracy and Upgrading Risk. Journal of Urological Surgery 2025. [CrossRef]
- Yamamichi, F.; Matsuoka, T.; Shigemura, K.; Kawabata, M.; Shirakawa, T.; Fujisawa, M. Potential establishment of lung metastatic xenograft model of androgen receptor-positive and androgen-independent prostate cancer (C4-2B). Urology 2012, 80, 951 e951-957. [CrossRef]
- Li, M.; Wang, Y.; Liu, M.; Lan, X. Multimodality reporter gene imaging: Construction strategies and application. Theranostics 2018, 8, 2954-2973. [CrossRef]
- Stuparu, A.D.; Meyer, C.A.L.; Evans-Axelsson, S.L.; Luckerath, K.; Wei, L.H.; Kim, W.; Poddar, S.; Mona, C.E.; Dahlbom, M.; Girgis, M.D.; et al. Targeted alpha therapy in a systemic mouse model of prostate cancer - a feasibility study. Theranostics 2020, 10, 2612-2620. [CrossRef] [PubMed]
- Han, M.; Yu, D.; Song, Q.; Wang, J.; Dong, P.; He, J. Polybrene: Observations on cochlear hair cell necrosis and minimal lentiviral transduction of cochlear hair cells. Neurosci Lett 2015, 600, 164-170. [CrossRef] [PubMed]
- Inada, E.; Saitoh, I.; Watanabe, S.; Aoki, R.; Miura, H.; Ohtsuka, M.; Murakami, T.; Sawami, T.; Yamasaki, Y.; Sato, M. PiggyBac transposon-mediated gene delivery efficiently generates stable transfectants derived from cultured primary human deciduous tooth dental pulp cells (HDDPCs) and HDDPC-derived iPS cells. Int J Oral Sci 2015, 7, 144-154. [CrossRef] [PubMed]
- Ali, F.Z. Prostate Cancer Imaging Beyond PSMA: Applications of GRPR, AR, and Amino Acid Tracers. Diagnostics (Basel) 2025, 15. [CrossRef]
- Sheehan, B.; Neeb, A.; Buroni, L.; Paschalis, A.; Riisnaes, R.; Gurel, B.; Gil, V.; Miranda, S.; Crespo, M.; Guo, C.; et al. Prostate-Specific Membrane Antigen Expression and Response to DNA Damaging Agents in Prostate Cancer. Clin Cancer Res 2022, 28, 3104-3115. [CrossRef]
- Zhang, C.; Liu, J.; Wu, J.; Ranjan, K.; Cui, X.; Wang, X.; Zhang, D.; Zhu, S. Key molecular DNA damage responses of human cells to radiation. Front Cell Dev Biol 2024, 12, 1422520. [CrossRef]
- Mahaney, B.L.; Meek, K.; Lees-Miller, S.P. Repair of ionizing radiation-induced DNA double-strand breaks by non-homologous end-joining. Biochem J 2009, 417, 639-650. [CrossRef]
- Bakht, M.K.; Beltran, H. Biological determinants of PSMA expression, regulation and heterogeneity in prostate cancer. Nat Rev Urol 2025, 22, 26-45. [CrossRef]
- Chen, Q.; Watson, J.T.; Marengo, S.R.; Decker, K.S.; Coleman, I.; Nelson, P.S.; Sikes, R.A. Gene expression in the LNCaP human prostate cancer progression model: progression associated expression in vitro corresponds to expression changes associated with prostate cancer progression in vivo. Cancer Lett 2006, 244, 274-288. [CrossRef] [PubMed]
- Mamouni, K.; Zhang, S.; Li, X.; Chen, Y.; Yang, Y.; Kim, J.; Bartlett, M.G.; Coleman, I.M.; Nelson, P.S.; Kucuk, O.; et al. A Novel Flavonoid Composition Targets Androgen Receptor Signaling and Inhibits Prostate Cancer Growth in Preclinical Models. Neoplasia 2018, 20, 789-799. [CrossRef]
- Johnson, T.J.; Hoti, N.; Liu, C.; Chowdhury, W.H.; Li, Y.; Zhang, Y.; Lupold, S.E.; Deweese, T.; Rodriguez, R. Bicalutamide-activated oncolytic adenovirus for the adjuvant therapy of high-risk prostate cancer. Cancer Gene Ther 2013, 20, 394-402. [CrossRef]
- Chen, Y.; Gera, L.; Zhang, S.; Li, X.; Yang, Y.; Mamouni, K.; Wu, A.Y.; Liu, H.; Kucuk, O.; Wu, D. Small molecule BKM1972 inhibits human prostate cancer growth and overcomes docetaxel resistance in intraosseous models. Cancer Lett 2019, 446, 62-72. [CrossRef]
- Podetz-Pedersen, K.M.; Vezys, V.; Somia, N.V.; Russell, S.J.; McIvor, R.S. Cellular immune response against firefly luciferase after sleeping beauty-mediated gene transfer in vivo. Hum Gene Ther 2014, 25, 955-965. [CrossRef]
- Zou, M.; Jiao, J.; Zou, Q.; Xu, Y.; Cheng, M.; Xu, J.; Zhang, Y. Multiple metastases in a novel LNCaP model of human prostate cancer. Oncol Rep 2013, 30, 615-622. [CrossRef]
- Smutova, V.; Para, C.; Foret, M.K.; Bennamoune, N.; Hung, S.; Spickler, C.; Riffon, R.; Rowe, J.; Festin, S.; Authier, S. Non-Clinical Cell Therapy Development Using the NCG Mouse Model as a Test System. Int J Toxicol 2023, 42, 232-253. [CrossRef] [PubMed]
- Ahmat Amin, M.K.B.; Shimizu, A.; Zankov, D.P.; Sato, A.; Kurita, S.; Ito, M.; Maeda, T.; Yoshida, T.; Sakaue, T.; Higashiyama, S.; et al. Epithelial membrane protein 1 promotes tumor metastasis by enhancing cell migration via copine-III and Rac1. Oncogene 2018, 37, 5416-5434. [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



