Submitted:
30 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Quantitative preclinical imaging enables non-invasive characterization of physiological, molecular, and functional processes across a variety of experimental models, providing metrics that inform longitudinal studies and translational research. This review synthesizes current strategies for quantitative imaging across modalities including Positron emission tomography (PET), Single Photon Emission Computed Tomography (SPECT), Magnetic resonance imaging (MRI), Computed Tomography (CT), optical imaging, and hybrid systems. We examine methodological frameworks for parameter extraction, reproducibility, and validation against biological reference standards, evaluating each modality through a cross-cutting analytical framework that distinguishes technical, biological, and computational sources of quantitative variance and identifies the current metrological maturity of harmonization infrastructure across platforms. Key challenges, such as protocol harmonization, cross-platform comparability, and integration across species, are analyzed, alongside computational advances including parametric mapping, and artificial intelligence–assisted pipelines. Emerging approaches that combine multimodal acquisition with standardized reconstruction and calibration strategies are also discussed, emphasizing their potential to enhance precision, reduce bias, and support biologically meaningful interpretation. Collectively, this review provides a comprehensive perspective on the design, implementation, and validation of quantitative preclinical imaging studies, offering practical guidance for generating reproducible, interpretable, and translationally relevant imaging biomarkers. By integrating methodological insights with advances in technology and analytics, it underscores the role of quantitative frameworks in bridging preclinical discovery with translational applications.
Keywords:
1. Introduction
2. Materials and Methods
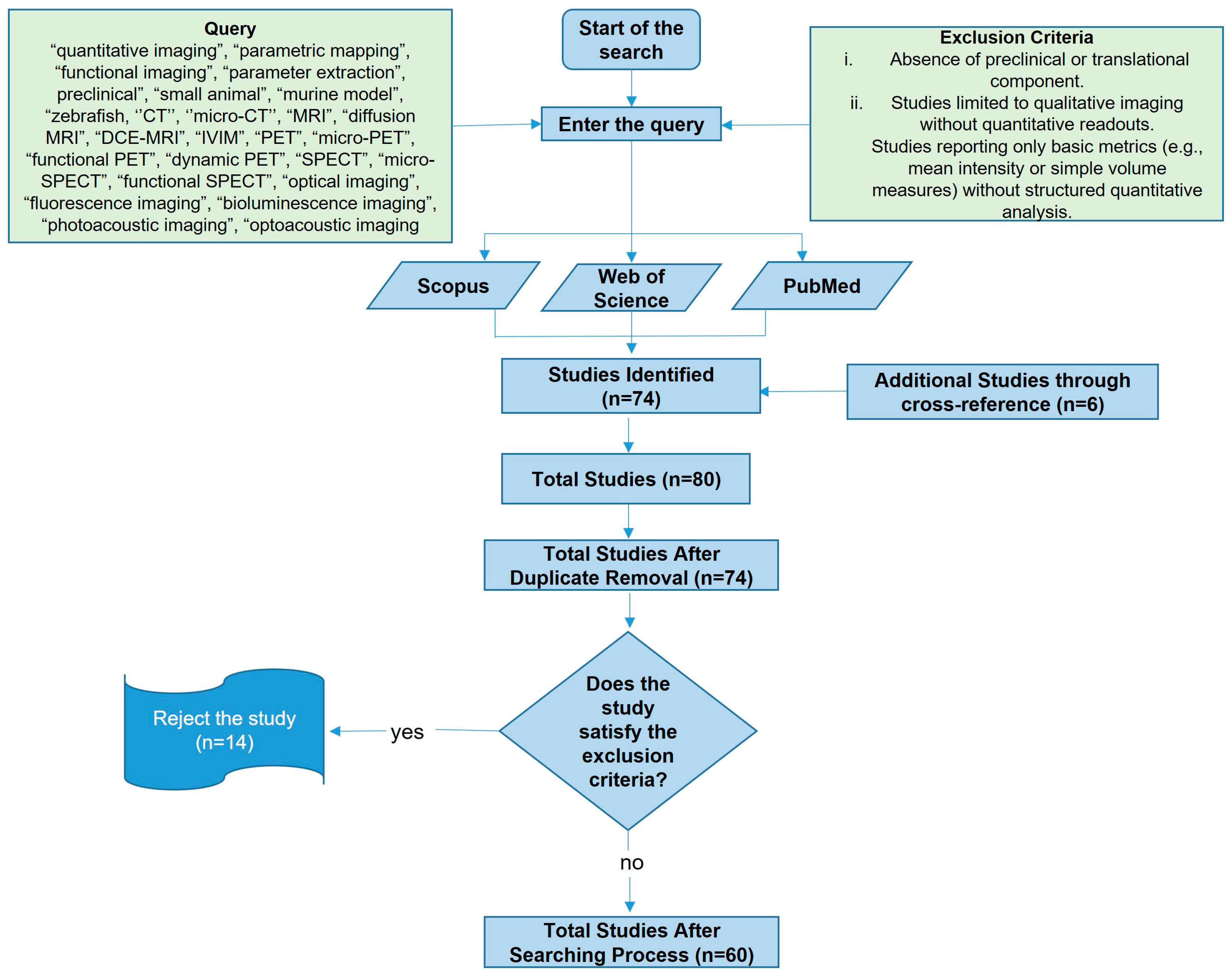
2.1. Literature Searching Strategy
2.2. Inclusion and Exclusion Criteria
- Implementation of quantitative imaging approaches with explicitly defined measurable endpoints, including model-based parameters (e.g., kinetic, diffusion, or perfusion models), calibrated physical measurements (e.g., attenuation coefficients or activity concentration), or imaging biomarkers supported by biological or histological validation.
- Application within preclinical experimental models, including small animal studies or ex vivo imaging frameworks relevant to translational research.
- Explicit consideration of methodological aspects related to quantification, such as reproducibility, standardization, calibration, or validation of imaging-derived parameters.
- Absence of clearly defined quantitative endpoints, including studies limited to qualitative image interpretation or descriptive analysis without measurable parameters.
- Lack of methodological description regarding the derivation, validation, or reproducibility of imaging-derived metrics.
- Studies focused exclusively on clinical imaging without a preclinical component.
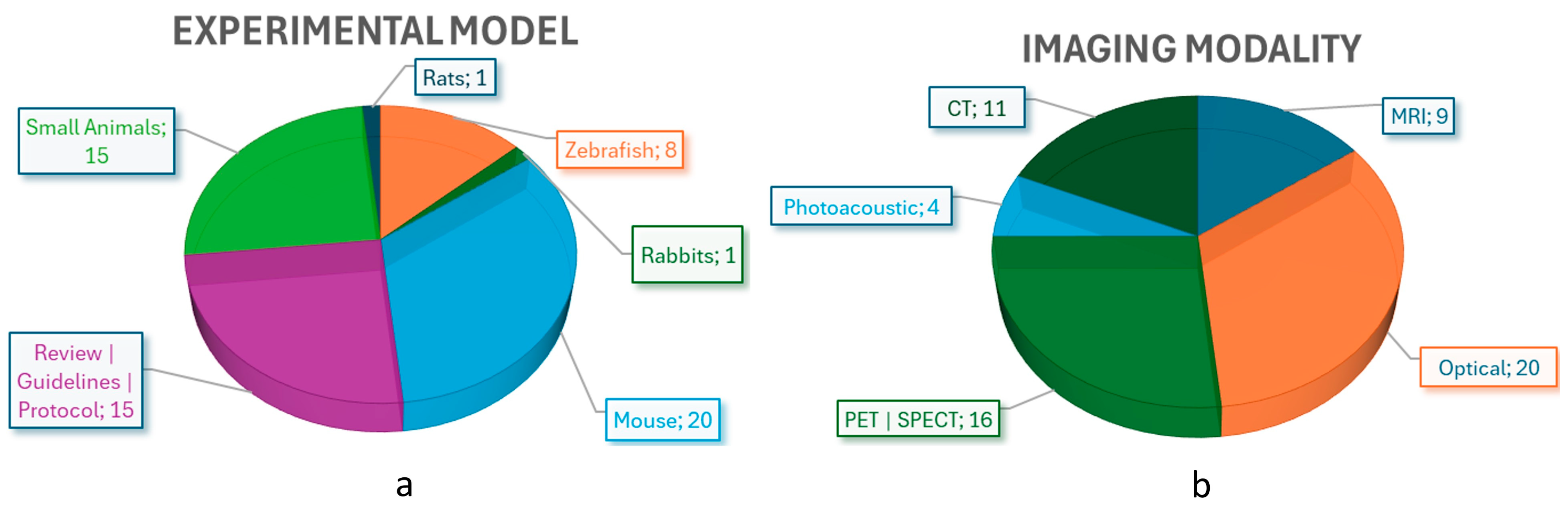
2.3. Classification of Studies
- Biological model: mouse, zebrafish, rabbit.
- Imaging modality: CT/micro- CT, MRI, PET, SPECT, optical imaging, photoacoustic imaging.
- Type of quantitative approach: diffusion-based metrics, perfusion modelling, relaxometry, spectroscopy, AI-assisted mapping, or multimodal integration.
| Database | PubMed | Scopus | Web of Science |
||||
| Period | 2015-2026 | ||||||
| Model | Mouse | Rabbit | Zebrafish | Small Animals |
|||
| Modality | CT | MRI | PET/SPECT | Optical | Photoacoustic | ||
| Purpose | Prediction | Bio- correlation |
Toxicology | ||||
| Study Type | Original | Reviews |
3. Results
3.1. Search Strategy
3.2. Overview of Biological Models and Imaging Modalities
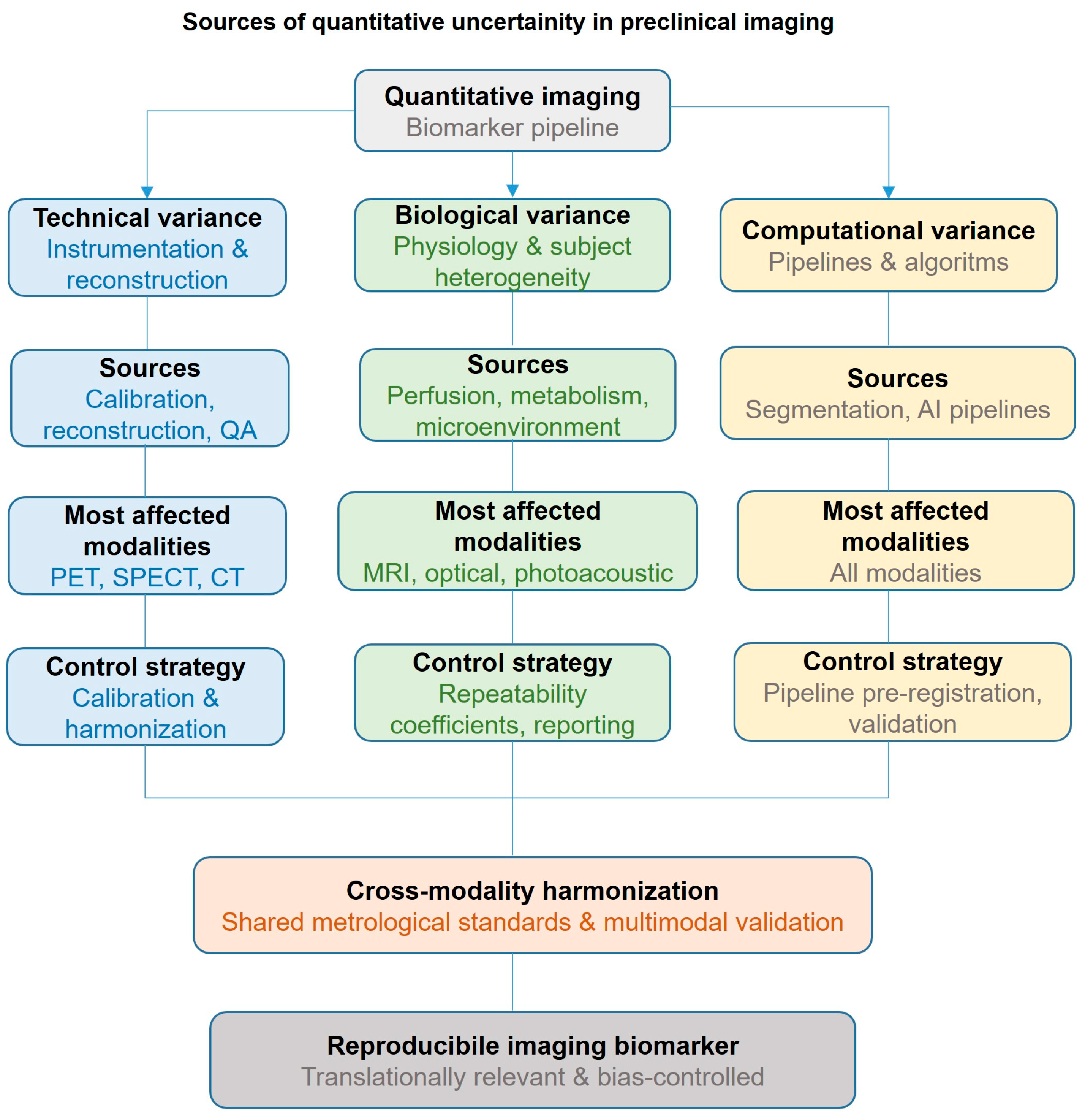
3.3. Variance, Bias, and Cross-Modality Harmonization in Preclinical Imaging
3.4. Quantitative CT in Preclinical Imaging
3.5. Quantitative MRI in Preclinical Models
3.6. Quantitative Preclinical Nuclear Imaging: PET and SPECT
3.7. Multimodal and Emerging Optical Imaging
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bruzgo-Grzybko, M.; Kalita, I.S.; Olichwier, A.J.; Bielicka, N.; Chabielska, E.; Gromotowicz-Poplawska, A. Preclinical PET and SPECT Imaging in Small Animals: Technologies, Challenges and Translational Impact. Cells 2025, 15, 73. [Google Scholar] [CrossRef]
- Miyaoka, R.S.; Lehnert, A.L. Small Animal PET: A Review of What We Have Done and Where We Are Going. Phys. Med. Biol. 2020, 65, 24TR04. [Google Scholar] [CrossRef]
- Mannheim, J.G.; Lan, W.; Conti, M.; Siedler, F.; Krueger, M.A.; Herfert, K.; la Fougère, C.; Schmidt, F.P. Feasibility of in Vivo Small Animal Imaging Using a Clinical Total-Body PET/CT System. EJNMMI Phys. 2025, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Chomet, M.; Schreurs, M.; Vos, R.; Verlaan, M.; Kooijman, E.J.; Poot, A.J.; Boellaard, R.; Windhorst, A.D.; van Dongen, G.A.; Vugts, D.J.; et al. Performance of NanoScan PET/CT and PET/MR for Quantitative Imaging of 18F and 89Zr as Compared with Ex Vivo Biodistribution in Tumor-Bearing Mice. EJNMMI Res. 2021, 11, 57. [Google Scholar] [CrossRef] [PubMed]
- Vanhove, C.; Koole, M.; Fragoso Costa, P.; Schottelius, M.; Mannheim, J.; Kuntner, C.; Warnock, G.; McDougald, W.; Tavares, A.; Bernsen, M. Preclinical SPECT and PET: Joint EANM and ESMI Procedure Guideline for Implementing an Efficient Quality Control Programme. Eur. J. Nucl. Med. Mol. Imaging 2024, 51, 3822–3839. [Google Scholar] [CrossRef]
- Iommelli, F.; De Rosa, V.; Terlizzi, C.; Fonti, R.; Del Vecchio, S. Preclinical Imaging in Targeted Cancer Therapies. Semin. Nucl. Med. 2019, 49, 369–381. [Google Scholar] [CrossRef]
- Balber, T.; Benčurová, K.; Mayrhofer, M.; Friske, J.; Haas, M.; Kuntner, C.; Helbich, T.H.; Hacker, M.; Mitterhauser, M.; Rausch, I. Quantitative Accuracy of Preclinical in Ovo PET/MRI: Influence of Attenuation and Quantification Methods. EJNMMI Phys. 2025, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Clark, D.P.; Badea, C.T. Advances in Micro-CT Imaging of Small Animals. Physica Medica 2021, 88, 175–192. [Google Scholar] [CrossRef]
- Brown, K.H.; Kerr, B.N.; Pettigrew, M.; Connor, K.; Miller, I.S.; Shiels, L.; Connolly, C.; McGarry, C.K.; Byrne, A.T.; Butterworth, K.T. A Comparative Analysis of Preclinical Computed Tomography Radiomics Using Cone-Beam and Micro-Computed Tomography Scanners. Phys. Imaging Radiat. Oncol. 2024, 31, 100615. [Google Scholar] [CrossRef]
- Kugler, E.C.; Frost, J.; Silva, V.; Plant, K.; Chhabria, K.; Chico, T.J.A.; Armitage, P.A. Zebrafish Vascular Quantification: A Tool for Quantification of Three-Dimensional Zebrafish Cerebrovascular Architecture by Automated Image Analysis. Development 2022, 149. [Google Scholar] [CrossRef]
- Sim, Y.; Lee, E.; Jeong, J. ZEMs: Zebrafish Embedding Molds for High-Throughput Imaging of Zebrafish Embryos and Larvae. Methods 2025, 244, 157–167. [Google Scholar] [CrossRef]
- K S, S.; Vithalkar, M.P.; Vishnusai, B.; Gupta, H.; Medishetti, R.; Mohamed, R.; Nayak, Y. Reimagining Preclinical Research: Zebrafish Models Driving Advances in Pharmacology and Toxicology. F1000Res. 2025, 14, 1342. [Google Scholar] [CrossRef]
- Omar, M.; Rebling, J.; Wicker, K.; Schmitt-Manderbach, T.; Schwarz, M.; Gateau, J.; López-Schier, H.; Mappes, T.; Ntziachristos, V. Optical Imaging of Post-Embryonic Zebrafish Using Multi Orientation Raster Scan Optoacoustic Mesoscopy. Light Sci. Appl. 2016, 6, e16186. [Google Scholar] [CrossRef]
- Mannheim, J.G.; Kara, F.; Doorduin, J.; Fuchs, K.; Reischl, G.; Liang, S.; Verhoye, M.; Gremse, F.; Mezzanotte, L.; Huisman, M.C. Standardization of Small Animal Imaging—Current Status and Future Prospects. Mol. Imaging Biol. 2018, 20, 716–731. [Google Scholar] [CrossRef] [PubMed]
- Mannheim, J.G.; Mamach, M.; Reder, S.; Traxl, A.; Mucha, N.; Disselhorst, J.A.; Mittelhäuser, M.; Kuntner, C.; Thackeray, J.T.; Ziegler, S.; et al. Reproducibility and Comparability of Preclinical PET Imaging Data: A Multicenter Small-Animal PET Study. Journal of Nuclear Medicine 2019, 60, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Kuntner, C.; Alcaide, C.; Anestis, D.; Bankstahl, J.P.; Boutin, H.; Brasse, D.; Elvas, F.; Forster, D.; Rouchota, M.G.; Tavares, A.; et al. Optimizing SUV Analysis: A Multicenter Study on Preclinical FDG-PET/CT Highlights the Impact of Standardization. Mol. Imaging Biol. 2024, 26, 668–679. [Google Scholar] [CrossRef]
- Kuntner, C.; Friske, J.; Stessl, A.; Haas, M.; Breyer, L.L.; Wanek, T.; Hacker, M.; Helbich, T.; Rausch, I. Assessing PET Performance, Image Quality, and Attenuation Correction in the Presence of RF Coils within a 9.4 T MRI for Preclinical Simultaneous Imaging. EJNMMI Phys. 2025, 12, 63. [Google Scholar] [CrossRef]
- McDougald, W.; Vanhove, C.; Lehnert, A.; Lewellen, B.; Wright, J.; Mingarelli, M.; Corral, C.A.; Schneider, J.E.; Plein, S.; Newby, D.E.; et al. Standardization of Preclinical PET/CT Imaging to Improve Quantitative Accuracy, Precision, and Reproducibility: A Multicenter Study. Journal of Nuclear Medicine 2020, 61, 461–468. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, G.; Gu, Y.; Zhao, W.; Lu, J.; Zhu, J.; MacAskill, C.J.; Dupuis, A.; Griswold, M.A.; Ma, D.; et al. 3D MR Fingerprinting for Dynamic Contrast-enhanced Imaging of Whole Mouse Brain. Magn. Reson. Med. 2025, 93, 67–79. [Google Scholar] [CrossRef]
- Sullivan, D.C.; Obuchowski, N.A.; Kessler, L.G.; Raunig, D.L.; Gatsonis, C.; Huang, E.P.; Kondratovich, M.; McShane, L.M.; Reeves, A.P.; Barboriak, D.P.; et al. Metrology Standards for Quantitative Imaging Biomarkers. Radiology 2015, 277, 813–825. [Google Scholar] [CrossRef]
- Pierce, T.T.; Sirlin, C.B.; Fowler, K.J.; Buckler, A.J.; Hall, T.J.; Obuchowski, N.A. Understanding Repeatability and Reproducibility Coefficients for Quantitative Imaging Biomarkers. Radiology 2025, 316. [Google Scholar] [CrossRef] [PubMed]
- Jelescu, I.O.; Grussu, F.; Ianus, A.; Hansen, B.; Barrett, R.L.C.; Aggarwal, M.; Michielse, S.; Nasrallah, F.; Syeda, W.; Wang, N.; et al. Considerations and Recommendations from the ISMRM Diffusion Study Group for Preclinical Diffusion MRI: Part 1: In Vivo Small-animal Imaging. Magn. Reson. Med. 2025, 93, 2507–2534. [Google Scholar] [CrossRef]
- Malyarenko, D.; Amouzandeh, G.; Pickup, S.; Zhou, R.; Manning, H.C.; Gammon, S.T.; Shoghi, K.I.; Quirk, J.D.; Sriram, R.; Larson, P.; et al. Evaluation of Apparent Diffusion Coefficient Repeatability and Reproducibility for Preclinical MRIs Using Standardized Procedures and a Diffusion-Weighted Imaging Phantom. Tomography 2023, 9, 375–386. [Google Scholar] [CrossRef]
- Gargiulo, S.; Albanese, S.; Mancini, M. State-of-the-Art Preclinical Photoacoustic Imaging in Oncology: Recent Advances in Cancer Theranostics. Contrast Media Mol. Imaging 2019, 2019, 1–24. [Google Scholar] [CrossRef]
- O’Connor, J.P.B.; Aboagye, E.O.; Adams, J.E.; Aerts, H.J.W.L.; Barrington, S.F.; Beer, A.J.; Boellaard, R.; Bohndiek, S.E.; Brady, M.; Brown, G.; et al. Imaging Biomarker Roadmap for Cancer Studies. Nat. Rev. Clin. Oncol. 2017, 14, 169–186. [Google Scholar] [CrossRef]
- Christiansen, B.A. Effect of Micro-Computed Tomography Voxel Size and Segmentation Method on Trabecular Bone Microstructure Measures in Mice. Bone Rep. 2016, 5, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Oliviero, S.; Cheong, V.S.; Roberts, B.C.; Orozco Diaz, C.A.; Griffiths, W.; Bellantuono, I.; Dall’Ara, E. Reproducibility of Densitometric and Biomechanical Assessment of the Mouse Tibia From In Vivo Micro-CT Images. Front. Endocrinol. (Lausanne). 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, E.; Buccardi, M.; Stellari, F.F. Vivo Micro-CT Imaging for Quantitative Longitudinal Assessment of Pulmonary Diseases in Small Animals; 2025; pp. 207–232. [Google Scholar]
- Vincenzi, E.; Fantazzini, A.; Basso, C.; Barla, A.; Odone, F.; Leo, L.; Mecozzi, L.; Mambrini, M.; Ferrini, E.; Sverzellati, N.; et al. A Fully Automated Deep Learning Pipeline for Micro-CT-Imaging-Based Densitometry of Lung Fibrosis Murine Models. Respir. Res. 2022, 23, 308. [Google Scholar] [CrossRef]
- Buccardi, M.; Ferrini, E.; Pennati, F.; Vincenzi, E.; Ledda, R.E.; Grandi, A.; Buseghin, D.; Villetti, G.; Sverzellati, N.; Aliverti, A.; et al. A Fully Automated Micro-CT Deep Learning Approach for Precision Preclinical Investigation of Lung Fibrosis Progression and Response to Therapy. Respir. Res. 2023, 24, 126. [Google Scholar] [CrossRef]
- Cheng, H.; Gao, T.; Sun, Y.; Huang, F.; Gu, X.; Shan, C.; Luo, S.; Wang, B. AI-Assisted Semiquantitative Measurement of Murine Bleomycin-Induced Lung Fibrosis Using in Vivo Micro-CT: An End-to-End Approach. American Journal of Physiology-Cell Physiology 2025, 329, C659–C674. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.; Clemmensen, A.; Hansen, J.G.; van Krimpen Mortensen, J.; Christensen, E.N.; Kjaer, A.; Ripa, R.S. 3D Whole Body Preclinical Micro-CT Database of Subcutaneous Tumors in Mice with Annotations from 3 Annotators. Sci. Data 2024, 11, 1021. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Rosa, A.; Oliveira, T.S.; Ferreira, M.S.; Vianna-Barbosa, R.J.; Wilmart-Gonçalves, T.C.; Ortiga, T.M.; Bloise, F.F. Non-Invasive Skeletal Muscle Quantification in Small Animals Using Micro-Computed Tomography. Journal of Visualized Experiments 2024. [Google Scholar] [CrossRef]
- Ashton, J.R.; West, J.L.; Badea, C.T. In Vivo Small Animal Micro-CT Using Nanoparticle Contrast Agents. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef]
- Albrecht, J.; Polenz, D.; Kühl, A.A.; Rogasch, J.M.M.; Leder, A.; Sauer, I.M.; Babos, M.; Mócsai, G.; Beindorff, N.; Steffen, I.G.; et al. Diffusion-Weighted Magnetic Resonance Imaging Using a Preclinical 1 T PET/MRI in Healthy and Tumor-Bearing Rats. EJNMMI Res. 2019, 9, 21. [Google Scholar] [CrossRef]
- Duan, Z.; Tao, J.; Liu, W.; Liu, Y.; Fang, S.; Yang, Y.; Liu, X.; Deng, X.; Song, Y.; Wang, S. Correlation of IVIM/DKI Parameters with Hypoxia Biomarkers in Fibrosarcoma Murine Models: Direct Control of MRI and Pathological Sections. Acad. Radiol. 2024, 31, 1014–1023. [Google Scholar] [CrossRef]
- Guo, J.; Sun, W.; Dong, C.; Wu, Z.; Li, X.; Zhou, R.; Xu, W. Intravoxel Incoherent Motion Imaging Combined with Diffusion Kurtosis Imaging to Assess the Response to Radiotherapy in a Rabbit VX2 Malignant Bone Tumor Model. Cancer Imaging 2022, 22, 47. [Google Scholar] [CrossRef]
- Pickup, S.; Romanello, M.; Gupta, M.; Song, H.; Zhou, R. Dynamic Contrast-Enhanced MRI in the Abdomen of Mice with High Temporal and Spatial Resolution Using Stack-of-Stars Sampling and KWIC Reconstruction. Tomography 2022, 8, 2113–2128. [Google Scholar] [CrossRef]
- Roudi, R.; Pisani, L.J.; Pisani, F.; Liang, T.; Daldrup-Link, H.E. Reproducibility and Repeatability of Quantitative T2 and T2* Mapping of Osteosarcomas in a Mouse Model. Eur. Radiol. Exp. 2024, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Xie, L.; Dibb, R.; Li, W.; Decker, K.; Zhang, Y.; Johnson, G.A.; Liu, C. Imaging Whole-Brain Cytoarchitecture of Mouse with MRI-Based Quantitative Susceptibility Mapping. Neuroimage 2016, 137, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K.; Erokwu, B.O.; Johansen, M.L.; Basilion, J.P.; Gulani, V.; Griswold, M.A.; Flask, C.A.; Brady-Kalnay, S.M. Dynamic Quantitative T1 Mapping in Orthotopic Brain Tumor Xenografts. Transl. Oncol. 2016, 9, 147–154. [Google Scholar] [CrossRef]
- Pavone, A.M.; Benfante, V.; Giaccone, P.; Stefano, A.; Torrisi, F.; Russo, V.; Serafini, D.; Richiusa, S.; Pometti, M.; Scopelliti, F.; et al. Biodistribution Assessment of a Novel 68Ga-Labeled Radiopharmaceutical in a Cancer Overexpressing CCK2R Mouse Model: Conventional and Radiomics Methods for Analysis. 2024, 14, 409. [Google Scholar] [CrossRef]
- Knyzeliene, A.; Shaw, R.; Balogh, V.; Tavares, A.A.S. Kinetic Modeling Methods in Preclinical Positron Emission Tomography Imaging; 2024; pp. 441–455. [Google Scholar]
- Kuttner, S.; Luppino, L.T.; Convert, L.; Sarrhini, O.; Lecomte, R.; Kampffmeyer, M.C.; Sundset, R.; Jenssen, R. Deep-Learning-Derived Input Function in Dynamic [18F]FDG PET Imaging of Mice. Frontiers in Nuclear Medicine 2024, 4. [Google Scholar] [CrossRef]
- Raccagni, I.; Belloli, S.; Valtorta, S.; Stefano, A.; Presotto, L.; Pascali, C.; Bogni, A.; Tortoreto, M.; Zaffaroni, N.; Daidone, M.G.; et al. [18F]FDG and [18F]FLT PET for the Evaluation of Response to Neo-Adjuvant Chemotherapy in a Model of Triple Negative Breast Cancer. PLoS One 2018, 13, e0197754. [Google Scholar] [CrossRef]
- Gargiulo, S.; Coda, A.R.; Panico, M.; Gramanzini, M.; Moresco, R.M.; Chalon, S.; Pappatà, S. Molecular Imaging of Neuroinflammation in Preclinical Rodent Models Using Positron Emission Tomography. The Quarterly Journal of Nuclear Medicine and Molecular Imaging 2017, 61. [Google Scholar] [CrossRef]
- Benfante, V.; Stefano, A.; Comelli, A.; Giaccone, P.; Cammarata, F.P.; Richiusa, S.; Scopelliti, F.; Pometti, M.; Ficarra, M.; Cosentino, S.; et al. A New Preclinical Decision Support System Based on PET Radiomics: A Preliminary Study on the Evaluation of an Innovative 64Cu-Labeled Chelator in Mouse Models. J. Imaging 2022, 8, 92. [Google Scholar] [CrossRef]
- Tucker, C.; Collins, R.; Denvir, M.A.; McDougald, W.A. PET/CT Technology in Adult Zebrafish: A Pilot Study Toward Live Longitudinal Imaging. Front. Med. (Lausanne). 2021, 8. [Google Scholar] [CrossRef]
- Gerdekoohi, S.K.; Vosoughi, N.; Tanha, K.; Assadi, M.; Ghafarian, P.; Rahmim, A.; Ay, M.R. Implementation of Absolute Quantification in Small-animal SPECT Imaging: Phantom and Animal Studies. J. Appl. Clin. Med. Phys. 2017, 18, 215–223. [Google Scholar] [CrossRef]
- Lukas, M.; Kluge, A.; Beindorff, N.; Brenner, W. Multi-Isotope Capabilities of a Small-Animal Multi-Pinhole SPECT System. Journal of Nuclear Medicine 2020, 61, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Prieto, E.; Irazola, L.; Collantes, M.; Ecay, M.; Cuenca, T.; Martí-Climent, J.M.; Peñuelas, I. Performance Evaluation of a Preclinical SPECT/CT System for Multi-Animal and Multi-Isotope Quantitative Experiments. Sci. Rep. 2022, 12, 18161. [Google Scholar] [CrossRef] [PubMed]
- Enninful, K.; Ahmed, F.; Girod, B.; Laforest, R.; Thorek, D.L.J.; Prasad, V.; Jha, A.K. Advances in SPECT and PET Reconstruction for Theranostics. PET Clin. 2026, 21, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Altunay, B.; Bauwens, M.; Schäfer, L.; Heesch, A.; Morgenroth, A.; Mottaghy, F.M.; Florea, A. Radiosynthesis, Quality Control, and Small Animal Positron Emission Tomography Imaging of <Sup>68</Sup>Ga-Labelled Nano Molecules. Journal of Visualized Experiments 2024. [Google Scholar] [CrossRef]
- Refaat, A.; Yap, M.L.; Pietersz, G.; Walsh, A.P.G.; Zeller, J.; del Rosal, B.; Wang, X.; Peter, K. In Vivo Fluorescence Imaging: Success in Preclinical Imaging Paves the Way for Clinical Applications. J. Nanobiotechnology 2022, 20, 450. [Google Scholar] [CrossRef] [PubMed]
- Chu, T.; Zheng, X.; Manshian, B.B.; Soenen, S.J. Optical Imaging in Biomedical Research: Guidelines and Practical Insights. Health Nanotechnology 2025, 1, 4. [Google Scholar] [CrossRef]
- Lo, P.-A.; Su, S.-P.; Chiang, H.K. Small-Animal 360-Deg Fluorescence Diffuse Optical Tomography Using Structural Prior Information from Ultrasound Imaging. J. Biomed. Opt. 2020, 25, 1. [Google Scholar] [CrossRef]
- An, Y.; Wang, K.; Tian, J. Recent Methodology Advances in Fluorescence Molecular Tomography. Vis. Comput. Ind. Biomed. Art 2018, 1, 1. [Google Scholar] [CrossRef]
- Zhang, P.; Ma, C.; Song, F.; Fan, G.; Sun, Y.; Feng, Y.; Ma, X.; Liu, F.; Zhang, G. A Review of Advances in Imaging Methodology in Fluorescence Molecular Tomography. Phys. Med. Biol. 2022, 67, 10TR01. [Google Scholar] [CrossRef]
- Nouizi, F.; Brooks, J.; Zuro, D.M.; Hui, S.K.; Gulsen, G. Development of a Theranostic Preclinical Fluorescence Molecular Tomography/Cone Beam CT-Guided Irradiator Platform. Biomed. Opt. Express 2022, 13, 6100. [Google Scholar] [CrossRef]
- Konovalov, A.B.; Vlasov, V. V.; Uglov, A.S. Early-photon Reflectance Fluorescence Molecular Tomography for Small Animal Imaging: Mathematical Model and Numerical Experiment. Int. J. Numer. Method. Biomed. Eng. 2021, 37. [Google Scholar] [CrossRef]
- Klose, A.D.; Paragas, N. Automated Quantification of Bioluminescence Images. Nat. Commun. 2018, 9, 4262. [Google Scholar] [CrossRef]
- Thompson, W.R.; Brecht, H.-P.F.; Ivanov, V.; Yu, A.M.; Dumani, D.S.; Lawrence, D.J.; Emelianov, S.Y.; Ermilov, S.A. Characterizing a Photoacoustic and Fluorescence Imaging Platform for Preclinical Murine Longitudinal Studies. J. Biomed. Opt. 2023, 28. [Google Scholar] [CrossRef]
- Deng, Z.; Xu, X.; Iordachita, I.; Dehghani, H.; Zhang, B.; Wong, J.W.; Wang, K.K.-H. Mobile Bioluminescence Tomography-Guided System for Pre-Clinical Radiotherapy Research. Biomed. Opt. Express 2022, 13, 4970. [Google Scholar] [CrossRef]
- Xu, X.; Deng, Z.; Sforza, D.; Tong, Z.; Tseng, Y.; Newman, C.; Reinhart, M.; Tsouchlos, P.; Devling, T.; Dehghani, H.; et al. Characterization of a Commercial Bioluminescence Tomography-guided System for Pre-clinical Radiation Research. Med. Phys. 2023, 50, 6433–6453. [Google Scholar] [CrossRef]
- Smith, J.T.; Sinsuebphon, N.; Rudkouskaya, A.; Michalet, X.; Intes, X.; Barroso, M. In Vivo Quantitative FRET Small Animal Imaging: Intensity versus Lifetime-Based FRET. Biophysical Reports 2023, 3, 100110. [Google Scholar] [CrossRef]
- Kim, S.J.; Lee, H.-Y. In Vivo Molecular Imaging in Preclinical Research. Lab. Anim. Res. 2022, 38, 31. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, S.; Gramanzini, M.; Bonente, D.; Tamborrino, T.; Inzalaco, G.; Gherardini, L.; Franci, L.; Bertelli, E.; Barone, V.; Chiariello, M. Preclinical Application of Computer-Aided High-Frequency Ultrasound (HFUS) Imaging: A Preliminary Report on the In Vivo Characterization of Hepatic Steatosis Progression in Mouse Models. J. Imaging 2025, 11, 369. [Google Scholar] [CrossRef] [PubMed]
- Lichtenegger, A.; Tamaoki, J.; Licandro, R.; Mori, T.; Mukherjee, P.; Bian, L.; Greutter, L.; Makita, S.; Wöhrer, A.; Matsusaka, S.; et al. Longitudinal Investigation of a Xenograft Tumor Zebrafish Model Using Polarization-Sensitive Optical Coherence Tomography. Sci. Rep. 2022, 12, 15381. [Google Scholar] [CrossRef] [PubMed]
- Bini, F.; Missori, E.; Pucci, G.; Pasini, G.; Marinozzi, F.; Forte, G.I.; Russo, G.; Stefano, A. Preclinical Implementation of MatRadiomics: A Case Study for Early Malformation Prediction in Zebrafish Model. J. Imaging 2024, 10, 290. [Google Scholar] [CrossRef]
- Li, K.; Liu, B.; Wang, Z.; Li, Y.; Li, H.; Wu, S.; Li, Z. Quantitative Characterization of Zebrafish Development Based on Multiple Classifications Using Mueller Matrix OCT. Biomed. Opt. Express 2023, 14, 2889. [Google Scholar] [CrossRef]
- Sturtzel, C.; Grissenberger, S.; Wenninger-Weinzierl, A.; Distel, M. High-Content Imaging-Based Screening for Anticancer Compounds in Zebrafish Xenografts; 2025; pp. 153–162. [Google Scholar]
- Mitovic, N.; Kovacevic, S.; Nesovic Ostojic, J.; Puflovic, D.; Stankovic, S.M. Functional Cardiac Imaging in Zebrafish Embryos Using Standard Microscopy and Video Analysis: Applications in Environmental and Biomedical Research. Journal of Visualized Experiments 2025. [Google Scholar] [CrossRef]
- Cani, O.; Zannotti, L.; De Vita, A.; Liverani, C.; Vanni, S.; Miserocchi, G. Zebrafish Tumor Xenograft Models for Drug-Screening: A Systematic Review of Methods for Treatment Assessment. J. Transl. Med. 2026. [Google Scholar] [CrossRef]
- Turrini, L.; Roschi, L.; de Vito, G.; Pavone, F.S.; Vanzi, F. Imaging Approaches to Investigate Pathophysiological Mechanisms of Brain Disease in Zebrafish. Int. J. Mol. Sci. 2023, 24, 9833. [Google Scholar] [CrossRef]
- Upputuri, P.K.; Pramanik, M. Recent Advances toward Preclinical and Clinical Translation of Photoacoustic Tomography: A Review. J. Biomed. Opt. 2016, 22, 041006. [Google Scholar] [CrossRef]
- Humbert, J.; Will, O.; Peñate-Medina, T.; Peñate-Medina, O.; Jansen, O.; Both, M.; Glüer, C.-C. Comparison of Photoacoustic and Fluorescence Tomography for the in Vivo Imaging of ICG-Labelled Liposomes in the Medullary Cavity in Mice. Photoacoustics 2020, 20, 100210. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, Y.; Li, W.; Li, C. Real-Time Dual-Modal Photoacoustic and Fluorescence Small Animal Imaging. Photoacoustics 2024, 36, 100593. [Google Scholar] [CrossRef]
- van der Heijden, W.R.P.; Groothuizen, S.; Zwezerijnen, G.J.C.; Schuit, R.C.; Hemke, R.; Boellaard, R.; van der Laken, C.J.; Yaqub, M. Na [18F]F PET/CT Quantification in Spondyloarthritis: Comparative Evaluation of SUV Normalization Approaches. EJNMMI Res. 2025, 15, 143. [Google Scholar] [CrossRef] [PubMed]
- Benfante, V.; Stefano, A.; Ali, M.; Laudicella, R.; Arancio, W.; Cucchiara, A.; Caruso, F.; Cammarata, F.P.; Coronnello, C.; Russo, G.; et al. An Overview of In Vitro Assays of 64Cu-, 68Ga-, 125I-, and 99mTc-Labelled Radiopharmaceuticals Using Radiometric Counters in the Era of Radiotheranostics. Diagnostics 2023, 13, 1210. [Google Scholar] [CrossRef] [PubMed]
- Willowson, K.P.; Bailey, D.L. Evolving SPECT-CT Technology. British Journal of Radiology 2024. [Google Scholar] [CrossRef]
- Lavielle, A.; Pinaud, N.; Zhang, B.; Crémillieux, Y. Quantitative Brain T1 Maps Derived from T1-Weighted MRI Acquisitions: A Proof-of-Concept Study. Eur. Radiol. Exp. 2024, 8, 109. [Google Scholar] [CrossRef]
- Arridge, S.; Hauptmann, A. Networks for Nonlinear Diffusion Problems in Imaging. J. Math. Imaging Vis. 2020, 62, 471–487. [Google Scholar] [CrossRef]
- Di Giacinto, F.; Riente, A.; Mignini, I.; Ainora, M.E.; Esposto, G.; Borriello, R.; Zocco, M.A.; Minordi, L.M.; Sala, E.; Scaldaferri, F.; et al. Advancing Multispectral Optoacoustic Tomography (MSOT): Phasor Analysis for Real-Time Spectral Unmixing. Comput. Biol. Med. 2025, 195, 110586. [Google Scholar] [CrossRef]
- Reader, A.J.; Pan, B. AI for PET Image Reconstruction. Br. J. Radiol. 2023, 96. [Google Scholar] [CrossRef]
- Stefano, A. Challenges and Limitations in Applying Radiomics to PET Imaging: Possible Opportunities and Avenues for Research. Comput. Biol. Med. 2024, 179, 108827. [Google Scholar] [CrossRef] [PubMed]
- Lauciello, N.; Russo, G.; Stefano, A. Radiomics in Preclinical Imaging: Current Trends and Future Directions. Clin. Transl. Imaging 2026. [Google Scholar] [CrossRef]
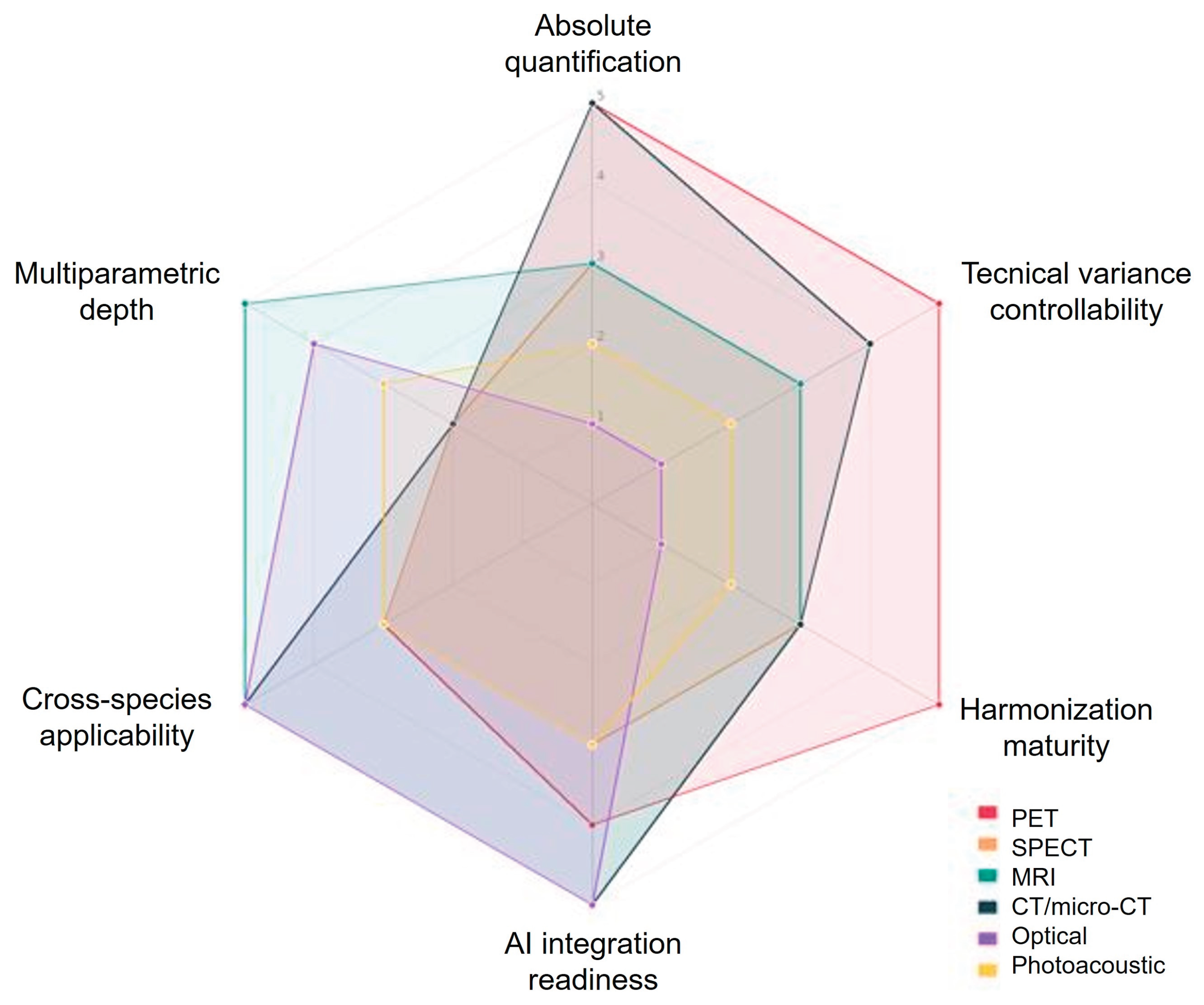
| Modality | Absolute quantification | Technical variance controllability | Harmonization maturity | AI integration readiness | Cross-species applicability | Multiparametric depth |
|---|---|---|---|---|---|---|
| PET | High | High | High | High | Medium | Low |
| SPECT | Medium | Medium | Medium | Medium | Medium | Low |
| MRI | Medium | Medium | Medium | High | High | High |
| CT / micro-CT | High | High | Medium | High | High | Low |
| Optical | Low | Low | Low | High | High | High |
| Photoacoustic | Low | Low | Low | Medium | Medium | Medium |
| Study | Year | Focus | Model | Key quantitative outcome | Quantitative performance |
|---|---|---|---|---|---|
| Clark et al. [8] |
2021 | Advanced micro-CT technologies: attenuation calibration, material decomposition, and automated segmentation | -Review -Small Animal | Multi-parameter structural metrics — Hounsfield Unit-based quantification, material-specific attenuation coefficients | High — comprehensive methodological framework; calibration and reconstruction standards reviewed |
| Christiansen et al. [26] |
2016 | Effect of voxel size and segmentation method on trabecular bone microstructure metrics | Mouse | BV/TV, Tb.Th, Tb.Sp — trabecular architecture descriptors sensitive to acquisition and segmentation parameters | Medium — systematic parameter sensitivity analysis; reproducibility dependent on protocol consistency |
| Oliviero et al. [27] |
2022 | Reproducibility of densitometric and biomechanical metrics from in vivo micro-CT tibia images | Mouse | BMD, cortical thickness, stiffness estimates — longitudinal skeletal biomarkers | High — repeatability and reproducibility coefficients formally reported; in vivo validation |
| Ferrini et al. [28] |
2025 | Longitudinal micro-CT for quantitative assessment of pulmonary disease in small animals | Small Animal | Total lung volume, aerated lung fraction, mean lung density — structural pulmonary biomarkers | High — standardized longitudinal workflow validated; quantitative descriptors reproducible across timepoints |
| Vincenzi et al. [29] |
2022 | Fully automated deep learning pipeline for micro-CT densitometry in pulmonary fibrosis models | Mouse | Lung density distribution — AI-derived densitometric descriptors with histopathological correlation | High — automated pipeline validated against manual annotations; deep learning segmentation accuracy reported |
| Buccardi et al. [30] |
2023 | Fully automated micro-CT deep learning approach for lung fibrosis progression and therapy response | Mouse | Lung fibrosis extent, density metrics — longitudinal therapy response indicators | High — prospective validation; automated quantification compared to expert assessment |
| Cheng et al. [31] |
2025 | AI-assisted semiquantitative measurement of bleomycin-induced lung fibrosis using in vivo micro-CT | Mouse | Fibrosis score, lung density — end-to-end AI-assisted quantification pipeline | Medium — AI-assisted approach validated; semiquantitative rather than fully quantitative output |
| Jensen et al. [32] |
2024 | Annotated whole-body micro-CT database of subcutaneous tumours with multi-annotator segmentation | Mouse | Tumour volume, segmentation consistency metrics — inter-observer reproducibility benchmarks | High — multi-annotator dataset enables reproducibility benchmarking; publicly available reference standard |
| Brown et al. [9] |
2024 | Comparative analysis of preclinical CT radiomics using cone-beam and micro-CT scanners | Mouse | Radiomic features, reproducibility metrics — cross-platform feature stability assessment | Medium — cross-platform reproducibility demonstrated; feature stability highly dependent on acquisition and reconstruction harmonization |
| Pereira-Rosa et al. [33] |
2024 | Non-invasive skeletal muscle quantification in small animals using micro-CT | Protocol (JoVE) - Small Animals |
Muscle volume, cross-sectional area — morphometric descriptors for musculoskeletal assessment | Medium — methodology demonstrated; formal reproducibility metrics not fully reported |
| Ashton et al. [34] |
2015 | In vivo micro-CT with nanoparticle contrast agents for vascular and perfusion imaging | -Review -Small Animals |
Vascular volume fraction, contrast distribution — semi-quantitative vascular architecture metrics | Medium — contrast-enhanced vascular quantification demonstrated; absolute quantification limited by agent pharmacokinetics |
| Study | Year | Focus | Model | Key quantitative outcome | Quantitative performance |
|---|---|---|---|---|---|
| Jelescu et al. [22] |
2025 | DWI — ADC quantification; ISMRM acquisition and modelling guidelines for quantitative robustness | Guidelines Small Animals |
ADC — structured recommendations for gradient calibration, motion management, and b-value selection | High — consensus-based framework; reproducibility benchmarks defined |
| Albrecht et al. [35] |
2019 | DWI — ADC quantification in healthy and tumour tissues on preclinical PET/MRI platform | Rats | ADC — compatibility with hybrid PET/MRI acquisition confirmed | Medium — single-centre validation; cross-platform reproducibility not assessed |
| Duan et al. [36] |
2024 | IVIM/DKI — separation of molecular diffusion and microvascular perfusion components | Mouse | D, D*, f (IVIM); K, D (DKI) — hypoxia biomarker correlation with histological endpoints | Medium — biological validation available; inter-session reproducibility not reported |
| Guo et al. [37] |
2022 | IVIM/DKI — monitoring radiotherapy response through perfusion-sensitive and microstructural parameters | Rabbit | D, D*, f, K — therapy response indicators with histopathological correlation | Medium — longitudinal design; single-centre; limited reproducibility metrics |
| Pickup et al. [38] |
2022 | DCE-MRI — optimized quantitative protocol for preclinical cancer models; AIF estimation | Mouse | Ktrans, ve — vascular permeability and extravascular volume fraction | High — optimised AIF strategy; protocol reproducibility validated in vivo |
| Zhu et al. [19] |
2025 | MR Fingerprinting — simultaneous T1/T2 mapping with dynamic contrast acquisition in single scan | Mouse | T1, T2, DCE parameters — simultaneous multiparametric estimation | High — dictionary-based validation; simultaneous parameter estimation confirmed in vivo |
| Roudi et al. [39] |
2024 | Relaxometry — T2/T2* mapping; edema, fibrosis, and tumour microenvironment assessment | Mouse | T2, T2* — between-session variability quantified under controlled conditions | Medium — repeatability coefficients reported; biological and technical variance partially separated |
| Wei et al. [40] |
2016 | QSM — tissue magnetic susceptibility for iron deposition, haemorrhage, and calcification | Mouse | chi (susceptibility) — iron deposition and haemorrhage quantification | Medium — methodology validated; preclinical-specific reproducibility data limited |
| Herrman et al. [41] |
2016 | 1H-MRS with T1 mapping — metabolic quantification combined with longitudinal relaxation assessment | Mouse | Metabolite concentrations, T1 — combined metabolic and structural characterization | Medium — multiparametric combination demonstrated; reproducibility metrics not fully reported |
| Study | Year | Focus | Model | Key quantitative outcome | Quantitative performance |
|---|---|---|---|---|---|
| Pavone et al. [42] |
2024 | PET as a tool to identify quantitative biomarkers in preclinical imaging | Mouse | SUV, metabolic rate — imaging biomarker identification and biological validation | Medium — biomarker framework proposed; multicentric validation not reported |
| Kuntner et al. [16] |
2024 | Harmonization of SUV acquisition and analysis to reduce variability and improve reproducibility | Small Animals |
SUV — multicentric variability reduction through standardized acquisition and analysis | High — multicentric study; SUV variability formally quantified and reduced through harmonized protocols |
| Knyzeliene et al. [43] |
2024 | Dynamic PET quantification using compartmental and graphical kinetic modelling | Small Animals |
Ki, k1-k4, DVR — kinetic rate constants from compartmental and graphical modelling | Medium — kinetic modelling framework validated; AIF sensitivity acknowledged |
| Kuttner et al. [44] |
2024 | AI-assisted arterial input function estimation to improve kinetic parameter accuracy | Mouse | AIF-derived kinetic parameters — AI-assisted estimation reduces operator dependency | Medium — AI-AIF approach validated in vivo; generalizability across tracers not fully established |
| Mannheim et al. [3] |
2025 | Cross-platform quantitative comparison between preclinical and clinical total-body PET/CT | Small Animals |
SUV, partial volume effects — cross-platform and cross-scale quantitative concordance | Medium — translational concordance demonstrated; scale-dependent challenges explicitly reported |
| Raccagni et al. [45] |
2018 | Evaluation of response to neo-adjuvant chemotherapy in a triple negative breast cancer (TNBC) mouse model | Mouse | PET, SUV, standard uptake metrics, tumor metabolic activity quantification | Medium — biological validation with histological endpoints; single-centre study |
| Gargiulo et al. [46] |
2017 | PET molecular imaging of neuroinflammation; quantitative microglial activation assessment | -Review -Rodents |
BPND, SUV — neuroinflammatory biomarker quantification | Medium — biological validation with histological correlation; single-centre |
| Benfante et al. [47] |
2022 | PET biodistribution analysis of 64Cu-chelator radiotracer in mouse models | Mouse | SUV, organ-specific uptake — radiotracer biodistribution and biological validation | Medium — biological validation available; quantitative reproducibility not formally reported |
| Tucker et al. [48] |
2021 | Radiotracer uptake quantification and longitudinal biodistribution in zebrafish | Zebrafish | SUV, whole-body biodistribution — cross-species feasibility of quantitative nuclear imaging | Limited — proof-of-concept; pronounced partial volume effects; metrological re-validation required |
| Gerdekoohi et al. [49] |
2017 | Absolute activity quantification with attenuation correction and iterative reconstruction in SPECT | Small Animals |
Absolute activity concentration — attenuation-corrected quantification with calibration factors | Medium — absolute quantification demonstrated; calibration stability not longitudinally assessed |
| Lukas et al. [50] |
2020 | Quantitative validation of simultaneous multi-radionuclide small-animal SPECT imaging | Small Animals |
Activity concentration per radionuclide — simultaneous multi-isotope quantitative validation | High — simultaneous multi-radionuclide validation; quantitative integrity formally assessed |
| Prieto et al. [51] |
2022 | Multi-isotope quantitative validation in preclinical SPECT/CT |
Small Animals |
Activity concentration, recovery coefficients — multi-isotope SPECT/CT quantitative accuracy | High — multi-isotope validation with recovery coefficient analysis; cross-calibration reported |
| Enninful et al. [52] |
2026 | Review of SPECT instrumentation and reconstruction strategies impacting quantitation | Review | Quantitative accuracy metrics — instrumentation and reconstruction impact on SPECT quantification | High — comprehensive methodological review; reconstruction and collimator impact systematically analysed |
| Altunay et al. [53] |
2024 | PET tracer radiochemistry and QC linked to quantitative imaging workflows | -Protocol (JoVE) -Small Animals |
Radiochemical purity, specific activity — radiopharmaceutical QC impact on quantitative PET | Medium — radiochemical QC framework described; direct link to imaging reproducibility partially established |
| Vanhove et al. [5] |
2024 | Quality control and harmonization standards for quantitative PET and SPECT | -Guidelines -Small Animals |
QA framework — structured calibration, acquisition standards, and analysis workflows for PET/SPECT | High — international guideline; EANM/ESMI endorsed; formally adopted quality assurance framework |
| Bruzgo-Grzybko et al. [1] |
2025 | Comprehensive review on quantitative performance and translational relevance of PET/SPECT | -Review -Small Animals |
Translational performance metrics — synthesis of quantitative PET/SPECT capabilities and limitations | High — comprehensive evidence synthesis; multicentric and translational perspective |
| Study | Year | Focus | Model | Key quantitative outcome | Quantitative performance |
|---|---|---|---|---|---|
| Refaat et al. [54] |
2022 | Fluorescence imaging standardization, calibration, and reproducibility frameworks | Review | Signal calibration standards — reproducibility requirements for quantitative fluorescence imaging | Medium — standardization framework proposed; formal validation across platforms limited |
| Chu et al. [55] |
2025 | Optical imaging harmonization, workflow standardization, cross-lab reproducibility | Review | Cross-lab reproducibility metrics — harmonization strategies for quantitative optical imaging | Medium — harmonization framework outlined; multicentric adoption not yet established |
| Lo et al. [56] |
2020 | Fluorescence Diffuse Optical Tomography with ultrasound priors; reconstruction accuracy | Small Animals |
Fluorophore concentration maps — depth-corrected volumetric fluorescence quantification | Medium — reconstruction accuracy improved with anatomical priors; absolute quantification partially validated |
| An et al. [57] |
2018 | Fluorescence Molecular Tomography (FMT); inverse problem regularization | Review | Volumetric fluorophore distribution — regularized inverse solution for FMT quantification | Medium — reconstruction methodology validated in phantoms; in vivo reproducibility limited |
| Zhang et al. [58] |
2022 | FMT reconstruction; diffusion-based modelling, volumetric quantification | Review | Volumetric fluorophore concentration — diffusion model-based FMT reconstruction | Medium — modelling framework validated; sensitivity to optical property assumptions acknowledged |
| Nouizi et al. [59] |
2022 | Hybrid FMT/CBCT; anatomical priors to improve inverse solution stability | Small Animals |
Fluorophore concentration — anatomically constrained FMT reconstruction with improved stability | Medium — hybrid approach improves quantitative stability; cross-platform validation not reported |
| Konovalov et al. [60] |
2021 | Early-photon and compressed sensing; sensitivity and spatial resolution improvement | Small Animals |
Spatial resolution, sensitivity metrics — early-photon approach for improved optical quantification | Limited — proof-of-concept in phantoms; in vivo quantitative validation limited |
| Klose et al. [61] |
2018 | 3D Bioluminescence imaging (BLI); volumetric reconstruction, geometric bias correction | Small Animals |
Volumetric bioluminescence emission — 3D BLI reconstruction with geometric bias reduction | Medium — volumetric reconstruction validated; substrate delivery variability acknowledged |
| Thompson et al. [62] |
2023 | Dual-modality photoacoustic and fluorescence imaging; dynamic perfusion quantification | Mouse | sO2, HbT, fluorescence signal — simultaneous structural and molecular dynamic quantification | Medium — dual-modality quantification demonstrated; reconstruction stability partially validated |
| Deng et al. [63] |
2022 | Mobile Bioluminescence Tomography (BLT); source localization and reconstruction accuracy | Small Animals |
Source position, bioluminescence intensity — volumetric source localization accuracy | Medium — high-precision source localization demonstrated; cross-system reproducibility not reported |
| Xu et al. [64] |
2023 | Commercial BLT platform; quantitative accuracy and performance assessment | Small Animals |
Bioluminescence flux, source reconstruction accuracy — commercial BLT quantitative performance | Medium — commercial platform performance assessed; biological variability sources acknowledged |
| Smith et al. [65] |
2023 | In vivo quantitative FRET; intensity vs lifetime analysis for molecular interactions | Small Animals |
FRET efficiency, donor lifetime — dynamic molecular interaction quantification | Medium — intensity vs lifetime comparison validated; depth-dependent limitations acknowledged |
| Kim et al. [66] |
2022 | Standardized injection protocols; reproducibility in optical imaging experiments | Review | Signal reproducibility metrics — injection protocol standardization impact on optical quantification | Medium — standardization recommendations provided; formal multicentric validation absent |
| Gargiulo et al [67] |
2025 | Quantitative HFUS assessment of hepatic steatosis in mice using computer-aided analysis | Mouse | Liver echogenicity, morphology metrics — reproducible longitudinal HFUS quantification | Medium — computer-aided analysis validated; cross-scanner reproducibility not assessed |
| Lichtenegger et al. [68] |
2022 | PS-OCT in zebrafish tumour xenografts; microstructural birefringence quantification | Zebrafish | Birefringence, retardation maps — polarimetric microstructural metrics for tumour characterization | Medium — quantitative polarimetric metrics validated in zebrafish; longitudinal reproducibility limited |
| Bini et al. [69] |
2024 | Brightfield imaging; high-dimensional feature extraction and texture quantification in zebrafish | Zebrafish | Morphometric and texture features — radiomics-based quantitative phenotyping | Medium — feature extraction pipeline validated; test-retest reproducibility not formally reported |
| Li et al. [70] |
2023 | Mueller matrix OCT with deep learning; structural-functional mapping in zebrafish | Zebrafish | Polarimetric tissue properties — deep learning-assisted structural-functional quantification | Medium — deep learning integration demonstrated; generalizability across developmental stages limited |
| Sturtzel et al. [71] |
2025 | High-content automated optical microscopy; multiplexed phenotypic quantification | Zebrafish | Volumetric tumour burden, phenotypic metrics — automated multiplexed quantification | High — high-content automated platform; multiplexed quantification validated across cohorts |
| Mitovic et al. [72] |
2025 | Optical microscopy for dynamic cardiac functional quantification in zebrafish | Zebrafish | Heart rate, contractility metrics — motion-tracking-based cardiac functional quantification | Medium — functional quantification validated; inter-session reproducibility not reported |
| Cani et al. [73] |
2026 | Optical/fluorescence xenograft workflows; tumour burden quantification in zebrafish | Zebrafish | Tumour burden, fluorescence intensity — reproducible endpoint harmonization in xenograft models | Medium — systematic review; endpoint harmonization strategies outlined; reproducibility data variable across included studies |
| Turrini et al. [74] |
2023 | Functional fluorescence imaging; dynamic neural activity quantification in zebrafish | Zebrafish | Neural activity maps, spatiotemporal signal metrics — functional fluorescence quantification | Medium — functional imaging framework validated; quantitative reproducibility across preparations limited |
| Upputuri et al. [75] |
2016 | Multispectral photoacoustic imaging (MSOT); spectral unmixing and fluence correction | Review | sO2, HbT — oxygen saturation and total haemoglobin quantification | Limited — methodology reviewed; depth-dependent fluence correction remains a key unresolved limitation |
| Humbert et al. [76] |
2020 | Photoacoustic vs fluorescence tomography; physiologically relevant metric comparison | Mouse | Fluorophore concentration, photoacoustic signal — cross-modality quantitative comparison | Medium — comparative validation available; absolute quantification limited by fluence heterogeneity |
| Sun et al. [77] |
2024 | Dual-modality photoacoustic and fluorescence imaging; structural and molecular quantification | Small Animals |
sO2, HbT, fluorescence signal — simultaneous dual-modality dynamic quantification | Medium — dual-modality approach validated; cross-session reproducibility not formally assessed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




