Submitted:
27 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
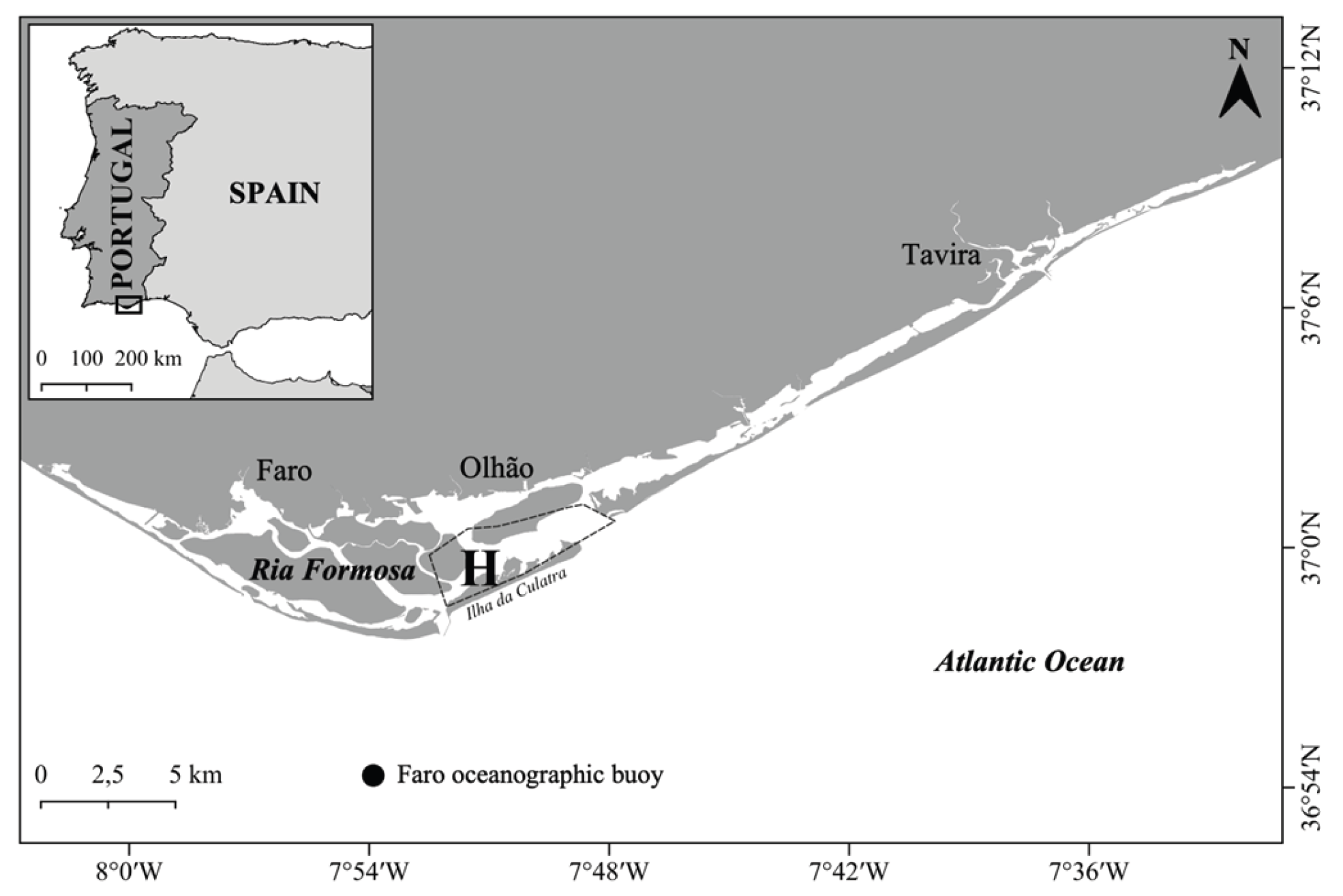
2. Materials and Methods
3. Results
3.1. Samples Size and Sex Ratio
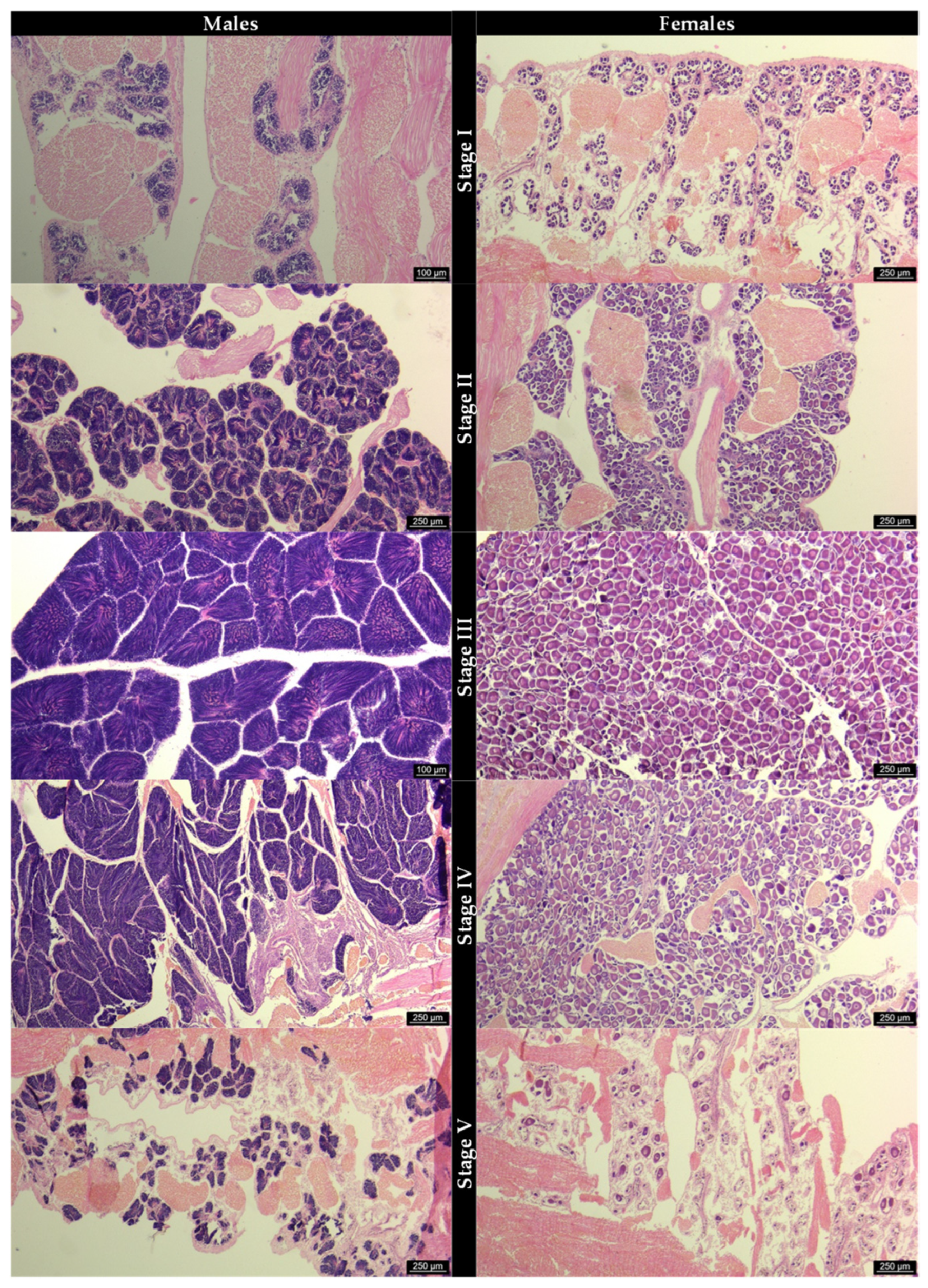
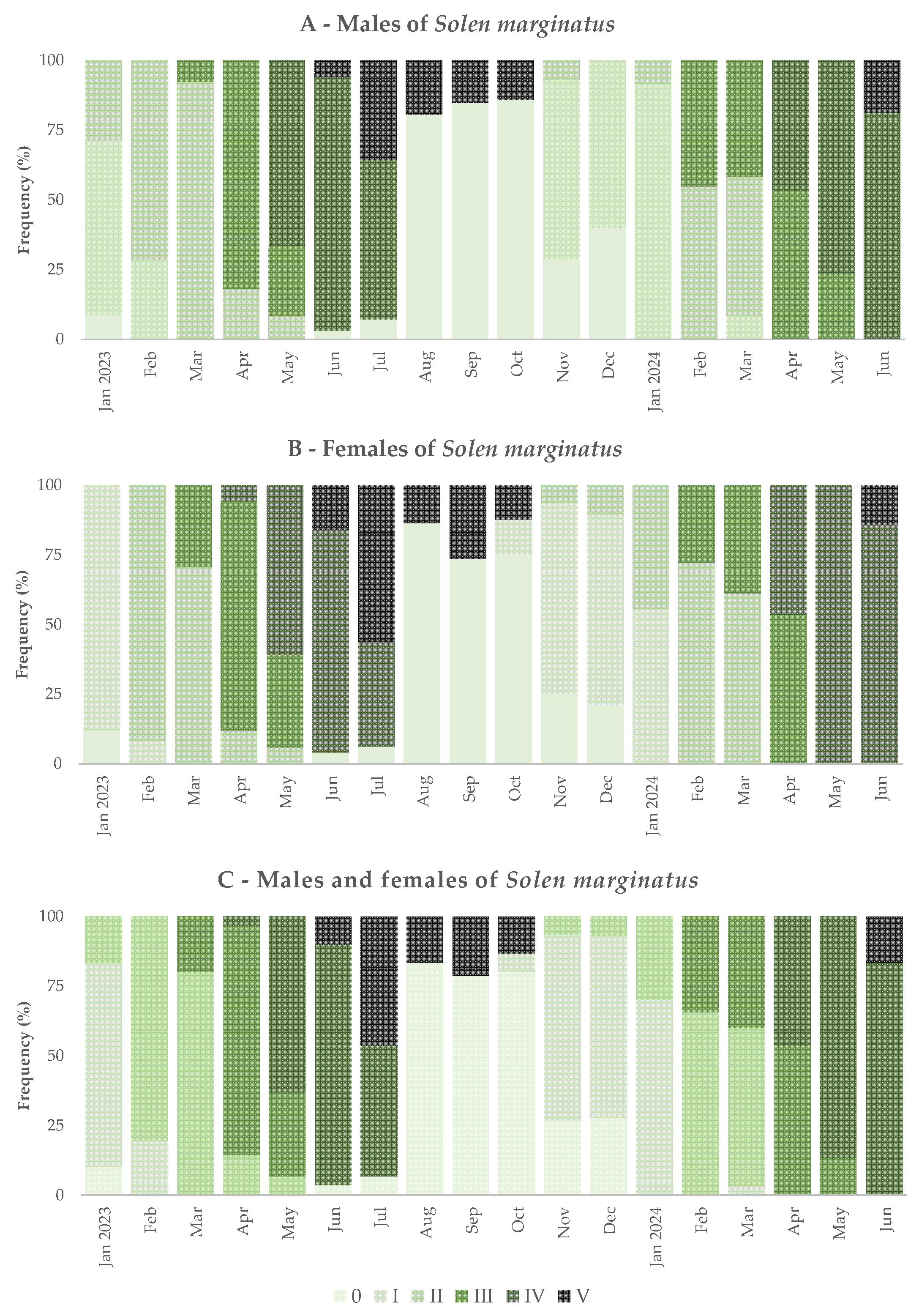
3.2. Gonad Histology and Reproductive Cycle
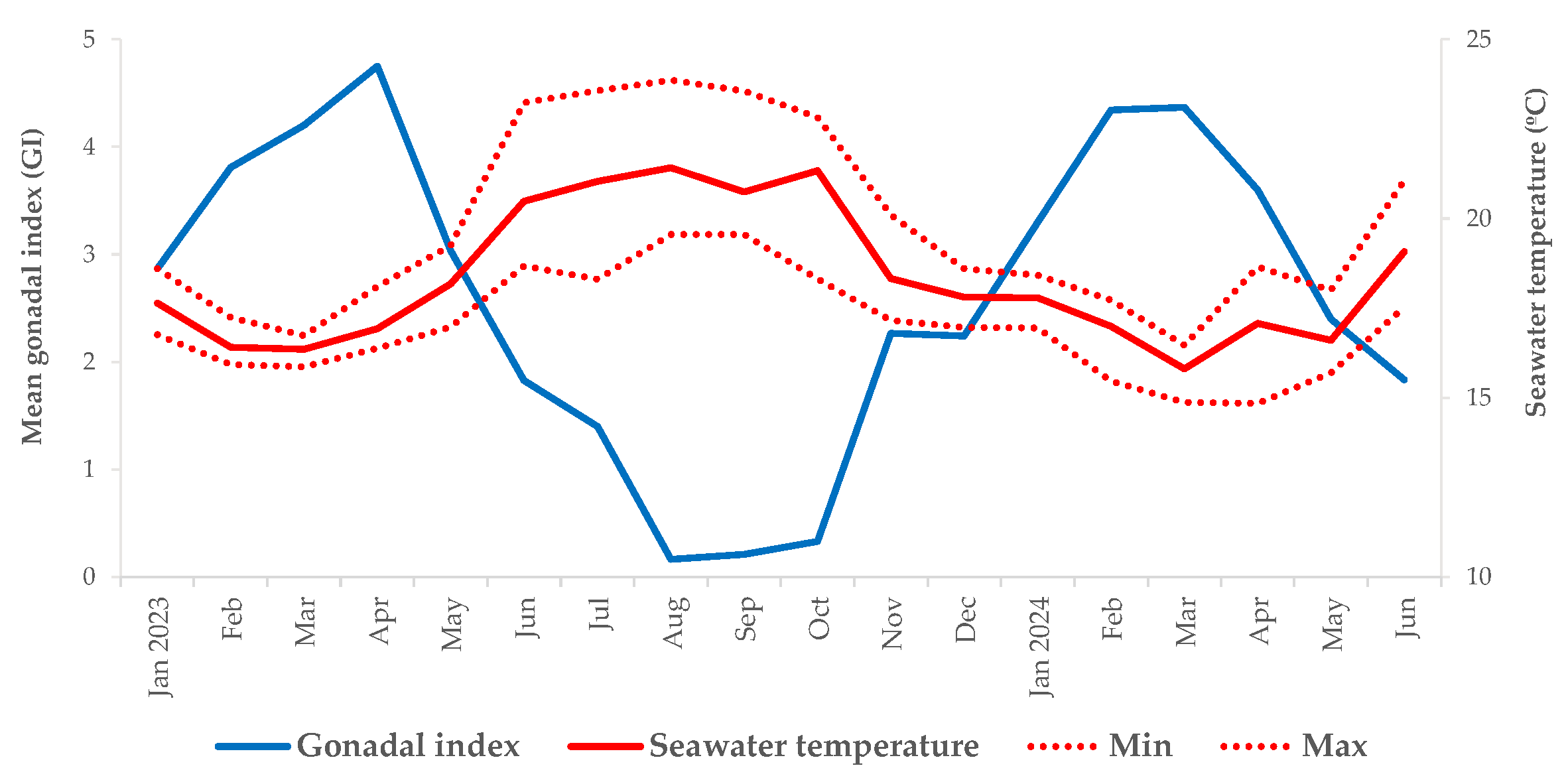
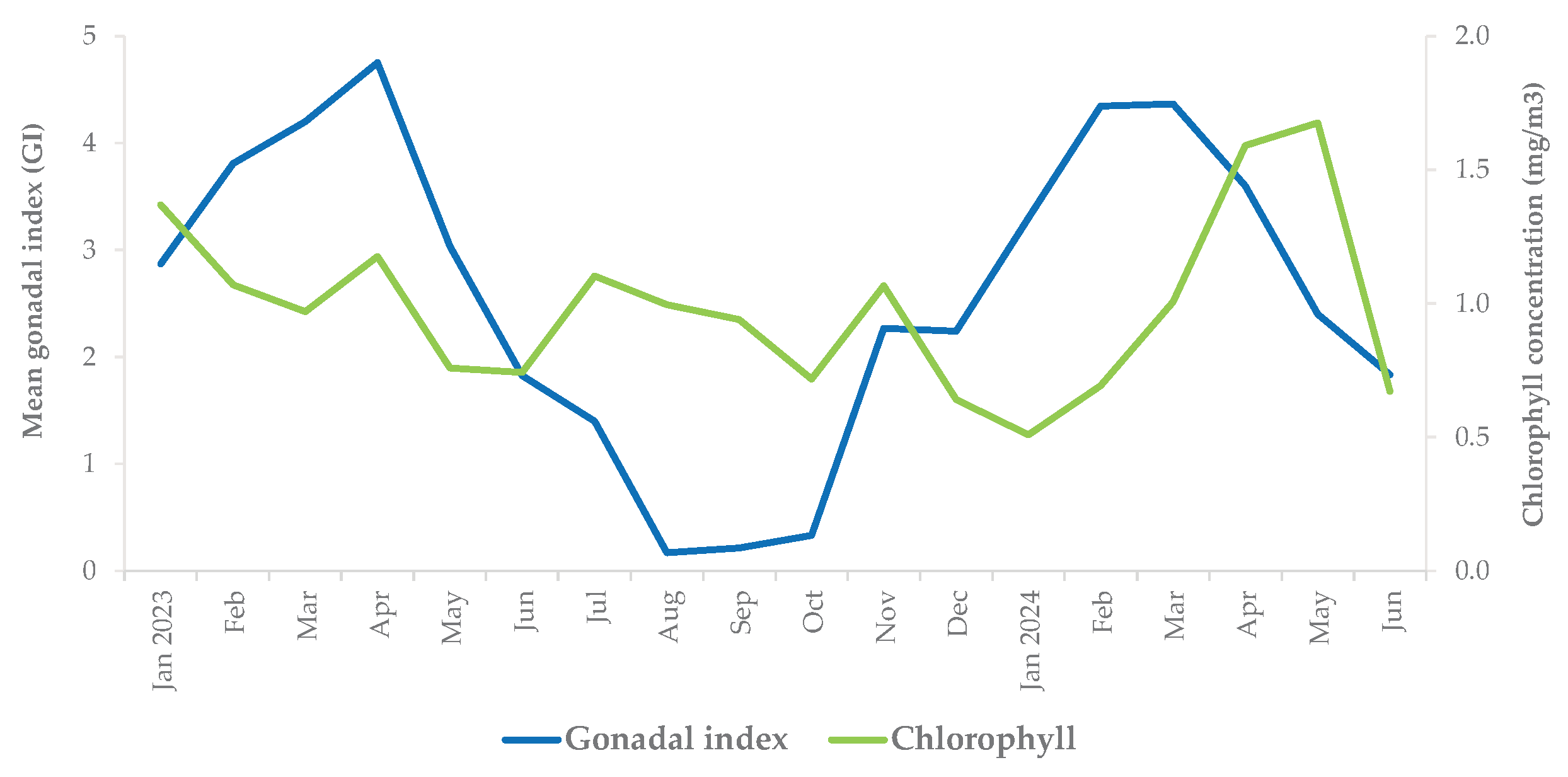
3.3. Environmental Parameters and Mean Gonadal Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, S.C.; Mills Flemming, J.; Watson, R.; Lotze, H.K. Rapid global expansion of invertebrate fisheries: trends, drivers, and ecosystem effects. PLoS ONE 2011, 6, e14735. [Google Scholar] [CrossRef] [PubMed]
- Berkes, F.; Hughes, T.P.; Steneck, R.S.; Wilson, J.A.; Bellwood, D.R.; Crona, B.; Folke, C.; Gunderson, L.H.; Leslie, H.M.; Norberg, J.; Nyström, M.; Olsson, P.; Österblom, H.; Scheffer, M.; Worm, B. Globalization, roving bandits, and marine resources. Science 2006, 311, 1557–1558. [Google Scholar] [CrossRef]
- Anderson, S.C.; Lotze, H.K.; Shackell, N.L. Evaluating the knowledge base for expanding low-trophic-level fisheries in Atlantic Canada. Can. J. Fish. Aquat. Sci. 2008, 65, 2553–2571. [Google Scholar] [CrossRef]
- FAO The State of Food and Agriculture. Technical report; Food and Agriculture Organization of the United Nations: Rome, Italy, 2009; 166p, Available online: https://www.fao.org/4/i0680e/i0680e.pdf (accessed on 6 January 2026).
- Gaspar, M.B.; Barracha, I.; Carvalho, S.; Vasconcelos, P. Clam fisheries worldwide: main species, harvesting methods and fishing impacts. In Clam Fisheries and Aquaculture; Costa, F., Ed.; Nova Science Publishers: New York, NY, USA, 2013; pp. 291–327. [Google Scholar]
- Tebble, N. British Bivalve Seashells; British Museum (Natural History): London, UK, 1966; 212. [Google Scholar]
- Nordsieck, F. Die europäischen Meeresmuscheln (Bivalvia) Von Eismer bis Kapverden, Mittelmeer und Schwarzes Meer; Gustav Fischer Verlag: Stuttgart, Germany, 1969; 256p. [Google Scholar]
- Parenzan, P. Carta d’identità delle conchiglie del Mediterraneo; Vol. 2 Part. 2 of Bivalvi. Bios. Taras Editrice: Taranto, Italy, 1976; 263p. [Google Scholar]
- Maia, F.; Sobral, M.P.; Gaspar, M. Ciclo reprodutivo e primeira maturação de Solen marginatus e Venerupis pullastra na Ria de Aveiro. Bases científicas para a gestão destes recursos. Relatório Científico Técnico IPIMAR 2006. [Google Scholar] [CrossRef]
- Tirado, C.; Rodríguez, F.A.; Bruzón, M.A.; López, J.I.; Salas, C.; Márquez, I. La reproducción de bivalvos y gasterópodos de interés pesquero en Andalucía; Junta de Andalucía, Consejería de Agricultura y Pesca.: Huelva, Spain, 2002; 129p. [Google Scholar]
- Moura, P.; Gaspar, M.B.; Monteiro, C.C. Gametogenic cycle of the smooth clam Callista chione on the south-western coast of Portugal. J. Mar. Biol. Assoc. UK 2008, 88, 161–167. [Google Scholar] [CrossRef]
- Santos, A.; Castro, J.; Raimundo, L. O canal de Tavira (“Ria” formosa – Algarve): Caracterização geral do meio e análise da estrutura, dinâmica e produção das populações de Cerastoderma edule (L.), Solen marginatus Montagu e Spisula solida (L.). Relatório de estágio científico. Universidade de Lisboa, Faculdade de Ciências, Departamento de Zoologia e Antropologia. Laboratório Marítimo da Guia, 1986.
- Newton, A.; Mudge, S.M. Temperature and salinity regimes in a shallow, mesotidal lagoon, the Ria Formosa, Portugal. Estuar. Coast. Shelf Sci. 2003, 57, 73–85. [Google Scholar] [CrossRef]
- Gamito, S.; Erzini, K. Trophic food web and ecosystem attributes of a water reservoir of the Ria Formosa (south Portugal). Ecol. Model. 2005, 181, 509–520. [Google Scholar] [CrossRef]
- Pilkey, O.H.; Neal, W.J.; Monteiro, J.H.; Dias, J.M.A. Algarve barrier islands: a noncoastal-plain system in Portugal. J. Coast. Res. 1989, 5, 239–261. [Google Scholar]
- Águas, M.P.N. Simulação da circulação hidrodinâmica na Ria Formosa. In Proceedings of the Seminário comemorativo do dia mundial do ambiente: Os Sistemas Lagunares do Algarve (pp. 78–90). Universidade do Algarve, Faro, Portugal, 5–7 June 1985; pp. 78–90. [Google Scholar]
- Falcão, M.; Vale, C. Fluxos de nutrientes em viveiros de amêijoa na Ria Formosa. In Proceedings of the VI Simposio Ibérico de Estudio del Bentos Marino, Palma de Mallorca, Spain, 18–22 September 1988. [Google Scholar]
- Duarte, D.N.R.; Permata, D.; Moreira da Silva, M.; Dores, T.M.P.; Alves, M.C.N.; Fernandes, F.A.B.; dos Santos, M.P.; Chícharo, L. Ria Formosa Hydrodynamics and the Best Location for Shellfish Beds. In Proceedings of the 2nd International Congress on Engineering and Sustainability in the XXI Century - INCREaSE, 9–11 October 2019. [Google Scholar] [CrossRef]
- Cravo, A.; Barbosa, A.; Correia, C.; Matos, A.; Caetano, S.; Lima, M.; Jacob, J. Unravelling the effects of treated wastewater discharges on the water quality in a coastal lagoon system (Ria Formosa, South Portugal): Relevance of hydrodynamic conditions. Mar. Pollut. Bull. 2022, 174, 113296. [Google Scholar] [CrossRef]
- Constantino, R.; Gaspar, M.B.; Pereira, F.; Carvalho, S.; Cúrdia, J.; Matias, D.; Monteiro, C.C. Environmental impact of razor clam harvesting using salt in Ria Formosa lagoon (Southern Portugal) and subsequent recovery of associated benthic communities. Aquatic Conserv: Mar. Freshw. Ecosyst. 2009, 19, 542–553. [Google Scholar] [CrossRef]
- Martínez-Patiño, D. Estudio de los solénidos, Solen marginatus (Pennant, 1777) y Ensis siliqua (Linné, 1758), de los bancos naturales de la Ria Ortigueira y la Ria del Barquero: Ciclo gametogénico, composición bioquímica y cultivo larvario. Ph.D. Thesis, Departamento de Bioquímica y Biologia Molecular, Universidad de Santiago de Compostela, Santiago de Compostela, Spain, 2002. [Google Scholar]
- Fahy, E.; Gaffney, J. Growth statistic of an exploited razor clam (Ensis siliqua) bed at Gormanstown, Co Meath, Ireland. Hydrobiologia 2001, 465, 139–151. [Google Scholar] [CrossRef]
- Pyke, M. Evaluation of good handling practice for razor clam. Seafish Report No. SR548. Seafish Industry Authorithy - Seafish Technology, 2002; 33p. Available online: https://www.seafish.org/media/q2md1dya/sr548.pdf (accessed on 15 January 2026).
- Gaspar, M.B.; Carvalho, A.N.; Pereira, F.; Martins, R.; Carneiro, M.; Moura, P.; Rufino, M.M.; Piló, D.; Moreno, A. Apanha e pesca apeada em Portugal continental: análise do número de licenças e das capturas registadas em lota; Gaspar, M.B., Carvalho, A.N., Eds.; Instituto Português do Mar e da Atmosfera, I.P., Programa MOPPA, 2025; 181p. [Google Scholar]
- Seed, R. Ecology. In Marine Mussels: Their Ecology and Physiology; Bayne, B.L., Ed.; Cambridge University Press: Cambridge, UK, 1976; pp. 13–65. [Google Scholar]
- Ayache, N.; Hmida, L.; Cardoso, J.F.M.F.; Haouas, Z.; Da Costa, F.; Romdhane, M.S. Reproductive cycle of the razor clam Solen marginatus (Pulteney 1799) in the Southern Mediterranean Sea (Gulf of Gabes, South Tunisia). J. Shellfish Res. 2016, 35, 389–397. [Google Scholar] [CrossRef]
- Doukilo, I.; Belhsen, O.K.; Hmimid, F.; Idhalla, M.; Manchih, K.; Mouttaki, B.; Errhif, A. Seasonal changes in reproductive biology and biochemical composition of the grooved razor shell Solen marginatus (Bivalvia: Solenidae) from Oualidia Lagoon, Morocco. Reg. Stud. Mar. Sci. 2022, 54, 102474. [Google Scholar] [CrossRef]
- López, J.; Rodríguez, C.; Carrasco, J.F. Comparación del ciclo reproductor de Solen marginatus (Pulteney, 1799) (Mollusca: Bivalvia) en las rías del Eo y Villaviciosa (Asturias, noroeste de España): relación con las variables ambientales. Bol. Inst. Esp. Oceanogr. 2005, 21, 317–327. [Google Scholar]
- Remacha-Triviño, A.; Anadón, N. Reproductive cycle of the razor clam Solen marginatus (Pulteney, 1799) in Spain: a comparative study in three different locations. J. Shellfish Res. 2006, 25, 869–876. [Google Scholar] [CrossRef]
- Pérez-Camacho, A.; Román, G. La reproducción en los moluscos bivalvos. In Reproducción en Acuicultura; Espinosa, J., Labarta, U., Eds.; Plan de Formación de Técnicos Superiores en Acuicultura (FEUGA): Madrid, Spain, 1987; pp. 133–175. [Google Scholar]
- Darriba, S. Biología de la navaja (Ensis arcuatus Jeffreys, 1865) de la Ría de Vigo (NO de España): Crecimiento y Reproducción. Ph.D. Thesis, Universidad de Vigo, Vigo (Pontevedra), Spain, 2001. [Google Scholar]
- Ojea, J.; Pazos, A.J.; Martinez, D.; Novoa, S.; Sanchez, J.L.; Abad, M. Seasonal variation in weight and biochemical composition of the tissues of Ruditapes decussatus in relation to the gametogenic cycle. Aquaculture 2004, 238, 451–468. [Google Scholar] [CrossRef]
- Joaquim, S.; Matias, D.; Matias, A.M.; Moura, P.; Arnold, W.S.; Chícharo, L.; Gaspar, M.B. Reproductive activity and biochemical composition of the pullet carpet shell Venerupis senegalensis (Gmelin, 1791) (Mollusca: Bivalvia) from Ria de Aveiro (northwestern coast of Portugal). Sci. Mar. 2011, 75, 217–226. [Google Scholar] [CrossRef]
- Himmelman, J.H. Spawning, marine invertebrates. In Encyclopedia of Reproduction; Knobil, E., Neill, J.D., Eds.; Academic Press: NY, USA, 1999; vol. 4, pp. 524–533. [Google Scholar]
- Lubet, P.E. Influence des facteurs externes sur la reproduction des lamellibranches. Oceanis 1980, 6, 469–489. [Google Scholar]
| Year | Month | M | F | I | % M | % F | % I |
|---|---|---|---|---|---|---|---|
| 2023 | January | 16 | 11 | 3 | 53.3 | 36.7 | 10.0 |
| February | 14 | 12 | 0 | 53.8 | 46.2 | 0.0 | |
| March | 13 | 17 | 0 | 43.3 | 56.7 | 0.0 | |
| April | 11 | 17 | 0 | 39.3 | 60.7 | 0.0 | |
| May | 12 | 18 | 0 | 40.0 | 60.0 | 0.0 | |
| June | 16 | 12 | 1 | 55.2 | 41.4 | 3.4 | |
| July | 13 | 15 | 2 | 43.3 | 50.0 | 6.7 | |
| August | 3 | 2 | 25 | 10.0 | 6.7 | 83.3 | |
| September | 2 | 4 | 22 | 7.1 | 14.3 | 78.6 | |
| October | 2 | 4 | 24 | 6.7 | 13.3 | 80.0 | |
| November | 10 | 12 | 8 | 33.3 | 40.0 | 26.7 | |
| December | 6 | 15 | 8 | 20.7 | 51.7 | 27.6 | |
| 2024 | January | 12 | 18 | 0 | 40.0 | 60.0 | 0.0 |
| February | 11 | 18 | 0 | 37.9 | 62.1 | 0.0 | |
| March | 12 | 18 | 0 | 40.0 | 60.0 | 0.0 | |
| April | 15 | 15 | 0 | 50.0 | 50.0 | 0.0 | |
| May | 17 | 13 | 0 | 56.7 | 43.3 | 0.0 | |
| June | 16 | 14 | 0 | 53.3 | 46.7 | 0.0 | |
| Total | 201 | 235 | 93 | 38.0 | 44.4 | 17.6 |
| Location Country |
Jan | Feb | Mar | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec | Related parameters |
Reference | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ria Formosa Portugal |
Present study | ||||||||||||||
| Ria Formosa Portugal |
[12] | ||||||||||||||
| Ria de Aveiro Portugal |
[9] | ||||||||||||||
| Ria of Ortigueira Spain |
[21] | ||||||||||||||
| San Pedro River Spain |
[10] | ||||||||||||||
| Ria de Villaviciosa Spain |
[27] | ||||||||||||||
| Eo Estuary Spain |
[27] | ||||||||||||||
| Eo Estuary Spain |
*[28] | ||||||||||||||
| Santander Bay Spain |
*[28] | ||||||||||||||
| Terrón Estuary Spain |
*[28] | ||||||||||||||
| Gulf of Gabès Tunisia |
**[25] | ||||||||||||||
| Oualidia Lagoon Morocco |
[26] | ||||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




