Submitted:
30 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Background/Objectives: The cochlea and vestibular organs develop concurrently during embryogenesis and share anatomical and functional pathways. As a result, congenital factors affecting the vestibulocochlear system may impair both hearing and vestibular function. Despite this, the relationship between congenital bilateral sensorineural hearing loss (SNHL) and vestibular dysfunction remains insufficiently defined. This study evaluated vestibular function in patients with congenital bilateral SNHL and investigated the association between hearing loss severity and vestibular function. Methods: A total of 202 participants aged 7–31 years were enrolled, including 102 patients with congenital bilateral SNHL and 100 healthy controls. Vestibular function was assessed using videonystagmography during three tests: sinusoidal harmonic acceleration (SHA) rotational testing, the video head impulse test (vHIT), and caloric testing according to the Fitzgerald–Hallpike protocol. Statistical analyses compared vestibular parameters between groups and assessed correlations with hearing loss severity. Results: Patients with congenital bilateral SNHL exhibited significantly lower vestibulo-ocular reflex (VOR) values in the SHA test compared to controls. Greater hearing loss severity was associated with lower VOR gain values. No statistically significant differences were observed between groups in caloric test results or vHIT VOR gain values. However, corrective saccades during vHIT were identified exclusively in patients with hearing loss and occurred in approximately 15% of cases. Furthermore, the age of independent walking was significantly delayed in the study group compared to controls. Conclusions: Congenital bilateral SNHL is associated with vestibular dysfunction, as evidenced by abnormal SHA test results and the presence of corrective saccades in vHIT. These patients may also experience delayed motor development. The findings underscore the importance of comprehensive vestibular assessment in individuals with congenital hearing loss.
Keywords:
1. Introduction
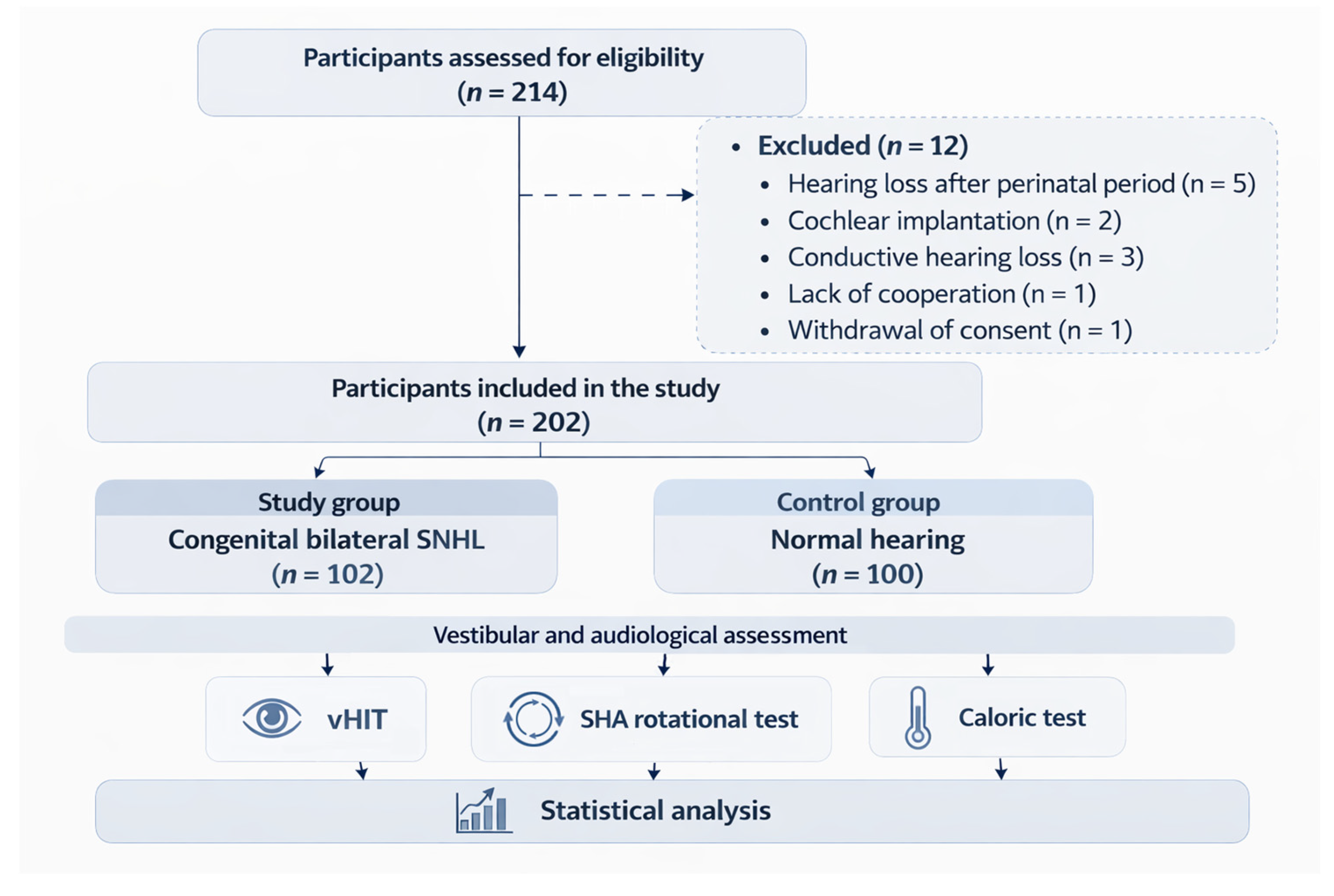
2. Materials and Methods
2.1. Study Population
2.2. Audiological Assessment
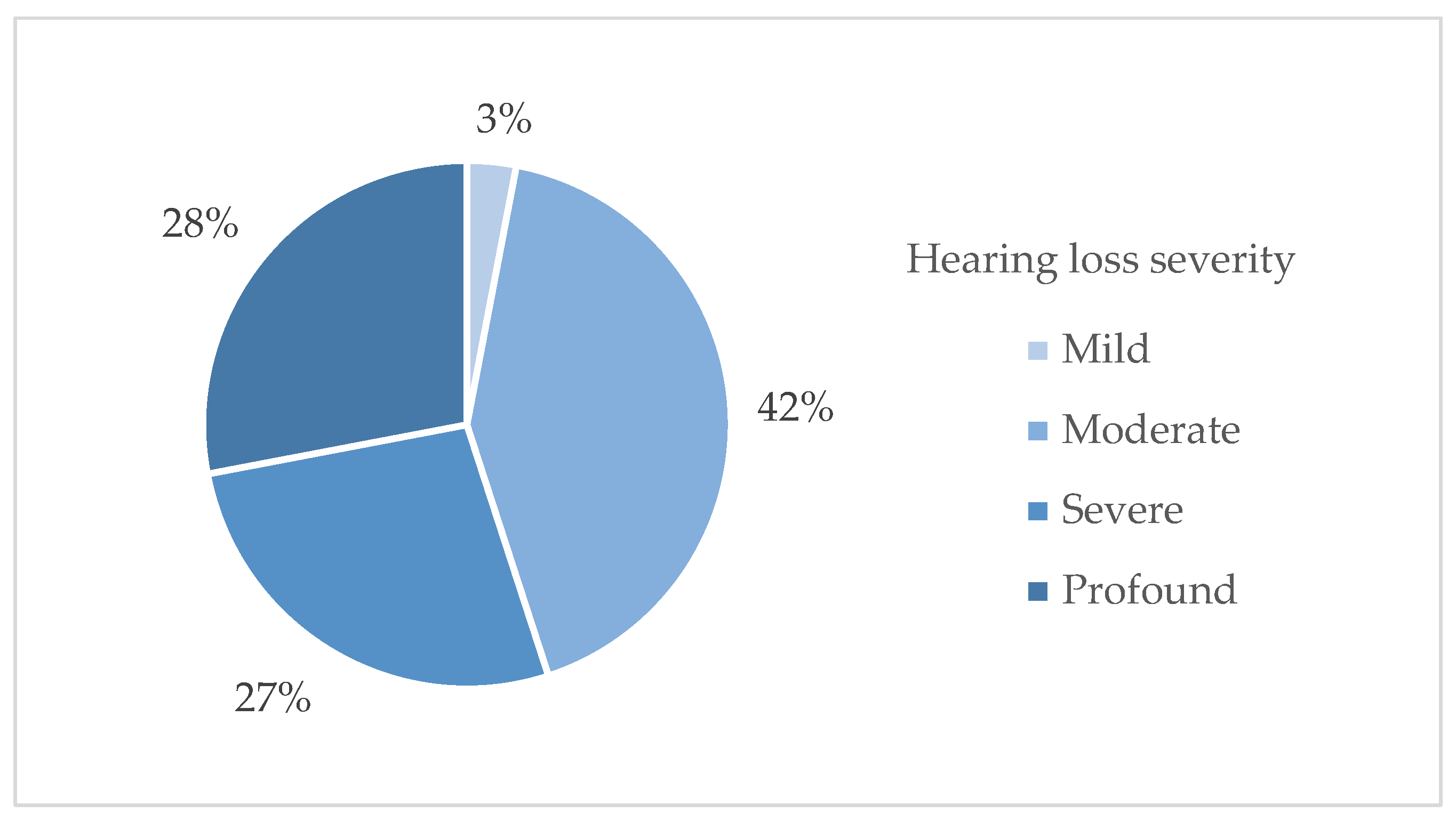
- mild hearing loss (25–40 dB HL),
- moderate hearing loss (45–60 dB HL),
- severe hearing loss (65–85 dB HL),
- profound hearing loss (≥90 dB HL).
2.3. Vestibular Testing
2.3.1. Video Head Impulse Test (vHIT)
2.3.2. Sinusoidal Harmonic Acceleration (SHA)
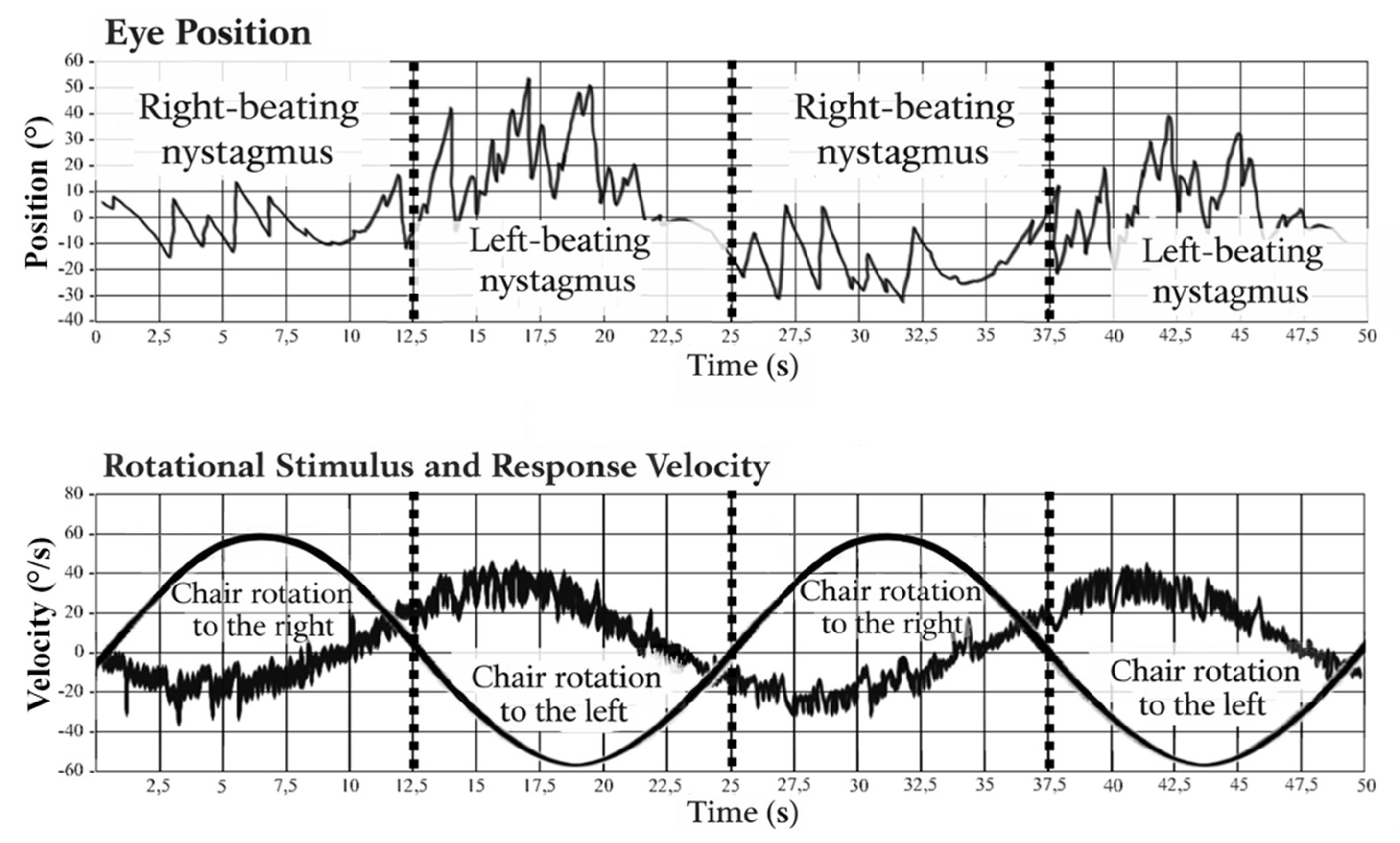
2.3.3. Caloric Test
2.4. Assessment of Motor Development
2.5. Statistical Analysis
2.6. Ethical Approval
3. Results
3.1. Study Population
3.2. Vestibular Function: Comparison Between Groups
3.2.1. Sinusoidal Harmonic Acceleration (SHA)
3.2.2. Video Head Impulse Test (vHIT)
3.2.3. Caloric Test
3.3. Motor Development
3.4. Neurological Disorders
3.5. Genetic Disorders
3.6. Risk Factors for Hearing Loss and Vestibular Dysfunction in the Studied Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SNHL | sensorineural hearing loss |
| VOR | vestibulo-ocular reflex |
| vHIT | video head impulse test |
| SHA | sinusoidal harmonic acceleration |
| VEMP | vestibular-evoked myogenic potentials |
| cVEMP | cervical vestibular-evoked myogenic potentials |
References
- Bouchlarhem S, Achraf S, Drissia B, Adil E, Azeddine L, Fahd EAEI. Congenital deafness: from screening to management. Ann Med Surg (Lond). 2025;87(6):3236-3243. Published 2025 May 21. [CrossRef]
- Zachreini, I., Bashiruddin, J., Zizlavsky, S., Tamin, S., Priyono, H., Mayangsari, I. D., … Fitria, H. (2022). Financing hearing aids for patients with congenital deafness in Indonesia. Oto Rhino Laryngologica Indonesiana, 52(1). [CrossRef]
- Jones SM, Jones TA, Mills KN, Gaines GC. Anatomical and Physiological Considerations in Vestibular Dysfunction and Compensation. Semin Hear. 2009;30(4):231-241. [CrossRef]
- Verbecque E, Marijnissen T, De Belder N, et al. Vestibular (dys)function in children with sensorineural hearing loss: a systematic review. Int J Audiol. 2017;56(6):361-381. [CrossRef]
- Ghai S, Hakim M, Dannenbaum E, Lamontagne A. Prevalence of Vestibular Dysfunction in Children With Neurological Disabilities: A Systematic Review. Front Neurol. 2019;10:1294. Published 2019 Dec 17. [CrossRef]
- Jefferies TJ, Jozsa F. Neuroanatomy, Vestibular Pathways. In: StatPearls. Treasure Island (FL): StatPearls Publishing; November 8, 2025.
- Gerdsen M, Hundscheid TM, Boudewyns A, Van Rompaey V, Van De Berg R, Widdershoven JCC. Vestibular assessment in children with sensorineural hearing loss: diagnostic accuracy and a proposed diagnostic algorithm. Front Neurol. 2024;15:1349554. Published 2024 Feb 1. [CrossRef]
- Gerdsen M, Schuurman BG, Boudewyns A, van de Berg R, Widdershoven JCC. Diagnosing Vestibular Hypofunction in Children with Sensorineural Hearing Loss: Using the Video Head Impulse Test or the Caloric Test First, Not the Cervical Vestibular Evoked Myogenic Potential. J Clin Med. 2025;14(8):2721. Published 2025 Apr 15. [CrossRef]
- Kjærsgaard JB, Kingma H. Opinion: the neural basis of vestibular evoked myogenic potentials. The cVEMP is a specific indicator of saccular function. Front Neurol. 2025;16:1705995. Published 2025 Nov 26. [CrossRef]
- Olusanya BO, Neumann KJ, Saunders JE. The global burden of disabling hearing impairment: a call to action. Bull World Health Organ. 2014;92(5):367-373. [CrossRef]
- Humes LE. The World Health Organization’s hearing-impairment grading system: an evaluation for unaided communication in age-related hearing loss. Int J Audiol. 2019;58(1):12-20. [CrossRef]
- van de Berg R, Rosengren S, Kingma H. Laboratory examinations for the vestibular system. Curr Opin Neurol. 2018;31(1):111-116. [CrossRef]
- MacDougall HG, Weber KP, McGarvie LA, Halmagyi GM, Curthoys IS. The video head impulse test: diagnostic accuracy in peripheral vestibulopathy. Neurology. 2009;73(14):1134-1141. [CrossRef]
- Rine RM, Wiener-Vacher S. Evaluation and treatment of vestibular dysfunction in children. NeuroRehabilitation. 2013;32(3):507-518. [CrossRef]
- Wiener-Vacher SR, Hamilton DA, Wiener SI. Vestibular activity and cognitive development in children: perspectives. Front Integr Neurosci. 2013;7:92. Published 2013 Dec 11. [CrossRef]
- Janky KL, Rodriguez AI. Quantitative Vestibular Function Testing in the Pediatric Population. Semin Hear. 2018;39(3):257-274. [CrossRef]
- Weber KP, Aw ST, Todd MJ, McGarvie LA, Curthoys IS, Halmagyi GM. Head impulse test in unilateral vestibular loss: vestibulo-ocular reflex and catch-up saccades. Neurology. 2008;70(6):454-463. [CrossRef]
- Blödow A, Pannasch S, Walther LE. Detection of isolated covert saccades with the video head impulse test in peripheral vestibular disorders. Auris Nasus Larynx. 2013;40(4):348-351. [CrossRef]
- Cushing SL, Papsin BC, Rutka JA, James AL, Gordon KA. Evidence of vestibular and balance dysfunction in children with profound sensorineural hearing loss using cochlear implants. Laryngoscope. 2008;118(10):1814-1823. [CrossRef]
- Franco Sánchez I., Uricel Y., Valencia Ramírez S., Castillo-Bustamante M., Madrigal J. Quality of life in patients with vestibular disorders: A narrative review. Int. J. Otorhinolaryngol. Head Neck Surg. 2023, 9, 426–434. [CrossRef]
- Śpiewak P., Śpiewak M. Regarding the Vestibular Infant Screening Flanders (VIS-Flanders) project—a critique. Pol. Otorhinolaryngol. Rev. 2021, 10, 55–58. [CrossRef]
- Bisdorff A, Von Brevern M, Lempert T, Newman-Toker DE. Classification of vestibular symptoms: towards an international classification of vestibular disorders. J Vestib Res. 2009;19(1-2):1-13. [CrossRef]
- Cherchi M, Yacovino DA. Histology and neuroanatomy suggest a unified mechanism to explain the distribution of lesion patterns in acute vestibular neuropathy. Exp Brain Res. 2021;239(5):1395-1399. [CrossRef]
- Kaga K. Vertigo and Balance Disorders in Children; Springer: Tokyo, Japan, 2014; pp. 28–31.
| Control group n=100 |
Moderate hearing loss n=43 |
Severe hearing loss n=29 |
Profound hearing loss n=27 |
p-value | ||||
|---|---|---|---|---|---|---|---|---|
| Age [years] | 15 (10;21.75) | 14 (10;18) | 13 (10.5;23) | 15 (11;24) | 0.72 | |||
| Sex | Female | 46 (46%) | 19 (44.2%) | 17 (58.6%) | 11 (40.7%) | 0.54 | ||
| Male | 54 (54%) | 24 (55.8%) | 12 (41.4%) | 16 (59.3%) | ||||
| Better-hearing ear | Right | 22 (22%) | 13 (30.2%) | 6 (20.7%) | 9 (33.3%) | 0.51 | <0.001 | |
| Left | 13 (13%) | 19 (44.2%) | 15 (51.7%) | 14 (51.9%) | <0.001 | |||
| Same hearing threshold | 65 (65%) | 11 (25.6%) | 8 (27.6%) | 4 (14.8%) | <0.001 | |||
| Hearing loss [dB] | 15 (10;20) | 60 (55;60) | 90 (80;90) | 110 (100;120) | <0.001 | |||
| Tympanogram type for right ear | A | 100 (100%) | 43 (100%) | 23 (79.3%) | 26 (96.3%) | <0.001 | <0.001 | |
| B | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |||
| C | 0 (0%) | 0 (0%) | 6 (20.7%) | 1 (3.7%) | <0.001 | |||
| Tympanogram type for the left ear | A | 100 (100%) | 43 (100%) | 23 (79.3%) | 26 (96.3%) | <0.001 | <0.001 | |
| B | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | - | |||
| C | 0 (0%) | 0 (0%) | 6 (20.7%) | 1 (3.7%) | <0.001 | |||
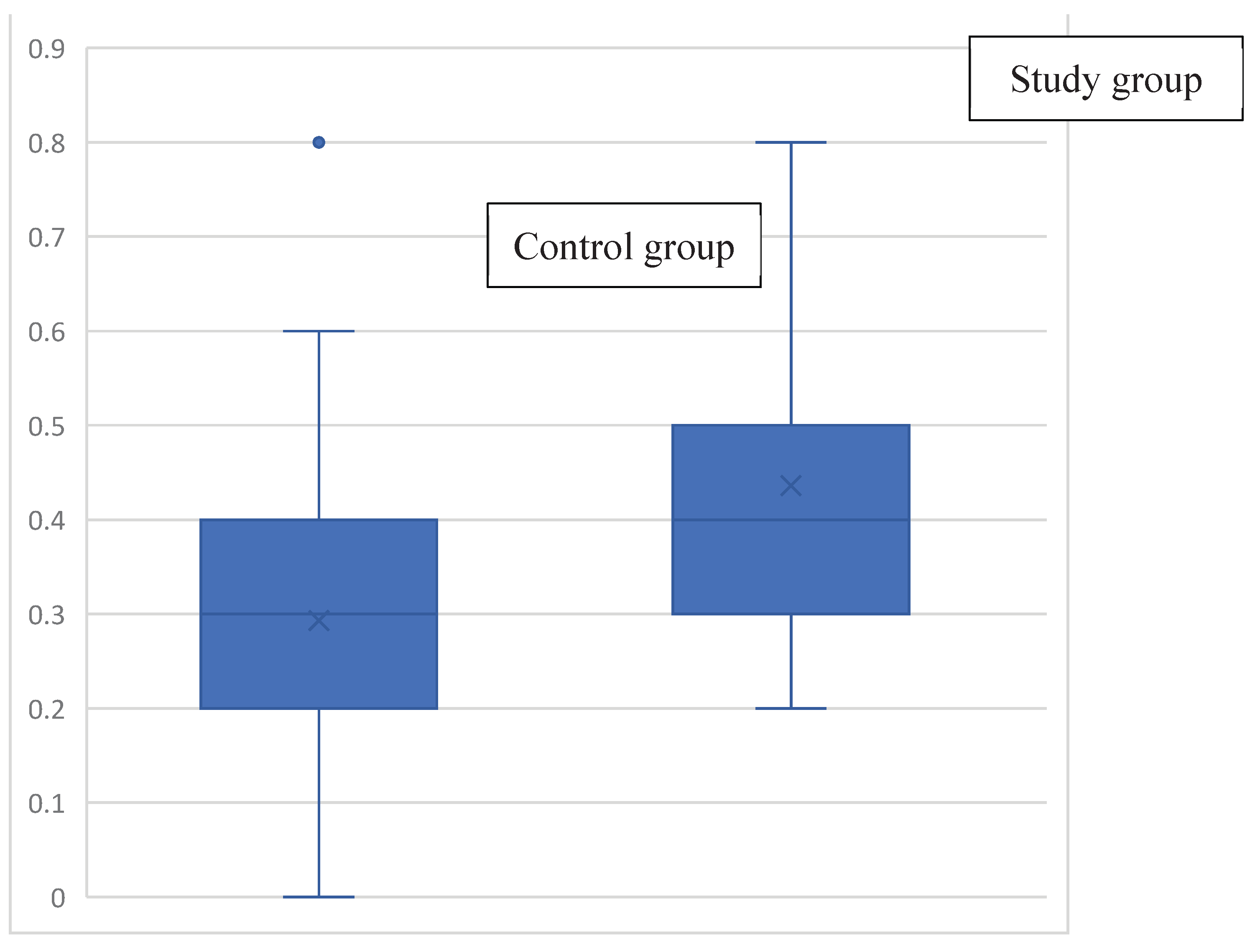
| VOR gain value in the SHA test | 0.4 (0.3;0.5) | 0.3 (0.2;0.5) | 0.2 (0.1;0.4) | 0.2 (0.1;0.3) | <0.001 | |||
| Unilateral Weakness in caloric test [%] | 14 (7;23) | 11 (6;20) | 14 (6.5;26.5) | 14 (6;20) | 0.54 | |||
| Directional Preponderance in caloric test [%] | 13 (6;19.5) | 10 (4;17) | 12 (7.5;17) | 13 (7;22) | 0.28 | |||
| Total response—right ear in caloric test [°/s] | 22.18 (14.88;31.6) | 28.54 (15.61;37.13) | 28.22 (17.99; 39.75) |
30.05 (24.64; 33.52) |
0.051 | |||
| Total response—left ear in caloric test [°/s] | 29.62 (16.5; 40.13) |
26.3 (15.78;31.46) | 29.83 (16.01; 37.04) |
34.53 (20.05; 43.82) |
0.06 | |||
| VOR gain in vHIT for the right side | 0.96 (0.845; 1.073) |
0.95 (0.85;1.05) | 0.95 (0.835;1.05) | 0.95 (0.88;1.03) | 0.92 | |||
| VOR gain in vHIT for the left side | 1 (0.888; 1.07) |
0.99 (0.95;1.11) | 0.95 (0.78;1.115) | 1.07 (0.89;1.15) | 0.23 | |||
| Saccades | None | 100 (100%) | 35 (81.4%) | 25 (86.2%) | 24 (88.9%) | <0.001 | <0.001 | |
| Rightward | 0 (0%) | 5 (11.6%) | 2 (6.9%) | 0 (0%) | 0.002 | |||
| Leftward | 0 (0%) | 0 (0%) | 1 (3.4%) | 1 (3.7%) | 0.077 | |||
| Bilateral | 0 (0%) | 3 (7%) | 1 (3.4%) | 2 (7.4%) | 0.019 | |||
| Age of independent walking [months] | 12 (12;12) | 12 (12;15) | 12 (11;14.5) | 15 (12;18) | <0.001 | |||
| Neurological disorders | 0 (0%) | 11 (25.6%) | 11 (37.9%) | 9 (33.3%) | <0.001 | |||
| Genetic disorders (confirmed and under diagnostic evaluation) |
0 (0%) | 6 (14%) | 6 (20.7%) | 3 (11.1%) | <0.001 | |||
| Genetic disorders (confirmed only) |
0 (0%) | 4 (9.3%) | 1 (3.4%) | 2 (7.4%) | 0.002 | |||
| Hearing loss severity | Mild hearing loss n=3 |
Moderate hearing loss n=43 |
Severe hearing loss n=29 |
Profound hearing loss n=27 |
Total | |
|---|---|---|---|---|---|---|
| The number of neurologically evaluated patients | 0 (0%) | 11 (25.6%) | 11 (37.9%) | 9 (33.3%) | 31 | |
| Indications for consultation | Hypotonia | 0 | 9 | 8 | 8 | 25 |
| Hypertonia | 0 | 1 | 1 | 1 | 3 | |
| Other | 0 | migraine with aura (n=1) | epilepsy (n=1) | vertigo (n=1) | 6 | |
| Parry–Romberg syndrome (n=1) | Impaired balance up to 2 years of age (n=1) | Antley–Bixler syndrome (n=1) | ||||
| Severity of hearing loss | Mild hearing loss | Moderate hearing loss | Severe hearing loss | Profound hearing loss |
|---|---|---|---|---|
| Confirmed mutations | none | ‣ 9p22 deletion | ‣ c.35delG in GJB2 gene | ‣ mutation p.Glu18 7 |
| ‣ p.L90P in GJB2 gene | ‣ 10q26 in the FGFR2 gene (Antley-Bixler syndrome) | |||
| ‣ 6p21.32 in COL11A2 gene (Stickler syndrome type 3) | ||||
| ‣ c.362dupC in FKBP14 gene | ||||
| ‣ p.E120del in GJB2 gene | ||||
| Number of patients undergoing genetic testing | 0 | 1 | 5 | 0 |
| Number of patients without available test results | 0 | 1 | 0 | 1 |
| Risk factors | Control group n=100 |
Mild hearing loss n=3 |
Moderate hearing loss n=43 | Severe hearing loss n=29 |
Profound hearing loss n=27 |
Total |
|---|---|---|---|---|---|---|
| Number of patients with confirmed risk factors | 9 (9%) |
1 (33.3%) | 9 (20.9%) | 8 (27.6%) | 8 (29.6%) | 35 (100%) |
| Genetically confirmed mutation | 0 | 0 | 4 | 1 | 2 | 7 (20%) |
| confirmed congenital TORCH infection | 0 | 1 | 2 | 4 | 2 | 9 (25.7%) |
| Maternal diabetes during pregnancy | 9 | 0 | 5 | 5 | 4 | 23 (65.7%) |
| Congenital anomaly of the head or neck | 0 | 0 | 1 | 0 | 1 | 2 (5.7%) |
| Exposure to ototoxic drugs | 0 | 0 | 0 | 1 | 0 | 1 (2.9%) |
| jaundice requiring exchange transfusion | 1 | 0 | 2 | 4 | 3 | 10 (28.6%) |
| Prematurity (<37 weeks of gestation) | 3 | 1 | 4 | 5 | 3 | 16 (45.7%) |
| Low birth weight (<2500 g) | 2 | 1 | 4 | 3 | 3 | 13 (37.1%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




