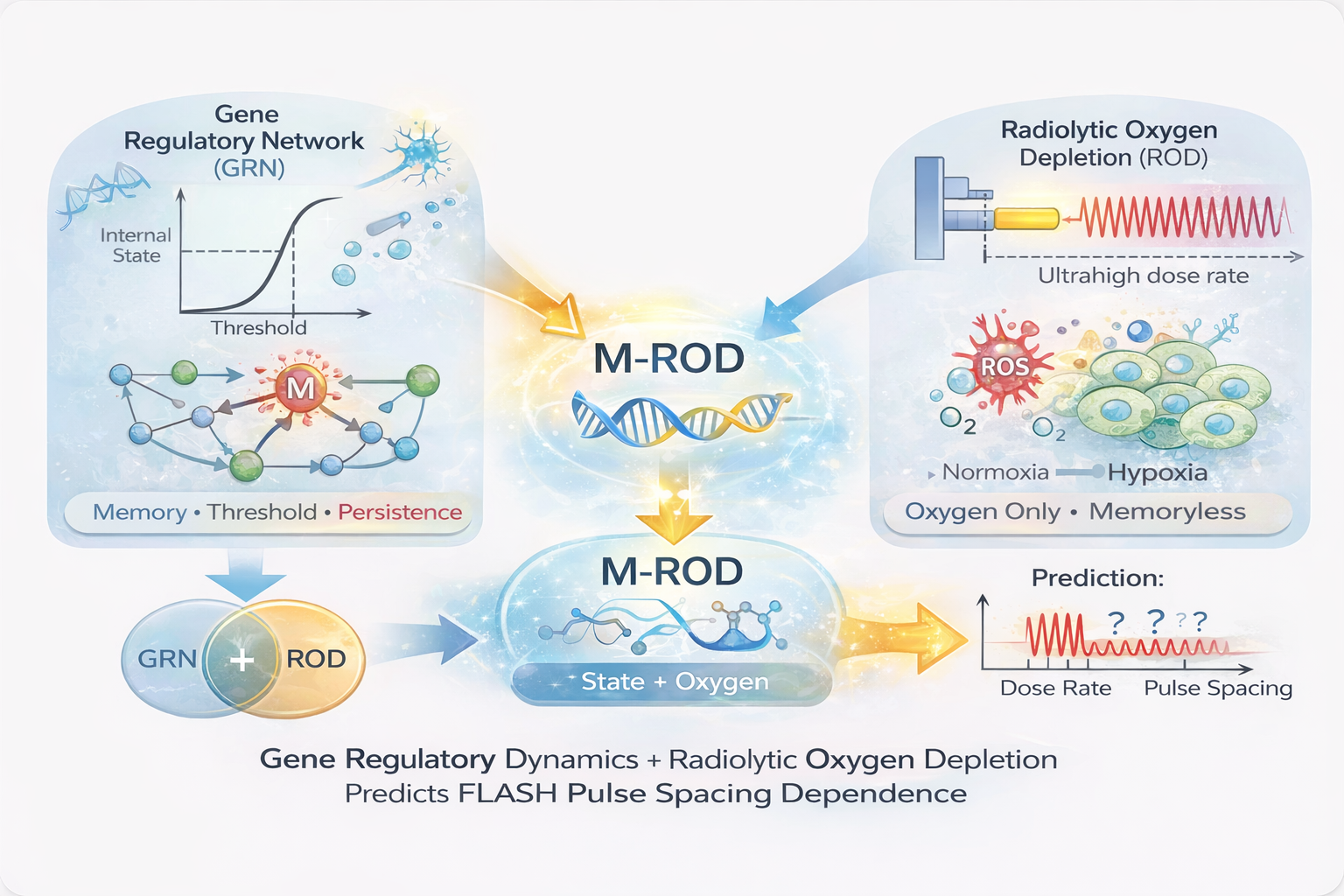
FLASH radiotherapy, characterized by ultra-high dose rates, has been shown to reduce normal tissue toxicity while preserving tumor control, yet its underlying mechanism remains unresolved. Existing models based on radiolytic oxygen depletion (ROD) successfully capture dose-rate dependence but fail to explain key experimental features, including threshold-like onset, saturation of the sparing effect, and sensitivity to temporal delivery structure. Here, we propose a mechanistic framework — Memory-modulated Radiolytic Oxygen Depletion (M-ROD) — that extends classical ROD by incorporating a bounded, history-dependent internal state. The dynamical structure of this state — cooperative activation, bounded feedback, and characteristic decay — is consistent with that of cooperative biological regulatory processes, including gene regulatory networks. In this framework, dose-rate–dependent stress activates a nonlinear biological state that evolves through induction, bounded feedback, and decay, modulating radiosensitivity alongside oxygen effects. We show that the framework reproduces the defining characteristics of FLASH, including sharp threshold-like transitions, plateau behavior, and strong dependence on pulse spacing, duty cycle, and irradiation sequence, while reducing to conventional radiobiology under low dose-rate conditions. The pulse-spacing sensitivity that distinguishes M-ROD from memoryless models requires the state to relax on a characteristic timescale τ_M of approximately 10–100 ms; we show that bioelectric membrane dynamics, treated as a passive RC system using parameter values from standard electrophysiology, naturally produce relaxation in this range without parameter tuning. The model predicts that the magnitude of the FLASH effect is governed by the extent of state activation rather than dose rate alone, providing a mechanistic explanation for variability across experiments. These results support the interpretation of FLASH as an emergent state transition in a dynamical biological system and offer experimentally testable predictions that distinguish it from memoryless models.