Submitted:
27 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
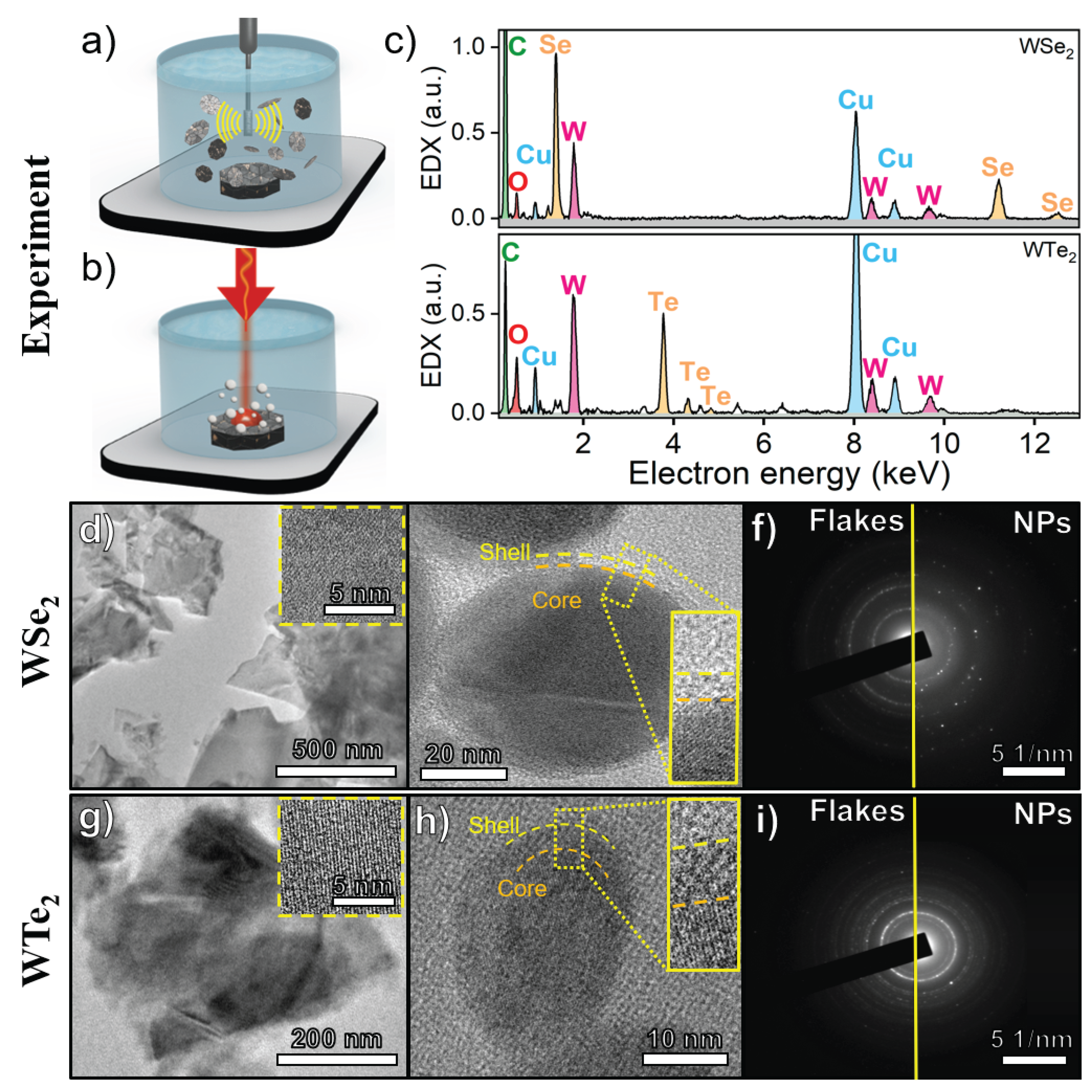
2.1. Synthesis of TMDC Nanomaterials
2.1.1. Nanoparticle Synthesis via Femtosecond PLAL
2.1.2. Flake Synthesis via Ultrasonic Exfoliation
2.2. Structural and Morphological Characterization
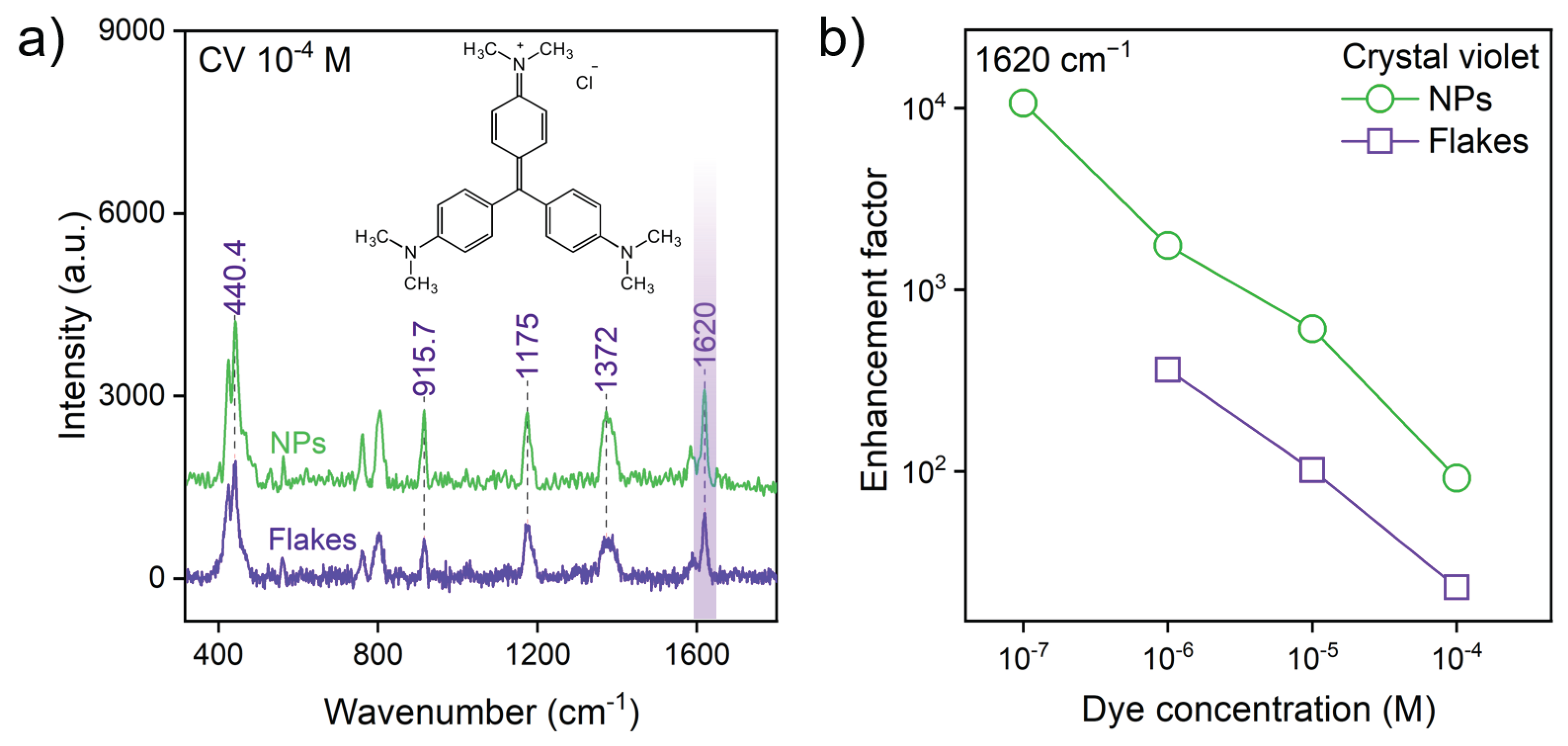
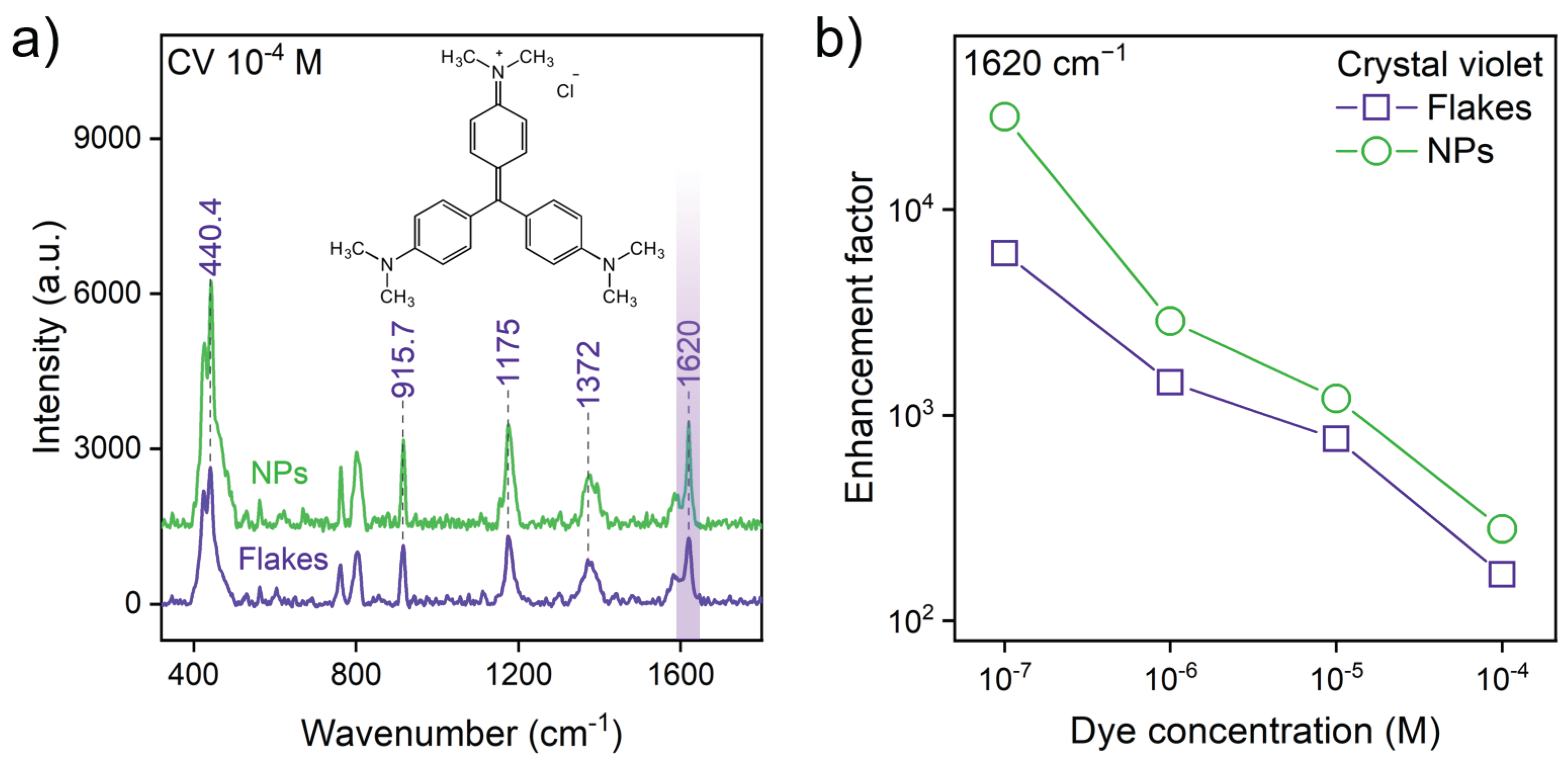
2.3. SERS Substrate Fabrication and Measurements
2.4. Enhancement Factor (EF) Calculation
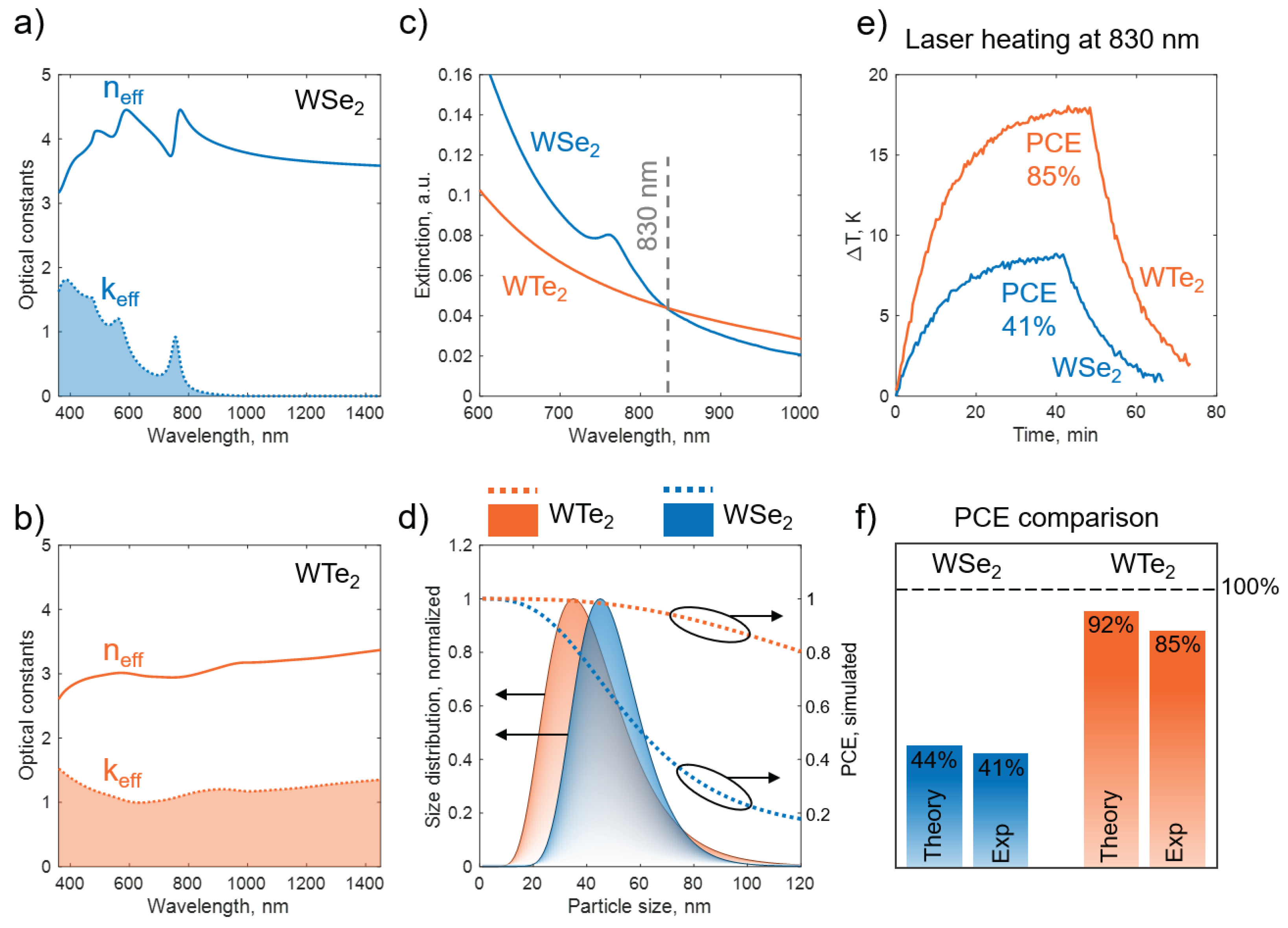
2.5. Photothermal Studies
3. Results and Discussion
3.1. Synthesis Dictates Nanomaterial Morphology while Preserving Crystallinity
3.2. Nanoparticle Morphology Unlocks Superior SERS Performance
3.3. Colloidal Photothermal Response Governed by Bulk Effective Optical Constants
Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| LSPR | Localized surface plasmon resonances |
| CM | Chemical mechanism |
| PICT | Photoinduced charge transfer |
| DI water | Deionized water |
| TMDC | Transition Metal Dichalcogenide |
| NP | Nanoparticle |
| PLAL | Pulsed Laser Ablation in Liquid |
| TEM | Transmission Electron Microscope |
| HRTEM | High Resolution Transmission Electron Microscope |
| SAED | Selected Area Electron Diffraction |
| EDX | Energy Dispersive X-ray Spectroscopy |
| SERS | Surface-Enhanced Raman Scattering |
| CV | Crystal violet |
| LOD | The limit of detection |
| EF | Enhancement Factor |
| NA | Numeric Aperture |
| PCE | Photothermal Conversion Efficiency |
References
- Onyemaobi, I.M.; Xie, Y.; Xu, L.; Zhang, J.; Xiang, L.; Lin, J.; Wu, A. Nanomaterials and clinical SERS technology: broad applications in disease diagnosis. Journal of Materials Chemistry B 2025, pp. 2890–2911.
- Zong, C.; Xu, M.; Xu, L.J.; Wei, T.; Ma, X.; Zheng, X.S.; Hu, R.; Ren, B. Surface-enhanced Raman spectroscopy for bioanalysis: reliability and challenges. Chemical reviews 2018, 118, 4946–4980. [Google Scholar] [CrossRef]
- Liu, Y.; Qin, Z.; Deng, J.; Zhou, J.; Jia, X.; Wang, G.; Luo, F. The advanced applications of 2D materials in SERS. Chemosensors 2022, 10, 455. [Google Scholar] [CrossRef]
- Yu, L.x.; Lv, R.t. Two-dimensional layer materials for highly efficient molecular sensing based on surface-enhanced Raman scattering. New Carbon Materials 2021, 36, 995–1012. [Google Scholar] [CrossRef]
- Tang, X.; Hao, Q.; Hou, X.; Lan, L.; Li, M.; Yao, L.; Zhao, X.; Ni, Z.; Fan, X.; Qiu, T. Exploring and engineering 2D transition metal dichalcogenides toward ultimate SERS performance. Advanced Materials 2024, 36, 2312348. [Google Scholar] [CrossRef] [PubMed]
- Nie, S.; Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. science 1997, 275, 1102–1106. [Google Scholar] [CrossRef] [PubMed]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced Raman scattering (SERS). Physical review letters 1997, 78, 1667. [Google Scholar] [CrossRef]
- Sharma, B.; Frontiera, R.R.; Henry, A.I.; Ringe, E.; Van Duyne, R.P. SERS: Materials, applications, and the future. Materials today 2012, 15, 16–25. [Google Scholar]
- Majumdar, D. 2D Material-Based Surface-Enhanced Raman Spectroscopy Platforms (Either Alone or in Nanocomposite Form) - From a Chemical Enhancement Perspective. ACS omega 2024, 9, 40242–40258. [Google Scholar]
- Langer, J.; Jimenez de Aberasturi, D.; Aizpurua, J.; Alvarez-Puebla, R.A.; Auguie, B.; Baumberg, J.J.; Bazan, G.C.; Bell, S.E.; Boisen, A.; Brolo, A.G.; et al. Present and future of surface-enhanced Raman scattering. ACS nano 2019, 14, 28–117. [Google Scholar] [CrossRef]
- Le Ru, E.; Etchegoin, P. Principles of Surface-Enhanced Raman Spectroscopy: and related plasmonic effects; Elsevier, 2008. [Google Scholar]
- Aroca, R. Surface-Enhanced Vibrational Spectroscopy; Wiley, 2006. [Google Scholar]
- Goel, R.; Vij, R.; Chakraborty, S.; Achanta, V.G.; Dubey, S.K. Application of plasmonic quasi crystal (PIQC) in surface enhanced Raman spectroscopy (SERS). In Proceedings of the Optical Sensors 2023. SPIE, 2023, Vol. 12572, pp. 84–88.
- Lombardi, J.R.; Birke, R.L. A unified approach to surface-enhanced Raman spectroscopy. The Journal of Physical Chemistry C 2008, 112, 5605–5617. [Google Scholar] [CrossRef]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A review on surface-enhanced Raman scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef]
- Karagianni, K.; Leontidou, T.; Constantinou, M.; Andreou, C. Bacterial detection with electrochemical, SERS, and electrochemical SERS sensors. Analyst 2025, 150, 3762–3787. [Google Scholar] [CrossRef]
- Tahir, M.A.; Dina, N.E.; Cheng, H.; Valev, V.K.; Zhang, L. Surface-enhanced Raman spectroscopy for bioanalysis and diagnosis. Nanoscale 2021, 13, 11593–11634. [Google Scholar] [CrossRef]
- Chaudhry, I.; Hu, G.; Ye, H.; Jensen, L. Toward modeling the complexity of the chemical mechanism in SERS. ACS nano 2024, 18, 20835–20850. [Google Scholar] [CrossRef]
- Chen, L.; Liu, H.; Gao, J.; Wang, J.; Jin, Z.; Lv, M.; Yan, S. Development and Biomedical Application of Non-Noble Metal Nanomaterials in SERS. Nanomaterials 2024, 14, 1654. [Google Scholar] [CrossRef]
- Chen, M.; Liu, D.; Du, X.; Lo, K.H.; Wang, S.; Zhou, B.; Pan, H. 2D materials: Excellent substrates for surface-enhanced Raman scattering (SERS) in chemical sensing and biosensing. TrAC Trends in Analytical Chemistry 2020, 130, 115983. [Google Scholar] [CrossRef]
- Ling, X.; Moura, L.; Pimenta, M.A.; Zhang, J. Charge-transfer mechanism in graphene-enhanced Raman scattering. The Journal of Physical Chemistry C 2012, 116, 25112–25118. [Google Scholar] [CrossRef]
- Ling, X.; Xie, L.; Fang, Y.; Xu, H.; Zhang, H.; Kong, J.; Dresselhaus, M.S.; Zhang, J.; Liu, Z. Can graphene be used as a substrate for Raman enhancement? Nano letters 2010, 10, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhao, Y.; Tao, L. Interface engineering in 2D materials for SERS sensing. Frontiers in Materials 2023, 10, 1272826. [Google Scholar] [CrossRef]
- Wan, J.; Lacey, S.D.; Dai, J.; Bao, W.; Fuhrer, M.S.; Hu, L. Tuning two-dimensional nanomaterials by intercalation: materials, properties and applications. Chemical Society Reviews 2016, 45, 6742–6765. [Google Scholar] [CrossRef]
- Akhound, M.A.; Jacobsen, K.W.; Thygesen, K.S. Activating the Basal Plane of 2D Transition Metal Dichalcogenides via High-Entropy Alloying. Journal of the American Chemical Society 2025, 147, 5743–5754. [Google Scholar] [CrossRef]
- Li, H.; Tsai, C.; Koh, A.L.; Cai, L.; Contryman, A.W.; Fragapane, A.H.; Zhao, J.; Han, H.S.; Manoharan, H.C.; Abild-Pedersen, F.; et al. Activating and optimizing MoS2 basal planes for hydrogen evolution through the formation of strained sulphur vacancies. Nature materials 2016, 15, 48–53. [Google Scholar] [CrossRef]
- Huang, B.; Li, N.; Ong, W.J.; Zhou, N. Single atom-supported MXene: how single-atomic-site catalysts tune the high activity and selectivity of electrochemical nitrogen fixation. Journal of Materials Chemistry A 2019, 7, 27620–27631. [Google Scholar] [CrossRef]
- Sim, Y.; Chae, Y.; Kwon, S.Y. Recent advances in metallic transition metal dichalcogenides as electrocatalysts for hydrogen evolution reaction. Iscience 2022, 25, 105098. [Google Scholar] [CrossRef]
- Song, G.; Cong, S.; Zhao, Z. Defect engineering in semiconductor-based SERS. Chemical Science 2022, 13, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, T.F.; Jorgensen, K.P.; Bonde, J.; Nielsen, J.H.; Horch, S.; Chorkendorff, I. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts. science 2007, 317, 100–102. [Google Scholar]
- Nicolosi, V.; Chhowalla, M.; Kanatzidis, M.G.; Strano, M.S.; Coleman, J.N. Liquid exfoliation of layered materials. Science 2013, 340, 1226419. [Google Scholar] [CrossRef]
- Hernandez, Y.; Nicolosi, V.; Lotya, M.; Blighe, F.M.; Sun, Z.; De, S.; McGovern, I.T.; Holland, B.; Byrne, M.; Gun’Ko, Y.K.; et al. High-yield production of graphene by liquid-phase exfoliation of graphite. Nature nanotechnology 2008, 3, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. Journal of Materials Chemistry A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Tselikov, G.I.; Ermolaev, G.A.; Popov, A.A.; Tikhonowski, G.V.; Panova, D.A.; Taradin, A.S.; Vyshnevyy, A.A.; Syuy, A.V.; Klimentov, S.M.; Novikov, S.M.; et al. Transition metal dichalcogenide nanospheres for high-refractive-index nanophotonics and biomedical theranostics. Proceedings of the National Academy of Sciences 2022, 119, e2208830119. [Google Scholar]
- Amendola, V.; Meneghetti, M. Laser ablation synthesis in solution and size manipulation of noble metal nanoparticles. Physical chemistry chemical physics 2009, 11, 3805–3821. [Google Scholar]
- Zhang, D.; Gokce, B.; Barcikowski, S. Laser synthesis and processing of colloids: fundamentals and applications. Chemical reviews 2017, 117, 3990–4103. [Google Scholar] [CrossRef]
- Forsythe, R.C.; Cox, C.P.; Wilsey, M.K.; Muller, A.M. Pulsed laser in liquids made nanomaterials for catalysis. Chemical Reviews 2021, 121, 7568–7637. [Google Scholar] [CrossRef]
- Rao, S.V.; Podagatlapalli, G.K.; Hamad, S. Ultrafast laser ablation in liquids for nanomaterials and applications. Journal of nanoscience and nanotechnology 2014, 14, 1364–1388. [Google Scholar] [CrossRef] [PubMed]
- Maiti, R.; Patil, C.; Saadi, M.; Xie, T.; Azadani, J.; Uluutku, B.; Amin, R.; Briggs, A.; Miscuglio, M.; Van Thourhout, D.; et al. Strain-engineered high-responsivity MoTe2 photodetector for silicon photonic integrated circuits. Nature Photonics 2020, 14, 578–584. [Google Scholar] [CrossRef]
- Kosmider, K.; Fernandez-Rossier, J. Electronic properties of the MoS 2-WS 2 heterojunction. Physical Review B—Condensed Matter and Materials Physics 2013, 87, 075451. [Google Scholar]
- Liu, F.; Ming, P.; Li, J. Ab initio calculation of ideal strength and phonon instability of graphene under tension. Physical Review B—Condensed Matter and Materials Physics 2007, 76, 064120. [Google Scholar] [CrossRef]
- Chavalekvirat, P.; Hirunpinyopas, W.; Deshsorn, K.; Jitapunkul, K.; Iamprasertkun, P. Liquid phase exfoliation of 2D materials and its electrochemical applications in the data-driven future. Precision Chemistry 2024, 2, 300–329. [Google Scholar] [CrossRef] [PubMed]
- Balati, A.; Tek, S.; Nash, K.; Shipley, H. Nanoarchitecture of TiO2 microspheres with expanded lattice interlayers and its heterojunction to the laser modified black TiO2 using pulsed laser ablation in liquid with improved photocatalytic performance under visible light irradiation. Journal of colloid and interface science 2019, 541, 234–248. [Google Scholar] [CrossRef]
- Zavidovskiy, I.A.; Martynov, I.V.; Tselikov, D.I.; Syuy, A.V.; Popov, A.A.; Novikov, S.M.; Kabashin, A.V.; Arsenin, A.V.; Tselikov, G.I.; Volkov, V.S.; et al. Leveraging femtosecond laser ablation for tunable near-infrared optical properties in MoS2-Gold nanocomposites. Nanomaterials 2024, 14, 1961. [Google Scholar] [CrossRef]
- Lasemi, N.; Rupprechter, G. Chemical and laser ablation synthesis of monometallic and bimetallic Ni-based nanoparticles. Catalysts 2020, 10, 1453. [Google Scholar] [CrossRef]
- Ushkov, A.; Dyubo, D.; Belozerova, N.; Kazantsev, I.; Yakubovsky, D.; Syuy, A.; Tikhonowski, G.V.; Tselikov, D.; Martynov, I.; Ermolaev, G.; et al. Tungsten diselenide nanoparticles produced via femtosecond ablation for SERS and theranostics applications. Nanomaterials 2024, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Munkhbat, B.; Wrobel, P.; Antosiewicz, T.J.; Shegai, T.O. Optical constants of several multilayer transition metal dichalcogenides measured by spectroscopic ellipsometry in the 300–1700 nm range: high index, anisotropy, and hyperbolicity. ACS photonics 2022, 9, 2398–2407. [Google Scholar] [CrossRef] [PubMed]
- Abu Serea, E.S.; Orue, I.; Garcia, J.A.; Lanceros-Mendez, S.; Reguera, J. Enhancement and tunability of plasmonic-magnetic hyperthermia through shape and size control of Au: Fe3O4 Janus nanoparticles. ACS Applied Nano Materials 2023, 6, 18466–18479. [Google Scholar] [CrossRef]
- Hohenester, U. Nano and quantum optics: an introduction to basic principles and theory; Springer Nature, 2019. [Google Scholar]
| Materials | Atomic composition (at.%) | Stoichiometry | ||
| W | Se/Te | |||
| WSe2 | Flakes | 30.51 | 61.77 | ∼ 1:2 |
| NPs | 31.08 | 56.31 | ∼ 1:1.8 | |
| WTe2 | Flakes | 29.54 | 59.15 | ∼ 1:2 |
| NPs | 26.20 | 44.38 | ∼ 1:1.7 | |
| Substrate | Synthesis Method | Morphology | Analyte | EF | LOD |
|---|---|---|---|---|---|
| WSe2 | Ultrasonic Exfoliation | Crystalline Flakes | CV | M | |
| WSe2 | Femtosecond PLAL | Crystalline Nanoparticles | CV | ∼ | M |
| WTe2 | Ultrasonic Exfoliation | Crystalline Flakes | CV | M | |
| WTe2 | Femtosecond PLAL | Crystalline Nanoparticles | CV | > | M |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




