Submitted:
28 March 2026
Posted:
31 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
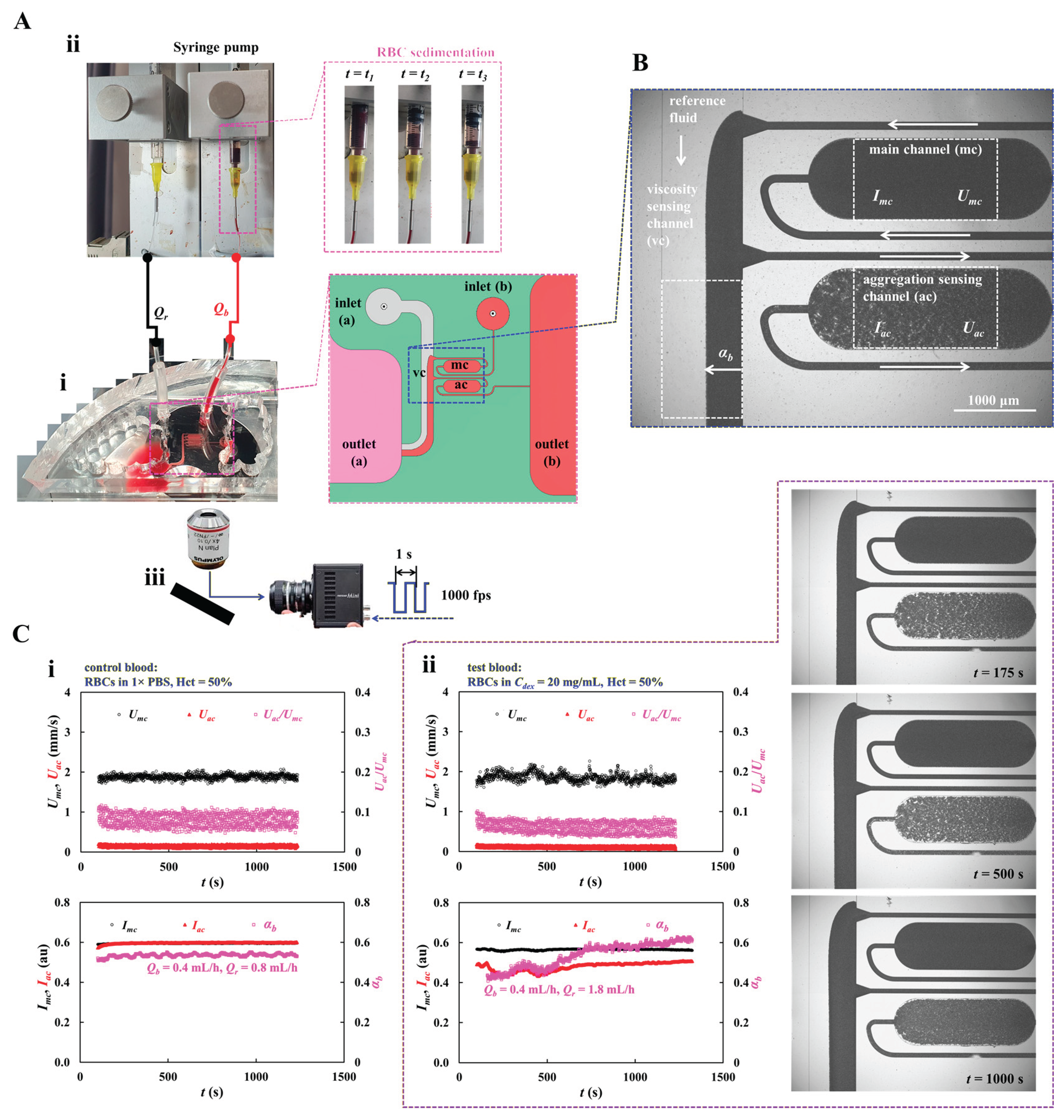
2.1. Microfluidic Chip and Experimental Setup
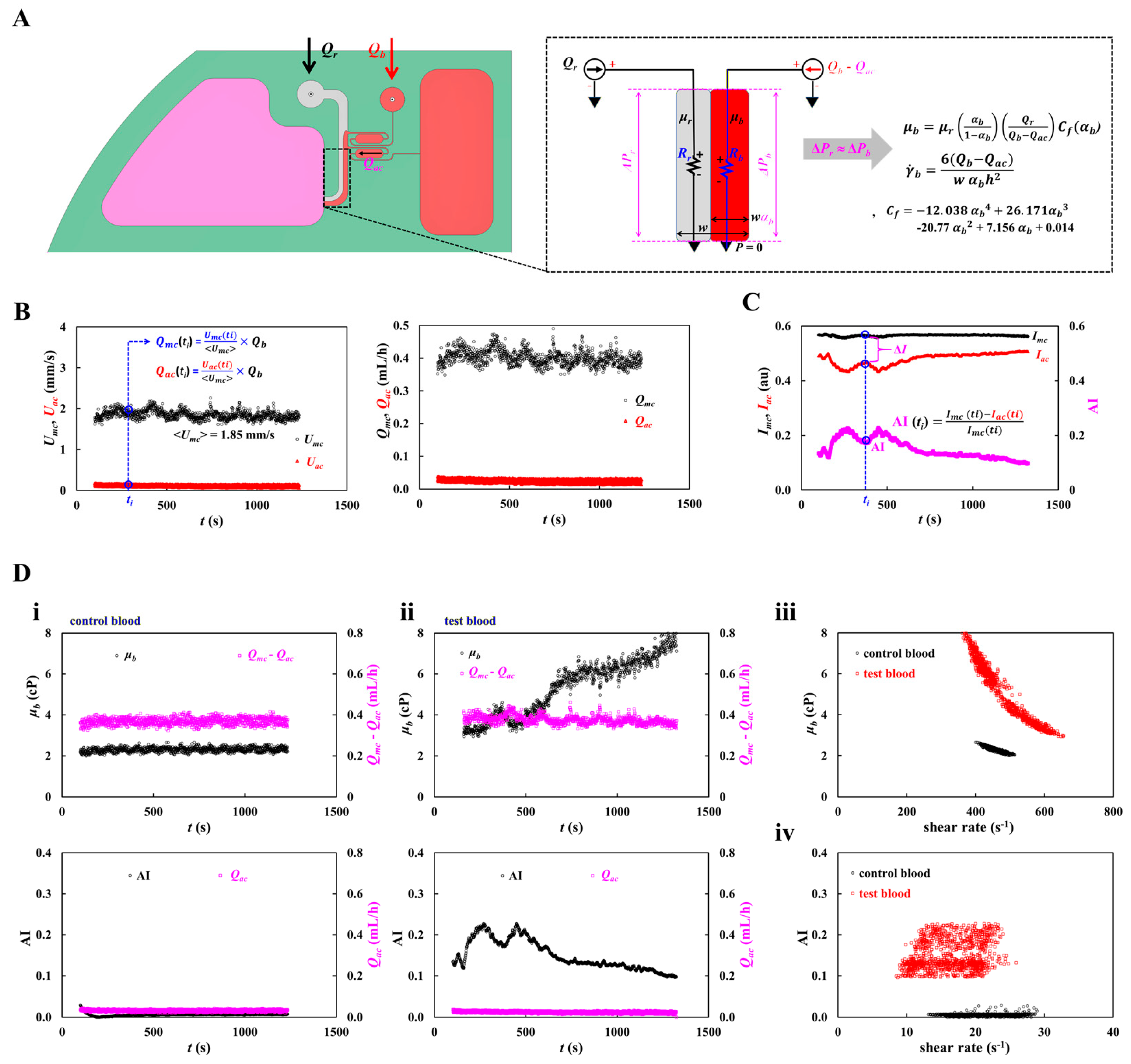
2.2. Quantification of Blood Velocity, Image Intensity, and Interface
2.3. Blood Sample Preparation
3. Results and Discussion
3.1. Quantitative Evaluation of the Proposed Method
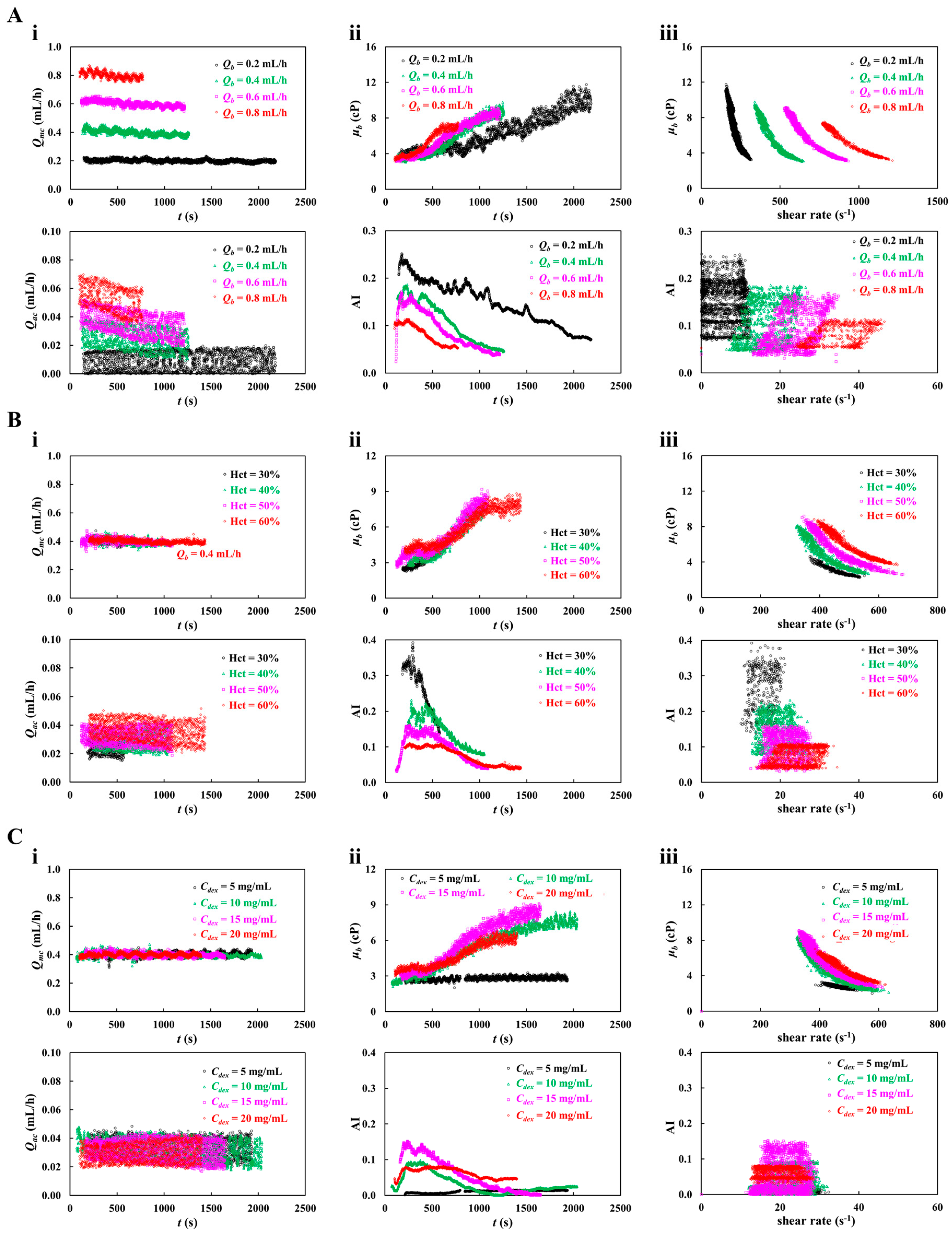
3.2. Contribution of Blood Flow-Rate, Hematocrit, and Suspending Medium
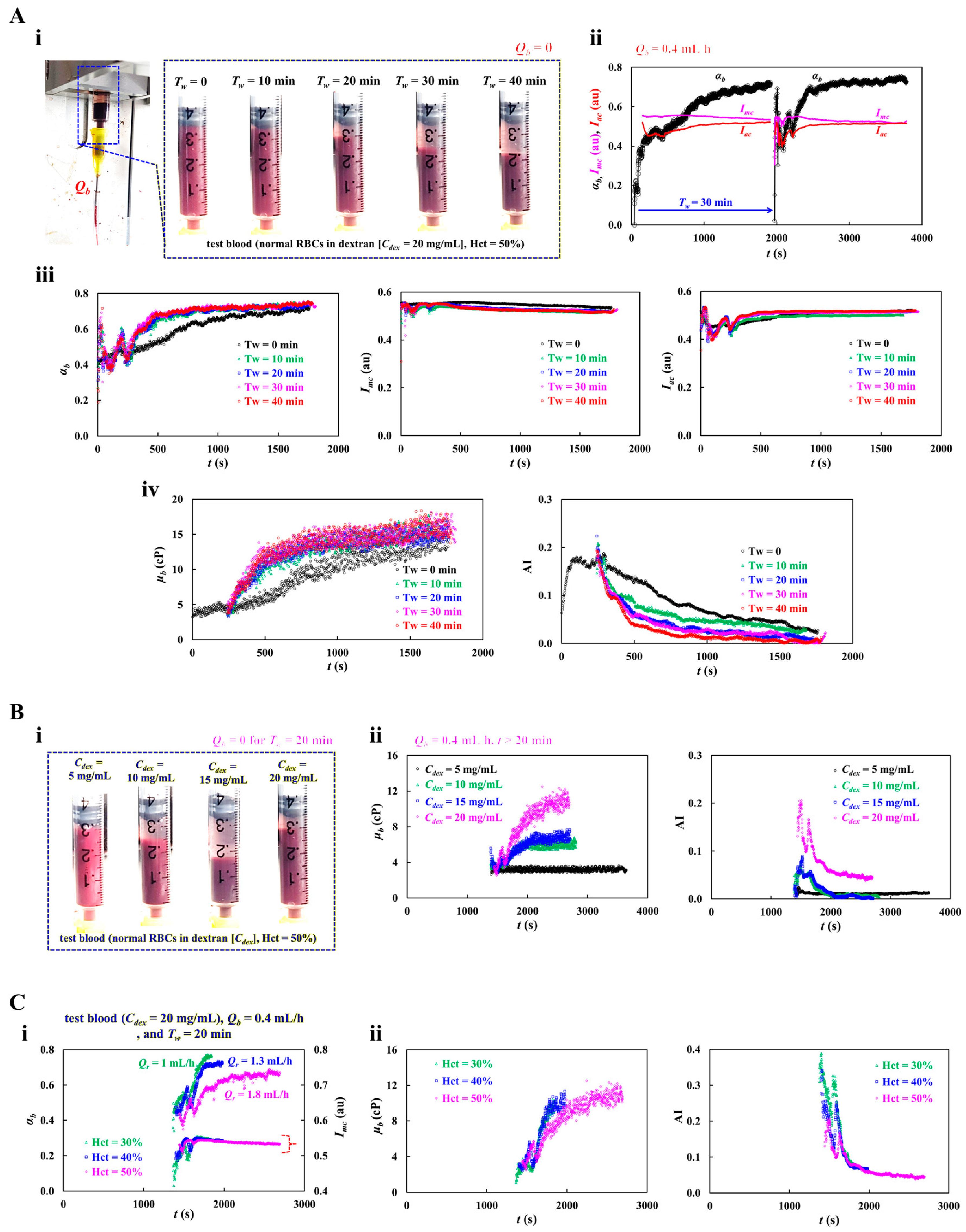
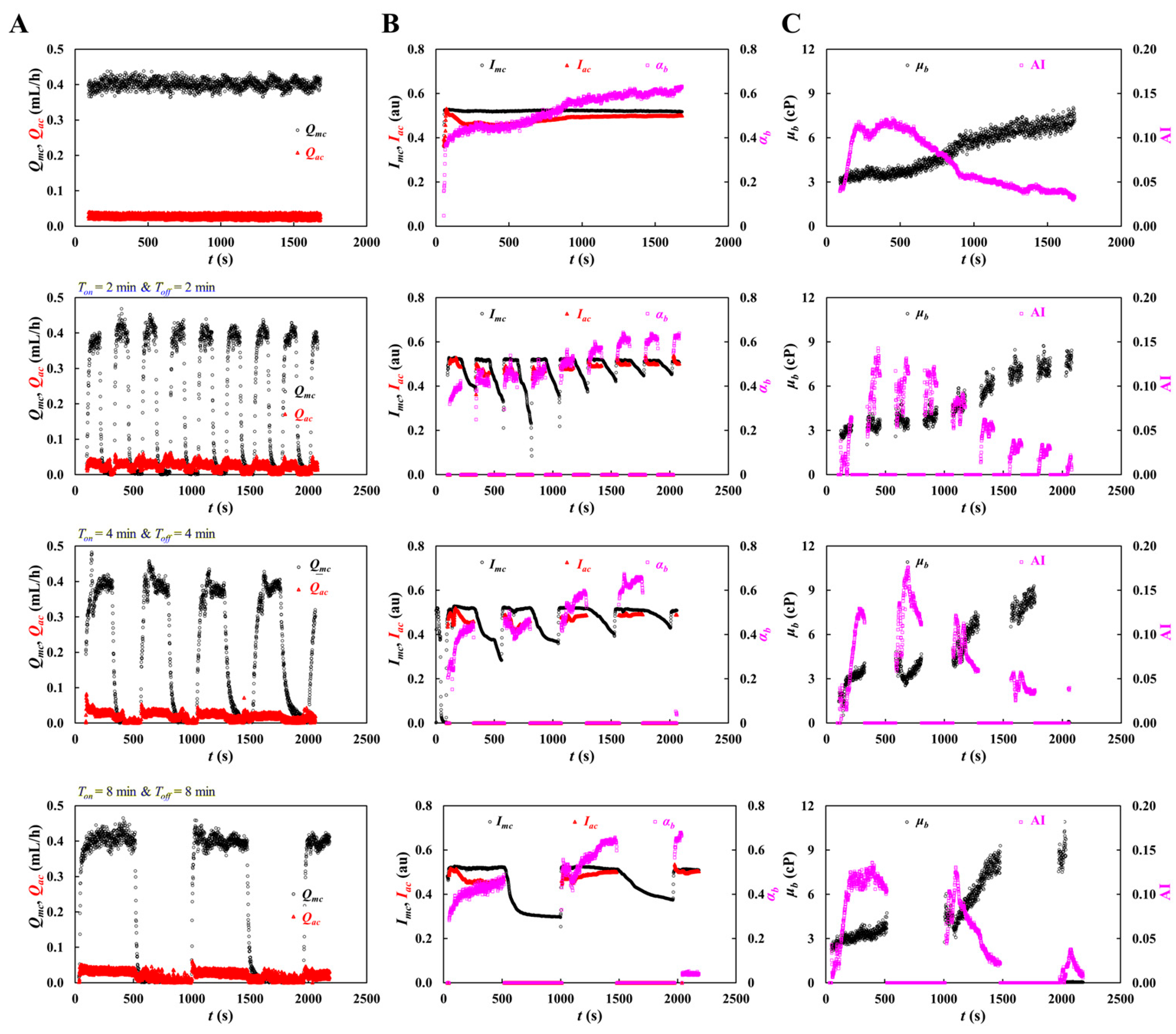
3.3. Evaluation of No-Delivery Waiting Time Under RBCs Sedimentation
3.4. Contribution of Blood Flow Condition in Quantification of Blood Properties
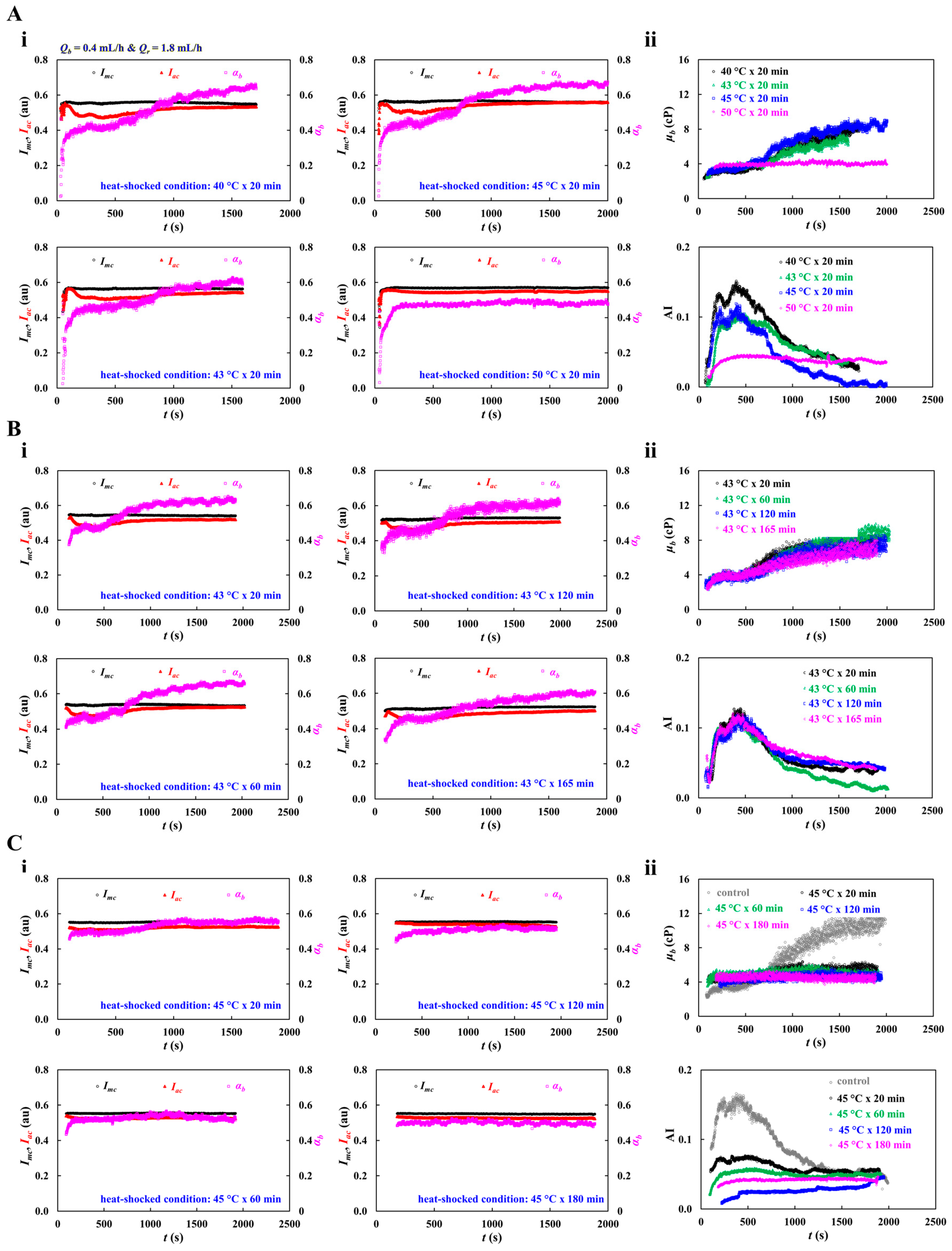
3.5. Quantitative Evaluation of Thermal-Exposed RBCs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cho, Y.I.; Cho, D.J. Hemorheology and microvascular disorders. Korean Circ. J. 2011, 41, 287–95. [Google Scholar] [CrossRef] [PubMed]
- Piagnerelli, M.; Boudjeltia, K.Z.; Vanhaeverbeek, M.; Vincent, J.L. Red blood cell rheology in sepsis. Intensive Care Med. 2003, 29, 1052–1061. [Google Scholar] [CrossRef] [PubMed]
- Rab, M.A.E.; Kanne, C.K.; Boisson, C.; Bos, J.; van Oirschot, B.A.; Houwing, M.E.; Renoux, C.; Bartels, M.; Rijneveld, A.W.; Nur, E.; Cnossen, M.H.; Joly, P.; Nader, E.; Fort, R.; Connes, P.; van Wijk, R.; Sheehan, V.A.; van Beers, E.J. Oxygen gradient ektacytometry-derived biomarkers are associated with acute complications in sickle cell disease. Blood Adv 2024, 8, 276–286. [Google Scholar] [CrossRef]
- Mahawar, M.; Soni, B.; Nayak, A.K. An efficient approach for blood transport in complex interlinked micro-circulatory network. Physics of Fluids 2024, 36, 121903. [Google Scholar] [CrossRef]
- Nader, E.; Skinner, S.; Romana, M.; Fort, R.; Lemonne, N.; Guillot, N.; Gauthier, A.; Antoine-Jonville, S.; Renoux, C.; Hardy-Dessources, M.D.; Stauffer, E.; Joly, P.; Bertrand, Y.; Connes, P. Blood rheology: key parameters, impact on blood flow, role in sickle cell disease and effects of exercise. Front Physiol 2019, 10, 1329. [Google Scholar] [CrossRef]
- Woo, H.G.; Kim, H.G.; Lee, K.M.; Ha, S.H.; Jo, H.; Heo, S.H.; Chang, D.I.; Kim, B.J. Blood viscosity associated with stroke mechanism and early neurological deterioration in middle cerebral artery atherosclerosis. Sci. Rep. 2023, 13, 9384. [Google Scholar] [CrossRef]
- Lu, M.; Rab, M.A.; Shevkoplyas, S.S.; Sheehan, V.A. Blood rheology biomarkers in sickle cell disease. Exp. Biol. Med. (Maywood) 2020, 245, 155–165. [Google Scholar] [CrossRef]
- Kucukal, E.; Man, Y.; Hill, A.; Liu, S.; Bode, A.; An, R.; Kadambi, J.; Little, J.A.; Gurkan, U.A. Whole blood viscosity and red blood cell adhesion: Potential biomarkers for targeted and curative therapies in sickle cell disease. Am. J. Hematol. 2020, 95, 1246–1256. [Google Scholar] [CrossRef]
- Choi, D.; Waksman, O.; Shaik, A.; Mar, P.; Chen, Q.; Cho, D.J.; Kim, H.; Smith, R.L.; Goonewardena, S.N.; Rosenson, R.S. Association of blood viscosity with mortality among patients hospitalized with COVID-19. J. Am. Coll. Cardiol. 2022, 80, 316–328. [Google Scholar] [CrossRef]
- Baskurt, O.K.; Meiselman, H.J. Blood rheology and hemodynamics. Semin. Thromb. Hemost. 2003, 29, 435–450. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I.; Milivojevic, M.; Barshtein, G.; Gural, A. The mechanical properties of erythrocytes are influenced by the conformational state of albumin. Cells 2025, 14, 1139. [Google Scholar] [CrossRef]
- Caglar, S.E.; Karakoc, Y.; Tanoglu, A.; Demirtunc, R.; Tanrikulu, S.; Kilickaya, H.; Ercan, M. Investigation of hemorheology in patients with hyperthyroidism via blood viscosity, erythrocyte deformability and aggregation. Thyroid. Res. 2025, 18, 11. [Google Scholar] [CrossRef]
- Gural, A.; Pajić-Lijaković, I.; Barshtein, G. Mechanical stimulation of red blood cells aging: focusing on the microfluidics application. Micromachines 2025, 16, 259. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Wang, W.S.; Vanapalli, S.A. Microfluidic viscometers for shear rheology of complex fluids and biofluids. Biomicrofluidics 2016, 10, 043402. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, F. A review of microfluidic devices for rheological characterisation. Micromachines (Basel) 2022, 13, 167. [Google Scholar] [CrossRef]
- Kapadia, W.; V Giri, N.; Qin, N.; Zhao, P.; Phan, C.-M.; Haines, L.; Jones, L.; Ren, C.L. A novel microfluidic viscometer for measuring viscosity of ultrasmall volumes of Newtonian and non-Newtonian liquids. Journal of Micromechanics and Microengineering 2025, 35, 055005. [Google Scholar] [CrossRef]
- Chen, W.; Xia, M.; Zhu, W.; Xu, Z.; Cai, B.; Shen, H. A bio-fabricated tesla valves and ultrasound waves-powered blood plasma viscometer. Front Bioeng Biotechnol 2024, 12, 1394373. [Google Scholar] [CrossRef]
- Uno, M.O.; Omori, M.; Sakamoto, K. Nonwoven-fabric-based microfluidic devices for solution viscosity measurements. Sensors & Diagnostics 2024, 3, 1551–1561. [Google Scholar]
- Illibauer, J.; Clodi-Seitz, T.; Zoufaly, A.; Aberle, J.H.; Weninger, W.J.; Foedinger, M.; Elsayad, K. Diagnostic potential of blood plasma longitudinal viscosity measured using Brillouin light scattering. Proc Natl Acad Sci U S A 2024, 121, e2323016121. [Google Scholar] [CrossRef]
- Chang, Y.N.; Yao, D.J. Development of a microfluidic viscometer for non-Newtonian blood analog fluid analysis. Bioengineering (Basel) 2024, 11, 1298. [Google Scholar] [CrossRef]
- Bakhtiaridoost, S.; Musuroi, C.; Volmer, M.; Florescu, M. Optoelectronic microfluidic device for point-of-care blood plasma viscosity measurement. Lab Chip 2024, 24, 3305–3314. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, A.; Haider, D.; Barua, A.; Tanyeri, M.; Erten, A.; Yalcin, O. Machine learning based microfluidic sensing device for viscosity measurements. Sensors & Diagnostics 2023, 2, 1509–1520. [Google Scholar] [CrossRef]
- Gautam, N.; Ram, R.; Bishnoi, V.; Sarkar, A. A low-cost and disposable capillary-based paper sensor for measuring blood-plasma viscosity using a smartphone app. Microfluidics and Nanofluidics 2023, 27, 41. [Google Scholar] [CrossRef]
- Trejo-Soto, C.; Lazaro, G.R.; Pagonabarraga, I.; Hernandez-Machado, A. Microfluidics approach to the mechanical Properties of Red Blood Cell Membrane and Their Effect on Blood Rheology. Membranes (Basel) 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Trejo-Soto, C.; Hernandez-Machado, A. Normalization of blood viscosity according to the hematocrit and the shear rate. Micromachines (Basel) 2022, 13, 357. [Google Scholar] [CrossRef]
- Kim, B.J.; Lee, Y.S.; Zhbanov, A.; Yang, S. A physiometer for simultaneous measurement of whole blood viscosity and its determinants: hematocrit and red blood cell deformability. Analyst 2019, 144, 3144–3157. [Google Scholar] [CrossRef]
- Charansonney, O.L.; Meseguer, E.; Goube, P.; Vicaut, E. Erythrocyte aggregation kinetics for studying the vascular phase of inflammation in patients with suspected acute coronary syndrome or acute stroke. Sci Rep 2025, 15, 38049. [Google Scholar] [CrossRef]
- Lee, C.A.; Farooqi, H.M.U.; Paeng, D.G. Axial shear rate: A hemorheological factor for erythrocyte aggregation under Womersley flow in an elastic vessel based on numerical simulation. Comput Biol Med 2023, 157, 106767. [Google Scholar] [CrossRef] [PubMed]
- Puthumana Melepattu, M.; Maitrejean, G.; Wagner, C.; Podgorski, T. Influence of erythrocyte density on aggregability as a marker of cell age: Dissociation dynamics in extensional flow. J Biomech 2025, 183, 112603. [Google Scholar] [CrossRef]
- Nam, J.H.; Xue, S.; Lim, H.; Shin, S. Study of erythrocyte aggregation at pulsatile flow conditions with backscattering analysis. Clin Hemorheol Microcirc 2012, 50, 257–66. [Google Scholar] [CrossRef]
- Semenov, A.; Lugovtsov, A.; Ermolinskiy, P.; Lee, K.; Priezzhev, A. Problems of red blood cell aggregation and deformation assessed by laser tweezers, diffuse light scattering and laser diffractometry. Photonics 2022, 9, 238. [Google Scholar] [CrossRef]
- You, J.; Park, C.-A.; Kim, A.-K.; Jeon, H.R.; Kim, D.-I.; Shin, S. Ultrasensitive microfluidic detection of red blood cell deformability: Age-related decline in deformability. Physics of Fluids 2025, 37. [Google Scholar]
- Liu, W.; Xie, L.; Yang, J.; Gong, X.; Sun, D.; Zhang, C. A microfluidic device for detecting the deformability of red blood cells. Biosensors (Basel) 2025, 15, 758. [Google Scholar] [CrossRef]
- Kajitani, K.; Ohtani, T.; Higuchi, R.; Chimura, M.; Sera, F.; Tsai, C.D.; Ueda, Y.; Nishimura, J.I.; Sakata, Y. An on-chip deformability checker demonstrates that the severity of iron deficiency is associated with increased deformability of red blood cells. Sci Rep 2025, 15, 19994. [Google Scholar] [CrossRef]
- Williams, D.C.; Wood, D.K. High-throughput quantification of red blood cell deformability and oxygen saturation to probe mechanisms of sickle cell disease. Proc Natl Acad Sci U S A 2023, 120, e2313755120. [Google Scholar] [CrossRef]
- Kang, Y.J. A microfluidic-based blood viscometer. Physics of Fluids 2025, 37, 091904. [Google Scholar] [CrossRef]
- Oh, K.W.; Lee, K.; Ahn, B.; Furlani, E.P. Design of pressure-driven microfluidic networks using electric circuit analogy. Lab Chip 2012, 12, 515–45. [Google Scholar] [CrossRef]
- Alexandrova-Watanabe, A.; Abadjieva, E.; Ivanova, M.; Gartcheva, L.; Langari, A.; Guenova, M.; Tiankov, T.; Nikolova, E.V.; Krumova, S.; Todinova, S. Quantitative assessment of red blood cell disaggregation in chronic lymphocytic leukemia via software image flow analysis. Fluids 2025, 10, 167. [Google Scholar] [CrossRef]
- Namgung, B.; Lee, T.; Tan, J.K.S.; Poh, D.K.H.; Park, S.; Chng, K.Z.; Agrawal, R.; Park, S.Y.; Leo, H.L.; Kim, S. Vibration motor-integrated low-cost, miniaturized system for rapid quantification of red blood cell aggregation. Lab Chip 2020, 20, 3930–3937. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Zhbanov, A.; Yang, S. Microfluidic systems for blood and blood cell characterization. Biosensors (Basel) 2022, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Microfluidic-based effective monitoring of bloods by measuring RBC aggregation and blood viscosity under stepwise varying shear rates. Korea-Aust. Rheol. J. 2020, 32, 15–27. [Google Scholar] [CrossRef]
- Shin, S.; Yang, Y.; Suh, J.S. Measurement of erythrocyte aggregation in a microchip stirring system by light transmission. Clin Hemorheol Microcirc 2009, 41, 197–207. [Google Scholar] [CrossRef]
- Zhbanov, A.; Yang, S. Effects of Aggregation on Blood Sedimentation and Conductivity. PLoS One 2015, 10, e0129337. [Google Scholar] [CrossRef]
- Baskurt, O.K.; Uyuklu, M.; Meiselman, H.J. Time course of electrical impedance during red blood cell aggregation in a glass tube: comparison with light transmittance. IEEE Trans Biomed Eng 2010, 57, 969–78. [Google Scholar] [CrossRef]
- Kang, Y.J. Microfluidic-based technique for measuring RBC aggregation and blood viscosity in a continuous and simultaneous fashion. Micromachines (Basel) 2018, 9, 467. [Google Scholar] [CrossRef] [PubMed]
- Antonova, N.; Khristov, K. Microrheological and microfluidic approaches for evaluation of the mechanical properties of blood cells. Applied Sciences 2025, 15, 8291. [Google Scholar] [CrossRef]
- Kang, Y.J. Continuous and simultaneous measurement of the biophysical properties of blood in a microfluidic environment. Analyst 2016, 141, 6583–6597. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Assessment of continuous flow-dependent red cell aggregation using a microfluidic chip. Applied Sciences 2025, 15, 11481. [Google Scholar] [CrossRef]
- Liang, M.; Ming, D.; Zhong, J.; Shannon, C.S.; Rojas-Carabali, W.; Agrawal, K.; Ai, Y.; Agrawal, R. Pathophysiological associations and measurement techniques of red blood cell deformability. Biosensors (Basel) 2025, 15, 566. [Google Scholar] [CrossRef]
- Higuchi, M.; Watanabe, N. A rapid and accurate method for estimating the erythrocyte sedimentation rate using a hematocrit-corrected optical aggregation index. PLoS One 2022, 17, e0270977. [Google Scholar] [CrossRef]
- Higuchi, M.; Watanabe, N. Determination of the erythrocyte sedimentation rate using the hematocrit-corrected aggregation index and mean corpuscular volume. J. Clin. Lab Anal. 2023, 37, e24877. [Google Scholar] [CrossRef] [PubMed]
- Flormann, D.; Aouane, O.; Kaestner, L.; Ruloff, C.; Misbah, C.; Podgorski, T.; Wagner, C. The buckling instability of aggregating red blood cells. Sci Rep 2017, 7, 7928. [Google Scholar] [CrossRef] [PubMed]
- Joo, J.B.; Kim, K.; Ro, W.B.; Lee, C.M. The erythrocyte sedimentation rate as a novel prognostic marker in canine inflammatory diseases. Animals (Basel) 2025, 16, 40. [Google Scholar] [CrossRef]
- Kang, Y.J. Red blood cell sedimentation index using shear stress of blood flow in microfluidic channel. Biosensors (Basel) 2022, 12, 547. [Google Scholar] [CrossRef]
- Kang, Y.J. Biomechanical investigation of red cell sedimentation using blood shear stress and blood flow image in a capillary chip. Micromachines (Basel) 2023, 14, 1594. [Google Scholar] [CrossRef]
- Thielicke, W.; Stamhuis, E.J. PIVlab – towards user-friendly, affordable and accurate digital particle image velocimetry in MATLAB. Journal of Open Research Software 2014, 2, e30. [Google Scholar] [CrossRef]
- Kloosterman, A.; Poelma, C.; Westerweel, J. Flow rate estimation in large depth-of-field micro-PIV. Experiments in Fluids 2010, 50, 1587–1599. [Google Scholar] [CrossRef]
- Kim, G.; Jeong, S.; Kang, Y.J. Ultrasound standing wave-based cell-to-liquid separation for measuring viscosity and aggregation of blood sample. Sensors (Basel) 2020, 20, 2284. [Google Scholar] [CrossRef]
- Guillot, P.; Panizza, P.; Salmon, J.-B.; Joanicot, M.; Colin, A. Viscosimeter on a microfluidic chip. Langmuir 2006, 22, 6438–6445. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Microfluidic chip for quantitatively assessing hemorheological parameters. Micromachines 2025, 16, 567. [Google Scholar] [CrossRef]
- Kang, Y.J. Periodic and simultaneous quantification of blood viscosity and red blood cell aggregation using a microfluidic platform under in-vitro closed-loop circulation. Biomicrofluidics 2018, 12, 024116. [Google Scholar] [CrossRef]
- Kang, Y.J. Microfluidic-based biosensor for blood viscosity and erythrocyte sedimentation rate using disposable fluid delivery system. Micromachines (Basel) 2020, 11, 215. [Google Scholar] [CrossRef]
- Squires, T.M.; Mason, T.G. Fluid Mechanics of Microrheology. Annual Review of Fluid Mechanics 2010, 42, 413–438. [Google Scholar] [CrossRef]
- Sebastian, B.; Dittrich, P.S. Microfluidics to Mimic Blood Flow in Health and Disease. Annual Review of Fluid Mechanics 2018, 50, 483–504. [Google Scholar] [CrossRef]
- Bosek, M.; Ziomkowska, B.; Pyskir, J.; Wybranowski, T.; Pyskir, M.; Cyrankiewicz, M.; Napiorkowska, M.; Durmowicz, M.; Kruszewski, S. Relationship between red blood cell aggregation and dextran molecular mass. Sci. Rep. 2022, 12, 19751. [Google Scholar] [CrossRef]
- Mehri, R.; Mavriplis, C.; Fenech, M. Red blood cell aggregates and their effect on non-Newtonian blood viscosity at low hematocrit in a two-fluid low shear rate microfluidic system. PLoS One 2018, 13, e0199911. [Google Scholar] [CrossRef] [PubMed]
- Brust, M.; Aouane, O.; Thiebaud, M.; Flormann, D.; Verdier, C.; Kaestner, L.; Laschke, M.W.; Selmi, H.; Benyoussef, A.; Podgorski, T.; Coupier, G.; Misbah, C.; Wagner, C. The plasma protein fibrinogen stabilizes clusters of red blood cells in microcapillary flows. Sci. Rep. 2014, 4, 4348. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Nam, J.H.; Hou, J.X.; Suh, J.S. A transient, microfluidic approach to the investigation of erythrocyte aggregation: the threshold shear-stress for erythrocyte disaggregation. Clin Hemorheol Microcirc 2009, 42, 117–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Neu, B. Role of macromolecular depletion in red blood cell adhesion. Biophys. J. 2009, 97, 1031–1037. [Google Scholar] [CrossRef]
- Dobbe, J.G.; Streekstra, G.J.; Strackee, J.; Rutten, M.C.; Stijnen, J.M.; Grimbergen, C.A. Syllectometry: the effect of aggregometer geometry in the assessment of red blood cell shape recovery and aggregation. IEEE Trans. Biomed. Eng. 2003, 50, 97–106. [Google Scholar] [CrossRef]
- Valerio de Arruda, M.; Cruz Silva, A.; Fernandes Galduroz, J.C.; Ferreira Galduroz, R. Standardization for obtaining blood viscosity: A systematic review. Eur J Haematol 2021, 106, 597–605. [Google Scholar] [CrossRef]
- Buono, M.J.; Krippes, T.; Kolkhorst, F.W.; Williams, A.T.; Cabrales, P. Increases in core temperature counterbalance effects of haemoconcentration on blood viscosity during prolonged exercise in the heat. Exp. Physiol. 2016, 101, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Matrai, A.A.; Varga, G.; Tanczos, B.; Barath, B.; Varga, A.; Horvath, L.; Bereczky, Z.; Deak, A.; Nemeth, N. In vitro effects of temperature on red blood cell deformability and membrane stability in human and various vertebrate species. Clin Hemorheol Microcirc 2021, 78, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.; Shin, T.; Lee, S.S.; Shim, W.; Lee, G.; Lee, S.J.; Kim, Y.; Kim, J.M. Cell stretching measurement utilizing viscoelastic particle focusing. Anal. Chem. 2012, 84, 10471–10477. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Yim, Y.; Ahn, K.H.; Lee, S.J. Extensional flow-based assessment of red blood cell deformability using hyperbolic converging microchannel. Biomed. Microdevices 2009, 11, 1021–1027. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



