Submitted:
25 March 2026
Posted:
26 March 2026
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
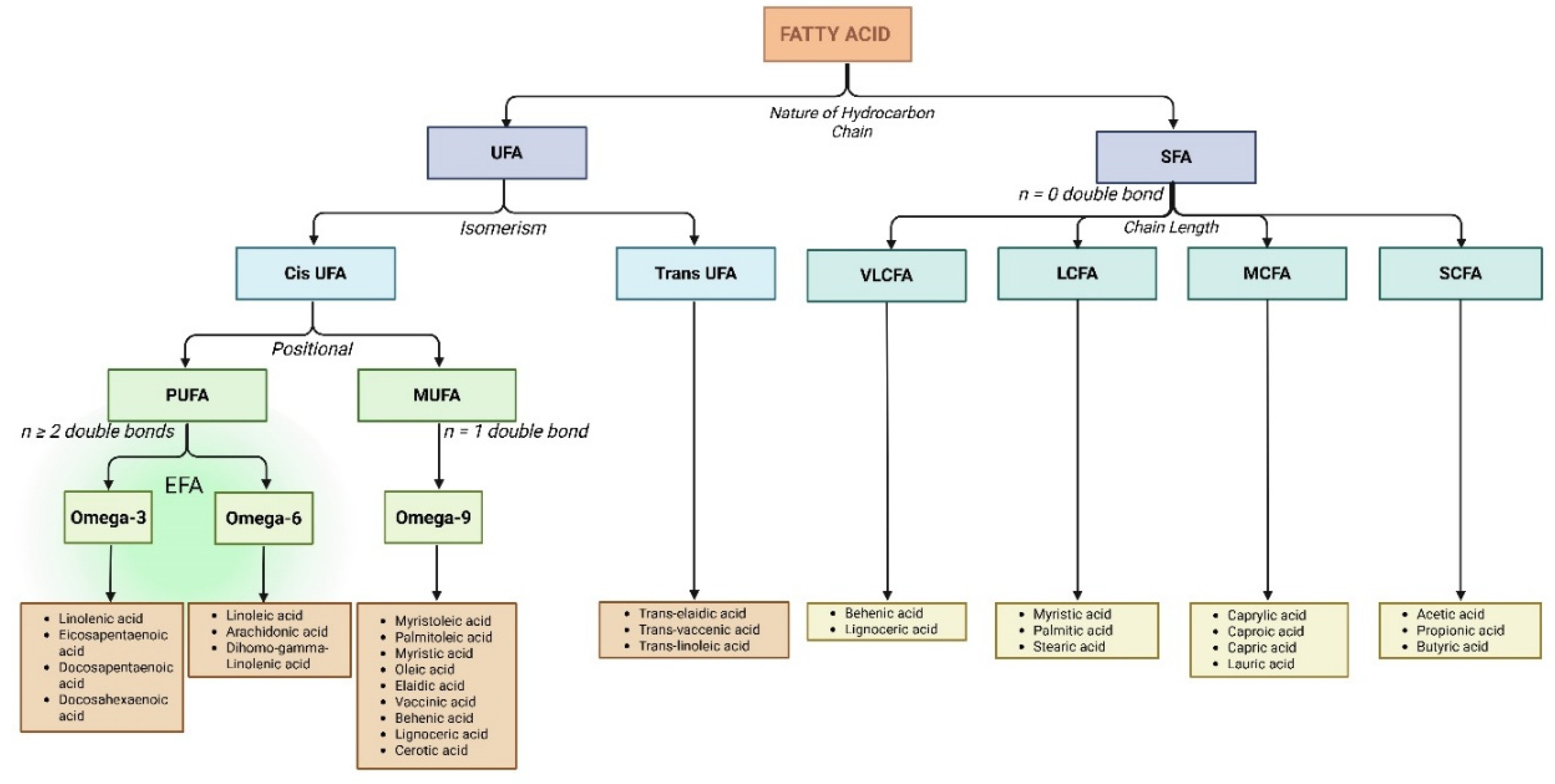
2. Types of Fatty Acids: Structure, Sources and Cardiac Relevance
3. Fatty Acids in Cardiac Physiology
3.1. Role of Fatty Acids in Cardiac Metabolism
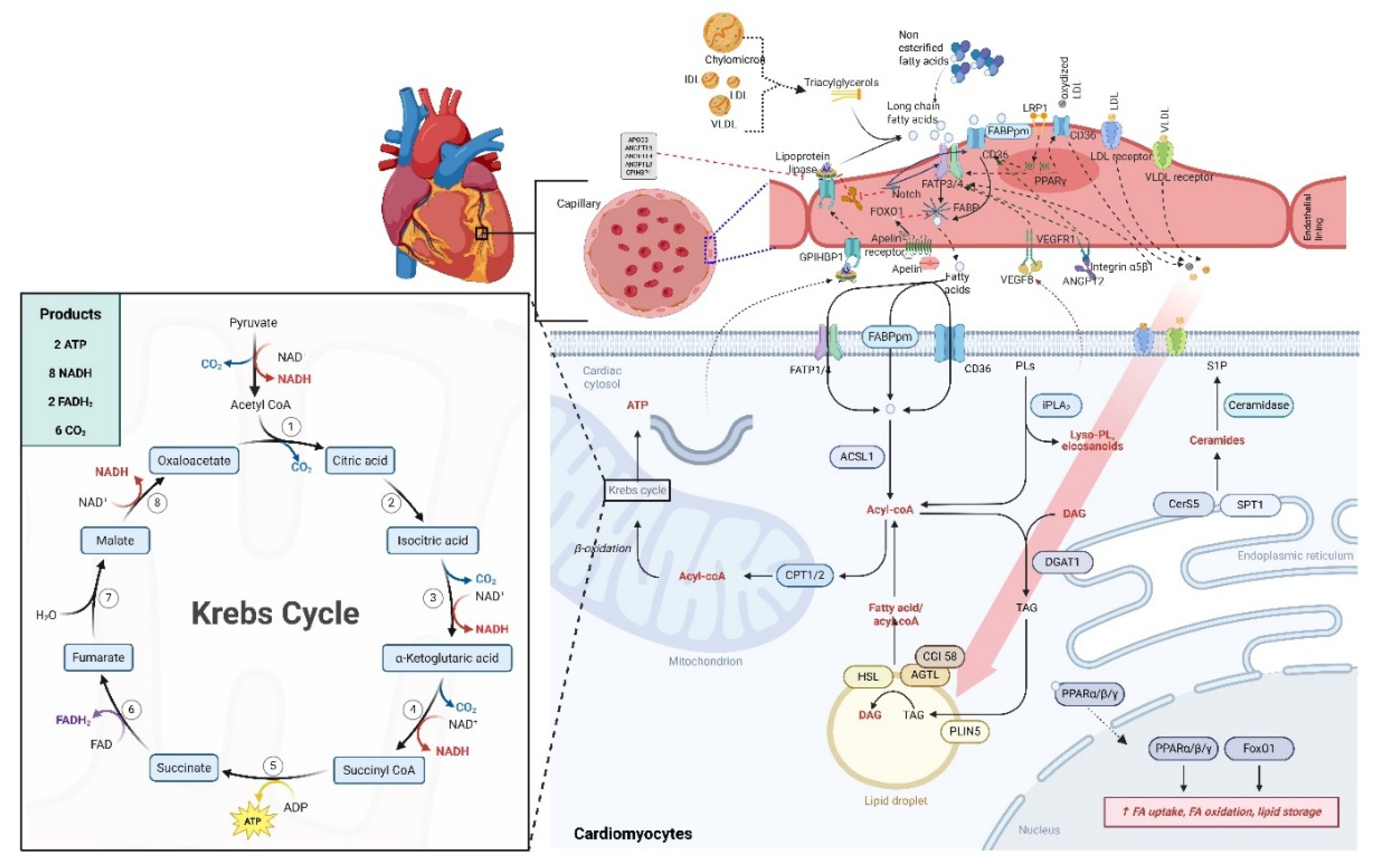
3.2. Fatty Acids as Energy Sources for the Heart
3.3. Mechanisms of Fatty Acid Uptake and Oxidation in Cardiac Cells
3.4. Impact of Different Types of Fatty Acids on Cardiac Function
4. Molecular Pathways and Emerging Research in Fatty Acid-Cardiac Interactions
4.1. Transcriptional and Nuclear Receptor Pathways
PPAR Regulation
AMP-Activated Protein Kinase (AMPK) Energy Sensing
Sirtuin Modulation
4.2. Inflammatory and Epigenetic Networks
Nuclear Factor Kappa B (NF-κB) Inflammatory Cascade
4.3. Membrane Receptors and Lipid Signaling
G-protein-Coupled Receptor (GPCR) Activation by FFAs
Lipid Raft Modulation
Calcium Handling
4.4. Mitochondrial Function and Dynamics
Fatty Acid Oxidation (FAO) Entry
Reactive Oxygen Species (ROS)
Mitochondrial Dynamics
4.5. Emerging Research Frontiers in Cardiac Lipid Biology
Membrane Lipid Remodeling and Microdomain Signaling
n-3 PUFA Incorporation
Cardiolipin (CL) Dynamics
Very Long-Chain SFA (VLSFA) Paradox
Mitochondrial Plasticity and Metabolic Flexibility
Fission-Fusion Balance
Alternative Fuels
Chrononutrition and Circadian Lipid Metabolism
Meal Timing
Time-Restricted Feeding
Gut-Heart Axis Modulation (PLEFA-Compliant)
SCFAs as Oxylipin Adjuvants
TMAO Counteraction
Omics-Driven Discoveries
Lipidomics
Transcriptomics
5. Pathological Effects of Fatty Acids
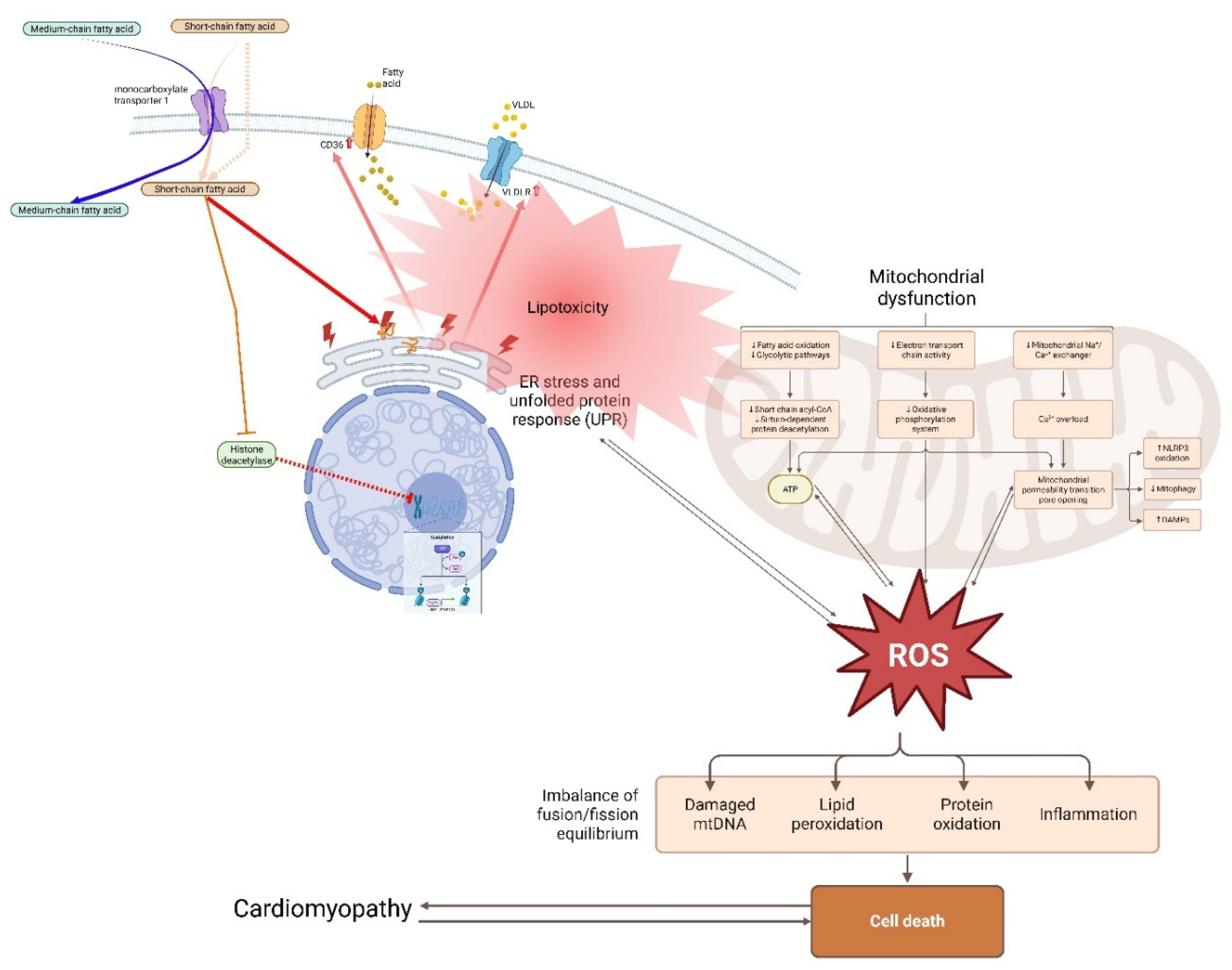
5.1. Lipotoxicity and Metabolic Stress
5.2. Fatty Acid Profiles in Heart Failure Phenotypes
5.3. Ischemia-Reperfusion Injury and Arrhythmia
5.4. Atherosclerosis and Vascular Interactions
6. Clinical Implications and Epidemiological Studies
6.1. Saturated Fatty Acids (SFAs)
6.2. Unsaturated Fatty Acids (UFAs)
6.3. Food Source Considerations
6.4. Timing of Intake
6.5. Reconciling Controversies: A Unifying Perspective
7. Interventional Studies
8. Future Directions
8.1. Future Therapeutic Directions
8.2. Innovative Dietary Strategies
9. Conclusions
- Prioritize whole foods: Choose fish (especially sea fish like sardines, salmon etc), nuts, and extra-virgin olive oil over supplements.
- Avoid industrial trans fats (iTFAs) and minimize consumption of processed meats, refined carbohydrates, sugar-sweetened beverages, and excessive sodium.
- Replace saturated fats with polyunsaturated fats from natural sources, not refined seed oils or processed alternatives.
- Adopt established dietary patterns, such as the Mediterranean or DASH diets, which have consistently been shown to lower CVD risk.
- Use supplements judiciously, only where whole-food alternatives are insufficient or contraindicated.
- Support emerging dietary approaches (e.g., KD, IF) with well-designed randomized trials before broad implementation.
Tables
| Targets | Inhibitors (drug bank ID) | Effects |
| Angiotensin-converting enzyme (ACE) inhibitor | Perindopril/ DB00790 |
Used in combination with Atorvastatin (DB01076) to prevent CVD events [200]. |
| Ramipril/ DB00178 |
Reduction of cardiovascular mortality, MI [201]. | |
| Quinapril/ DB00881 |
Treat hypertension, congestive heart failure [202]. | |
| Lisinopril/ DB00722 |
Treat hypertension, heart failure, and acute MI [203]. | |
| Trandolapril/ DB00519 |
Treat hypertension, congestive heart failure, and improve survival following a MI [204]. | |
| Fosinopril/ DB00492 |
Used to treat mild to moderate hypertension, congestive heart failure [205]. | |
| Enalapril/ DB00584 |
Used to treat mild to moderate hypertension, congestive heart failure [205]. | |
| Acyl-CoA:1,2-diacylglycerol acyltransferase (DGAT) inhibitor | Icosapent ethyl/ DB08887 |
Reduce the risk of MI, stroke, coronary revascularization, elevated triglycerides (≥150 mg/dL) and established cardiovascular disease [174]. |
| Aldo-keto reductase family 1 member C2 inhibitor | Ursodeoxycholic acid/ DB01586 | Reduces cholesterol levels in the blood [206]. |
| Antithrombin-III inhibitor | Fondaparinux/ DB00569 |
Prevent venous thromboembolism to improve survival following MI [207]. |
| Apolipoprotein C-III (APOC3, apoC-III) inhibitor | Olezarsen/ DB18728 | Reduce triglyceride levels in adults with familial chylomicronemia syndrome [208]. |
| ATP citrate lyase (ACLY) inhibitor |
Bempedoic acid/ DB11936 | Reduces cholesterol levels, Prevents MI [209]. |
| β-tubulin inhibitor | Colchicine/ DB01394 | Cardiovascular mortality, Coronary revascularization, MI, Stroke [210,211]. |
| Beta-1 adrenergic receptor agonist | Dobutamine/ DB00841 |
Treat cardiac decompensation [212]. |
| Beta-1 adrenergic receptor antagonist | Nebivolol/ DB04861 |
Treat hypertension and aid in the management of heart failure [213]. |
| Bisoprolol/ DB00612 |
Prevent MI and heart failure and treat mild to moderate hypertension [214]. | |
| Carvedilol/ DB01136 |
Treat mild to severe heart failure, left ventricular dysfunction after MI [215]. | |
| Metoprolol/ DB00264 |
Treat heart failure, MI [216]. | |
| Propranolol/ DB00571 |
Used to treat hypertension, MI [217]. | |
| Atenolol/ DB00335 |
Secondary prevention of MI [218]. | |
| Bile acid sequestrant | Cholestyramine/DB01432 | Reduce elevated serum cholesterol in patients with primary hypercholesterolemia [219]. |
| Colesevelam/ DB00930 |
Used to lower LDL-C in adults with hyperlipidemia and pediatric patients with heterozygous familial hypercholesterolemia [220]. | |
| Colestipol/ DB00375 |
Used as an adjunct to diet and exercise to reduce LDL-C cholesterol levels in patients with primary hypercholesterolemia [219]. | |
| COX-1 inhibitor | Aspirin/ DB00945 |
Reducing the risk of major adverse cardiovascular events [221]. |
| Endothelin-1 (ET-1) antagonist | Bosentan/ DB00559 |
Used to treat pulmonary arterial hypertension [222]. |
| Fatty acid binding protein 1 (FABP-1), [Peroxisome proliferator receptor alpha (PPAR-alpha) agonist] |
Fenofibric acid/ DB13873 | Treat severe hypertriglyceridemia, primary hypercholesterolemia, or mixed dyslipidemia [223]. |
| Glucagon-like peptide 1 receptor agonist | Semaglutide/ DB13928 |
Reduces the risk of major adverse cardiovascular events in selected adults [224]. |
| Liraglutide/ DB06655 |
Prevention of cardiovascular complications associated with diabetes and obesity [225]. | |
| Hepatocyte diacylglycerol acyltransferase-2 inhibitor | Niacin/ DB00627 |
Treat hyperlipidemia, dyslipidemia, hypertriglyceridemia, and reduce the risk of MIs [226]. |
| Plasminogen activator | Tenecteplase/ DB00031 |
Used in the emergency treatment of MI [227]. |
| mRNA that codes for apolipoprotein B—100 (apoB-100) | Mipomersen/ DB05528 |
Used for the treatment of homozygous familial hypercholesterolemia [228]. |
| Microsomal triglyceride transfer protein (MTP) inhibitor | Lomitapide/ DB08827 |
Used in homozygous familial hypercholesterolemia (HoFH) patients to reduce low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), apolipoprotein B (apo B), and non-high-density lipoprotein cholesterol (non-HDL-C) [219]. |
| Mineralocorticoid receptor antagonist | Finerenone/ DB16165 |
Used to treat Cardiovascular mortality, non-fatal MI [229]. |
| Niemann-Pick C1 (NPC1)-like intracellular cholesterol transporter 1 inhibitor, Sterol O-acyltransferase 1 inhibitor |
Ezetimibe/ DB00973 |
Used to lower total cholesterol, LDL-C, Apo-B, and non-HDL-C in primary hyperlipidemia and familial cholesterolemia [230]. |
| Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor | Evolocumab/ DB09303 | Coronary revascularization, MI [231]. |
| Protease-activated receptor 1 (PAR-1) antagonist | Vorapaxar/ DB09030 |
Reducing the number of thrombotic cardiovascular events in patients with a history of MI (MI) or peripheral arterial disease (PAD) [232]. |
| Potassium/sodium hyperpolarization-activated cyclic nucleotide-gated channel 2 inhibitor | Ivabradine/ DB09083 |
Reduce the risk of chronic heart failure [233]. |
| Potentiates antithrombin-III (ATIII) inhibitor | Dalteparin/ DB06779 |
Prophylaxis of Cardiovascular event ischemic complications of unstable angina and non-Q-wave MI [234]. |
| Enoxaparin/ DB01225 |
Prophylaxis of Cardiovascular event ischemic complications of unstable angina and non-Q-wave MI [235]. | |
| Platelet glycoprotein (GP) IIb/IIIa receptor antagonist | Tirofiban/ DB00775 |
Prevents Cardiovascular event [236]. |
| Peroxisome proliferator activated receptor alpha (PPARα) agonist | Fenofibrate/ DB01039 |
Used to lower LDL-C, total-C, triglycerides, and Apo B, while increasing HDL-C in hypercholesterolemia, dyslipidemia, and hypertriglyceridemia [237]. |
| Gemfibrozil/ DB01241 |
Reduction of serum triglyceride levels in high-risk patients with hyperlipidemia [238]. | |
| Peroxisome proliferator activated receptor alpha (PPARα) activator | N-3 fatty acids/ DB11133 |
Prevention of recurrent events after MI in addition to treatment of hypertriglyceridemia [239]. |
| P2Y Purinoceptor 12 antagonist | Ticagrelor/ DB08816 |
Used to lower the rate of cardiovascular events such as heart attack and stroke [240]. |
| Prasugrel/ DB06209 |
Used to reduce risk of thrombotic cardiovascular events in unstable angina or non-ST-elevation MI (NSTEMI) [241]. | |
| Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor | Alirocumab/ DB09302 |
Used as an adjunct to manage heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease in patients who require additional lowering of LDL-cholesterol (LDL-C) [242]. |
| Recombinant tissue plasminogen activator (rt-PA) | Alteplase/ DB00009 |
Used for emergency treatment of MI, ischemic stroke [243]. |
| SGLT1 and, or SGLT2 inhibitor | Sotagliflozin/ DB12713 |
Used to treat cardiovascular mortality and heart failure [244]. |
| Type-1 angiotensin II receptor antagonist | Irbesartan/ DB01029 |
Treat congestive heart failure [245]. |
| Transthyretin (TTR) inhibitor | Acoramidis/ DB17999 |
Treatment of Cardiomyopathy caused by transthyretin mediated amyloidosis [246]. |
| 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor | Rosuvastatin/ DB01098 | Reduces the risk of CVD including heart attacks and stroke [247]. |
| Lovastatin /DB00227 |
Reduces the risk of CVD including heart attacks and stroke [247]. | |
| Atorvastatin/ DB01076 |
Reduces the risk of CVD including myocardial infarction (MI) and stroke [247]. | |
| Fluvastatin/ DB01095 |
Reduces the risk of CVD including MI and stroke [248]. | |
| Pitavastatin/ DB08860 |
Reduces the risk of CVD including MI and stroke [249]. | |
| Pravastatin/ DB00175 |
Reduces the risk of CVD including MI and stroke [247]. | |
| Simvastatin/ DB00641 |
Used to lower lipid levels and reduce the risk of cardiovascular events including MI and stroke [250]. |
Abbreviations
References
- Duttaroy, A.K.; Mallick, R. Comparative Effects of Fatty Acid and Glucose in Cardiac Remodeling. In Cellular, Molecular, and Environmental Contribution in Cardiac Remodeling; Elsevier, 2024; pp. 89–100.
- Stanley, W.C.; Recchia, F.A.; Lopaschuk, G.D. Myocardial Substrate Metabolism in the Normal and Failing Heart. Physiol. Rev. 2005, 85.
- Spector, A.A.; Yorek, M.A. Membrane Lipid Composition and Cellular Function. J. Lipid Res. 1985, 26.
- Calder, P.C. Marine Omega-3 Fatty Acids and Inflammatory Processes: Effects, Mechanisms and Clinical Relevance. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2015, 1851.
- Mozaffarian, D.; Wu, J.H.Y. Omega-3 Fatty Acids and Cardiovascular Disease: Effects on Risk Factors, Molecular Pathways, and Clinical Events. J. Am. Coll. Cardiol. 2011, 58.
- Elagizi, A.; Lavie, C.J.; O’keefe, E.; Marshall, K.; O’keefe, J.H.; Milani, R. V. An Update on Omega-3 Polyunsaturated Fatty Acids and Cardiovascular Health. Nutrients 2021, 13.
- Yoshida, A.; Sekine, W.; Homma, J.; Sekine, H.; Itoyama, Y.Y.; Sasaki, D.; Matsuura, K.; Kobayashi, E.; Shimizu, T. Development of Appropriate Fatty Acid Formulations to Raise the Contractility of Constructed Myocardial Tissues. Regen. Ther. 2022, 21. [CrossRef]
- Mallick, R.; Basak, S.; Duttaroy, A.K. Fatty Acids and Evolving Roles of Their Proteins in Neurological, Cardiovascular Disorders and Cancers. Prog. Lipid Res. 2021, 83.
- Balta, I.; Stef, L.; Pet, I.; Iancu, T.; Stef, D.; Corcionivoschi, N. Essential Fatty Acids as Biomedicines in Cardiac Health. Biomedicines 2021, 9.
- Listenberger, L.L.; Han, X.; Lewis, S.E.; Cases, S.; Farese, R. V.; Ory, D.S.; Schaffer, J.E. Triglyceride Accumulation Protects against Fatty Acid-Induced Lipotoxicity. Proceedings of the National Academy of Sciences 2003, 100, 3077–3082. [CrossRef]
- Annevelink, C.E.; Sapp, P.A.; Petersen, K.S.; Shearer, G.C.; Kris-Etherton, P.M. Diet-Derived and Diet-Related Endogenously Produced Palmitic Acid: Effects on Metabolic Regulation and Cardiovascular Disease Risk. J. Clin. Lipidol. 2023, 17.
- Glass, C.K.; Olefsky, J.M. Inflammation and Lipid Signaling in the Etiology of Insulin Resistance. Cell Metab. 2012, 15.
- Emken, E. Metabolism of Dietary Stearic Acid Relative to Other Fatty Acids in Human Subjects. Am. J. Clin. Nutr. 1994, 60, 1023S-1028S. [CrossRef]
- BRUCE, J.S.; SALTER, A.M. Metabolic Fate of Oleic Acid, Palmitic Acid and Stearic Acid in Cultured Hamster Hepatocytes. Biochemical Journal 1996, 316, 847–852. [CrossRef]
- van Rooijen, M.; Mensink, R. Palmitic Acid Versus Stearic Acid: Effects of Interesterification and Intakes on Cardiometabolic Risk Markers—A Systematic Review. Nutrients 2020, 12, 615. [CrossRef]
- Shahidi, F.; Senanayake, S.P.J.N. Fatty Acids. In International Encyclopedia of Public Health; Elsevier, 2008; pp. 594–603.
- Fan, H.; Wang, Y.; Ren, Z.; Liu, X.; Zhao, J.; Yuan, Y.; Fei, X.; Song, X.; Wang, F.; Liang, B. Mediterranean Diet Lowers All-Cause and Cardiovascular Mortality for Patients with Metabolic Syndrome. Diabetol. Metab. Syndr. 2023, 15, 107. [CrossRef]
- Gonzalez-Soto, M.; Mutch, D.M. Diet Regulation of Long-Chain PUFA Synthesis: Role of Macronutrients, Micronutrients, and Polyphenols on Δ-5/Δ-6 Desaturases and Elongases 2/5. Advances in Nutrition 2021, 12.
- Salsinha, A.S.; Socodato, R.; Relvas, J.B.; Pintado, M. The Pro- and Antiinflammatory Activity of Fatty Acids. In Bioactive Lipids; Elsevier, 2023; pp. 51–75.
- Mohebi-Nejad, A.; Bikdeli, B. Omega-3 Supplements and Cardiovascular Diseases. Tanaffos 2014, 13, 6–14.
- Masud Parvez, G.M.; Akanda, K.M. Foods and Arthritis: An Overview. In Bioactive Food as Dietary Interventions for Arthritis and Related Inflammatory Diseases; Elsevier, 2019; pp. 3–22.
- Wolf, C.; Steller, R.N. Introduction to the Nutrients and Their Association with Common Gastrointestinal Disorders. Physician Assist. Clin. 2022, 7.
- Poli, A.; Agostoni, C.; Visioli, F. Dietary Fatty Acids and Inflammation: Focus on the n-6 Series. Int. J. Mol. Sci. 2023, 24.
- Anderson, B.M.; Ma, D.W.L. Are All N-3 Polyunsaturated Fatty Acids Created Equal? Lipids Health Dis. 2009, 8.
- Djuricic, I.; Calder, P.C. N-3 Fatty Acids (EPA and DHA) and Cardiovascular Health—Updated Review of Mechanisms and Clinical Outcomes. Curr. Atheroscler. Rep. 2025, 27.
- Delgado, G.E.; Kleber, M.E. Trans Fatty Acids and Mortality. In The Molecular Nutrition of Fats; Elsevier, 2019; pp. 335–345.
- Gyamfi, D.; Ofori Awuah, E.; Owusu, S. Classes, Nomenclature, and Functions of Lipids and Lipid-Related Molecules and the Dietary Lipids. In The Molecular Nutrition of Fats; Elsevier, 2019; pp. 3–16.
- Da Dalt, L.; Cabodevilla, A.G.; Goldberg, I.J.; Norata, G.D. Cardiac Lipid Metabolism, Mitochondrial Function, and Heart Failure. Cardiovasc. Res. 2023, 119.
- O’Connell, T.D.; Mason, R.P.; Budoff, M.J.; Navar, A.M.; Shearer, G.C. Mechanistic Insights into Cardiovascular Protection for Omega-3 Fatty Acids and Their Bioactive Lipid Metabolites. European Heart Journal, Supplement 2020, 22.
- Dufau, J.; Recazens, E.; Bottin, L.; Bergoglio, C.; Mairal, A.; Chaoui, K.; Marques, M.A.; Jimenez, V.; García, M.; Wang, T.; et al. Nuclear Hormone-Sensitive Lipase Regulates Adipose Tissue Mass and Adipocyte Metabolism. Cell Metab. 2025, 37. [CrossRef]
- Herbst, E.A.F.; Paglialunga, S.; Gerling, C.; Whitfield, J.; Mukai, K.; Chabowski, A.; Heigenhauser, G.J.F.; Spriet, L.L.; Holloway, G.P. Omega-3 Supplementation Alters Mitochondrial Membrane Composition and Respiration Kinetics in Human Skeletal Muscle. Journal of Physiology 2014, 592. [CrossRef]
- Abumrad, N.A.; Goldberg, I.J. CD36 Actions in the Heart: Lipids, Calcium, Inflammation, Repair and More? Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2016, 1861.
- Bharadwaj, K.G.; Hiyama, Y.; Hu, Y.; Huggins, L.A.; Ramakrishnan, R.; Abumrad, N.A.; Shulman, G.I.; Blaner, W.S.; Goldberg, I.J. Chylomicron- and VLDL-Derived Lipids Enter the Heart through Different Pathways: In Vivo Evidence for Receptor- and Non-Receptor-Mediated Fatty Acid Uptake. Journal of Biological Chemistry 2010, 285. [CrossRef]
- Hames, K.C.; Vella, A.; Kemp, B.J.; Jensen, M.D. Free Fatty Acid Uptake in Humans with CD36 Deficiency. Diabetes 2014, 63. [CrossRef]
- Fillmore, N.; Hou, V.; Sun, J.; Springer, D.; Murphy, E. Cardiac Specific Knock-down of Peroxisome Proliferator Activated Receptor α Prevents Fasting-Induced Cardiac Lipid Accumulation and Reduces Perilipin 2. PLoS One 2022, 17. [CrossRef]
- Prosdocimo, D.A.; John, J.E.; Zhang, L.; Efraim, E.S.; Zhang, R.; Liao, X.; Jain, M.K. KLF15 and PPAR α Cooperate to Regulate Cardiomyocyte Lipid Gene Expression and Oxidation. PPAR Res. 2015, 2015. [CrossRef]
- Tien, Y.T.; Chang, M.H.; Chu, P.Y.; Lin, C.S.; Liu, C.H.; Liao, A.T. Downregulation of the KLF4 Transcription Factor Inhibits the Proliferation and Migration of Canine Mammary Tumor Cells. Veterinary Journal 2015, 205. [CrossRef]
- Drosatos, K.; Pollak, N.M.; Pol, C.J.; Ntziachristos, P.; Willecke, F.; Valenti, M.C.; Trent, C.M.; Hu, Y.; Guo, S.; Aifantis, I.; et al. Cardiac Myocyte KLF5 Regulates Ppara Expression and Cardiac Function. Circ. Res. 2016, 118. [CrossRef]
- Kucharski, M.; Kaczor, U. PPARα and PPARγ as Main Regulators of Fatty Acid Metabolism. Postepy Hig. Med. Dosw. 2018, 72.
- Savarese, G.; Becher, P.M.; Lund, L.H.; Seferovic, P.; Rosano, G.M.C.; Coats, A.J.S. Global Burden of Heart Failure: A Comprehensive and Updated Review of Epidemiology. Cardiovasc. Res. 2022, 118.
- Li, Z.; Lei, H.; Jiang, H.; Fan, Y.; Shi, J.; Li, C.; Chen, F.; Mi, B.; Ma, M.; Lin, J.; et al. Saturated Fatty Acid Biomarkers and Risk of Cardiometabolic Diseases: A Meta-Analysis of Prospective Studies. Front. Nutr. 2022, 9.
- Maki, K.C.; Dicklin, M.R.; Kirkpatrick, C.F. Saturated Fats and Cardiovascular Health: Current Evidence and Controversies. J. Clin. Lipidol. 2021, 15.
- Elnwasany, A.; Ewida, H.A.; Menendez-Montes, I.; Mizerska, M.; Fu, X.; Kim, C.-W.; Horton, J.D.; Burgess, S.C.; Rothermel, B.A.; Szweda, P.A.; et al. Reciprocal Regulation of Cardiac β-Oxidation and Pyruvate Dehydrogenase by Insulin. Journal of Biological Chemistry 2024, 300, 107412. [CrossRef]
- Yarmohammadi, F.; Hayes, A.W.; Karimi, G. Natural and Chemical Compounds as Protective Agents against Cardiac Lipotoxicity. Biomedicine and Pharmacotherapy 2022, 145.
- Silva-Soto, M.; Carrillo-Fernández, P.; Saez Lancellotti, E.; Medina-Jiménez, E.; Mogaburo Alba, J.; Catena-Granados, N.; López-Carmona, M.; Pérez-Belmonte, L.; Prieto Lain, N.; Gómez Hernández, A.; et al. Extra Virgin Olive Oil Phenolic Compounds: Modulating Mitochondrial Function and Protecting Against Chronic Diseases—A Narrative Review. Nutrients 2025, 17, 1443. [CrossRef]
- Seifi, K.; Rezaei, M.; Yansari, A.T.; Zamiri, M.J.; Riazi, G.H.; Heidari, R. Short Chain Fatty Acids May Improve Hepatic Mitochondrial Energy Efficiency in Heat Stressed-Broilers. J. Therm. Biol. 2020, 89. [CrossRef]
- Endo, J.; Arita, M. Cardioprotective Mechanism of Omega-3 Polyunsaturated Fatty Acids. J. Cardiol. 2016, 67.
- Mallick, R.; Basak, S.; Duttaroy, A.K. Docosahexaenoic Acid,22:6n-3: Its Roles in the Structure and Function of the Brain. International Journal of Developmental Neuroscience 2019, 79. [CrossRef]
- Kang, J.X. Reduction of Heart Rate by Omega-3 Fatty Acids and the Potential Underlying Mechanisms. Front. Physiol. 2012, 3 OCT.
- Matacchione, G.; Piacenza, F.; Pimpini, L.; Rosati, Y.; Marcozzi, S. The Role of the Gut Microbiota in the Onset and Progression of Heart Failure: Insights into Epigenetic Mechanisms and Aging. Clin. Epigenetics 2024, 16, 175. [CrossRef]
- Macdonald-Ramos, K.; Martínez-Ibarra, A.; Monroy, A.; Miranda-Ríos, J.; Cerbón, M. Effect of Dietary Fatty Acids on MicroRNA Expression Related to Metabolic Disorders and Inflammation in Human and Animal Trials. Nutrients 2021, 13, 1830. [CrossRef]
- Ahn, S.; Kaipparettu, B.A. G-Protein Coupled Receptors in Metabolic Reprogramming and Cancer. Pharmacol. Ther. 2025, 270, 108849. [CrossRef]
- Kaemmerer, E.; Plum, P.; Klaus, C.; Weiskirchen, R.; Liedtke, C.; Adolf, M.; Schippers, A.; Wagner, N.; Reinartz, A.; Gassler, N. Fatty Acid Binding Receptors in Intestinal Physiology and Pathophysiology. World J. Gastrointest. Pathophysiol. 2010, 1, 147–153. [CrossRef]
- Fredriksson, R.; Höglund, P.J.; Gloriam, D.E.I.; Lagerström, M.C.; Schiöth, H.B. Seven Evolutionarily Conserved Human Rhodopsin G Protein-Coupled Receptors Lacking Close Relatives. FEBS Lett. 2003, 554, 381–388. [CrossRef]
- Hirasawa, A.; Tsumaya, K.; Awaji, T.; Katsuma, S.; Adachi, T.; Yamada, M.; Sugimoto, Y.; Miyazaki, S.; Tsujimoto, G. Free Fatty Acids Regulate Gut Incretin Glucagon-like Peptide-1 Secretion through GPR120. Nat. Med. 2005, 11, 90–94. [CrossRef]
- Davie, J.R. Inhibition of Histone Deacetylase Activity by Butyrate. Journal of Nutrition 2003, 133. [CrossRef]
- Wu, Y.; Dong, Y.; Song, P.; Zou, M.H. Activation of the AMP-Activated Protein Kinase (AMPK) by Nitrated Lipids in Endothelial Cells. PLoS One 2012, 7, e31056. [CrossRef]
- Canbolat, E.; Cakıroglu, F.P. The Importance of AMPK in Obesity and Chronic Diseases and the Relationship of AMPK with Nutrition: A Literature Review. Crit. Rev. Food Sci. Nutr. 2023, 63, 449–456. [CrossRef]
- Monsalve, F.A.; Pyarasani, R.D.; Delgado-Lopez, F.; Moore-Carrasco, R. Peroxisome Proliferator-Activated Receptor Targets for the Treatment of Metabolic Diseases. Mediators Inflamm. 2013, 2013. [CrossRef]
- Kota, B.P.; Huang, T.H.W.; Roufogalis, B.D. An Overview on Biological Mechanisms of PPARs. Pharmacol. Res. 2005, 51, 85–94. [CrossRef]
- Shu, L.; Chan, A.; Wells, R.A. Cross-Talk between PPARs and the Partners of RXR: A Molecular Perspective. 2009, 9. [CrossRef]
- König, B.; Koch, A.; Spielmann, J.; Hilgenfeld, C.; Stangl, G.I.; Eder, K. Activation of PPARα Lowers Synthesis and Concentration of Cholesterol by Reduction of Nuclear SREBP-2. Biochem. Pharmacol. 2007, 73, 574–585. [CrossRef]
- Steinberg, G.R.; Hardie, D.G. New Insights into Activation and Function of the AMPK. Nature Reviews Molecular Cell Biology 2022 24:4 2022, 24, 255–272. [CrossRef]
- Jung, E.J.; Kwon, S.W.; Jung, B.H.; Oh, S.H.; Lee, B.H. Role of the AMPK/SREBP-1 Pathway in the Development of Orotic Acid-Induced Fatty Liver. J. Lipid Res. 2011, 52, 1617–1625. [CrossRef]
- Li, N.; Li, X.; Ding, Y.; Liu, X.; Diggle, K.; Kisseleva, T.; Brenner, D.A. SREBP Regulation of Lipid Metabolism in Liver Disease, and Therapeutic Strategies. Biomedicines 2023, Vol. 11, Page 3280 2023, 11, 3280. [CrossRef]
- Myers, M.N.; Chirivi, M.; dos Santos Neto, J.M.; Parales-Girón, J.; Worden, L.C.; Lock, A.L.; Contreras, G.A. Dynamics of Oxylipin Biosynthesis in Systemic Inflammation: Insights from a Large Animal Model of Endotoxemia. Front. Immunol. 2025, 16. [CrossRef]
- Verdin, E.; Hirschey, M.D.; Finley, L.W.S.; Haigis, M.C. Sirtuin Regulation of Mitochondria: Energy Production, Apoptosis, and Signaling. Trends Biochem. Sci. 2010, 35, 669–675. [CrossRef]
- Singh, C.K.; Chhabra, G.; Ndiaye, M.A.; Garcia-Peterson, L.M.; MacK, N.J.; Ahmad, N. The Role of Sirtuins in Antioxidant and Redox Signaling. Antioxid. Redox Signal. 2018, 28, 643–661. [CrossRef]
- Han, Y.; Zhou, S.; Coetzee, S.; Chen, A. Sirt4 and Its Roles in Energy and Redox Metabolism in Health, Disease and during Exercise. Front. Physiol. 2019, 10. [CrossRef]
- Purushotham, A.; Schug, T.T.; Li, X. SIRT1 Performs a Balancing Act on the Tight-Rope toward Longevity. Aging 2009, 1, 669–673. [CrossRef]
- Picard, F.; Kurtev, M.; Chung, N.; Topark-Ngarm, A.; Senawong, T.; De Oliveira, R.M.; Leid, M.; McBurney, M.W.; Guarente, L. Sirt1 Promotes Fat Mobilization in White Adipocytes by Repressing PPAR-γ. Nature 2004, 429, 771–776. [CrossRef]
- Walker, A.K.; Yang, F.; Jiang, K.; Ji, J.Y.; Watts, J.L.; Purushotham, A.; Boss, O.; Hirsch, M.L.; Ribich, S.; Smith, J.J.; et al. Conserved Role of SIRT1 Orthologs in Fasting-Dependent Inhibition of the Lipid/Cholesterol Regulator SREBP. Genes Dev. 2010, 24. [CrossRef]
- Lim, J.H.; Gerhart-Hines, Z.; Dominy, J.E.; Lee, Y.; Kim, S.; Tabata, M.; Xiang, Y.K.; Puigserver, P. Oleic Acid Stimulates Complete Oxidation of Fatty Acids through Protein Kinase A-Dependent Activation of SIRT1-PGC1α Complex. Journal of Biological Chemistry 2013, 288, 7117–7126. [CrossRef]
- Son, S.H.; Lee, S.M.; Lee, M.H.; Son, Y.K.; Kim, S.E.; An, W.S. Omega-3 Fatty Acids Upregulate SIRT1/3, Activate PGC-1α via Deacetylation, and Induce Nrf1 Production in 5/6 Nephrectomy Rat Model. Mar. Drugs 2021, 19, 182. [CrossRef]
- Tornatore, L.; Thotakura, A.K.; Bennett, J.; Moretti, M.; Franzoso, G. The Nuclear Factor Kappa B Signaling Pathway: Integrating Metabolism with Inflammation. Trends Cell Biol. 2012, 22, 557–566. [CrossRef]
- Yang, X.; Zhang, X.; Heckmann, B.L.; Lu, X.; Liu, J. Relative Contribution of Adipose Triglyceride Lipase and Hormone-Sensitive Lipase to Tumor Necrosis Factor-α (TNF-α)-Induced Lipolysis in Adipocytes. Journal of Biological Chemistry 2011, 286, 40477–40485. [CrossRef]
- Edwards, M.; Mohiuddin, S.S. Biochemistry, Lipolysis. StatPearls 2023.
- Cho, Y.Y.; Kim, S.; Kim, P.; Jo, M.J.; Park, S.E.; Choi, Y.; Jung, S.M.; Kang, H.J. G-Protein-Coupled Receptor (GPCR) Signaling and Pharmacology in Metabolism: Physiology, Mechanisms, and Therapeutic Potential. Biomolecules 2025, Vol. 15, Page 291 2025, 15, 291. [CrossRef]
- Gao, H.; Tian, K.; Feng, X.; Yan, M.; Gao, C.; Jiang, Y.; Zhu, C.; Zhu, H.; Liu, X.; Peng, Y. Free Fatty Acid Receptor 2 Promotes Cardiomyocyte Hypertrophy by Activating STAT3 and GATA4. Food Science and Human Wellness 2022, 11, 405–417. [CrossRef]
- Das, M.; Das, D.K. Lipid Raft in Cardiac Health and Disease. Curr. Cardiol. Rev. 2009, 5, 105. [CrossRef]
- O’Connell, K.M.S.; Martens, J.R.; Tamkun, M.M. Localization of Ion Channels to Lipid Raft Domains within the Cardiovascular System. Trends Cardiovasc. Med. 2004, 14, 37–42. [CrossRef]
- Maguy, A.; Hebert, T.E.; Nattel, S. Involvement of Lipid Rafts and Caveolae in Cardiac Ion Channel Function. Cardiovasc. Res. 2006, 69, 798–807. [CrossRef]
- Epand, R.M.; Epand, R.F.; Ahmed, N.; Chen, R. Promotion of Hexagonal Phase Formation and Lipid Mixing by Fatty Acids with Varying Degrees of Unsaturation. Chem. Phys. Lipids 1991, 57, 75–80. [CrossRef]
- Cooper, G.M. Cell Membranes. 2000.
- Ibarguren, M.; López, D.J.; Escribá, P. V. The Effect of Natural and Synthetic Fatty Acids on Membrane Structure, Microdomain Organization, Cellular Functions and Human Health. Biochimica et Biophysica Acta (BBA)—Biomembranes 2014, 1838, 1518–1528. [CrossRef]
- Fearnley, C.J.; Llewelyn Roderick, H.; Bootman, M.D. Calcium Signaling in Cardiac Myocytes. Cold Spring Harb. Perspect. Biol. 2011, 3, a004242. [CrossRef]
- Zhihao, L.; Jingyu, N.; Lan, L.; Michael, S.; Rui, G.; Xiyun, B.; Xiaozhi, L.; Guanwei, F. SERCA2a: A Key Protein in the Ca2+ Cycle of the Heart Failure. Heart Fail. Rev. 2020, 25, 523–535. [CrossRef]
- Giroud, S.; Frare, C.; Strijkstra, A.; Boerema, A.; Arnold, W.; Ruf, T. Membrane Phospholipid Fatty Acid Composition Regulates Cardiac SERCA Activity in a Hibernator, the Syrian Hamster (Mesocricetus Auratus). PLoS One 2013, 8. [CrossRef]
- Li, Y.; Ge, M.; Ciani, L.; Kuriakose, G.; Westover, E.J.; Dura, M.; Covey, D.F.; Freed, J.H.; Maxfield, F.R.; Lytton, J.; et al. Enrichment of Endoplasmic Reticulum with Cholesterol Inhibits Sarcoplasmic-Endoplasmic Reticulum Calcium ATPase-2b Activity in Parallel with Increased Order of Membrane Lipids: Implications for Depletion of Endoplasmic Reticulum Calcium Stores and Apoptosis in Cholesterol-Loaded Macrophages. Journal of Biological Chemistry 2004, 279, 37030–37039. [CrossRef]
- Yamamoto, T.; Endo, J.; Kataoka, M.; Matsuhashi, T.; Katsumata, Y.; Shirakawa, K.; Yoshida, N.; Isobe, S.; Moriyama, H.; Goto, S.; et al. Decrease in Membrane Phospholipids Unsaturation Correlates with Myocardial Diastolic Dysfunction. PLoS One 2018, 13, e0208396. [CrossRef]
- Asp, M.L.; Martindale, J.J.; Heinis, F.I.; Wang, W.; Metzger, J.M. Calcium Mishandling in Diastolic Dysfunction: Mechanisms and Potential Therapies. Biochimica et Biophysica Acta (BBA)—Molecular Cell Research 2013, 1833, 895–900. [CrossRef]
- Rodríguez-Rodríguez, R.; Fosch, A.; Garcia-Chica, J.; Zagmutt, S.; Casals, N. Targeting Carnitine Palmitoyltransferase 1 Isoforms in the Hypothalamus: A Promising Strategy to Regulate Energy Balance. J. Neuroendocrinol. 2023, 35, e13234. [CrossRef]
- Lopaschuk, G.D.; Ussher, J.R.; Folmes, C.D.L.; Jaswal, J.S.; Stanley, W.C. Myocardial Fatty Acid Metabolism in Health and Disease. Physiol. Rev. 2010, 90, 207–258. [CrossRef]
- Tirosh, O.; Ilan, E.; Anavi, S.; Ramadori, G.; Madar, Z. Nutritional Lipid-Induced Oxidative Stress Leads to Mitochondrial Dysfunction Followed by Necrotic Death in FaO Hepatocytes. Nutrition 2009, 25, 200–208. [CrossRef]
- Jain, A.; Casanova, D.; Padilla, A.V.; Paniagua Bojorges, A.; Kotla, S.; Ko, K.A.; Samanthapudi, V.S.K.; Chau, K.; Nguyen, M.T.H.; Wen, J.; et al. Premature Senescence and Cardiovascular Disease Following Cancer Treatments: Mechanistic Insights. Front. Cardiovasc. Med. 2023, 10. [CrossRef]
- Heshmati, J.; Morvaridzadeh, M.; Maroufizadeh, S.; Akbari, A.; Yavari, M.; Amirinejad, A.; Maleki-Hajiagha, A.; Sepidarkish, M. Omega-3 Fatty Acids Supplementation and Oxidative Stress Parameters: A Systematic Review and Meta-Analysis of Clinical Trials. Pharmacol. Res. 2019, 149, 104462. [CrossRef]
- Li, Q.; Yu, Q.; Na, R.; Liu, B. Omega-3 Polyunsaturated Fatty Acids Prevent Murine Dilated Cardiomyopathy by Reducing Oxidative Stress and Cardiomyocyte Apoptosis. Exp. Ther. Med. 2017, 14, 6152. [CrossRef]
- Kyriakoudi, S.; Drousiotou, A.; Petrou, P.P. When the Balance Tips: Dysregulation of Mitochondrial Dynamics as a Culprit in Disease. Int. J. Mol. Sci. 2021, 22, 4617. [CrossRef]
- Smirnova, E.; Griparic, L.; Shurland, D.L.; Van der Bliek, A.M. Dynamin-Related Protein Drp1 Is Required for Mitochondrial Division in Mammalian Cells. Mol. Biol. Cell 2001, 12, 2245–2256. [CrossRef]
- Santel, A.; Fuller, M.T. Control of Mitochondrial Morphology by a Human Mitofusin. J. Cell Sci. 2001, 114, 867–874. [CrossRef]
- Chen, H.; Detmer, S.A.; Ewald, A.J.; Griffin, E.E.; Fraser, S.E.; Chan, D.C. Mitofusins Mfn1 and Mfn2 Coordinately Regulate Mitochondrial Fusion and Are Essential for Embryonic Development. Journal of Cell Biology 2003, 160, 189–200. [CrossRef]
- Hoppins, S.; Lackner, L.; Nunnari, J. The Machines That Divide and Fuse Mitochondria. Annu. Rev. Biochem. 2007, 76, 751–780. [CrossRef]
- Sherratt, S.C.R.; Juliano, R.A.; Copland, C.; Bhatt, D.L.; Libby, P.; Mason, R.P. EPA and DHA Containing Phospholipids Have Contrasting Effects on Membrane Structure. J. Lipid Res. 2021, 62, 100106. [CrossRef]
- Pal, A.; Metherel, A.H.; Fiabane, L.; Buddenbaum, N.; Bazinet, R.P.; Shaikh, S.R. Do Eicosapentaenoic Acid and Docosahexaenoic Acid Have the Potential to Compete against Each Other? Nutrients 2020, 12, 1–12. [CrossRef]
- Fuentes, J.; Morcillo, P. The Role of Cardiolipin in Mitochondrial Function and Neurodegenerative Diseases. Cells 2024, 13, 609. [CrossRef]
- Lin, J.; Duan, J.; Wang, Q.; Xu, S.; Zhou, S.; Yao, K. Mitochondrial Dynamics and Mitophagy in Cardiometabolic Disease. Front. Cardiovasc. Med. 2022, 9.
- Lemaitre, R.N.; King, I.B. Very Long-Chain Saturated Fatty Acids and Diabetes and Cardiovascular Disease. Curr. Opin. Lipidol. 2022, 33.
- Wang, C.; Han, D.; Feng, X.; Hu, L.; Wu, J. Docosahexaenoic Acid Alleviates LPS-Induced Cytotoxicity in HL-1 Cardiac Cells via Improving Stress-Induced Mitochondrial Fragmentation. Heliyon 2023, 9. [CrossRef]
- Elezaby, A.; Sverdlov, A.L.; Tu, V.H.; Soni, K.; Luptak, I.; Qin, F.; Liesa, M.; Shirihai, O.S.; Rimer, J.; Schaffer, J.E.; et al. Mitochondrial Remodeling in Mice with Cardiomyocyte-Specific Lipid Overload. J. Mol. Cell. Cardiol. 2015, 79, 275–283. [CrossRef]
- Tsushima, K.; Bugger, H.; Wende, A.R.; Soto, J.; Jenson, G.A.; Tor, A.R.; McGlauflin, R.; Kenny, H.C.; Zhang, Y.; Souvenir, R.; et al. Mitochondrial Reactive Oxygen Species in Lipotoxic Hearts Induces Post-Translational Modifications of AKAP121, DRP1 and OPA1 That Promote Mitochondrial Fission. Circ. Res. 2017, 122, 58. [CrossRef]
- Gao, J.; Li, C.; Chen, H.; Li, Z.-H.; You, F.-F.; Song, W.-Q.; Zhong, W.-F.; Chen, P.-L.; Yang, J.; Huang, Q.-M.; et al. Effect of Meal-Timing on the Association of Unsaturated Fatty Acids with All-Cause and Cardiovascular Mortality among Adults: A Prospective Cohort Study with 10-Year Follow-Up. Nutrients 2024, 16, 2071. [CrossRef]
- Manoogian, E.N.C.; Zadourian, A.; Lo, H.C.; Gutierrez, N.R.; Shoghi, A.; Rosander, A.; Pazargadi, A.; Ormiston, C.K.; Wang, X.; Sui, J.; et al. Feasibility of Time-Restricted Eating and Impacts on Cardiometabolic Health in 24-h Shift Workers: The Healthy Heroes Randomized Control Trial. Cell Metab. 2022, 34, 1442-1456.e7. [CrossRef]
- Pakhomov, N.; Baugh, J.A. The Role of Diet-Derived Short-Chain Fatty Acids in Regulating Cardiac Pressure Overload. Am. J. Physiol. Heart Circ. Physiol. 2021, 320.
- Miao, H.; Li, B.; Wang, Z.; Mu, J.; Tian, Y.; Jiang, B.; Zhang, S.; Gong, X.; Shui, G.; Lam, S.M. Lipidome Atlas of the Developing Heart Uncovers Dynamic Membrane Lipid Attributes Underlying Cardiac Structural and Metabolic Maturation. Research 2022, 2022. [CrossRef]
- Finck, B.N.; Kelly, D.P. Peroxisome Proliferator-Activated Receptor α (PPARα) Signaling in the Gene Regulatory Control of Energy Metabolism in the Normal and Diseased Heart. J. Mol. Cell. Cardiol. 2002, 34, 1249–1257. [CrossRef]
- Kimura, I.; Ichimura, A.; Ohue-Kitano, R.; Igarashi, M. Free Fatty Acid Receptors in Health and Disease. Physiol. Rev. 2020, 100, 171–210. [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-ΚB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [CrossRef]
- Beauloye, V.; Zech, F.; Mong, H.T.T.; Clapuyt, P.; Maes, M.; Brichard, S.M. Determinants of Early Atherosclerosis in Obese Children and Adolescents. Journal of Clinical Endocrinology and Metabolism 2007, 92. [CrossRef]
- Jaca, A.; Durão, S.; Harbron, J. Omega-3 Fatty Acids for the Primary and Secondary Prevention of Cardiovascular Disease. South African Medical Journal 2020, 110. [CrossRef]
- Eclov, J.A.; Qian, Q.; Redetzke, R.; Chen, Q.; Wu, S.C.; Healy, C.L.; Ortmeier, S.B.; Harmon, E.; Shearer, G.C.; O’Connell, T.D. EPA, Not DHA, Prevents Fibrosis in Pressure Overload-Induced Heart Failure: Potential Role of Free Fatty Acid Receptor 4. J. Lipid Res. 2015, 56. [CrossRef]
- Kain, V.; Ingle, K.A.; Colas, R.A.; Dalli, J.; Prabhu, S.D.; Serhan, C.N.; Joshi, M.; Halade, G. V. Resolvin D1 Activates the Inflammation Resolving Response at Splenic and Ventricular Site Following Myocardial Infarction Leading to Improved Ventricular Function. J. Mol. Cell. Cardiol. 2015, 84. [CrossRef]
- Lipsky, P.E.; Brooks, P.; Crofford, L.J.; DuBois, R.; Graham, D.; Simon, L.S.; Van De Putte, L.B.A.; Abramson, S.B. Unresolved Issues in the Role of Cyclooxygenase-2 in Normal Physiologic Processes and Disease. Arch. Intern. Med. 2000, 160.
- Cheng, Y.; Austin, S.C.; Rocca, B.; Koller, B.H.; Coffman, T.M.; Grosser, T.; Lawson, J.A.; FitzGerald, G.A. Role of Prostacyclin in the Cardiovascular Response to Thromboxane A2. Science (1979). 2002, 296. [CrossRef]
- Mehta, J.; Mehta, P.; Feldman, R.L. Severe Intracoronary Thromboxane Release Preceding Acute Coronary Artery Occlusion. Prostaglandins Leukot. Med. 1982, 8.
- Barone, F.C.; Schmidt, D.B.; Hillegass, L.M.; Price, W.J.; White, R.F.; Feuerstein, G.Z.; Clark, R.K.; Lee, E. V.; Griswold, D.E.; Sarau, H.M. Reperfusion Increases Neutrophils and Leukotriene B4 Receptor Binding in Rat Focal Ischemia. Stroke 1992, 23. [CrossRef]
- Kadletz, M.; Dignan, R.J.; Loesser, K.E.; Hess, M.L.; Wechsler, A.S. Ischemia and Activated Neutrophils Alter Coronary Microvascular but Not Epicardial Coronary Artery Reactivity. Journal of Thoracic and Cardiovascular Surgery 1994, 108. [CrossRef]
- Zhang, P.; Wang, J.; Wang, X.; Wang, L.; Xu, S.; Gong, P. Protectin D1 Alleviates Myocardial Ischemia/Reperfusion Injury by Regulating PI3K/AKT Signaling Pathway. J. Cardiovasc. Transl. Res. 2024, 17. [CrossRef]
- Li, X.W.; Li, C.L.; Liang, W.D.; Bi, Y.T.; Chen, M.H.; Dong, S. Protectin D1 Promotes Resolution of Inflammation in a Murine Model of Lipopolysaccharide-Induced Acute Lung Injury via Enhancing Neutrophil Apoptosis. Chin. Med. J. (Engl). 2014, 127. [CrossRef]
- Colas, R.A.; Shinohara, M.; Dalli, J.; Chiang, N.; Serhan, C.N. Identification and Signature Profiles for Pro-Resolving and Inflammatory Lipid Mediators in Human Tissue. Am. J. Physiol. Cell Physiol. 2014, 307. [CrossRef]
- Friedrich, E.B.; Tager, A.M.; Liu, E.; Pettersson, A.; Owman, C.; Munn, L.; Luster, A.D.; Gerszten, R.E. Mechanisms of Leukotriene B4-Triggered Monocyte Adhesion. Arterioscler. Thromb. Vasc. Biol. 2003, 23. [CrossRef]
- Hara, T.; Sata, M. Roles of Perivascular Adipose Tissue in the Pathogenesis of Atherosclerosis ― an Update on Recent Findings. Front. Physiol. 2024, 15.
- Wahrburg, U. What Are the Health Effects of Fat? Eur. J. Nutr. 2004, 43 Suppl 1. [CrossRef]
- WHITE, B. Dietary Fatty Acids. Am. Fam. Physician 2009, 80, 345–350.
- Hu, F.B.; Stampfer, M.J.; Manson, J.A.E.; Ascherio, A.; Colditz, G.A.; Speizer, F.E.; Hennekens, C.H.; Willett, W.C. Dietary Saturated Fats and Their Food Sources in Relation to the Risk of Coronary Heart Disease in Women. Am. J. Clin. Nutr. 1999, 70, 1001–1008. [CrossRef]
- Lemaitre, R.N.; King, I.B. Very Long-Chain Saturated Fatty Acids and Diabetes and Cardiovascular Disease. Curr. Opin. Lipidol. 2022, 33, 76–82. [CrossRef]
- Steur, M.; Johnson, L.; Sharp, S.J.; Imamura, F.; Sluijs, I.; Key, T.J.; Wood, A.; Chowdhury, R.; Guevara, M.; Jakobsen, M.U.; et al. Dietary Fatty Acids, Macronutrient Substitutions, Food Sources and Incidence of Coronary Heart Disease: Findings from the EPIC-CVD Case-Cohort Study across Nine European Countries. J. Am. Heart Assoc. 2021, 10, 19814. [CrossRef]
- Praagman, J.; De Jonge, E.A.L.; Kiefte-De Jong, J.C.; Beulens, J.W.J.; Sluijs, I.; Schoufour, J.D.; Hofman, A.; Van Der Schouw, Y.T.; Franco, O.H. Dietary Saturated Fatty Acids and Coronary Heart Disease Risk in a Dutch Middle-Aged and Elderly Population. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2011–2018. [CrossRef]
- Unhapipatpong, C.; Shantavasinkul, P.C.; Kasemsup, V.; Siriyotha, S.; Warodomwichit, D.; Maneesuwannarat, S.; Vathesatogkit, P.; Sritara, P.; Thakkinstian, A. Tropical Oil Consumption and Cardiovascular Disease: An Umbrella Review of Systematic Reviews and Meta Analyses. Nutrients 2021, 13.
- Valk, R.; Hammill, J.; Grip, J. Saturated Fat: Villain and Bogeyman in the Development of Cardiovascular Disease? Eur. J. Prev. Cardiol. 2022, 29, 2312–2321. [CrossRef]
- Rucker, D.; Dhamoon, A.S. Physiology, Thromboxane A2; 2019;
- Wone, B.W.M.; Donovan, E.R.; Cushman, J.C.; Hayes, J.P. Metabolic Rates Associated with Membrane Fatty Acids in Mice Selected for Increased Maximal Metabolic Rate. Comparative Biochemistry and Physiology—A Molecular and Integrative Physiology 2013, 165. [CrossRef]
- Praagman, J.; Beulens, J.W.; Alssema, M.; Zock, P.L.; Wanders, A.J.; Sluijs, I.; van der Schouw, Y.T. The Association between Dietary Saturated Fatty Acids and Ischemic Heart Disease Depends on the Type and Source of Fatty Acid in the European Prospective Investigation into Cancer and Nutrition–Netherlands Cohort. Am. J. Clin. Nutr. 2016, 103, 356–365. [CrossRef]
- Mehrabani, A.; Jalalzadeh, M.; Jannati, N.; Lotfi, K.; Arzhang, P.; Azadbakht, L. Association Between Monounsaturated Fatty Acid Intake and Risk of Total Stroke and Its Subtypes: A Systematic Review and Dose–Response Meta-Analysis of Prospective Cohort Studies. Nutr. Rev. 2024. [CrossRef]
- Lotfi, K.; Salari-Moghaddam, A.; Yousefinia, M.; Larijani, B.; Esmaillzadeh, A. Dietary Intakes of Monounsaturated Fatty Acids and Risk of Mortality from All Causes, Cardiovascular Disease and Cancer: A Systematic Review and Dose-Response Meta-Analysis of Prospective Cohort Studies. Ageing Res. Rev. 2021, 72, 101467. [CrossRef]
- Hooper, L.; Martin, N.; Jimoh, O.F.; Kirk, C.; Foster, E.; Abdelhamid, A.S. Reduction in Saturated Fat Intake for Cardiovascular Disease. Cochrane Database of Systematic Reviews 2020, 2020. [CrossRef]
- Sheashea, M.; Xiao, J.; Farag, M.A. MUFA in Metabolic Syndrome and Associated Risk Factors: Is MUFA the Opposite Side of the PUFA Coin? Food Funct. 2021, 12, 12221–12234. [CrossRef]
- Yamada, M.H.; Fujihara, K.; Kodama, S.; Sato, T.; Osawa, T.; Yaguchi, Y.; Yamamoto, M.; Kitazawa, M.; Matsubayashi, Y.; Yamada, T.; et al. Associations of Systolic Blood Pressure and Diastolic Blood Pressure With the Incidence of Coronary Artery Disease or Cerebrovascular Disease According to Glucose Status. Diabetes Care 2021, 44, 2124–2131. [CrossRef]
- Cao, X.; Xia, J.; Zhou, Y.; Wang, Y.; Xia, H.; Wang, S.; Liao, W.; Sun, G. The Effect of MUFA-Rich Food on Lipid Profile: A Meta-Analysis of Randomized and Controlled-Feeding Trials. Foods 2022, 11, 1982. [CrossRef]
- Li, Y.; Hruby, A.; Bernstein, A.M.; Ley, S.H.; Wang, D.D.; Chiuve, S.E.; Sampson, L.; Rexrode, K.M.; Rimm, E.B.; Willett, W.C.; et al. Saturated Fats Compared With Unsaturated Fats and Sources of Carbohydrates in Relation to Risk of Coronary Heart Disease. J. Am. Coll. Cardiol. 2015, 66, 1538–1548. [CrossRef]
- Critselis, E.; Tsiampalis, T.; Damigou, E.; Georgousopoulou, E.; Barkas, F.; Chrysohoou, C.; Skoumas, J.; Pitsavos, C.; Liberopoulos, E.; Tsioufis, C.; et al. High Fish Intake Rich in N-3 Polyunsaturated Fatty Acids Reduces Cardiovascular Disease Incidence in Healthy Adults: The ATTICA Cohort Study (2002-2022). Front. Physiol. 2023, 14. [CrossRef]
- Park, G.-H.; Cho, J.-H.; Lee, D.; Kim, Y. Association between Seafood Intake and Cardiovascular Disease in South Korean Adults: A Community-Based Prospective Cohort Study. Nutrients 2022, 14, 4864. [CrossRef]
- Petersen, K.S.; Maki, K.C.; Calder, P.C.; Belury, M.A.; Messina, M.; Kirkpatrick, C.F.; Harris, W.S. Perspective on the Health Effects of Unsaturated Fatty Acids and Commonly Consumed Plant Oils High in Unsaturated Fat. British Journal of Nutrition 2024, 132, 1039–1050. [CrossRef]
- Bork, C.S.; Lundbye-Christensen, S.; Venø, S.K.; Lasota, A.N.; Schmidt, E.B.; Overvad, K. Plant N-3 PUFA Intake May Lower the Risk of Atherosclerotic Cardiovascular Disease Only among Subjects with a Low Intake of Marine n-3 PUFAs. Eur. J. Nutr. 2022, 61, 557–559. [CrossRef]
- Nestel, P.J.; Mori, T.A. Dietary Patterns, Dietary Nutrients and Cardiovascular Disease. Rev. Cardiovasc. Med. 2022, 23. [CrossRef]
- Wang, Y.; Liu, B.; Han, H.; Hu, Y.; Zhu, L.; Rimm, E.B.; Hu, F.B.; Sun, Q. Associations between Plant-Based Dietary Patterns and Risks of Type 2 Diabetes, Cardiovascular Disease, Cancer, and Mortality—a Systematic Review and Meta-Analysis. Nutr. J. 2023, 22, 46. [CrossRef]
- Rizzo, G.; Baroni, L.; Lombardo, M. Promising Sources of Plant-Derived Polyunsaturated Fatty Acids: A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20.
- Duvall, M.G.; Levy, B.D. DHA- and EPA-Derived Resolvins, Protectins, and Maresins in Airway Inflammation. Eur. J. Pharmacol. 2016, 785. [CrossRef]
- Wang, Z.; Yuan, C.; Huang, T.; Lu, B. Early Nutritional Interventions for Chronic Low-Grade Inflammation. Trends in Endocrinology and Metabolism 2025.
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Gambell, J.; Madden, J.; Calder, P.C.; Jebb, S.A. Compared with Daily, Weekly n-3 PUFA Intake Affects the Incorporation of Eicosapentaenoic Acid and Docosahexaenoic Acid into Platelets and Mononuclear Cells in Humans. Journal of Nutrition 2014, 144. [CrossRef]
- Lindqvist, H.M.; Winkvist, A.; Gjertsson, I.; Calder, P.C.; Armando, A.M.; Quehenberger, O.; Coras, R.; Guma, M. Influence of Dietary N-3 Long Chain Polyunsaturated Fatty Acid Intake on Oxylipins in Erythrocytes of Women with Rheumatoid Arthritis. Molecules 2023, 28. [CrossRef]
- Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes: From Molecules to Man. Biochem. Soc. Trans. 2017, 45.
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. New England Journal of Medicine 2018, 378. [CrossRef]
- Filippou, C.; Tatakis, F.; Polyzos, D.; Manta, E.; Thomopoulos, C.; Nihoyannopoulos, P.; Tousoulis, D.; Tsioufis, K. Overview of Salt Restriction in the Dietary Approaches to Stop Hypertension (DASH) and the Mediterranean Diet for Blood Pressure Reduction. Rev. Cardiovasc. Med. 2022, 23. [CrossRef]
- Onwuzo, C.; Olukorode, J. o; Omokore, O.A.; Odunaike, O.S.; Omiko, R.; Osaghae, O. w; Sange, W.; Orimoloye, D.A.; Kristilere, H.O.; Addeh, E.; et al. DASH Diet: A Review of Its Scientifically Proven Hypertension Reduction and Health Benefits. Cureus 2023. [CrossRef]
- de Souza, P.A.L.; Marcadenti, A.; Portal, V.L. Effects of Olive Oil Phenolic Compounds on Inflammation in the Prevention and Treatment of Coronary Artery Disease. Nutrients 2017, 9.
- Bercea, C.I.; Cottrell, G.S.; Tamagnini, F.; McNeish, A.J. Omega-3 Polyunsaturated Fatty Acids and Hypertension: A Review of Vasodilatory Mechanisms of Docosahexaenoic Acid and Eicosapentaenoic Acid. Br. J. Pharmacol. 2021, 178.
- S, B.; L, R.; P, S.; KS, L. Novel SGC Stimulators and SGC Activators for the Treatment of Heart Failure. Handb. Exp. Pharmacol. 2017, 243.
- Satija, A.; Bhupathiraju, S.N.; Spiegelman, D.; Chiuve, S.E.; Manson, J.E.; Willett, W.; Rexrode, K.M.; Rimm, E.B.; Hu, F.B. Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. J. Am. Coll. Cardiol. 2017, 70, 411–422. [CrossRef]
- Gan, Z.H.; Cheong, H.C.; Tu, Y.-K.; Kuo, P.-H. Association between Plant-Based Dietary Patterns and Risk of Cardiovascular Disease: A Systematic Review and Meta-Analysis of Prospective Cohort Studies. Nutrients 2021, 13, 3952. [CrossRef]
- Choi, Y.; Larson, N.; Steffen, L.M.; Schreiner, P.J.; Gallaher, D.D.; Duprez, D.A.; Shikany, J.M.; Rana, J.S.; Jacobs, D.R. Plant-Centered Diet and Risk of Incident Cardiovascular Disease During Young to Middle Adulthood. J. Am. Heart Assoc. 2021, 10. [CrossRef]
- Yang, Q.; Cao, W.; Zhou, X.; Cao, W.; Xie, Y.; Wang, S. Anti-Thrombotic Effects of α-Linolenic Acid Isolated from Zanthoxylum Bungeanum Maxim Seeds. BMC Complement. Altern. Med. 2014, 14. [CrossRef]
- Singer, P.; Berger, I.; Moritz, V.; Förster, D.; Taube, C. N-6 and N-3 PUFA in Liver Lipids, Thromboxane Formation and Blood Pressure from SHR during Diets Supplemented with Evening Primrose, Sunflowerseed or Fish Oil. Prostaglandins Leukot. Essent. Fatty Acids 1990, 39. [CrossRef]
- Guasch-Ferré, M.; Liu, X.; Malik, V.S.; Sun, Q.; Willett, W.C.; Manson, J.A.E.; Rexrode, K.M.; Li, Y.; Hu, F.B.; Bhupathiraju, S.N. Nut Consumption and Risk of Cardiovascular Disease. J. Am. Coll. Cardiol. 2017, 70. [CrossRef]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. New England Journal of Medicine 2019, 380, 11–22. [CrossRef]
- Goldberg, I.J.; Trent, C.M.; Schulze, P.C. Lipid Metabolism and Toxicity in the Heart. Cell Metab. 2012, 15.
- Shu, H.; Peng, Y.; Hang, W.; Nie, J.; Zhou, N.; Wang, D.W. The Role of CD36 in Cardiovascular Disease. Cardiovasc. Res. 2022, 118.
- Nakatani, K.; Masuda, D.; Kobayashi, T.; Sairyo, M.; Zhu, Y.; Okada, T.; Naito, A.T.; Ohama, T.; Koseki, M.; Oka, T.; et al. Pressure Overload Impairs Cardiac Function in Long-Chain Fatty Acid Transporter CD36-Knockout Mice. Int. Heart J. 2019, 60, 159–167. [CrossRef]
- Umbarawan, Y.; Syamsunarno, M.R.A.A.; Koitabashi, N.; Obinata, H.; Yamaguchi, A.; Hanaoka, H.; Hishiki, T.; Hayakawa, N.; Sano, M.; Sunaga, H.; et al. Myocardial Fatty Acid Uptake through CD36 Is Indispensable for Sufficient Bioenergetic Metabolism to Prevent Progression of Pressure Overload-Induced Heart Failure. Sci. Rep. 2018, 8, 12035. [CrossRef]
- Kim, T.T.; Dyck, J.R.B. The Role of CD36 in the Regulation of Myocardial Lipid Metabolism. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2016, 1861.
- Zhang, M.J.; Karachenets, S.; Gyberg, D.J.; Puccini, S.; Healy, C.L.; Wu, S.C.; Shearer, G.C.; O’Connell, T.D. Free Fatty Acid Receptor 4 in Cardiac Myocytes Ameliorates Ischemic Cardiomyopathy 2024.
- Anbazhagan, A.N.; Priyamvada, S.; Gujral, T.; Bhattacharyya, S.; Alrefai, W.A.; Dudeja, P.K.; Borthakur, A. A Novel Anti-Inflammatory Role of GPR120 in Intestinal Epithelial Cells. Am. J. Physiol. Cell Physiol. 2016, 310. [CrossRef]
- Park, M.H.; Lee, M.; Nam, G.; Kim, M.; Kang, J.; Choi, B.J.; Jeong, M.S.; Park, K.H.; Han, W.H.; Tak, E.; et al. N,N′-Diacetyl-p-Phenylenediamine Restores Microglial Phagocytosis and Improves Cognitive Defects in Alzheimer’s Disease Transgenic Mice. Proc. Natl. Acad. Sci. U. S. A. 2019, 116. [CrossRef]
- Shewale, S. V.; Brown, A.L.; Bi, X.; Boudyguina, E.; Sawyer, J.K.; Alexander-Miller, M.A.; Parks, J.S. In Vivo Activation of Leukocyte GPR120/FFAR4 by PUFAs Has Minimal Impact on Atherosclerosis in LDL Receptor Knockout Mice. J. Lipid Res. 2017, 58. [CrossRef]
- Hirschey, M.D.; Shimazu, T.; Goetzman, E.; Jing, E.; Schwer, B.; Lombard, D.B.; Grueter, C.A.; Harris, C.; Biddinger, S.; Ilkayeva, O.R.; et al. SIRT3 Regulates Mitochondrial Fatty-Acid Oxidation by Reversible Enzyme Deacetylation. Nature 2010, 464, 121–125. [CrossRef]
- Knox, C.; Wilson, M.; Klinger, C.M.; Franklin, M.; Oler, E.; Wilson, A.; Pon, A.; Cox, J.; Chin, N.E. (Lucy); Strawbridge, S.A.; et al. DrugBank 6.0: The DrugBank Knowledgebase for 2024. Nucleic Acids Res. 2024, 52, D1265–D1275. [CrossRef]
- Mallick, R.; Bhowmik, P.; Duttaroy, A.K. Targeting Fatty Acid Uptake and Metabolism in Cancer Cells: A Promising Strategy for Cancer Treatment. Biomedicine & Pharmacotherapy 2023, 167, 115591. [CrossRef]
- Spite, M.; Clària, J.; Serhan, C.N. Resolvins, Specialized Proresolving Lipid Mediators, and Their Potential Roles in Metabolic Diseases. Cell Metab. 2014, 19.
- Al-Shaer, A.E.; Buddenbaum, N.; Shaikh, S.R. Polyunsaturated Fatty Acids, Specialized pro-Resolving Mediators, and Targeting Inflammation Resolution in the Age of Precision Nutrition. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866. [CrossRef]
- Davis, C.R.; Hodgson, J.M.; Woodman, R.; Bryan, J.; Wilson, C.; Murphy, K.J. A Mediterranean Diet Lowers Blood Pressure and Improves Endothelial Function: Results from the MedLey Randomized Intervention Trial. American Journal of Clinical Nutrition 2017, 105. [CrossRef]
- Salehin, S.; Rasmussen, P.; Mai, S.; Mushtaq, M.; Agarwal, M.; Hasan, S.M.; Salehin, S.; Raja, M.; Gilani, S.; Khalife, W.I. Plant Based Diet and Its Effect on Cardiovascular Disease. Int. J. Environ. Res. Public Health 2023, 20.
- Vogtschmidt, Y.D.; Soedamah-Muthu, S.S.; Imamura, F.; Givens, D.I.; Lovegrove, J.A. Replacement of Saturated Fatty Acids from Meat by Dairy Sources in Relation to Incident Cardiovascular Disease: The European Prospective Investigation into Cancer and Nutrition (EPIC)-Norfolk Study. Am. J. Clin. Nutr. 2024, 119, 1495–1503. [CrossRef]
- Hayes, K.C. Dietary Fat and Heart Health: In Search of the Ideal Fat. Asia Pac. J. Clin. Nutr. 2002, 11 Suppl 7.
- Wilkinson, M.J.; Manoogian, E.N.C.; Zadourian, A.; Lo, H.; Fakhouri, S.; Shoghi, A.; Wang, X.; Fleischer, J.G.; Navlakha, S.; Panda, S.; et al. Ten-Hour Time-Restricted Eating Reduces Weight, Blood Pressure, and Atherogenic Lipids in Patients with Metabolic Syndrome. Cell Metab. 2020, 31. [CrossRef]
- Djuricic, I.; Calder, P.C. Omega-3 (n-3) Fatty Acid–Statin Interaction: Evidence for a Novel Therapeutic Strategy for Atherosclerotic Cardiovascular Disease. Nutrients 2024, 16, 962. [CrossRef]
- Fan, H.; Zhou, J.; Yuan, Z. Meta-Analysis Comparing the Effect of Combined Omega-3 + Statin Therapy Versus Statin Therapy Alone on Coronary Artery Plaques. American Journal of Cardiology 2021, 151.
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. New England Journal of Medicine 2018, 378. [CrossRef]
- Plewes, M.R.; Burns, P.D.; Graham, P.E.; Bruemmer, J.E.; Engle, T.E. Influence of Omega-3 Polyunsaturated Fatty Acids from Fish Oil or Meal on the Structure of Lipid Microdomains in Bovine Luteal Cells. Anim. Reprod. Sci. 2018, 193. [CrossRef]
- Cottin, S.C.; Sanders, T.A.; Hall, W.L. The Differential Effects of EPA and DHA on Cardiovascular Risk Factors. In Proceedings of the Proceedings of the Nutrition Society; 2011; Vol. 70.
- McManus, S.; Tejera, N.; Awwad, K.; Vauzour, D.; Rigby, N.; Fleming, I.; Cassidy, A.; Minihane, A.M. Differential Effects of EPA versus DHA on Postprandial Vascular Function and the Plasma Oxylipin Profile in Men. J. Lipid Res. 2016, 57. [CrossRef]
- [Fixed Combination Atorvastatin-Perindopril (Lipercosyl®) for Substitution Treatment of Cardiovascular Risk Management]—PubMed Available online: https://pubmed.ncbi.nlm.nih.gov/33543860/ (accessed on 5 June 2025).
- Teo, K.K.; Mitchell, L.B.; Pogue, J.; Bosch, J.; Dagenais, G.; Yusuf, S. Effect of Ramipril in Reducing Sudden Deaths and Nonfatal Cardiac Arrests in High-Risk Individuals without Heart Failure or Left Ventricular Dysfunction. Circulation 2004, 110, 1413–1417. [CrossRef]
- Sedman, A.J.; Posvar, E. Clinical Pharmacology of Quinapril in Healthy Volunteers and in Patients with Hypertension and Congestive Heart Failure. Angiology 1989, 40, 360–369. [CrossRef]
- Simpson, K.; Jarvis, B. Lisinopril: A Review of Its Use in Congestive Heart Failure. Drugs 2000, 59, 1149–1167. [CrossRef]
- Peters, D.C.; Noble, S.; Plosker, G.L. Trandolapril. An Update of Its Pharmacology and Therapeutic Use in Cardiovascular Disorders. Drugs 1998, 56, 871–893. [CrossRef]
- Zannad, F.; Chati, Z.; Guest, M.; Plat, F.; Baille, N.; Baillie, S.; Tyneside, N.; Baksi, A.; Bloomfield, P.; Bocadamo, I.; et al. Differential Effects of Fosinopril and Enalapril in Patients with Mild to Moderate Chronic Heart Failure. Am. Heart J. 1998, 136, 672–680. [CrossRef]
- Woollett, L.A.; Buckley, D.D.; Yao, L.; Jones, P.J.H.; Granholm, N.A.; Tolley, E.A.; Heubi, J.E. Effect of Ursodeoxycholic Acid on Cholesterol Absorption and Metabolism in Humans. J. Lipid Res. 2003, 44, 935–942. [CrossRef]
- Eikelboom, J.W.; Quinlan, D.J.; O’Donnell, M. Major Bleeding, Mortality, and Efficacy of Fondaparinux in Venous Thromboembolism Prevention Trials. Circulation 2009, 120, 2006–2011. [CrossRef]
- Stroes, E.S.G.; Alexander, V.J.; Karwatowska-Prokopczuk, E.; Hegele, R.A.; Arca, M.; Ballantyne, C.M.; Soran, H.; Prohaska, T.A.; Xia, S.; Ginsberg, H.N.; et al. Olezarsen, Acute Pancreatitis, and Familial Chylomicronemia Syndrome. New England Journal of Medicine 2024, 390, 1781–1792. [CrossRef]
- Albosta, M.; Grant, J.K.; Michos, E.D. Bempedoic Acid: Lipid Lowering for Cardiovascular Disease Prevention. Heart Int. 2023, 17, 27. [CrossRef]
- Chang, Y.C.; Yeh, L.C.; Lin, T.T.; Huang, C.C.; Hung, C.S.; Yang, Y.Y.; Chuang, S.L.; Lin, L.Y.; Lin, L.C.; Kao, H.L.; et al. Colchicine to Prevent Cardiovascular Death after an Acute Myocardial Infarction. Journal of the Formosan Medical Association 2024. [CrossRef]
- Al-Atta, A.; Kuzemczak, M.; Alkhalil, M. Colchicine for the Prevention of Ischemic Stroke. Brain Circ. 2021, 7, 187–193. [CrossRef]
- Ahmad, T.; Manohar, S.A.; Stencel, J.D.; Le Jemtel, T.H. Dobutamine in the Management of Advanced Heart Failure. Journal of Clinical Medicine 2024, Vol. 13, Page 3782 2024, 13, 3782. [CrossRef]
- Olawi, N.; Krüger, M.; Grimm, D.; Infanger, M.; Wehland, M. Nebivolol in the Treatment of Arterial Hypertension. Basic Clin. Pharmacol. Toxicol. 2019, 125, 189–201. [CrossRef]
- Wang, Y. [Retracted] The Efficacy and Safety of Bisoprolol in the Treatment of Myocardial Infarction with Cardiac Insufficiency. Comput. Math. Methods Med. 2022, 2022, 3098726. [CrossRef]
- Packer, M.; Colucci, W.S.; Sackner-Bernstein, J.D.; Liang, C.S.; Goldscher, D.A.; Freeman, I.; Kukin, M.L.; Kinhal, V.; Udelson, J.E.; Klapholz, M.; et al. Double-Blind, Placebo-Controlled Study of the Effects of Carvedilol in Patients with Moderate to Severe Heart Failure: The PRECISE Trial. Circulation 1996, 94, 2793–2799. [CrossRef]
- Yan, C.; Duan, G.; Zhang, Y.; Wu, F.X.; Pan, Y.; Wang, J. Predicting Drug-Drug Interactions Based on Integrated Similarity and Semi-Supervised Learning. IEEE/ACM Trans. Comput. Biol. Bioinform. 2022, 19, 168–179. [CrossRef]
- Laviolette, S.R.; Nalepa, I.; Zelek-Molik, A.; Szeleszczuk, Ł.; Fr, D. Propranolol versus Other Selected Drugs in the Treatment of Various Types of Anxiety or Stress, with Particular Reference to Stage Fright and Post-Traumatic Stress Disorder. International Journal of Molecular Sciences 2022, Vol. 23, Page 10099 2022, 23, 10099. [CrossRef]
- Mahmood, A.; Eqan, M.; Pervez, S.; Javed, R.; Ullah, R.; Islam, A.; Khan, A.; Baig, A.A.; Kumar, G.; Abaid-Ullah, M.; et al. Drugs Resistance in Heart Diseases. Biochemistry of Drug Resistance 2021, 295–334. [CrossRef]
- Feingold, K.R. Cholesterol Lowering Drugs. Endotext 2024.
- Butt, W.Z.; Yee, J.K. The Role of Non-Statin Lipid-Lowering Medications in Youth with Hypercholesterolemia. Curr. Atheroscler. Rep. 2022, 24, 379–389. [CrossRef]
- Calderone, D.; Greco, A.; Ingala, S.; Agnello, F.; Franchina, G.; Scalia, L.; Buccheri, S.; Capodanno, D. Efficacy and Safety of Aspirin for Primary Cardiovascular Risk Prevention in Younger and Older Age: An Updated Systematic Review and Meta-Analysis of 173,810 Subjects from 21 Randomized Studies. Thromb. Haemost. 2022, 122, 445–455. [CrossRef]
- Kuang, H. yu; Li, Q.; Du, H. an; Chen, M.; Yin, Y. hui Efficacy and Safety of Long-Term Oral Bosentan in Different Types of Pulmonary Arterial Hypertension: A Systematic Review and Meta-Analysis. American Journal of Cardiovascular Drugs 2021, 21, 181–191. [CrossRef]
- Biswas, K.; Tiwari, A.; Jadhav, P.; Goel, A.; Chanukya, G. V Rosuvastatin and Fenofibrate Combination in The Treatment of Mixed Hyperlipidemia: A Narrative Review. Journal of Current Medical Research and Opinion 2021, 4, 867–877. [CrossRef]
- Lusk, J.B.; Glover, L.S.; Soneji, S.; Granger, C.B.; O’Brien, E.; Pagidipati, N. Cardiovascular Event Reduction among a US Population Eligible for Semaglutide per the SELECT Trial. Am. Heart J. 2024, 276, 110–114. [CrossRef]
- Drucker, D.J. Prevention of Cardiorenal Complications in People with Type 2 Diabetes and Obesity. Cell Metab. 2024, 36, 338–353. [CrossRef]
- Suda, N.; Leon-Martinez, D.; Peter, P.R.; Flannery, C.A.; Irani, R.A. Management of Severe Hypertriglyceridemia in Pregnancy With Niacin: Reevaluating Safety and Therapeutic Benefits. Case Rep. Endocrinol. 2025, 2025, 2644678. [CrossRef]
- Forry, J.; Chappell, A.; Alexander, E.; Allen, J. Tenecteplase: A Review of Its Pharmacology and Uses. AACN Adv. Crit. Care 2023, 34, 77–83. [CrossRef]
- Chambergo-Michilot, D.; Alur, A.; Kulkarni, S.; Agarwala, A. Mipomersen in Familial Hypercholesterolemia: An Update on Health-Related Quality of Life and Patient-Reported Outcomes. Vasc. Health Risk Manag. 2022, 18, 73–80. [CrossRef]
- Vaduganathan, M.; Filippatos, G.; Claggett, B.L.; Desai, A.S.; Jhund, P.S.; Henderson, A.; Brinker, M.; Kolkhof, P.; Schloemer, P.; Lay-Flurrie, J.; et al. Finerenone in Heart Failure and Chronic Kidney Disease with Type 2 Diabetes: The FINE-HEART Pooled Analysis of Cardiovascular, Kidney, and Mortality Outcomes. Nat. Med. 2024, 30, 3758–3764. [CrossRef]
- Kiaei, A.A.; Boush, M.; Abadijou, S.; Momeni, S.; Safaei, D.; Bahadori, R.; Salari, N.; Mohammadi, M. Recommending Drug Combinations Using Reinforcement Learning Targeting Genes/Proteins Associated with Heterozygous Familial Hypercholesterolemia: A Comprehensive Systematic Review and Net-Work Meta-Analysis. 2023. [CrossRef]
- Oyama, K.; Furtado, R.H.M.; Fagundes, A.; Zelniker, T.A.; Tang, M.; Kuder, J.; Murphy, S.A.; Hamer, A.; Wang, H.; Keech, A.C.; et al. Effect of Evolocumab on Complex Coronary Disease Requiring Revascularization. J. Am. Coll. Cardiol. 2021, 77, 259–267. [CrossRef]
- Chaudhary, R.; Mohananey, A.; Sharma, S.P.; Singh, S.; Singh, A.; Kondur, A. Improving Outcomes in Cardiovascular Diseases: A Review on Vorapaxar. Cardiol. Rev. 2022, 30, 241–246. [CrossRef]
- Bryan Richard, S.; Huang, B.; Liu, G.; Yang, Y.; Luo, S. Impact of Ivabradine on the Cardiac Function of Chronic Heart Failure Reduced Ejection Fraction: Meta-Analysis of Randomized Controlled Trials. Clin. Cardiol. 2021, 44, 463–471. [CrossRef]
- Chen, W.-H.; Lau, C.-P.; Lau, C.A. Recent Advances in Antithrombin Therapy for Acute Coronary Syndromes. Journal of the Hong Kong College of Cardiology 2022, 7, 109–118. [CrossRef]
- Imbalzano, E.; Orlando, L.; Dattilo, G.; Gigliotti De Fazio, M.; Camporese, G.; Russo, V.; Perrella, A.; Bernardi, F.F.; Di Micco, P. Update on the Pharmacological Actions of Enoxaparin in Nonsurgical Patients. Medicina 2024, Vol. 60, Page 156 2024, 60, 156. [CrossRef]
- Wang, J.; Zou, D. Tirofiban-Induced Thrombocytopenia. Ann. Med. 2023, 55, 2233425. [CrossRef]
- Ganda, O.P. Triglyceride-Rich Lipoproteins, Remnant-Cholesterol, and Atherosclerotic Cardiovascular Disease. Curr. Opin. Lipidol. 2023, 34, 105–113. [CrossRef]
- Patel, K.; Patil, J.; Girase, T.; Tatiya, A.; Patil, D. An Overview of the Applications of Gemfibrozil Nano-Formulation in Hyperlipidemia. Materials Proceedings 2023, Vol. 14, Page 45 2023, 14, 45. [CrossRef]
- Kalstad, A.A.; Myhre, P.L.; Laake, K.; Tveit, S.H.; Schmidt, rik B.; Smith, P.; Nilsen, D.W.T.; Tveit, A.; Fagerland, M.W.; Solheim, S.; et al. Effects of N-3 Fatty Acid Supplements in Elderly Patients after Myocardial Infarction: A Randomized, Controlled Trial. Circulation 2021, 143, 528–539. [CrossRef]
- Sanderson, N.C.; Parker, W.A.E.; Storey, R.F. Ticagrelor: Clinical Development and Future Potential. Rev. Cardiovasc. Med. 2021, 22, 373–394. [CrossRef]
- Mitsis, A.; Gragnano, F. Myocardial Infarction with and without ST-Segment Elevation: A Contemporary Reappraisal of Similarities and Differences. Curr. Cardiol. Rev. 2020, 17, 49–58. [CrossRef]
- Santos, R.D.; Wiegman, A.; Caprio, S.; Cariou, B.; Averna, M.; Poulouin, Y.; Scemama, M.; Manvelian, G.; Garon, G.; Daniels, S. Alirocumab in Pediatric Patients With Heterozygous Familial Hypercholesterolemia: A Randomized Clinical Trial. JAMA Pediatr. 2024, 178, 283–293. [CrossRef]
- Murphy, L.R.; Hill, T.P.; Paul, K.; Talbott, M.; Golovko, G.; Shaltoni, H.; Jehle, D. Tenecteplase Versus Alteplase for Acute Stroke: Mortality and Bleeding Complications. Ann. Emerg. Med. 2023, 82, 720–728. [CrossRef]
- Pitt, B.; Bhatt, D.L.; Szarek, M.; Cannon, C.P.; Leiter, L.A.; McGuire, D.K.; Lewis, J.B.; Riddle, M.C.; Voors, A.A.; Metra, M.; et al. Effect of Sotagliflozin on Early Mortality and Heart Failure-Related Events: A Post Hoc Analysis of SOLOIST-WHF. JACC Heart Fail. 2023, 11, 879–889. [CrossRef]
- Yoo, Y.G.; Lim, M.J.; Kim, J.S.; Jeong, H.E.; Ko, H.; Shin, J.Y. Risk of Myocardial Infarction, Heart Failure, and Cerebrovascular Disease with the Use of Valsartan, Losartan, Irbesartan, and Telmisartan in Patients. Medicine (United States) 2023, 102, E36098. [CrossRef]
- Gillmore, J.D.; Judge, D.P.; Cappelli, F.; Fontana, M.; Garcia-Pavia, P.; Gibbs, S.; Grogan, M.; Hanna, M.; Hoffman, J.; Masri, A.; et al. Efficacy and Safety of Acoramidis in Transthyretin Amyloid Cardiomyopathy. New England Journal of Medicine 2024, 390, 132–142. [CrossRef]
- Liu, T.; Zuo, R.; Wang, J.; Huangtao, Z.; Wang, B.; Sun, L.; Wang, S.; Li, B.; Zhu, Z.; Pan, Y. Cardiovascular Disease Preventive Effects of Aspirin Combined with Different Statins in the United States General Population. Sci. Rep. 2023, 13, 1–7. [CrossRef]
- Cai, T.; Abel, L.; Langford, O.; Monaghan, G.; Aronson, J.K.; Stevens, R.J.; Lay-Flurrie, S.; Koshiaris, C.; McManus, R.J.; Richard Hobbs, F.D.; et al. Associations between Statins and Adverse Events in Primary Prevention of Cardiovascular Disease: Systematic Review with Pairwise, Network, and Dose-Response Meta-Analyses. BMJ 2021, 374, 1537. [CrossRef]
- Grinspoon, S.K.; Fitch, K. V.; Zanni, M. V.; Fichtenbaum, C.J.; Umbleja, T.; Aberg, J.A.; Overton, E.T.; Malvestutto, C.D.; Bloomfield, G.S.; Currier, J.S.; et al. Pitavastatin to Prevent Cardiovascular Disease in HIV Infection. New England Journal of Medicine 2023, 389, 687–699. [CrossRef]
- Yang, X.H.; Zhang, B.L.; Cheng, Y.; Fu, S.K.; Jin, H.M. Statin Use and the Risk of CVD Events, Stroke, and All-Cause Mortality in Patients with Diabetes: A Systematic Review and Meta-Analysis. Nutrition, Metabolism and Cardiovascular Diseases 2022, 32, 2470–2482. [CrossRef]
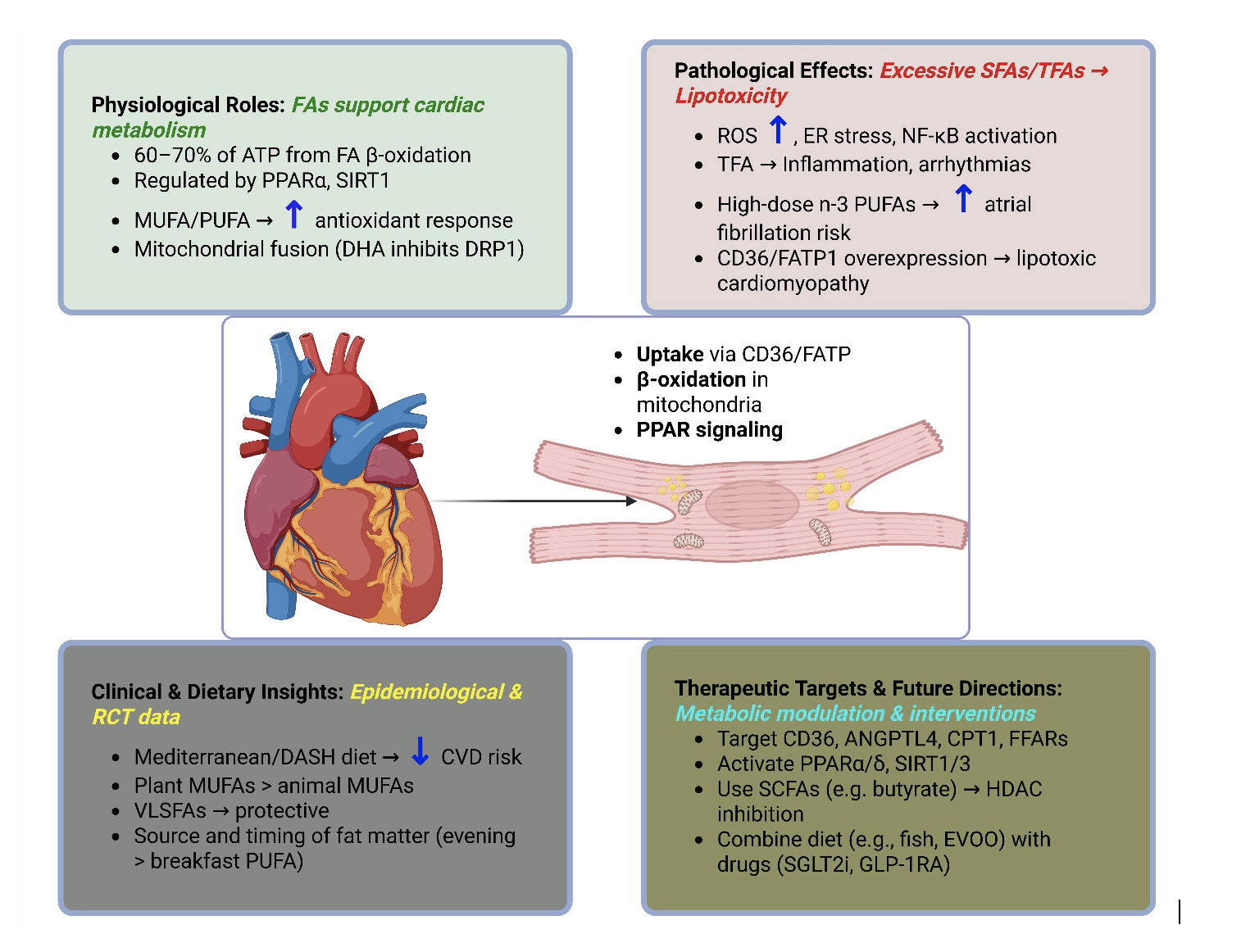
| Fatty Acid Types | Key Pathogenic Mechanisms | Associated CVD Outcomes |
| Saturated FAs (SFAs)(e.g., palmitate, stearate) | • ER stress → UPR activation (PERK/IRE1 → CHOP) → caspase-3–mediated apoptosis • Mitochondrial dysfunction (ceramide accumulation, impaired electron transport) • Lipid intermediate buildup (ceramides, DAG) → insulin resistance, fibrosis |
• Hypertrophic cardiomyopathy • Left ventricular dysfunction • Increased risk of heart failure • Atherosclerosis progression |
| Trans FAs (TFAs)(e.g., industrial elaidic acid) | • NF-κB–mediated inflammation (↑ TNF-α, IL-6) • NADPH oxidase → ↑ superoxide → oxidative damage • Endothelial dysfunction (↓ eNOS), macrophage infiltration • Impaired phospholipid membrane integrity → pro-arrhythmic remodeling |
• Accelerated atherosclerosis • Coronary artery disease • Exacerbated ischemia-reperfusion injury • Higher incidence of arrhythmias |
| n-3 PUFAs(e.g., EPA, DHA) | • Low-Moderate Dose (1–2 g/day):—↓ NF-κB activation, ↓ proinflammatory eicosanoids—↑ antioxidant enzymes (SOD, catalase)—Membrane stabilization → improved ion channel function • High Dose (>3 g/day):—Altered ion currents → prolonged repolarization—Potential imbalance of ROS if excess incorporation |
• ↓ Major adverse cardiovascular events (MACE) at moderate intake • ↓ post-MI inflammation • ↓ sudden cardiac death • ↑ risk of atrial fibrillation at high dose |
| MUFAs (Plant-Derived)(e.g., oleic acid) | • ↑ HDL, ↓ oxidized LDL • ↑ eNOS activity → vasodilation • Redirect SFA-induced lipids into triglycerides rather than ceramides • Anti-inflammatory via NF-κB suppression |
• ↓ CVD mortality • ↓ progression of atherosclerosis • Improved diastolic function |
| MUFAs (Animal-Derived)(e.g., palmitoleic acid) | • May contribute to low-grade inflammation in insulin-resistant states • In conditions of metabolic syndrome, can be converted to ceramides (via salvage pathways) → mild ER stress • Less favorable lipid profile modulation compared with plant MUFAs |
• Neutral to slightly ↑ CVD risk when part of high-SFA diet • Effects largely depend on overall dietary pattern |
| Intervention | Key Trials | Patient Population | CVD Benefit | Risks / Limitations |
| Icosapent ethyl (4 g/day) | REDUCE-IT (2018) | CVD/diabetes + high triglecerides | ↓ 25% MACE; ↓ MI, stroke, CV death |
|
| EPA+DHA (4 g/day, CA) | STRENGTH (2020) | High CVD risk + low HDL/high triglecerides | No significant CV benefit |
|
| EPA+DHA (1 g/day, EE) | ASCEND, VITAL (2019) | Primary prevention (no CVD) | No significant primary prevention benefit |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




