Submitted:
25 March 2026
Posted:
26 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Search Strategy
3. Immunologic Landscape of the Receptive Endometrium
3.1. Uterine Natural Killer Cells
3.2. Regulatory T Cells
3.3. Macrophages and Dendritic Cells
3.4. Cytokine Networks
4. Molecular and Intracellular Signaling Pathways
5. Embryo–Endometrium Communication
5.1. Embryo-Derived Signals
5.2. Extracellular Vesicles
5.3. Endometrium as a Biosensor
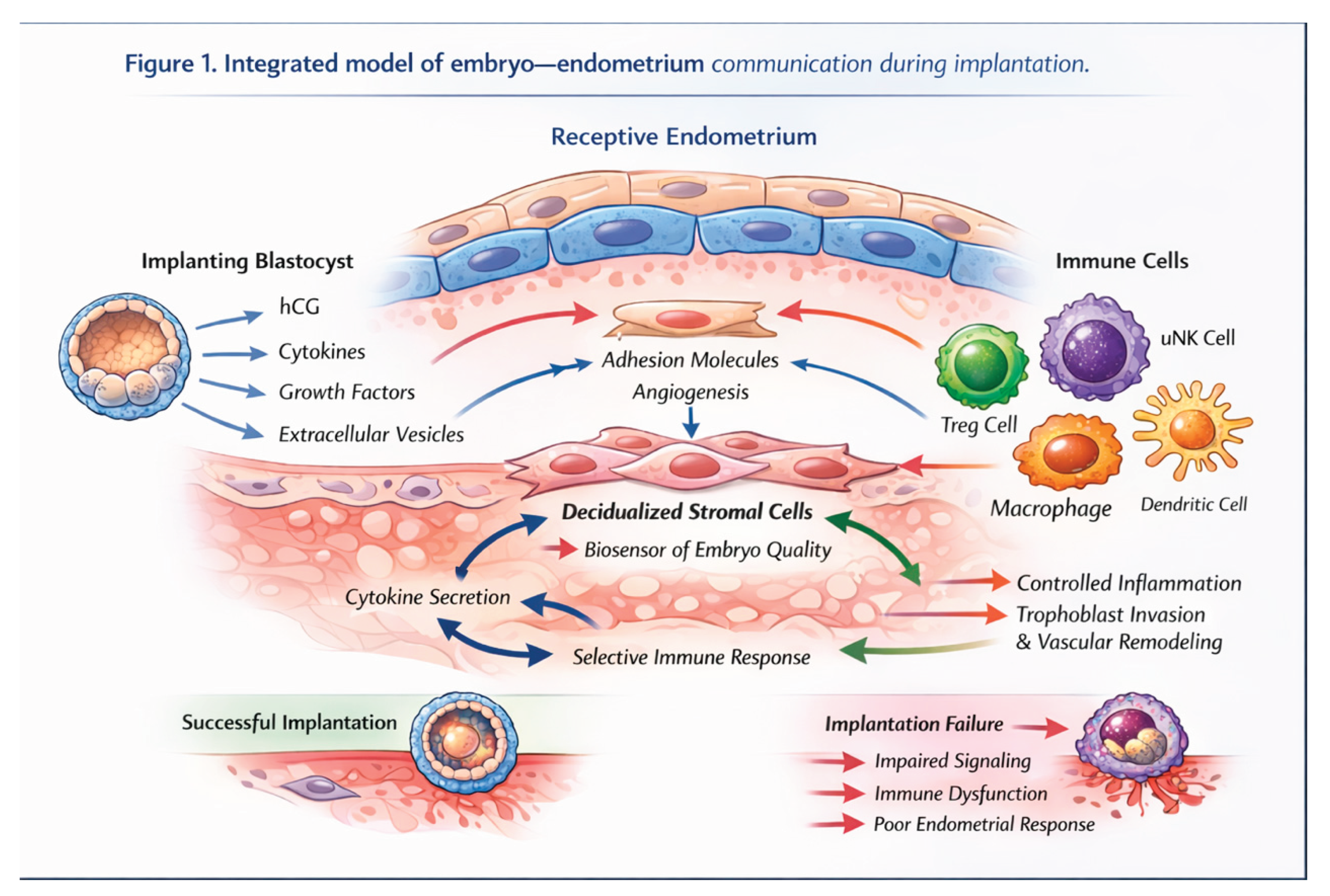
6. Clinical Implications and Therapeutic Perspectives
7. Discussion
| Component | Function | Key Molecules | Clinical Relevance |
|---|---|---|---|
| uNK cells | Angiogenesis, trophoblast regulation | VEGF, PlGF, KIR | Implantation failure, preeclampsia |
| Tregs | Immune tolerance | FOXP3, IL-10, TGF-β | Recurrent implantation failure |
| Macrophages | Tissue remodeling | IL-10, growth factors | Decidualization defects |
| Dendritic cells | Immune regulation | Cytokines | Immune imbalance |
| Cytokines | Signaling regulation | LIF, TNF-α | Implantation outcomes |
| EVs | Intercellular communication | miRNAs, proteins | Emerging biomarkers |
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKT: protein kinase B; |
| ART, assisted reproductive technologies; |
| CSF-1, colony-stimulating factor-1; |
| DC, dendritic cell; |
| ERA, Endometrial Receptivity Array; |
| EVs, extracellular vesicles; |
| FOXO, forkhead box O transcription factor; |
| FOXP3, forkhead box P3; |
| HB-EGF, heparin-binding epidermal growth factor-like growth factor; |
| hCG, human chorionic gonadotropin; |
| HLA, human leukocyte antigen; |
| ICAM-1, intercellular adhesion molecule-1; |
| IGF-1, insulin-like growth factor-1; |
| IL-1β, interleukin 1 beta; |
| IL-6, interleukin 6; |
| IL-10, interleukin 10; |
| IVIG, intravenous immunoglobulin; |
| IVF, in vitro fertilization; |
| JAK, Janus kinase; |
| KIR, killer-cell immunoglobulin-like receptor; |
| LHCGR, luteinizing hormone/chorionic gonadotropin receptor; |
| LIF, leukemia inhibitory factor; |
| MAPK, mitogen-activated protein kinase; |
| miRNA, microRNA; |
| MMPs, matrix metalloproteinases; |
| mRNA, messenger RNA; |
| NF-κB, nuclear factor kappa B; |
| NK, natural killer; |
| PI3K, phosphoinositide 3-kinase; |
| PlGF, placental growth factor; |
| STAT3, signal transducer and activator of transcription 3; |
| TGF-β, transforming growth factor beta; |
| Th1, T helper type 1; |
| Th2, T helper type 2; |
| TNF-α, tumor necrosis factor alpha; |
| Tregs, regulatory T cells; |
| uNK, uterine natural killer; |
| VEGF, vascular endothelial growth factor. |
References
- Psychoyos, A. Uterine receptivity for nidation. Ann N Y Acad Sci. 1986, 476, 36–42. [Google Scholar] [CrossRef]
- Dey, SK; Lim, H; et al. Molecular cues to implantation. Endocr Rev. 2004, 25, 341–373. [Google Scholar] [CrossRef] [PubMed]
- Macklon, NS; Brosens, JJ. Resolving recurrent implantation failure. Reprod Biomed Online. 2025, 50, 104827. [Google Scholar] [CrossRef]
- Cimadomo, D; et al. ESHRE good practice recommendations on recurrent implantation failure. Hum Reprod Open. 2023, 2023, hoad023. [Google Scholar]
- Mor, G; Cardenas, I. The immune system in pregnancy. Am J Reprod Immunol. 2010, 63, 425–433. [Google Scholar] [CrossRef]
- Erlebacher, A. Immunology of the maternal–fetal interface. Annu Rev Immunol. 2013, 31, 387–411. [Google Scholar] [CrossRef]
- Zenclussen, AC. Adaptive immune responses during pregnancy. Am J Reprod Immunol. 2013, 69, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Lessey, BA. Adhesion molecules and implantation. J Reprod Immunol. 2002, 55, 101–112. [Google Scholar] [CrossRef]
- Díaz-Gimeno, P; et al. A genomic diagnostic tool for human endometrial receptivity based on the transcriptomic signature. Fertil Steril. 2011, 95, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Male, V; Moffett, A. Natural Killer Cells in the Human Uterine Mucosa. Annu Rev Immunol. 2023, 41, 127–151. [Google Scholar] [CrossRef]
- King, A; et al. Functions of human decidual NK cells. Am J Reprod Immunol. 1996, 35, 258–260. [Google Scholar] [CrossRef]
- Hanna, J; et al. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med. 2006, 12, 1065–1074. [Google Scholar] [CrossRef]
- Lash, GE; et al. Review: Functional role of uterine natural killer (uNK) cells in human early pregnancy decidua. Placenta. 2010, 31, 87–92. [Google Scholar] [CrossRef]
- Moffett, A; Colucci, F. Uterine NK cells: active regulators at the maternal–fetal interface. J Clin Investig. 2014, 124, 1872–1879. [Google Scholar] [CrossRef]
- Hiby, SE; et al. Combinations of maternal KIR and fetal HLA-C genes influence the risk of preeclampsia and reproductive success. J Exp Med. 2004, 200, 957–965. [Google Scholar] [CrossRef]
- Parham, P. MHC class I molecules and KIRs in human history, health and survival. Nat Rev Immunol. 2005, 5, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, S; Sunkara, SK. Natural killer cells in female infertility and recurrent miscarriage: a systematic review and meta-analysis. Hum Reprod Update. 2014, 20, 429–438. [Google Scholar] [CrossRef]
- Sakaguchi, S; et al. Regulatory T cells and immune tolerance. Cell. 2008, 133, 775–787. [Google Scholar] [CrossRef]
- Robertson, SA; Care, AS; Moldenhauer, LM. Regulatory T cells in embryo implantation and the immune response to pregnancy. J Clin Investig. 2018, 128, 4224–4235. [Google Scholar] [CrossRef] [PubMed]
- Shima, T; et al. Uterine CD11c+ cells induce the development of paternal antigen-specific Tregs via seminal plasma priming. J Reprod Immunol. 2020, 141, 103165. [Google Scholar] [CrossRef] [PubMed]
- Mor, G; Aldo, P; Alvero, AB. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol. 2017, 17, 469–482. [Google Scholar] [CrossRef]
- Plaks, V; et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J Clin Invest. 2008, 118, 3954–65. [Google Scholar] [CrossRef]
- Robertson, SA; et al. Immune determinants of endometrial receptivity: a biological perspective. Fertil Steril. 2022, 117, 1107–1120. [Google Scholar] [CrossRef]
- Lim, HJ; Dey, SK. HB-EGF: a unique mediator of embryo-uterine interactions during implantation. Exp Cell Res. 2009, 315, 619–626. [Google Scholar] [CrossRef]
- Etezadi, A; et al. The evaluation role of T helper 17 cells in pregnancy and abortion: A molecular and future perspective: Narrative review. Int J Reprod Biomed. 2025, 23, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, JS; Rosler, KM; Harrison, DA. The JAK/STAT signaling pathway. J Cell Sci. 2004, 117, 1281–1283. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z; et al. Key factors mediated by PI3K signaling pathway and related genes in endometrial carcinoma. J Bioenerg Biomembr. 2020, 52, 465–473. [Google Scholar] [CrossRef]
- Tang, Y; et al. P38α MAPK is a gatekeeper of uterine progesterone responsiveness at peri-implantation via Ube3c-mediated PGR degradation. Proc Natl Acad Sci U S A. 2022, 119, e2206000119. [Google Scholar] [CrossRef]
- Gellersen, B; Brosens, JJ. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr Rev. 2014, 35, 851–905. [Google Scholar] [CrossRef]
- Macklon, NS; Brosens, JJ. The endometrium as a sensor of embryo quality. Biol Reprod. 2014, 91, 98. [Google Scholar] [CrossRef] [PubMed]
- Voros, C; et al. Molecular Crosstalk Between Intrauterine hCG and Endometrial Receptivity: Signalling Pathways, Immune Modulation, and Translational Perspectives in IVF. Int J Mol Sci. 2025, 27, 278. [Google Scholar] [CrossRef]
- Simon, C; et al. Interleukin-1 system crosstalk between embryo and endometrium in implantation. Hum Reprod. 1995, 2, 43–54. [Google Scholar] [CrossRef]
- Chen, K; et al. The Role of Extracellular Vesicles in Embryo Implantation. Front. Endocrinol (Lausanne) 2022, 13, 809596. [Google Scholar] [CrossRef]
- Vilella, F; et al. Hsa-miR-30d, secreted by the human endometrium, is taken up by the pre-implantation embryo and might modify its transcriptome. Development. 2015, 142, 3210–21. [Google Scholar] [CrossRef] [PubMed]
- Teklenburg, G; et al. Natural selection of human embryos: decidualizing endometrial stromal cells serve as sensors of embryo quality upon implantation. PLoS One 2010, 5, e10258. [Google Scholar] [CrossRef]
- Altmäe, S; et al. Meta-signature of human endometrial receptivity: a meta-analysis and validation study of transcriptomic biomarkers. Sci Rep. 2017, 7, 10077. [Google Scholar] [CrossRef]
- Siristatidis, C; et al. Immunotherapy in IVF. Cochrane Database Syst Rev 2018, 3, CD011963. [Google Scholar]
- Santos, ACFF; et al. Immunosuppressants in women with repeated implantation failure in assisted reproductive techniques: a systematic review and meta-analysis. Rev Bras Ginecol Obstet. 2024, 46, e-rbgo70. [Google Scholar] [CrossRef] [PubMed]
- Moreno, I; et al. Evidence that the endometrial microbiota has an effect on implantation success or failure. Am J Obstet Gynecol. 2016, 215, 684–703. [Google Scholar] [CrossRef]
- Mlodzik, N; et al. Endometrial microbiota - do they mean more than we have expected? Ginekol Pol. 2020, 91, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, S; Whiteside, S; Li, SY; et al. Implantation and Decidualization in PCOS: Unraveling the Complexities of Pregnancy. Int J Mol Sci. 2024, 25, 1203. [Google Scholar] [CrossRef]
- Segura-Benítez, M; Bas-Rivas, A; Juárez-Barber, E; et al. Human blastocysts uptake extracellular vesicles secreted by endometrial cells containing miRNAs related to implantation. Hum Reprod. 2023, 38, 1547–1559. [Google Scholar] [CrossRef]
- Poh, QH; Rai, A; Greening, DW. Omics insights into extracellular vesicles in embryo implantation and their therapeutic utility. Proteomics 2023, 23, e2200107. [Google Scholar] [CrossRef]
- Muter, J; Lynch, VJ; McCoy, RC; Brosens, JJ. Human embryo implantation. Development 2023, 150, dev201507. [Google Scholar] [CrossRef] [PubMed]
- Kovalevska, L; Shcherbak, S; Kheifetz, I. The Role of the Endometrium in Implantation: A Modern View. Int J Mol Sci. 2024, 25, 9746. [Google Scholar]
- Liu, S; Diao, L; Li, S; et al. Time-series single-cell transcriptomic profiling of luteal-phase endometrium uncovers dynamic characteristics and its dysregulation in recurrent implantation failures. Nat Commun. 2025, 16, 44. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




