Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Anthropometric and Blood Pressure Assessment
Blood Sampling
Chemiluminescence Immunoassay (CLIA)
Multiplex Assay
Statistical Analyses
Results
Sex-Based Demographic, Anthropometric, and Blood Pressure Characteristics
Sex-Based Glucoregulatory and Lipid Profile Markers
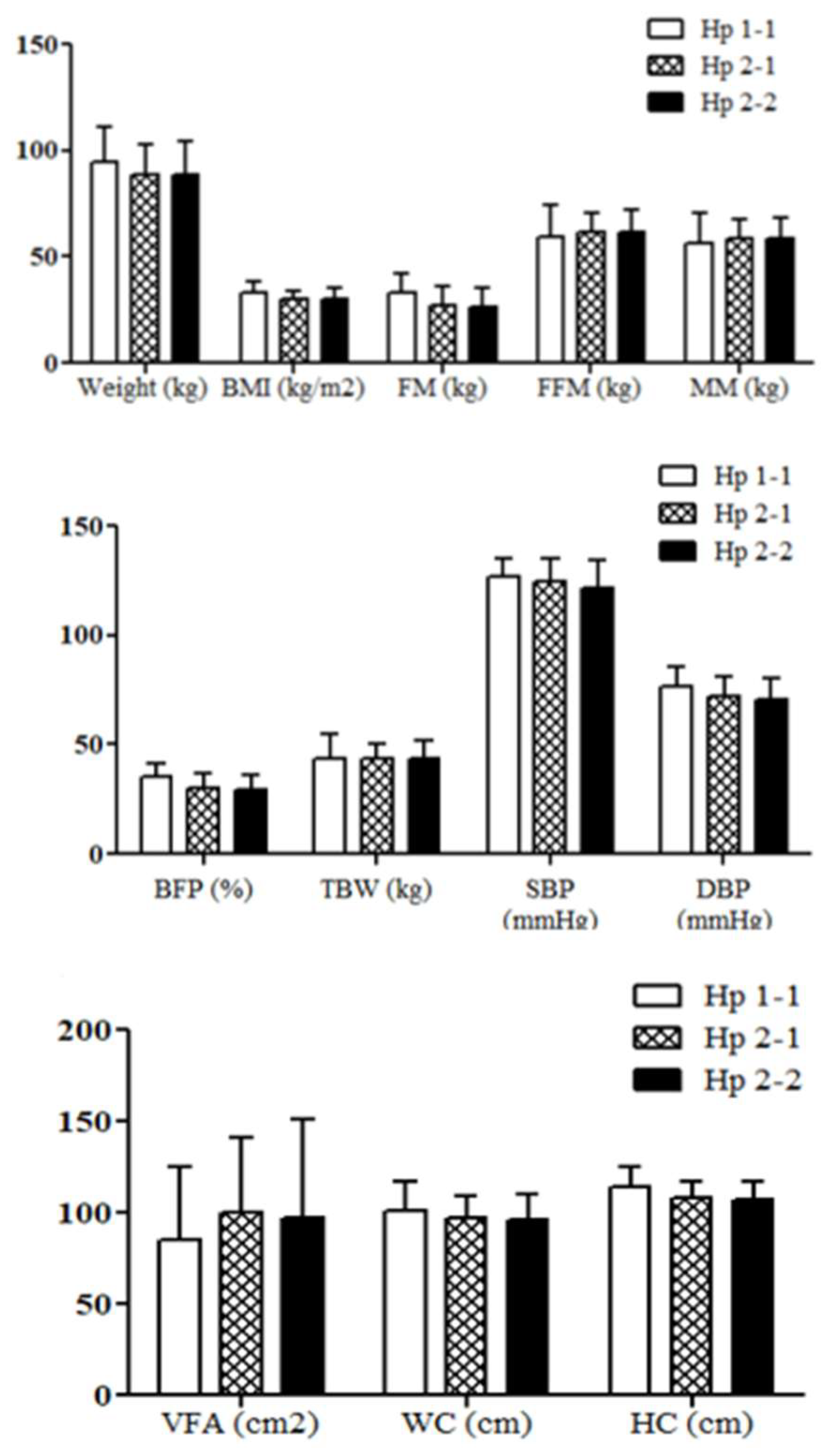
Hp Genotype-Based Anthropometric and Blood Pressure Measures
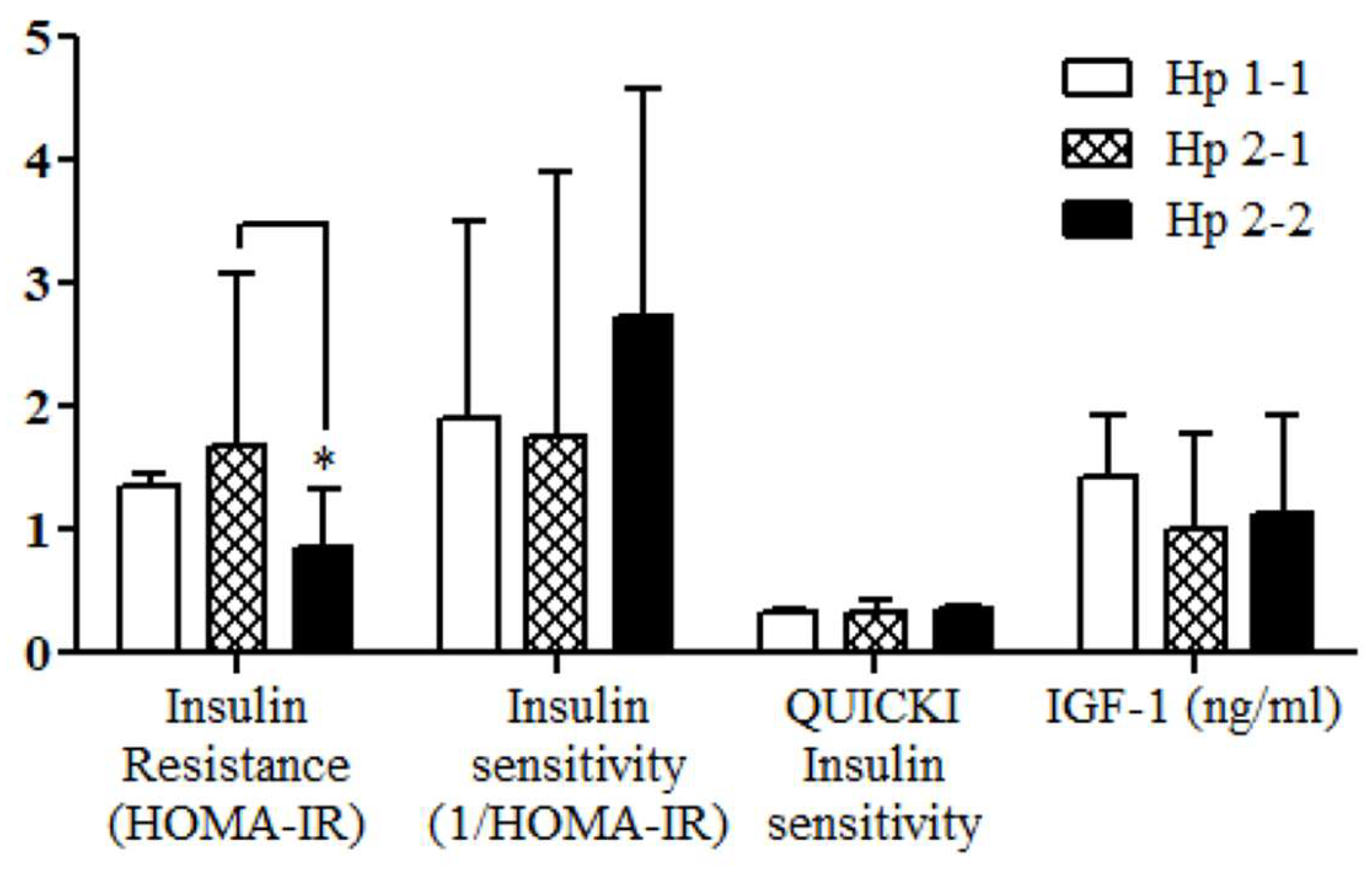
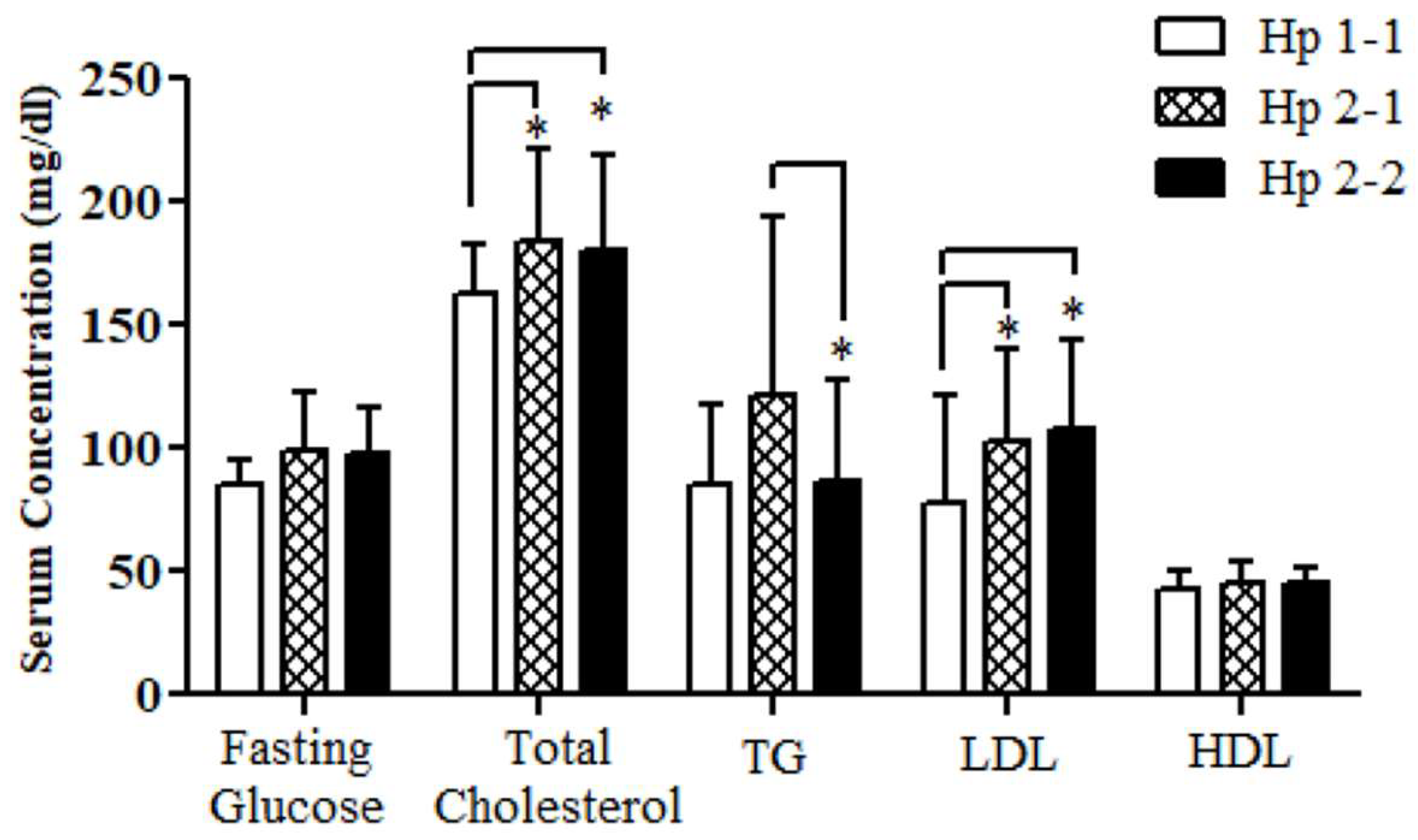
Hp Genotype-Based Glucoregulatory and Lipid Profile Parameters
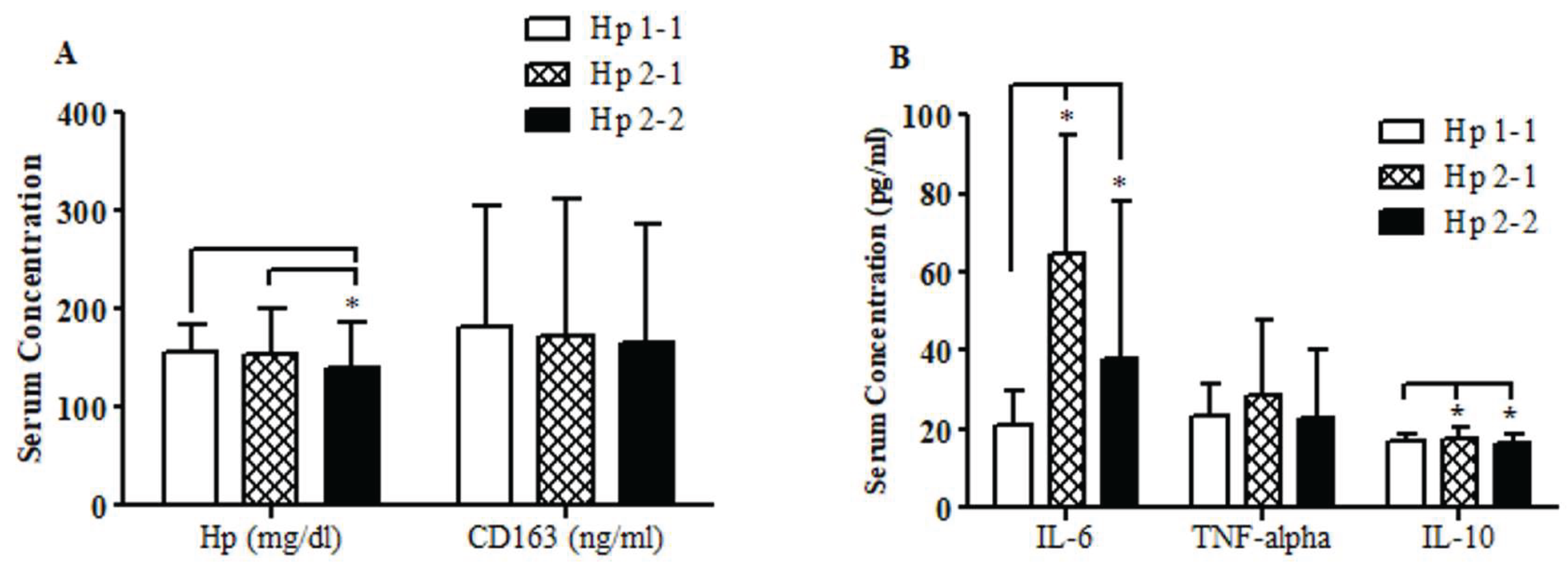
Hp Genotype-Based Serum CD163, Haptoglobin, and Inflammatory Markers
Discussion
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Afolabi HA, Zakaria Z, Salleh SM, Ch’ng ES, Nafi SNM, Aziz AABA, et al. Obesity: a prerequisite for major chronic illnesses. Obesity-Recent Insights and Therapeutic Options: IntechOpen; 2023.
- Sarzani R, Landolfo M, Di Pentima C, Ortensi B, Falcioni P, Sabbatini L, et al. Adipocentric origin of the common cardiometabolic complications of obesity in the young up to the very old: pathophysiology and new therapeutic opportunities. Frontiers in Medicine. 2024;11. [CrossRef]
- Powell-Wiley TM, Poirier P, Burke LE, Després JP, Gordon-Larsen P, Lavie CJ, et al. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation. 2021;143:e984-e1010.
- Wand H, Moodley J, Reddy T, Vujovich-Dunn C, Naidoo S. Quantifying impact of obesity on cardiometabolic comorbidities and public health implications: Insights from South Africa. Obesity Research & Clinical Practice. 2023;17:122-9. [CrossRef]
- Damanti S, Citterio L, Zagato L, Brioni E, Magnaghi C, Simonini M, et al. DNA polymorphisms in inflammatory and endocrine signals linked to frailty are also associated with obesity: data from the FRASNET cohort. Front Endocrinol (Lausanne). 2024;15:1412160.
- Chang C-S, Lu Y-J, Chang H-H, Hsu S-H, Kuo P-H, Shieh C-C, et al. Role of adiponectin gene variants, adipokines and hydrometry-based percent body fat in metabolically healthy and abnormal obesity. Obesity Research & Clinical Practice. 2018;12:49-61.
- An S-M, Cho S-H, Yoon JC. Adipose tissue and metabolic health. Diabetes & Metabolism Journal. 2023;47:595-611.
- Wen X, Zhang B, Wu B, Xiao H, Li Z, Li R, et al. Signaling pathways in obesity: mechanisms and therapeutic interventions. Signal Transduction and Targeted Therapy. 2022;7:298.
- Rodríguez-Hernández H, Simental-Mendía LE, Rodríguez-Ramírez G, Reyes-Romero MA. Obesity and inflammation: epidemiology, risk factors, and markers of inflammation. Int J Endocrinol. 2013;2013:678159.
- Schleh MW, Ameka MK, Rodriguez AS, Hasty AH. Deficiency of the Hemoglobin-Haptoglobin Receptor, CD163, Worsens Insulin Sensitivity in Obese Male Mice. Diabetes. 2024;73:1990-2002.
- Chintagari NR, Nguyen J, Belcher JD, Vercellotti GM, Alayash AI. Haptoglobin attenuates hemoglobin-induced heme oxygenase-1 in renal proximal tubule cells and kidneys of a mouse model of sickle cell disease. Blood Cells, Molecules, and Diseases. 2015;54:302-6. [CrossRef]
- Agyemang AA, Kvist SV, Brinkman N, Gentinetta T, Illa M, Ortenlöf N, et al. Cell-free oxidized hemoglobin drives reactive oxygen species production and pro-inflammation in an immature primary rat mixed glial cell culture. Journal of Neuroinflammation. 2021;18:1-13.
- Marvasti TB, Moody AR, Singh N, Maraj T, Tyrrell P, Afshin M. Haptoglobin 2-2 genotype is associated with presence and progression of MRI depicted atherosclerotic intraplaque hemorrhage. IJC heart & vasculature. 2018;18:96-100.
- Costacou T, Levy AP. Haptoglobin genotype and its role in diabetic cardiovascular disease. Journal of cardiovascular translational research. 2012;5:423-35.
- Kessel I, Molad M, Nemirovsky R, Lavie-Nevo K, Levy AP, Blum S, et al. Haptoglobin Polymorphism and Morbidity in Children Born Prematurely. American Journal of Pediatrics. 2020;6:73-7.
- Asleh R, Blum S, Kalet-Litman S, Alshiek J, Miller-Lotan R, Asaf R, et al. Correction of HDL dysfunction in individuals with diabetes and the haptoglobin 2-2 genotype. Diabetes. 2008;57:2794-800.
- Mostaza JM, de Dios O, Lahoz C, Arribas M, Arroyo AP, Salinero-Fort MA, et al. Phenotype of haptoglobin and presence of subclinical vascular disease: Population study. Clínica e Investigación en Arteriosclerosis (English Edition). 2020;32:1-7. [CrossRef]
- Nakhoul FM, Miller-Lotan R, Awaad H, Asleh R, Levy AP. Hypothesis—haptoglobin genotype and diabetic nephropathy. Nature clinical practice Nephrology. 2007;3:339-44.
- Maffei M, Funicello M, Vottari T, Gamucci O, Costa M, Lisi S, et al. The obesity and inflammatory marker haptoglobin attracts monocytes via interaction with chemokine (CC motif) receptor 2 (CCR2). BMC biology. 2009;7:1-14. [CrossRef]
- Jin X, Qiu T, Li L, Yu R, Chen X, Li C, et al. Pathophysiology of obesity and its associated diseases. Acta Pharmaceutica Sinica B. 2023;13:2403-24.
- Okunogbe A, Nugent R, Spencer G, Ralston J, Wilding J. Economic impacts of overweight and obesity: current and future estimates for eight countries. BMJ global health. 2021;6:e006351.
- Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. The Lancet. 2007;370:1453-7.
- Weir CB, Jan A. BMI classification percentile and cut-off points. 2019.
- Madkour MI, Hassan RE, Sherif NM, Awadallah S, Abdelrahim DN, Jahrami HA, et al. Haptoglobin polymorphism modulates cardiometabolic impacts of four consecutive weeks, dawn to sunset Ramadan intermittent fasting among subjects with overweight/obesity. Diabetes Res Clin Pract. 2022;190:110024.
- Laemmli UK. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature. 1970;227:680-5.
- Boettger LM, Salem RM, Handsaker RE, Peloso GM, Kathiresan S, Hirschhorn JN, et al. Recurring exon deletions in the HP (haptoglobin) gene contribute to lower blood cholesterol levels. Nature genetics. 2016;48:359-66.
- Guthrie PA, Rodriguez S, Gaunt TR, Lawlor DA, Smith GD, Day IN. Complexity of a complex trait locus: HP, HPR, haemoglobin and cholesterol. Gene. 2012;499:8-13.
- Bjornsson E, Helgason H, Halldorsson G, Helgadottir A, Gylfason A, Kehr B, et al. A rare splice donor mutation in the haptoglobin gene associates with blood lipid levels and coronary artery disease. Human molecular genetics. 2017;26:2364-76.
- Wang S, Wang J, Zhang R, Wang T, Yan D, He Z, et al. Mendelian randomization analysis to assess a causal effect of haptoglobin on macroangiopathy in Chinese type 2 diabetes patients. Cardiovascular diabetology. 2018;17:14. [CrossRef]
- Froguel P, Ndiaye NC, Bonnefond A, Bouatia-Naji N, Dechaume A, Siest G, et al. A genome-wide association study identifies rs2000999 as a strong genetic determinant of circulating haptoglobin levels. PLoS One. 2012;7.
- Soejima M, Munkhtulga L, Furukawa K, Iwamoto S, Koda Y. Serum haptoglobin correlates positively with cholesterol and triglyceride concentrations in an obese Mongolian population. Clinica Chimica Acta. 2020. [CrossRef]
- Asleh R, Blum S, Kalet-Litman S, Alshiek J, Miller-Lotan R, Asaf R, et al. Correction of HDL Dysfunction in Individuals With Diabetes and the Haptoglobin 2-2 Genotype. Diabetes. 2008;57:2794-800.
- De Pergola G, Di Roma P, Paoli G, Guida P, Pannacciulli N, Giorgino R. Haptoglobin serum levels are independently associated with insulinemia in overweight and obese women. Journal of endocrinological investigation. 2007;30:399-403.
- Uchegbu E, Kopelman P. Cardiovascular risks in obesity. Journal of endocrinological investigation. 2002;25:915-8.
- Montani J, Antic V, Yang Z, Dulloo A. Pathways from obesity to hypertension: from the perspective of a vicious triangle. International journal of obesity. 2002;26:S28-S38.
- Engström G, Janzon L, Berglund Gr, Lind P, Stavenow L, Hedblad B, et al. Blood pressure increase and incidence of hypertension in relation to inflammation-sensitive plasma proteins. Arteriosclerosis, thrombosis, and vascular biology. 2002;22:2054-8. [CrossRef]
- Nakagawa T, Muramoto Y, Hori M, Mihara S, Marubayashi T, Nakagawa K. A preliminary investigation of the association between haptoglobin polymorphism, serum ferritin concentration and fatty liver disease. Clinica Chimica Acta. 2008;398:34-8.
- Alvarez-Blasco F, Martínez-García MÁ, Luque-Ramírez M, Parraza N, San Millan JL, Escobar-Morreale HF. Role of haptoglobin in polycystic ovary syndrome (PCOS), obesity and disorders of glucose tolerance in premenopausal women. PLoS One. 2009;4:e5606.
- Braeckman L, De Bacquer D, Delanghe J, Claeys L, De Backer G. Associations between haptoglobin polymorphism, lipids, lipoproteins, and inflammatory variables. Atherosclerosis. 1999;143:383-8.
- Chiellini C, Santini F, Marsili A, Berti P, Bertacca A, Pelosini C, et al. Serum haptoglobin: a novel marker of adiposity in humans. The Journal of Clinical Endocrinology & Metabolism. 2004;89:2678-83. [CrossRef]
- Rodrigues KF, Pietrani NT, Carvalho LML, Bosco AA, Sandrim VC, Ferreira CN, et al. Haptoglobin levels are influenced by Hp1–Hp2 polymorphism, obesity, inflammation, and hypertension in type 2 diabetes mellitus. Endocrinología, Diabetes y Nutrición (English ed). 2019;66:99-107.
- Doumatey AP, Lashley KS, Huang H, Zhou J, Chen G, Amoah A, et al. Relationships among obesity, inflammation, and insulin resistance in African Americans and West Africans. Obesity. 2010;18:598-603.
- Chiellini C, Bertacca A, Novelli SE, Görgün CZ, Ciccarone A, Giordano A, et al. Obesity modulates the expression of haptoglobin in the white adipose tissue via TNFα. Journal of cellular physiology. 2002;190:251-8. [CrossRef]
- Fain JN, Bahouth SW, Madan AK. Haptoglobin release by human adipose tissue in primary culture. Journal of lipid research. 2004;45:536-42.
- Lazalde B, Huerta-Guerrero HM, Simental-Mendía LE, Rodríguez-Morán M, Guerrero-Romero F. Haptoglobin 2-2 genotype is associated with TNF-α and IL-6 levels in subjects with obesity. Disease markers. 2014;2014.
- Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW. Obesity is associated with macrophage accumulation in adipose tissue. The Journal of Clinical Investigation. 2003;112:1796-808.
- Oliviero S, Cortese R. The human haptoglobin gene promoter: interleukin-6-responsive elements interact with a DNA-binding protein induced by interleukin-6. The EMBO journal. 1989;8:1145-51.
- MacKellar M, Vigerust DJ. Role of haptoglobin in health and disease: a focus on diabetes. Clinical Diabetes. 2016;34:148-57.
- Graves KL, Vigerust DJ. Hp: an inflammatory indicator in cardiovascular disease. Future cardiology. 2016;12:471-81.
- Mostaza JM, De Dios O, Lahoz C, Arribas M, Pérez Arroyo A, Salinero-Fort MA, et al. Phenotype of haptoglobin and presence of subclinical vascular disease: Population study. Clínica e Investigación en Arteriosclerosis (English Edition). 2020;32:1-7. [CrossRef]
- Liu J-J, Liu S, Wong MD, Gurung RL, Lim SC. Urinary haptoglobin predicts rapid renal function decline in Asians with type 2 diabetes and early kidney disease. The Journal of Clinical Endocrinology & Metabolism. 2016;101:3794-802. [CrossRef]
- Minović I, Eisenga MF, Riphagen IJ, van den Berg E, Kootstra-Ros J, Frenay A-RS, et al. Circulating haptoglobin and metabolic syndrome in renal transplant recipients. Scientific reports. 2017;7:1-9.
| Parameter | All participants (n=114) | Males (n=75) | Females (n=39) | P-value |
|---|---|---|---|---|
| Age (year) | 37.32± 11.8 | 38.77 ± 11.75 | 34.53 ± 11.64 | NS |
| Weight (kg) | 88.99 ± 15.51 | 92.96 ± 12.44 | 81.34 ± 17.98 | ** |
| BMI (kg/m2) | 30.41± 5.09 | 30.3 ± 3.9 | 30.5 ± 5.5 | NS |
| FM (kg) | 26.94 ± 9.46 | 25.61 ± 7.9 | 29.52 ± 11.56 | * |
| BFP (%) | 29.98 ± 7.02 | 27.01 ± 5.2 | 35.69 ± 6.50 | ** |
| FFM (kg) | 61.41 ± 10.52 | 67.09 ± 6.15 | 50.47 ± 8.29 | ** |
| MM (kg) | 58.34 ± 10.02 | 63.76 ± 5.87 | 47.92 ± 7.88 | ** |
| TBW (kg) | 43.82 ±7.42 | 47.79 ± 4.45 | 36.2 ± 5.88 | ** |
| VFA (cm2) | 97.85 ± 48.0 | 114.6 ± 43.71 | 65.64 ± 38.91 | ** |
| WC (cm) | 96.84 ± 13.48 | 99.68 ± 11.05 | 91.38 ± 16.0 | ** |
| HC (cm) | 108.0 ± 10.14 | 107.56 ± 8.13 | 109 ± 13.27 | NS |
| SBP (mmHg) | 123.43±11.65 | 125.8 ± 10.63 | 118.7 ± 12.18 | ** |
| DBP (mmHg) | 71.73 ± 9.57 | 74.23 ± 9.03 | 66.92 ± 8.82 | ** |
| Parameter | All participants (n=114) | Males (n=75) | Females (n=39) | P-value |
|---|---|---|---|---|
| FBG (mg/dl) | 97.97 ± 20.93 | 102.3 ± 22.3 | 89.5 ± 14.72 | ** |
| Fasting insulin (ng/ml) | 14.45 ± 14.17 | 15.85 ± 17.0 | 12.21 ± 7.64 | NS |
| HOMA-IR | 1.24 ± 1.08 | 1.40 ± 1.29 | 0.98 ± 0.54 | NS |
| Insulin sensitivity (1/HOMA-IR) | 2.26 ± 2.02 | 2.0 ± 1.62 | 2.68 ± 2.51 | NS |
| QUICKI Insulin sensitivity | 0.35 ± 0.06 | 0.34 ± 0.06 | 0.36 ± 0.06 | NS |
| IGF-1 (ng/ml) | 1.08 ± 0.79 | 1.18 ± 0.86 | 0.91 ± 0.66 | NS |
| Total cholesterol (mg/dl) | 180.6 ± 37.6 | 186.1 ± 39.3 | 169.9 ± 31.9 | * |
| TG (mg/dl) | 102.3 ± 60.4 | 114.8 ± 62.6 | 78.5 ± 48.3 | ** |
| LDL-C (mg/dl) | 115.1 ± 31.7 | 114.9 ± 32.6 | 115.2 ± 29.8 | NS |
| HDL-C (mg/dl) | 45.1 ± 7.73 | 45.3 ± 7.67 | 44.6 ± 7.93 | NS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




