Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Search and Study Selection
3. The Biological Pressure Toward Weight Regain
3.1. Neuroendocrine Regulation of Appetite in Weight Regain
4. Can Nutrition Counteract Appetite Dysregulation?
4.1. Dietary Protein and Satiety
4.2. Dietary Fiber and the Gut–Brain Axis
4.3. Glycemic Responses and Appetite Regulation
4.4. Dietary Patterns and Appetite Regulation
5. Clinical Evidence of Weight Regain
6. Clinical Implications for Preventing Weight Regain
6.1. Future Research Directions
7. Limitations
8. Conclusion
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Johnson, B.V.; Milstead, M.; Kreider, R.; Jones, R. Dietary supplement considerations during glucagon-like Peptide-1 receptor agonist treatment: A narrative review. Obes. Pillars 2025, 16. [CrossRef]
- Yao, H.; Zhang, A.; Li, D.; Wu, Y.; Wang, C.-Z.; Wan, J.-Y.; Yuan, C.-S. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ 2024, 384, e076410. [CrossRef]
- Hall, K.D.; Kahan, S. Maintenance of Lost Weight and Long-Term Management of Obesity. Med Clin. North Am. 2018, 102, 183–197. [CrossRef]
- Liu, J.; Jing, C.; Guo, Y.; Shang, Z.; Zhang, B.; Zhou, X.; Zhang, J.; Lian, G.; Tian, F.; Li, L.; et al. The central signaling pathways related to metabolism-regulating hormones of the gut-brain axis: a review. J. Transl. Med. 2025, 23, 1–18. [CrossRef]
- Melby, C.L.; Paris, H.L.; Foright, R.M.; Peth, J. Attenuating the Biologic Drive for Weight Regain Following Weight Loss: Must What Goes Down Always Go Back Up?. Nutrients 2017, 9, 468. [CrossRef]
- van Baak MA, Mariman ECM. Physiology of Weight Regain after Weight Loss: Latest Insights. Curr Obes Rep. 2025 Mar 31;14(1):28.
- Blüher, M. Understanding Adipose Tissue Dysfunction. J. Obes. Metab. Syndr. 2024, 33, 275–288. [CrossRef]
- Hinte, L.C.; Castellano-Castillo, D.; Ghosh, A.; Melrose, K.; Gasser, E.; Noé, F.; Massier, L.; Dong, H.; Sun, W.; Hoffmann, A.; et al. Adipose tissue retains an epigenetic memory of obesity after weight loss. Nature 2024, 636, 457–465. [CrossRef]
- Hall, K.D.; Guo, J. Obesity Energetics: Body Weight Regulation and the Effects of Diet Composition. Gastroenterology 2017, 152, 1718–1727.e3. [CrossRef]
- Rosenbaum, M.; Nicolson, M.; Hirsch, J.; Murphy, E.; Chu, F.; Leibel, R.L. Effects of Weight Change on Plasma Leptin Concentrations and Energy Expenditure1. J. Clin. Endocrinol. Metab. 1997, 82, 3647–3654. [CrossRef]
- Rosenbaum, M.; Murphy, E.M.; Heymsfield, S.B.; Matthews, D.E.; Leibel, R.L. Low Dose Leptin Administration Reverses Effects of Sustained Weight-Reduction on Energy Expenditure and Circulating Concentrations of Thyroid Hormones. J. Clin. Endocrinol. Metab. 2002, 87, 2391–2394. [CrossRef]
- Montez, J.M.; Soukas, A.; Asilmaz, E.; Fayzikhodjaeva, G.; Fantuzzi, G.; Friedman, J.M. Acute leptin deficiency, leptin resistance, and the physiologic response to leptin withdrawal. Proc. Natl. Acad. Sci. 2005, 102, 2537–2542. [CrossRef]
- Miller, G.D. Appetite Regulation: Hormones, Peptides, and Neurotransmitters and Their Role in Obesity. Am. J. Lifestyle Med. 2017, 13, 586–601. [CrossRef]
- Żurawski, T.; Bartosiewicz, A. Dietary and Behavioral Strategies for Weight Loss and Weight Loss Maintenance: A Narrative Review. Nutrients 2025, 18, 12. [CrossRef]
- MacLean, P.S.; Bergouignan, A.; Cornier, M.-A.; Jackman, M.R. Biology's response to dieting: the impetus for weight regain. Am. J. Physiol. Integr. Comp. Physiol. 2011, 301, R581–R600. [CrossRef]
- Flore, G.; Preti, A.; Carta, M.G.; Deledda, A.; Fosci, M.; Nardi, A.E.; Loviselli, A.; Velluzzi, F. Weight Maintenance after Dietary Weight Loss: Systematic Review and Meta-Analysis on the Effectiveness of Behavioural Intensive Intervention. Nutrients 2022, 14, 1259. [CrossRef]
- Courcoulas, A.P.; King, W.C.; Belle, S.H.; Berk, P.; Flum, D.R.; Garcia, L.; Gourash, W.; Horlick, M.; Mitchell, J.E.; Pomp, A.; et al. Seven-Year Weight Trajectories and Health Outcomes in the Longitudinal Assessment of Bariatric Surgery (LABS) Study. JAMA Surg. 2018, 153, 427–434. [CrossRef]
- Brissman, M.; Beamish, A.J.; Olbers, T.; Marcus, C. Prevalence of insufficient weight loss 5 years after Roux-en-Y gastric bypass: metabolic consequences and prediction estimates: a prospective registry study. BMJ Open 2021, 11, e046407. [CrossRef]
- Al-Ghabeesh, S.; Rayan, A.; Assaf, E.A.; Fawaz, M.; AlBay, A.; Alyami, H. Understanding weight regain: Insights from Saudi patients on factors influencing post-metabolic and bariatric surgery outcomes: A qualitative study. PLOS ONE 2026, 21, e0340120. [CrossRef]
- Akhlaghi, M. The role of dietary fibers in regulating appetite, an overview of mechanisms and weight consequences. Crit. Rev. Food Sci. Nutr. 2022, 64, 3139–3150. [CrossRef]
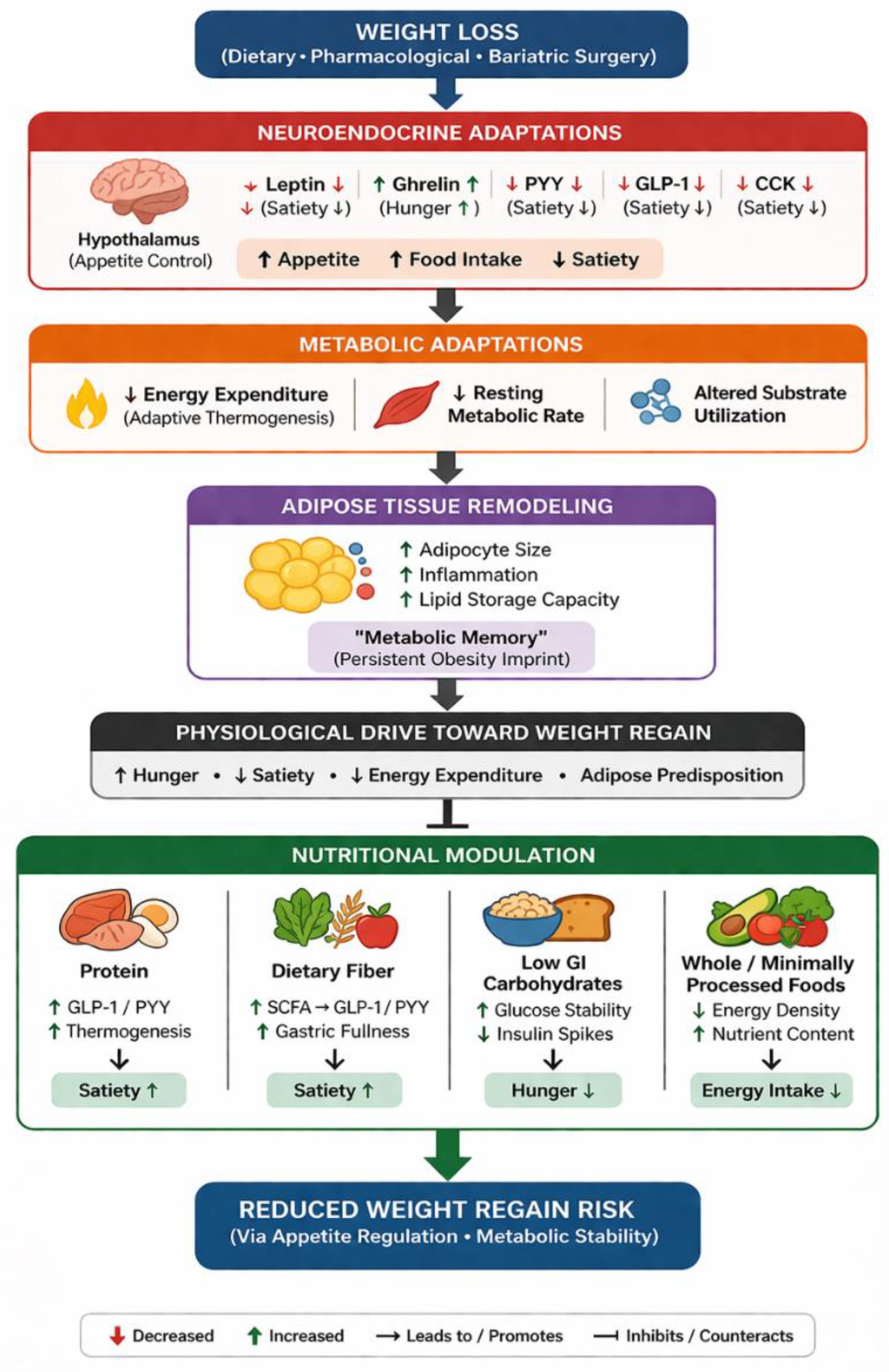
| Nutritional factor | Mechanism | Potential effect |
| Protein | Increased GLP-1 and PYY secretion; higher thermic effect | Increased satiety; reduced energy intake |
| Dietary fiber | SCFA production; modulation of gut microbiota; delayed gastric emptying | Enhanced gut–brain signaling; increased satiety |
| Low glycemic index carbohydrates | Stabilized postprandial glucose and insulin responses | Reduced hunger; improved appetite control |
| Whole / minimally processed foods | Lower energy density; higher nutrient quality | Reduced energy intake; improved weight maintenance |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




