Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
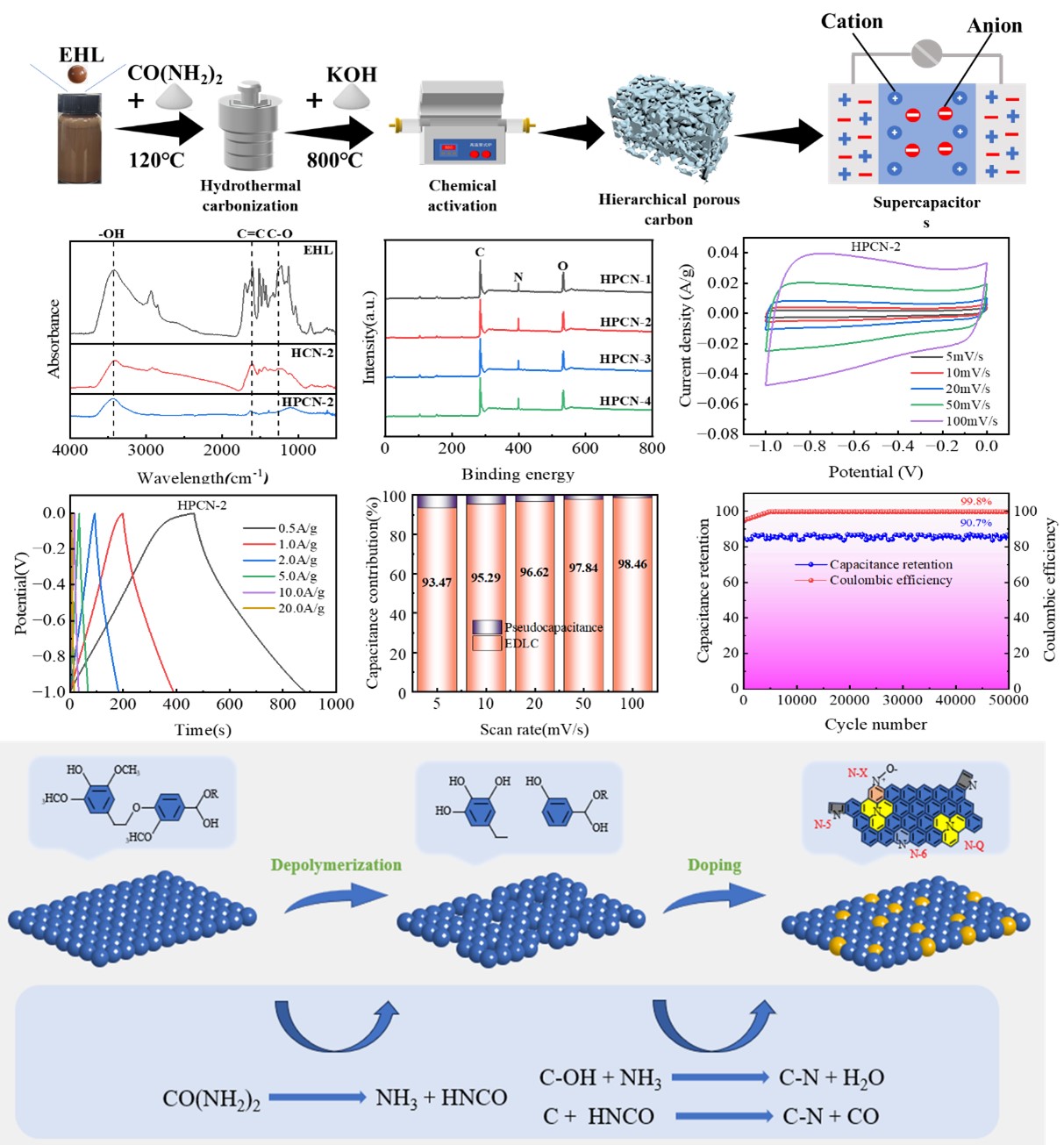
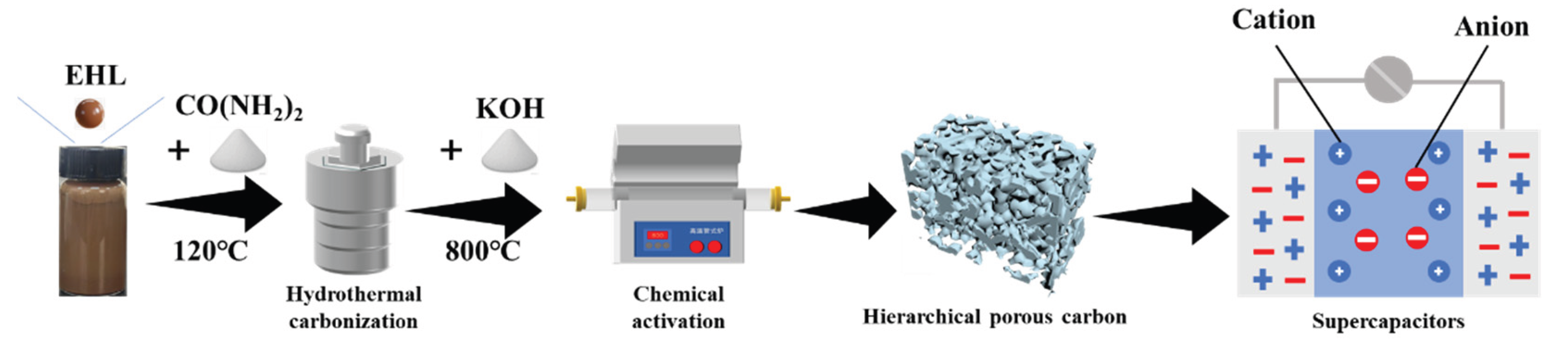
2.1. Preparation of Activated Carbon
2.2. Materials Characterizations
2.3. Electrochemical Measurements
3. Results and Discussion
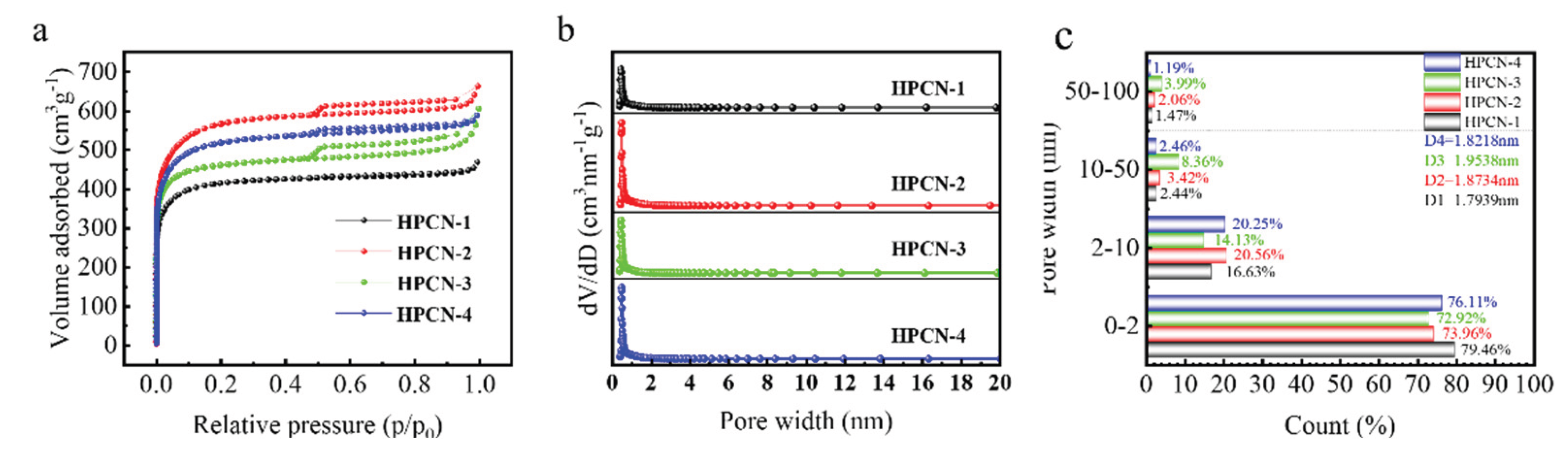
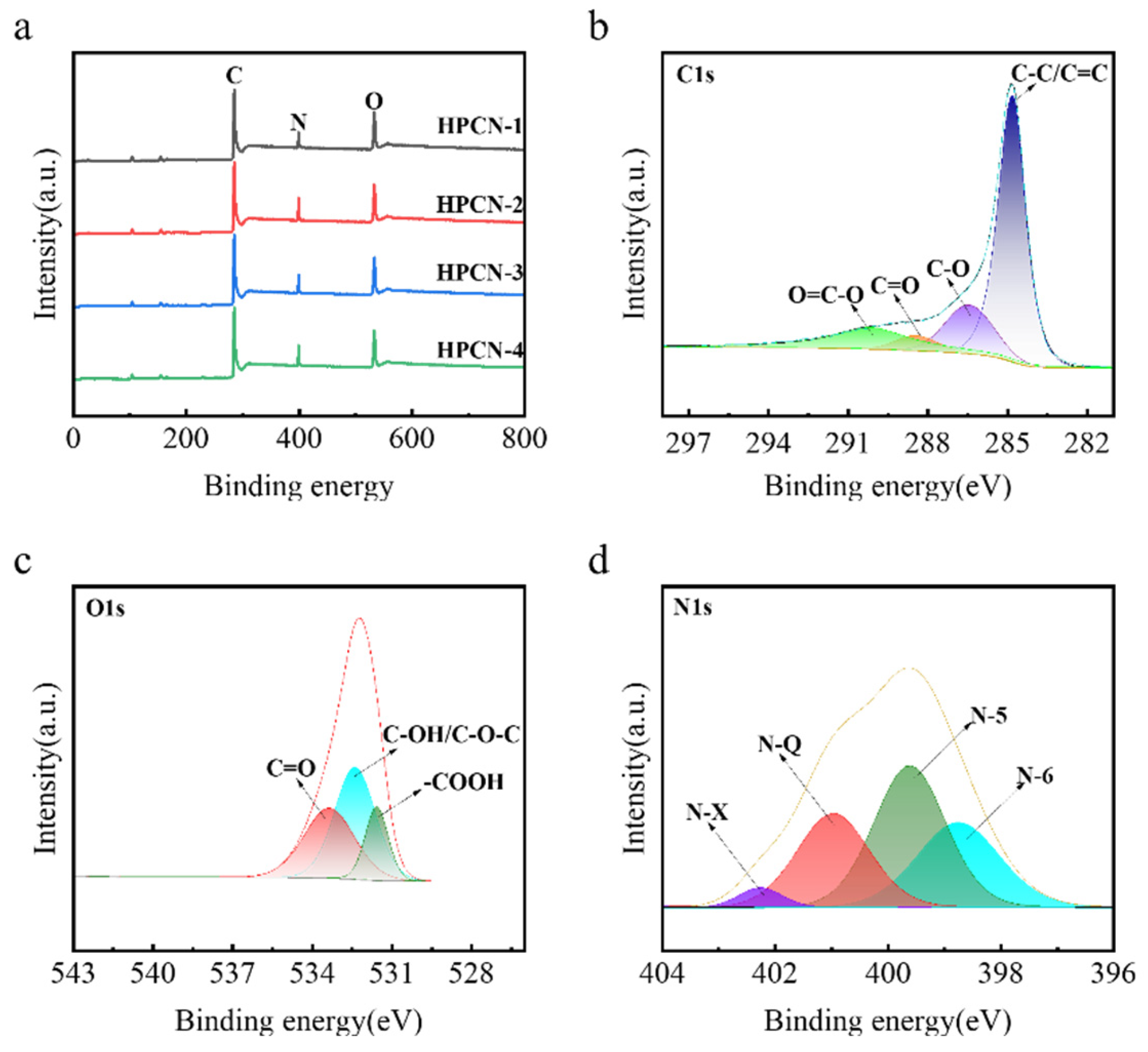
3.1. Morphology and Structure Properties of Hierarchical Porous Carbon
| HPCN Samples |
SBET (m2/g) |
Smicro (m2/g) |
Smeso (m2/g) |
Cmeso (%) |
Vtotal (cm3/g) |
Vmicro (cm3/g) |
Vmeso (cm3/g) |
Wmeso (%) |
dpore (nm) |
|---|---|---|---|---|---|---|---|---|---|
| HPCN-1 | 1823.87 | 1577.13 | 245.91 | 13.48 | 0.8183 | 0.6502 | 0.1561 | 19.08 | 1.7939 |
| HPCN-2 | 2569.45 | 2117.98 | 450.18 | 17.52 | 1.2062 | 0.8921 | 0.2893 | 23.98 | 1.8734 |
| HPCN-3 | 1990.89 | 1750.01 | 238.93 | 12.00 | 0.9690 | 0.7066 | 0.2237 | 23.06 | 1.9538 |
| HPCN-4 | 2294.64 | 1926.01 | 367.68 | 16.02 | 1.0456 | 0.7958 | 0.2374 | 22.70 | 1.8218 |
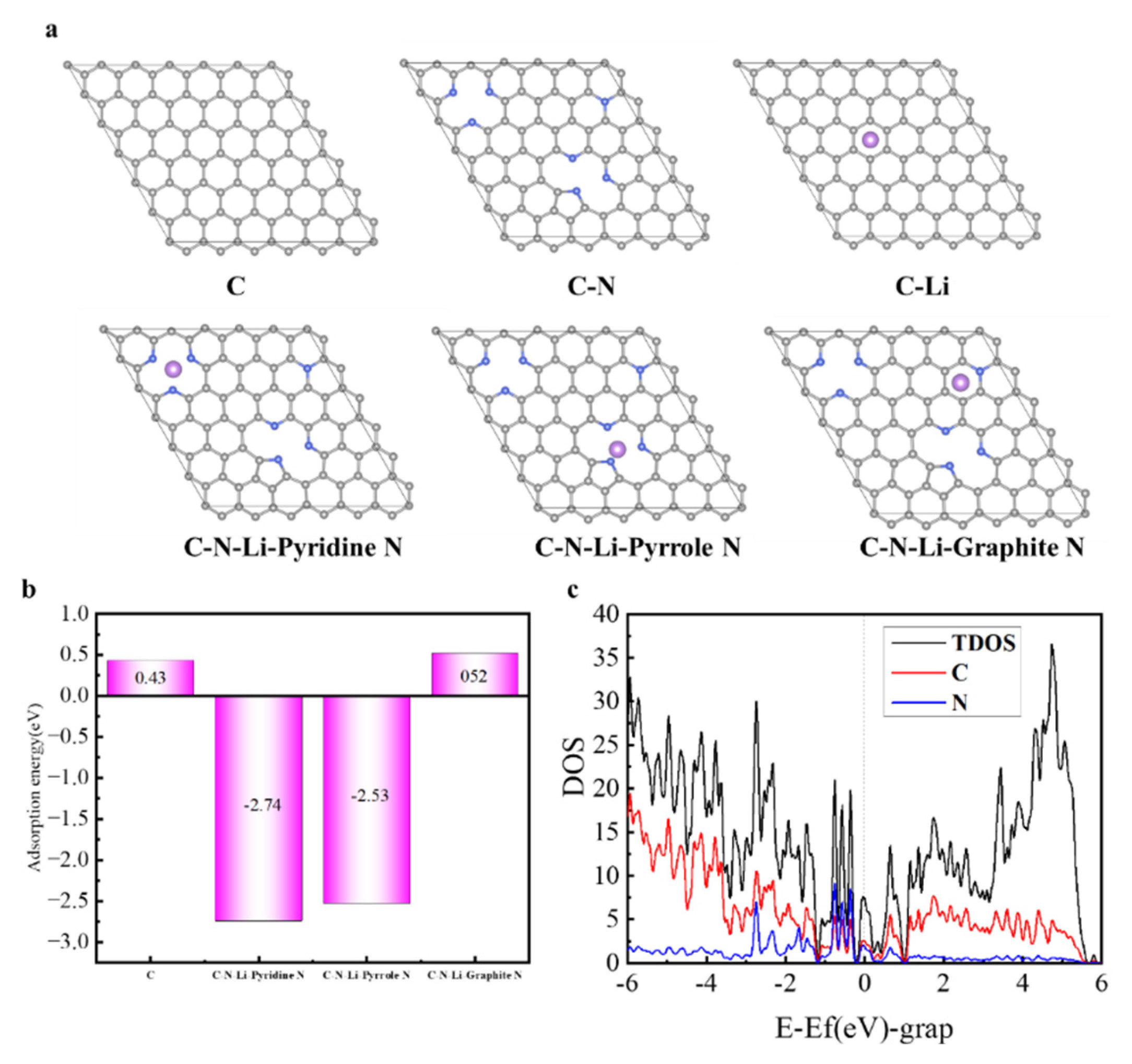
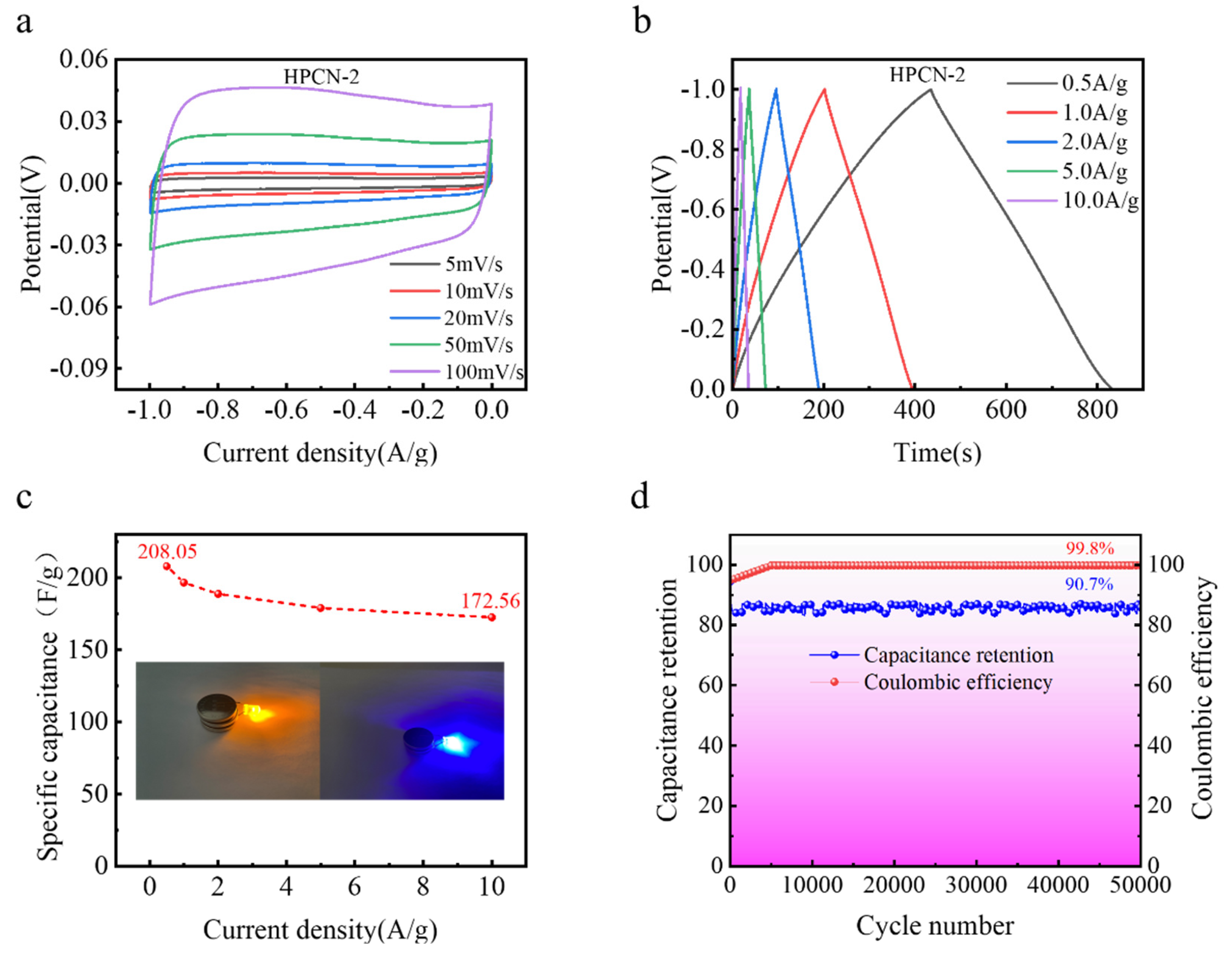
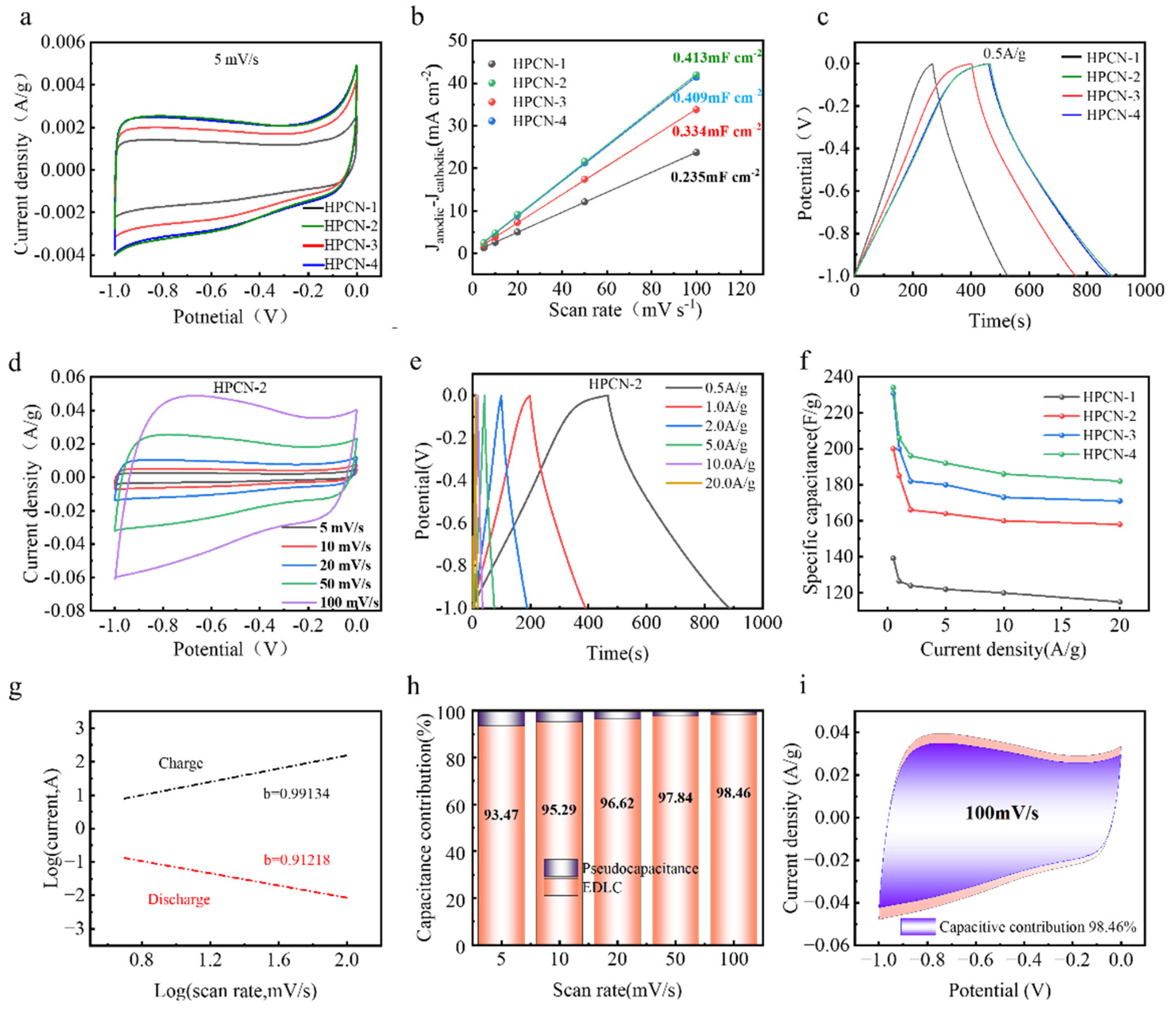
3.2. Electrochemicaltests
| Sample | Total N (at%) | N-6 (%) | N-5 (%) | N-Q (%) | Capacitance (F/g) | Retention (%) |
| HPCN-1 | 2.91 | 31.5 | 26.3 | 22.1 | 129 | 76.2 |
| HPCN-2 | 3.84 | 35.2 | 28.7 | 24.1 | 221 | 84.4 |
| HPCN-3 | 3.25 | 29.8 | 30.2 | 25.4 | 188 | 79.1 |
| HPCN-4 | 3.57 | 27.6 | 31.9 | 26.8 | 218 | 81.3 |
4. Conclusions
References
- Tong, Y; Yang, J; Li, J; et al. Lignin-derived electrode materials for supercapacitor applications: progress and perspectives[J]. Journal of Materials Chemistry A 2023, 11(3), 1061–1082. [Google Scholar] [CrossRef]
- Li, W; Zhang, W; Xu, Y; et al. Heteroatom-doped lignin derived carbon materials with improved electrochemical performance for advanced supercapacitors[J]. Chemical Engineering Journal 2024, 497, 154829. [Google Scholar] [CrossRef]
- Zhang, W; Yin, J; Wang, C; et al. Lignin derived porous carbons: synthesis methods and supercapacitor applications[J]. Small Methods 2021, 5(11), 2100896. [Google Scholar] [CrossRef]
- Rajasekaran, S J; Grace, A N; Jacob, G; et al. Investigation of different aqueous electrolytes for biomass-derived activated carbon-based supercapacitors[J]. Catalysts 2023, 13(2), 286. [Google Scholar] [CrossRef]
- Yi, M; Budarin, V; Yue, H; et al. Lignin-derived porous carbons via nitrogen-enhanced activation for efficient supercapacitors[J]. Journal of Colloid and Interface Science 2025, 138456. [Google Scholar] [CrossRef]
- Khalid, M; De, B S; Singh, A; et al. Dual action of lignin: electrode and electrolyte for sustainable supercapacitor application[J]. ACS Applied Energy Materials 2023, 6(15), 7857–7864. [Google Scholar] [CrossRef]
- Nargotra, P; Sharma, V; Wang, H M D; et al. Biocatalysis for lignin conversion and valorization: driving sustainability in the circular economy[J]. Catalysts 2025, 15(1), 91. [Google Scholar] [CrossRef]
- Zhao, X; Gao, P; Shen, B; et al. Recent advances in lignin-derived mesoporous carbon based-on template methods[J]. Renewable and Sustainable Energy Reviews 2023, 188, 113808. [Google Scholar] [CrossRef]
- Kolavada, H; Gajjar, P N; Gupta, S K. Unraveling quantum capacitance in supercapacitors: Energy storage applications[J]. Journal of Energy Storage 2024, 81, 110354. [Google Scholar] [CrossRef]
- Banerjee, S; Mordina, B; Sinha, P; et al. Recent advancement of supercapacitors: A current era of supercapacitor devices through the development of electrical double layer, pseudo and their hybrid supercapacitor electrodes[J]. Journal of Energy Storage 2025, 108, 115075. [Google Scholar] [CrossRef]
- Wu, X; Liu, R; Zhao, J; et al. Advanced carbon materials with different spatial dimensions for supercapacitors[J]. Nano Materials Science 2021, 3(3), 241–267. [Google Scholar] [CrossRef]
- Liu, H; Xu, T; Cai, C; et al. Multifunctional superelastic, superhydrophilic, and ultralight nanocellulose-based composite carbon aerogels for compressive supercapacitor and strain sensor[J]. Advanced Functional Materials 2022, 32(26), 2113082. [Google Scholar] [CrossRef]
- Kesavan, K S; Surya, K; Michael, M S. High powered hybrid supercapacitor with microporous activated carbon[J]. Solid State Ionics 2018, 321, 15–22. [Google Scholar] [CrossRef]
- Liu, W; Liu, K; Du, H; et al. Cellulose nanopaper: fabrication, functionalization, and applications[J]. Nano-Micro Letters 2022, 14(1), 104. [Google Scholar] [CrossRef]
- Zhong, M; Zhang, M; Li, X. Carbon nanomaterials and their composites for supercapacitors[J]. Carbon Energy 2022, 4(5), 950–985. [Google Scholar] [CrossRef]
- Jiang, C; Gao, M; Zhang, S; et al. Chitosan/graphene oxide hybrid hydrogel electrode with porous network boosting ultrahigh energy density flexible supercapacitor[J]. International Journal of Biological Macromolecules 2023, 225, 1437–1448. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W; Su, W; Li, P; et al. Preparation and research progress of lignin-based supercapacitor electrode materials[J]. International Journal of Biological Macromolecules 2024, 259, 128942. [Google Scholar] [CrossRef] [PubMed]
- Hao, R; Wu, Y; JIANG, N. Recent progress on the use of lignin-based porous carbon in supercapacitors[J]. New Carbon Materials 2025, 40(1), 50–80. [Google Scholar]
- Li, L; Wang, J; Chen, Z; et al. Preparation of sodium lignosulfonate-based porous carbon for supercapacitors with outstanding rate capacity and high voltage[J]. Chemical Engineering Journal 2025, 507, 160760. [Google Scholar] [CrossRef]
- Zhang, X; Liu, S; Zhao, Y; et al. Honeycomb-like hierarchical porous carbon from lignosulphonate by enzymatic hydrolysis and alkali activation for high-performance supercapacitors[J]. Energies 2023, 16(9), 3824. [Google Scholar] [CrossRef]
- Feng, P; Wang, H; Huang, P; et al. Nitrogen-doped lignin-derived porous carbons for supercapacitors: Effect of nanoporous structure[J]. Chemical Engineering Journal 2023, 471, 144817. [Google Scholar] [CrossRef]
- Tian, J; Liu, C; Lin, C; et al. Constructed nitrogen and sulfur codoped multilevel porous carbon from lignin for high-performance supercapacitors[J]. Journal of Alloys and Compounds 2019, 789, 435–442. [Google Scholar] [CrossRef]
- Liu, F; He, J; Liu, X; et al. MoC nanoclusters anchored Ni@ N-doped carbon nanotubes coated on carbon fiber as three-dimensional and multifunctional electrodes for flexible supercapacitor and self-heating device[J]. Carbon Energy 2021, 3(1), 129–141. [Google Scholar] [CrossRef]
- Du, L; Quan, B; Xu, Z; et al. Biomass-derived laser-induced graphene doped with nitrogen and sulfur for enhanced supercapacitor performance[J]. Carbon 2025, 238, 120225. [Google Scholar] [CrossRef]
- Wang, S; Feng, J; Pan, H. Facile preparation of nitrogen-doped hierarchical porous carbon derived from lignin with KCl for supercapacitors[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022, 651, 129622. [Google Scholar] [CrossRef]
- Sun, H; Rong, Y; Li, J; et al. Constructing hierarchical nanosheet frameworks of nitrogen-doped carbon for enhanced energy storage performance of supercapacitors[J]. Applied Surface Science 2025, 165041. [Google Scholar] [CrossRef]
- Jiang, Q; Cai, Y; Sang, X; et al. Nitrogen-doped carbon materials as supercapacitor electrodes: a mini review[J]. Energy & Fuels 2024, 38(12), 10542–10559. [Google Scholar]
- Ma, X; Deng, J; Zhang, R; et al. Hierarchical porous carbon nanoarchitectonics with honeycomb-like and N, P co-doped features for flexible symmetric supercapacitors and high-efficiency dye removal[J]. Journal of Energy Storage 2023, 65, 107272. [Google Scholar] [CrossRef]
- Ran, F; Yang, X; Xu, X; et al. Green activation of sustainable resources to synthesize nitrogen-doped oxygen-riched porous carbon nanosheets towards high-performance supercapacitor[J]. Chemical Engineering Journal 2021, 412, 128673. [Google Scholar] [CrossRef]
- Liu, A; Yan, L; Zhang, Y; et al. Nitrogen-doped coal-based microporous carbon material co-activated by HCOOK and urea for high performance supercapacitors[J]. Surfaces and Interfaces 2024, 44, 103754. [Google Scholar] [CrossRef]
- Tian, X L; Yu, J H; Qiu, L; et al. Structural changes and electrochemical properties of mesoporous activated carbon derived from Eucommia ulmoides wood tar by KOH activation for supercapacitor applications[J]. Industrial Crops and Products 2023, 197, 116628. [Google Scholar] [CrossRef]
- Feng, S; Fan, Q; Ouyang, Q; et al. Morphology and structure control of lignin-derived hierarchical porous carbon for high-performance supercapacitors[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2024, 685, 133292. [Google Scholar] [CrossRef]
- Rustamaji, H; Prakoso, T; Devianto, H; et al. Urea nitrogenated mesoporous activated carbon derived from oil palm empty fruit bunch for high-performance supercapacitor[J]. Journal of Energy Storage 2022, 52, 104724. [Google Scholar] [CrossRef]
- Wang, T; Guo, J; Guo, Y; et al. Nitrogen-doped carbon derived from deep eutectic solvent as a high-performance supercapacitor[J]. ACS Applied Energy Materials 2021, 4(3), 2190–2200. [Google Scholar] [CrossRef]
- Li, P; Yang, C; Yi, D; et al. Preparation of spherical porous carbon from lignin-derived phenolic resin and its application in supercapacitor electrodes[J]. International Journal of Biological Macromolecules 2023, 252, 126271. [Google Scholar] [CrossRef]
- Liu, G; Chen, X; Ahmed, S F; et al. Urea assisted melamine N/O co-doping Activated carbon for high performance energy storage materials[J]. Journal of Power Sources 2025, 640, 236652. [Google Scholar] [CrossRef]
- Zhang, D; He, Q; Wang, D; et al. Preparation of nitrogen-doped lignin-based porous carbon materials and their application in a supercapacitor[J]. Journal of Materials Science 2024, 59(40), 19161–19174. [Google Scholar] [CrossRef]
- Chen, J; Lin, Y; Liu, J; et al. Outstanding supercapacitor performance of nitrogen-doped activated carbon derived from shaddock peel[J]. Journal of Energy Storage 2021, 39, 102640. [Google Scholar] [CrossRef]
- Cao, L; Li, H; Xu, Z; et al. Comparison of the heteroatoms-doped biomass-derived carbon prepared by one-step nitrogen-containing activator for high performance supercapacitor[J]. Diamond and Related Materials 2021, 114, 108316. [Google Scholar] [CrossRef]
- Liu, X; Sun, B; Lin, L; et al. Urea-Tris doped 3D graphene for high-stability supercapacitors[J]. Electrochemistry Communications 2025, 108004. [Google Scholar] [CrossRef]
- Yin, L; Hu, P; Liang, C; et al. Construction of self-supporting ultra-micropores lignin-based carbon nanofibers with high areal desalination capacity[J]. International Journal of Biological Macromolecules 2023, 225, 1415–1425. [Google Scholar] [CrossRef]
- Khamnantha, P; Homla-or, C; Suttisintong, K; et al. Stable lignin-rich nanofibers for binder-free carbon electrodes in supercapacitors[J]. ACS Applied Nano Materials 2021, 4(12), 13099–13111. [Google Scholar] [CrossRef]
- Wang, B; Xu, J; Lu, F; et al. Efficient utilization of lignin heterogeneity: Employing a simple approach to modulate the structure-effect relationship of lignin microspheres for its high-quality applications in supercapacitors[J]. Industrial Crops and Products 2024, 221, 119360. [Google Scholar] [CrossRef]
- Li, W; Wang, G; Sui, W; et al. Facile and scalable preparation of cage-like mesoporous carbon from lignin-based phenolic resin and its application in supercapacitor electrodes[J]. Carbon 2022, 196, 819–827. [Google Scholar] [CrossRef]
- Li, W; Li, C; Xu, Y; et al. Heteroatom-doped and graphitization-enhanced lignin-derived hierarchically porous carbon via facile assembly of lignin-Fe coordination for high-voltage symmetric supercapacitors[J]. Journal of Colloid and Interface Science 2024, 659, 374–384. [Google Scholar] [CrossRef]
- Wang, L; Feng, X; Li, X; et al. Hydrothermal, KOH-assisted synthesis of lignin-derived porous carbon for supercapacitors: value-added of lignin and constructing texture properties/specific capacitance relationships[J]. Journal of Materials Research and Technology 2022, 16, 570–580. [Google Scholar] [CrossRef]
- Srinivasan, S B; Devendiran, S; Savunthari, K V; et al. Insights into multifarious heteroatom-doped/enriched carbon-based materials and their composites: Synthesis and Supercapacitor applications− A crucial review[J]. Progress in Materials Science 2025, 153, 101470. [Google Scholar] [CrossRef]
- Cai, X; Zhang, K; Chen, W; et al. Deciphering the capacitive behavior of heteroatom-doped carbon materials with small mesopores[J]. Energy Storage Materials 2025, 104584. [Google Scholar] [CrossRef]
- Lei, T; Wang, Y; Wang, H; et al. Sustainable self-doping engineering: Transforming pulping black liquor into hierarchical sulfur-doped porous carbon architectures for advanced supercapacitor electrodes[J]. Journal of Electroanalytical Chemistry 2025, 119448. [Google Scholar] [CrossRef]
- Lei, T; Gai, X; Wang, H; et al. Petaloid-shaped hierarchical porous carbon sub-microspheres derived from lignin for high-performance supercapacitor electrodes[J]. Journal of Power Sources 2025, 658, 238304. [Google Scholar] [CrossRef]

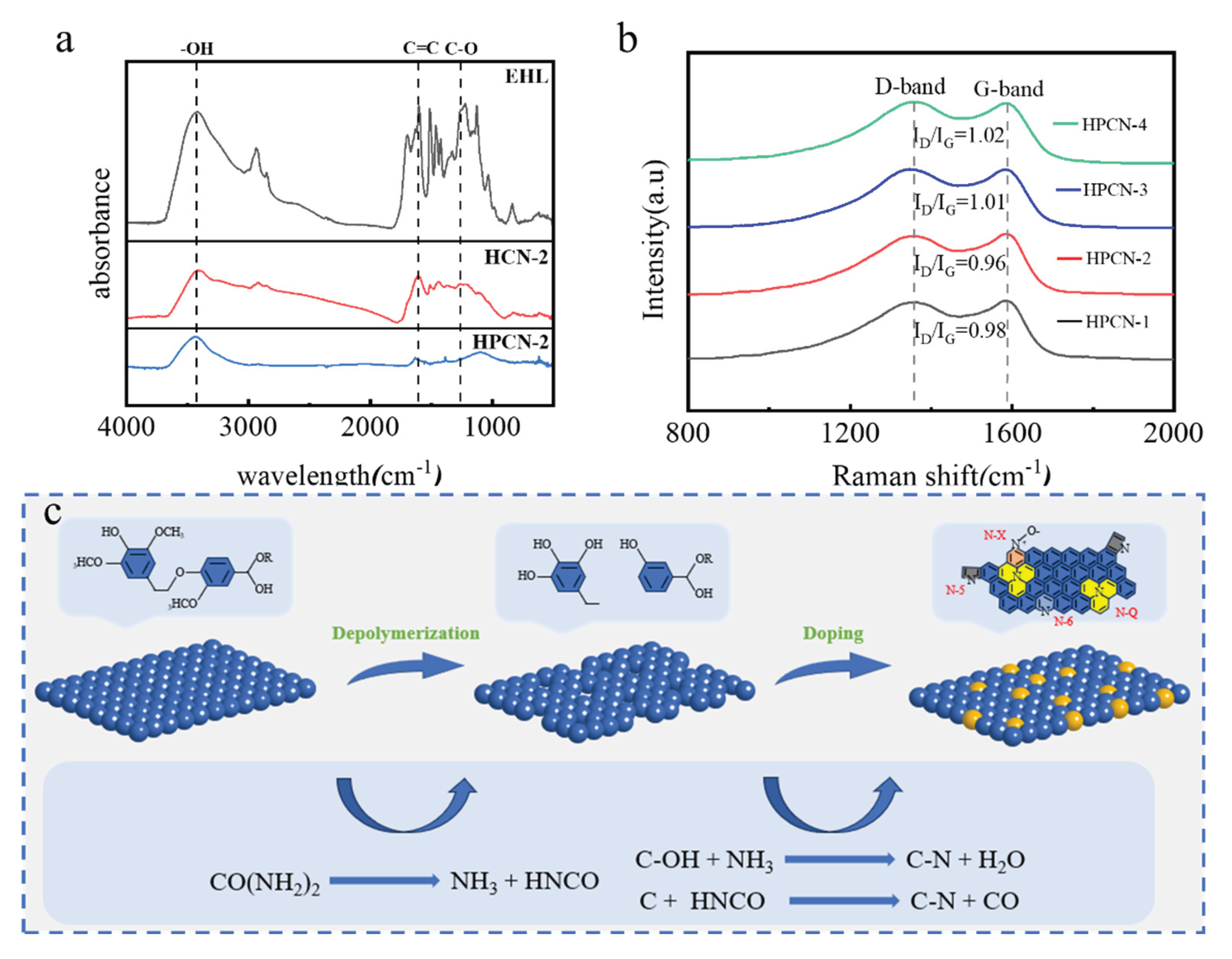
| Sample | N (%) | C (%) | H (%) | S (%) | O (%) | |
|---|---|---|---|---|---|---|
| CHNS mode | EHL | 0.605 | 58.849 | 3.755 | 0 | 32.953 |
| O mode | EHL | 0.576 | 58.414 | 3.752 | 0 | 32.801 |
| Sample | Moisture Mad (%) |
Ash A (%) |
Volatile matter V (%) |
Fixed carbon FCad (%) |
|||
|---|---|---|---|---|---|---|---|
| Aad | Ad | Vad | Vd | Vdaf | |||
| EHL | 6.00 | 2.89 | 3.07 | 60.06 | 63.89 | 65.91 | 31.05 |
| Sample | Electrolytes |
Test condition |
Specific capacitance | References |
| KL-8 | 6M KOH | 0.25A/g | 88F/g | [6] |
| ACNFs | 1M H2SO4 | 0.1 A/g | 182 F/g | [42] |
| LMSC-L1 | 6M KOH | 1A/g | 200F/g | [43] |
| LPRAC-20% | 6M KOH | 0.5A/g | 217.3F/g | [44] |
| Fe-NLC | 6M KOH | 0.5A/g | 264 F/g | [45] |
| LPC-150 | 6M KOH | 0.5A/g | 214.03 F/g | [46] |
| HPCN-2 | 6M KOH | 0.5A/g | 221F/g | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




