Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
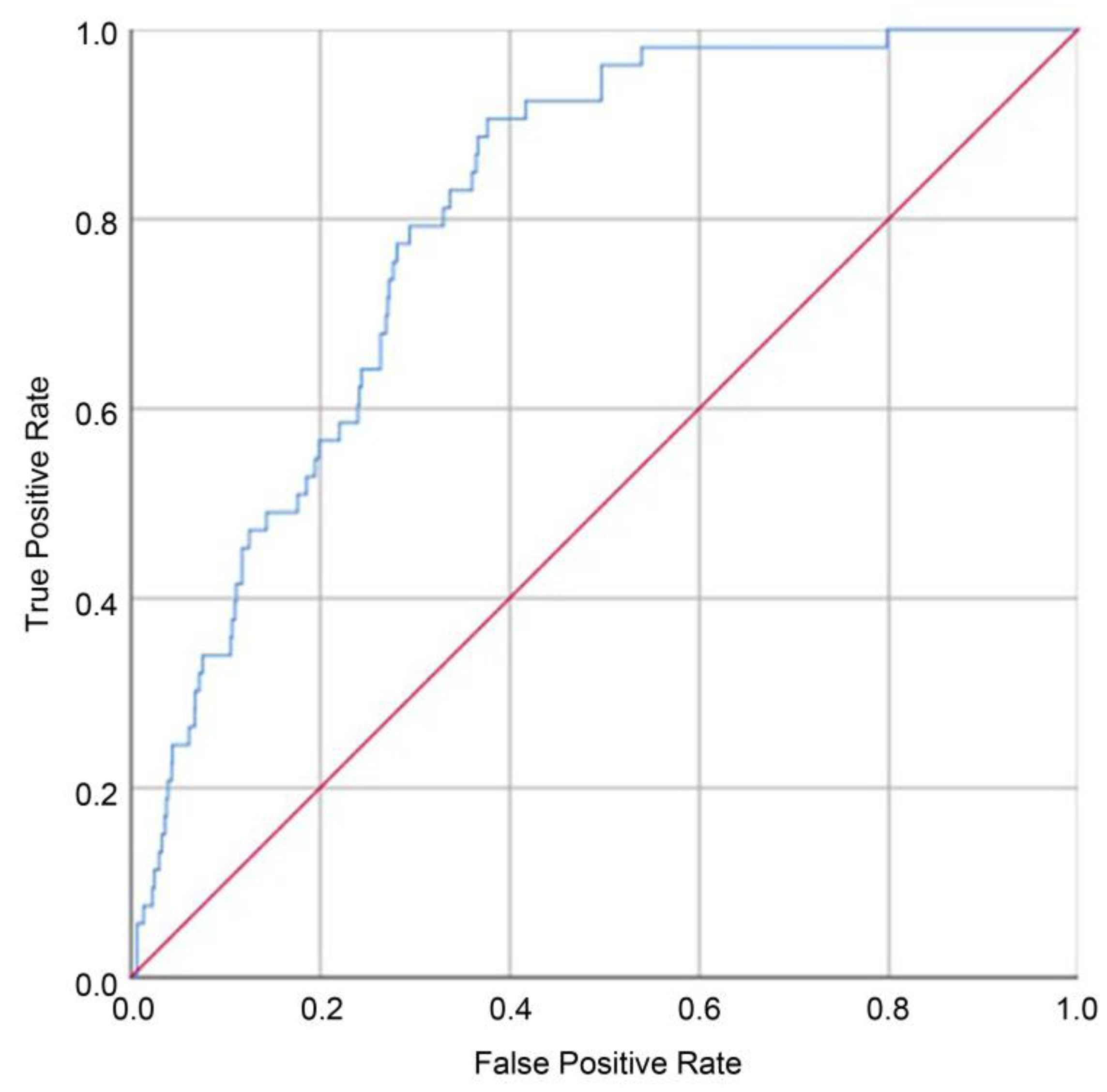
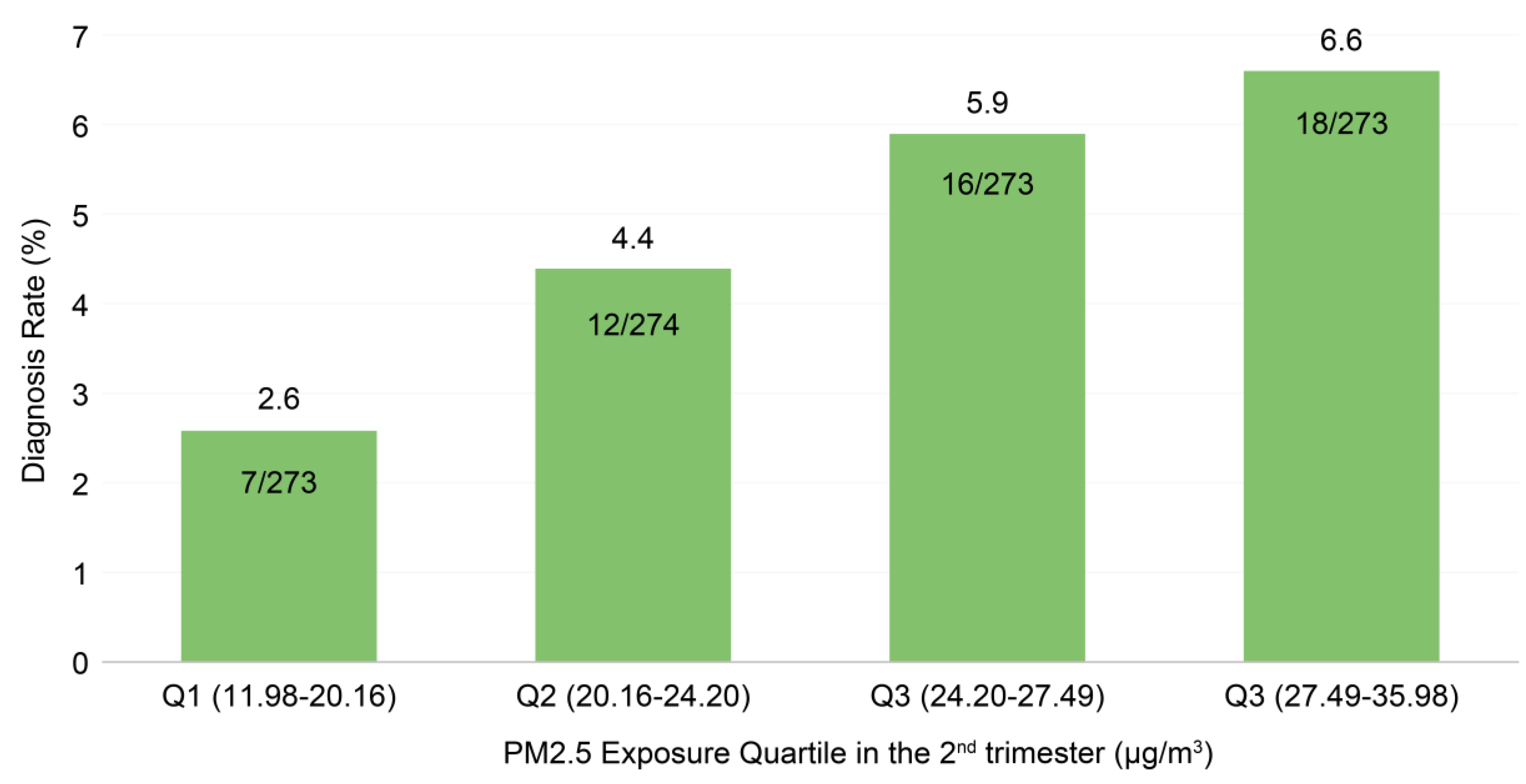
Background/Objectives: Inguinal hernia and hydrocele are common pediatric surgical conditions resulting from failed obliteration of the processus vaginalis during fetal development. Although prenatal exposure to fine particulate matter (PM2.5) has been linked to adverse perinatal outcomes and congenital anomalies, its role in structurally defined pediatric surgical diseases remains unclear. We examined the association between maternal PM2.5 exposure during pregnancy and the risk of inguinal hernia or hydrocele in offspring. Methods: We performed a retrospective cohort study of 1,093 mother–offspring pairs delivering at a tertiary referral center (July 2016–June 2019). Monthly residential PM2.5 levels were estimated at geocoded maternal addresses using kriging interpolation from fixed-site monitoring stations. Offspring diagnosed with inguinal hernia or hydrocele through March 2024 were identified using ICD-10 codes. Perinatal characteristics were compared using t-tests and chi-square tests, and multivariable logistic regression assessed trimester-specific PM2.5 exposure and risk. Results: During follow-up, 53 offspring (4.85%) developed inguinal hernia or hydrocele. Male sex (odds ratio [OR], 24.71; 95%CI, 5.95–102.54; p<0.001) and second-trimester PM2.5 exposure (OR, 1.07 per µg/m³; 95%CI, 1.01–1.14; p=0.028) were independent risk factors. A dose–response pattern was observed across quartiles of second-trimester exposure; interquartile range increase was associated with a 64% higher risk (OR, 1.64). The model showed good discrimination (AUC, 0.804). Conclusions: Elevated maternal PM2.5 exposure during the second trimester was independently associated with increased risk of inguinal hernia or hydrocele in offspring. Prenatal air pollution may contribute to persistence of the processus vaginalis and represents a potentially modifiable environmental risk factor.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
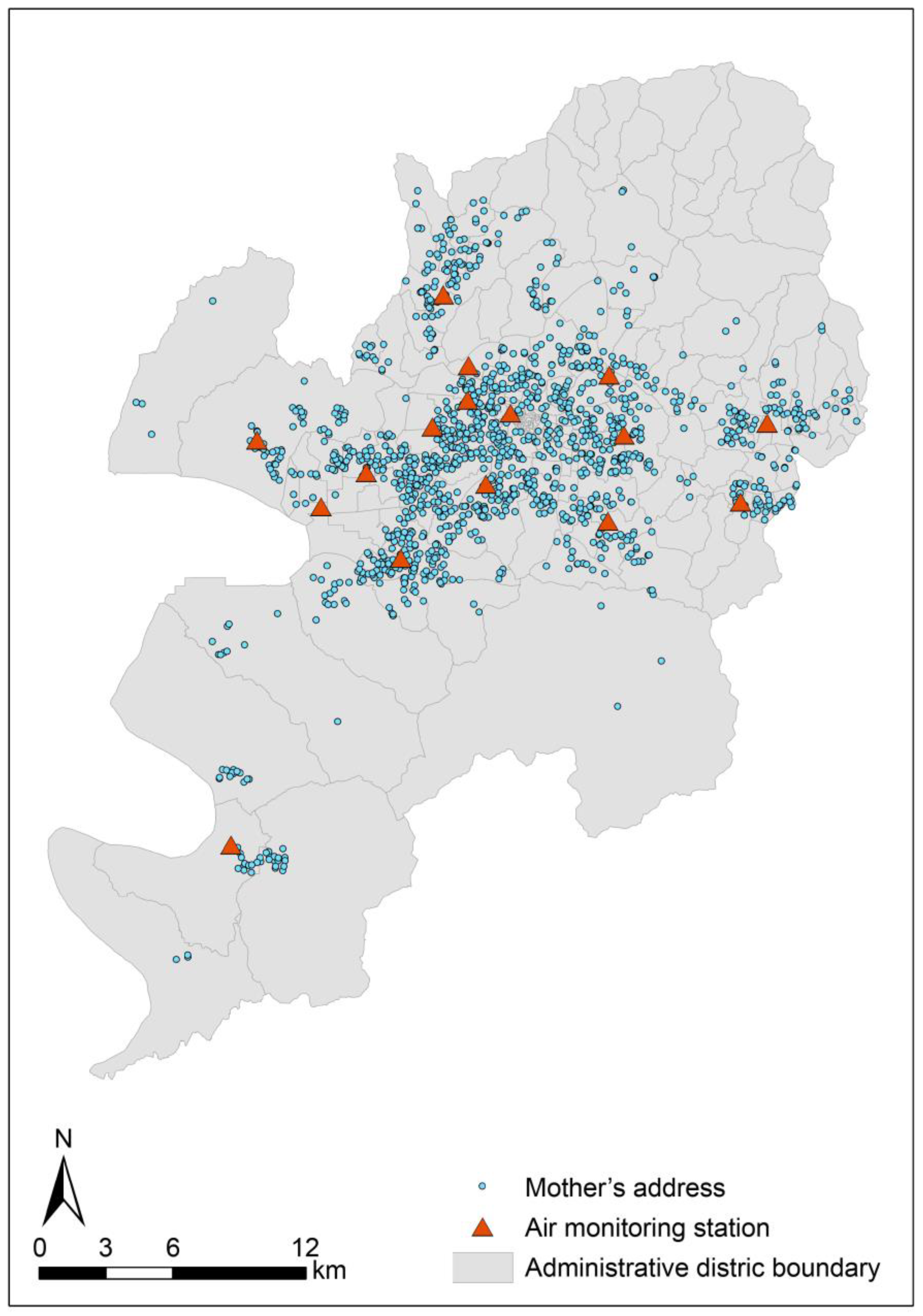
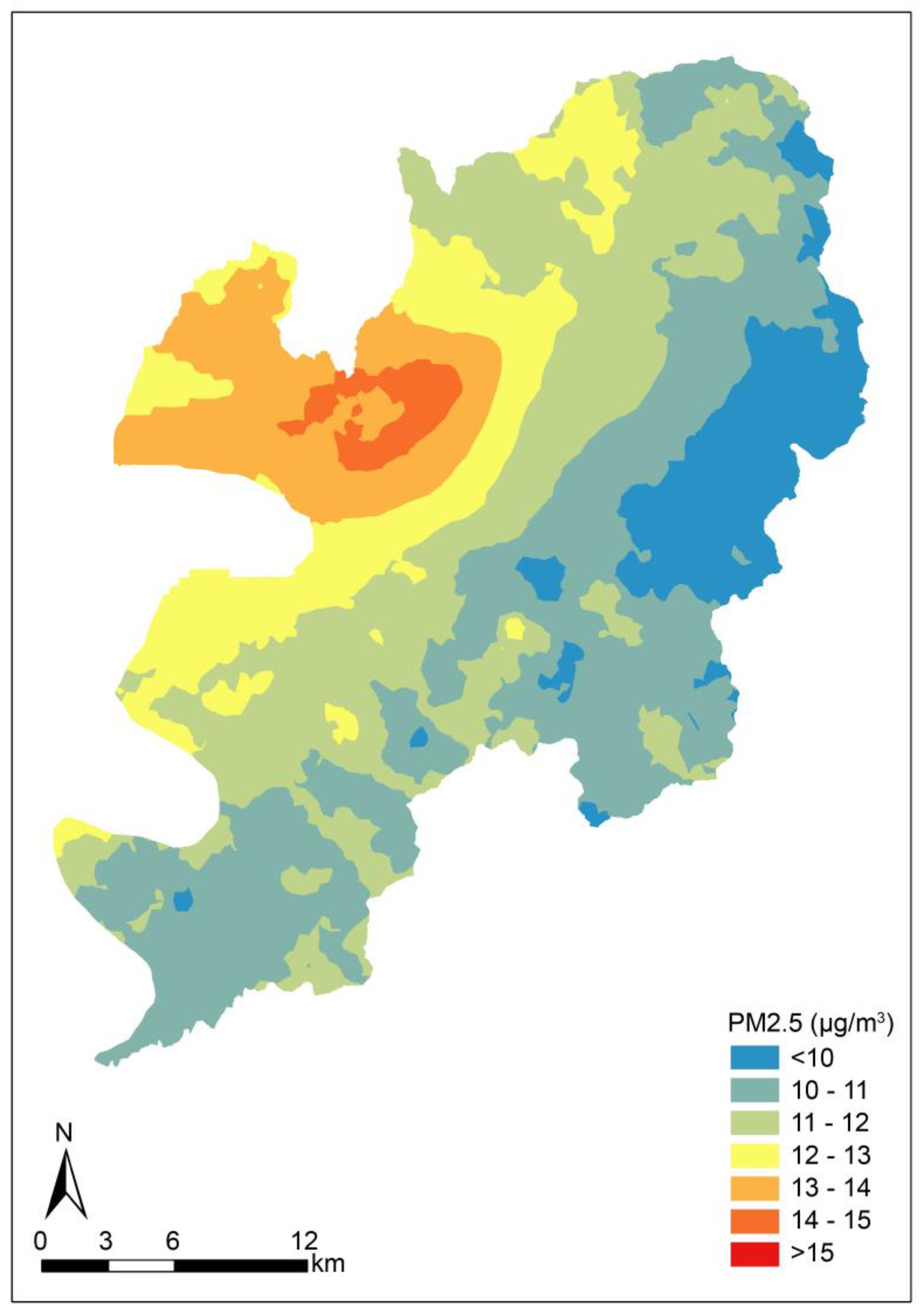
2.2. Exposure Assessment
2.3. Outcome Definition
- Inguinal hernia: K40.00–K40.90
- Hydrocele: N43.00–N43.30, P83.5
2.4. Covariates and Statistical Analysis
2.5. Ethics Statement
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | Area Under the Curve |
| ICD-10 | International Classification of Diseases, 10th Revision |
| IQR | Interquartile Range |
| IRB | Institutional Review Board |
| PM2.5 | Fine particulate matter ≤2.5 μm in diameter |
| PPV | Persistent Processus Vaginalis |
| ROC | Receiver Operating Characteristic |
References
- Svenningsson, A.; Hernias, W.T. Pediatric Surgery: Diagnosis and Management; Puri, P., Höllwarth, M.E., Eds.; Springer International Publishing: Cham, 2023; pp. 1001–1014. [Google Scholar]
- Raitio, A.; Kalliokoski, N.; Syvänen, J.; Harju, S.; Tauriainen, A.; Hyvärinen, A.; Gissler, M.; Helenius, I.; Sankilampi, U. High incidence of inguinal hernias among patients with congenital abdominal wall defects: a population-based case–control study. Eur J Pediatr 2021, 180, 2693–2698. [Google Scholar] [CrossRef] [PubMed]
- Wolf, L.L.; Sonderman, K.A.; Kwon, N.K.; Armstrong, L.B.; Weil, B.R.; Koehlmoos, T.P.; Losina, E.; Ricca, R.L.; Weldon, C.B.; Haider, A.H.; et al. Epidemiology of abdominal wall and groin hernia repairs in children. Pediatr Surg Int 2021, 37, 587–595. [Google Scholar] [CrossRef]
- Dolgin, S. Inguinal hernia in girls: a retrospective analysis of over 1000 patients. J Pediatr Surg 2021, 56, 203. [Google Scholar] [CrossRef]
- Chang, S.J.; Chen, J.Y.; Hsu, C.K.; Chuang, F.C.; Yang, S.S. The incidence of inguinal hernia and associated risk factors of incarceration in pediatric inguinal hernia: a nation-wide longitudinal population-based study. Hernia 2016, 20, 559–563. [Google Scholar] [CrossRef]
- Salari, N.; Beiromvand, M.; Abdollahi, R.; Hemmatabadi, F.K.; Daneshkhah, A.; Ghaderi, A.; Asgari, M.; Mohammadi, M. Global prevalence of hydrocele in infants and children: a systematic review and meta-analysis. BMC Pediatr 2025, 25, 128. [Google Scholar] [CrossRef]
- Osifo, O.D.; Osaigbovo, E.O. Congenital hydrocele: prevalence and outcome among male children who underwent neonatal circumcision in Benin City, Nigeria. J Pediatr Urol 2008, 4, 178–182. [Google Scholar] [CrossRef]
- Brainwood, M.; Beirne, G.; Fenech, M. Persistence of the processus vaginalis and its related disorders. Australas J Ultrasound Med 2020, 23, 22–29. [Google Scholar] [CrossRef]
- Palmer, L.S. Hernias and hydroceles. Pediatr Rev 2013, 34, 457–64; quiz 464. [Google Scholar] [CrossRef]
- Alexeeff, S.E.; Liao, N.S.; Liu, X.; Van Den Eeden, S.K.; Sidney, S. Long-term PM2.5 exposure and risks of ischemic heart disease and stroke events: review and meta-analysis. J Am Heart Assoc 2021, 10, e016890. [Google Scholar] [CrossRef] [PubMed]
- Thangavel, P.; Park, D.; Lee, Y.C. Recent insights into particulate matter (PM2.5)-mediated toxicity in humans: an overview. Int J Environ Res Public Health 2022, 19, 7507. [Google Scholar] [CrossRef] [PubMed]
- Jiao, A.; Reilly, A.N.; Benmarhnia, T.; Sun, Y.; Avila, C.; Chiu, V.; Slezak, J.; Sacks, D.A.; Molitor, J.; Li, M.; et al. Fine particulate matter, its constituents, and spontaneous preterm birth. JAMA Netw Open 2024, 7, e2444593. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, P.; Zhou, Y.; Xia, B.; Zhu, Q.; Ge, W.; Li, J.; Shi, H.; Xiao, X.; Zhang, Y. Prenatal fine particulate matter exposure, placental DNA methylation changes, and fetal growth. Environ Int 2021, 147, 106313. [Google Scholar] [CrossRef] [PubMed]
- Zhen, S.; Li, N.; Li, Y.; Li, Q.; Zheng, L.; Yin, Z.; Dou, Y.; Wu, S.; Liu, Y.; Zhang, X.; et al. Maternal exposure to fine particulate matter before and during pregnancy, and the risk of birth defects: a population-based study. Ecotoxicol Environ Saf 2025, 289, 117408. [Google Scholar] [CrossRef] [PubMed]
- Koo, E.J.; Bae, J.G.; Kim, E.J.; Cho, Y.H. Correlation between exposure to fine particulate matter (PM2.5) during pregnancy and congenital anomalies: its surgical perspectives. J Korean Med Sci 2021, 36, e236. [Google Scholar] [CrossRef]
- Jung, E.M.; Kim, K.N.; Park, H.; Shin, H.H.; Kim, H.S.; Cho, S.J.; Kim, S.T.; Ha, E.H. Association between prenatal exposure to PM2.5 and the increased risk of specified infant mortality in South Korea. Environ Int 2020, 144, 105997. [Google Scholar] [CrossRef]
- Michikawa, T.; Morokuma, S.; Fukushima, K.; Kato, K.; Nitta, H.; Yamazaki, S. Maternal exposure to air pollutants during the first trimester and foetal growth in Japanese term infants. Environ Pollut 2017, 230, 387–393. [Google Scholar] [CrossRef]
- Ren, S.; Haynes, E.; Hall, E.; Hossain, M.; Chen, A.; Muglia, L.; Lu, L.; DeFranco, E. Periconception exposure to air pollution and risk of congenital malformations. J Pediatr 2018, 193, 76–84.e6. [Google Scholar] [CrossRef]
- Morillo, M.C.; Martínez-Cuevas, S.; García-Aranda, C.; Molina, I.; Querol, J.J.; Martínez, E. Spatial analysis of the particulate matter (PM10) an assessment of air pollution in the region of Madrid (Spain): spatial interpolation comparisons and results. Int J Environ Stud 2024, 81, 1501–1511. [Google Scholar] [CrossRef]
- Masroor, K.; Fanaei, F.; Yousefi, S.; Raeesi, M.; Abbaslou, H.; Shahsavani, A.; Hadei, M. Spatial modelling of PM2.5 concentrations in Tehran using kriging and inverse distance weighting (IDW) methods. J Air Pollut Health 2020, 5, 115–124. [Google Scholar] [CrossRef]
- Nguyen, D.A.; Duong, S.H.; Tran, P.A.; Cao, H.H.; Ho, B.Q. Combination of data-driven models and interpolation technique to develop PM10 map for Hanoi, Vietnam. Sci Rep 2020, 10, 19268. [Google Scholar] [CrossRef]
- Canadian Association of Paediatric Surgeons. Inguinal hernias and hydroceles in infancy and childhood: a consensus statement of the Canadian Association of Paediatric Surgeons. Paediatr Child Health 2000, 5, 461–462. [Google Scholar] [CrossRef]
- Krishnan, N.; Kakkar, A.; Nag, T.C.; Agarwala, S.; Goel, P.; Dhua, A.K. A comparative study of smooth muscle cell characteristics and myofibroblasts in processus vaginalis of pediatric inguinal hernia, hydrocele and undescended testis. BMC Urol 2024, 24, 115. [Google Scholar] [CrossRef] [PubMed]
- Hutson, J.M.; Southwell, B.R.; Li, R.; Lie, G.; Ismail, K.; Harisis, G.; Chen, N. The regulation of testicular descent and the effects of cryptorchidism. Endocr Rev 2013, 34, 725–752. [Google Scholar] [CrossRef]
- Favorito, L.A.; Costa, W.S.; Sampaio, F.J. Relationship between the persistence of the processus vaginalis and age in patients with cryptorchidism. Int Braz J Urol 2005, 31, 57–61. [Google Scholar] [CrossRef]
- Fussell, J.C.; Jauniaux, E.; Smith, R.B.; Burton, G.J. Ambient air pollution and adverse birth outcomes: a review of underlying mechanisms. BJOG 2024, 131, 538–550. [Google Scholar] [CrossRef]
- Parasin, N.; Amnuaylojaroen, T.; Saokaew, S. Prenatal PM2.5 exposure and its association with low birth weight: a systematic review and meta-analysis. Toxics 2024, 12, 446. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Oliver, B.G.; Pant, A.; Olivera, A.; Poronnik, P.; Pollock, C.A.; Saad, S. Particulate matter, an intrauterine toxin affecting foetal development and beyond. Antioxidants (Basel) 2021, 10, 732. [Google Scholar] [CrossRef] [PubMed]
- Kilcoyne, K.R.; Mitchell, R.T. Effect of environmental and pharmaceutical exposures on fetal testis development and function: a systematic review of human experimental data. Hum Reprod Update 2019, 25, 397–421. [Google Scholar] [CrossRef]
- Plunk, E.C.; Richards, S.M. Endocrine-disrupting air pollutants and their effects on the hypothalamus–pituitary–gonadal axis. Int J Mol Sci 2020, 21, 8902. [Google Scholar] [CrossRef]
- Gaudriault, P.; Mazaud-Guittot, S.; Lavoué, V.; Coiffec, I.; Lesné, L.; Dejucq-Rainsford, N.; Scholze, M.; Kortenkamp, A.; Jégou, B. Endocrine disruption in human fetal testis explants by individual and combined exposures to selected pharmaceuticals, pesticides, and environmental pollutants. Environ Health Perspect 2017, 125, 087004. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.L.; Yu, C.J.; Han, R.; Chen, L.; Liu, M.L.; Sun, M.; Zeng, Z.Y.; Wang, Q.; Xu, X.M.; et al. Associations between maternal periconceptional exposure to PM2.5 and the risk of cryptorchidism: a case-control study of 4274. Sci Rep 2024, 14, 29703. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.S.; Toft, G.; Thulstrup, A.M.; Bonde, J.P.; Olsen, J. Cryptorchidism according to maternal gestational smoking. Epidemiology 2007, 18, 220–225. [Google Scholar] [CrossRef]
- Gurney, J.K.; McGlynn, K.A.; Stanley, J.; Merriman, T.; Signal, V.; Shaw, C.; Edwards, R.; Richiardi, L.; Hutson, J.; Sarfati, D. Risk factors for cryptorchidism. Nat Rev Urol 2017, 14, 534–548. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.W.; Pan, M.L.; Hsu, Y.J.; Chin, T.W. A nationwide survey of incidence rates and risk factors of inguinal hernia in preterm children. Pediatr Surg Int 2018, 34, 91–95. [Google Scholar] [CrossRef] [PubMed]
| Study population | Characteristic | Values |
| Mother (n=1,093) | Age (years) | 34.1 ± 4.5 (18–46) |
| Diabetes mellitus, n (%) | 118 (10.8) | |
| Hypertension, n (%) | 75 (6.9) | |
| Smoking history, n (%) | 6 (0.5) | |
| Offspring (n=1,093) | Gestational age (weeks) | 38.6 ± 1.0 (37–42) |
| Sex, male: female, n (%) | 574:519 (52.5:47.5) | |
| Birth weight (g) | 3,181 ± 377 (2,060–4,610) | |
| Apgar score (1 min) | 7.8 ± 0.5 | |
| Apgar score (5 min) | 8.9 ± 0.3 |
| Variable | No hernia/hydrocele (N=1,040) | Hernia/hydrocele (N=53) | p-value |
| Sex, male: female, n (%) | 523:517 (50.3:49.7) | 51:2 (96.2:3.8) | <0.001 |
| Gestational age (weeks) | 38.6 ± 1.0 | 38.5 ± 0.9 | 0.352 |
| Birth weight (g) | 3,176 ± 377 | 3,296 ± 375 | 0.023 |
| Apgar score at 1 min | 7.8 ± 0.5 | 7.8 ± 0.4 | 0.362 |
| Apgar score at 5 min | 8.9 ± 0.3 | 8.9 ± 0.3 | 0.857 |
| Maternal age (years) | 34.0 ± 4.5 | 34.8 ± 4.4 | 0.222 |
| Maternal diabetes mellitus, n (%) | 112 (10.8) | 6 (11.3) | 0.822 |
| Maternal hypertension, n (%) | 73 (7.0) | 2 (3.8) | 0.518 |
| Maternal smoking, n (%) | 6 (0.6) | 0 (0.0) | 1.000 |
| Trimester | No hernia/hydrocele (mean ± SD) | Hernia/hydrocele (mean ± SD) | p-value |
| 1st trimester | 24.9 ± 4.3 | 24.0 ± 4.9 | 0.151 |
| 2nd trimester | 23.9 ± 4.9 | 25.4 ± 4.6 | 0.032 |
| 3rd trimester | 24.0 ± 5.3 | 24.9 ± 5.0 | 0.265 |
| Variable | Odds ratio (OR) | 95% CI | p-value |
| Sex of offspring (male) | 24.71 | 5.95–102.54 | <0.001 |
| Birth weight (g) | 1.001 | 1.000–1.001 | 0.121 |
| PM2.5 in 2nd trimester (µg/m³) | 1.070 | 1.01–1.14 | 0.028 |
| Gestational age (weeks) | 0.783 | 0.57–1.09 | 0.143 |
| Maternal age (years) | 1.036 | 0.97–1.11 | 0.301 |
| Maternal diabetes mellitus | 0.781 | 0.31–1.96 | 0.599 |
| Maternal hypertension | 2.389 | 0.57–1.09 | 0.143 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




