Submitted:
24 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
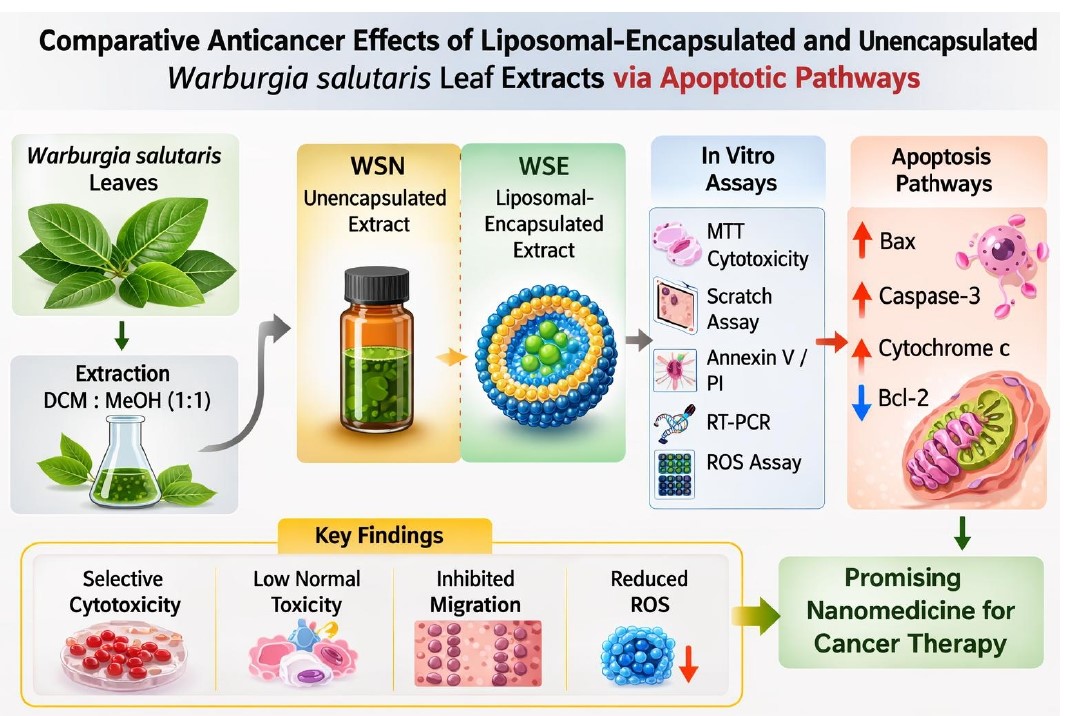
1. Introduction
2. Results
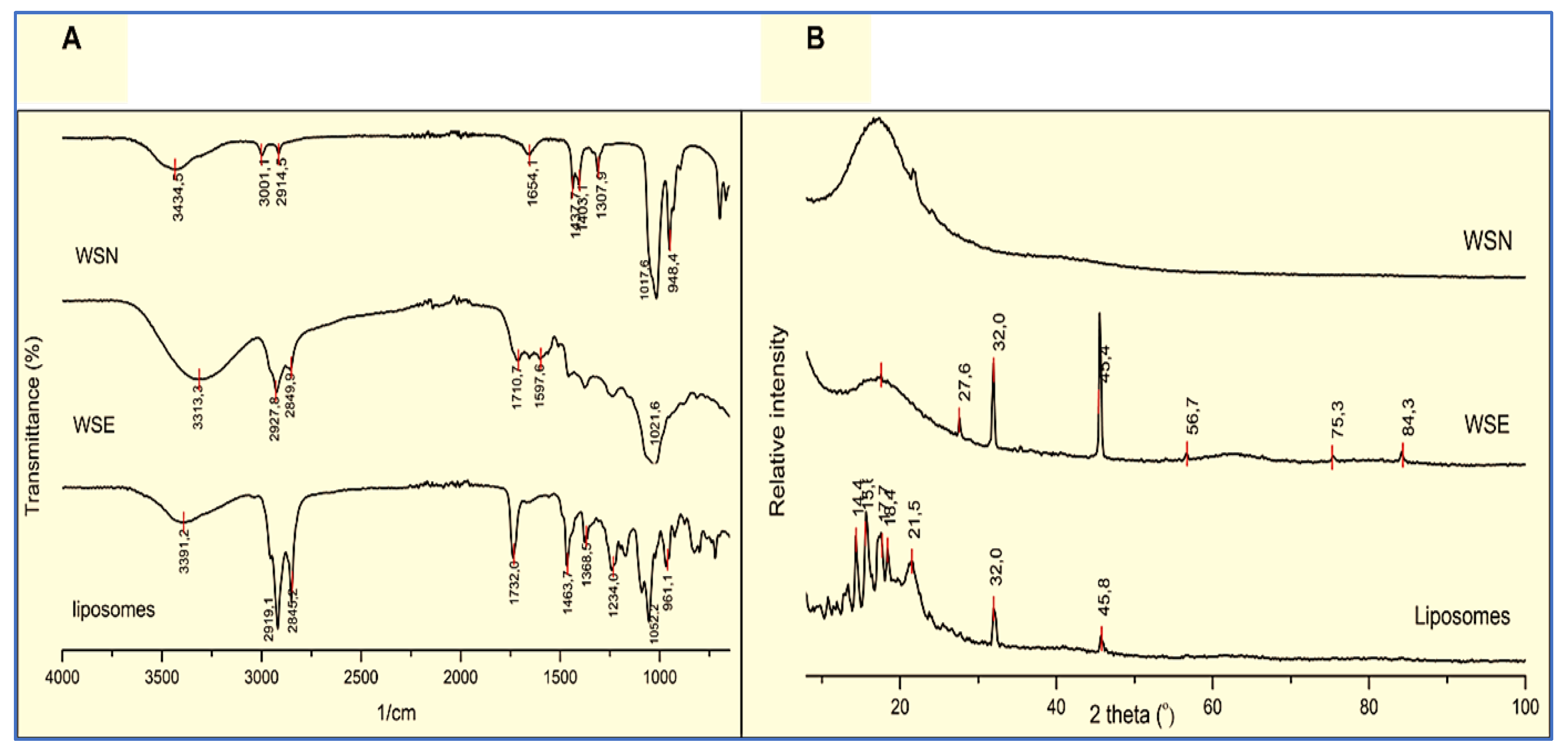
2.1. Phytochemical Characteristics Analysed by FTIR
2.2. Powder X-Ray Diffraction (PXRD)
2.3. Particle Size and Zeta Potential
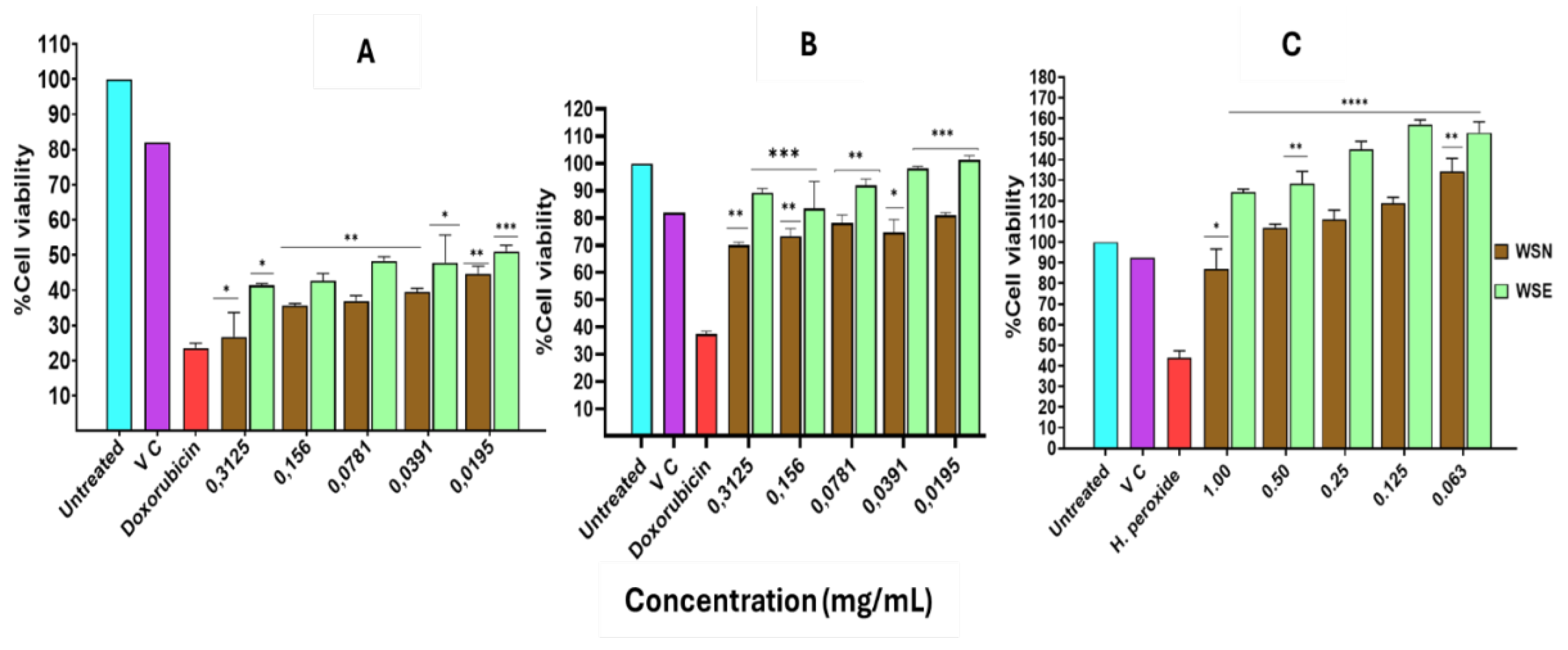
2.4. MTT Assay
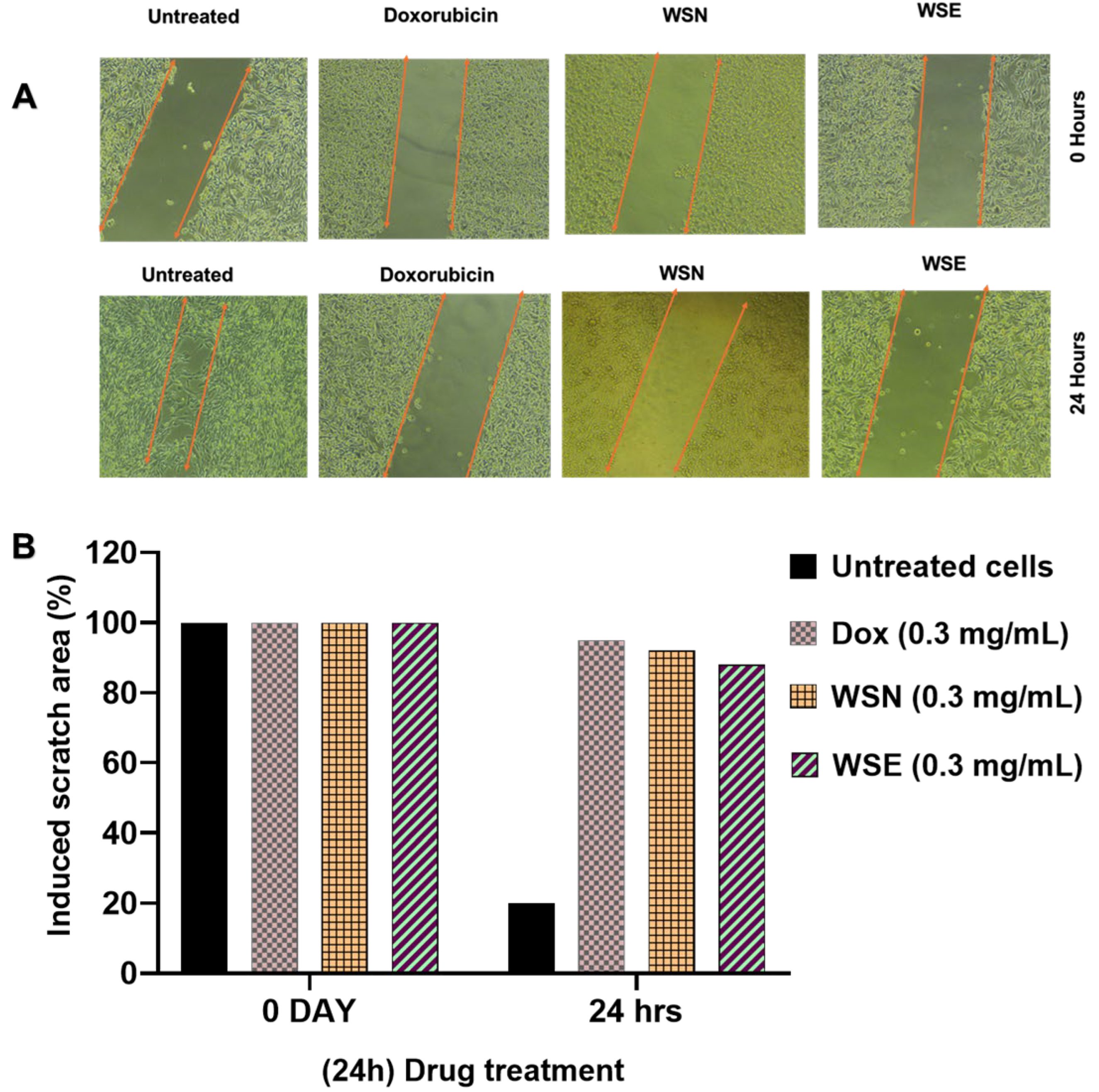
2.5. Anti-Metastatic Effect on MCF-7 Cells Treated with WSN and WSE Leaf Extracts
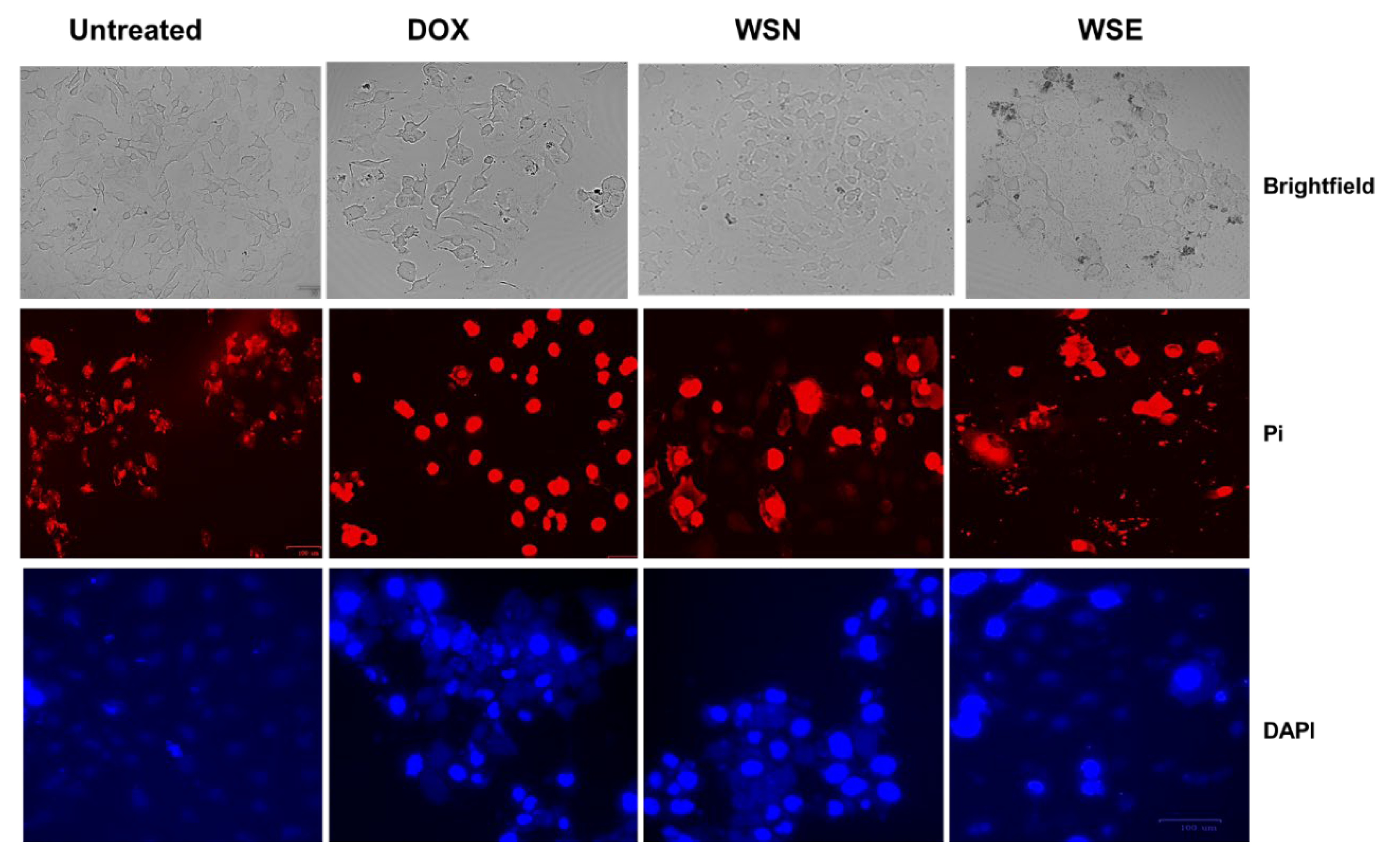
2.6. Determination of W. salutaris Induced Apoptotic Features Using DAPI/PI Staining
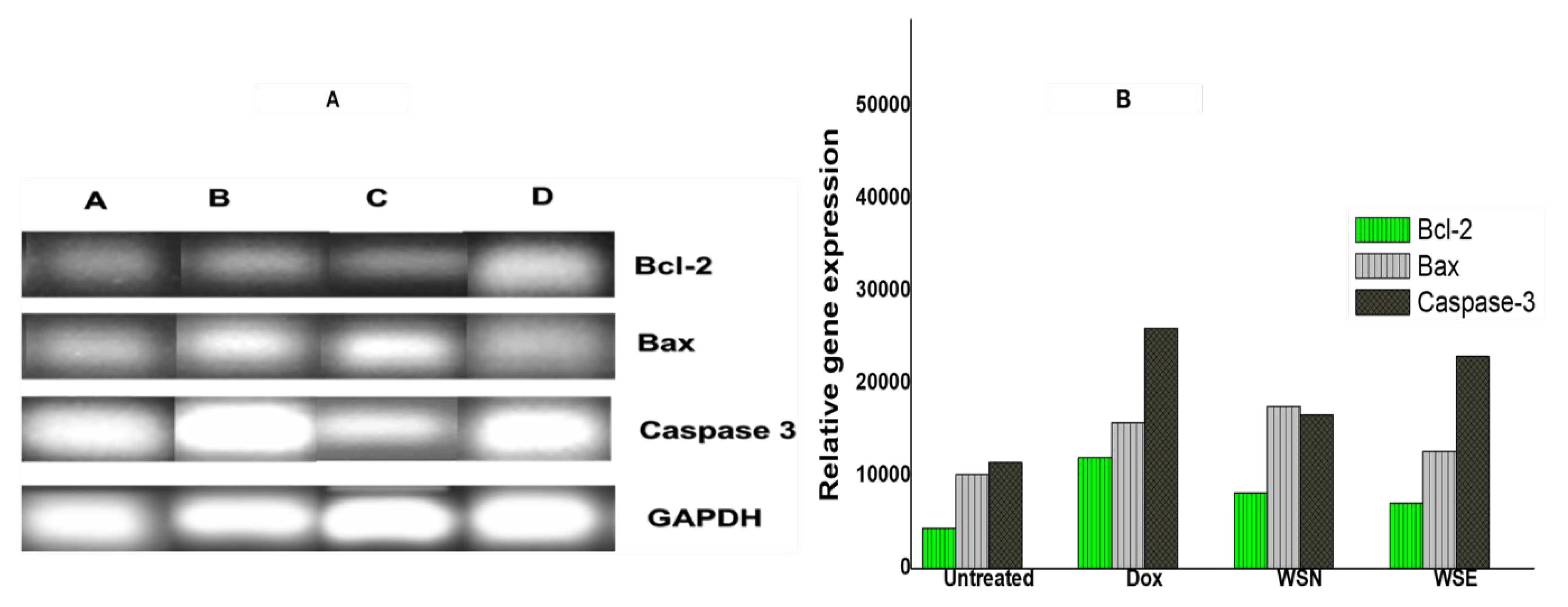
2.7. Apoptotic Gene Expression Levels in MCF-7 Cells Analysed by RT-PCR
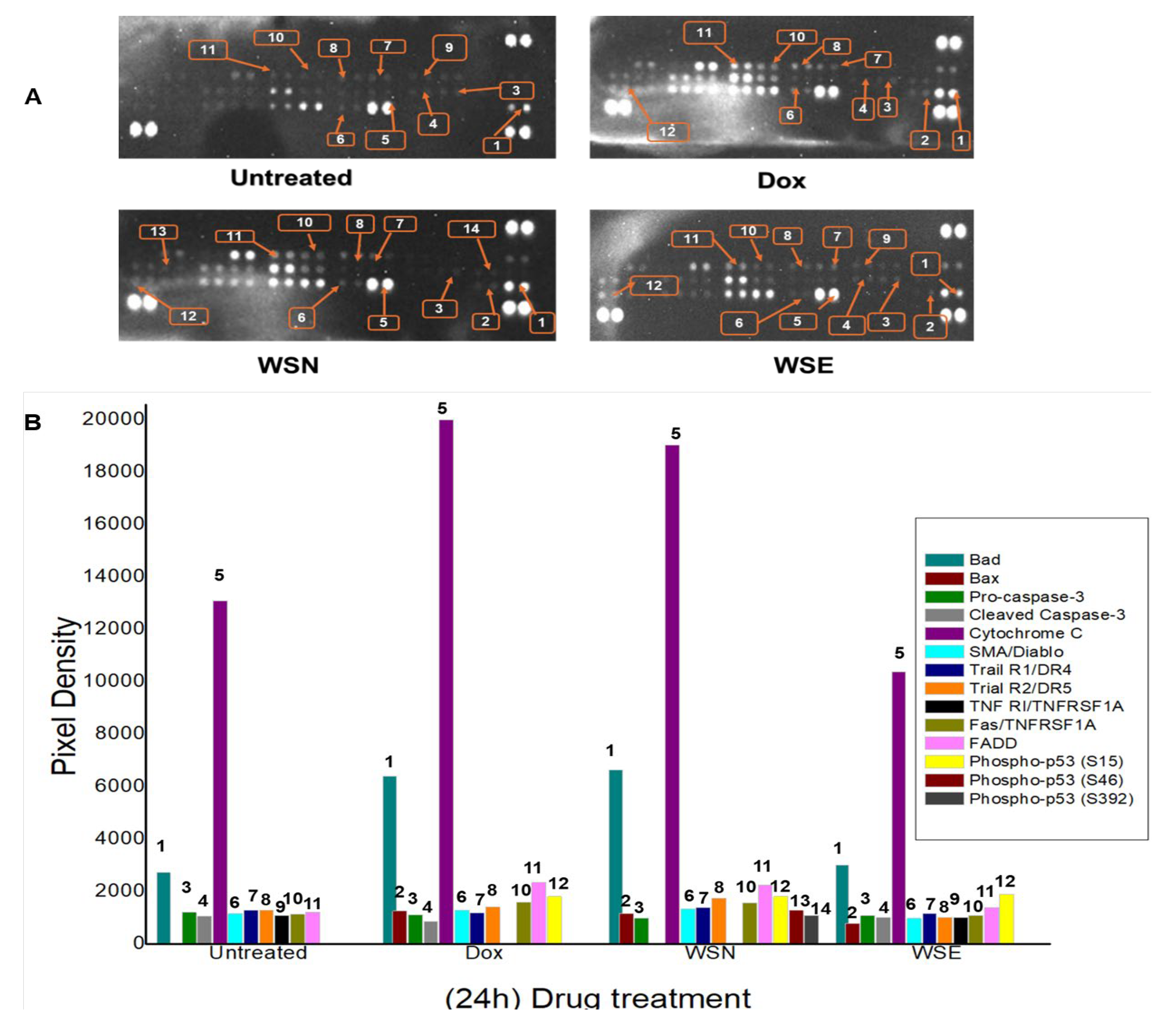
2.8. Analysis of the Human Apoptotic Proteome Profiling
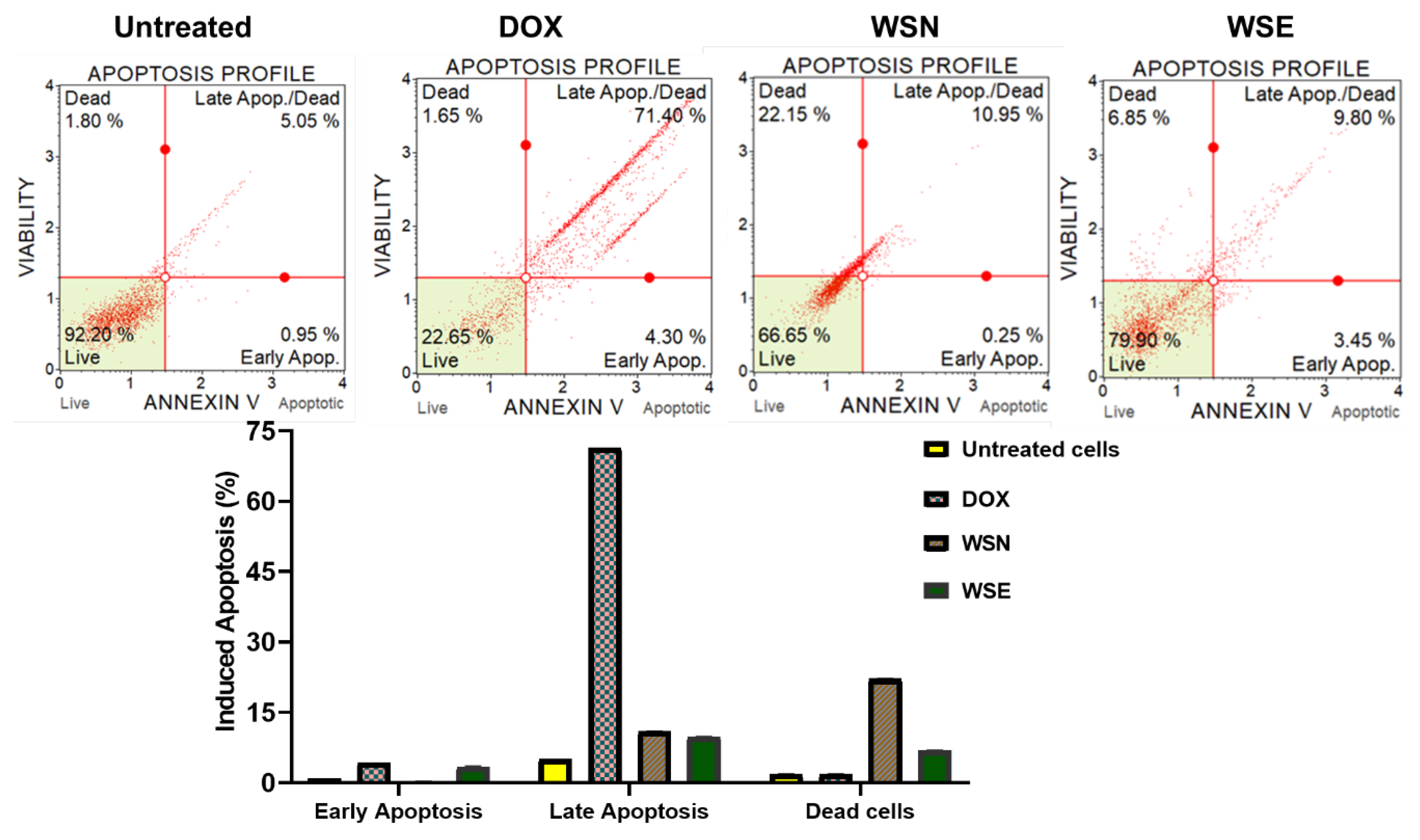
2.9. Analysis of Apoptosis Profile in MCF-7 Using the Annexin V/PI Staining Method
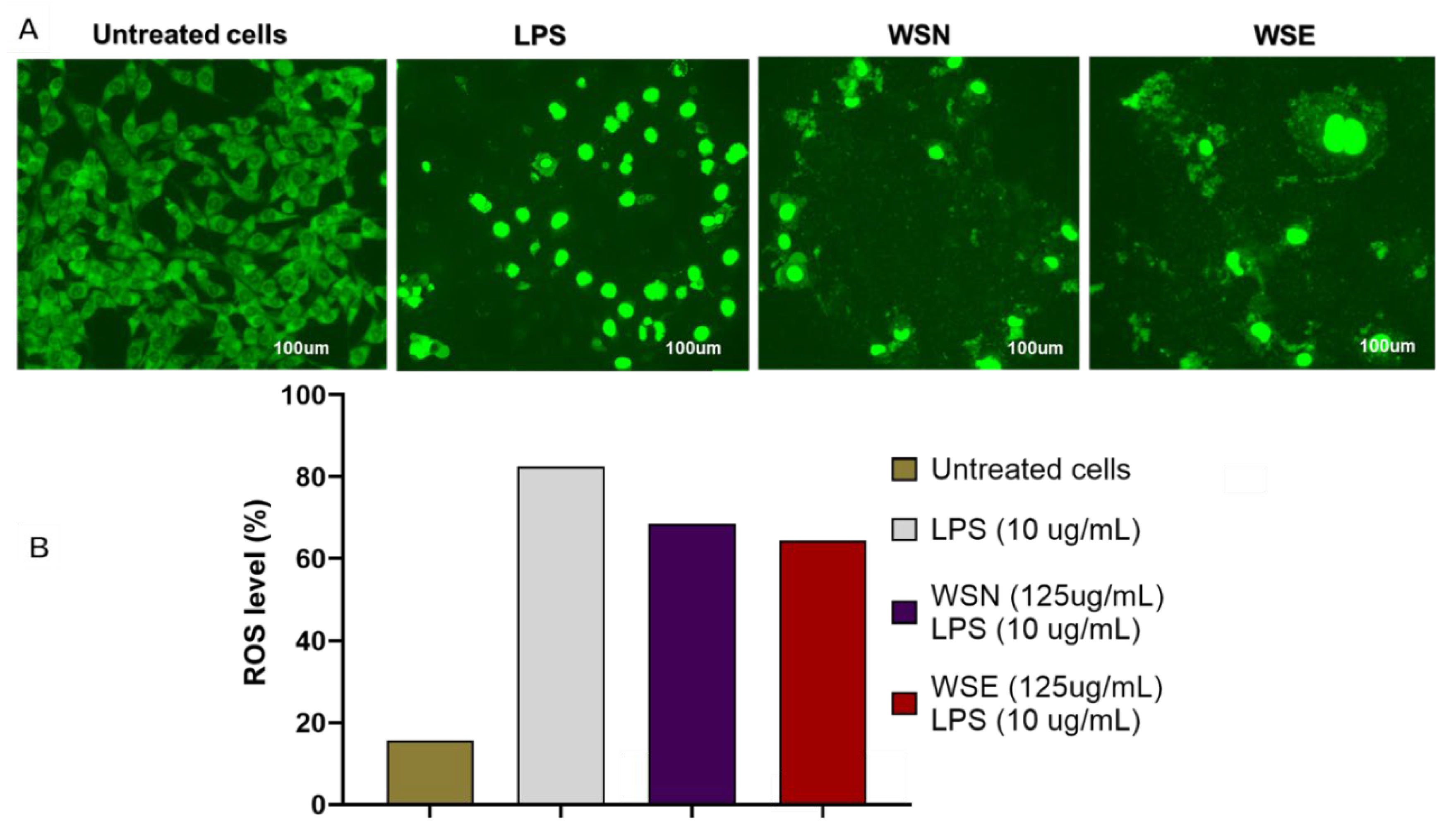
2.10. The Effect of WSN and WSE Crude Leaf Extract on ROS Production Measured with H2DCF-DA (ROS Probe)
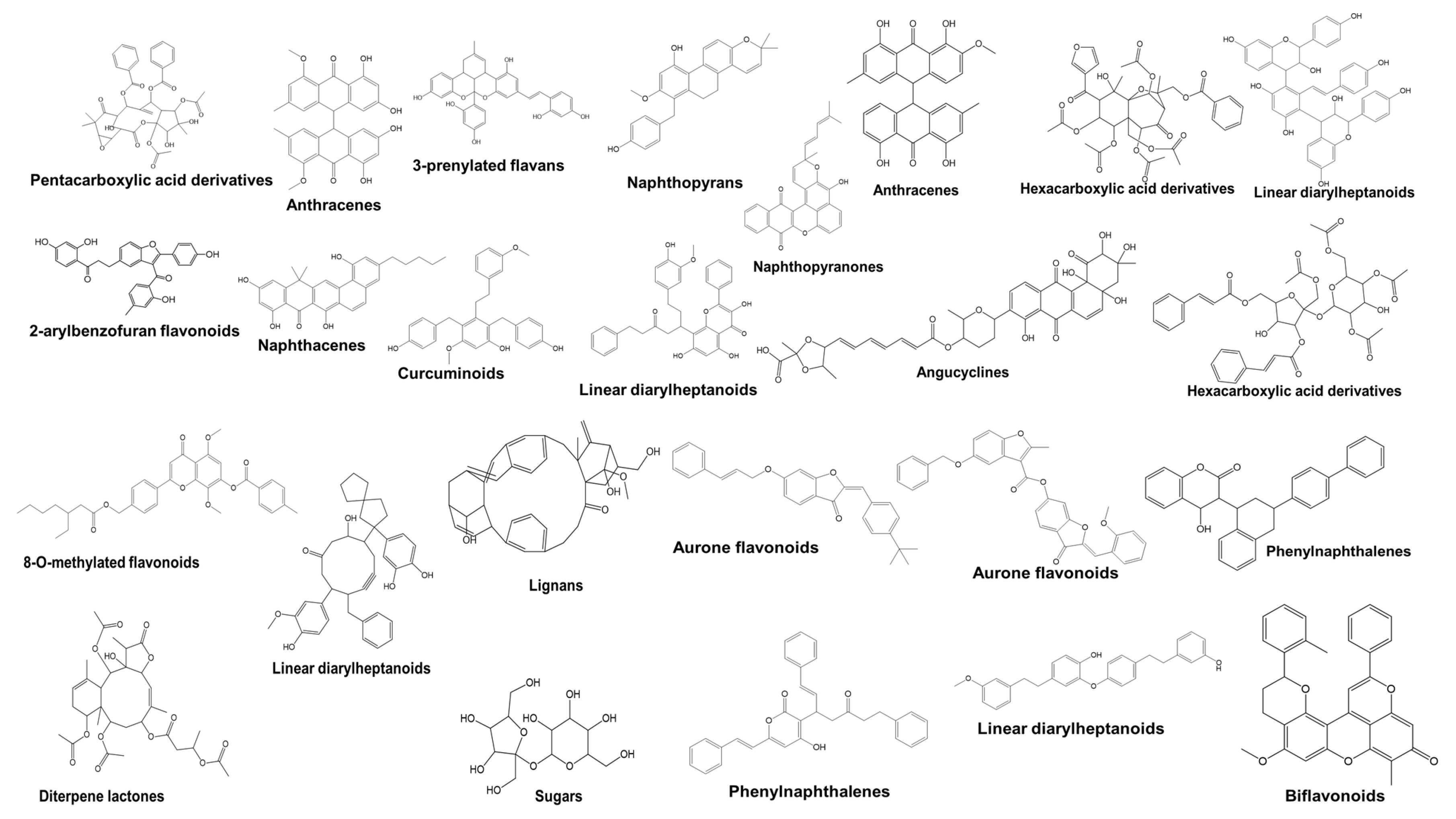
2.11. Liquid Chromatography-Mass Spectrometry (LC-MS)
3. Discussion
4. Materials and Methods
4.1. Synthesis of Liposomal Nanoparticles
4.2. FTIR Spectroscopy
4.3. Powder X-Ray Diffraction (pXRD)
4.4. Zeta Potential
4.5. Cell Culture
4.6. Cell Viability Assay
4.7. Anti-Metastatic Assay in MCF-7 Cells
4.8. H2DCF-DA Assay
4.9. RNA Extraction and Purification
4.10. First-Strand cDNA Synthesis
4.11. Polymerase Chain Reaction (PCR)
4.12. Annexin V-FITC and Propidium Iodide (PI), Analysis by Muse® Cell Analyser
4.13. Human Apoptosis Proteome Array
4.14. Liquid Chromatography-Mass Spectrometry (LC-MS)
4.15. Statistical Analysis
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Bax | Bcl-2-associated X protein |
| Bcl-2 | B-cell lymphoma 2 |
| DLS | Dynamic Light Scattering |
| FTIR | Fourier Transform Infrared Spectroscopy |
| H2DCF-DA | 2′,7′-Dichlorodihydrofluorescein diacetate |
| IC50 | Half-maximal inhibitory concentration |
| LC–MS | Liquid Chromatography–Mass Spectrometry |
| LPS | Lipopolysaccharide |
| PDI | Polydispersity Index; |
| PXRD | Powder X-ray Diffraction |
| RT-PCR | Reverse Transcription Polymerase Chain Reaction |
| WSE | Warburgia salutaris liposomal-encapsulated crude leaf extract |
| WSN | Warburgia salutaris unencapsulated crude leaf extract |
References
- Yadav, AR; Mohite, SK. Cancer-A silent killer: An overview. Asian Journal of Pharmaceutical Research 2020, 10(3), 213–216. [Google Scholar] [CrossRef]
- Santibáñez-Andrade, M; Chirino, YI; González-Ramírez, I; Sánchez-Pérez, Y; García-Cuellar, CM. Deciphering the code between air pollution and disease: the effect of particulate matter on cancer hallmarks. International journal of molecular sciences 2019, 21(1), 136. [Google Scholar] [CrossRef]
- Ali, A; Manzoor, MF; Ahmad, N; et al. The burden of cancer, government strategic policies, and challenges in Pakistan: A comprehensive review. Frontiers in nutrition 2022, 9, 940514. [Google Scholar] [CrossRef]
- Halperin, DT. Coping with COVID-19: learning from past pandemics to avoid pitfalls and panic. Global Health: Science and Practice 2020, 8(2), 155–165. [Google Scholar] [CrossRef]
- Finestone, E; Wishnia, J. Estimating the burden of cancer in South Africa. SA Journal of Oncology 2022, 6(0), 220. [Google Scholar] [CrossRef]
- Hamdi, Y; Abdeljaoued-Tej, I; Zatchi, AA; et al. Cancer in Africa: the untold story. Frontiers in oncology 2021, 11, 650117. [Google Scholar] [CrossRef]
- Lombe, DC; Mwamba, M; Msadabwe, S; et al. Delays in seeking, reaching and access to quality cancer care in sub-Saharan Africa: a systematic review. BMJ open. 2023, 13(4), e067715. [Google Scholar] [CrossRef]
- Ndlovu, BH. Awareness, knowledge and experiences of women regarding cervical cancer in rural Kwazulu-Natal, South Africa; University of Stellenbosch: Stellenbosch, 2011. [Google Scholar]
- Goswami, N. A dual burden dilemma: Navigating the global impact of communicable and non-communicable diseases and the way forward. International Journal of Medical Research 2024, 12(3), 65–77. [Google Scholar] [CrossRef]
- Schirrmacher, V. From chemotherapy to biological therapy: A review of novel concepts to reduce the side effects of systemic cancer treatment. International journal of oncology 2019, 54(2), 407–419. [Google Scholar] [CrossRef]
- Tripathi, D; Pandey, P; Sharma, S; Rai, AK; BH, MP. Advances in nanomaterials for precision drug delivery: Insights into pharmacokinetics and toxicity. BioImpacts: BI 2024, 15, 30573. [Google Scholar] [CrossRef]
- Mao, JJ; Pillai, GG; Andrade, CJ; et al. Integrative oncology: Addressing the global challenges of cancer prevention and treatment. CA: a cancer journal for clinicians 2022, 72(2), 144–164. [Google Scholar] [CrossRef]
- Kutova, OM; Guryev, EL; Sokolova, EA; Alzeibak, R; Balalaeva, IV. Targeted delivery to tumors: multidirectional strategies to improve treatment efficiency. Cancers 2019, 11(1), 68. [Google Scholar] [CrossRef]
- Dehelean, CA; Marcovici, I; Soica, C; et al. Plant-derived anticancer compounds as new perspectives in drug discovery and alternative therapy. Molecules 2021, 26(4), 1109. [Google Scholar] [CrossRef]
- Hashim, GM; Shahgolzari, M; Hefferon, K; Yavari, A; Venkataraman, S. Plant-Derived Anti-Cancer Therapeutics and Biopharmaceuticals. Bioengineering 2024, 12(1), 7. [Google Scholar] [CrossRef]
- Salehi, B; Fokou, PVT; Yamthe, LRT; et al. Phytochemicals in prostate cancer: from bioactive molecules to upcoming therapeutic agents. Nutrients 2019, 11(7), 1483. [Google Scholar] [CrossRef]
- Ayaz, M; Nawaz, A; Ahmad, S; et al. Underlying anticancer mechanisms and synergistic combinations of phytochemicals with cancer chemotherapeutics: potential benefits and risks. Journal of Food Quality 2022, 2022(1), 1189034. [Google Scholar] [CrossRef]
- Viljoen, A; Sandasi, M; Fouche, G; Combrinck, S; Vermaak, I. The South African Herbal Pharmacopoeia: Monographs of Medicinal and Aromatic Plants; Academic Press, 2022. [Google Scholar]
- Kyriakoudi, A; Spanidi, E; Mourtzinos, I; Gardikis, K. Innovative Delivery Systems Loaded with Plant Bioactive Ingredients: Formulation Approaches and Applications. Plants 2021, 10, 1238, s Note: MDPI stays neutral with regard to jurisdictional claims in published …; 2021. [Google Scholar] [CrossRef]
- Leonard, C; Chen, W; Kamatou, G. Warburgia salutaris. The South African Herbal Pharmacopoeia; Elsevier, 2023; pp. 531–556. [Google Scholar]
- Garcia-Oliveira, P; Otero, P; Pereira, AG; et al. Status and challenges of plant-anticancer compounds in cancer treatment. Pharmaceuticals 2021, 14(2), 157. [Google Scholar] [CrossRef]
- Emeihe, EV; Nwankwo, EI; Ajegbile, MD; Olaboye, JA; Maha, CC. Revolutionizing drug delivery systems: Nanotechnology-based approaches for targeted therapy. Int J Life Sci Res Arch. 2024, 7(1), 40–58. [Google Scholar] [CrossRef]
- Eloff, J. Which extractant should be used for the screening and isolation of antimicrobial components from plants? Journal of ethnopharmacology 1998, 60(1), 1–8. [Google Scholar] [CrossRef]
- Umbarkar, M; Thakare, S; Surushe, T; Giri, A; Chopade, V. Formulation and evaluation of liposome by thin film hydration method. J Drug Deliv Ther. 2021, 11(1), 72–76. [Google Scholar] [CrossRef]
- Jovanović, AA; Balanč, B; Petrović, PM; et al. Design and characterization of liposomal-based carriers for the encapsulation of rosa canina fruit extract: in vitro gastrointestinal release behavior. Plants 2024, 13(18), 2608. [Google Scholar] [CrossRef]
- Ajeeshkumar, KK; Aneesh, PA; Raju, N; Suseela, M; Ravishankar, CN; Benjakul, S. Advancements in liposome technology: Preparation techniques and applications in food, functional foods, and bioactive delivery: A review. Comprehensive Reviews in Food Science and Food Safety 2021, 20(2), 1280–1306. [Google Scholar] [CrossRef]
- Bononi, G; Masoni, S; Di Bussolo, V; Tuccinardi, T; Granchi, C; Minutolo, F. Historical perspective of tumor glycolysis: a century with Otto Warburg; Elsevier, 2022; pp. 325–333. [Google Scholar]
- Jakob, CH; Dominelli, B; Schlagintweit, JF; et al. Improved antiproliferative activity and fluorescence of a dinuclear gold (I) bisimidazolylidene complex via anthracene-modification. Chemistry–An Asian Journal. 2020, 15(24), 4275–4279. [Google Scholar] [CrossRef]
- Hameed, R. Diarylheptanoids: Potent Anticancer Agents. Clinical Cancer Drugs 2021, 8(1), 18–26. [Google Scholar] [CrossRef]
- Esquivel-Campos, A; Pérez-Gutiérrez, S; Sánchez-Pérez, L; Campos-Xolalpa, N; Pérez-Ramos, J. Cytotoxicity and Antitumor Action of Lignans and Neolignans. In Secondary Metabolites-Trends and Reviews; 2022. [Google Scholar]
- Ahmed Borik, RM; Amri, NJ; Mukhrish, YE; et al. Design, synthesis, reactions, molecular docking, antitumor activities of novel naphthopyran, naphthopyranopyrimidines, and naphthoyranotriazolopyrimidine derivatives. Current Organic Chemistry 2023, 27(19), 1717–1727. [Google Scholar] [CrossRef]
- Lima, CAd; Maquedano, LK; Jaalouk, LS; Santos, DCd; Longato, GB. Biflavonoids: preliminary reports on their role in prostate and breast cancer therapy. Pharmaceuticals 2024, 17(7), 874. [Google Scholar] [CrossRef]
- Weber, F; Weber, A; Schmitt, L; et al. From the Total Synthesis of Semi–Viriditoxin, Semi–Viriditoxic Acid and Dimeric Naphthopyranones to their Biological Activities in Burkitt B Cell Lymphoma. Chemistry–A European Journal 2024, 30(25), e202400559. [Google Scholar] [CrossRef]
- Jantip, P; Singh, CK; Klu, YAK; Ahmad, N; Bolling, BW. Peanut skin polyphenols inhibit proliferation of leukemia cells in vitro, and its A-type procyanidins selectively pass through a Caco-2 intestinal barrier. Journal of Food Science 2025, 90(2), e70018. [Google Scholar] [CrossRef]
- Sk, S; Chakrovorty, A; Samadder, A; Bera, M. A family of zinc compounds of an anthracene-appended new multifunctional organic scaffold as potent chemotherapeutics against cervical cancer. Materials Advances 2025, 6(4), 1478–1496. [Google Scholar] [CrossRef]
- Zhu, Q; Zheng, X; Tan, Y; Luo, Z; Yao, X; Chen, H. Biological Activities of Aurones: A Brief Summary. Mini-Reviews in Organic Chemistry 2025, 22(2), 226–243. [Google Scholar] [CrossRef]
- Jang, WY; Kim, M-Y; Cho, JY. Antioxidant, anti-inflammatory, anti-menopausal, and anti-cancer effects of lignans and their metabolites. International journal of molecular sciences 2022, 23(24), 15482. [Google Scholar] [CrossRef]
- Elrayess, RA; Elshihawy, H. Naphthalene: an overview. Records of Pharmaceutical and Biomedical Sciences 2023, 7(1), 145–153. [Google Scholar] [CrossRef]
- Bermejo-Casadesús, C; Gonzalo-Navarro, C; Organero, JA; et al. New encapsulated bis-cyclometalated Ir (III) complexes with very potent anticancer PDT activity. Inorganic Chemistry Frontiers 2025, 12(22), 7304–7332. [Google Scholar] [CrossRef]
- Liu, H-S; Chen, H-R; Huang, S-S; Li, Z-H; Wang, C-Y; Zhang, H. Bioactive Angucyclines/Angucyclinones Discovered from 1965 to 2023. Marine Drugs 2025, 23(1), 25. [Google Scholar] [CrossRef]
- Monama, LV; Tswaledi, DL; Hadzhi, TM; et al. Identification of Bioactive Compounds in Warburgia salutaris Leaf Extracts and Their Pro-Apoptotic Effects on MCF-7 Breast Cancer Cells. International Journal of Molecular Sciences 2025, 26(16), 8065. [Google Scholar] [CrossRef]
- Mustafa, M; Ahmad, R; Tantry, IQ; et al. Apoptosis: a comprehensive overview of signaling pathways, morphological changes, and physiological significance and therapeutic implications. Cells 2024, 13(22), 1838. [Google Scholar] [CrossRef]
- Wani, AK; Akhtar, N; Mir, TuG; et al. Targeting apoptotic pathway of cancer cells with phytochemicals and plant-based nanomaterials. Biomolecules 2023, 13(2), 194. [Google Scholar] [CrossRef]
- Maurent, K; Vanucci-Bacque, C; Baltas, M; Negre-Salvayre, A; Auge, N; Bedos-Belval, F. Synthesis and biological evaluation of diarylheptanoids as potential antioxidant and anti-inflammatory agents. European Journal of Medicinal Chemistry 2018, 144, 289–299. [Google Scholar] [CrossRef]
- Motiur Rahman, A; Lu, Y; Lee, H-J; et al. Linear diarylheptanoids as potential anticancer therapeutics: synthesis, biological evaluation, and structure–activity relationship studies. Archives of pharmacal research 2018, 41(12), 1131–1148. [Google Scholar] [CrossRef]
- Bharath, B; Perinbam, K; Devanesan, S; AlSalhi, MS; Saravanan, M. Evaluation of the anticancer potential of Hexadecanoic acid from brown algae Turbinaria ornata on HT–29 colon cancer cells. Journal of Molecular Structure 2021, 1235, 130229. [Google Scholar] [CrossRef]
- Sudarshan, K; Yarlagadda, S; Sengupta, S. Recent advances in the synthesis of diarylheptanoids. Chemistry–An Asian Journal 2024, 19(15), e202400380. [Google Scholar] [CrossRef]
- Covarrubias, A; Byles, V; Horng, T. ROS sets the stage for macrophage differentiation. Cell research 2013, 23(8), 984–985. [Google Scholar] [CrossRef]
- Xie, Y; Liu, F; Wu, Y; et al. Inflammation in cancer: therapeutic opportunities from new insights. Molecular cancer 2025, 24(1), 51. [Google Scholar] [CrossRef]
- Aggarwal, V; Tuli, HS; Varol, A; et al. Role of reactive oxygen species in cancer progression: molecular mechanisms and recent advancements. Biomolecules 2019, 9(11), 735. [Google Scholar] [CrossRef]
- Nwosu, SN; Obagbemisoye, OV; Onuba, CO; Balogun, SE. Advances in Anticancer Drug Discovery: Exploring the Untapped Potential of Natural Products for Targeted Therapies. Journal of Advances in Medical and Pharmaceutical Sciences 2024, 26(11), 1–22. [Google Scholar] [CrossRef]
- Karonen, M. Insights into polyphenol–lipid interactions: Chemical methods, molecular aspects and their effects on membrane structures. Plants 2022, 11(14), 1809. [Google Scholar] [CrossRef]
- López-Lázaro, M. The warburg effect: why and how do cancer cells activate glycolysis in the presence of oxygen? Anti-Cancer Agents in Medicinal Chemistry-Anti-Cancer Agents 2008, 8(3), 305–312. [Google Scholar] [CrossRef]
- Mukhija, M; Joshi, BC; Bairy, PS; Bhargava, A; Sah, AN. Lignans: a versatile source of anticancer drugs. Beni-suef university journal of basic and applied sciences 2022, 11(1), 76. [Google Scholar] [CrossRef]
- Chang, C-F; Ke, C-Y; Wu, Y-C; Chuang, T-H. Structure-activity relationship of synthetic 2-phenylnaphthalenes with hydroxyl groups that inhibit proliferation and induce apoptosis of MCF-7 cancer cells. PLoS One 2015, 10(10), e0141184. [Google Scholar] [CrossRef]
- Zhao, H; Wu, L; Yan, G; et al. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal transduction and targeted therapy 2021, 6(1), 263. [Google Scholar] [CrossRef]
- Greten, FR; Grivennikov, SI. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 2019, 51(1), 27–41. [Google Scholar] [CrossRef]
- Luo, R; Yue, Z; Yang, Q; et al. In situ repolarization of tumor-associated macrophages with synergic nanoformulation to reverse immunosuppressive TME in mouse breast cancer for cancer therapy. Journal of Pharmaceutical Analysis 2024, 14(8), 100941. [Google Scholar] [CrossRef]
- Altemimi, A; Lakhssassi, N; Baharlouei, A; Watson, DG; Lightfoot, DA. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 2017, 6(4), 42. [Google Scholar] [CrossRef] [PubMed]
- Edwards, RL; Luis, PB; Varuzza, PV; et al. The anti-inflammatory activity of curcumin is mediated by its oxidative metabolites. Journal of Biological Chemistry 2017, 292(52), 21243–21252. [Google Scholar] [CrossRef] [PubMed]
- Rani, P; Dhillon, S; Kumari, G; et al. 2-Arylidene-6-methyl-2H-furo [3, 2-c] pyran-3, 4-diones: Design, synthesis, and evaluation of Anti-inflammatory, Anti-malarial, and Anti-cancer efficacy. Journal of Molecular Structure 2025, 1340, 142371. [Google Scholar] [CrossRef]
| Parameter | Value | Comments |
|---|---|---|
| Average Particle size (nm) | 159.4 | Nanoscale vesicle suitable for drug delivery |
| Polydispersity Index (PDI) | 0.114 | A narrow size distribution indicates a monodisperse vesicle population |
| Zeta potential (mV) | +79.3 | Strong positive surface charge, strong electrostatic repulsion, enhances colloidal stability and prevents aggregation |
| ID | Retention time (min) | Measured (m/z) | Compound name | Biological Activity | Molecular Formula | Ref |
|---|---|---|---|---|---|---|
| 545 | 5.4 | 507.14468 | 2-arylbenzofuran flavonoids | Antioxidant, Anti-inflammatory | C31H24O7 | (uk et at., 2023). |
| 605 | 5.464 | 563.17297 | 3-prenylated flavans | Anticancer, Cytotoxic | C34H28O8 |
(Jantip et al., 2025) |
| 435 | 7.079 | 455.18509 | Naphthacenes | Antimicrobial, Antitumor | C29H28O5 |
(Bermejo-Casadesús et al., 2025) |
| 573 | 7.311 | 537.15637 | Anthracenes | Cytotoxic, Antiproliferative | C32H26O8 | (Jakob et al., 2020) |
| 315 | 7.473 | 405.22162 | Linear diarylheptanoids | Antioxidant, Anti-inflammatory | C30H30O | (Fang et al., 2024). |
| 327 | 7.761 | 413.17435 | Naphthopyrans | Anti-inflammatory, Cytotoxic | C27H26O4 | (Ahmed Borik et al., 2023). |
| 474 | 9.048 | 469.19977 | Curcuminoids | Anticancer, Anti-inflammatory | C30H30O5 |
(Kaur et al., 2024) |
| 631 | 9.175 | 579.20422 | Linear diarylheptanoids | Anti-inflammatory, Cytotoxic | C35H32O8 |
(Hameed, 2021). |
| 318 | 9.578 | 409.17892 | Aurone flavonoids | Antioxidant, Anticancer | C28H26O3 |
(kte, Ungureanu, and Zaharia, 2023). |
| 572 | 9.802 | 531.14600 | Aurone flavonoids | Anti-inflammatory, Cytotoxic | C33H24O7 |
(Zhu et al., 2025) |
| 409 | 9.893 | 445.17993 | Phenylnaphthalenes | Antiproliferative, anti-cancer | C31H26O3 |
(Elrayess and Elshihawy, 2023) |
| 494 | 10.749 | 475.19086 | Linear diarylheptanoids | Anticancer, Anti-inflammatory | C32H28O4 |
(Sudarshan, Yarlagadda and Sengupta, 2024) |
| 394 | 11.486 | 439.18967 | Lignans | Anticancer, Cytotoxic | C29H28O4 |
(Esquivel-Campos et al., 2022). |
| 538 | 11.585 | 501.17010 | Biflavonoids | Antiproliferative, anti-cancer, Cytotoxic | C33H26O5 | (Lima et al., 2024) |
| 452 | 11.592 | 461.13800 | Naphthopyranones | Cytotoxic, Anticancer | C30H22O5 |
(Weber, F et al., 2024) |
| 544 | 11.592 | 507.14429 | Anthracenes | Cytotoxic, anti-cancer, Antiproliferative | C31H24O7 |
(Sk et al., 2025) |
| 758 | 11.684 | 755.21619 | Hexacarboxylic acid derivatives | Anti-inflammatory, anti-cancer, Cytotoxic | C37H40O17 |
(Bharath et al., 2021) |
| 751 | 11.959 | 739.22125 | Linear diarylheptanoids | Anticancer, Anti-inflammatory | C44H36O11 | (Motiur et al., 2018) |
| 748 | 12.986 | 735.22589 | Angucyclines | Antimicrobial, Antitumor | C38H40O15 |
(Liu et al., 2025). |
| 640 | 13.126 | 585.24927 | 8-O-methylated flavonoids | Anticancer, Cytotoxic | C35H38O8 |
(Berim and Gang, 2016) |
| 674 | 13.574 | 607.30713 | Linear diarylheptanoids | Anticancer, Anti-inflammatory | C39H44O6 |
(Maurent et al., 2018). |
| 619 | 13.588 | 575.27985 | Lignans | Antiproliferative, anti-cancer, Cytotoxic | C38H40O5 |
(Jang, Kim and Cho, 2022) |
| 700 | 13.941 | 635.26794 | Diterpene lactones | Cytotoxic, Antitumor | C32H44O13 | (Islam et al., 2018) |
| 246 | 13.377 | 341.10751 | Sugars | Nutrient, not bioactive, anti-cancer | C12H22O11 |
(Akl and Ahmed, 2023) |
| Gene | Primer sequence |
|---|---|
| caspase-3 | Forward: 5’CCATGGGTAGCAGCCTCCTTC 3’ Reverse: 3’ TGCGCTGCTCTGCCTTCT 5’ |
| Bax | Forward: 5’TCCCCCCAGAGGTCTTTT 3’ Reverse: 3’CGGCCCCAGTTGAAGTTG 5’ |
| bcl-2 | Forward: 5’CTGCACCTGACGCCCTTCACC 3’ Reverse: 3’CACATGACCCCACCGAACTCAAAGA 5’ |
| GAPDH | Forward: 5’TGCGCTGCTGCTCTGCCTTCT 3’ Reverse: 3’CCATGGGTAGCAGCTCCTTC 5’ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




