Submitted:
19 March 2026
Posted:
25 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Biological Mechanisms Linking ADT to Myosteatosis and Sarcopenic Obesity
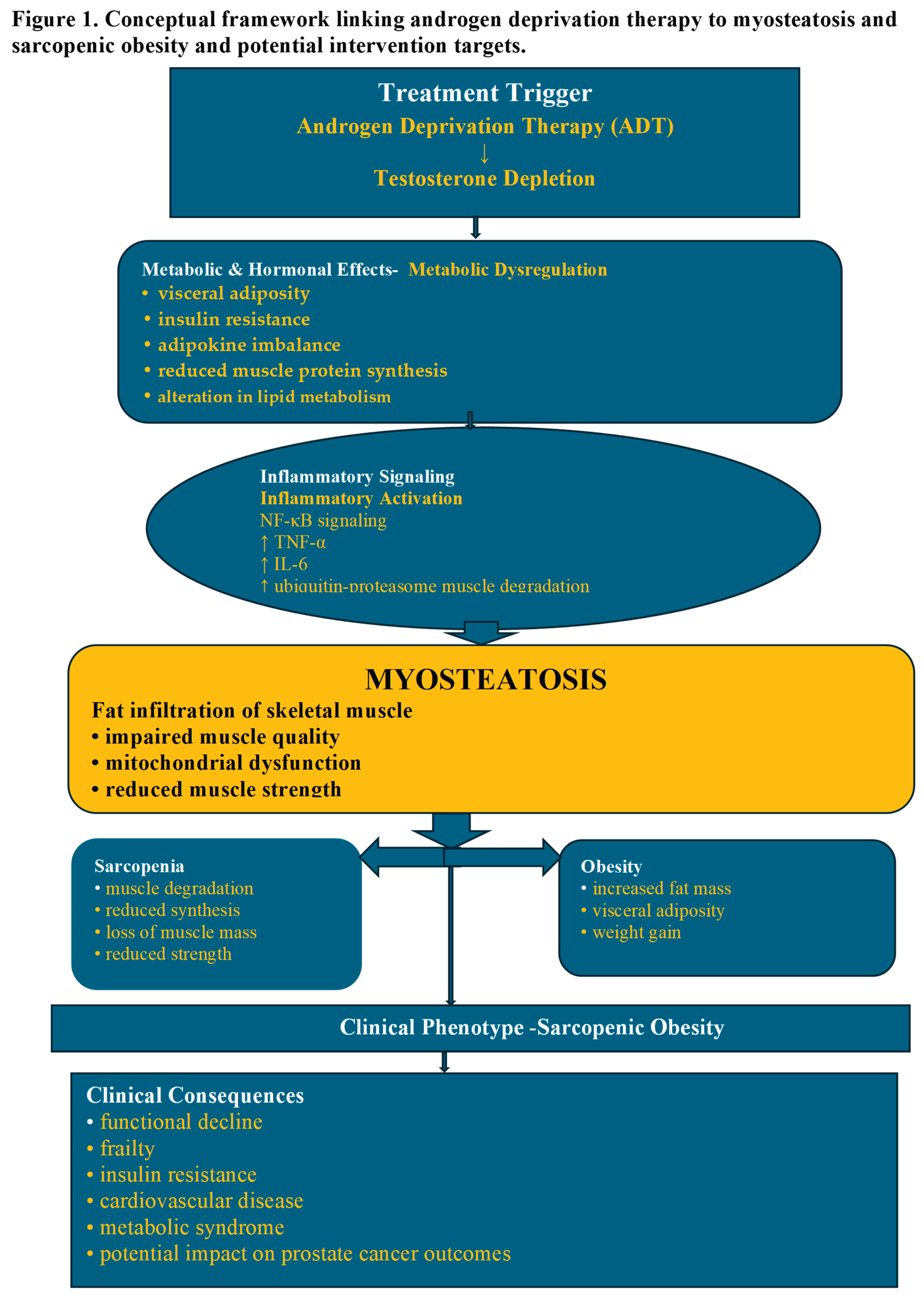
3.2. Conceptual Framework (Figure 1)
3.3. Current Strategies to Manage Sarcopenia, Obesity, and Other Metabolic Consequences of ADT
3.4. Promising Approaches to Ameliorate SO and Other Metabolic Consequences of ADT
3.4.1. Exercise
3.4.2. Protein and Micronutrient Support
3.4.3. Omega-3 Fatty Acids
3.4.4. Plant-Rich, Phytochemical-Based Dietary Strategies
3.4.5. Time-Restricted Eating and Fasting-Based Approaches
4. Conclusions
5. Future Directions
 |
 |
Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SO | Sarcopenic obesity |
| MYO | Myosteatosis |
| CC | Cancer cachexia |
| BMD | Bone mineral density |
| PCa | Prostate cancer |
| IGF-1 | Insulin-like Growth Factor 1 |
| IGFBP-1 | Insulin-like growth factor-binding protein 1 |
| MTOR | Mammalian target of rapamycin |
| EGCG | Epigallocatechin gallate |
| PIF | Proteolytic inducing factor |
| TRE | Time restricted eating |
| FMD | Fasting Mimicking Diet |
| TRF | Time restricted feeding |
| Kcal | Kilocalories |
| BP | Blood pressure |
| EPA | Eicosapentaenoic acid |
| DHA | Docosahexaenoic acid |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| TNFα | Tumor necrosis factor-alpha |
| Il | Interleukins |
| CT | Computer Tomography |
| L3 | Lumbar vertebrae 3 |
| SMI | Skeletal muscle index (SMI) |
| SMD | Skeletal muscle radiodensity |
| ADT | Androgen deprivation therapy |
References
- American Cancer Society. Cancer Facts & Figures 2026; American Cancer Society: Atlanta, 2025. [Google Scholar]
- Huggins, C; Hodges, CV. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. J Urol 1941, 168, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, SM; fang KuoY; Shahinian, VB. Prevalent and incident use of androgen deprivation therapy among men with prostate cancer in the United States. Urol Oncol 2011, 29, 647–653. [Google Scholar] [CrossRef]
- Payne, H; Mason, M. Androgen deprivation therapy as adjuvant/neoadjuvant to radiotherapy for high-risk localised and locally advanced prostate cancer: Recent developments. Br J Cancer 2011, 105, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Kovač, MB; Pavlin, T; Čavka, L; Ribnikar, D; Spazzapan, S; Templeton, AJ; Šeruga, B. The trajectory of sarcopenia following diagnosis of prostate cancer: A systematic review and meta-analysis. J Geriatr Oncol. 2023, 14(7), 101594. [Google Scholar] [CrossRef] [PubMed]
- Stangl-Kremser, J; Mari, A; Lai, LY; Lee, CT; Vince, R; Zaslavsky, A; Salami, SS; Fajkovic, H; Shariat, SF; Palapattu, GS. Sarcopenic Obesity and its Prognostic Impact on Urological Cancers: A Systematic Review. J Urol. 2021, 206(4), 854–865. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T; Mao, W; Hu, M; Yu, Q; Peng, X; Ji, J; Qiu, J; Wu, J. Advances in sarcopenia and urologic disorders. Front Nutr. 2024, 11, 1475977. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y; Yamada, M; Ohji, S; Ishiyama, D; Nishio, N; Otobe, Y; Koyama, S; Suzuki, M; Ichikawa, T; Ito, D; Maehori, N; Nagae, H. Presence of sarcopenic obesity and evaluation of the associated muscle quality in Japanese older men with prostate cancer undergoing androgen deprivation therapy. J Geriatr Oncol. 2019, 10(5), 835–838. [Google Scholar] [CrossRef] [PubMed]
- Focht, BC; Lucas, AR; Grainger, E; Simpson, C; Thomas-Ahner, JM; Clinton, SK. The Individualized Diet and Exercise Adherence Pilot Trial (IDEA-P) in prostate cancer patients undergoing androgen deprivation therapy: study protocol for a randomized controlled trial. Trials 2014, 15, 354. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stelmach-Mardas, M; Warchoł, W; Garczyk, A; Warchoł, E; Korczak, J; Litwiniuk, M; Brajer-Luftmann, B; Mardas, M. Influence of Androgen Deprivation Therapy on the Development of Sarcopenia in Patients with Prostate Cancer: A Systematic Review. Nutrients 2024, 16(5), 656. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Donini, LM; Busetto, L; Bischoff, SC; Cederholm, T; Ballesteros-Pomar, MD; Batsis, JA; Bauer, JM; Boirie, Y; Cruz-Jentoft, AJ; Dicker, D; Frara, S; Frühbeck, G; Genton, L; Gepner, Y; Giustina, A; Gonzalez, MC; Han, HS; Heymsfield, SB; Higashiguchi, T; Laviano, A; Lenzi, A; Nyulasi, I; Parrinello, E; Poggiogalle, E; Prado, CM; Salvador, J; Rolland, Y; Santini, F; Serlie, MJ; Shi, H; Sieber, CC; Siervo, M; Vettor, R; Villareal, DT; Volkert, D; Yu, J; Zamboni, M; Barazzoni, R. Definition and Diagnostic Criteria for Sarcopenic Obesity: ESPEN and EASO Consensus Statement. Obes Facts 2022, 15(3), 321–335. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dolan, R.D.; Almasaudi, A.S.; Dieu, L.B.; Horgan, P.G.; McSorley, S.T.; McMillan, D.C. The relationship between computed tomography-derived body composition, systemic inflammatory response, and survival in patients undergoing surgery for colorectal cancer. J Cachexia Sarcopenia Muscle 2019, 10, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Derweesh, IH; Diblasio, CJ; Kincade, MC; Malcolm, JB; Lamar, KD; Patterson, AL; Kitabchi, AE; Wake, RW. Risk of new-onset diabetes mellitus and worsening glycaemic variables for established diabetes in men undergoing androgen-deprivation therapy for prostate cancer. BJU Int. 2007, 100(5), 1060–5. [Google Scholar] [CrossRef] [PubMed]
- Harborg, S; Kjærgaard, KA; Thomsen, RW; Borgquist, S; Cronin-Fenton, D; Hjorth, CF. New Horizons: Epidemiology of Obesity, Diabetes Mellitus, and Cancer Prognosis. J Clin Endocrinol Metab. 2024, 109(4), 924–935. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D; Toker, M; Shyr, W; Fram, E; Watts, KL; Agalliu, I. Association of Obesity and Diabetes With Prostate Cancer Risk Groups in a Multiethnic Population. Clin Genitourin Cancer 2022, 20(3), 299–299.e10. [Google Scholar] [CrossRef] [PubMed]
- Wilson, RL; Taaffe, DR; Newton, RU; Hart, NH; Lyons-Wall, P; Galvão, DA. Using Exercise and Nutrition to Alter Fat and Lean Mass in Men with Prostate Cancer Receiving Androgen Deprivation Therapy: A Narrative Review. Nutrients 2021, 13(5), 1664. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Batsis, JA; Villareal, DT. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nat Rev Endocrinol 2018, 14(9), 513–537. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rivera-Izquierdo, M; Pérez de Rojas, J; Martínez-Ruiz, V; Arrabal-Polo, MÁ; Pérez-Gómez, B; Jiménez-Moleón, JJ. Obesity and biochemical recurrence in clinically localised prostate cancer: a systematic review and meta-analysis of 86,490 patients. Prostate Cancer Prostatic Dis. 2022, 25(3), 411–421. [Google Scholar] [CrossRef] [PubMed]
- Troeschel, AN; Hartman, TJ; Jacobs, EJ; Stevens, VL; Gansler, T; Flanders, WD; McCullough, LE; Wang, Y. Postdiagnosis Body Mass Index, Weight Change, and Mortality From Prostate Cancer, Cardiovascular Disease, and All Causes Among Survivors of Nonmetastatic Prostate Cancer. J Clin Oncol. 2020, 38(18), 2018–2027. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mitsuzuka, K; Arai, Y. Metabolic changes in patients with prostate cancer during androgen deprivation therapy. Int J Urol 2018, 25(1), 45–53. [Google Scholar] [CrossRef] [PubMed]
- Lifshitz, K; Ber, Y; Margel, D. Role of Metabolic Syndrome in Prostate Cancer Development. Eur Urol Focus 2021, 7(3), 508–512. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, M; Mazzali, G; Fantin, F; Rossi, A; Di Francesco, V. Sarcopenic obesity: a new category of obesity in the elderly. Nutr Metab Cardiovasc Dis. 2008, 18(5), 388–95. [Google Scholar] [CrossRef] [PubMed]
- Veen, J; Montiel-Rojas, D; Nilsson, A; Kadi, F. Engagement in Muscle-Strengthening Activities Lowers Sarcopenia Risk in Older Adults Already Adhering to the Aerobic Physical Activity Guidelines. Int J Environ Res Public Health 2021, 18(3), 989. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, JW; Kim, R; Choi, H; Lee, SJ; Bae, GU. Understanding of sarcopenia: from definition to therapeutic strategies. Arch Pharm Res 2021, 44(9-10), 876–889. [Google Scholar] [CrossRef] [PubMed]
- Kumar, NB; Kazi, A; Smith, T; Crocker, T; Yu, D; Reich, RR; Reddy, K; Hastings, S; Exterman, M; Balducci, L; Dalton, K; Bepler, G. Cancer cachexia: traditional therapies and novel molecular mechanism-based approaches to treatment. Curr Treat Options Oncol 2010, 11(3-4), 107–17. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Villareal, JA. DT. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nat Rev Endocrinol 2018, 14(9), 513–537. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Whitehouse, AS; Tisdale, MJ. Increased expression of the ubiquitin-proteasome pathway in murine myotubes by proteolysis-inducing factor (PIF) is associated with activation of the transcription factor NF-kappaB. Br J Cancer 2003, 89(6), 1116–22. [Google Scholar] [CrossRef] [PubMed]
- Melstrom, LG; Melstrom, KA, Jr.; Ding, XZ; Adrian, TE. Mechanisms of skeletal muscle degradation and its therapy in cancer cachexia. Histol Histopathol 2007, 22(7), 805–14. [Google Scholar]
- Mitch, WE; Price, SR. Transcription factors and muscle cachexia: is there a therapeutic target? Lancet 2001, 357(9258), 734–5. [Google Scholar] [CrossRef] [PubMed]
- Ciechanover, A; Orian, A; Schwartz, AL. The ubiquitin-mediated proteolytic pathway: mode of action and clinical implications. J Cell Biochem Suppl. 2000, 34, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Voges, D; Zwickl, P; Baumeister, W. The 26S proteasome: a molecular machine designed for controlled proteolysis. Annu Rev Biochem. 1999, 68, 1015–68. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S; Karin, M. Missing pieces in the NF-kappaB puzzle. Cell. 2002, 109 Suppl, S81–96. [Google Scholar] [CrossRef]
- Cartwright, MJ; Tchkonia, T; Kirkland, JL. Aging in adipocytes: potential impact of inherent, depot-specific mechanisms. Exp Gerontol 2007, 42(6), 463–71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jiang, Z; Marriott, K; Maly, MR. Impact of Inter- and Intramuscular Fat on Muscle Architecture and Capacity. Crit Rev Biomed Eng. 2019, 47(6), 515–533. [Google Scholar] [CrossRef] [PubMed]
- Daniell, HW; Dunn, SR; Ferguson, DW; et al. Progressive osteoporosis during androgen deprivation therapy for prostate cancer. J Urol 2000, 163, 181–186. [Google Scholar] [CrossRef]
- Smith, MR; McGovern, FJ; Zietman, AL; et al. Pamidronate to prevent bone loss during androgen-deprivation therapy for prostate cancer. N Engl J Med 2001, 345, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Gudenkauf, LM; Gray, S; Gonzalez, BD; Sachdeva, A; Autio, K. Balancing Hormone Therapy: Mitigating Adverse Effects of Androgen-Deprivation Therapy and Exploring Alternatives in Prostate Cancer Management. Am Soc Clin Oncol Educ Book 2024, 44(3), e433126. [Google Scholar] [CrossRef] [PubMed]
- Daniell, HW; Dunn, SR; Ferguson, DW; et al. Progressive osteoporosis during androgen deprivation therapy for prostate cancer. J Urol 2000, 163, 181–186. [Google Scholar] [CrossRef]
- Lin, KL; Talmor, B; Crumbaker, M; Joshua, A. A Review of Hot Flash Management in Patients with Prostate Cancer. J Clin Endocrinol Metab. Epub ahead of print. 2025, dgaf302. [Google Scholar] [CrossRef] [PubMed]
- Datta, M; Schwartz, GG. Calcium and vitamin D supplementation during androgen deprivation therapy for prostate cancer: A critical review. Oncologist 2012, 17, 1171–1179. [Google Scholar] [CrossRef]
- Galvão, DA; Newton, RU; Taaffe, DR; Spry, N. Can exercise ameliorate the increased risk of cardiovascular disease and diabetes associated with ADT? Nat Clin Pract Urol 2008, 5(6), 306–7. [Google Scholar] [CrossRef] [PubMed]
- Galvão, DA; Taaffe, DR; Newton, RU. Body weight, fat mass and metabolic complications during androgen deprivation therapy: should urologists recommend exercise and diet to help patients overcome toxicities? Prostate Cancer Prostatic Dis 2021, 24(3), 591–593. [Google Scholar] [CrossRef] [PubMed]
- Lopez, P; Radaelli, R; Taaffe, DR; Newton, RU; Galvão, DA; Trajano, GS; Teodoro, JL; Kraemer, WJ; Häkkinen, K; Pinto, RS. Resistance Training Load Effects on Muscle Hypertrophy and Strength Gain: Systematic Review and Network Meta-analysis. Med Sci Sports Exerc. Erratum in: Med Sci Sports Exerc. 2022 Feb 1;54(2):370. doi: 10.1249/MSS.0000000000002838. PMID: 33433148; PMCID: PMC8126497. 2021, 53(6), 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Lopez, P; Newton, RU; Taaffe, DR; Singh, F; Buffart, LM; Spry, N; Tang, C; Saad, F; Galvão, DA. Associations of fat and muscle mass with overall survival in men with prostate cancer: a systematic review with meta-analysis. Prostate Cancer Prostatic Dis. 2022, 25(4), 615–626. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lopez, P; Newton, RU; Taaffe, DR; Singh, F; Lyons-Wall, P; Buffart, LM; Tang, C; Hayne, D; Galvão, DA. Interventions for Improving Body Composition in Men with Prostate Cancer: A Systematic Review and Network Meta-analysis. Med Sci Sports Exerc. 2022, 54(5), 728–740. [Google Scholar] [CrossRef] [PubMed]
- Wilson, RL; Taaffe, DR; Newton, RU; Hart, NH; Lyons-Wall, P; Galvão, DA. Using Exercise and Nutrition to Alter Fat and Lean Mass in Men with Prostate Cancer Receiving Androgen Deprivation Therapy: A Narrative Review. Nutrients 2021, 13(5), 1664. [Google Scholar] [CrossRef] [PubMed]
- Focht, BC; Lucas, AR; Grainger, E; Simpson, C; Fairman, CM; Thomas-Ahner, JM; Buell, J; Monk, JP; Mortazavi, A; Clinton, SK. Effects of a Group-Mediated Exercise and Dietary Intervention in the Treatment of Prostate Cancer Patients Undergoing Androgen Deprivation Therapy: Results From the IDEA-P Trial. Ann Behav Med 2018, 52(5), 412–428. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Houben, LHP; Overkamp, M; VAN Kraaij, P; Trommelen, J; VAN Roermund, JGH; DE Vries, P; DE Laet, K; VAN DER Meer, S; Mikkelsen, UR; Verdijk, LB; VAN Loon, LJC; Beijer, S; Beelen, M. Resistance Exercise Training Increases Muscle Mass and Strength in Prostate Cancer Patients on Androgen Deprivation Therapy. Med Sci Sports Exerc. 2023, 55(4), 614–624. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wall, BA; GALVãO, DA; Fatehee, N; Taaffe, DR; Spry, N; Joseph, D; Hebert, JJ; Newton, RU. Exercise Improves V˙O2max and Body Composition in Androgen Deprivation Therapy-treated Prostate Cancer Patients. Med Sci Sports Exerc. 2017, 49(8), 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Villalobos, A; Chambers, DA. Advancing the science of integrating multiple interventions by blending and bundling. JNCI Cancer Spectr 2023, 7(5), pkad070. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zamboni, M; Mazzali, G; Fantin, F; Rossi, A; Di Francesco, V. Sarcopenic obesity: a new category of obesity in the elderly. Nutr Metab Cardiovasc Dis. 2008, 18(5), 388–95. [Google Scholar] [CrossRef] [PubMed]
- Veen, J; Montiel-Rojas, D; Nilsson, A; Kadi, F. Engagement in Muscle-Strengthening Activities Lowers Sarcopenia Risk in Older Adults Already Adhering to the Aerobic Physical Activity Guidelines. Int J Environ Res Public Health 2021, 18(3), 989. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, JW; Kim, R; Choi, H; Lee, SJ; Bae, GU. Understanding of sarcopenia: from definition to therapeutic strategies. Arch Pharm Res 2021, 44(9-10), 876–889. [Google Scholar] [CrossRef] [PubMed]
- Paris, MT; Bell, KE; Mourtzakis, M. Myokines and adipokines in sarcopenia: understanding cross-talk between skeletal muscle and adipose tissue and the role of exercise. Curr Opin Pharmacol 2020, 52, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Batsis, JA; Villareal, DT. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nat Rev Endocrinol 2018, 14(9), 513–537. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cartwright, MJ; Tchkonia, T; Kirkland, JL. Aging in adipocytes: potential impact of inherent, depot-specific mechanisms. Exp Gerontol 2007, 42(6), 463–71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kimura, Y; Yamada, M; Ohji, S; Ishiyama, D; Nishio, N; Otobe, Y; Koyama, S; Suzuki, M; Ichikawa, T; Ito, D; Maehori, N; Nagae, H. Presence of sarcopenic obesity and evaluation of the associated muscle quality in Japanese older men with prostate cancer undergoing androgen deprivation therapy. J Geriatr Oncol. 2019, 10(5), 835–838. [Google Scholar] [CrossRef] [PubMed]
- Jeromson, S; Gallagher, IJ; Galloway, SD; Hamilton, DL. Omega-3 Fatty Acids and Skeletal Muscle Health. Mar Drugs 2015, 13(11), 6977–7004. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, JH; Jeon, JH; Lee, MJ. Docosahexaenoic Acid, a Potential Treatment for Sarcopenia, Modulates the Ubiquitin-Proteasome and the Autophagy-Lysosome Systems. Nutrients 2020, 12(9), 2597. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Taheri, M; Chilibeck, PD; Cornish, SM. A Brief Narrative Review of the Underlying Mechanisms Whereby Omega-3 Fatty Acids May Influence Skeletal Muscle: From Cell Culture to Human Interventions. Nutrients 2023, 15(13), 2926. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kamolrat, T; Gray, SR. The effect of eicosapentaenoic and docosahexaenoic acid on protein synthesis and breakdown in murine C2C12 myotubes. Biochem Biophys Res Commun. 2013, 432, 593–598. [Google Scholar] [CrossRef]
- Dewey, A; Baughan, C; Dean, T; et al. Eicosapentaenoic acid (EPA, an omega-3 fatty acid from fish oils) for the treatment of cancer cachexia. Cochrane Database Syst Rev 2007, 24(1), CD004597. [Google Scholar] [CrossRef]
- Hamerman, D. Molecular-based therapeutic approaches in treatment of anorexia of aging and cancer cachexia. J Gerontol A Biol Sci Med Sci 2002, 57(8), M511–M518. [Google Scholar] [CrossRef]
- Wang, Y; Wang, Y; Shehzad, Q; Su, Y; Xu, L; Yu, L; Zeng, W; Fang, Z; Wu, G; Wei, W; Jin, Q; Zhang, H; Wang, X. Does omega-3 PUFAs supplementation improve metabolic syndrome and related cardiovascular diseases? A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. Epub. 2024, 64(26), 9455–9482. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y; Liu, R; Chang, M; Wei, W; Guo, Y; Jin, Q; Wang, X. Does omega-3 PUFA-enriched oral nutritional intervention benefit cancer patients receiving chemo (radio) therapy? A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. Epub. 2023, 63(18), 3081–3096. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y; Wang, C. Effect of ω-3 polyunsaturated fatty acid-supplemented parenteral nutrition on inflammatory and immune function in postoperative patients with gastrointestinal malignancy: A meta-analysis of randomized control trials in China. Medicine (Baltimore) 2018, 97(16), e0472. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Guo, Y; Ma, B; Li, X; Hui, H; Zhou, Y; Li, N; Xie, X. n-3 PUFA can reduce IL-6 and TNF levels in patients with cancer. Br J Nutr. 2023, 129(1), 54–65. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y; Meng, Y; Li, N; Wang, Q; Chen, L. The effects of low-ratio n-6/n-3 PUFA on biomarkers of inflammation: a systematic review and meta-analysis. Food Funct 2021, 12(1), 30–40. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J; et al. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J Am Med Dir Assoc 2013, 14, 542–559. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, RR; Miller, SL; Miller, KB. Optimal protein intake in the elderly. Clin Nutr 2008, 27, 675–684. [Google Scholar] [CrossRef]
- Deutz, NE; et al. Protein intake and exercise for optimal muscle function with aging: recommendations from the ESPEN Expert Group. Clin Nutr 2014, 33, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Bouillanne, O; et al. Impact of protein pulse feeding on lean mass in malnourished and at-risk hospitalized elderly patients: a randomized controlled trial. Clin Nutr 2013, 32, 186–192. [Google Scholar] [CrossRef]
- Verreijen, AM; et al. Effect of a high protein diet and/or resistance exercise on the preservation of fat free mass during weight loss in overweight and obese older adults: a randomized controlled trial. Nutr J 2017, 16, 10. [Google Scholar] [CrossRef]
- Hassan-Smith, ZK; et al. 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 exert distinct effects on human skeletal muscle function and gene expression. PLoS ONE 2017, 12, e0170665. [Google Scholar] [CrossRef]
- Li, CW; Yu, K; Shyh-Chang, N; Jiang, Z; Liu, T; Ma, S; Luo, L; Guang, L; Liang, K; Ma, W; Miao, H; Cao, W; Liu, R; Jiang, LJ; Yu, SL; Li, C; Liu, HJ; Xu, LY; Liu, RJ; Zhang, XY; Liu, GS. Pathogenesis of sarcopenia and the relationship with fat mass: descriptive review. J Cachexia Sarcopenia Muscle 2022, 13(2), 781–794. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kalinkovich, A; Livshits, G. Sarcopenic obesity or obese sarcopenia: A cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of pathogenesis. Ageing Res Rev. 2017, 35, 200–221. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B; Dabur, R. Role of Pro-inflammatory Cytokines in Regulation of Skeletal Muscle Metabolism: A Systematic Review. Curr Med Chem. 2020, 27(13), 2161–2188. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z; Zhang, T; Liu, H; Li, Z; Peng, L; Wang, C; Wang, T. Inflammaging: The ground for sarcopenia? Exp Gerontol 2022, 168, 111931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N; Zhai, L; Wong, RMY; Cui, C; Law, SW; Chow, SK; Goodman, SB; Cheung, WH. Harnessing immunomodulation to combat sarcopenia: current insights and possible approaches. Immun Ageing 2024, 21(1), 55. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sears, B. Anti-inflammatory Diets. J Am Coll Nutr. 2015, 34 Suppl 1, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Block, KI; Gyllenhaal, C; Lowe, L; Amedei, A; Amin, AR; Amin, A; Aquilano, K; Arbiser, J; Arreola, A; Arzumanyan, A; Ashraf, SS; Azmi, AS; Benencia, F; Bhakta, D; Bilsland, A; Bishayee, A; Blain, SW; Block, PB; Boosani, CS; Carey, TE; Carnero, A; Carotenuto, M; Casey, SC; Chakrabarti, M; Chaturvedi, R; Chen, GZ; Chen, H; Chen, S; Chen, YC; Choi, BK; Ciriolo, MR; Coley, HM; Collins, AR; Connell, M; Crawford, S; Curran, CS; Dabrosin, C; Damia, G; Dasgupta, S; DeBerardinis, RJ; Decker, WK; Dhawan, P; Diehl, AM; Dong, JT; Dou, QP; Drew, JE; Elkord, E; El-Rayes, B; Feitelson, MA; Felsher, DW; Ferguson, LR; Fimognari, C; Firestone, GL; Frezza, C; Fujii, H; Fuster, MM; Generali, D; Georgakilas, AG; Gieseler, F; Gilbertson, M; Green, MF; Grue, B; Guha, G; Halicka, D; Helferich, WG; Heneberg, P; Hentosh, P; Hirschey, MD; Hofseth, LJ; Holcombe, RF; Honoki, K; Hsu, HY; Huang, GS; Jensen, LD; Jiang, WG; Jones, LW; Karpowicz, PA; Keith, WN; Kerkar, SP; Khan, GN; Khatami, M; Ko, YH; Kucuk, O; Kulathinal, RJ; Kumar, NB; Kwon, BS; Le, A; Lea, MA; Lee, HY; Lichtor, T; Lin, LT; Locasale, JW; Lokeshwar, BL; Longo, VD; Lyssiotis, CA; MacKenzie, KL; Malhotra, M; Marino, M; Martinez-Chantar, ML; Matheu, A; Maxwell, C; McDonnell, E; Meeker, AK; Mehrmohamadi, M; Mehta, K; Michelotti, GA; Mohammad, RM; Mohammed, SI; Morre, DJ; Muralidhar, V; Muqbil, I; Murphy, MP; Nagaraju, GP; Nahta, R; Niccolai, E; Nowsheen, S; Panis, C; Pantano.
- Mohammad, RM; Muqbil, I; Lowe, L; Yedjou, C; Hsu, HY; Lin, LT; Siegelin, MD; Fimognari, C; Kumar, NB; Dou, QP; Yang, H; Samadi, AK; Russo, GL; Spagnuolo, C; Ray, SK; Chakrabarti, M; Morre, JD; Coley, HM; Honoki, K; Fujii, H; Georgakilas, AG; Amedei, A; Niccolai, E; Amin, A; Ashraf, SS; Helferich, WG; Yang, X; Boosani, CS; Guha, G; Bhakta, D; Ciriolo, MR; Aquilano, K; Chen, S; Mohammed, SI; Keith, WN; Bilsland, A; Halicka, D; Nowsheen, S; Azmi, AS. Broad targeting of resistance to apoptosis in cancer. Semin Cancer Biol. 2015, 35 Suppl, S78–103. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kumar, NB. Contemporary Strategies for Clinical Chemoprevention of Localized Prostate Cancer. In Cancer Control; November 2024. [Google Scholar] [CrossRef]
- Kumar, N. B.; Pow-Sang, J.; Spiess, P.; Dickinson, S.; Schell, M. J. A phase II randomized clinical trial using aglycone isoflavones to treat patients with localized prostate cancer in the pre-surgical period prior to radical prostatectomy. Oncotarget 2020, 11, 1218–1234. [Google Scholar] [CrossRef] [PubMed]
- Leone, A; Diorio, G; Sexton, W; Schell, M; Alexandrow, M; Fahey, JW; Kumar, NB. Sulforaphane for the chemoprevention of bladder cancer: molecular mechanism targeted approach. Oncotarget 2017, 8(21), 35412–35424. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kumar, NB. The Promise of Nutrient-Derived Bioactive Compounds and Dietary Components to Ameliorate Symptoms of Chemotherapy-Related Cognitive Impairment in Breast Cancer Survivors. Curr Treat Options Oncol 2021, 22(8), 67. [Google Scholar] [CrossRef] [PubMed]
- Barreca, MM; Alessandro, R; Corrado, C. Effects of Flavonoids on Cancer, Cardiovascular and Neurodegenerative Diseases: Role of NF-κB Signaling Pathway. Int J Mol Sci. 2023, 24(11), 9236. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Prasad, S; Phromnoi, K; Yadav, VR; Chaturvedi, MM; Aggarwal, BB. Targeting inflammatory pathways by flavonoids for prevention and treatment of cancer. Planta Med. 2010, 76(11), 1044–63. [Google Scholar] [CrossRef] [PubMed]
- Al-Khayri, JM; Sahana, GR; Nagella, P; Joseph, BV; Alessa, FM; Al-Mssallem, MQ. Flavonoids as Potential Anti-Inflammatory Molecules: A Review. Molecules 2022, 27(9), 2901. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Serafini, M; Peluso, I; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc Nutr Soc. 2010, 69(3), 273–8. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A; Munir, S; Badshah, SL; Khan, N; Ghani, L; Poulson, BG; Emwas, AH; Jaremko, M. Important Flavonoids and Their Role as a Therapeutic Agent. Molecules 2020, 25(22), 5243. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saleh, HA; Yousef, MH; Abdelnaser, A. The Anti-Inflammatory Properties of Phytochemicals and Their Effects on Epigenetic Mechanisms Involved in TLR4/NF-κB-Mediated Inflammation. Front Immunol 2021, 12, 606069. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Laurindo, LF; Santos, AROD; Carvalho, ACA; Bechara, MD; Guiguer, EL; Goulart, RA; Vargas Sinatora, R; Araújo, AC; Barbalho, SM. Phytochemicals and Regulation of NF-kB in Inflammatory Bowel Diseases: An Overview of In Vitro and In Vivo Effects. Metabolites 2023, 13(1), 96. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Connors, SK; Chornokur, G; Kumar, NB. New insights into the mechanisms of green tea catechins in the chemoprevention of prostate cancer. Nutr Cancer 2012, 64(1), 4–22. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kumar, NB; Pow-Sang, J; Egan, KM; Spiess, PE; Dickinson, S; Salup, R; Helal, M; McLarty, J; Williams, CR; Schreiber, F; Parnes, HL; Sebti, S; Kazi, A; Kang, L; Quinn, G; Smith, T; Yue, B; Diaz, K; Chornokur, G; Crocker, T; Schell, MJ. Randomized, Placebo-Controlled Trial of Green Tea Catechins for Prostate Cancer Prevention. Cancer Prev Res (Phila) 2015, 8(10), 879–87. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, SJ; Amankwah, E; Connors, S; Park, HY; Rincon, M; Cornnell, H; Chornokur, G; Hashim, AI; Choi, J; Tsai, YY; Engelman, RW; Kumar, N; Park, JY. Safety and chemopreventive effect of Polyphenon E in preventing early and metastatic progression of prostate cancer in TRAMP mice. Cancer Prev Res (Phila) 2014, 7(4), 435–44. [Google Scholar] [CrossRef]
- Kumar, NB; Pow-Sang, J; Spiess, PE; Park, J; Salup, R; Williams, CR; Parnes, H; Schell, MJ. Randomized, placebo-controlled trial evaluating the safety of one-year administration of green tea catechins. Oncotarget 2016, 7(43), 70794–70802. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maphetu, N; Unuofin, JO; Masuku, NP; Olisah, C; Lebelo, SL. Medicinal uses, pharmacological activities, phytochemistry, and the molecular mechanisms of Punica granatum L. (pomegranate) plant extracts: A review. Biomed Pharmacother 2022, 153, 113256. [Google Scholar] [CrossRef] [PubMed]
- Cordiano, R; Gammeri, L; Di Salvo, E; Gangemi, S; Minciullo, PL. Pomegranate (Punica granatum L.) Extract Effects on Inflammaging. Molecules 2024, 29(17), 4174. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hong, MY; Seeram, NP; Heber, D. Pomegranate polyphenols down-regulate expression of androgen-synthesizing genes in human prostate cancer cells overexpressing the androgen receptor. J Nutr Biochem 2008, 19, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, FH; Li, Y. Indole-3-carbinol and prostate cancer. J Nutr. 2004, 134((12) Suppl, 3493S–3498S. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P; Kim, JS. Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules 2018, 23(11), 2983. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Joshi, P; Verma, K; Kumar Semwal, D; Dwivedi, J; Sharma, S. Mechanism insights of curcumin and its analogues in cancer: An update. Phytother Res. 2023, 37(12), 5435–5463. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, AB; Bordoloi, D; Harsha, C; Banik, K; Gupta, SC; Aggarwal, BB. Curcumin mediates anticancer effects by modulating multiple cell signaling pathways. Clin Sci (Lond) 2017, 131(15), 1781–1799. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, PMP; Rodrigues, LARL; de Alencar Carnib, LP; de Lima Sousa, PV; Nolasco Lugo, LM; Nunes, NMF; do Nascimento Silva, J; da Silva Araûjo, L; de Macêdo Gonçalves Frota, K. Cruciferous Vegetables as Antioxidative, Chemopreventive and Antineoplasic Functional Foods: Preclinical and Clinical Evidences of Sulforaphane Against Prostate Cancers. Curr Pharm Des. 2018, 24(40), 4779–4793. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, TL; Saha, S; Bernuzzi, F; Savva, GM; Troncoso-Rey, P; Traka, MH; Mills, RD; Ball, RY; Mithen, RF. Accumulation of Sulforaphane and Alliin in Human Prostate Tissue. Nutrients 2022, 14(16), 3263. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mattson, MP; Longo, VD; Harvie, M. Impact of intermittent fasting on health and disease processes. Ageing Res Rev. 2017, 39, 46–58. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Longo, VD; Panda, S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metab 2016, 23(6), 1048–1059. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Brandhorst, S.; Choi, I.Y.; Wei, M.; Cheng, C.W.; Sedrakyan, S.; Navarrete, G.; Dubeau, L.; Yap, L.P.; Park, R.; Vinciguerra, M.; et al. APeriodic Diet that Mimics Fasting Promotes Multi-System Regeneration, Enhanced Cognitive Performance, and Healthspan. Cell Metab. 2015, 22, 86–99. [Google Scholar] [CrossRef]
- Wei, M.; Brandhorst, S.; Shelehchi, M.; Mirzaei, H.; Cheng, C.W.; Budniak, J.; Groshen, S.; Mack, W.J.; Guen, E.; Di Biase, S.; et al. Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef]
- Gill, S; Panda, S. A Smartphone App Reveals Erratic Diurnal Eating Patterns in Humans that Can Be Modulated for Health Benefits. Cell Metab 2015, 22(5), 789–98. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Marinac, CR; Nelson, SH; Breen, CI; Hartman, SJ; Natarajan, L; Pierce, JP; Flatt, SW; Sears, DD; Patterson, RE. Prolonged Nightly Fasting and Breast Cancer Prognosis. JAMA Oncol 2016, 2(8), 1049–55. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Caffa, I; Spagnolo, V; Vernieri, C; Valdemarin, F; Becherini, P; Wei, M; Brandhorst, S; Zucal, C; Driehuis, E; Ferrando, L; Piacente, F; Tagliafico, A; Cilli, M; Mastracci, L; Vellone, VG; Piazza, S; Cremonini, AL; Gradaschi, R; Mantero, C; Passalacqua, M; Ballestrero, A; Zoppoli, G; Cea, M; Arrighi, A; Odetti, P; Monacelli, F; Salvadori, G; Cortellino, S; Clevers, H; De Braud, F; Sukkar, SG; Provenzani, A; Longo, VD; Nencioni, A. Fasting-mimicking diet and hormone therapy induce breast cancer regression. Nature Erratum in: Nature. 2020 Dec;588(7839):E33. doi: 10.1038/s41586-020-2957-6. PMID: 32669709; PMCID: PMC7881940. 2020, 583(7817), 620–624. [Google Scholar] [CrossRef] [PubMed]
- Galvão, DA; Taaffe, DR; Newton, RU. Body weight, fat mass and metabolic complications during androgen deprivation therapy: should urologists recommend exercise and diet to help patients overcome toxicities? Prostate Cancer Prostatic Dis 2021, 24(3), 591–593. [Google Scholar] [CrossRef] [PubMed]
- Rejeski, K; Cordas Dos Santos, DM; Parker, NH; Bücklein, VL; Winkelmann, M; Jhaveri, KS; Liu, L; Trinkner, P; Günther, S; Karschnia, P; Blumenberg, V; Schmidt, C; Kunz, WG; von Bergwelt-Baildon, M; Jain, MD; Theurich, S; Subklewe, M. Influence of Adipose Tissue Distribution, Sarcopenia, and Nutritional Status on Clinical Outcomes After CD19 CAR T-cell Therapy. Cancer Immunol Res. 2023, 11(6), 707–719. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Parker, Nathan; Bean, Morgan; De Vera, Mart Theodore; Carmella, Rachel; McCoy, Jessica; Corallo, Salvatore; Delgado, Luis Cuadrado; Menges, Meghan; Castaneda, Omar; Chavez, Julio C.; Faramand, Rawan; Jain, Michael D.; Jim, Heather S.L.; Lazaryan, Aleksandr; Mirza, Sayeef; Oswald, Laura B.; Shah, Bijal D; Locke, Frederick L.; Freeman, Ciara Louise. Exercise Prehabilitation for Older Adults Undergoing CAR-T: Feasibility and Preliminary Outcomes from a Single-Arm Pilot Study. Blood 2024, 144 (Supplement 1), 7224. [Google Scholar] [CrossRef]
- Ngo-Huang, AT; Parker, NH; Xiao, L; Schadler, KL; Petzel, MQB; Prakash, LR; Kim, MP; Tzeng, CD; Lee, JE; Ikoma, N; Wolff, RA; Javle, MM; Koay, EJ; Pant, SD; Folloder, JP; Wang, X; Cotto, AM; Ju, YR; Garg, N; Wang, H; Bruera, ED; Basen-Engquist, KM; Katz, MHG. Effects of a Pragmatic Home-based Exercise Program Concurrent With Neoadjuvant Therapy on Physical Function of Patients With Pancreatic Cancer: The PancFit Randomized Clinical Trial. Ann Surg. 2023, 278(1), 22–30. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kurz, E; Hirsch, CA; Dalton, T; Shadaloey, SA; Khodadadi-Jamayran, A; Miller, G; Pareek, S; Rajaei, H; Mohindroo, C; Baydogan, S; Ngo-Huang, A; Parker, N; Katz, MHG; Petzel, M; Vucic, E; McAllister, F; Schadler, K; Winograd, R; Bar-Sagi, D. Exercise-induced engagement of the IL-15/IL-15Rα axis promotes anti-tumor immunity in pancreatic cancer. Cancer Cell 2022, 40(7), 720–737.e5. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Parker, NH; Arlinghaus, KR; Johnston, CA. Integrating Physical Activity Into Clinical Cancer Care. Am J Lifestyle Med 2018, 12(3), 220–223. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kumar, NB; Bahl, S; Lemay, D; Dhillon, J; Poch, M; Manley, B; Li, R; Pow-Sang, J; Yu, A; Whiting, J; Schell, MJ. Nutritional intake of ω-3 fatty acid intake and clinical grade of prostate cancer. Cancer Epidemiol 2025, 99, 102959. [Google Scholar] [CrossRef] [PubMed]
- Prokopidis, K; Cervo, MM; Gandham, A; Scott, D. Impact of Protein Intake in Older Adults with Sarcopenia and Obesity: A Gut Microbiota Perspective. Nutrients 2020, 12(8), 2285. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, M; Ren, F; Zhou, Y; He, Y; Du, T; Tan, Y. Age-related sarcopenia and altered gut microbiota: A systematic review. Microb Pathog 2024, 195, 106850. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y; de Crom, TOE; Chen, Z; Benz, E; van der Schaft, N; Pinel, A; Boirie, Y; Eglseer, D; Topinkova, E; Schoufour, JD; Weijs, PJM; Rivadeneira, F; Boer, CG; van Meurs, JBJ; Voortman, T. Dietary protein intake and body composition, sarcopenia and sarcopenic obesity: A prospective population-based study. Clin Nutr. 2025, 53, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Daniell, HW. Osteoporosis due to androgen deprivation therapy in men with prostate cancer. Urology 2001, 58((2) Suppl 1, 101–7. [Google Scholar] [CrossRef] [PubMed]
- Jones, JM; Tsang, DS; Zheng, S; Yeheskel, A; Catton, CN; Cheung, AM; Hamilton, R; Alibhai, SMH. Implementing and Evaluating the Impact of BoneRx: A Healthy Bone Prescription for Men with Prostate Cancer Initiating Androgen Deprivation Therapy. J Clin Med. 2022, 11(10), 2703. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).