Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Structural inadequacy: Simple mesothelial apposition (fusion) alone cannot account for the highly organized multilayered (lamellar) architecture consistently observed in high-resolution micro-anatomical studies.
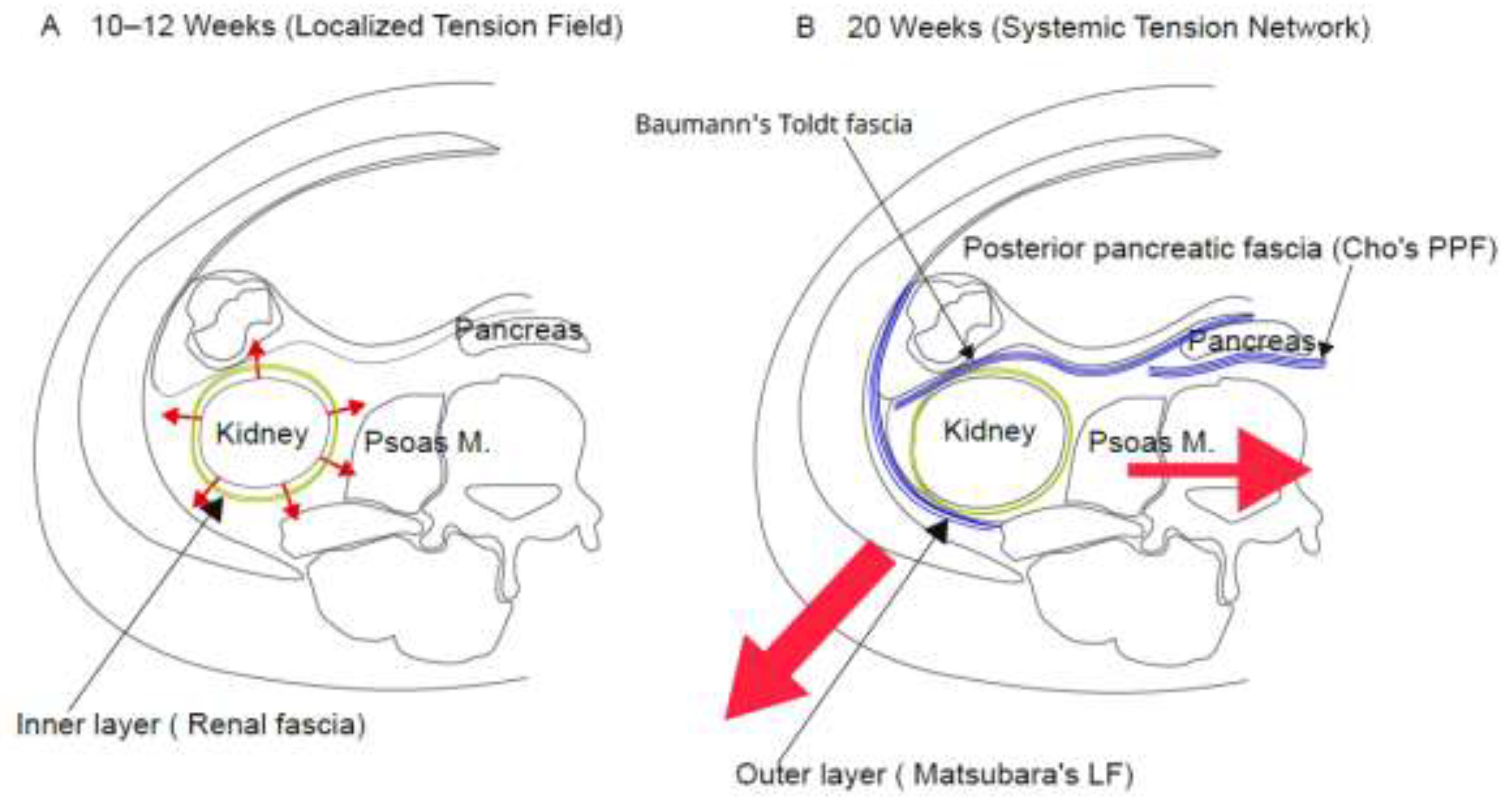
- Temporal paradox: Classical models assume that fascial lamination occurs concurrently with visceral fixation. However, a pronounced temporal lag exists. In the posterior pancreatic region, early visceral fixation (gestational week 10) precedes definitive fascial lamination by 10 weeks (Cho et al., 2009). Similarly, in the renal region, anatomical fixation (completion of ascent) occurs by weeks 7–9, followed by the formation of the organ-dependent inner fascial layer at weeks 10–12. However, the definitive multi-layered outer lamination is delayed until the week-20 threshold (Matsubara et al., 2009). This 10-week developmental lag represents the central paradox that classical descriptive embryology cannot explain. This asynchronous development suggests that definitive outer lamination is governed not by initial organ fixation, but by a later, systemic trigger.
- Clinical contradiction: Classical gross anatomy suggested that the renal fascia is entirely absent when the kidney is missing (Tobin, 1944), whereas modern cross-sectional imaging clearly demonstrates a bilayered posterior renal fascia in the normal state (Raptopoulos et al., 1986). The behavior of connective tissue meshwork in the absence of the principal organ remains an unresolved question.
2. Methods: Conceptual and Analytical Approach
3. Results
3.1. Radiological Subtraction Experiment (Adult Renal Absence)
3.2. Temporal Integration of Fetal Fascial Development
4. Theoretical Integration and Discussion
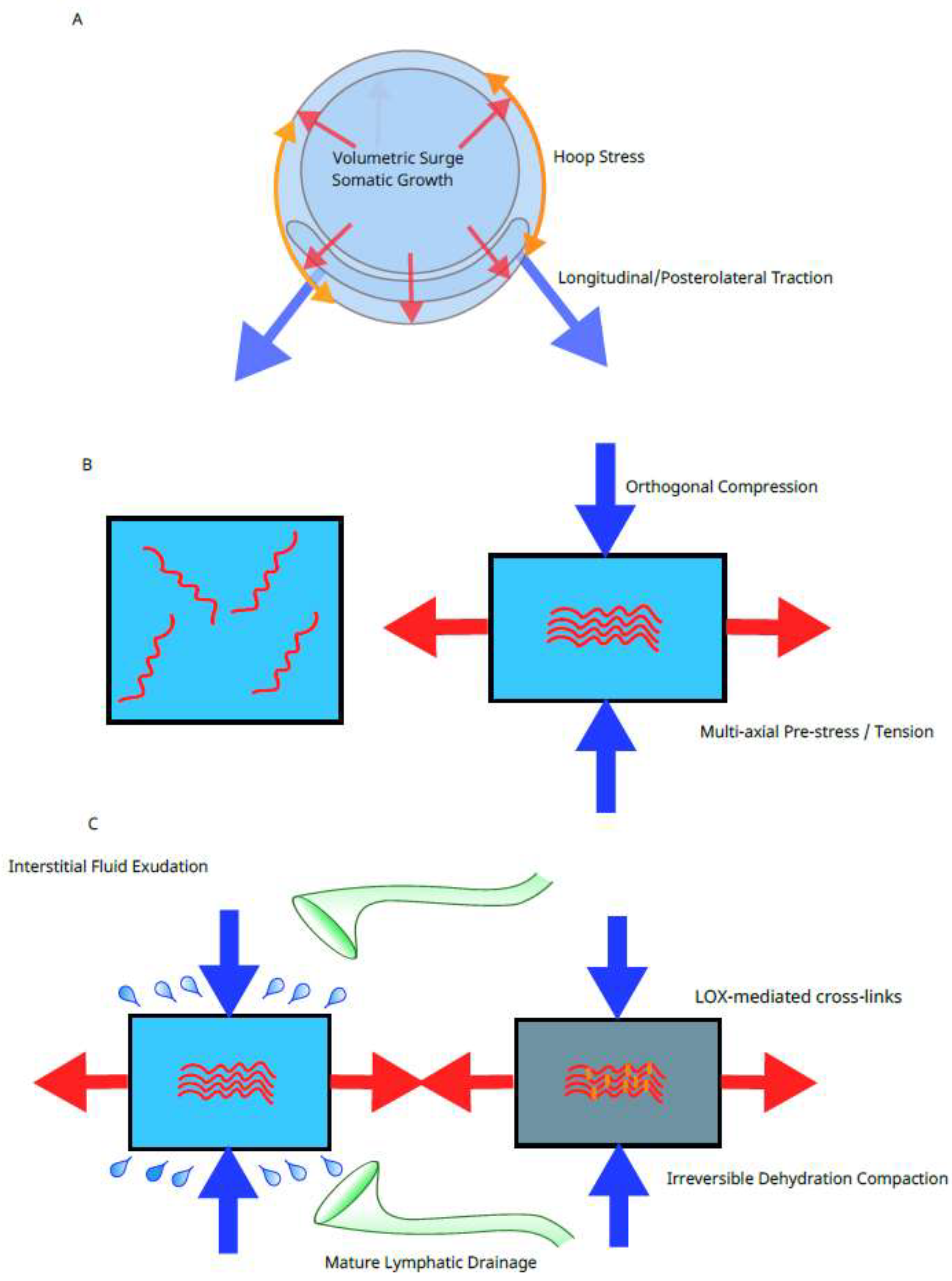
4.1. Mechanobiological Interpretation of the Week-20 Inflection Point
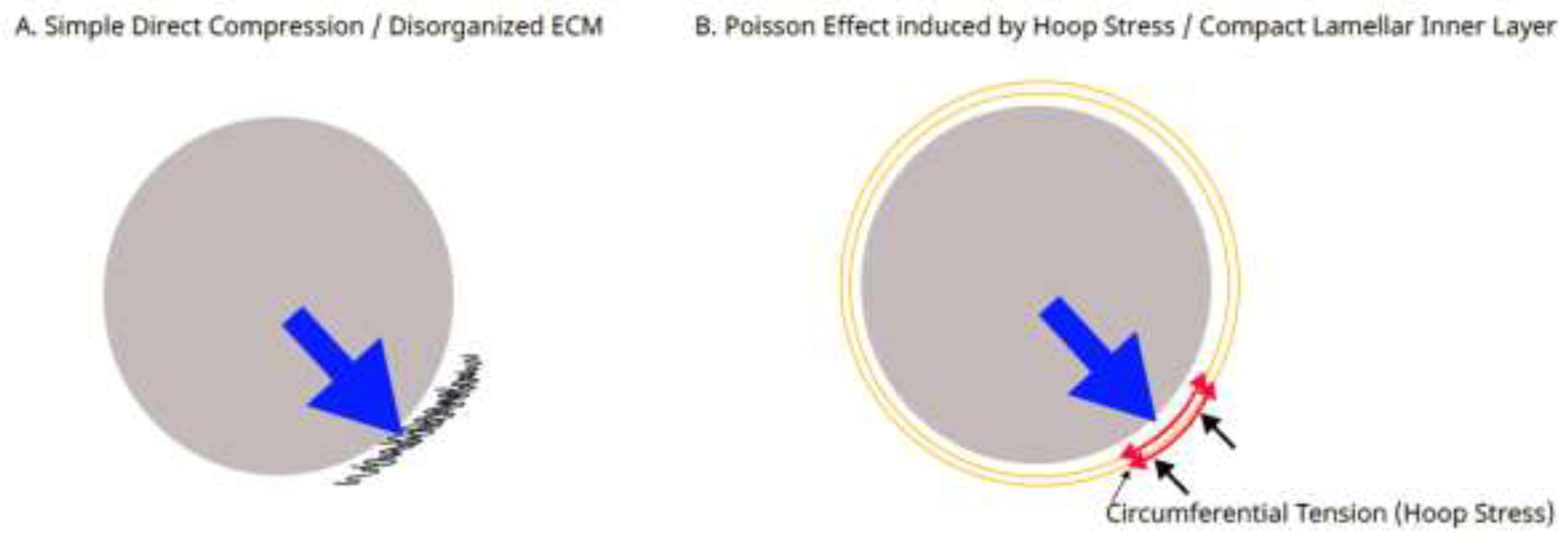
4.2. Systemic Tension Field and Poisson Effect-Driven Lamination
4.3. Terminology and Clinical Relevance
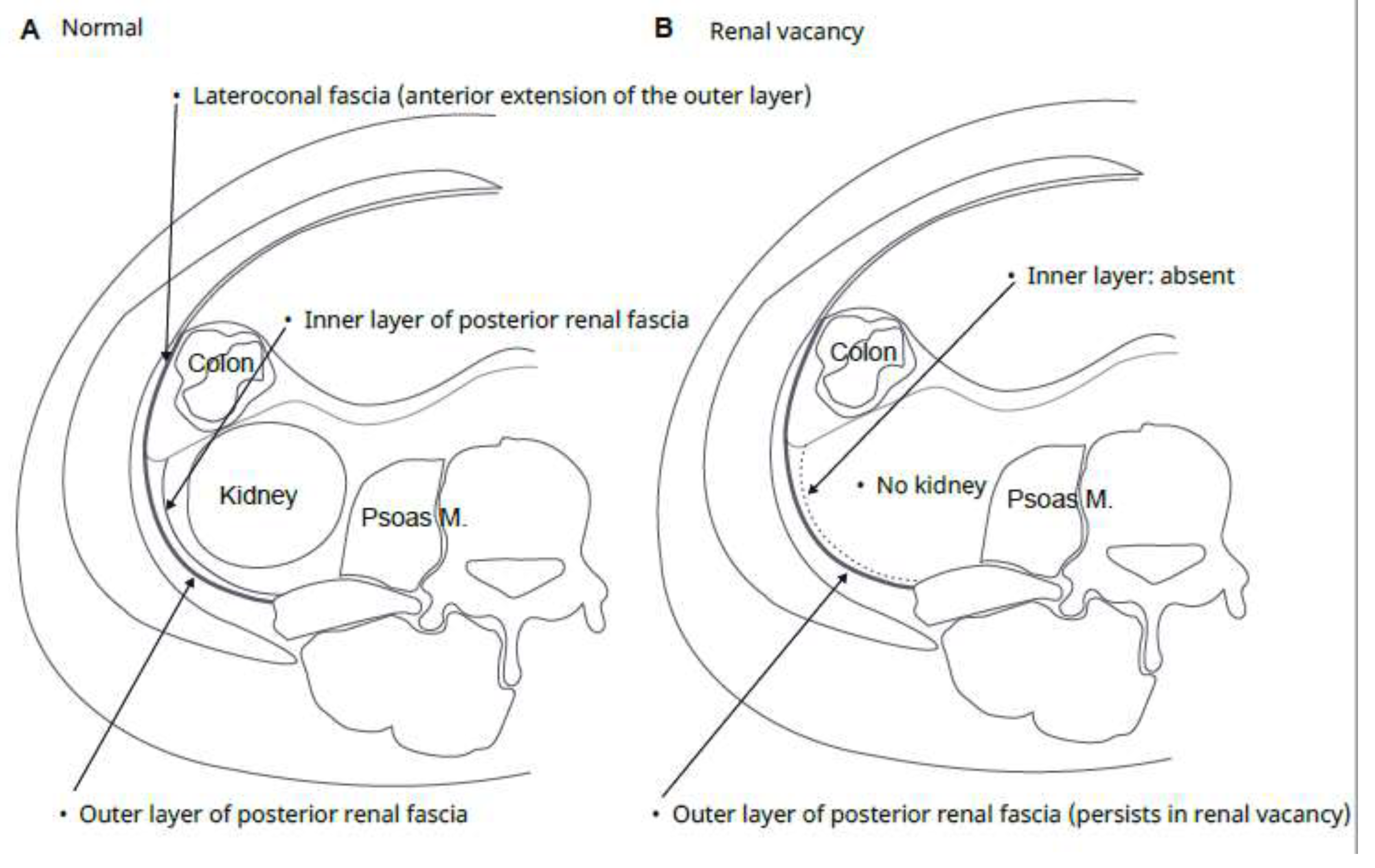
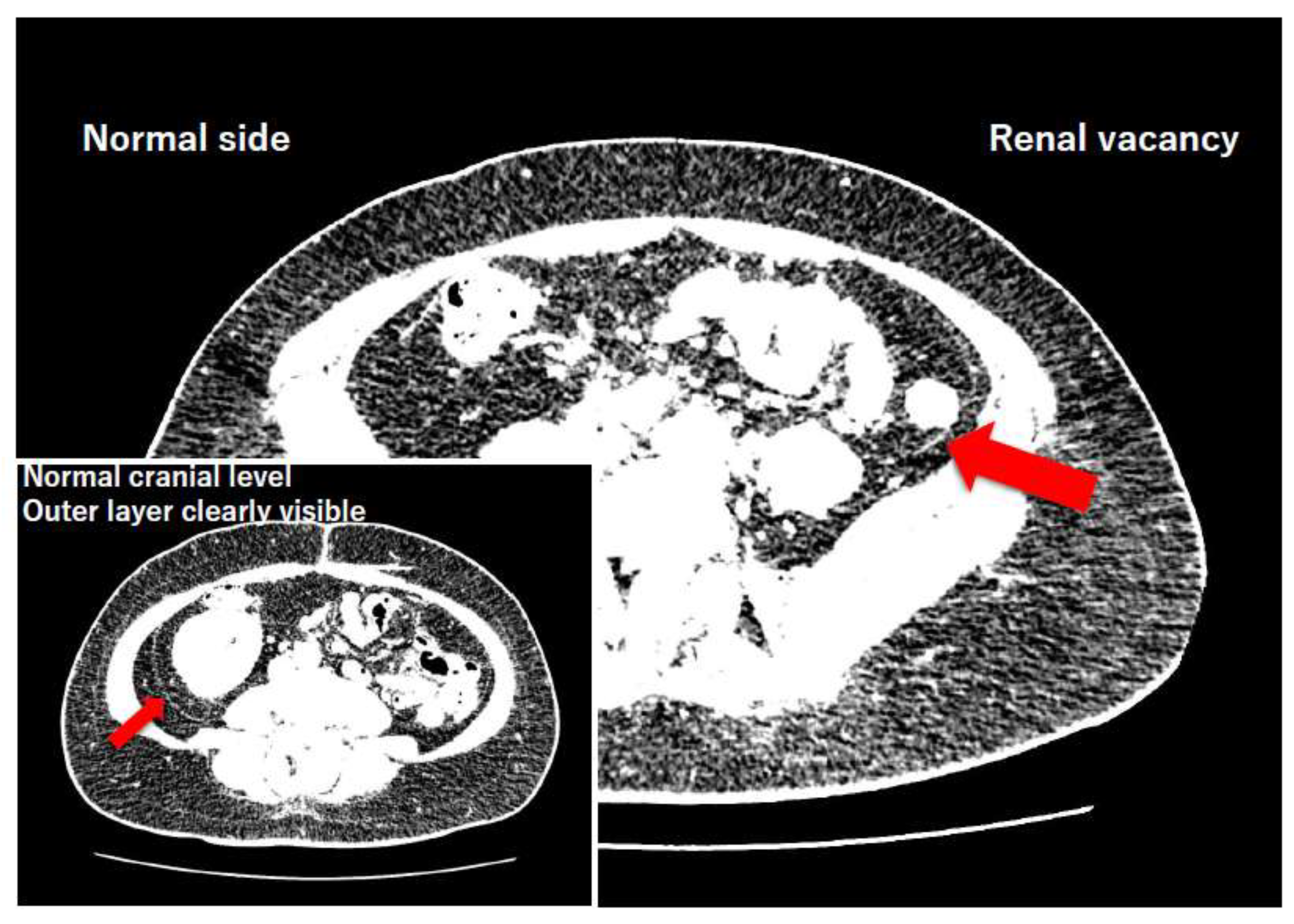
4.4. Resolution of the Historical Controversy Regarding the Bilaminar Renal Fascia
4.5. The Square-Cube Law and the Emergence of a Fetal Tensegrity-like System
4.5.1. Embryological Prerequisites Derived from Pelvic Morphogenesis
4.6. Lymphatic Maturation, Poroelastic Consolidation, and Cross-Linking
4.7. Implications for Comparative Anatomy
4.8. Limitations and Future Directions
5. Conclusion
| Gestational Age | Anatomical/Biomechanical Event | Mechanobiological Significance | Key References |
|---|---|---|---|
| Weeks 10–12 | • Early pancreatic fixation to the posterior wall • Circumferential appearance of the inner renal fascial layer • Morphogenetic blueprint of iliac flare geometry established via cartilaginous migration | Local tension and spatial vector priming: viscera are asynchronously integrated into the nascent tension network. Pelvic cartilage establishes future posterolateral traction vectors but remains too compliant to generate systemic tension. | Cho et al. (2009); Matsubara et al. (2009); Senevirathne et al. (2025) |
| Weeks 14–16 | • Functional maturation of the fetal lymphatic system (connection to terminal veins) | Physiological preparation: systemic drainage capacity required for poroelastic fluid exudation during subsequent Poisson effect compression is established. | Berger (1999); Bekker et al. (2005) |
| Weeks 18–20 | • Progressive vertebral ossification • Epidermal keratinization and completion of the inextensible cutaneous envelope • Onset of exponential volumetric growth (square-cube law) | Dynamic priming and the square-cube law: the vertebral column stiffens into a central pillar; volumetric growth outpaces surface area, generating immense outward pressure that collides with the maturing epidermis to produce powerful whole-body hoop stress. | Bagnall et al. (1977); Singh & Archana (2008); Hadlock et al. (1991) |
| ~Week 20 (temporal nexus) | • Trunk stiffening: cumulative 3D pelvic expansion and ossification reach a mechanical threshold • Fascial lamination: multilayered architecture of the Toldt fascia, posterior pancreatic fascia, and outer posterior renal fascia simultaneously becomes definitive | Temporal and mechanical trigger with Poisson effect: once anchors stiffen into rigid levers, growth forces are transmitted rather than dissipated. The resulting tension spike induces orthogonal compression (Poisson effect), forcing fluid exudation and subsequent LOX cross-linking that completes fascial lamination. | Baumann (1945); Cho et al. (2009); Matsubara et al. (2009); Verbruggen & Nowlan (2017); (present model) |
| Anatomical System | Event at ~Week 20 | Biomechanical Significance | Key References |
|---|---|---|---|
| Skeletal system | Vertebral ossification | Rigid central pillar for tension transmission | Bagnall et al. (1977) |
| Iliac flare expansion/ossification | Establishment of lateral levers for systemic tension | Baumgart et al. (2018); Senevirathne et al. (2025) | |
| Cutaneous envelope | Epidermal keratinization | Inextensible shell generating whole-body hoop stress | Hardman et al. (1999) |
| Trunk growth | Exponential volumetric increase | Square-cube law drives systemic tension | Hadlock et al. (1991) |
| Musculoskeletal movement | Intensification of fetal kicking and gross body movement | Dynamic tension spikes actively align fibroblasts and accelerate poroelastic consolidation | Nowlan (2015); de Vries et al. (1982); Patrick et al. (1982) |
| Respiratory physiology | Surge in fetal breathing movements (FBM) | Rhythmic loading reinforces tension network | Nowlan (2015) |
| Increasing thoracic rigidity | Thorax becomes a stable mechanical frame | Bagnall et al. (1977); Verbruggen & Nowlan (2017) | |
| Onset of surfactant production | Alveolar stabilization → stable FBM | Avery & Fletcher (1974); Clements (1957) | |
| Lymphatic system | Maturation of whole-body drainage capacity | Facilitates irreversible poroelastic consolidation | Bekker et al. (2005) |
| Fascial structures | Lamination of the Toldt fascia | Reflects systemic tension threshold | Baumann (1945) |
| Lamination of the posterior pancreatic fascia | Occurs after the 10-week lag | Cho et al. (2009) | |
| Lamination of the outer posterior renal fascia | Multiaxial tension + Poisson effect compression | Matsubara et al. (2009) | |
| Peripheral nervous system | Compaction of the sciatic nerve sheath | Pelvic traction induces Poisson compression | Pummi et al. (2004) |
| Case | Age/Sex | Radiological Diagnosis | Adrenal Morphology | Fascial Thickness: Affected Side | Fascial Thickness: Normal Side | Difference (Δ) |
|---|---|---|---|---|---|---|
| 1 | 53F | True left renal agenesis | “Pancake” (recumbent) | 1.49 mm | 1.88 mm | −0.39 mm |
| 2 | 47F | Severe left renal dysplasia/involution (renal remnant) | Normal | 1.46 mm | 1.82 mm | −0.36 mm |
| 3 | 89M | True left renal agenesis | “Pancake” (recumbent) | 1.62 mm | Excluded* | N/A |
| Mean | 1.52 mm | 1.85 mm | −0.38 mm |
Author Contributions
Data Availability
Ethics Statement
Acknowledgments
Conflicts of Interest Statement
References
- Avery, M.E.; Fletcher, B.D. The Lung and Its Disorders in the Newborn Infant, 3rd ed.; WB Saunders: Philadelphia, 1974. [Google Scholar]
- Bagnall, K.M.; Harris, P.F.; Jones, P.R. The appearance of ossification centers in the human fetal spine. J Anat 1977, 124, 791–802. [Google Scholar] [PubMed]
- Baumann, J.A. Développement et anatomie de la loge rénale chez l’homme. Acta Anat (Basel) 1945, 1, 15–65. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, M.; Wiśniewski, M.; Grzonkowska, M.; Badura, M.; Biernacki, M.; Siedlecki, Z.; et al. Quantitative anatomy of the ilium’s primary ossification center in the human fetus. Surg Radiol Anat 2018, 40, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Bekker, M.N.; van den Akker, N.M.; de Ruiter, M.C.; Gittenberger-de Groot, A.C. The human fetal lymphatic system: A morphological study. Anat Embryol (Berl) 2005, 210, 167–175. [Google Scholar]
- Berger, G. First-trimester nuchal translucency and the fetal lymphatic system. Ultrasound Obstet Gynecol 1999, 13, 12–17. [Google Scholar]
- Carlson, B.M. Human Embryology and Developmental Biology, 5th ed.; Elsevier: Philadelphia, 2014. [Google Scholar]
- Chau, Y.Y.; Bandiera, R.; Serrels, A.; Saunders, P.T.K.; Hastie, N.D. Visceral adipose tissue formation depends on mesothelial cells. Development 2014, 141, 2752–2762. [Google Scholar]
- Cho, B.H.; Kimura, W.; Song, C.H.; Fujimiya, M.; Murakami, G. Investigation into the embryological development of the fasciae used as the basis for pancreatoduodenal mobilization. J Hepatobiliary Pancreat Surg 2009, 16, 824–831. [Google Scholar] [CrossRef]
- Clements, J.A. Surface tension of lung extracts. Proc Soc Exp Biol Med 1957, 95, 170–172. [Google Scholar] [CrossRef]
- Coffey, J.C.; O’Leary, D.P. The mesentery: Structure, function, and role in disease. Nat Rev Gastroenterol Hepatol 2016, 13, 477–486. [Google Scholar] [CrossRef]
- Congdon, E.D.; Edson, J.N. The cone of renal fascia in the adult white male. Anat Rec 1941, 80, 289–313. [Google Scholar] [CrossRef]
- de Vries, J.I.; Visser, G.H.; Prechtl, H.F. The emergence of fetal behaviour. I. Qualitative aspects. Early Hum Dev 1982, 7, 301–322. [Google Scholar] [CrossRef] [PubMed]
- Exposito, J.Y.; Lethias, C.; Ricard-Blum, F. The fibrillar collagen family. Structure 2010, 18, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Feldberg, M.A.M. Computed Tomography of the Retroperitoneum: An Anatomical and Pathological Atlas with Emphasis on the Fascial Planes; Martinus Nijhoff: Boston, 1983. [Google Scholar]
- Fung, Y.C. Biomechanics: Motion, Flow, Stress, and Growth; Springer-Verlag: New York, 1990. [Google Scholar]
- Gerota, D. Beiträge zur Kenntniss des Befestigungsapparates der Niere. Arch Anat Entwicklungsgesch 1895, 265–285. [Google Scholar]
- Hadlock, F.P.; Harrist, R.B.; Martinez-Poyer, J. In utero analysis of fetal growth: A sonographic weight standard. Radiology 1991, 181, 129–133. [Google Scholar] [CrossRef]
- Han, J.; Lee, J.E.; Jin, J.; Lim, J.S.; Oh, N.; Kim, K.; et al. The origin and development of mesenteric adipose tissue. Cell Metab 2011, 13, 376–388. [Google Scholar]
- Hardman, M.J.; Sisi, P.; Banbury, D.N.; Byrne, C. Barrier formation in the human fetus is patterned. J Invest Dermatol 1999, 113, 1106–1113. [Google Scholar] [CrossRef]
- Hayes, M.A. Abdominopelvic fasciae. Am J Anat 1950, 87, 119–161. [Google Scholar] [CrossRef]
- Humphrey, J.D. Continuum biomechanics of soft biological tissues. Proc Math Phys Eng Sci 2003, 459, 3–46. [Google Scholar] [CrossRef]
- Ingber, D.E. Tensegrity I: Cell structure and hierarchical systems biology. J Cell Sci 2003, 116, 1157–1173. [Google Scholar] [CrossRef]
- Ishikawa, K.; Idoguchi, K.; Tanaka, H.; Tohma, Y.; Ukai, I.; Watanabe, H.; et al. Classification of acute pancreatitis based on retroperitoneal extension: Application of the concept of interfascial planes. Eur J Radiol 2006, 60, 445–452. [Google Scholar] [CrossRef]
- Ishikawa, K.; Nakao, S.; Nakamuro, M.; Huang, T.P.; Nakano, H. The retroperitoneal interfascial planes: Current overview and future perspectives. Acute Med Surg 2016, 3, 219–229. [Google Scholar] [CrossRef]
- Kagan, H.M.; Li, W. Lysyl oxidase: Properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem 2003, 88, 660–672. [Google Scholar] [CrossRef]
- Kinugasa, Y.; Niikura, H.; Murakami, G.; Suzuki, D.; Saito, S.; Tatsumi, H.; et al. Development of the human hypogastric nerve sheath with special reference to the topohistology between the nerve sheath and other prevertebral fascial structures. Clin Anat 2008, 21, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Lakes, R. Deformation mechanisms in negative Poisson’s ratio materials: Structural aspects. J Mater Sci 1991, 26, 2287–2292. [Google Scholar] [CrossRef]
- Marks, S.C., Jr.; Raptopoulos, V.; Kleinman, P.; Snyder, M. The anatomical basis for retrorenal extensions of pancreatic effusions: The role of the renal fasciae. Surg Radiol Anat 1986, 8, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, A.; Kinugasa, Y.; Murakami, G.; Suzuki, D.; Fujimiya, M.; Sugihara, K. Development of the lateroconal fascia in human fetuses. Cells Tissues Organs 2009, 190, 286–296. [Google Scholar] [CrossRef]
- Molmenti, E.P.; Balfe, D.M.; Kanterman, R.Y.; Bennett, H.F. Anatomy of the retroperitoneum: Observations of the distribution of pathologic fluid collections. Radiology 1996, 200, 95–103. [Google Scholar] [CrossRef]
- Mow, V.C.; Kuei, S.C.; Lai, W.M.; Armstrong, C.G. Biphasic creep and stress relaxation of articular cartilage in compression: Theory and experiments. J Biomech Eng 1980, 102, 73–84. [Google Scholar] [CrossRef]
- Nowlan, N.C. Biomechanics of fetal movement. Eur Cell Mater 2015, 29, 1–21. [Google Scholar] [CrossRef]
- Partin, A.W.; Dmochowski, R.R.; Kavoussi, L.R.; Peters, C.A. (Eds.) Campbell-Walsh-Wein Urology, 12th ed.; Elsevier: Philadelphia, 2020. [Google Scholar]
- Patrick, J.; Campbell, K.; Carmichael, L.; Natale, R.; Richardson, B. Patterns of gross fetal body movements over 24-hour observation intervals during the last 10 weeks of pregnancy. Am J Obstet Gynecol 1982, 142, 363–371. [Google Scholar] [CrossRef]
- Provenzano, P.P.; Vanderby, R. Collagen fibril morphology and organization: Implications for force transmission in ligament and tendon. Matrix Biol 2006, 25, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Pummi, K.; Heape, A.M.; Grenell, S.; Peltonen, J.; Peltonen, S. Tight junction proteins ZO-1, occludin, and claudins in developing and adult human perineurium. J Histochem Cytochem 2004, 52, 1037–1046. [Google Scholar] [CrossRef] [PubMed]
- Raptopoulos, V.; Kleinman, P.K.; Marks, S.; Snyder, M.; Silverman, P.M. Renal fascial pathway: Posterior extension of pancreatic effusions within the anterior pararenal space. Radiology 1986, 158, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Senevirathne, G.; Fernandopulle, S.C.; Richard, D.; Baumgart, S.L.; Christensen, A.L.; Fabbri, M.; et al. The evolution of hominin bipedalism in two steps. Nature 2025, 645, 952–963. [Google Scholar] [CrossRef]
- Singh, G.; Archana, G. Unraveling the mystery of vernix caseosa. Indian J Dermatol 2008, 53, 54–60. [Google Scholar] [CrossRef]
- Tobin, C.E. The renal fascia and its relation to the transversalis fascia. Anat Rec 1944, 89, 295–311. [Google Scholar] [CrossRef]
- Toldt, C. Bau und Wachsthumsveränderungen der Gekröse des menschlichen Darmkanales. Denkschr Akad Wiss Wien 1879, 41, 1–56. [Google Scholar]
- Verbruggen, S.W.; Nowlan, N.C. Ontogeny of the human pelvis. Anat Rec (Hoboken) 2017, 300, 643–652. [Google Scholar] [CrossRef]
- Wedel, T.; Heimke, M.; Fletcher, J.; Miskovic, D.; Benz, S.; Stelzner, S.; et al. The retrocolic fascial system revisited for right hemicolectomy with complete mesocolic excision based on anatomical terminology: Do we need the eponyms Toldt, Gerota, Fredet and Treitz? Colorectal Dis 2022, 24, 1612–1622. [Google Scholar] [CrossRef]
- Zuckerkandl, E. Ueber den Fixationsapparat der Nieren. Med Jahrb 1883, 1883, 59–67. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




