Submitted:
23 March 2026
Posted:
24 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Growth Conditions
2.2. Peptide Design, Synthesis, and Preparation
2.3. Determination of Inhibitory Concentrations of 50% (IC50) and 90% (IC90) Values by Dose–Response Analysis
2.4. Fluorescence-Based Assessment of Membrane Permeabilization
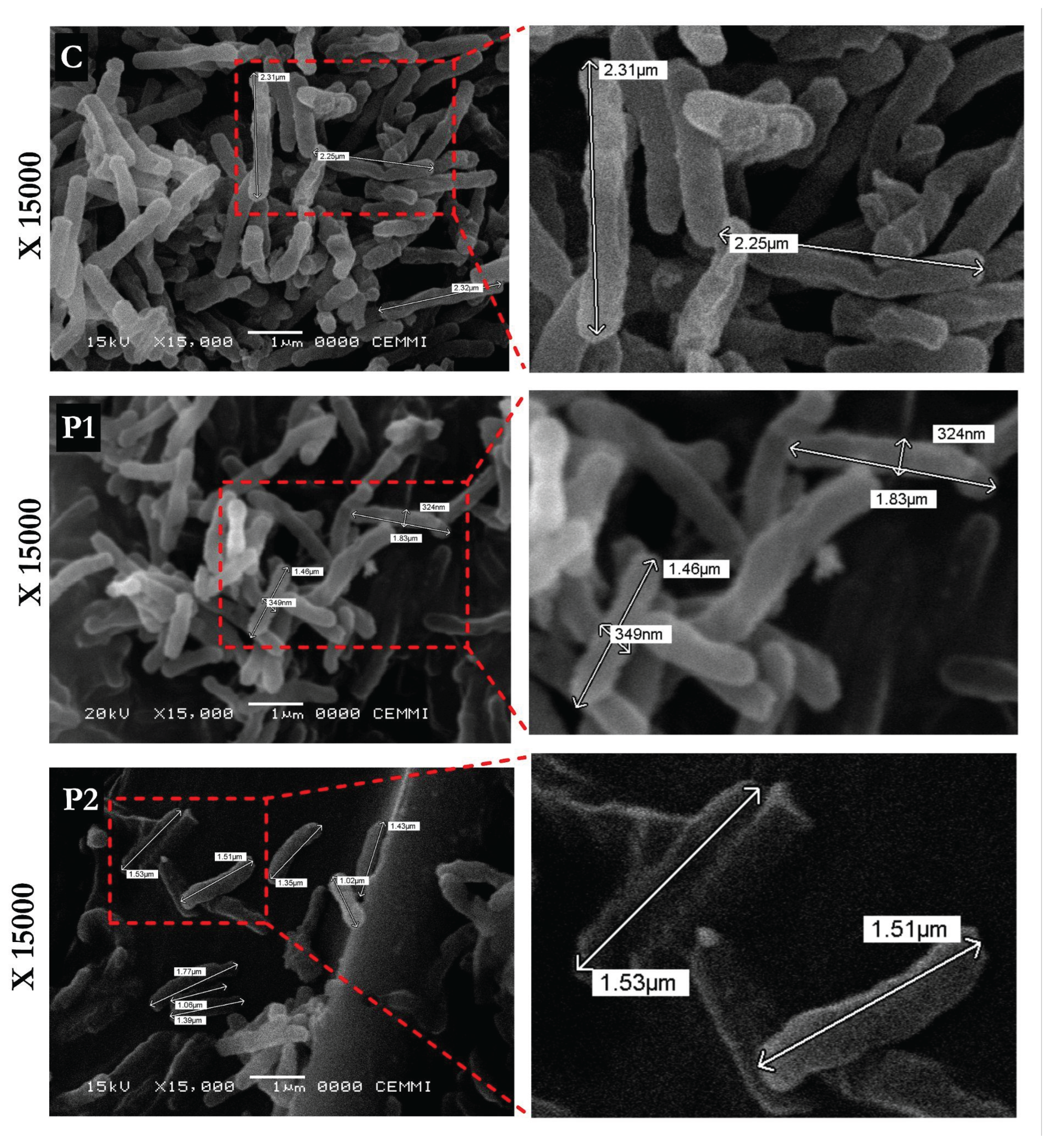
2.5. Scanning Electron Microscopy (SEM) for Evaluation of Peptide-Induced Morphological Alterations
2.6. Quantitative Real-Time PCR (qRT-PCR) Analysis of P-Type ATPase Transcriptional Response
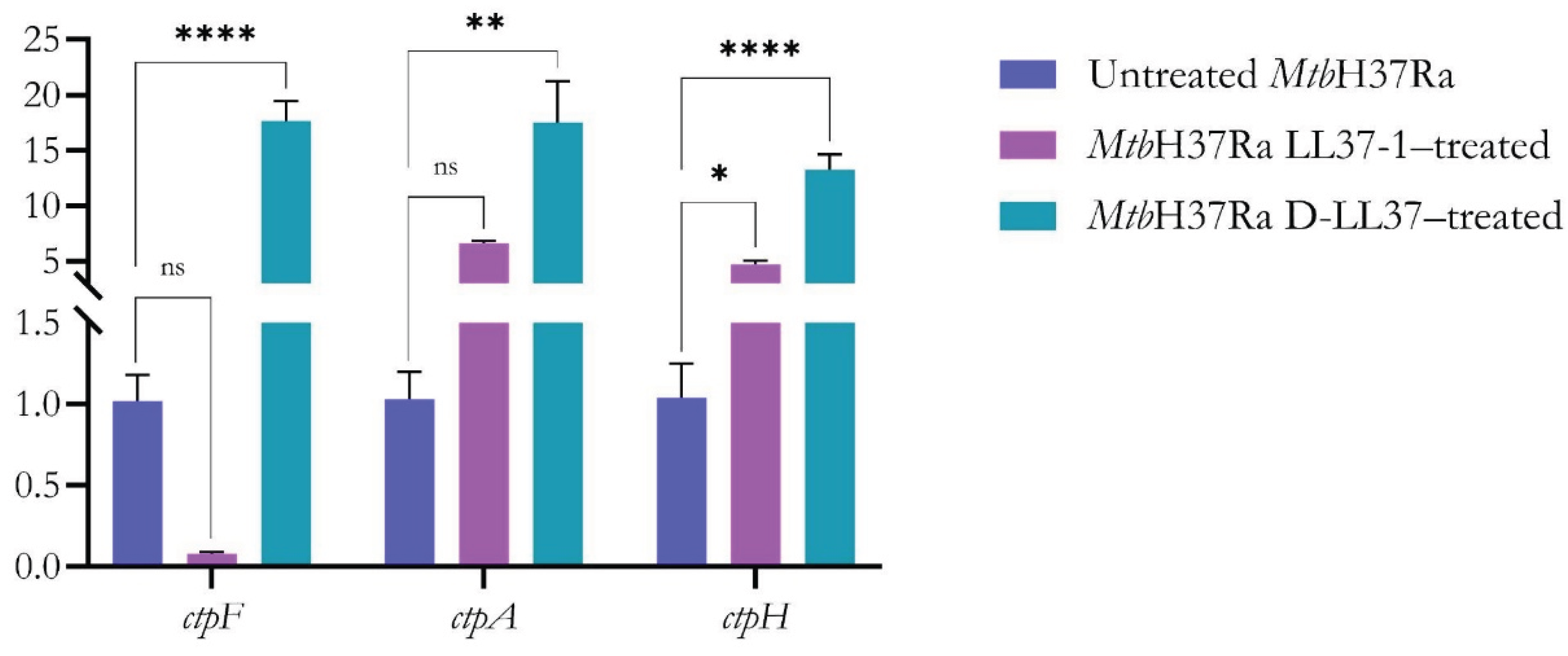
3. Results
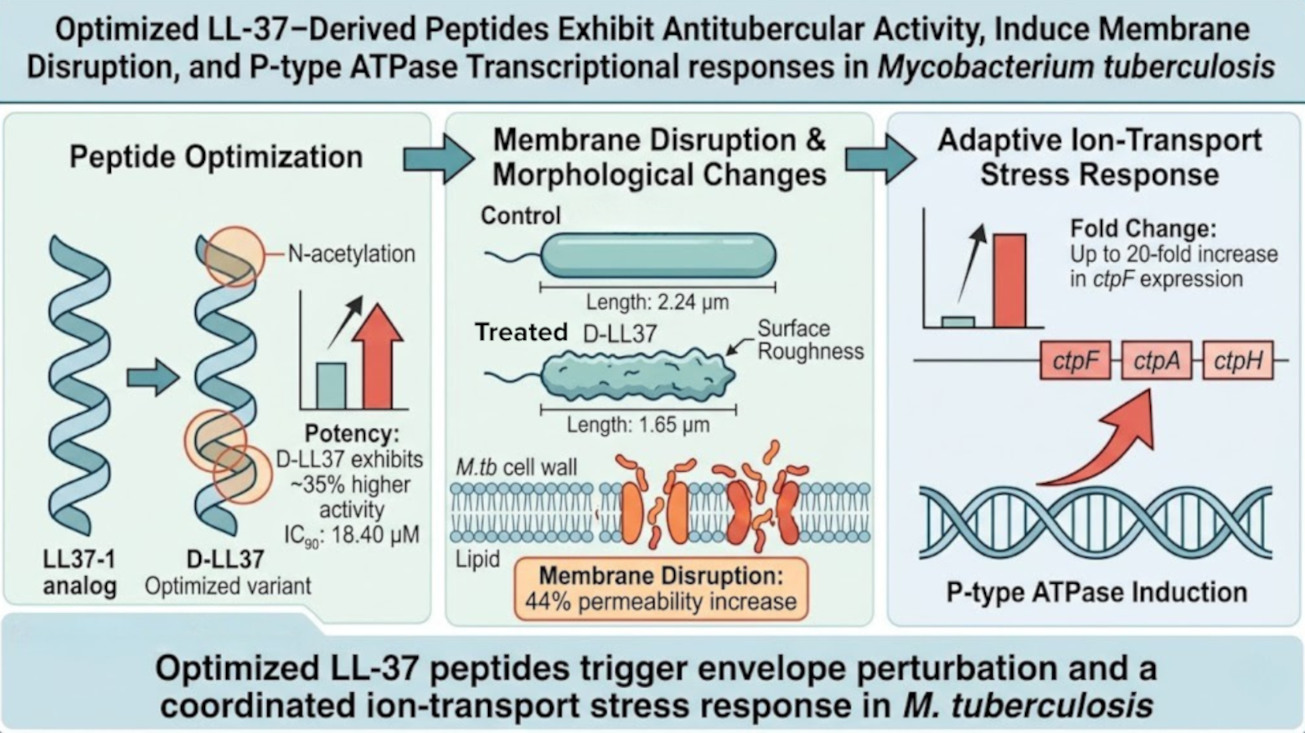
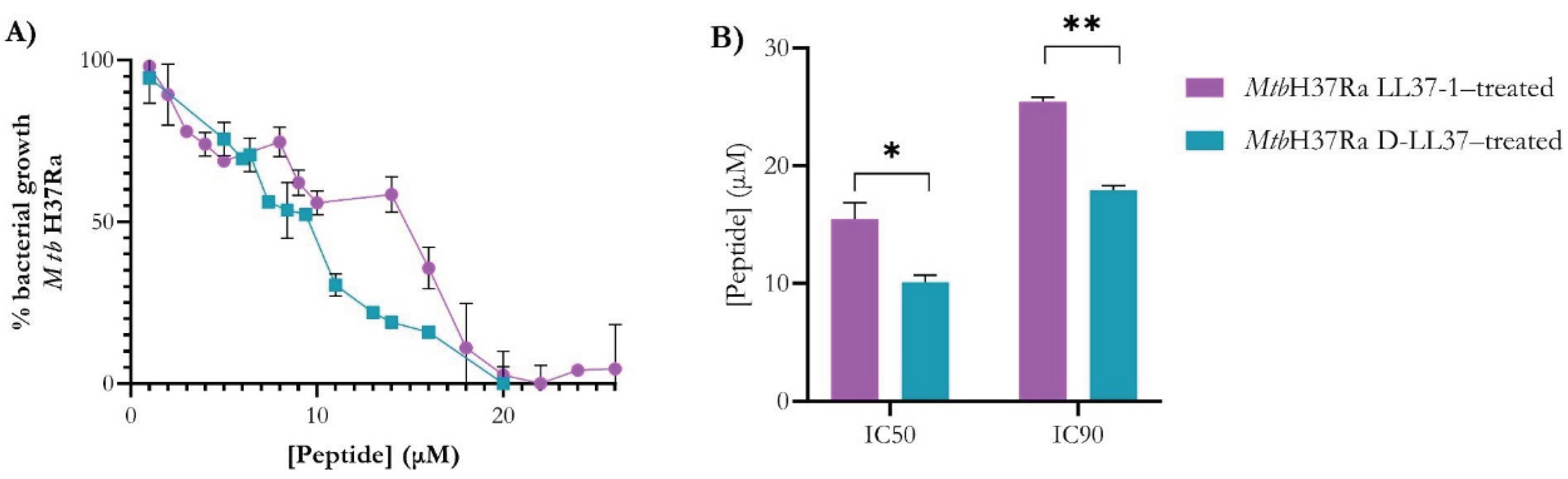
3.1. Dose–Response Analysis Reveals Enhanced Inhibitory Potency of D-LL37
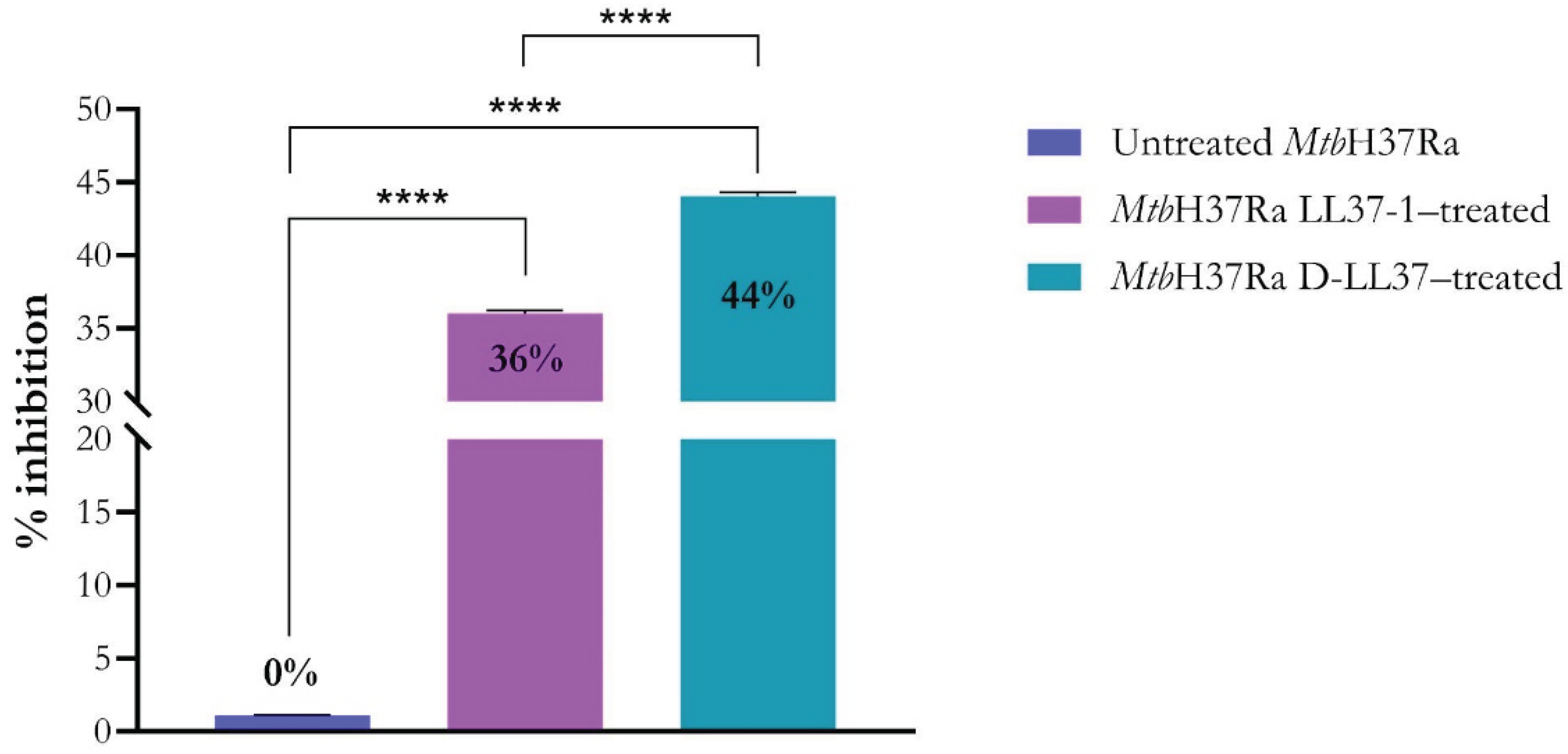
3.2. LL-37–Derived Peptides Induce Membrane Permeabilization in Mtb
3.3. LL-37-Derived Peptides Induce Ultrastructural Alterations in Mtb
3.4. D-LL37 Induces Transcriptional Upregulation of P-Type ATPase Genes in Mtb
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2025; 2025. [Google Scholar]
- Dheda, K.; Gumbo, T.; Maartens, G.; Dooley, K.E.; McNerney, R.; Murray, M.; Furin, J.; Nardell, E.A.; London, L.; Lessem, E.; et al. The Epidemiology, Pathogenesis, Transmission, Diagnosis, and Management of Multidrug-Resistant, Extensively Drug-Resistant, and Incurable Tuberculosis. Lancet Respir. Med. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, D.; Liu, G. Application of Antimicrobial Drugs in Mycobacterium Tuberculosis and Research Progress. Microb. Pathog. 2025, 206. [Google Scholar] [CrossRef] [PubMed]
- Mookherjee, N.; Anderson, M.A.; Haagsman, H.P.; Davidson, D.J. Antimicrobial Host Defence Peptides: Functions and Clinical Potential. Nat. Rev. Drug Discov. 2020, 19. [Google Scholar] [CrossRef] [PubMed]
- Magana, M.; Pushpanathan, M.; Santos, A.L.; Leanse, L.; Fernandez, M.; Ioannidis, A.; Giulianotti, M.A.; Apidianakis, Y.; Bradfute, S.; Ferguson, A.L.; et al. The Value of Antimicrobial Peptides in the Age of Resistance. Lancet Infect. Dis. 2020, 20. [Google Scholar] [CrossRef]
- Dwivedi, M.; Parmar, M.D.; Mukherjee, D.; Yadava, A.; Yadav, H.; Saini, N.P. Biochemistry, Mechanistic Intricacies, and Therapeutic Potential of Antimicrobial Peptides: An Alternative to Traditional Antibiotics. Curr. Med. Chem. 2023, 31. [Google Scholar] [CrossRef]
- Daffé, M.; Marrakchi, H. Unraveling the Structure of the Mycobacterial Envelope. Microbiol. Spectr. 2019, 7. [Google Scholar] [CrossRef]
- Dulberger, C.L.; Rubin, E.J.; Boutte, C.C. The Mycobacterial Cell Envelope — a Moving Target. Nat. Rev. Microbiol. 2020, 18, 47–59. [Google Scholar] [CrossRef]
- Ridyard, K.E.; Overhage, J. The Potential of Human Peptide Ll-37 as an Antimicrobial and Anti-Biofilm Agent. Antibiotics 2021, 10. [Google Scholar] [CrossRef]
- Memariani, H.; Memariani, M. Antibiofilm Properties of Cathelicidin LL-37: An in-Depth Review. World J. Microbiol. Biotechnol. 2023, 39. [Google Scholar] [CrossRef]
- Ito, T.; Matsunaga, N.; Kurashima, M.; Demizu, Y.; Misawa, T. Enhancing Chemical Stability through Structural Modification of Antimicrobial Peptides with Non-Proteinogenic Amino Acids. Antibiotics 2023, 12. [Google Scholar] [CrossRef]
- Alba, M.L.S.; Durán-Rodriguez, A.T.; Pulido, L.M.S.; Escobar-Pérez, J.; Gutiérrez, S.A.; Ospina, J.N.; Bermúdez, G.P.; Molina, L.C.M. Peptides DLL37-1 and LL37-1, an Alternative to Inhibit Biofilm Formation in Clinical Isolates of Staphylococcus Aureus and Staphylococcus Epidermidis. An. Acad. Bras. Cienc. 2022, 94. [Google Scholar] [CrossRef] [PubMed]
- Chingaté, S.; Delgado, G.; Salazar, L.M.; Soto, C.Y. The ATPase Activity of the Mycobacterial Plasma Membrane Is Inhibited by the LL37-Analogous Peptide LLAP. Peptides 2015, 71. [Google Scholar] [CrossRef] [PubMed]
- Novoa-Aponte, L.; Soto Ospina, C.Y. Mycobacterium Tuberculosis P-Type Atpases: Possible Targets for Drug or Vaccine Development. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Maya-Hoyos, M.; Mata-Espinosa, D.; López-Torres, M.O.; Tovar-Vázquez, B.; Barrios-Payán, J.; León-Contreras, J.C.; Ocampo, M.; Hernández-Pando, R.; Soto, C.Y. The ctpF Gene Encoding a Calcium P-Type ATPase of the Plasma Membrane Contributes to Full Virulence of Mycobacterium Tuberculosis. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef]
- López-R, M.; Maya-Hoyos, M.; León-Torres, A.; Cruz-Cacais, A.; Castillo, E.; Soto, C.Y. The Copper P-Type ATPase CtpA Is Involved in the Response of Mycobacterium Tuberculosis to Redox Stress. Biochimie 2024, 221, 137–146. [Google Scholar] [CrossRef]
- Hoyos, M.M. ATPasas Tipo P2 Como Blancos Para La Atenuación de Mycobacterium Tuberculosis; Universidad Nacional de Colombia, 2021. [Google Scholar]
- López-Ruíz, M.; Barrios-Payán, J.; Maya-Hoyos, M.; Hernández-Pando, R.; Ocampo, M.; Soto, C.Y.; Mata-Espinosa, D. The Plasma Membrane P-Type ATPase CtpA Is Required for Mycobacterium Tuberculosis Virulence in Copper-Activated Macrophages in a Mouse Model of Progressive Tuberculosis. Biomedicines 2025, 13, 439. [Google Scholar] [CrossRef]
- A.Varon, H.; Santos, P.; Lopez-Vallejo, F.; Y.Soto, C. Novel Scaffolds Targeting Mycobacterium Tuberculosis Plasma Membrane Ca2+ Transporter CtpF by Structure-Based Strategy. Bioorganic Chem. 2023, 138, 106648. [Google Scholar] [CrossRef]
- Rustad, T.R.; Roberts, D.M.; Liao, R.P.; Sherman, D.R. Isolation of Mycobacterial RNA. Methods Mol. Biol. 2008, 465. [Google Scholar] [CrossRef]
- Pfaffl, M.W.; Tichopad, A.; Prgomet, C.; Neuvians, T.P. Determination of Stable Housekeeping Genes, Differentially Regulated Target Genes and Sample Integrity: BestKeeper – Excel-Based Tool Using Pair-Wise Correlations. Biotechnol. Lett. 2004, 26, 509–515. [Google Scholar] [CrossRef]
- Zhang, Z.-T.; Wu, Y.-C.; Dong, C.-M. Cathelicidin LL-37: Mechanisms of Action and Research Progress. Infect. Dis. Res. 2025, 6, 19. [Google Scholar] [CrossRef]
- Simonetti, O.; Cirioni, O.; Goteri, G.; Lucarini, G.; Kamysz, E.; Kamysz, W.; Orlando, F.; Rizzetto, G.; Molinelli, E.; Morroni, G.; et al. Efficacy of Cathelicidin LL-37 in an MRSA Wound Infection Mouse Model. Antibiotics 2021, 10, 1210. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Li, J.; Wei, G.; Shen, Z.; Li, B.; Wu, J.; Liu, J. Exploring the Antimicrobial Potential of LL-37 Derivatives: Recent Developments and Challenges. ACS Biomater. Sci. Eng. 2025, 11, 3145–3164. [Google Scholar] [CrossRef] [PubMed]
- Voronko, O.E.; Khotina, V.A.; Kashirskikh, D.A.; Lee, A.A.; Gasanov, V.A.O. Antimicrobial Peptides of the Cathelicidin Family: Focus on LL-37 and Its Modifications. Int. J. Mol. Sci. 2025, 26, 8103. [Google Scholar] [CrossRef]
- Chen, Z.; Yu, X.; Zhang, A.; Wang, F.; Xing, Y. De Novo Hydrocarbon-Stapling Design of Single-Turn α-Helical Antimicrobial Peptides. Int. J. Pept. Res. Ther. 2020, 26, 1711–1719. [Google Scholar] [CrossRef]
- Habibie, A.; Putri, R.A.; Swasono, R.T.; Retnaningrum, E.; Dhar, P.; Kuczera, K.; Raharjo, T.J.; Siahaan, T.J. Improving Conformational Stability and Bacterial Membrane Interactions of Antimicrobial Peptides with Amphipathic Helical Structure. Med. Chem. Res. 2025, 34, 2593–2609. [Google Scholar] [CrossRef]
- Högel, P.; Götz, A.; Kuhne, F.; Ebert, M.; Stelzer, W.; Rand, K.D.; Scharnagl, C.; Langosch, D. Glycine Perturbs Local and Global Conformational Flexibility of a Transmembrane Helix. Biochemistry 2018, 57, 1326–1337. [Google Scholar] [CrossRef]
- Yokoo, H.; Hirano, M.; Ohoka, N.; Misawa, T.; Demizu, Y. Structure–Activity Relationship Study of Amphipathic Antimicrobial Peptides Using Helix-destabilizing Sarcosine. J. Pept. Sci. 2021, 27, e3360. [Google Scholar] [CrossRef]
- Schahl, A.; Réat, V.; Malaga, W.; Birbes, C.; Czaplicki, G.; Jolibois, F.; Yamamoto, E.; Ramos, P.; Milon, A.; Saurel, O.; et al. How PGL Finds a Sweet Spot in Phospholipid Membranes: A Combined Multiscale MD and NMR Study. Biophys. J. 2026, 125, 457–470. [Google Scholar] [CrossRef]
- Cui, S.; Guo, C.; Yan, L.; He, Y.; Wu, L. Research on Enhancing Enzymatic Degradation of Anti-Digestive Peptides Containing D-Amino Acids through N-Terminal Acetylation. Bioorganic Chem. 2025, 158, 108337. [Google Scholar] [CrossRef]
- Kuzmin, D.V.; Emelianova, A.A.; Kalashnikova, M.B.; Panteleev, P.V.; Ovchinnikova, T.V. Effect of N- and C-Terminal Modifications on Cytotoxic Properties of Antimicrobial Peptide Tachyplesin I. Bull. Exp. Biol. Med. 2017, 162, 754–757. [Google Scholar] [CrossRef]
- Wątły, J.; Szarszoń, K.; Sabieraj, M.; Kola, A.; Wieczorek, R.; Janek, T.; Valensin, D. Modulating Copper(II) Coordination and Antimicrobial Activity: Effects of d -Amino Acid Substitution and Retro-Inverso Modification in Human Saliva MUC7 Peptide. Inorg. Chem. 2025, 64, 6365–6377. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, D. Antimicrobial Peptides. In Antibiotics and Their Mechanisms of Action; Nagarajan, D., Ed.; Springer Nature Singapore: Singapore, 2024; pp. 53–65. ISBN 978-981-97-6850-9. [Google Scholar]
- Sharma, A.; Gaur, A.; Kumar, V.; Sharma, N.; Patil, S.A.; Verma, R.K.; Singh, A.K. Antimicrobial Activity of Synthetic Antimicrobial Peptides Loaded in Poly-Ɛ-Caprolactone Nanoparticles against Mycobacteria and Their Functional Synergy with Rifampicin. Int. J. Pharm. 2021, 608, 121097. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Xu, H.; Xia, J.; Ma, J.; Xu, J.; Li, Y.; Feng, J. D- and Unnatural Amino Acid Substituted Antimicrobial Peptides With Improved Proteolytic Resistance and Their Proteolytic Degradation Characteristics. Front. Microbiol. 2020, 11, 563030. [Google Scholar] [CrossRef] [PubMed]
- Khara, J.S.; Mojsoska, B.; Mukherjee, D.; Langford, P.R.; Robertson, B.D.; Jenssen, H.; Ee, P.L.R.; Newton, S.M. Ultra-Short Antimicrobial Peptoids Show Propensity for Membrane Activity Against Multi-Drug Resistant Mycobacterium Tuberculosis. Front. Microbiol. 2020, 11, 417. [Google Scholar] [CrossRef]
- Tan, T.; Wu, D.; Li, W.; Zheng, X.; Li, W.; Shan, A. High Specific Selectivity and Membrane-Active Mechanism of Synthetic Cationic Hybrid Antimicrobial Peptides Based on the Peptide FV7. Int. J. Mol. Sci. 2017, 18, 339. [Google Scholar] [CrossRef]
- Lai, C.-W.; Lin, C.-Y.; Tsai, M.-C.; Chen, W.-J.; Hsieh, C.-C.; Lin, Z.-J.; Shen, L.-J.; Chen, Y.-L.; Lai, L.-J.; Chen, S.-H.; et al. From AI to Action: Antimicrobial Peptides Engineered by Generative Adversarial Networks (GANs)-A Novel Approach to Combat Resistant Bacteria. Chem. Eng. J. 2025, 519, 164905. [Google Scholar] [CrossRef]
- Sharma, D.; Poonam; Shrivastava, R.; Bisht, G.S. In Vitro Efficacy of Lipid Conjugated Peptidomimetics Against Mycobacterium Smegmatis. Int. J. Pept. Res. Ther. 2020, 26, 531–537. [Google Scholar] [CrossRef]
- Casanova, M.; Maresca, M.; Poncin, I.; Point, V.; Olleik, H.; Boidin-Wichlacz, C.; Tasiemski, A.; Mabrouk, K.; Cavalier, J.-F.; Canaan, S. Promising Antibacterial Efficacy of Arenicin Peptides against the Emerging Opportunistic Pathogen Mycobacterium Abscessus. J. Biomed. Sci. 2024, 31, 18. [Google Scholar] [CrossRef]
- Ricaurte, S.N. Características Microbiológicas Asociadas a La Virulencia de Mutantes Defectivos En Los Transportadores de Membrana CtpF y MmpL7 de Mycobacterium Tuberculosis. 2025. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



