Submitted:
23 March 2026
Posted:
24 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
3. Biomass Characterization
3.1. Volatiles
3.2. Moisture
3.3. Ash
3.4. Biomass Physical and Chemical Characteristics
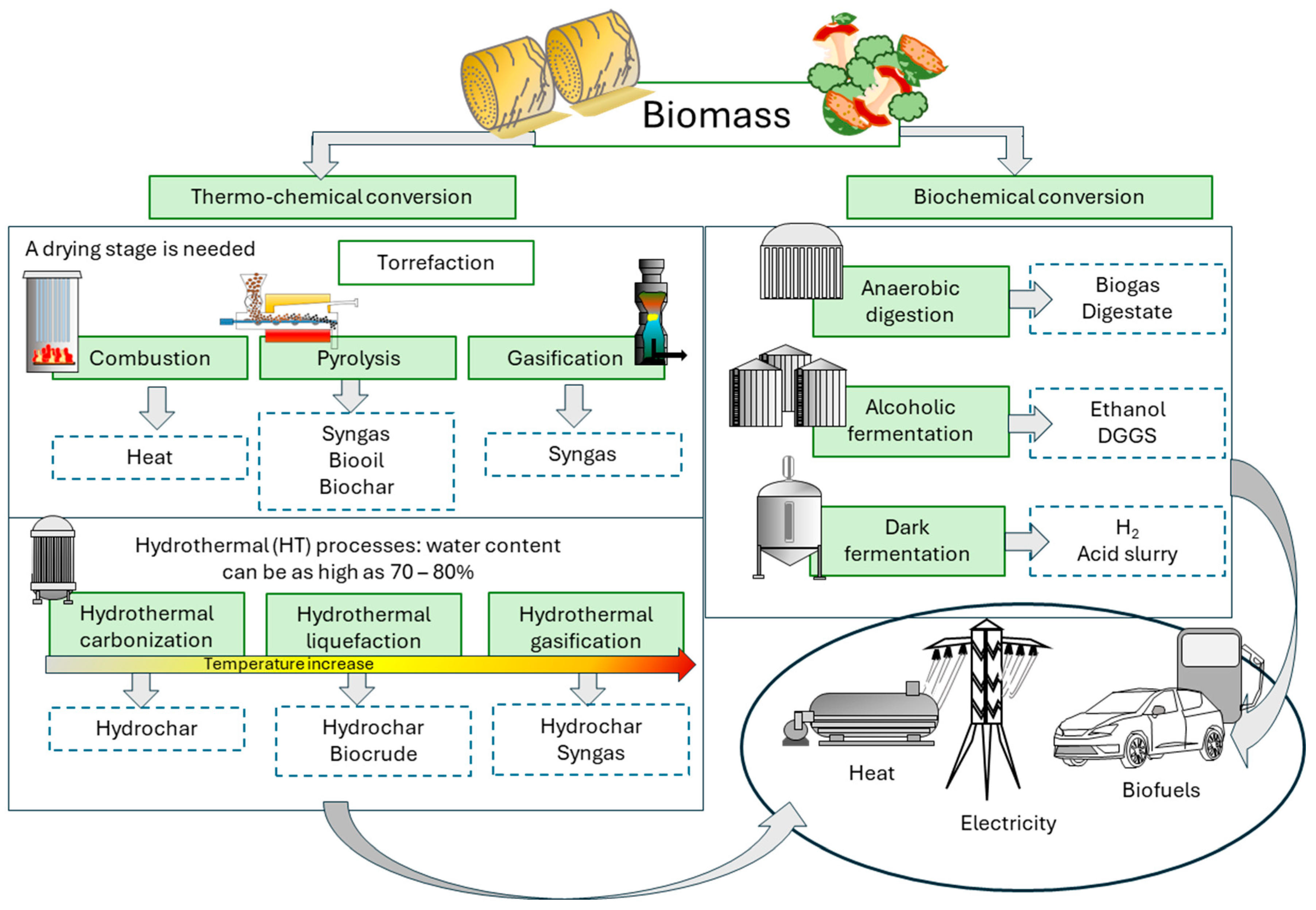
4. Biomass Conversion Following Different Energy Pathways
4.1. Main Biological Conversion Pathways for Fuel Production
4.2. Physical-Chemical Conversion for Biodiesel Production
4.3. Thermochemical Conversions for Fuel Production
- Combustion: It is an ancient and widely used process that burns biomass in the presence of oxygen to release heat, which can be directly used in domestic and industrial processes, for generating steam to cover heat demand or to produce electricity [100].
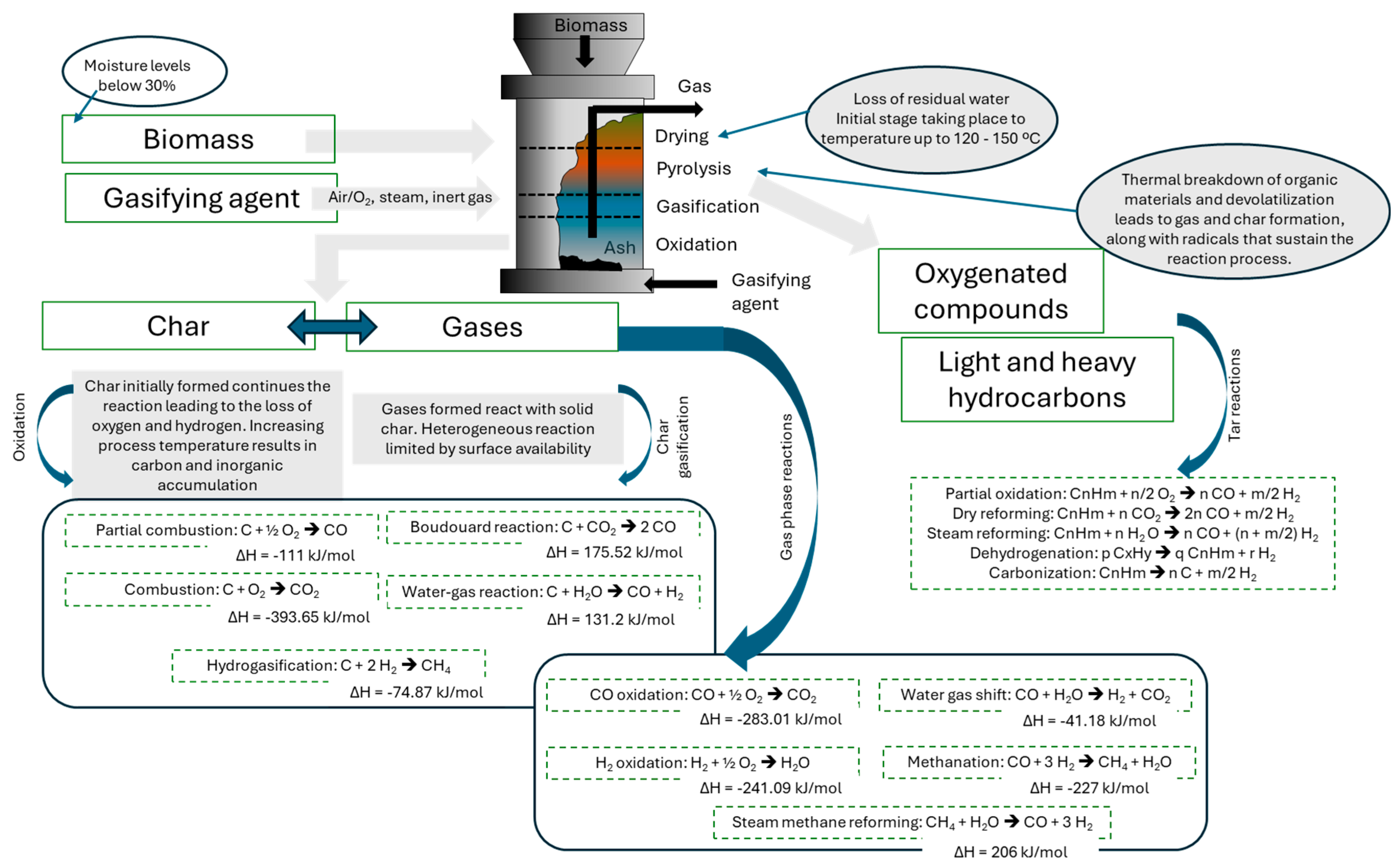
- Pyrolysis: biomass is converted at high temperatures (300 and 1000 °C) in an oxygen-free environment, producing three main products: syngas, bio-oil, and biochar [101,102]. The heating rate and temperature greatly influence the product yield. Table 2 shows different pyrolysis processes classified by the heating rate and process temperature.
| Pyrolysis process | Heating rate (°C/s) | Temperature range (°C) | Residence time | Particle size (mm) | Reference | |
| Vapor residence time (s) | Solid residence time | |||||
| Slow | 0.1 – 1.0 | 300 – 950 | 330 – 550 | 5.5 – 9.2 min | 5 – 50 | [105,106] |
| Intermediate | 1 – 10 | 400 – 500 | 10 – 30 | 0.7 – 0.5 min | 0.3 – 0.75 | [107,108] |
| Fast | 10 – 200 | Up to 1000 | ≤5 | <0.5 – 10 s | ≤1 | [105,106,109] |
| Flash | 13,000 – 21,000 | 900 – 1200 | 0.5 – 0.025 | 0.1 | [11,110] | |
- Gasification: The process converts biomass into syngas through partial oxidation. Syngas is a mixture of CO, H2, and CH4 that can be used to generate electricity or as a raw material for subsequent chemical processes to produce liquid fuels [101,112]. Gasification offers a competitive technical alternative to combustion. The combustion of syngas is cleaner than the direct combustion of biomass, reducing the need for excess air and, consequently, heat losses. Besides, it ensures homogeneous combustion, enabling better process control. The volumetric flow rate is lower than that of the effluent gases in direct combustion, providing a cost-effective gas-cleaning process [113].
- Hydrothermal processes: The technology involves the thermal cracking of biomass in the presence of water and below the supercritical point. Under high temperature and pressure, hydroxyl ions enable the degradation of organics, similarly to conventional thermal processes, but with the added advantage of avoiding a prior drying stage of the material, thereby reducing the energy demand [114]. The process can be classified by temperature range: hydrothermal carbonization is limited to temperatures below 260 – 280 °C, while higher temperatures, close to the critical point (374 °C), encompass hydrothermal liquefaction, where the main product of interest is a biocrude. In contrast, at lower temperatures, hydrochar is the main product. At temperatures above the supercritical point, the process is called hydrothermal gasification, and as its name suggests, syngas is the main product. Hydrothermal processes may also involve an additional category associated with oxidation or organic matter (wet oxidation) under conditions below or around the supercritical point [115]. The addition of an oxidant (air or oxygen) significantly favors the conversion of organics into small molecules. When the process is carried out at temperatures above the supercritical point, it is known as supercritical water gasification [116].
5. Biomass Gasification: Process Stages
6. Factors Influencing Biomass Gasification
6.1. Feedstock Characteristics
6.1.1. Particle Size
6.1.2. Moisture Content
6.1.3. Inorganic Content
6.1.4. Activation Energy
6.2. Gasification Operating Conditions
6.2.1. Temperature
6.2.2. Pressure
6.2.3. Equivalence Ratio
6.2.4. Residence Time
6.2.4. Gasifying Agents
6.2.5. The Use of CO2 as a Gasifying Agent Brings Additional Benefits
7. Biomass Gasification Technologies
8. Gasification of Low-Quality Biomass
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DOE. International Energy Outlook 2023. U.S. Department of Energy. 2023. Available online: https://www.eia.gov/outlooks/ieo/ (accessed on 3 February 2026).
- IEA. World Energy Outlook 2024 (No. 4.0). 2023. Available online: https://www.iea.org/reports/world-energy-outlook-2024 (accessed on 3 February 2026).
- Cevik, E.I.; Yıldırım, D.Ç.; Dibooglu, S. Renewable and non-renewable energy consumption and economic growth in the US: A Markov-Switching VAR analysis. Energy Environ. 2021, 32, 519–541. [Google Scholar] [CrossRef]
- Ramasesha, S.K. Challenges in the quest for clean energies: 4. Other renewable resources and conclusion. Resonance 2013, 18, 1110–1126. [Google Scholar] [CrossRef]
- Prokhorov, O.; Dreisbach, D. The Impact of Renewables on the Incidents of Negative Prices in the Energy Spot Markets. Energy Policy 2022, 167, 113073. [Google Scholar] [CrossRef]
- El-Sayed, A.H.A.; Khalil, A.; Yehia, M. Energy storage systems impact on Egypt’s future energy mix with high renewable energy penetration: A long-term analysis. J. Energy Storage 2024, 95, 112583. [Google Scholar] [CrossRef]
- González, R.; Gómez, X. Simulation of Conventional WWTPs Acting as Mediators in H2/CO2 Conversion into Methane. Environments 2025, 12, 245. [Google Scholar] [CrossRef]
- Chuang, M.C.; Ma, H.W. Energy security and improvements in the function of diversity indices—Taiwan energy supply structure case study. Renew. Sustain. Energy Rev. 2013, 24, 9–20. [Google Scholar] [CrossRef]
- Le, T.-H.; Nguyen, C.P. Is energy security a driver for economic growth? Evidence from a global sample. Energy Policy 2019, 129, 436–451. [Google Scholar] [CrossRef]
- Oveisi, E.; Sokhansanj, S.; Lau, A.; Lim, J.; Bi, X.; Preto, F.; Mui, C. Characterization of Recycled Wood Chips, Syngas Yield, and Tar Formation in an Industrial Updraft Gasifier. Environments 2018, 5, 84. [Google Scholar] [CrossRef]
- Mehmeti, A.; Angelis-Dimakis, A.; Arampatzis, G.; McPhail, S.J.; Ulgiati, S. Life Cycle Assessment and Water Footprint of Hydrogen Production Methods: From Conventional to Emerging Technologies. Environments 2018, 5, 24. [Google Scholar] [CrossRef]
- Kirkels, A.F.; Verbong, G.P. Biomass gasification: Still promising? A 30-year global overview. Renew. Sustain. Energy Rev. 2010, 15, 471–481. [Google Scholar] [CrossRef]
- Makepa, D.C.; Chihobo, C.H. Sustainable pathways for biomass production and utilization in carbon capture and storage—A review. Biomass Convers. Biorefin. 2025, 15, 11397–11419. [Google Scholar] [CrossRef]
- Henrich, E.; Dahmen, N.; Dinjus, E.; Sauer, J. The role of biomass in a future world without fossil fuels. Chem. Ing. Tech. 2015, 87, 1667–1685. [Google Scholar] [CrossRef]
- Dufour, A. Thermochemical Conversion of Biomass for Energy and Chemicals Production; John Wiley & Sons, Inc.: New Jersey, United States, 2016; pp. 1–28. [Google Scholar] [CrossRef]
- He, M.; Sun, Y.; Han, B. Green Carbon Science: Efficient Carbon Resource Processing, Utilization, and Recycling towards Carbon Neutrality. Angew. Chem. Int. Ed. 2022, 61, e202112835. [Google Scholar] [CrossRef] [PubMed]
- Matin, B.; Leto, J.; Antonović, A.; Brandić, I.; Jurišić, V.; Matin, A.; Krička, T.; Grubor, M.; Kontek, M.; Bilandžija, N. Energetic Properties and Biomass Productivity of Switchgrass (Panicum virgatum L.) under Agroecological Conditions in Northwestern Croatia. Agronomy 2023, 13, 1161. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Ali, S.; Shah, T.A.; Afzal, A.; Tabassum, R. Exploring lignocellulosic biomass for bio-methane potential by anaerobic digestion and its economic feasibility. Energy Environ. 2018, 29, 742–751. [Google Scholar] [CrossRef]
- Grippi, D.; Clemente, R.; Bernal, M.P. Chemical and Bioenergetic Characterization of Biofuels from Plant Biomass: Perspectives for Southern Europe. Appl. Sciences 2020, 10, 3571. [Google Scholar] [CrossRef]
- Kim, J.; Sen, S.M.; Maravelias, C.T. An optimization-based assessment framework for biomass-to-fuel conversion strategies. Energy Environ. Sci. 2013, 6, 1093–1104. [Google Scholar] [CrossRef]
- Dai, J.; Saayman, J.; Grace, J.R.; Ellis, N. Gasification of Woody Biomass. Annu. Rev. Chem. Biomol. Eng. 2015, 6, 77–99. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.P.; Teixeira, S.; Teixeira, J.C. Characterization of the physicochemical and thermal properties of different forest residues. Biomass Bioenerg. 2023, 175, 106870. [Google Scholar] [CrossRef]
- Dida, G. Biotechnology towards energy crops. CABI Agric. Biosci. 2024, 5, 45. [Google Scholar] [CrossRef]
- Channiwala, S.A.; Parikh, P.P. A unified correlation for estimating HHV of solid, liquid and gaseous fuels. Fuel 2002, 81, 1051–1063. [Google Scholar] [CrossRef]
- Basu, P.; Basu, P. Chapter 3 - Biomass Characteristics. In Biomass Gasification, Pyrolysis and Torrefaction, 2nd ed.; Academic Press: London, United Kingdom, 2013; pp. 47–86. [Google Scholar] [CrossRef]
- Cordero, T.; Marquez, F.; Rodriguez-Mirasol, J.; Rodriguez, J.J. Predicting heating values of lignocellulosics and carbonaceous materials from proximate analysis. Fuel 2001, 80, 1567–1571. [Google Scholar] [CrossRef]
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of heating values of biomass fuel from elemental composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Parikh, J.; Channiwala, S.A.; Ghosal, G.K. A correlation for calculating HHV from proximate analysis of solid fuels. Fuel 2005, 84, 487–494. [Google Scholar] [CrossRef]
- Sheng, C.; Azevedo, J.L.T. Estimating the higher heating value of biomass fuels from basic analysis data. Biomass Bioenerg. 2005, 28, 499–507. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, Y.; Zhu, J.; Cao, Y.; Pan, W. Coal and biomass partial gasification and soot properties in an atmospheric fluidized bed. Energy Fuels 2011, 25, 1964–1969. [Google Scholar] [CrossRef]
- Howaniec, N.; Smoliński, A. Effect of fuel blend composition on the efficiency of hydrogen-rich gas production in co-gasification of coal and biomass. Fuel 2014, 128, 442–450. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 3): Gasification technologies. Bioresour. Technol. 2002, 83, 55–63. [Google Scholar] [CrossRef] [PubMed]
- González-Vázquez, M.P.; García, R.; Gil, M.V.; Pevida, C.; Rubiera, F. Comparison of the gasification performance of multiple biomass types in a bubbling fluidized bed. Energy Convers. Manag. 2018, 176, 309–323. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, M.; Raheem, A.; Wang, F.; Wei, J.; Xu, D.; Song, X.; Bao, W.; Huang, A.; Zhang, S.; Zhang, H. Syngas production from biomass gasification: Influences of feedstock properties, reactor type, and reaction parameters. ACS omega 2023, 8, 31620–31631. [Google Scholar] [CrossRef] [PubMed]
- Naryanto, R.F.; Enomoto, H.; Vo Cong, A.; Fukadu, K.; Zong, Z.; Delimayanti, M.K.; Chunti, C.; Noda, R. The Effect of Moisture Content on the Tar Characteristic of Wood Pellet Feedstock in a Downdraft Gasifier. Appl. Sciences 2020, 10, 2760. [Google Scholar] [CrossRef]
- Dong, J.; Chi, Y.; Tang, Y.; Ni, M.; Nzihou, A.; Weiss-Hortala, E.; Huang, Q. Effect of operating parameters and moisture content on municipal solid waste pyrolysis and gasification. Energy Fuels 2016, 30, 3994–4001. [Google Scholar] [CrossRef]
- Lindström, E.; Larsson, S.H.; Boström, D.; Öhman, M. Slagging Characteristics during Combustion of Woody Biomass Pellets Made from a Range of Different Forestry Assortments. Energy Fuels 2010, 24, 3456–3461. [Google Scholar] [CrossRef]
- Font Palma, C. Model for biomass gasification including tar formation and evolution. Energy Fuels 2013, 27, 2693–2702. [Google Scholar] [CrossRef]
- Milne, T.A.; Evans, R.J.; Abatzoglou, N. Biomass Gasifier “Tars”: Their Nature, Formation, and Conversion (No. NREL/TP-570-25357). National Renewable Energy Laboratory, 1998. Available online: https://www.nrel.gov/docs/fy99osti/25357.pdf (accessed on 3 February 2026).
- Littlejohns, J.V.; Butler, J.; Luque, L.; Austin, K. Experimental Investigation of Bioenergy Production from Small-Scale Gasification of Landfill-Diverted Wood Wastes. Waste Biomass Valori. 2020, 11, 6885–6901. [Google Scholar] [CrossRef]
- Kim, K.; Jahan, S.A.; Kabir, E.; Brown, R.J. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ. Int. 2013, 60, 71–80. [Google Scholar] [CrossRef]
- Claude, V.; Courson, C.; Köhler, M.; Lambert, S.D. Overview and essentials of biomass gasification technologies and their catalytic cleaning methods. Energy Fuels 2016, 30, 8791–8814. [Google Scholar] [CrossRef]
- Narnaware, S. L.; Panwar, N.L. Catalysts and their role in biomass gasification and tar abetment: a review. Biomass Convers. Biorefin. 2021, 1–31. [Google Scholar] [CrossRef]
- Telmo, C.; Lousada, J.; Moreira, N. Proximate analysis, backwards stepwise regression between gross calorific value, ultimate and chemical analysis of wood. Bioresour. Technol. 2010, 101, 3808–3815. [Google Scholar] [CrossRef]
- Zevenhoven-Onderwater, M.; Backman, R.; Skrifvars, B.-J.; Hupa, M. The Ash Chemistry in Fluidised Bed Gasification of Biomass Fuels. Part I: Predicting the Chemistry of Melting Ashes and Ash–Bed Material Interaction. Fuel 2001, 80, 1489–1502. [Google Scholar] [CrossRef]
- Mlonka-Mędrala, A.; Magdziarz, A.; Gajek, M.; Nowińska, K.; Nowak, W. Alkali metals association in biomass and their impact on ash melting behaviour. Fuel 2020, 261, 116421. [Google Scholar] [CrossRef]
- Troiano, M.; Montagnaro, F.; Salatino, P.; Solimene, R. Experimental characterization of particle-wall interaction relevant to entrained-flow gasification of biomass. Fuel 2017, 209, 674–684. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, L.; Pu, J.; Zhao, J. Melting characteristics of coal ash and properties of fly ash to understand the slag formation in the shell gasifier. ACS omega 2021, 6, 16066–16075. [Google Scholar] [CrossRef]
- Pushp, M.; Gall, D.; Davidsson, K.; Seemann, M.; Pettersson, J.B. Influence of bed material, additives, and operational conditions on alkali metal and tar concentrations in fluidized bed gasification of biomass. Energy Fuels 2018, 32, 6797–6806. [Google Scholar] [CrossRef]
- Al-Rahbi, A.S.; Williams, P.T. Waste ashes as catalysts for the pyrolysis–catalytic steam reforming of biomass for hydrogen-rich gas production. J. Mater. Cycles Waste Manag. 2019, 21, 1224–1231. [Google Scholar] [CrossRef]
- Schmid, J.C.; Wolfesberger, U.; Koppatz, S.; Pfeifer, C.; Hofbauer, H. Variation of feedstock in a dual fluidized bed steam gasifier—Influence on product gas, tar content, and composition. Environ. Prog. Sustain. Energy 2012, 31, 205–215. [Google Scholar] [CrossRef]
- Saddawi, A.; Jones, J.M.; Williams, A.; Le Coeur, C. Commodity fuels from biomass through pretreatment and torrefaction: effects of mineral content on torrefied fuel characteristics and quality. Energy Fuels 2012, 26, 6466–6474. [Google Scholar] [CrossRef]
- Chen, Q.; Zhou, J.; Liu, B.; Mei, Q.; Luo, Z. Influence of torrefaction pretreatment on biomass gasification technology. Chin. Sci. Bull. 2011, 56, 1449–1456. [Google Scholar] [CrossRef]
- Raj, R.S.; Jain, S.; Sharma, A.K. Optimized torrefaction of municipal solid waste for enhanced energy recovery. Biofuels 2025, 1–19. [Google Scholar] [CrossRef]
- Fajimi, L.I.; Chrisostomou, J.; Oboirien, B.O. A techno-economic analysis (TEA) of a combined process of torrefaction and gasification of lignocellulose biomass (bagasse) for methanol and electricity production. Biomass Convers. Biorefin. 2024, 14, 12501–12516. [Google Scholar] [CrossRef]
- Stolarski, M.; Mleczek, M.; Szczukowski, S.; Goliński, P.; Waliszewska, B.; Szentner, K.; Rutkowski, P.; Krzyżaniak, M. Characteristics of Thermophysical Parameters of Selected Salix Taxa with Elemental Analysis. Int. J. Green Energy 2015, 12, 1272–1279. [Google Scholar] [CrossRef]
- Brown, R.C.; Brown, T.R.; Brown, R.C.; Brown, T.R. Appendice A: Description of Biorenewable Resources. In Biorenewable Resources: Engineering New Products from Agriculture, 1st ed.; John Wiley & Sons, Inc: New Jersey, USA, 2014; pp. 341–366. [Google Scholar] [CrossRef]
- Ul Hai, I.; Sher, F.; Yaqoob, A.; Liu, H. Assessment of biomass energy potential for SRC willow woodchips in a pilot scale bubbling fluidized bed gasifier. Fuel 2019, 258, 116143. [Google Scholar] [CrossRef]
- Nega, T.; Tesfaye, A.; Paramasivam, P. Design and CFD modeling of gasifier stove combined with heat exchanger for water heating application. AIP Advances 2022, 12, 045121. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, L.; Zhao, M.; Ma, H.; Chen, D.; Zhang, Y.; Zhou, J. Comparative Study on the Pyrolysis Behaviors of Pine Cone and Pretreated Pine Cone by Using TGA–FTIR and Pyrolysis-GC/MS. ACS Omega 2021, 6, 3490–3498. [Google Scholar] [CrossRef]
- Velázquez Martí, B.; Gaibor-Chávez, J.; Franco Rodríguez, J.E.; López Cortés, I. Biomass Identification from Proximate Analysis: Characterization of Residual Vegetable Materials in Andean Areas. Agronomy 2023, 13, 2347. [Google Scholar] [CrossRef]
- Rizzo, A.M.; Pettorali, M.; Nistri, R.; Chiaramonti, D. Mass and energy balances of an autothermal pilot carbonization unit. Biomass Bioenerg. 2019, 120, 144–155. [Google Scholar] [CrossRef]
- Sutanto, H.; Suyitno, S.; Juwana, W.E.; Nurrohim, T. G. Design, Production Cost, and Air Flow Distribution of Biomass Pellet Furnace. Mekanika 2021, 20, 68. [Google Scholar] [CrossRef]
- Rezaei, H.; Lim, J.; Sokhansanj, S. Comparison of Drying Rates of Ground Western Red Cedar with Hemlock, Birch, Aspen, and Spruce/Pine/Douglas Fir. Appl. Eng. Agric. 2020, 36, 159–165. [Google Scholar] [CrossRef]
- Díez, H.E.; Pérez, J.F. Effects of wood biomass type and airflow rate on fuel and soil amendment properties of biochar produced in a top-lit updraft gasifier. Environ. Prog. Sustain. Energy 2019, 38, 13105. [Google Scholar] [CrossRef]
- Granados, D.A.; Ruiz, R.A.; Vega, L.Y.; Chejne, F. Study of reactivity reduction in sugarcane bagasse as consequence of a torrefaction process. Energy 2017, 139, 818–827. [Google Scholar] [CrossRef]
- Senneca, O.; Ciaravolo, S.; Nunziata, A. Composition of the gaseous products of pyrolysis of tobacco under inert and oxidative conditions. J. Anal. Appl. Pyrolysis 2007, 79, 234–243. [Google Scholar] [CrossRef]
- Patel, A.; Agrawal, B.; Rawal, B.R. Elemental Composition Of Biodiesel Produced By Fast Pyrolysis Of Eucalyptus Leaves. ICIPPSD Conference. J. Eng. Res. [CrossRef]
- Safaei, Z.; Zolanvari, R.; Adloo, H.; Sarhadi, H.; Khoramdel, S.; Gharacheh, A. Effects of Various Biomass Materials on LHV and Composition of Produced Syngas in a Downdraft Gasifier. Proceedings of The 11th International Chemical Engineering Congress & Exhibition (IChEC 2020), Fouman, Iran, 15-17 April, 2020; Available online: https://www.researchgate.net/profile/Roshanak-Zolanvari/publication/350739810_Effects_of_Various_Biomass_Materials_on_LHV_and_Composition_of_Produced_Syngas_in_a_Downdraft_Gasifier/links/606f470f299bf1c911ba0efc/Effects-of-Various-Biomass-Materials-on-LHV-and-Composition-of-Produced-Syngas-in-a-Downdraft-Gasifier.pdf (accessed on 4 February 2026).
- Kazemi, F.; Younesi, H.; Ghoreyshi, A.A.; Bahramifar, N.; Heidari, A. Thiol-incorporated activated carbon derived from fir wood sawdust as an efficient adsorbent for the removal of mercury ion: Batch and fixed-bed column studies. Process Saf. Environ. Prot. 2016, 100, 22–35. [Google Scholar] [CrossRef]
- Diker, İ.; Ozkan, G.M. An investigation on implementing wet torrefaction to dewatered poultry sludge. Biomass Convers. Biorefi. 2024, 14, 5517–5530. [Google Scholar] [CrossRef]
- Hejna, M.; Świechowski, K.; Rasaq, W.A.; Białowiec, A. Study on the Effect of Hydrothermal Carbonization Parameters on Fuel Properties of Chicken Manure Hydrochar. Materials 2022, 15, 5564. [Google Scholar] [CrossRef]
- Janković, B. On-line pyrolysis kinetics of swine manure solid samples collected from rearing farm: Pyrolytic behavior in dynamic heating mode. J. Therm. Anal. Calorim. 2016, 123, 2103–2120. [Google Scholar] [CrossRef]
- Kaur, H.; Kommalapati, R.R. Optimizing anaerobic co-digestion of goat manure and cotton gin trash using biochemical methane potential (BMP) test and mathematical modeling. SN Appl. Sci. 2021, 3, 724. [Google Scholar] [CrossRef]
- Hadroug, S.; Jellali, S.; Leahy, J.J.; Kwapinska, M.; Jeguirim, M.; Hamdi, H.; Kwapinski, W. Pyrolysis Process as a Sustainable Management Option of Poultry Manure: Characterization of the Derived Biochars and Assessment of their Nutrient Release Capacities. Water 2019, 11, 2271. [Google Scholar] [CrossRef]
- Tańczuk, M.; Junga, R.; Kolasa-Więcek, A.; Niemiec, P. Assessment of the energy potential of chicken manure in Poland. Energies 2019, 12. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, Z.; Xu, J.; Liu, C.; Qing, M.; Jiang, L.; Wang, Y.; Hu, S.; Luo, Z.; Xiang, J. (A study on the devolatilization and combustion characteristics of yak manure under simulated plateau conditions. Biomass Bioenerg. 2025, 199, 107919. [Google Scholar] [CrossRef]
- Shen, X.; Huang, G.; Yang, Z.; Han, L. Compositional characteristics and energy potential of Chinese animal manure by type and as a whole. Appl. Energy 2015, 160, 108–119. [Google Scholar] [CrossRef]
- Hernandez, J.J.; Aranda-Almansa, G.; Serrano, C. Co-gasification of biomass wastes and coal− coke blends in an entrained flow gasifier: an experimental study. Energy Fuels 2010, 24, 2479–2488. [Google Scholar] [CrossRef]
- Fermoso, J.; Arias, B.; Moghtaderi, B.; Pevida, C.; Plaza, M.G.; Pis, J.J.; Rubiera, F. Effect of co-gasification of biomass and petroleum coke with coal on the production of gases. Greenhouse Gases: Sci. Technol. 2012, 2, 304–313. [Google Scholar] [CrossRef]
- Adeyemi, I.; Khan, H.; Ghenai, C.; Janajreh, I. Techno-economic analysis of the co-gasification of sewage sludge and petroleum coke. Front. Sustain. 2023, 4, 1234760. [Google Scholar] [CrossRef]
- Haykırı-Açma, H. Combustion characteristics of different biomass materials. Energy Convers. Manag. 2003, 44, 155–162. [Google Scholar] [CrossRef]
- Lapuerta, M.; Hernández, J.J.; Pazo, A.; López, J. Gasification and co-gasification of biomass wastes: Effect of the biomass origin and the gasifier operating conditions. Fuel Process. Technol. 2008, 89, 828–837. [Google Scholar] [CrossRef]
- Barot, S. Biomass and bioenergy: Resources, conversion and application. In Renewable Energy for Sustainable Growth Assessment; Kumar, N., Prabhansu, Eds.; Wiley-Scrivener: Massachusetts, USA, 2022; pp. 243–262. [Google Scholar] [CrossRef]
- Cheng, J.J. Kinetics and microbiology of biological processes. In Biomass to Renewable Energy Processes, 2nd Ed.; Cheng, J., Ed.; CRC Press: Boca Raton, USA, 2017; pp. 125–141. [Google Scholar] [CrossRef]
- Ellacuriaga, M.; García-Cascallana, J.; Gómez, X. Biogas Production from Organic Wastes: Integrating Concepts of Circular Economy. Fuels 2021, 2, 144–167. [Google Scholar] [CrossRef]
- De Carolis, C.; Iori, V.; Narciso, A.; Gentile, D.; Casentini, B.; Pietrini, F.; Grenni, P.; Barra Caracciolo, A.; Iannelli, M.A. The Effects of Different Combinations of Cattle Organic Soil Amendments and Copper on Lettuce (cv. Rufus) Plant Growth. Environments 2024, 11, 134. [Google Scholar] [CrossRef]
- Schiel-Bengelsdorf, B.; Montoya, J.; Linder, S.; Dürre, P. Butanol fermentation. Environ. Technol. 2013, 34(13-14), 1691–1710. [Google Scholar] [CrossRef]
- Lignin & Lignosulfonates. Available online: https://www.borregaard.com/product-areas/lignin?utm_term=lignin%20biopolymer&utm_campaign=Product+Area+%7C+Lignin+Biopolymers+%7C+Global&utm_source=adwords&utm_medium=ppc&hsa_acc=3642184401&hsa_cam=13555587785&hsa_grp=126240300920&hsa_ad=655182154449&hsa_src=g&hsa_tgt=kwd-1371649915331&hsa_kw=lignin%20biopolymer&hsa_mt=p&hsa_net=adwords&hsa_ver=3&gad_source=1&gclid=Cj0KCQjwhMq-BhCFARIsAGvo0Kdjk2MwX9COmiEgAcjot4SX2O6wWkGm2fN1g8PxEBAIyluykrEez8IaAlfhEALw_wcB (accessed on 4 February 2026).
- Rødsrud, G.; Lersch, M.; Sjöde, A. History and future of world's most advanced biorefinery in operation. Biomass Bioenerg. 2012, 46, 46–59. [Google Scholar] [CrossRef]
- Neste. Available online: https://www.neste.com/ (accessed on 4 February 2026).
- Combustibles renovables y su impacto en la movilidad. Available online: https://www.repsol.com/es/energia-futuro/movilidad-sostenible/combustibles-renovables/index.cshtml (accessed on 4 February 2026).
- Alazaiza, M.Y.D.; Ahmad, Z.; Albahnasawi, A.; Nassani, D.E.; Alenezi, R.A. Biomass processing technologies for bioenergy production: factors for future global market. Int. J. Environ. Sci. Technol. 2024, 21, 2307–2324. [Google Scholar] [CrossRef]
- Balagurumurthy, B.; Singh, R.; Ohri, P.; Prakash, A.; Bhaskar, T. Thermochemical Biorefinery. In Recent Advances in Thermochemical Conversion of Biomass; Pandey, A., Bhaskar, T., Stöcker, M., Sukumaran, R.K., Eds.; Elsevier Science & Technology: Amsterdam, Netherlands, 2015; pp. 157–174. [Google Scholar] [CrossRef]
- Komandur, J.; Das, A.; Mohanty, K. Thermochemical conversion of woody biomass to energy and high-value products. In Sustainable Biorefining of Woody Biomass to Biofuels and Biochemicals; Kumar, D., Kumar, S., Rajendran, K., Eds.; Woodhead Publishing: Cambridge, UK, 2023; pp. 125–162. [Google Scholar] [CrossRef]
- Sikarwar, V.S.; Zhao, M.; Clough, P.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.J.; Fennell, P.S. An overview of advances in biomass gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar] [CrossRef]
- Balat, M.; Balat, M.; Kirtay, E.; Balat, H. Main routes for the thermo-conversion of biomass into fuels and chemicals. Part 2: Gasification systems. In Energy Convers. Manag.; Scopus, 2009; Volume 50, pp. 3158–3168. [Google Scholar] [CrossRef]
- Sharma, A.; Pareek, V.; Zhang, D. Biomass pyrolysis—A review of modelling, process parameters and catalytic studies. Renew. Sustain. Energy Rev. 2015, 50, 1081–1096. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Baxter, L.L.; Koppejan, J. Biomass combustion. In Thermochemical processing of biomass: conversion into fuels, chemicals and power, 2nd ed.; Brown, R.C., Ed.; Wiley-VCH: Weinheim, Germany, 2019; pp. 49–83. [Google Scholar] [CrossRef]
- Banik, R.K.; Das, S.; Bordoloi, U.; Das, H.J.; Das, B.; Basumatary, S.; Das, B.; Kalita, P. The Promising Role of Thermochemical Conversion in Sustainable Power Generation. In Challenges and Opportunities of Distributed Renewable Power. Energy, Environment, and Sustainability; De, S., Agarwal, A.K., Kalita, P., Eds.; Springer: Singapore, 2024; pp. 101–140. [Google Scholar] [CrossRef]
- Patel, A.; Agrawal, B.; Rawal, B.R. Pyrolysis of biomass for efficient extraction of biofuel. Energ. Sources, Part A 2020, 42, 1649–1661. [Google Scholar] [CrossRef]
- Panwar, N.L.; Divyangkumar, N. An overview of recent advancements in biomass torrefaction. Environ. Dev. Sustain. 2024. [Google Scholar] [CrossRef]
- Chandra C.S., J.; Sasi, S.; Sharmila T.K., B.; Varghese, J.R. Modern Biomass Conversion Technologies. In Handbook of Biomass; Thomas, S., Hosur, M., Pasquini, D., Jose Chirayil, C., Eds.; Springer: Singapore, 2024; pp. 1037–1067. [Google Scholar] [CrossRef]
- Aboelela, D.; Saleh, H.; Attia, A.M.; Elhenawy, Y.; Majozi, T.; Bassyouni, M. Recent Advances in Biomass Pyrolysis Processes for Bioenergy Production: Optimization of Operating Conditions. Sustainability 2023, 15, 11238. [Google Scholar] [CrossRef]
- Pahnila, M.; Koskela, A.; Sulasalmi, P.; Fabritius, T. A Review of Pyrolysis Technologies and the Effect of Process Parameters on Biocarbon Properties. Energies 2023, 16, 6936. [Google Scholar] [CrossRef]
- Jerzak, W.; Reinmöller, M.; Magdziarz, A. Estimation of the heat required for intermediate pyrolysis of biomass. Clean Techn. Environ. Policy 2022, 24, 3061–3075. [Google Scholar] [CrossRef]
- Torri, C.; Fabbri, D. Biochar enables anaerobic digestion of aqueous phase from intermediate pyrolysis of biomass. Bioresour. Technol. 2014, 172, 335–341. [Google Scholar] [CrossRef]
- Tippayawong, N.; Kinorn, J.; Thavornun, S. Yields and Gaseous Composition from Slow Pyrolysis of Refuse-derived Fuels. Energ. Sources, Part A 2008, 30, 1572–1580. [Google Scholar] [CrossRef]
- Shuangning, X.; Zhihe, L.; Baoming, L.; Weiming, Y.; Xueyuan, B. Devolatilization characteristics of biomass at flash heating rate. Fuel 2006, 85(5–6), 664–670. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis: A review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Chianese, S.; Musmarra, D. Biofuels Production by Biomass Gasification: A Review. Energies 2018, 11, 811. [Google Scholar] [CrossRef]
- Molino, A.; Chianese, S.; Musmarra, D. Biomass gasification technology: The state of the art overview. J. Energy Chem. 2016, 25, 10–25. [Google Scholar] [CrossRef]
- Peterson, A.A.; Vogel, F.; Lachance, R.P.; Fröling, M.; Antal, M.J., Jr.; Tester, J. W. Thermochemical biofuel production in hy-drothermal media: a review of sub-and supercritical water technologies. Energy Environ. Sci. 2008, 1, 32–65. [Google Scholar] [CrossRef]
- Yin, F.; Chen, H.; Xu, G.; Wang, G.; Xu, Y. A detailed kinetic model for the hydrothermal decomposition process of sewage sludge. Bioresour. Technol. 2015, 198, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Kruse, A.; Bernolle, P.; Dahmen, N.; Dinjus, E.; Maniam, P. Hydrothermal gasification of biomass: consecutive reactions to long-living intermediates. Energy Environ. Sci. 2010, 3, 136–143. [Google Scholar] [CrossRef]
- Basu, P. Pyrolysis and Torrefaction. In Biomass Gasification and Pyrolysis, Practical Design; Basu, P., Ed.; Academic Press: London, United Kingdom, 2010; pp. 65–96. [Google Scholar] [CrossRef]
- Sánchez, J.; Curt, M.D.; Robert, N.; Fernández, J.; Lago, C.; Caldés, N.; Lechón, Y. Biomass Resources. In The Role of Bioenergy in the Bioeconomy: Resources, Technologies, Sustainability and Policy; Academic Press: London, United Kingdom, 2019; pp. 25–111. [Google Scholar] [CrossRef]
- Sukthang, N.; Liplap, P.; Arjharn, W.; Hinsui, T. The Potential of High Moisture Biomass for Energy Production Using Plasma – Assisted Gasification. Orient. J. Chem. 2017, 33, 1805–1814. [Google Scholar] [CrossRef]
- Basu, P.; Kaushal, P. Gasification theory. In Biomass Gasification, Pyrolysis, and Torrefaction, Practical Design, Theory, and Climate Change Mitigation, 4th ed.; Basu, P., Kaushal, P., Eds.; Academic Press: London, United Kingdom, 2024; pp. 207–258. [Google Scholar] [CrossRef]
- Biagini, E.; Simone, M.; Nicolella, C.; Tognotti, L. Comparative Process Study of Biomass Gasification for Power and Chemical Production. In Proceedings of the 19th European Biomass Conference and Exhibition, Berlin, Germany, 6-10 June 2011; p. 6 Pages. [Google Scholar] [CrossRef]
- Ren, J.; Cao, J.P.; Zhao, X.Y.; Yang, F.L.; Wei, X.Y. Recent advances in syngas production from biomass catalytic gasification: A critical review on reactors, catalysts, catalytic mechanisms and mathematical models. Renew. Sustain. Energy Rev. 2019, 116, 109426. [Google Scholar] [CrossRef]
- Huang, Y.F.; Kuan, W.H.; Chiueh, P.T.; Lo, S.L. Pyrolysis of biomass by thermal analysis–mass spectrometry (TA–MS). Bioresour. Technol. 2011, 102, 3527–3534. [Google Scholar] [CrossRef]
- Broer, K.M.; Peterson, C. Gasification. In Thermochemical Processing of Biomass: Conversion into Fuels, Chemicals and Power, 2nd ed.; Brown, R.C., Ed.; Wiley-VCH: Weinheim, Germany, 2019; pp. 85–123. [Google Scholar] [CrossRef]
- Kirsanovs, V.; Zandeckis, A. Investigation of Biomass Gasification Process with Torrefaction Using Equilibrium Model. Energy Procedia 2015, 72, 329–336. [Google Scholar] [CrossRef]
- Glushkov, D.; Nyashina, G.; Shvets, A.; Pereira, A.; Ramanathan, A. Current Status of the Pyrolysis and Gasification Mechanism of Biomass. Energies 2021, 14, 7541. [Google Scholar] [CrossRef]
- Shahabuddin, M.; Alam, T. Gasification of Solid Fuels (Coal, Biomass and MSW): Overview, Challenges and Mitigation Strategies. Energies 2022, 15, 4444. [Google Scholar] [CrossRef]
- Lv, D.; Xu, M.; Liu, X.; Zhan, Z.; Li, Z.; Yao, H. Effect of cellulose, lignin, alkali and alkaline earth metallic species on biomass pyrolysis and gasification. Fuel Process. Technol. 2010, 91, 903–909. [Google Scholar] [CrossRef]
- Esteves, B.; Sen, U.; Pereira, H. Influence of Chemical Composition on Heating Value of Biomass: A Review and Bibliometric Analysis. Energies 2023, 16, 4226. [Google Scholar] [CrossRef]
- Wang, S.; Song, T.; Jarolin, K.; Dymala, T.; Dosta, M.; Heinrich, S.; Shen, L.; htt James, R.A.; Yuan, W.; Boyette, M. Mixing evolution behavior of raw and gasified biomass pellets in a fluidized bed reactor. Chem. Eng. Sci. 2022, 264, 118161. htt James R., A.; Yuan, W.; Boyette, M. The Effect of Biomass Physical Properties on Top-Lit Updraft Gasification of Woodchips. Energies 2016, 9, 283. [Google Scholar] [CrossRef]
- James R., A.; Yuan, W.; Boyette, M. The Effect of Biomass Physical Properties on Top-Lit Updraft Gasification of Woodchips. Energies 2016, 9, 283. [Google Scholar] [CrossRef]
- Siddiqui, M.Z.; Sheraz, M.; Toor, U.A.; Anus, A.; Mahmood, A.; Haseeb, M.; Ibrahim, M.; Khoo, K.S.; Devadas, V.V.; Mubashir, M.; Ullah, S.; Show, P.L. Recent approaches on the optimization of biomass gasification process parameters for product H2 and syngas ratio: A review. Environ. Dev. Sustain. 2025, 27, 22605–22633. [Google Scholar] [CrossRef]
- Luo, S.; Xiao, B.; Hu, Z.; Liu, S.; Guo, X.; He, M. Hydrogen-rich gas from catalytic steam gasification of biomass in a fixed bed reactor: Influence of temperature and steam on gasification performance. Int. J. Hydrog. Energy 2009, 34, 2191–2194. [Google Scholar] [CrossRef]
- Sharma, M.; Kaushal, R. Advances and challenges in the generation of bio-based fuels using gasifiers: A comprehensive review. Int. J. Ambient Energy 2020, 41, 1645–1663. [Google Scholar] [CrossRef]
- Tremel, A.; Becherer, D.; Fendt, S.; Gaderer, M.; Spliethoff, H. Performance of entrained flow and fluidised bed biomass gasifiers on different scales. Energy Convers. Manag. 2013, 69, 95–106. [Google Scholar] [CrossRef]
- Tacuri, D.; Andrade, C.; Álvarez, P.; Abril-González, M.; Zalamea, S.; Pinos-Vélez, V.; Jara, L.; Montero-Izquierdo, A. Design and Development of a Catalytic Fixed-Bed Reactor for Gasification of Banana Biomass in Hydrogen Production. Catalysts 2022, 12, 395. [Google Scholar] [CrossRef]
- Carmo-Calado, L.; Hermoso-Orzáez, M.J.; Diaz-Perete, D.; Cal-Herrera, J.L.; Brito, P.; Terrados-Cepeda, J. Experimental Research on the Production of Hydrogen-Rich Synthesis Gas via the Air-Gasification of Olive Pomace: A Comparison between an Updraft Bubbling Bed and a Downdraft Fixed Bed. Hydrogen 2023, 4, 726–745. [Google Scholar] [CrossRef]
- Miao, Z.; Grift, T.; Hansen, A.; Ting, K. Energy requirement for comminution of biomass in relation to particle physical properties. Ind. Crops Prod. 2011, 33, 504–513. [Google Scholar] [CrossRef]
- Mak, J.; Landry, H.; Grieger, L.; Agnew, J.; Krigstin, S.; Helmeste, C.; Wetzel, S.; Madrali, S.; Volpé, S. An Assessment of Ambient and Heated Forced Air Drying Pre-treatments for Enhancing the Quality of Various Forest Biomass Feedstocks. Front. Energy Res. 2020, 8, 7. [Google Scholar] [CrossRef]
- Plis, P.; Wilk, R. Theoretical and experimental investigation of biomass gasification process in a fixed bed gasifier. Energy 2011, 36, 3838–3845. [Google Scholar] [CrossRef]
- Hughes, W.E.M.; Larson, E.D. Effect of Fuel Moisture Content on Biomass-IGCC Performance. J. Eng. Gas Turbines Power 1998, 120, 455–459. [Google Scholar] [CrossRef]
- Ruiz, J.; Juárez, M.; Morales, M.; Muñoz, P.; Mendívil, M. Biomass gasification for electricity generation: Review of current technology barriers. Renew. Sustain. Energy Rev. 2013, 18, 174–183. [Google Scholar] [CrossRef]
- Vamvuka, D.; Pitharoulis, M.; Alevizos, G.; Repouskou, E.; Pentari, D. Ash effects during combustion of lignite/biomass blends in fluidized bed. Renew. Energy 2009, 34, 2662–2671. [Google Scholar] [CrossRef]
- Bennici, S.; Jeguirim, M.; Limousy, L.; Haddad, K.; Vaulot, C.; Michelin, L.; Josien, L.; Zorpas, A.A. Influence of CO2 Concentration and Inorganic Species on the Gasification of Lignocellulosic Biomass Derived Chars. Waste Biomass Valori. 2019, 10, 3745–3752. [Google Scholar] [CrossRef]
- Dayton, D. Review of the Literature on Catalytic Biomass Tar Destruction: Milestone Completion Report (No. NREL/TP-510-32815, 15002876; p. NREL/TP-510-32815, 15002876). 2002. Available online: https://doi.org/10.2172/15002876 (accessed on 4 February 2026).
- Trubetskaya, A. Reactivity Effects of Inorganic Content in Biomass Gasification: A Review. Energies 2022, 15, 3137. [Google Scholar] [CrossRef]
- Huang, X.; Li, Y.; Ren, J.; Yang, K.; Li, R.; Bai, Y.; Deng, L.; Che, D. Experimental study on potassium catalyzed gasification of large particle size biomass with CO2. J. Energy Inst. 2023, 111, 101397. [Google Scholar] [CrossRef]
- Gil, M.; González-Vázquez, M.; García, R.; Rubiera, F.; Pevida, C. Assessing the influence of biomass properties on the gasification process using multivariate data analysis. Energy Convers. Manag. 2019, 184, 649–660. [Google Scholar] [CrossRef]
- Hirohata, O.; Wakabayashi, T.; Tasaka, K.; Fushimi, C.; Furusawa, T.; Kuchonthara, P.; Tsutsumi, A. Release behavior of tar and alkali and alkaline earth metals during biomass steam gasification. Energy Fuels 2008, 22, 4235–4239. [Google Scholar] [CrossRef]
- Phounglamcheik, A.; Vila, R.; Kienzl, N.; Wang, L.; Hedayati, A.; Brostrom, M.; Ramser, K.; Engvall, K.; Skreiberg, Ø.; Robinson, R.; Umeki, K. CO2 gasification reactivity of char from high-ash biomass. ACS omega 2021, 6, 34115–34128. [Google Scholar] [CrossRef]
- Ma, C.; Weiland, F.; Hedman, H.; Boström, D.; Backman, R.; Ohman, M. Characterization of reactor ash deposits from pilot-scale pressurized entrained-flow gasification of woody biomass. Energy Fuels 2013, 27, 6801–6814. [Google Scholar] [CrossRef]
- Ma, C.; Carlborg, M.; Hedman, H.; Wennebro, J.; Weiland, F.; Wiinikka, H.; Backman, R.; Ohman, M. Ash formation in pilot-scale pressurized entrained-flow gasification of bark and a bark/peat mixture. Energy Fuels 2016, 30, 10543–10554. [Google Scholar] [CrossRef]
- Lin, Y.; Ge, Y.; He, Q.; Chen, P.; Xiao, H. The redistribution and migration mechanism of chlorine during hydrothermal carbonization of waste biomass and fuel properties of hydrochars. Energy 2022, 244, 122578. [Google Scholar] [CrossRef]
- Corella, J.; Toledo, J.M.; Padilla, R. Olivine or dolomite as in-bed additive in biomass gasification with air in a fluidized bed: which is better? Energy Fuels 2004, 18, 713–720. [Google Scholar] [CrossRef]
- Chiang, K.Y.; Lin, M.H.; Lu, C.H.; Chien, K.L.; Lin, Y.H. Improving the synthesis gas quality in catalytic gasification of rice straw by an integrated hot-gas cleaning system. Int. J. Green Energy 2015, 12, 1005–1011. [Google Scholar] [CrossRef]
- Berdugo Vilches, T.; Seemann, M.; Thunman, H. Influence of in-bed catalysis by ash-coated olivine on tar formation in steam gasification of biomass. Energy Fuels 2018, 32, 9592–9604. [Google Scholar] [CrossRef]
- Kuba, M.; Fürsatz, K.; Janisch, D.; Aziaba, K.; Chlebda, D.; Łojewska, J.; Forsberg, F.; Umeki, K.; Hofbauer, H. Surface characterization of ash-layered olivine from fluidized bed biomass gasification. Biomass Convers. Biorefin. 2021, 11, 29–38. [Google Scholar] [CrossRef]
- Vanisree, G.S.; Chandran, A.M.; Aparna, K. Investigation on thermochemical characteristics and pyrolysis kinetics of lignocellulosic biomass for biofuel production feasibility. Biomass Convers. Biorefin. 2024, 15, 28667–28685. [Google Scholar] [CrossRef]
- Jha, P. Characterization of Biomass and Studies of Pyrolysis on Rice Husk in a Lab-Scale Pyrolyzer: A Step Toward Environmental and Energy Sustainability. In Solid Waste Management. Sustainable Development Goals Series; Nasr, M., Negm, A., Eds.; Springer: Cham, Switzerland, 2024; pp. 75–90. [Google Scholar] [CrossRef]
- Lyons Cerón, A.; Konist, A.; Lees, H.; Järvik, O. Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas. Sustainability 2021, 13, 11763. [Google Scholar] [CrossRef]
- Montagnaro, F. Biomass Gasification for Energy Production. In Sustainable Energy Production Using Solid Materials; Montagnaro, F., Ed.; Springer: Cham, Switzerland, 2024; pp. 125–142. [Google Scholar] [CrossRef]
- Yu, H.; Wu, Z.; Chen, G. Catalytic gasification characteristics of cellulose, hemicellulose and lignin. Renew. Energy 2018, 121, 559–567. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, Z.; Li, Z.; Chen, D. Characteristics of tar formation during cellulose, hemicellulose and lignin gasification. Fuel 2014, 118, 250–256. [Google Scholar] [CrossRef]
- Zhu, L.; Zhong, Z. Effects of cellulose, hemicellulose and lignin on biomass pyrolysis kinetics. Korean J. Chem. Eng. 2020, 37, 1660–1668. [Google Scholar] [CrossRef]
- Benac, D.J.; Olson, D.B.; Urzendowski, M. High-Temperature Stress Relaxation Cracking and Stress Rupture Observed in a Coke Gasifier Failure. J. Fail. Anal. and Preven. 2011, 11, 251–264. [Google Scholar] [CrossRef]
- Marcantonio, V.; Di Paola, L.; De Falco, M.; Capocelli, M. Modeling of Biomass Gasification: From Thermodynamics to Process Simulations. Energies 2023, 16, 7042. [Google Scholar] [CrossRef]
- Yao, X.; Zhou, H.; Qi, P.; Xu, K. Insights into synergistic effects of oxygen content and reaction temperature on gas product emissions and solid product deposition behaviors during biomass gasification process Biomass Convers. Biorefin 2025, 15, 4109–4124. [Google Scholar] [CrossRef]
- Lackner, M.; Fei, Q.; Guo, S.; Yang, N.; Guan, X.; Hu, P. Biomass Gasification as a Scalable, Green Route to Combined Heat and Power (CHP) and Synthesis Gas for Materials: A Review. Fuels 2024, 5, 625–649. [Google Scholar] [CrossRef]
- Guizani, C.; Escudero Sanz, F.J.; Salvador, S. Influence of temperature and particle size on the single and mixed atmosphere gasification of biomass char with H2O and CO2. Fuel Process. Technol. 2015, 134, 175–188. [Google Scholar] [CrossRef]
- Maiti, R.; Ghosh, S.; De, S. Cracking of tar by steam reforming and hydrogenation: An equilibrium model development. Biomass Convers. Biorefin. 2013, 3, 103–111. [Google Scholar] [CrossRef]
- Loweski Feliz, M.; Abdelouahed, L.; Taouk, B. Comparative and Descriptive Study of Biomass Gasification Simulations Using Aspen Plus. Energies 2024, 17, 4443. [Google Scholar] [CrossRef]
- Al-Zareer, M.; Dincer, I.; Rosen, M.A. Influence of Selected Gasification Parameters on Syngas Composition From Biomass Gasification. J. Energy Resour. Technol. 2018, 140, 041803. [Google Scholar] [CrossRef]
- Mayerhofer, M.; Mitsakis, P.; Meng, X.; De Jong, W.; Spliethoff, H.; Gaderer, M. Influence of pressure, temperature and steam on tar and gas in allothermal fluidized bed gasification. Fuel 2012, 99, 204–209. [Google Scholar] [CrossRef]
- Ramalingam, S.; Rajendiran, B.; Subramiyan, S. Recent advances in the performance of Co-Current gasification technology: A review. Int. J. Hydrog. Energy 2020, 45, 230–262. [Google Scholar] [CrossRef]
- Mishra, S.; Upadhyay, R.K. Review on biomass gasification: Gasifiers, gasifying mediums, and operational parameters. Mater. Sci. Energy Technol. 2021, 4, 329–340. [Google Scholar] [CrossRef]
- Dasappa, S. Thermochemical Conversion of Biomass. In Transformation of Biomass: Theory to Practice; Hornung, A., Ed.; John Wiley & Sons, Ltd: New Jersey, USA, 2014; pp. 133–157. [Google Scholar] [CrossRef]
- Jangsawang, W.; Laohalidanond, K.; Kerdsuwan, S. Optimum Equivalence Ratio of Biomass Gasification Process Based on Thermodynamic Equilibrium Model. Energy Procedia 2015, 79, 520–527. [Google Scholar] [CrossRef]
- Jayathilake, R.; Rudra, S. Numerical and Experimental Investigation of Equivalence Ratio (ER) and Feedstock Particle Size on Birchwood Gasification. Energies 2017, 10, 1232. [Google Scholar] [CrossRef]
- Beheshti, S.M.; Ghassemi, H.; Shahsavan-Markadeh, R.J.E.C. Process simulation of biomass gasification in a bubbling fluidized bed reactor. Energy Convers. Manag. 2015, 94, 345–352. [Google Scholar] [CrossRef]
- Abdoulmoumine, N.; Kulkarni, A.; Adhikari, S. Effects of temperature and equivalence ratio on pine syngas primary gases and contaminants in a bench-scale fluidized bed gasifier. Ind. Eng. Chem. Res. 2014, 53, 5767–5777. [Google Scholar] [CrossRef]
- Ismail, T.M.; El-Salam, M.A. Parametric studies on biomass gasification process on updraft gasifier high temperature air gasification. Appl. Therm. Eng. 2017, 112, 1460–1473. [Google Scholar] [CrossRef]
- Rupesh, S.; Muraleedharan, C.; Arun, P. Energy and exergy analysis of syngas production from different biomasses through air-steam gasification. Front. Energy 2020, 14, 607–619. [Google Scholar] [CrossRef]
- Latifi, M.; Ferrante, L.; Briens, C.; Berruti, F. Effect of Residence Time and Temperature on Thermal Cracking of Bio-oil for Syngas Production. Bioenergy- II: Fuels and Chemical from Renewable Resources, 2009. Available online: https://publications.polymtl.ca/50681/ (accessed on 4 February 2026).
- Hernández, J.J.; Aranda-Almansa, G.; Bula, A. Gasification of biomass wastes in an entrained flow gasifier: Effect of the particle size and the residence time. Fuel Process. Technol. 2010, 91, 681–692. [Google Scholar] [CrossRef]
- Guo, L.; Cao, C.; Lu, Y. Supercritical Water Gasification of Biomass and Organic Wastes. In Biomass; Benteke Momba, M.N., Ed.; IntechOpen Limited: London, United Kingdom, 2010; pp. 1–20. [Google Scholar] [CrossRef]
- Mehmood, A.; Tahir, M.W.; Saeed, M.A.; Arshad, M.Y.; Hussain, H.; Mularski, J.; Niedzwiecki, L. Optimization of Gasifying Agents in 3D Downdraft Gasification for Enhanced Gas Composition, Combustion, and CO2 Utilization. Fire 2023, 6, 361. [Google Scholar] [CrossRef]
- Natarajan, A.; Venugopal, D.; Thangavelu, L. Investigation of biomass gasification simulation using air, steam and oxygen as gasifying agent. Thermal Science 2022, 26, 5109–5119. [Google Scholar] [CrossRef]
- Salem, A.M.; Paul, M.C. Effects of throat sizing and gasification agents in a biomass downdraft gasifier: Towards CO2 -free syngas production. RSC Advances 2023, 13, 10221–10238. [Google Scholar] [CrossRef]
- Sandeep, K.; Dasappa, S. First and second law thermodynamic analysis of air and oxy-steam biomass gasification. Int. J. Hydrog. Energy 2014, 39, 19474–19484. [Google Scholar] [CrossRef]
- Butterman, H.C.; Castaldi, M.J. Influence of CO2 injection on biomass gasification. Ind. Eng. Chem. Res. 2007, 46(26), 8875–8886. [Google Scholar] [CrossRef]
- Mauerhofer, A.M.; Müller, S.; Bartik, A.; Benedikt, F.; Fuchs, J.; Hammerschmid, M.; Hofbauer, H. Conversion of CO2 during the DFB biomass gasification process. Biomass Convers. Biorefin. 2021, 11, 15–27. [Google Scholar] [CrossRef]
- Prabowo, B.; Umeki, K.; Yan, M.; Nakamura, M.R.; Castaldi, M.J.; Yoshikawa, K. CO2–steam mixture for direct and indirect gasification of rice straw in a downdraft gasifier: Laboratory-scale experiments and performance prediction. Appl. Energy 2014, 113, 670–679. [Google Scholar] [CrossRef]
- Sadhwani, N.; Adhikari, S.; Eden, M.R. Biomass gasification using carbon dioxide: effect of temperature, CO2/C ratio, and the study of reactions influencing the process. Ind. Eng. Chem. Res. 2016, 55(10), 2883–2891. [Google Scholar] [CrossRef]
- Kogler, A.; Rauch, N.; Bönisch, S.; Derntl, M.; Kofler, I.; Haider, A. Post-combustion CO2 Capture Using Amine-scrubbing at an Integrated Steel Mill. Berg Huettenmaenn Monatsh 2025, 170, 428–433. [Google Scholar] [CrossRef]
- Romano, M.C.; Spinelli, M.; Campanari, S.; Consonni, S.; Cinti, G.; Marchi, M.; Borgarello, E. The Calcium Looping Process for Low CO2 Emission Cement and Power. Energy Procedia 2013, 37, 7091–7099. [Google Scholar] [CrossRef]
- Schmid, M.; Beirow, M.; Schweitzer, D.; Waizmann, G.; Spörl, R.; Scheffknecht, G. Product gas composition for steam-oxygen fluidized bed gasification of dried sewage sludge, straw pellets and wood pellets and the influence of limestone as bed material. Biomass Bioenerg. 2018, 117, 71–77. [Google Scholar] [CrossRef]
- Wei, L.; Xu, S.; Liu, J.; Liu, C.; Liu, S. Hydrogen production in steam gasification of biomass with CaO as a CO2 absorbent. Energy Fuels 2008, 22, 1997–2004. [Google Scholar] [CrossRef]
- Shang, Q.; Feng, D.; Yan, P.; Li, Y.; Dong, L.; Wang, S.; Zhang, X.; Zhao, Y.; Sun, S. In-situ CO2 capture by DFMs to enhance hydrogen production and regeneration performance of biomass-H2O gasification. Appl. Energy Combust. Sci. 2025, 24, 100420. [Google Scholar] [CrossRef]
- Long, Y.; Wang, C.; Xu, R.; Liu, L.; Zeng, P.; Zhou, Z.; Xu, M. Dual Effects of In Situ Coal Combustion on CaO Pellets for CO2 Capture: High-Temperature Sintering and Ash Stabilization. Int. J. Mol. Sci. 2025, 26, 8535. [Google Scholar] [CrossRef]
- Zhang, S.; Gao, N.; Quan, C.; Wang, F.; Wu, C. Autothermal CaO looping biomass gasification to increase process energy efficiency and reduce ash sintering. Fuel 2020, 277, 118199. [Google Scholar] [CrossRef]
- Chen, M.; Li, H.; Wang, L.; Bi, X. Experimental Study on Enhanced Gasification of Biomass and Simulation of a CO2 Adsorption Mechanism Based on a Modified Ca-Based Catalyst. Energy Fuels 2025, 39, 3157–3168. [Google Scholar] [CrossRef]
- Cao, Y.; Bai, Y.; Du, J. CaO-based sorption-enhanced steam gasification of biomass for high purity H2 production: a modeling approach. Biomass Convers. Biorefin. 2025, 15, 13817–13829. [Google Scholar] [CrossRef]
- Santos, S.M.; Assis, A.C.; Gomes, L.; Nobre, C.; Brito, P. Waste Gasification Technologies: A Brief Overview. Waste 2023, 1, 140–165. [Google Scholar] [CrossRef]
- Frolov, S.M. Organic Waste Gasification: A Selective Review. Fuels 2021, 2, 556–650. [Google Scholar] [CrossRef]
- Allesina, G.; Pedrazzi, S. Barriers to Success: A Technical Review on the Limits and Possible Future Roles of Small Scale Gasifiers. Energies 2021, 14, 6711. [Google Scholar] [CrossRef]
- Gubin, V.; Kadlez, D.; Bartik, A.; Steiner, L.; Zeitlhofer, J.; Thelen, F.; Hofbauer, H.; Müller, S. Hydrogen production from woody biomass via fixed-bed gasification at pilot-scale. Int. J. Hydrogen Energ. 2025, 114, 462–474. [Google Scholar] [CrossRef]
- HajiHashemi, M.; Mazhkoo, S.; Dadfar, H.; Livani, E.; Varnosefaderani, A.N.; Pourali, O.; Nobar, S.N.; Dutta, A. Combined heat and power production in a pilot-scale biomass gasification system: experimental study and kinetic simulation using ASPEN Plus. Energy 2023, 276, 127506. [Google Scholar] [CrossRef]
- Mazhkoo, S.; Dadfar, H.; HajiHashemi, M.; Pourali, O. A comprehensive experimental and modeling investigation of walnut shell gasification process in a pilot-scale downdraft gasifier integrated with an internal combustion engine. Energy Convers. Manag. 2021, 231, 113836. [Google Scholar] [CrossRef]
- Gunarathne, D.S.; Mueller, A.; Fleck, S.; Kolb, T.; Chmielewski, J.K.; Yang, W.; Blasiak, W. Gasification characteristics of steam exploded biomass in an updraft pilot scale gasifier. Energy 2014, 71, 496–506. [Google Scholar] [CrossRef]
- Guerrero, J.; Carmona-Martínez, A.; Jarauta-Córdoba, C. Renewable syngas production and technoeconomic validation in a pilot-scale reactor of air and air-steam gasification of biomass. Int. J. Hydrogen Energ. 2024, 93, 273–284. [Google Scholar] [CrossRef]
- Pio, D.; Gomes, H.; Tarelho, L.; Vilas-Boas, A.; Matos, M.; Lemos, F. Superheated steam injection as primary measure to improve producer gas quality from biomass air gasification in an autothermal pilot-scale gasifier. Renew. Energy 2022, 181, 1223–1236. [Google Scholar] [CrossRef]
- Di Carlo, A.; Savuto, E.; Foscolo, P.U.; Papa, A.A.; Tacconi, A.; Del Zotto, L.; Aydin, B.; Bocci, E. Preliminary Results of Biomass Gasification Obtained at Pilot Scale with an Innovative 100 kWth Dual Bubbling Fluidized Bed Gasifier. Energies 2022, 15, 4369. [Google Scholar] [CrossRef]
- Emiola-Sadiq, T.; Zhang, L.; Dalai, A.; Gerspacher, R.; Campbell, B.; Evitts, R. Parametric and hydrodynamics studies on gasification performance of biomass pellets in a pilot-scale fluidized bed gasifier. Biomass Convers. Biorefin. 2024, 14, 16339–16361. [Google Scholar] [CrossRef]
- Ismail, T.M.; Ramos, A.; Monteiro, E.; El-Salam, M.A.; Rouboa, A. Parametric studies in the gasification agent and fluidization velocity during oxygen-enriched gasification of biomass in a pilot-scale fluidized bed: Experimental and numerical assessment. Renew. Energy 2020, 147, 2429–2439. [Google Scholar] [CrossRef]
- Koppatz, S.; Pfeifer, C.; Hofbauer, H. Comparison of the performance behaviour of silica sand and olivine in a dual fluidised bed reactor system for steam gasification of biomass at pilot plant scale. Chem. Eng. J. 2011, 175, 468–483. [Google Scholar] [CrossRef]
- Pitkäoja, A.; Ritvanen, J. Steam-oxygen blown circulating fluidised bed gasification for synthetic biofuel production: Pilot-scale reactor modelling, model-based reactor scale-up and analysis for power-biomass-to-liquid processes. Biomass Bioenerg. 2025, 193, 107540. [Google Scholar] [CrossRef]
- Freda, C.; Nanna, F.; Villone, A.; Barisano, D.; Brandani, S.; Cornacchia, G. Air gasification of digestate and its co-gasification with residual biomass in a pilot scale rotary kiln. Int. J. Energy Environ. Eng. 2019, 10, 335–346. [Google Scholar] [CrossRef]
- Placido, A.; Challman, D.; Liu, K.; Andrews, R.; Jacobs, G.; Davis, B.; Ma, W.; Darkwah, K. Small-scale pilot plant for gasification of coal and coal/biomass blends and conversion of derived syngas to liquid fuels via Fischer-Tropsch synthesis (No. DOE-UK-10482). Univ. of Kentucky, Lexington, KY (United States) 2018. Available online: https://www.osti.gov/servlets/purl/1458389 (accessed on 4 February 2026).
- Heyne, S.; Liliedahl, T.; Marklund, M. Biomass gasification-a synthesis of technical barriers and current research issues for deployment at large scale, 2013. Available online: https://www.osti.gov/etdeweb/biblio/22138974 (accessed on 4 February 2026).
- Asadullah, M. Barriers of commercial power generation using biomass gasification gas: A review. Renew. Sustain. Energy Rev. 2014, 29, 201–215. [Google Scholar] [CrossRef]
- Aries Clean Energy. Available online: https://ariescleantech.com/wp-content/uploads/2019/01/Aries-Clean-Energy-Lebanon-Case-Study-2019.pdf (accessed on 4 February 2026).
- URBAS Energy Technology. Available online: https://www.urbas.at/wp-content/uploads/2020/09/p_URBAS_DE_KWK_7.8.web_WF.PDF-en.pdf (accessed on 4 February 2026).
- Teislev, B. Harboore – woodchips updraft gasifier and 1500 kw gas-engines operating at 32% power efficiency in chp configuration, 2022. Available online: https://task33.ieabioenergy.com/wp-content/uploads/sites/33/2022/06/BWVgasifiersDOC.pdf (accessed on 4 February 2026).
- Neoelectra. Available online: https://neoelectra.es/?lang=en (accessed on 4 February 2026).
- Valmet. Available online: https://www.valmet.com/energyproduction/gasification/ (accessed on 4 February 2026).
- Sumitomo SHI/FW. Available online: https://www.shi-fw.com/our-solutions/gasification/ (accessed on 4 February 2026).
- GEMCO Energy. Turn Waste/Biomass Materials into Green Energy. Available online: https://gasificationplant.com/ (accessed on 4 February 2026).
- SynTech Bioenergy. Available online: https://www.syntechbioenergy.com/biomax (accessed on 4 February 2026).
- Ankur Scientific Energy Technologies. Available online: https://www.ankurscientific.com/ankur-gasifiers-biomass-woody.html (accessed on 4 February 2026).
- CMN Industry Inc. Available online: https://regenerativethermaloxidiser.com/product/biomass-gasifier/ (accessed on 4 February 2026).
- Powermax Renewable Energy. Available online: https://www.powermaxgasifiers.com/ (accessed on 4 February 2026).
- Compact Syngas Solutions. Available online: https://www.syngas-solutions.co.uk/ (accessed on 4 February 2026).
- SPANNER RE² GMBH. Available online: https://re2.energy/en/ (accessed on 4 February 2026).
- Meva Energy. Available online: https://mevaenergy.com/ (accessed on 4 February 2026).
- Meva Energy. Available online: https://mevaenergy.com/news/ (accessed on 4 February 2026).
- Syncraft®. Available online: https://www.syncraft.at/en/ (accessed on 4 February 2026).
- The Stadtwerke Rosenheim. Available online: https://www.swro.de/de/unternehmen/erzeugungsanlagen/holzvergaser (accessed on 4 February 2026).
- Kombi Power System. Available online: https://regawatt.de/en/kps-uebersicht/ (accessed on 4 February 2026).
- Lipro Energy. Available online: https://lipro-energy.de/en/products/technology/ (accessed on 4 February 2026).
- Holzenergie. Available online: https://www.hw-powerplants.de/technologie (accessed on 4 February 2026).
- Burkhardt GmbH. Available online: https://burkhardt-gruppe.de/en/power-engineering/heat-and-power-from-wood/wood-gas-generator/wood-gasifier-v-3-90s/ (accessed on 4 February 2026).
- CMD (Costruzioni Motori Diesel SPA). Available online: https://eco20cmd.com/en/eco20x/ (accessed on 4 February 2026).
- ESPE Energy expertise. Available online: https://www.espe.it/en/product/chip50-biomass-cogenerator/ (accessed on 4 February 2026).
- RESET. Available online: https://www.reset-energy.com/en/syngasmart/ (accessed on 4 February 2026).
- VOLTER. Available online: https://volter.fi/en/volter-products/technology/ (accessed on 4 February 2026).
- Xylergy SA. Available online: https://xylergy-group.com/en/ (accessed on 4 February 2026).
- Yadav, S.; Choudhary, A.K.; Yadav, P.; Pal, D.B. Lignocellulosic biomass gasification for bio-circular economy sustainability: a multiple criteria analysis framework. Biomass Convers. Biorefin. 2025, 15, 30667–30677. [Google Scholar] [CrossRef]
- Dhamodharan, K.; Konduru, T.; Kannan, M.; Malyan, S.K. Techno-economic feasibility and hurdles on agricultural waste management. In Emerging trends to approaching zero waste; Hussain, C.M., Singh, S., Goswami, L., Eds.; Elsevier Inc: Amsterdam, Netherlands, 2022; pp. 243–264. [Google Scholar] [CrossRef]
- Tian, Y.; Luo, Z.; He, D.; Yang, Y.; Liang, S.; Liu, W.; Yuan, L. Co-gasification of biomass and polyethylene: a simulation study by considering tar formation. Biomass Convers. Biorefin. 2024, 14, 4081–4089. [Google Scholar] [CrossRef]
- Liu, Q.; Sun, J.; Gu, Y.; Zhong, W.; Gao, K. Experimental study on CO2 co-gasification characteristics of biomass and waste plastics: Insight into interaction and targeted regulation method. Energy 2024, 292, 130509. [Google Scholar] [CrossRef]
- Patuzzi, F.; Basso, D.; Vakalis, S.; Antolini, D.; Piazzi, S.; Benedetti, V.; Cordioli, E.; Baratieri, M. State-of-the-art of small-scale biomass gasification systems: An extensive and unique monitoring review. Energy 2021, 223, 120039. [Google Scholar] [CrossRef]
- Eberhard, M.; Santo, U.; Michelfelder, B.; Günther, A.; Weigand, P.; Matthes, J.; Waibel, P.; Hagenmeyer, V.; Kolb, T. The bioliq® Entrained-Flow Gasifier – A Model for the German Energiewende. ChemBioEng Rev. 2020, 7, 106–118. [Google Scholar] [CrossRef]
- Menin, L.; Paolillo, A.; Piazzi, S.; Antolini, D.; Ravazzolo, F.; Baratieri, M. Biomass Derived Combined Heat and Power from Decentralized Small-Scale Gasification: Updated Cost Conditions for the Italian Mountain Context and Competitiveness in Future Energy Markets. Waste Biomass Valor. 2025, 16, 4009–4025. [Google Scholar] [CrossRef]
- Desclaux, L.; Pereira, A.O., Jr. Residual Biomass Gasification for Small-Scale Decentralized Electricity Production: Business Models for Lower Societal Costs. Energies 2024, 17, 1868. [Google Scholar] [CrossRef]
- Sharara, M.; Kim, D.; Sadaka, S.; Thoma, G. Consequential Life Cycle Assessment of Swine Manure Management within a Thermal Gasification Scenario. Energies 2019, 12, 4081. [Google Scholar] [CrossRef]
- González, R.; González-Rojo, S.; Gómez, X. Integrating Gasification into Conventional Wastewater Treatment Plants: Plant Performance Simulation. Eng 2025, 6, 100. [Google Scholar] [CrossRef]
- Yao, Z.; Li, W.; Kan, X.; Dai, Y.; Tong, Y.W.; Wang, C. Anaerobic digestion and gasification hybrid system for potential energy recovery from yard waste and woody biomass. Energy 2017, 124, 133–145. [Google Scholar] [CrossRef]
- Sanke, N.; Ravi Kumar, R.; Pasha, M.A.; Ammineni, S.P.; Saidulu, P. Influence of operational parameters on downdraft gasifier performance using municipal solid waste (MSW)-biomass blends. Energy Sources, Part A 2025, 47, 1–17. [Google Scholar] [CrossRef]
- Ani, P.C.; Al-Abedi, H.J.; Smith, J.D.; Zeitoun, Z. Comparative Morphological and Thermal Analysis of Biochar from Oak, and from Oak, Pine and RDF Blends, in a Downdraft Gasifier. Fuels 2025, 6, 73. [Google Scholar] [CrossRef]
- Migliaccio, R.; Brachi, P.; Montagnaro, F.; Papa, S.; Tavano, A.; Montesarchio, P.; Ruoppolo, G.; Urciuolo, M. Sewage sludge gasification in a fluidized bed: Experimental investigation and modeling. Ind. Eng. Chem. Res. 2021, 60, 5034–5047. [Google Scholar] [CrossRef]
- Ewald, A.; Spliethoff, H.; Fendt, S. Entrained Flow Gasification of Sewage Sludge─ Performance Parameters and the Fate of Phosphorus, Potassium, Sulfur, and Heavy Metals. Energy Fuels 2025, 39, 2616–2629. [Google Scholar] [CrossRef]
- Werle, S.; Sobek, S. Gasification of sewage sludge within a circular economy perspective: a Polish case study. Environ. Sci. Pollut. Res. 2019, 26, 35422–35432. [Google Scholar] [CrossRef]
- Moles, S.; Martinez, I.; Soledad Callén, M.; Gómez, J.; Manuel López, J.; Murillo, R. Pilot-scale study of sorption-enhanced gasification of sewage sludge. Fuel 2024, 360, 130611. [Google Scholar] [CrossRef]
- Schmid, M.; Hafner, S.; Scheffknecht, G. Experimental Parameter Study on Synthesis Gas Production by Steam-Oxygen Fluidized Bed Gasification of Sewage Sludge. Appl. Sci. 2021, 11, 579. [Google Scholar] [CrossRef]
- Ewald, A.; Roeder, G.; Spliethoff, H.; Fendt, S. Pulverized Sewage Sludge Combustion with Potassium Chloride Addition─ Fate of Phosphorus, Potassium, Sulfur, and Heavy Metals. ACS omega 2025, 10, 25733–25745. [Google Scholar] [CrossRef]
- Chen, G.; Guo, X.; Cheng, Z.; Yan, B.; Dan, Z.; Ma, W. Air gasification of biogas-derived digestate in a downdraft fixed bed gasifier. Waste Manag. 2017, 69, 162–169. [Google Scholar] [CrossRef]
- Chang, S.; Zhang, Z.; Cao, L.; Ma, L.; You, S.; Li, W. Co-gasification of digestate and lignite in a downdraft fixed bed gasifier: Effect of temperature. Energy Convers. Manag. 2020, 213, 112798. [Google Scholar] [CrossRef]
- Elbl, P.; Baláš, M.; Lisý, M.; Lisá, H. Sewage sludge and digestate gasification in an atmospheric fluidized bed gasifier. Biomass Convers. Biorefi. 2024, 14, 21821–21829. [Google Scholar] [CrossRef]
- Nam, H.; Maglinao, A.L.; Capareda, S.C.; Rodriguez-Alejandro, D.A. Enriched-air fluidized bed gasification using bench and pilot scale reactors of dairy manure with sand bedding based on response surface methods. Energy 2016, 95, 187–199. [Google Scholar] [CrossRef]
- Ongen, A.; Ozcan, H.K.; Ozbaş, E.E.; Aydin, S.; Yesildag, I. Co-gasification of oily sludge and chicken manure in a laboratory-scale updraft fixed bed gasifier. Clean Technol. Environ. Policy 2022, 24, 2229–2239. [Google Scholar] [CrossRef]
- Ng, W.C.; You, S.; Ling, R.; Gin, K.Y.H.; Dai, Y.; Wang, C. Co-gasification of woody biomass and chicken manure: Syngas production, biochar reutilization, and cost-benefit analysis. Energy 2017, 139, 732–742. [Google Scholar] [CrossRef]
- De Priall, O.; Brandoni, C.; Gogulancea, V.; Jaffar, M.; Hewitt, N.J.; Zhang, K.; Huang, Y. Gasification of biowaste based on validated computational simulations: a circular economy model to handle poultry litter waste. Waste Biomass Valori. 2022, 13, 3899–3911. [Google Scholar] [CrossRef]
- Judex, J.W.; Gaiffi, M.; Burgbacher, H.C. Gasification of dried sewage sludge: Status of the demonstration and the pilot plant. Waste Manag. 2012, 32, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, S.; Karaeva, J.; Kovalev, A.; Kovalev, D.; Litti, Y. Steam gasification of digestate after anaerobic digestion and dark fermentation of lignocellulosic biomass to produce syngas with high hydrogen content. Int. J. Hydrogen Energ. 2023, 48, 7559–7568. [Google Scholar] [CrossRef]
- Gómez, X.; Blanco, D.; Lobato, A.; Calleja, A.; Martínez-Núñez, F.; Martin-Villacorta, J. Digestion of cattle manure under mesophilic and thermophilic conditions: characterization of organic matter applying thermal analysis and 1H NMR. Biodegradation 2011, 22, 623–635. [Google Scholar] [CrossRef]
- Fernández-Domínguez, D.; Guilayn, F.; Patureau, D.; Jimenez, J. Characterising the stability of the organic matter during anaerobic digestion: A selective review on the major spectroscopic techniques. Rev. Environ. Sci. Biotechnol. 2022, 21, 691–726. [Google Scholar]
- Shahbaz, M.; Yusup, S.; Inayat, A.; Patrick, D.O.; Ammar, M.; Pratama, A. Cleaner production of hydrogen and syngas from catalytic steam palm kernel shell gasification using CaO sorbent and coal bottom ash as a catalyst. Energy Fuels 2017, 31, 13824–13833. [Google Scholar] [CrossRef]
- Bunma, T.; Kuchonthara, P. Synergistic study between CaO and MgO sorbents for hydrogen rich gas production from the pyrolysis-gasification of sugarcane leaves. Process Saf. Environ. Prot. 2018, 118, 188–194. [Google Scholar] [CrossRef]
- Asadullah, M.; Ito, S.I.; Kunimori, K.; Tomishige, K. Role of catalyst and its fluidization in the catalytic gasification of biomass to syngas at low temperature. Ind. Eng. Chem. Res. 2002, 41, 4567–4575. [Google Scholar] [CrossRef]
- Ruoppolo, G.; Landi, G. Towards Biomass Gasification Enhanced by Structured Iron-Based Catalysts. Fuels 2021, 2, 546–555. [Google Scholar] [CrossRef]
- Lysne, A.; Saxrud, I.; Madsen, K.Ø.; Blekkan, E.A. Steam Reforming of Tar Impurities from Biomass Gasification with Ni-Co/Mg(Al)O Catalysts—Operating Parameter Effects. Fuels 2024, 5, 458–475. [Google Scholar] [CrossRef]
- Silva, J.; Rocha, C.; Soria, M.A.; Madeira, L.M. Catalytic Steam Reforming of Biomass-Derived Oxygenates for H2 Production: A Review on Ni-Based Catalysts. ChemEngineering 2022, 6, 39. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, H.; Liu, J.; Chen, D. Catalytic characteristics of innovative Ni/slag catalysts for syngas production and tar removal from biomass pyrolysis. Int. J. Hydrogen Energ. 2019, 44, 11848–11860. [Google Scholar] [CrossRef]
- Valizadeh, S.; Hakimian, H.; Cho, E.H.; Ko, C.H.; Lee, S.H.; Rhee, G.H.; Jung, S.; Yoo, K.; Park, Y. Production of H2- and CO-rich syngas from the CO2 gasification of cow manure over (Sr/Mg)-promoted-Ni/Al2O3 catalysts. Int. J. Hydrogen Energ. 2022, 47, 37218–37226. [Google Scholar] [CrossRef]
- Li, S.; Yue, O.; Liu, Y.; Liu, J.; Cheng, Q. Co-pyrolysis of rice husk and cattle manure in N2/CO2 atmosphere. Energy Sources, Part A 2024, 46, 12206–12217. [Google Scholar] [CrossRef]
- Cerone, N.; Zimbardi, F.; Contuzzi, L.; Striūgas, N.; Eimontas, J.; Zito, G.D. Production of hydrogen-rich syngas from biomass gasification by double step steam catalytic tar reforming. Int. J. Hydrogen Energ. 2024, 95, 1215–1221. [Google Scholar] [CrossRef]
- Soomro, A.; Chen, S.; Ma, S.; Xiang, W. Catalytic activities of nickel, dolomite, and olivine for tar removal and H2-enriched gas production in biomass gasification process. Energy Environ. 2018, 29, 839–867. [Google Scholar] [CrossRef]
- Klinghoffer, N.B.; Castaldi, M.J.; Nzihou, A. Catalyst properties and catalytic performance of char from biomass gasification. Ind. Eng. Chem. Res. 2012, 51, 13113–13122. [Google Scholar] [CrossRef]
- Shen, Y.; Areeprasert, C.; Prabowo, B.; Takahashi, F.; Yoshikawa, K. Metal nickel nanoparticles in situ generated in rice husk char for catalytic reformation of tar and syngas from biomass pyrolytic gasification. RSC Adv. 2014, 4, 40651–40664. [Google Scholar] [CrossRef]
- Yang, H.; Cui, Y.; Jin, Y.; Lu, X.; Han, T.; Sandstrom, L.; Jönsson, P.G.; Yang, W. Evaluation of engineered biochar-based catalysts for syngas production in a biomass pyrolysis and catalytic reforming process. Energy Fuels 2023, 37, 5942–5952. [Google Scholar] [CrossRef]
- Ramadhani, B.; Kivevele, T.; Kihedu, J.H.; Jande, Y.A. Catalytic tar conversion and the prospective use of iron-based catalyst in the future development of biomass gasification: a review. Biomass Convers. Biorefin. 2022, 12, 1369–1392. [Google Scholar] [CrossRef]
- Jothiprakash, G.; Balasubramaniam, P.; Sundaram, S.; Ramesh, D. Catalytic Biomass Gasification for Syngas Production: Recent Progress in Tar Reduction and Future Perspectives. Biomass 2025, 5, 37. [Google Scholar] [CrossRef]
- Gebhard, W.; Zant, S.; Neidel, J.; Apfelbacher, A.; Daschner, R. Conversion of Sewage Sludge with Combined Pyrolysis and Gasification via the Enhanced Carbon-To-X-Output Technology. Biomass 2025, 5, 28. [Google Scholar] [CrossRef]
| HHV Correlations | References |
|---|---|
| HHV (MJ/kg) = 0.3491 C + 1.1783 H + 0.1005 S – 0.1034 O – 0.0151 N – 0.0211 Ash | [25] |
| HHV (kJ/kg) = 35430 – 183.5 VM 1 – 354.3 Ash | [27] |
| HHV (kJ/kg) = 1.87 C2 – 144 C – 2820 H + 63.8 C H + 129 N + 20147 HHV (kJ/kg) = 5.22 C2 – 319 C – 1647 H + 38.6 C H + 133 N + 21028 Average model for plant biomass: HHV (kJ/kg) = 3.55 C2 – 232 C – 2230 H + 51.2 C H + 131 N + 20600 |
[28] |
| HHV (MJ/kg) = 0.3536 FC 2 + 0.1559 VM – 0.0078 Ash | [29] |
| HHV (MJ/kg) = 19.914 – 0.2324 Ash HHV (MJ/kg) = -1.3675 + 0.3137 C + 0.7009 H + 0.0318 O |
[30] |
| Biomass | Moisture (%) | Proximate analysis (%) | LHV (MJ/kg) | HHV (MJ/kg) | Ultimate analysis (%) | Refences | ||||||
| VM | Ash | FC | C | H | N | O | S | |||||
| Salix Taxa | 7.48±1.26 | 74.84±0.94 | 2.8±0.62 | 20.31±0.68 | 17.76±0.44 | 19.35±0.33 | 49.97±0.78 | 5.76±0.32 | 0.05±0.01 | [57] | ||
| Black locust | 80.94 | 0.8 | 18.26 | 19.71 | 50.73 | 5.71 | 0.57 | 41.93 | 0.01 | [58] | ||
| SRC Willow Chips | 2.9 | 82.5 | 1.7 | 12.9 | 4.4 1 | 45.4 | 5.7 | 0.8 | 48 | 0.1 | [59] | |
| Eucalyptus Urosemente | 12.40 | 59.36 | 11.28 | 16.96 | 53.24 | 6.36 | 0.12 | 40.14 | 0.14 | [60] | ||
| Pine Trunk | 11.5 | 83.7 | 0.8 | 15.5 | 47.3 | 6.4 | 0.13 | 45.18 | 0.99 | [23] | ||
| Eucalyptus (Bark) | 3.4 | 89.6 | 4.2 | 6.2 | 13.8 | 15.2 | 43.10 | 6.46 | 0..25 | 50.18 | 0.01 | [23] |
| Eucalyptus (Trunk) | 2.1 | 88.9 | 1.0 | 10.1 | 16.0 | 17.6 | 47.20 | 7.03 | 0.11 | 45.65 | 0.01 | [23] |
| Eucalyptus (Branches) | 6.7 | 82.9 | 3.2 | 13.9 | 20.7 | 22.3 | 55.90 | 7.55 | 1.44 | 35.10 | 0.01 | [23] |
| Pine Cone | 73.98 | 2.61 | 23.41 | 43.99 | 3.65 | 1.67 | 50.18 | 0.51 | [61] | |||
| Poplar | 82.21 ±0.32 | 1.29 ±0.04 | 16.5±0.317 | 16.94±0.37 | 18.61±0.55 | 44.15 ±1.04 | 3.92± 0.23 | 0.642 ±03 | [62] | |||
| Hardwood, Chestnut | 74.2 | 0.7 | 25.1 | 18.44 | 49.74 | 5.28 | 0.19 | 44.45 | [63] | |||
| Wood Pellet | 77.82 | 1.12 | 18.635 | 46.80 | 5.61 | 0.26 | 47.33 | 0.06 | [64] | |||
| Western red cedar (WRC), | 93.35±0.51 | 0.45±0.05 | 6.20±0.32 | 19.68±0.20 | 49.35±0.61 | 6.02±0.05 | <0.5% | 44.64±0.66 | <0.5% | [65] | ||
| Forest wood/Gua Zumaumlifolia (Northern Colombia) | 7.99 | 80.92 | 1.8 | 17.28 | 16.82±0.91 | 49.1±0.7 | 5.8±0.2 | ND | 45.1±0.5 | [66] | ||
| Forest wood Cordia alliodora (Northern Colombia) | 7.41 | 81.75 | 1.53 | 16.72 | 18.16±3.8 | 49.2±0.2 | 5.8±0.1 | ND | 45.1±0.1 | [66] | ||
| Forest wood Eucalyptus Grandis (Central Colombia) | 10.28 | 81.66 | 1.17 | 17.17 | 17.80±0.9 | 51.0±0.2 | 6.1±0.1 | ND | 42.9±0.3 | [66] | ||
| Firewood Pinus Patula (Southern Colombia) | 8.55 | 84.11 | 0.4 | 15.49 | 17.64±1.43 | 47.2±0.6 | 6.2±0.1 | 0.3±0.01 | 46.3±0.4 | [66] | ||
| Sugarcane Bagasse | 81.86±1.01 | 2.04±0.01 | 15.98±0.32 | 16.79±0.04 | 42.09±0.21 | 5.42±0.08 | 0.18±0.04 | 51.50±0.33 | 0.12±0.01 | [67] | ||
| Tobacco | 1.9-5.4 | 61.9-68.0 | 15.9-18.1 | 9.9-16.0 | 35.8-42.5 | 5.4-6.2 | 2.6 | [68] | ||||
| Eucalyptus Leaves | 3.57 | 61.7 | 8.36 | 26.37 | 32.81 | 89.17 | 7.56 | 1.01 | 1.98 | 0.28 | [69] | |
| Sawdust | 7.0 | 70.0 | 2.3 | 20.7 | 18.2 | 46.23 | 6.14 | 2.2 | 45.42 | <0.01 | [70] | |
| Firewood Sawdust | 12.18 | 74.69 | 0.42 | 12.71 | 17.7 | 45.51 | 5.83 | 0.001 | 48.66 | [71] | ||
| dewatered poultry sludge | 67.50 | 25.09 | 7.41 | 51.13 | 7.67 | 4.24 | 11.53 | 0.34 | [72] | |||
| Chicken Manure | 70.35±0.23 | 21.12±0.47 | 8.53±0.38 | 15.55 | 37.46±1.19 | 5.22±0.12 | 8.28±0.23 | 26.00±2.31 | 1.92±0.09 | [73] | ||
| Swine Manure | 77.70±0.47 | 7.13±0.39 | 15.17±0.67 | 12.92±0.08 | 33.52 | 6.17 | 2.8 | 56.69 | 0.82 | [74] | ||
| Cattle Manure | 18 | 49 | 26 | 7 | 10.4 | 33.7 | 4.91 | 3.07 | 58.31 | <0.01 | [70] | |
| Goat Manure | 37.7±0.3 | 52.8±0.4 | 10.0 ± 0 | 43.9±0.3 | 1.5± 0.2 | 2.8± 0.1 | 51.3± 0.2 | 0.6 ± 0 | [75] | |||
| Raw Poultry Manure | 16.69± 0.04 | 36.34± 0.18 | 51.35± 0.38 | 11.99± 0.05 | 25.56± 1.39 | 3.27 ±0.79 | 2.19± 0.17 | 69.35 ±2.43 | 0.69± 0.0.17 | [76] | ||
| Chicken Manure (cage) | 70.9 | 67.5 | 15.6 | 16.9 | 12.744 | 39.67 | 4.72 | 5.49 | 34.12 | 0.4 | [77] | |
| Chicken Manure (Litter) | 18.1 | 69.9 | 12 | 18.1 | 16.55 | 42.86 | 5.57 | 5.50 | 33.39 | 0.68 | [77] | |
| Chicken Manure (Free-range) | 54.9 | 50.4 | 44.4 | 5.2 | 8.58 | 21.85 | 2.50 | 1.73 | 29.24 | 0.28 | [77] | |
| Yak Manure | 7.64 | 51.78 | 27.27 | 13.31 | 13.37 | 33.24 | 4.29 | 1.82 | 30.90 | 0.22 | [78] | |
| Pig Manure | 71.99±9.65 | 66.12±9.08 | 24.18±11.14 | 10.54±3.83 | 37.74±6.43 | 5.62±1.00 | 2.79±0.71 | 28.90±5.68 | 0.63±0.30 | [79] | ||
| Dairy Manure | 75.59±9.22 | 60.60±12.55 | 28.20±16.2 | 11.73±4.54 | 34.42±8.96 | 4.91±1.39 | 1.92±0.50 | 30.44±8.54 | 0.65±0.41 | [79] | ||
| Beef Manure | 75.66±7.82 | 64.58±8.14 | 22.64±11.88 | 13.73±4.05 | 37.64±6.16 | 5.26±1.12 | 2.16±0.64 | 31.90±6.81 | 0.59±0.28 | [79] | ||
| Layer Manure | 72.26±9.95 | 62.56±7.09 | 32.44±9.80 | 6.48±6.51 | 33.02±6.18 | 4.81±1.14 | 3.39±1.25 | 25.74±6.95 | 0.81±0.39 | [79] | ||
| Broiler Manure | 63.88±8.79 | 62.47±11.02 | 27.76±13.56 | 10.45±5.90 | 33.62±8.83 | 5.06±1.94 | 3.70±1.26 | 30.75±7.29 | 0.89±0.55 | [79] | ||
| Types of gasifiers | Biomass | Experimental characteristics | Reference |
|---|---|---|---|
| Downdraft fixed bed | Wood chips | 50 kWth gasifier coupled with a pilot-scale gas cleaning unit (water gas shift and pressure swing adsorption unit). High H2 purity (>99.977% vol.). H2 yield of 55.1 – 58.9 g H2/kg biomass (dry basis). | [206] |
| Pomegranate wood chips, walnut shell | Assessed process performance under different electrical power loads. Carbon conversion efficiency increased with increasing electrical power load. Higher ERs were applied when increasing the electrical power load, resulting in lower syngas energy density. | [207,208] | |
| Updraft high temperature agent (HTAG) | Black pellets (based on 75% soft wood and 25% hard wood pretreated with steam explosion) and gray pellets (woody based roadside scrub cuts, no pre-treatment applied) | Use of preheated air/steam. The preheater oxidizer unit supplies additional heat to the gasifier. Syngas post-combustion unit. Feeding rate: 50 – 70 kg/h, ER: 0.2, steam:biomass ratio: 1.2 Highest temperature at the bed bottom: around 980 – 1150 °C. Temperature at the top: 600 – 900 °C. Syngas LHV (air gasification): 6. – 7.3 MJ/m3, Syngas LHV (steam gasification): 8.2 - 10 MJ/m3. CGE 1: 74 – 77%. Steam gasification produced less tar (11.4 g/m3). |
[209] |
| Bubbling fluidized bed | Pine chips | Air/Air-steam gasification. Steam to biomass ratio of 0.5 and 1.05, ER: 0.15 – 0.25. No information regarding biomass loading conditions. CGE increased to 95% when adding steam as gasifying agent. Syngas yield: 3 m3/kg biomass. Air gasification showed higher profitability than air/steam gasification. | [210] |
| Pine and eucalyptus residues | Gasifier 80 kWth. ER: 0.24 – 0.26. Syngas LHV: 3.9 – 3.6 MJ/m3. CH4 content ranged from 2.9 to 4.8 %vol. Syngas yield: 1.5 – 1.8 m3/kg biomass (dry syngas/dry biomass). The use of high-density biomass and char formation during gasification improved performance when steam was added (char-steam reforming reactions), resulting in higher syngas LHV and CGE. Optimum steam to biomass ratio: 0.5. |
[211] | |
| Dual bubbling fluidized bed (DBFB) | Hazelnut shells | 100 kWth using steam as a single gasifying agent to avoid N2 dilution. Syngas LHV: 10.5 MJ/m3 (H2 content: 34.8%). Syngas yield: 1.33 m3/kg biomass. High tar production: 12 g/m3. This value was reduced to 3 g/m3 after conditioning using a cyclone and water scrubber. Carbon conversion: 73%. |
[212] |
| Fluidized bed gasifier | Soy hull pellets | Feeding rate: 5.5 kg/min. Temperature (750 – 950 °C), ER (0.2 – 0.4). Biomass loading and fluidification velocity were assessed. Lowering ER improved CGE. Higher fluidification velocity improved performance, particularly at higher biomass loading. | [213] |
| Agricultural waste (mainly straw) | Air gasification with O2 enrichment. ER: 0.11 – 0.24, O2 content: 21 – 45%. Syngas LHV: 9.35 MJ/m3, CGE: 51.62% at ER: 0.16. Increasing oxygen content resulted in higher CO and H2 levels in syngas, thanks to the increase in gasification temperature, positively affecting performance. | [214] | |
| Dual fluidized bed gasifier (combining bubbling fluidized bed (BFB) and fast fluidized combustion reactor) | Wood pellets | 100 kW fuel input (20 kg/h). Temperature: 770 - 850 °C in gasifier and 920 °C in combustion reactor. Gasifying agent in BFB: steam (steam to fuel ratio: 0.74 – 1.10), gasifying agent in combustion reactor: air. Syngas yield: 0.99 – 1.13 m3/kg biomass. Tar decomposition takes place at temperatures above 800 °C. |
[215] |
| circulating fluidized bed | Wood pellets | 500 kW pilot gasifier: comparison of model and pilot scale results previously published. H2:CO ratio of 2 was obtained by coupling an electrolyzer to produce H2 and using the O2 released to achieve a desired oxidant proportion. | [216] |
| Rotary kiln plant | Digestate and almond shells | Feeding rate: 20 kg/h. ER: 0.22 – 0.39 Syngas LHV: 4 – 5 MJ/m3, maximum CGE of 55% at ER: 0.3. Steam was added to increase the H2/CO ratio in syngas, but the CGE dropped to 17%. A heat recovery unit can increase CGE to 72% for the mixture, but only to 35% when using steam as the gasification agent. |
[217] |
| Coal-water slurry gasification technology with opposed multi-burners (OMB) |
Torrefied biomass and coal (water containing slurry) | Pilot plant containing: Slurry unit, gasification, water scrubber, WGS 2 reactor (producing syngas with H2/CO ratio of 11:1), acid gas removal unit (CO2 and H2S removal), Fischer-Tropsch synthesis reactor. The coal-torrefied biomass blend required a much higher water content to produce a pumpable slurry, thus reducing energy efficiency. Feeding: 0.37 t/day of coal and 0.02 t/day torrefied biomass. 0.12 t/day of natural gas was required to keep the process running. 0.36 t/day of water was added to generate the slurry. Gasification temperature: 1400 °C H2 content was 22%, and CO content was 20% in syngas |
[218] |
| Type of gasifier | Company | Plant location | Raw materials | Characteristics | Reference |
|---|---|---|---|---|---|
| Fixed bed downdraft | Aries Clean Energy (Tennessee, USA) | Lebanon, Tennessee, US | Biosolids, commercial wood waste, and scrap tires | 420 kWe, Feedstock throughput capacity: 64 t/d | [221] |
| Fixed bed downdraft | URBAS Energy Technology (Völkermarkt, Austria) | 15 installed plants in Austria, Italy, Japan, Croatia and Bosnia and Herzegovina | Wood biomass | 150 – 450 kWe | [222] |
| Fixed bed updraft | Harboøre updraft biomass gasification plant constructed by Babcock & Wilcox Vølund (now part of Babcock & Wilcox) | Harboøre, Denmark | Wood chips | No longer in operation. Demonstration plant. 3.5 – 4 MWth | [223] |
| Fixed bed | Neoelectra (Sant Just Desvern, Barcelona) using Careco Technology: “gasógeno” no specification of flow direction conditions 1 | Villacañas, Toledo, Spain | Industrial wood residue and forest residue | 380 000 t/year, 8 Mwe. | [224] |
| Santa Perpétua de Mogoda (Barcelona) | Woodchips, Plastics, rubber, Residual Biomass and combustible solid residue | 20 MWth | [224] | ||
| Fluidized bed | Aries Clean En-ergy (Tennessee, USA) | Linden, New Jersey, USA | Biosolids | Feedstock throughput capacity: 430 t/d. Commercialize ashes as Bio-Fly-Ash™ to be used as a concrete additive. | [221] |
| Circulating fluidized bed gasification | Valmet (Spoo, Finland) | 4 gasifiers in Finland, and other 3 located in Indonesia, China and Brazil | Mainly bark and wood residues and waste (solid residue fuel) | Thermal capacity 50 – 150 MW | [225] |
| Air fluidized bed | Sumitomo SHI/FW (Spoo, Finland) | Varkaus, Finland | Biomass, forestry residues | 12 MWth | [226] |
| Company | Gasification technology | Plants | Characteristics | Reference |
|---|---|---|---|---|
| GEMCO Energy, (Anyang, Henan, China) | Fixed bed (up-draft and down-draft) and circulating fluidized bed (CFB) | No installed gasification plants reported | The company offers modular biomass power stations (small scale), with an installed capacity ranging between 200 and 500 kW. The CFB is for large-scale and can operate on wood powder, sawdust, rice husks, bagasse, and crushed straw with a biomass flow range of 1500 – 5000 kg/h | [227] |
| SynTech Bioenergy (Englewood, Colorado, USA) | BioMax® system: Modular units. Using downdraft gasifiers operating at above 800 °C | Installed plants in California, Texas and United Kingdom (West Midlands) | Feedstock: agricultural waste. Offers the production of certified biochar and low level of tars in syngas using a dry filter for gas clean-up without releasing wastewater. 165 kW – 2 MW | [228] |
| Ankur Scientific Energy Technologies (Sama Vadodara, Gujarat, India) | downdraft gasifiers generating combustible gases for thermal applications (boilers, furnaces or CHP grid/off grid) | Installed plants in more than 35 countries | Feedstock: MSW, poultry litter, empty fruit bunch (EFB), fecal/sewage sludge, agricultural residues, paper. A wide range of plants with a treatment capacity of 5 – 100 t/day in the case of MSW and 16 – 2000 kg/h for biomass material (EFB and agricultural residues) | [229] |
| CMN Industry Inc. (Sacramento, California, USA) | No description given of gasification technology used. Process temperature 700 – 1200 °C | Installed plants located in USA, Germany, India and Brazil | Feedstock: Crop straw and forestry waste. No description of treatment capacity | [230] |
| Powermax Renewable Energy (Wuxi, Jiangsu, China) | Fixed bed (up-draft and downdraft), CFB and twin fire-fixed bed gasifier. Modular plants (50 kW – 2 MW) and large-scale units | Myanmar gasification plant. Collaboration in gasification plant project in Canada | Feedstock: rice husk Power: 12 MW |
[231] |
| Compact Syngas Solutions (Sandycroft, Flintshire, UK) | MicroHub 500 and MicroHub 1000: modular waste management technology | No installation plants reported. Participation in project for tea biomass gasification in Kericho district (Kenya) | Feedstock: Biomass and solid recovered fuel. Treatment capacity of 3570 t/year. Power: 500 and 1000 kWe | [232] |
| SPANNER RE² GMBH (Neufahrn, Niederbayern, Germany) | Gasification plants for heat and electricity production | Installed plants in Slovenia, Germany, Austria, Italy (South Tyrol region) | Feedstock: wood biomass 35 – 700 kWe | [233] |
| Meva Energy (Hisings Backa, Sweden) | Large- and small-scale gasification plants. Entrained gasification technology. | Demonstration plant. Agreement with Elcowire Group AB group for providing 9 MW of syngas. | Feedstock: Organic residues from agriculture, industry, and forestry | [234,235] |
| Syncraft® (Schwaz, Austria) | floating fixed-bed gasification technology. Small gasification plants with CHP engines | Installed plants in Austria (Tyrol region, Vorarlberg), Croatia and Switzerland | Feedstock: wood biomass 270, 400 and 550kWe | [236] |
| The Stadtwerke Rosenheim (Rosenheim Municipal Utilities) (Rosenheim, Germany) | Double stage fluidized bed reactor | Demonstration plant | Feedstock: wood gasification, 60 and 250 kWe | [237] |
| Kombi Power System (Regawatt GbmH) (Abensberg, Germany) | Updraft gasifier. The system can be implemented for heat (boiler) or electricity production (CHP). It can also be used to produce syngas or bio-oil | Installed plants in Netherlands, Japan, Switzerland and Germany | Feedstock: waste wood, forest chips, landscape wood, screenings from composting plants. 250 – 2000 kWe | [238] |
| Lipro Energy (Wardenburg, Germany) | Multi-stage gasification | Several installed plants in Germany | Feedstock: wood biomass and waste biomass. 50 – 85 kWe | [239] |
| Holzenergie Wegscheid GmbH (Sonnen, Germany). Merger with WegscheidEntrenco GmbH since 2023 | Down-draft gasifier with CHP engines | Several installed plants in Germany | Feedstock: wood biomass. 70 kW – 2 MWe | [240] |
| Burkhardt GmbH (Mühlhausen, Germany) | Multi-stage gasification system: Updraught co-current flow gasification forming a stationary fluidized bed | Several plants in Austria, Germany and Japan | Feedstock: wood biomass (wood pellets and wood chips). 190 – 390 kWe | [241] |
| CMD (Costruzioni Motori Diesel SPA) (Caserta, Italia) | ECO20x gasifier: Pyrogasification process | Demonstration plant in Salerno (Campania), Italy | Feedstock: Wood biomass (wood chips). 20 kWe (40kWth) | [242] |
| ESPE Energy expertise (Padua, Italy) | CHiP50 gasifier: Pyrogasification process | 3 plants installed (Italy, Japan) | Feedstock: Wood biomass (wood chips) | [243] |
| RESET (Rieti, Italy) | SyngaSmart gasifier. configurations available: Electricity production using CHP, heat production using thermal power units and syngas production (biomass to biofuel generator) | 1 medium size (250 kg/h input) gas producing plant in Rome (Italy) for the treatment of briquetted organic fraction of MSW) | Feedstock: Agroforestry residues, organic byproducts, and waste. 19 – 200 kWe (28 – 292 kWth). Biomass consumption rate: 22.8 – 240 kg/h | [244] |
| VOLTER (Tupos, Finland) | Walter CHP Modular gasification: Gasifier + CHP (gas cleaning and energy recovery) | No reports available of installed gasification plants | Feedstock: Wood chips. 50 kWe (130 kWth) | [245] |
| Xylergy SA (Lovain-La-Nueve, Belgium) | NOTAR®: downdraft gasifier with CHP unit | 1 demonstration plant in Belgium and 2 Plants installed in Belgium and France | Feedstock: Wood chips. 200 - 750 kWe | [246] |
| Raw material | Gasification characteristics | Main results | Reference |
|---|---|---|---|
| Sewage sludge. Ash content: 24.3 – 30.6% | Fluidized bed reactor. Temperature: 850 °C. Bench scale. ER: 0.1 – 0.2. Feed rate: 68 and 138 g/h | Syngas LHV: 5.8 MJ/m3 (ER: 0.2) and 12.1 MJ/m3 (ER: 0.1). CGE: 50 – 57%. The study focused on tar and bottom ash quality. Operating conditions had a greater influence on the types of tar components obtained than the characteristics of the raw sludge had. Lower tar production was obtained at an ER of 0.2, but operating at a lower ER resulted in ash with a higher surface area. | [260] |
| Sewage sludge. Ash content: 24.3 – 30.6% | Entrained flow gasifier (90 kW). Evaluated the effect of gasifying agents (air and O2) | Syngas LHV: 8.4 MJ/m3 (using O2). Syngas volume was reduced by 60%. CGE: 45 – 51%. The presence of KCl increased performance by favoring carbon conversion. | [261] |
| Sewage sludge. Ash content: 25.6% | Fluidized bed gasifier. ER: 0.12 – 0.27 | Syngas LHV: 3.1 – 4.8 MJ/m3. Reactor profile temperature range: 690 – 1000 °C |
[262] |
| Sewage sludge. Ash content: 49 – 51% | Bubbling fluidized bed gasifier. Input rate: 2 kg/h. ER: 0.2. Steam to carbon ratio: 1.0 – 1.8. CaO used for CO2 absorption during gasification | The combination of steam injection and CaO (used as bed material) enabled tripling syngas H2 content. Air addition improved carbon conversion but reduced H2 yield. | [263] |
| Sewage sludge. Ash content: 47.6% | Bubbling fluidized bed gasifier (20 kW fuel input). Steam-O2 gasification. Temperature: 650 – 900 °C. ER: 0.2 – 0.28. Steam to carbon ratio: 0.6 – 2.0 | The addition of limestone helps reduce H2S and COS species. Fluidization needs to be carefully controlled to avoid undesired hot spots that could lead to particle agglomeration. | [264] |
| Sewage sludge and torrefied sludge. Ash content: 39.3 – 43 | Pilot-scale entrained flow gasifier (90 kw fuel input). Air and O2 gasification. Temperature: 1000 – 1050 °C | Adding KCl as a catalyst increased carbon conversion from 90% to 95%. The CGE ranged from 45 to 51% across all operating conditions tested. Syngas LHV > 8 MJ/m3 (O2-based gasification). The use of air as a gasifying agent produced syngas with LHV < 4 MJ/m3. | [265] |
| Digestate and almond shells. Digestate ash content: 32%, almond shell ash content: 1.65% | Rotary kiln pilot scale gasifier. Gasification of digestate pellets as single raw material and mixture with almond shells. Digestate provided 60% of the mix. Feeding rate: 14 – 27.5 kg/h. ER: 0.26 – 0.39 |
The CGE of digestate gasification was 47% and increased to 55% when almond shells were added to the mixture. Steam injection enabled the production of syngas with a higher H2/CO ratio, but CGE dropped to 17%. A heat recovery unit can increase CGE to 72% for the mixture, but only to 35% when using steam as a gasification agent. | [217] |
| Digestate from a plant treating manure and straw (fresh weight ratio of 1:0.3). Ash content: 23.03% | Downdraft fixed bed gasifier. Temperature: 600 – 800 °C. ER: 0.25 – 0.30. | Syngas LHV: 4.78 MJ/m3, CGE: 67% at ER: 0.28. CGE efficiency increased from 30 to 67% with increasing temperature from 600 to 800 °C | [266] |
| Digestate and lignite (mass ratio 1:1). Digestate ash content: 41.5% | Downdraft fixed bed gasifier. Laboratory scale with a test capacity of 20 g of feedstock. Temperature: 650 – 950 °C. CO2 atmosphere. | Syngas LHV: 4.0 – 6.5 MJ/m3. Increasing temperature led to higher LHV. Increasing temperature reduced tar content in syngas. | [267] |
| Digestate and sewage sludge. Digestate ash content: 13.5%, sewage sludge ash content: 47.1% | Pilot-scale fluidized bed gasifier (thermal output of 100 kWth). Temperature: 750 °C. Steam addition. | Syngas LHV (from digestate): 4.06 MJ/m3. Syngas LHV (from sewage sludge): 4.11 MJ/m3. Adding steam helps reduce tar concentration in syngas. | [268] |
| Diary manure. Ash content: 48.8% | Bench and pilot scale reactor. Application of RSM 1 for process optimization. Factors: Temperature, ER and O2 concentration |
Syngas LHV: 8 MJ/m3, maximum value obtained at 800 °C, ER: 0.25 and O2 content: 40%. CGE increased with oxygen enrichment. |
[269] |
| Chicken manure (CM) – oily sludge (50% mixture) CM Ash content: 27.7% |
Fixed-bed (up-draft) gasifier. T: 600 – 800 °C. Batch process (laboratory scale) |
Syngas LHV: 2487 - 2838 kcal/m3 (10.4 – 11.9 MJ/m3). Methane content was increased by using oily sludge as a raw material, thereby increasing syngas energy density. | [270] |
| Chicken manure (CM) – woody biomass (30% CM in mixture) CM Ash content: 25.3% |
Fixed-bed (downdraft 10 kW) gasifier. Pilot plant. Temperature: 850 – 900 °C Feedstock rate: 10 kg/h. | Syngas LHV: 5.23 MJ/m3. CGE improvement (6.7%). The mixture containing 30 wt% chicken manure produced syngas with quality comparable to wood gasification. The char obtained was tested as low-cost activated carbon. | [271] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




