Submitted:
20 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
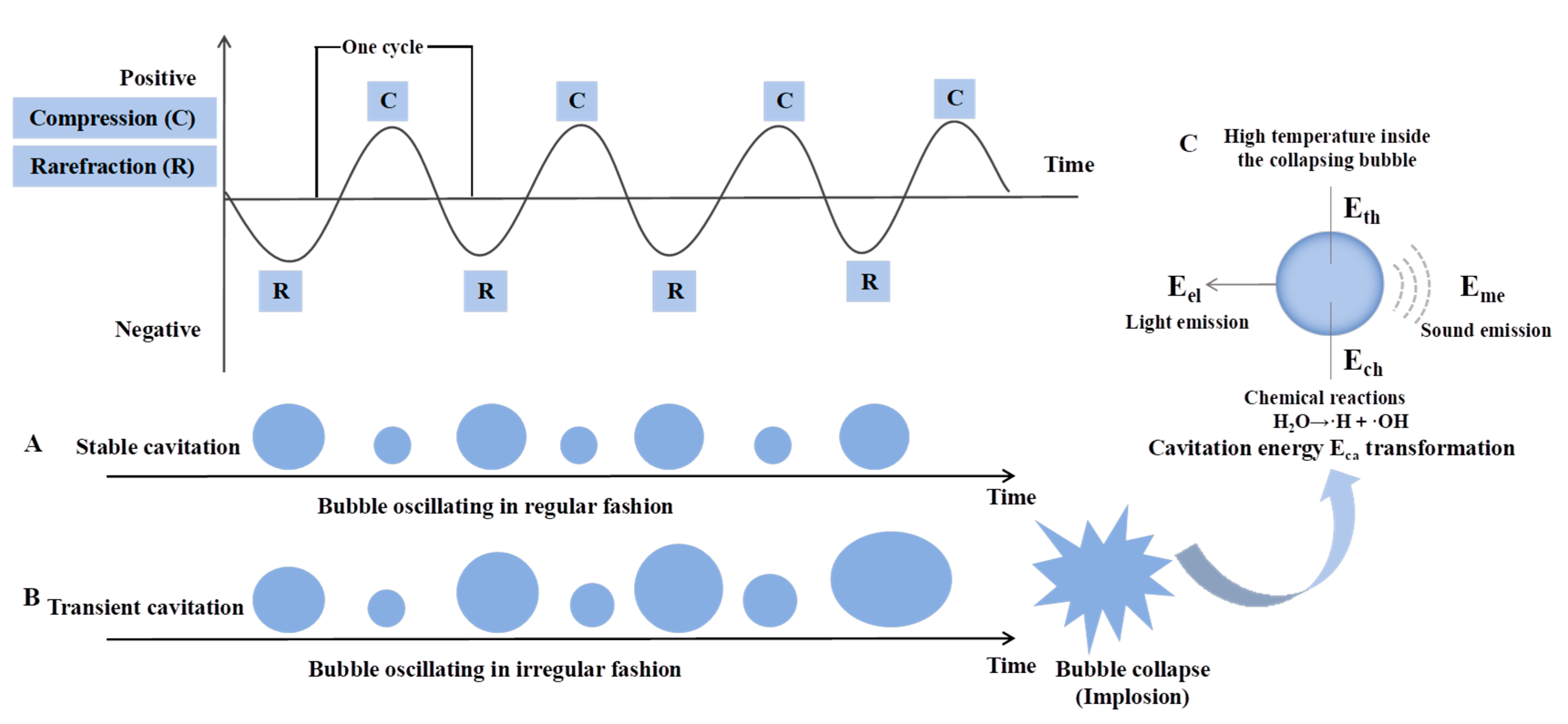
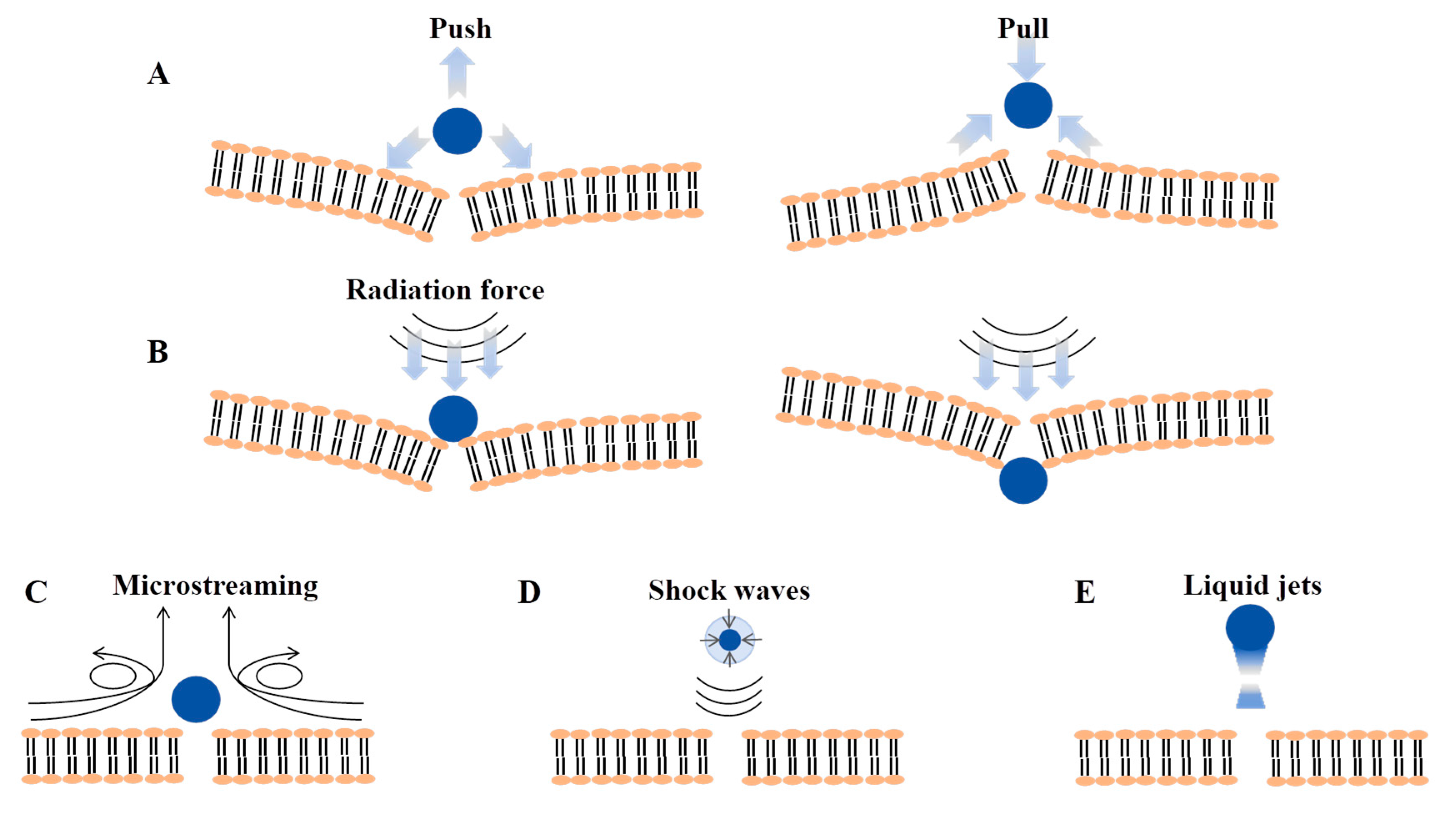
2. Ultrasound Overview
3. Application of Ultrasound in Meat Processing
3.1. Ultrasound-Assisted Meat Tenderization
3.1.1. Mechanism of Ultrasound-Assisted Meat Tenderization
3.1.2. Effects of Ultrasound-Assisted Tenderization on Meat Quality
3.2. Ultrasound-Assisted Meat Cooking
3.2.1. Mechanism of Ultrasound-Assisted Meat Cooking
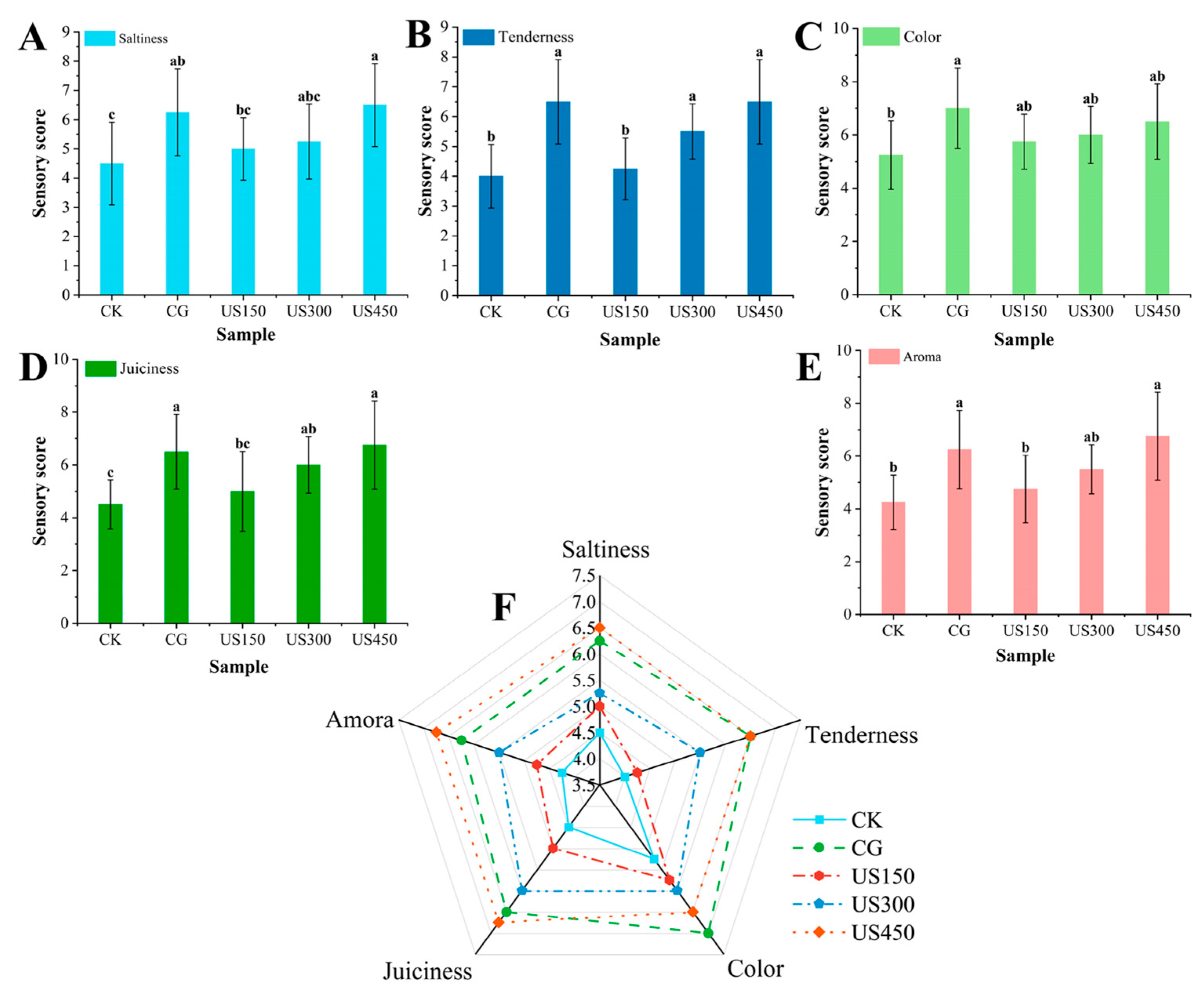
3.2.2. Effect of Ultrasound-Assisted Meat Cooking on the Quality of Meat Products
3.3. Ultrasound-Assisted Meat Brining
3.3.1. Mechanism of Ultrasound-Assisted Meat Brining
3.3.2. Effect of Ultrasound-Assisted Brining on the Quality of Meat Products
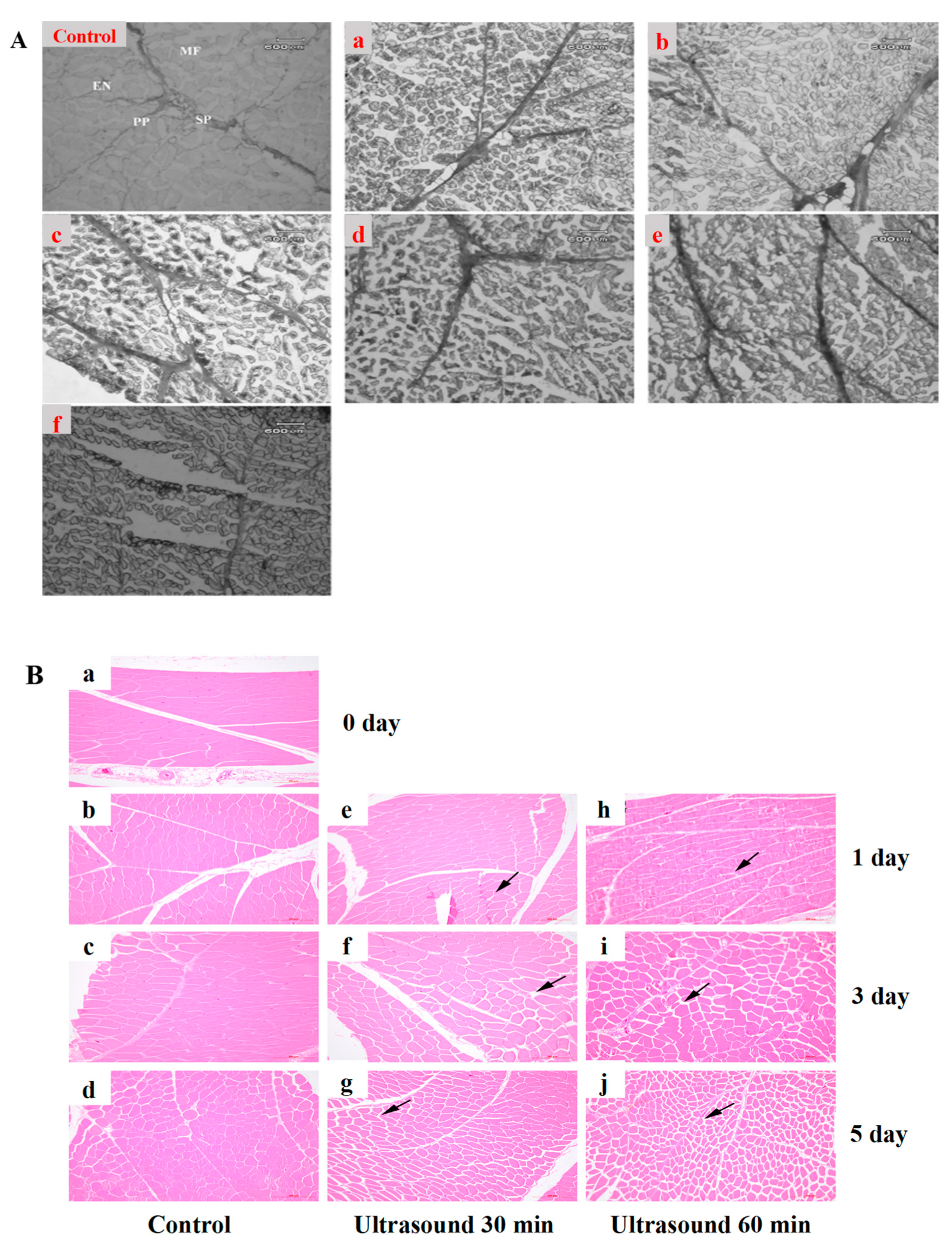
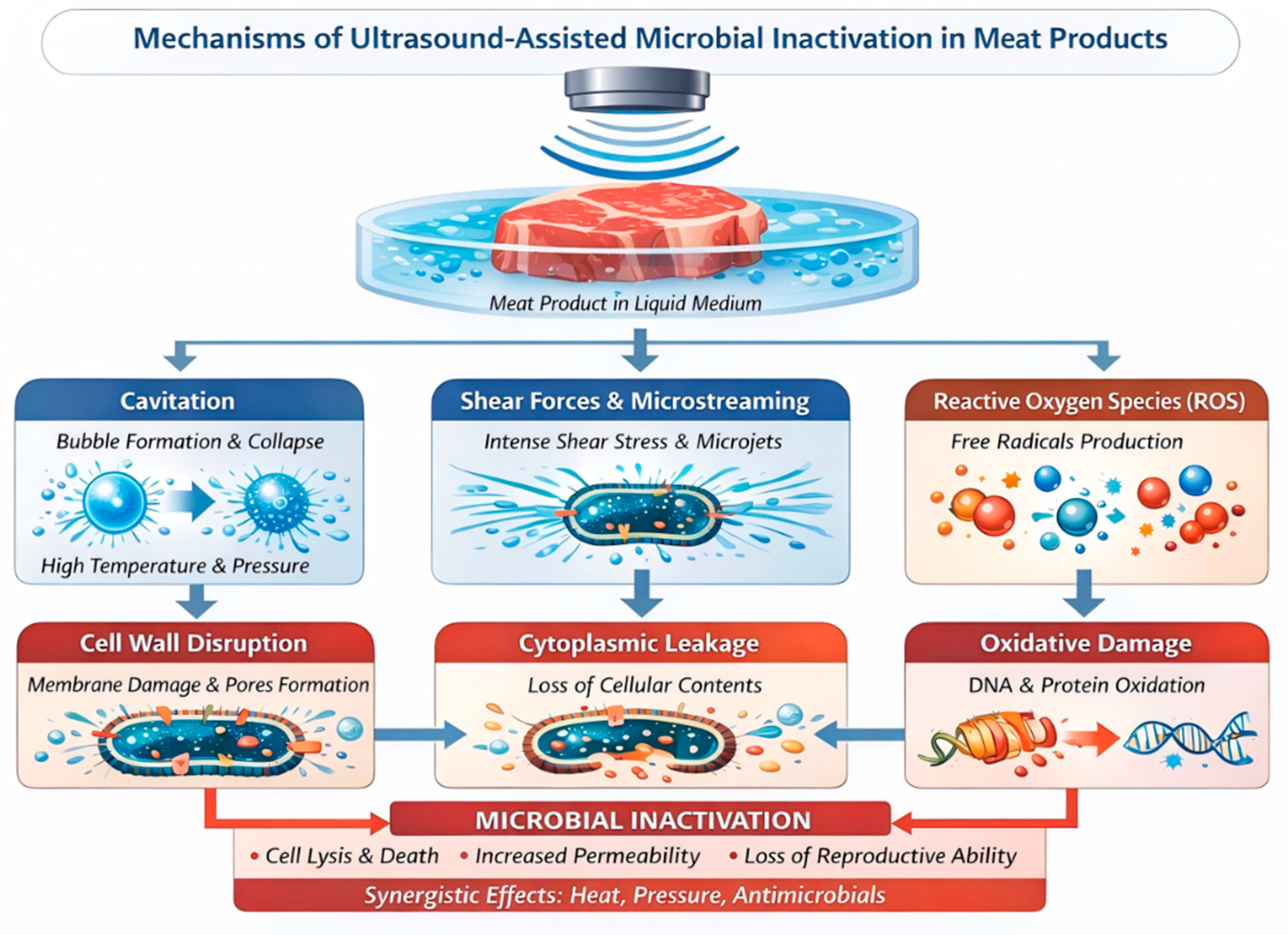
3.4. Ultrasound-Assisted Meat Sterilization
3.4.1. Mechanism of Ultrasound-Assisted Meat Sterilization
3.4.2. Effect of Ultrasound-Assisted Sterilization on the Quality of Meat Products
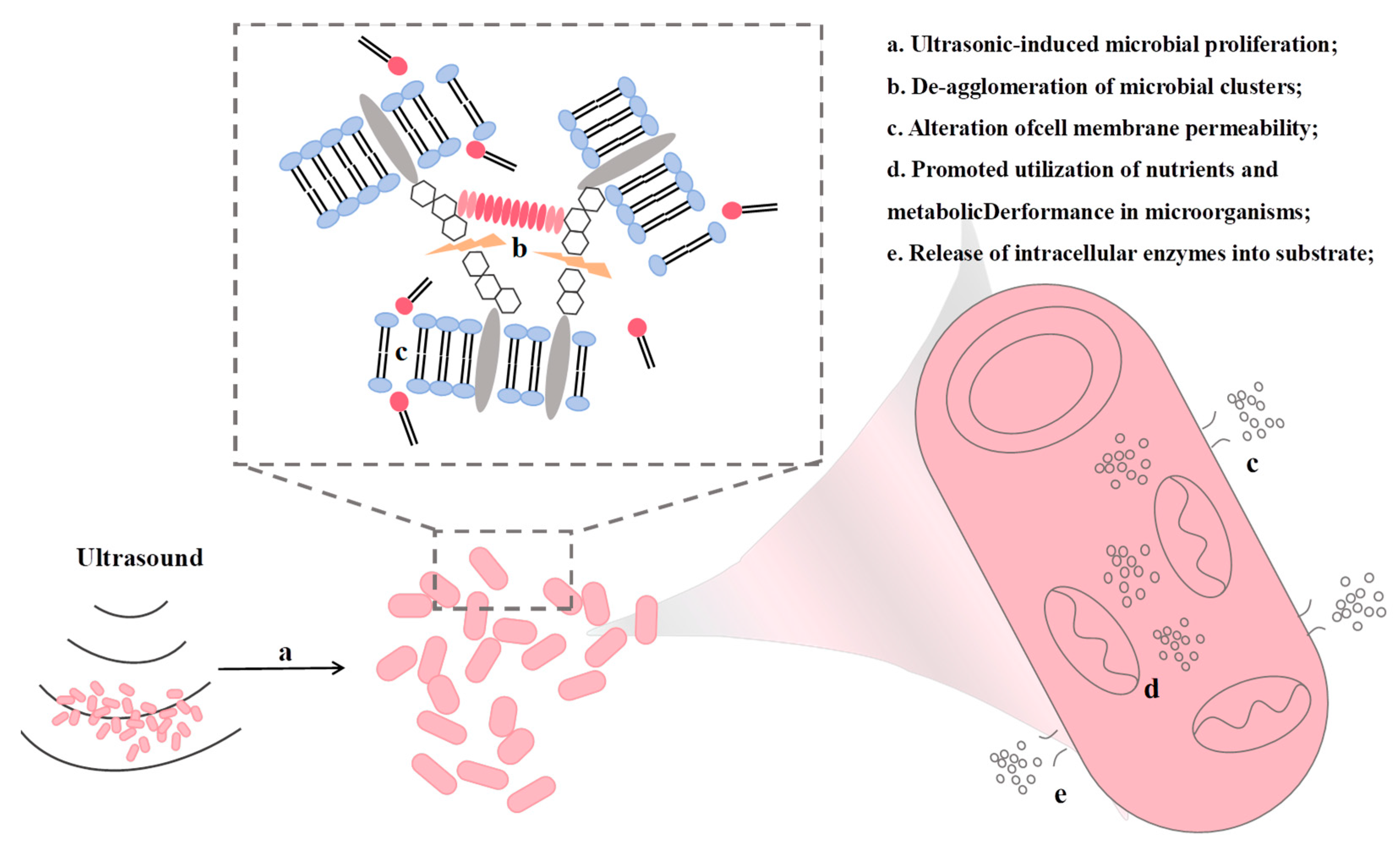
3.5. Ultrasound-Assisted Meat Fermentation
3.5.1. Mechanism of Ultrasound-Assisted Meat Fermentation
3.5.2. Effect of Ultrasound-Assisted Fermentation on the Quality of Meat Products
3.6. Ultrasound-Assisted Meat Freezing
3.6.1. Mechanism of Ultrasound-Assisted Meat Freezing
3.6.2. Effect of Ultrasound-Assisted Freezing on the Quality of Meat Products
3.7. Ultrasound-Assisted Meat Thawing
3.7.1. Mechanism of Ultrasound-Assisted Meat Thawing
3.7.2. Effect of Ultrasound-Assisted Thawing on the Quality of Meat Products
3.8. Ultrasonic Application in Other Meat Processing Procedures
3.8.1. Drying
3.8.2. Extraction
4. Disadvantages of Ultrasound-Assisted Meat Processing
5. Conclusions and Future Trends
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gastaldello, A.; Giampieri, F.; De Giuseppe, R.; Grosso, G.; Baroni, L.; Battino, M. The Rise of Processed Meat Alternatives: A Narrative Review of the Manufacturing, Composition, Nutritional Profile and Health Effects of Newer Sources of Protein, and Their Place in Healthier Diets. Trends in Food Science & Technology 2022, 127, 263–271. [Google Scholar] [CrossRef]
- Zhihua, L.; Xucheng, Z.; Kun, W.; Xiaobo, Z.; Jiyong, S.; Xiaowei, H.; Holmes, M. A Novel Sensor for Determination of Dopamine in Meat Based on ZnO-Decorated Reduced Graphene Oxide Composites. Innovative Food Science & Emerging Technologies 2015, 31, 196–203. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, C.; Cui, H.; Lin, L. Plasma Enhanced-Nutmeg Essential Oil Solid Liposome Treatment on the Gelling and Storage Properties of Pork Meat Batters. Journal of Food Engineering 2020, 266, 109696. [Google Scholar] [CrossRef]
- Huang, J.; Guo, Q.; Manzoor, M.F.; Chen, Z.; Xu, B. Evaluating the Sterilization Effect of Wheat Flour Treated with Continuous High-Speed-Stirring Superheated Steam. Journal of Cereal Science 2021, 99, 103199. [Google Scholar] [CrossRef]
- Cai, M.; Zhang, G.; Wang, J.; Li, C.; Cui, H.; Lin, L. Application of Glycyrrhiza Polysaccharide Nanofibers Loaded with Tea Tree Essential Oil/Gliadin Nanoparticles in Meat Preservation. Food Bioscience 2021, 43, 101270. [Google Scholar] [CrossRef]
- Nasiru, M.M.; Frimpong, E.B.; Muhammad, U.; Qian, J.; Mustapha, A.T.; Yan, W.; Zhuang, H.; Zhang, J. Dielectric Barrier Discharge Cold Atmospheric Plasma: Influence of Processing Parameters on Microbial Inactivation in Meat and Meat Products. Comp Rev Food Sci Food Safe 2021, 20, 2626–2659. [Google Scholar] [CrossRef]
- Li, J.; Shi, J.; Huang, X.; Zou, X.; Li, Z.; Zhang, D.; Zhang, W.; Xu, Y. Effects of Pulsed Electric Field on Freeze-Thaw Quality of Atlantic Salmon. Innovative Food Science & Emerging Technologies 2020, 65, 102454. [Google Scholar] [CrossRef]
- Cheng, Y.; Ofori Donkor, P.; Yeboah, G.B.; Ayim, I.; Wu, J.; Ma, H. Modulating the in Vitro Digestion of Heat-Set Whey Protein Emulsion Gels via Gelling Properties Modification with Sequential Ultrasound Pretreatment. LWT 2021, 149, 111856. [Google Scholar] [CrossRef]
- Zhang, R.; Cheng, Z.; Liang, Y.; Hu, X.; Shen, T.; Li, Y.; Han, Z.; Zhang, X.; Zou, X. A Novel Strategy for Accelerating Pumpable Ice Slurry Production with Ozone Micro–Nano Bubbles and Extending the Shelf Life of Larimichthys Polyactis. Foods 2023, 12, 2206. [Google Scholar] [CrossRef]
- Huang, G.; Sun, W.; Dai, C.; Sun, L.; Tang, Y.; He, R.; Ma, H. Sterilization of Bacillus Tequilensis Isolated from Aerogenic Vinegar by Intense Pulsed Light. LWT 2020, 118, 108811. [Google Scholar] [CrossRef]
- Xu, F.; Wang, B.; Hong, C.; Telebielaigen, S.; Nsor-Atindana, J.; Duan, Y.; Zhong, F. Optimization of Spiral Continuous Flow-through Pulse Light Sterilization for Escherichia Coli in Red Grape Juice by Response Surface Methodology. Food Control 2019, 105, 8–12. [Google Scholar] [CrossRef]
- Zhang, J.; Toldrá, F.; Kang, D.; Zhou, L.; Wang, J.; Zhang, W.; Hu, Y. Benefits of Ultrasonic Technology Application in Meat Field and Its Influential Mechanism: A Review. Critical Reviews in Food Science and Nutrition 2025, 65, 5726–5751. [Google Scholar] [CrossRef]
- Umego, E.C.; He, R.; Huang, G.; Dai, C.; Ma, H. Ultrasound-assisted Fermentation: Mechanisms, Technologies, and Challenges. J. Food Process. Preserv. 2021, 45. [Google Scholar] [CrossRef]
- Wali, A.; Ma, H.; Aadil, R.M.; Zhou, C.; Rashid, M.T.; Liu, X. Effects of Multifrequency Ultrasound Pretreatment on the Enzymolysis, ACE Inhibitory Activity, and the Structure Characterization of Rapeseed Protein. J Food Process Preserv 2017, 41, e13413. [Google Scholar] [CrossRef]
- Xu, B.; Chen, J.; Azam, S.M.R.; Feng, M.; Wei, B.; Yan, W.; Zhou, C.; Ma, H.; Bhandari, B.; Ren, G.; et al. Flat Dual-Frequency Sweeping Ultrasound Enhances the Inactivation of Polyphenol Oxidase in Strawberry Juice. Food Measure 2022, 16, 762–771. [Google Scholar] [CrossRef]
- Sun, J.; Sun, L.; Chen, X.; Raza, H.; Wu, G.; Liang, Q.; Ren, X. Characterization of Arrowhead-Derived Type 3 Resistant Starch Prepared by Ultrasound-Assisted α-Amylase Degradation. Journal of Food Quality 2023, 2023, 1–11. [Google Scholar] [CrossRef]
- Sun, Y.; Ji, D.; Ma, H.; Chen, X. Ultrasound Accelerated γ-Aminobutyric Acid Accumulation in Coffee Leaves through Influencing the Microstructure, Enzyme Activity, and Metabolites. Food Chemistry 2022, 385, 132646. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, F.; Liu, T.; Jing, H.; Huang, Y.; Obadi, M.; Xu, B. Ultrasound-Enhanced Egg White Proteins Conjugated with Polyphenols: The Structure of the Polyphenols on Their Functional Properties. LWT 2022, 164, 113600. [Google Scholar] [CrossRef]
- Hua, Y.; Zhang, H.; Fu, Q.; Feng, Y.; Duan, Y.; Ma, H. Effects of Ultrasound Modification with Different Frequency Modes on the Structure, Chain Conformation, and Immune Activity of Polysaccharides from Lentinus Edodes. Foods 2022, 11, 2470. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, W.; Ding, X.; Wu, Z.; Li, Y. Effects of Dual-Frequency Ultrasound with Different Energy Irradiation Modes on the Structural and Emulsifying Properties of Soy Protein Isolate. Food and Bioproducts Processing 2020, 123, 419–426. [Google Scholar] [CrossRef]
- Liang, Q.; Chen, X.; Ren, X.; Yang, X.; Raza, H.; Ma, H. Effects of Ultrasound-Assisted Enzymolysis on the Physicochemical Properties and Structure of Arrowhead-Derived Resistant Starch. LWT 2021, 147, 111616. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, X.; Yagoub, A.E.A.; Owusu-Ansah, P.; Wahia, H.; Ma, H.; Zhou, C. Effects of Low Frequency Multi-Mode Ultrasound and It’s Washing Solution’s Interface Properties on Freshly Cut Cauliflower. Food Chemistry 2022, 366, 130683. [Google Scholar] [CrossRef] [PubMed]
- Amanor-Atiemoh, R.; Zhou, C.; Mujumdar, A.; Osae, R.; Taiye Mustapha, A.; Wahia, H.; Sampson, G.; Amoa-Owusu, A.; Ma, H. Effect of Simultaneous Dual-frequency Ultrasound Aided Ethanolic Pretreatment on Drying Kinetics, Bioactive Compounds, Antioxidant Activity, and Physicochemical Properties of Apple Slices Using Pulsed Vacuum Dryer. J Food Process Engineering 2020, 43, e13535. [Google Scholar] [CrossRef]
- Liang, Q.; Ren, X.; Zhang, X.; Hou, T.; Chalamaiah, M.; Ma, H.; Xu, B. Effect of Ultrasound on the Preparation of Resveratrol-Loaded Zein Particles. Journal of Food Engineering 2018, 221, 88–94. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L.; Chen, L.; Wang, Y.; Okonkwo, C.E.; Yagoub, A.E.A.; Wahia, H.; Zhou, C. Application of Ultrasound and Its Real-time Monitoring of the Acoustic Field during Processing of Tofu: Parameter Optimization, Protein Modification, and Potential Mechanism. Comp Rev Food Sci Food Safe 2023, 22, 2747–2772. [Google Scholar] [CrossRef]
- Zhang, Z.; Shan, P.; Zhang, Z.-H.; He, R.; Xing, L.; Liu, J.; He, D.; Ma, H.; Wang, Z.; Gao, X. Efficient Degradation of Soybean Protein B3 Subunit in Soy Sauce by Ultrasound-Assisted Prolyl Endopeptidase and Its Primary Mechanism. Food Chemistry 2023, 429, 136972. [Google Scholar] [CrossRef]
- Azam, S.M.R.; Ma, H.; Xu, B.; Devi, S.; Siddique, M.A.B.; Stanley, S.L.; Bhandari, B.; Zhu, J. Efficacy of Ultrasound Treatment in the Removal of Pesticide Residues from Fresh Vegetables: A Review. Trends in Food Science & Technology 2020, 97, 417–432. [Google Scholar] [CrossRef]
- Dong, Y.; Zhang, H.; Mei, J.; Xie, J.; Shao, C. Advances in Application of Ultrasound in Meat Tenderization: A Review. Front. Sustain. Food Syst. 2022, 6, 969503. [Google Scholar] [CrossRef]
- Zhou, C.; Okonkwo, C.E.; Inyinbor, A.A.; Yagoub, A.E.A.; Olaniran, A.F. Ultrasound, Infrared and Its Assisted Technology, a Promising Tool in Physical Food Processing: A Review of Recent Developments. Critical Reviews in Food Science and Nutrition 2023, 63, 1587–1611. [Google Scholar] [CrossRef]
- Raza, H.; Ameer, K.; Ren, X.; Liang, Q.; Chen, X.; Chen, H.; Ma, H. Physicochemical Properties and Digestion Mechanism of Starch-Linoleic Acid Complex Induced by Multi-Frequency Power Ultrasound. Food Chemistry 2021, 364, 130392. [Google Scholar] [CrossRef]
- Al-Hilphy, A.R.; Al-Temimi, A.B.; Al Rubaiy, H.H.M.; Anand, U.; Delgado-Pando, G.; Lakhssassi, N. Ultrasound Applications in Poultry Meat Processing: A Systematic Review. Journal of Food Science 2020, 85, 1386–1396. [Google Scholar] [CrossRef]
- Luo, F.; Zhang, Z.; Lu, F.; Li, D.; Zhou, C.; Li, Y.; Niu, L.; Xu, Y.; Feng, L.; Dai, Z.; et al. Ultrasound Modification of Pectin and the Mechanism of Its Interaction with Cyanidin-3-O-Glucoside. Food Hydrocolloids 2024, 152, 109898. [Google Scholar] [CrossRef]
- Wang, Q.; Mei, S.; Manivel, P.; Ma, H.; Chen, X. Zinc Oxide Nanoparticles Synthesized Using Coffee Leaf Extract Assisted with Ultrasound as Nanocarriers for Mangiferin. Current Research in Food Science 2022, 5, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Zhong, H.; Hussain, K.; Manzoor, M.F.; Qayum, A.; Liu, X.; Xu, J.; Hussain, A.; Ahsan, H.M.; Guan, R. Emerging High Intensity Ultrasound for Soymilk in Boosting Bioactivity, Amino Acids Release, Reducing Anti-Nutritional Factors and Allergenicity. Food Bioscience 2024, 59, 104138. [Google Scholar] [CrossRef]
- Abdualrahman, M.A.Y.; Zhou, C.; Zhang, Y.; ElGasim Ahmed Yagoub, A.; Ma, H.; Mao, L.; Wang, K. Effects of Ultrasound Pretreatment on Enzymolysis of Sodium Caseinate Protein: Kinetic Study, Angiotensin-Converting Enzyme Inhibitory Activity, and the Structural Characteristics of the Hydrolysates. J Food Process Preserv 2017, 41, e13276. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, J.; Li, S.; Ma, H.; Zhang, H. Mechanism Study of Multimode Ultrasound Pretreatment on the Enzymolysis of Wheat Gluten. J Sci Food Agric 2018, 98, 1530–1538. [Google Scholar] [CrossRef]
- Li, C.; Niu, Z.; Zuo, M.; Wang, T.; Zou, X.; Sun, Z. Detection and Identification of Foreign Bodies in Conditioned Steak Based on Ultrasound Imaging. FSTR 2024, 30, 269–280. [Google Scholar] [CrossRef]
- Leong, T.; Ashokkumar, M.; Kentish, S. THE FUNDAMENTALS OF POWER ULTRASOUND – A REVIEW.
- Wang, Y.; Zhu, X.; Gong, Y.; Liu, N.; Li, Z.; Long, Z.; Teng, J. Combination of Transverse and Longitudinal Ultrasonic Waves for Plane Stress Measurement of Steel Plates. Applied Acoustics 2022, 188, 108500. [Google Scholar] [CrossRef]
- Wieja, K.; Kiełczyński, P.; Szymański, P.; Szalewski, M.; Balcerzak, A.; Ptasznik, S. Identification and Investigation of Mechanically Separated Meat (MSM) with an Innovative Ultrasonic Method. Food Chemistry 2021, 348, 128907. [Google Scholar] [CrossRef]
- Li, K.; Ma, H.; Li, S.; Wang, C. Numerical Simulation of Three Physical Fields in Counter-current Ultrasound. J Food Process Engineering 2024, 47, e14676. [Google Scholar] [CrossRef]
- Wen, C.; Zhang, J.; Yao, H.; Zhou, J.; Duan, Y.; Zhang, H.; Ma, H. Advances in Renewable Plant-Derived Protein Source: The Structure, Physicochemical Properties Affected by Ultrasonication. Ultrasonics Sonochemistry 2019, 53, 83–98. [Google Scholar] [CrossRef]
- Wu, P.; Bai, L.; Lin, W. On the Definition of Cavitation Intensity. Ultrasonics Sonochemistry 2020, 67, 105141. [Google Scholar] [CrossRef] [PubMed]
- Riesz, P.; Kondo, T. Free Radical Formation Induced by Ultrasound and Its Biological Implications. Free Radical Biology and Medicine 1992, 13, 247–270. [Google Scholar] [CrossRef] [PubMed]
- Alarcon-Rojo, A.D.; Janacua, H.; Rodriguez, J.C.; Paniwnyk, L.; Mason, T.J. Power Ultrasound in Meat Processing. Meat Science 2015, 107, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Mohd Azmi, S.; Kumar, P.; Sharma, N.; Sazili, A.; Lee, S.; Ismail-Fitry, M. Application of Plant Proteases in Meat Tenderization: Recent Trends and Future Prospects. Foods 2023, 12, 1336. [Google Scholar] [CrossRef]
- Stadnik, J.; Dolatowski, Z.J. Influence of Sonication on Warner-Bratzler Shear Force, Colour and Myoglobin of Beef (m. Semimembranosus). Eur Food Res Technol 2011, 233, 553–559. [Google Scholar] [CrossRef]
- Wang, L.; Wang, X.; Qin, X.; Wang, Z.; Zou, Y.; Wang, D.; Xu, W. Restructured Ground Chicken Quality Study by Ultrasound Combined with Plasma Protein Treatment. Food Bioscience 2023, 56, 103289. [Google Scholar] [CrossRef]
- Du, X.; Li, H.; Nuerjiang, M.; Shi, S.; Kong, B.; Liu, Q.; Xia, X. Application of Ultrasound Treatment in Chicken Gizzards Tenderization: Effects on Muscle Fiber and Connective Tissue. Ultrasonics Sonochemistry 2021, 79, 105786. [Google Scholar] [CrossRef]
- Hu, J.; Ge, S.; Huang, C.; Cheung, P.C.K.; Lin, L.; Zhang, Y.; Zheng, B.; Lin, S.; Huang, X. Tenderization Effect of Whelk Meat Using Ultrasonic Treatment. Food Science & Nutrition 2018, 6, 1848–1857. [Google Scholar] [CrossRef]
- Hu, Y.; Yu, H.; Dong, K.; Yang, S.; Ye, X.; Chen, S. Analysis of the Tenderisation of Jumbo Squid (Dosidicus Gigas) Meat by Ultrasonic Treatment Using Response Surface Methodology. Food Chemistry 2014, 160, 219–225. [Google Scholar] [CrossRef]
- Barekat, S.; Soltanizadeh, N. Improvement of Meat Tenderness by Simultaneous Application of High-Intensity Ultrasonic Radiation and Papain Treatment. Innovative Food Science & Emerging Technologies 2017, 39, 223–229. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, L.; Song, L.; Song, L.; Shi, S.; Liu, H.; Wu, J.; Si, K.; Gong, T.; Liu, H. Combined Treatment of Lactic Acid-Ultrasound-Papain on Yak Meat and Its Tenderization Mechanism. Meat Science 2023, 196, 109043. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wu, Z.; Bian, T.; Zhao, X. Ultrasonic-Assisted Tumbling Improves Water Retention and Tenderness of Wooden Breast Chicken Meat. Ultrasonics Sonochemistry 2025, 119, 107377. [Google Scholar] [CrossRef] [PubMed]
- Demir, H.; Çelik, S.; Sezer, Y.Ç. Effect of Ultrasonication and Vacuum Impregnation Pretreatments on the Quality of Beef Marinated in Onion Juice a Natural Meat Tenderizer. Food sci. technol. int. 2022, 28, 340–352. [Google Scholar] [CrossRef]
- Shi, H.; Zhang, X.; Chen, X.; Fang, R.; Zou, Y.; Wang, D.; Xu, W. How Ultrasound Combined with Potassium Alginate Marination Tenderizes Old Chicken Breast Meat: Possible Mechanisms from Tissue to Protein. Food Chemistry 2020, 328, 127144. [Google Scholar] [CrossRef]
- Carrillo-Lopez, L.M.; Cruz-Garibaldi, B.Y.; Huerta-Jimenez, M.; Garcia-Galicia, I.A.; Alarcon-Rojo, A.D. The Physicochemical, Microbiological, and Structural Changes in Beef Are Dependent on the Ultrasound System, Time, and One-Side Exposition. Molecules 2022, 27, 541. [Google Scholar] [CrossRef]
- Caraveo-Suarez, R.O.; Garcia-Galicia, I.A.; Santellano-Estrada, E.; Carrillo-Lopez, L.M.; Huerta-Jimenez, M.; Morales-Rodriguez, S.; Vargas-Bello-Pérez, E.; Alarcon-Rojo, A.D. Ultrasound as a Potential Technology to Improve the Quality of Meat Produced from a Mexican Autochthonous Bovine Breed. Sustainability 2022, 14, 3886. [Google Scholar] [CrossRef]
- Cao, C.; Xiao, Z.; Tong, H.; Tao, X.; Gu, D.; Wu, Y.; Xu, Z.; Ge, C. Effect of Ultrasound-Assisted Enzyme Treatment on the Quality of Chicken Breast Meat. Food and Bioproducts Processing 2021, 125, 193–203. [Google Scholar] [CrossRef]
- Maltin, C.; Balcerzak, D.; Tilley, R.; Delday, M. Determinants of Meat Quality: Tenderness. Proc. Nutr. Soc. 2003, 62, 337–347. [Google Scholar] [CrossRef]
- Bhat, Z.F.; Morton, J.D.; Mason, S.L.; Bekhit, A.E.A. Applied and Emerging Methods for Meat Tenderization: A Comparative Perspective. Comp Rev Food Sci Food Safe 2018, 17, 841–859. [Google Scholar] [CrossRef]
- Geeves, M.A. The Dynamics of Actin and Myosin Association and the Crossbridge Model of Muscle Contraction. Biochemical Journal 1991, 274, 1–14. [Google Scholar] [CrossRef]
- Linsenmayer, T.F. Collagen. In Cell Biology of Extracellular Matrix; Hay, E.D., Ed.; Springer US: Boston, MA, 1991; pp. 7–44. ISBN 978-1-4613-6680-5. [Google Scholar]
- Chang, H.; Wang, Q.; Tang, C.; Zhou, G. Effects of Ultrasound Treatment on Connective Tissue Collagen and Meat Quality of Beef Semitendinosus Muscle. Journal of Food Quality 2015, 38, 256–267. [Google Scholar] [CrossRef]
- Chen, L.; Chai, Y.; Luo, J.; Wang, J.; Liu, X.; Wang, T.; Xu, X.; Zhou, G.; Feng, X. Apoptotic Changes and Myofibrils Degradation in Post-Mortem Chicken Muscles by Ultrasonic Processing. LWT 2021, 142, 110985. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Sun, Y.Y.; Pan, D.D.; Cao, J.X. The Effect of Ultrasound Treatments on the Tenderizing Pathway of Goose Meat during Conditioning. Poultry Science 2018, 97, 2957–2965. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.A.; Campbell, P.; Kondo, T. The Role of Ca2+ in Ultrasound-Elicited Bioeffects: Progress, Perspectives and Prospects. Drug Discovery Today 2010, 15, 892–906. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Xu, Z.; Wang, H.; Xu, A.; Huan, C.; Guo, X.; Liu, R.; Wu, P.; Meng, X. Effect of Ultrasonic Processing on Beef Tenderness in Longissimus Lumborum during Aging by Proteomics Analysis. Journal of Food Composition and Analysis 2024, 131, 106220. [Google Scholar] [CrossRef]
- Zou, Y.; Jiang, D.; Xu, P.; Huang, Y.; Fang, R.; Wang, D.; Xu, W. Evaluation of the Postmortem Ageing Process of Beef M. Semitendinosus Based on Ultrasound-Assisted l-Histidine Treatment. Ultrasonics Sonochemistry 2020, 69, 105265. [Google Scholar] [CrossRef]
- Amiri, A.; Sharifian, P.; Soltanizadeh, N. Application of Ultrasound Treatment for Improving the Physicochemical, Functional and Rheological Properties of Myofibrillar Proteins. International Journal of Biological Macromolecules 2018, 111, 139–147. [Google Scholar] [CrossRef]
- Zou, Y.; Shi, H.; Xu, P.; Jiang, D.; Zhang, X.; Xu, W.; Wang, D. Combined Effect of Ultrasound and Sodium Bicarbonate Marination on Chicken Breast Tenderness and Its Molecular Mechanism. Ultrasonics Sonochemistry 2019, 59, 104735. [Google Scholar] [CrossRef]
- Roobab, U.; Chen, B.-R.; Madni, G.M.; Guo, S.-M.; Zeng, X.-A.; Abdi, G.; Aadil, R.M. Enhancing Chicken Breast Meat Quality through Ultrasonication: Physicochemical, Palatability, and Amino Acid Profiles. Ultrasonics Sonochemistry 2024, 104, 106824. [Google Scholar] [CrossRef] [PubMed]
- Cichoski, A.J.; Da Silva, J.S.; Leães, Y.S.V.; Robalo, S.S.; Dos Santos, B.A.; Reis, S.R.; Nehring, P.; Santos, S.P.; Wagner, R.; De Menezes, C.R.; et al. Effects of Ultrasonic-Assisted Cooking on the Volatile Compounds, Oxidative Stability, and Sensory Quality of Mortadella. Ultrasonics Sonochemistry 2021, 72, 105443. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.S.; Voss, M.; De Menezes, C.R.; Barin, J.S.; Wagner, R.; Campagnol, P.C.B.; Cichoski, A.J. Is It Possible to Reduce the Cooking Time of Mortadellas Using Ultrasound without Affecting Their Oxidative and Microbiological Quality? Meat Science 2020, 159, 107947. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Sun, X.; Lai, B.; Liu, R.; Wu, M.; Ge, Q.; Yu, H. Effects of Ultrasound-Assisted Cooking on the Physicochemical Properties and Microstructure of Pork Meatballs. Meat Science 2024, 208, 109382. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Zou, Y.; Zhang, W. Effects of Ultrasound-Assisted Cooking on Quality Characteristics of Spiced Beef during Cold Storage. LWT 2021, 136, 110359. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Wang, Y.; Xing, L.; Zhang, W. Influences of Ultrasonic-Assisted Frying on the Flavor Characteristics of Fried Meatballs. Innovative Food Science & Emerging Technologies 2020, 62, 102365. [Google Scholar] [CrossRef]
- Oladejo, A.O.; Ma, H.; Qu, W.; Zhou, C.; Wu, B.; Yang, X. Influence of Ultrasound Pretreatments on Diffusion Coefficients, Texture and Colour of Osmodehydrated Sweet Potato ( Ipomea Batatas ). Int J of Food Sci Tech 2017, 52, 888–896. [Google Scholar] [CrossRef]
- Oladejo, A.O.; Ma, H.; Qu, W.; Zhou, C.; Wu, B.; Yang, X.; Onwude, D.I. Effects of Ultrasound Pretreatments on the Kinetics of Moisture Loss and Oil Uptake during Deep Fat Frying of Sweet Potato (Ipomea Batatas). Innovative Food Science & Emerging Technologies 2017, 43, 7–17. [Google Scholar] [CrossRef]
- Wang, H.; Gao, Z.; Guo, X.; Gao, S.; Wu, D.; Liu, Z.; Wu, P.; Xu, Z.; Zou, X.; Meng, X. Changes in Textural Quality and Water Retention of Spiced Beef under Ultrasound-Assisted Sous-Vide Cooking and Its Possible Mechanisms. Foods 2022, 11, 2251. [Google Scholar] [CrossRef]
- Roobab, U.; Chen, B.-R.; Madni, G.M.; Tong, Z.G.; Zeng, X.-A.; Abdi, G.; Hussain, S.; Aadil, R.M. Evaluation of Ultrasound and Pulsed Electric Field Combinations on the Cooking Losses, Texture Profile, and Taste-Related Amino Acids of Chicken Breast Meat. Ultrasonics Sonochemistry 2024, 107, 106919. [Google Scholar] [CrossRef]
- Yang, W.; Wu, Z.; Song, X.; Zhao, Z. Ultrasonic Processing in Rabbit Leg Braising Advances Microstructure, Water Retention, and Flavor Development. Sci Rep 2025, 15, 36642. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Yu, Y.; Saleh, A.S.M.; Yang, X.; Ma, J.; Gao, Z.; Li, W.; Wang, Z.; Zhang, D. Structural Changes Induced by Ultrasound Improve the Ability of the Myofibrillar Protein to Bind Flavor Compounds from Spices. Ultrasonics Sonochemistry 2023, 98, 106510. [Google Scholar] [CrossRef] [PubMed]
- Tkacz, K.; Nogalski, Z.; Modzelewska-Kapituła, M. Sous-Vide Processing as a Method for Standardising the Quality of Beef from Holstein-Friesian Bulls: The Effect of Time on Tenderness. Applied Sciences 2025, 15, 12282. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, J.; Liu, E.; Yang, M.; Chen, S.; Hu, F.; Ma, H.; Liu, Z.; Yu, X. Enhancing the Taste of Raw Soy Sauce Using Low Intensity Ultrasound Treatment during Moromi Fermentation. Food Chemistry 2019, 298, 124928. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, Y.; Pan, Q.; Wang, X.; Li, P.; Cai, K.; Chen, C. Effect of Salt Mixture on Flavor of Reduced-sodium Restructured Bacon with Ultrasound Treatment. Food Science & Nutrition 2020, 8, 3857–3871. [Google Scholar] [CrossRef]
- Xi, L.; Sun, Y.; Jiang, S.; Wen, C.; Ding, W. Evaluation of Effects of Ultrasound-Assisted Curing on the Flavor of Chinese Bacon. Ultrasonics Sonochemistry 2023, 96, 106424. [Google Scholar] [CrossRef]
- Zou, Y.; Kang, D.; Liu, R.; Qi, J.; Zhou, G.; Zhang, W. Effects of Ultrasonic Assisted Cooking on the Chemical Profiles of Taste and Flavor of Spiced Beef. Ultrasonics Sonochemistry 2018, 46, 36–45. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, Y.; Hu, X.; Li, Z.; Huang, X.; Liang, J.; Zhang, X.; Zhang, D.; Zou, X.; Shi, J. Quantitative Characterization of the Diffusion Behavior of Sucrose in Marinated Beef by HSI and FEA. Meat Science 2023, 195, 109002. [Google Scholar] [CrossRef]
- Aheto, J.H.; Huang, X.; Xiaoyu, T.; Bonah, E.; Ren, Y.; Alenyorege, E.A.; Chunxia, D. Investigation into Crystal Size Effect on Sodium Chloride Uptake and Water Activity of Pork Meat Using Hyperspectral Imaging. J Food Process Preserv 2019, 43. [Google Scholar] [CrossRef]
- Liu, J.; Xie, C.; Ma, W.; Xiao, X.; Dong, W.; Chen, Y.; Hu, Y.; Feng, Y.; Yu, X. Effects of Ultrasound-Assisted Low-Salt Curing on Water Retention, Tenderness and in Vitro Digestive Characteristics of Grass Carp (Ctenopharyngodon Idellus). Ultrasonics Sonochemistry 2025, 113, 107214. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, L.; Chen, Z.; Ma, H. Tri-frequency Simultaneous Ultrasound Pickling for the Acceleration of the NaCl Content and Quality Improvement of Pork ( Longissimus Dorsi ). J Sci Food Agric 2024, 104, 6242–6251. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, X.; Hong, C.; Liu, N.; Ouyang, N.; Chen, J.; Ashokkumar, M.; Ma, H. Application of Ultrasound Treatment in Pork Marination: Effects on Moisture Migration and Microstructure. Food Chemistry 2024, 447, 138950. [Google Scholar] [CrossRef] [PubMed]
- Visy, A.; Jónás, G.; Szakos, D.; Horváth-Mezőfi, Z.; Hidas, K.I.; Barkó, A.; Friedrich, L. Evaluation of Ultrasound and Microbubbles Effect on Pork Meat during Brining Process. Ultrasonics Sonochemistry 2021, 75, 105589. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Hong, C.; Wang, W.; Zhang, X.; Chen, J.; Chen, Z.; Ashokkumar, M.; Ma, H. Evaluation of Low-Temperature Ultrasonic Marination of Pork Meat at Various Frequencies on Physicochemical Properties, Myoglobin Levels, and Volatile Compounds. Meat Science 2024, 217, 109606. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, N.; Guo, L.; Hong, C.; Wang, Y.; Guo, Y.; He, R.; Ma, H. Effects of Different Multi-Frequency Ultrasonic Modes Assisted Low-Temperature Marination On NaCl Diffusion and Protein Structure of Beef Tenderloins. Food Bioprocess Technol 2025, 18, 10948–10967. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Zhou, C.; Sun, Q.; Zhang, L.; Niu, Y.; Yao, D.; Song, L.; Okonkwo, C.E.; Phyllis, O.; et al. Ultrasonic Vacuum Synergistic Assisted Ethanol Extraction of Steviol Glycosides and Kinetic Studies. Industrial Crops and Products 2024, 221, 119385. [Google Scholar] [CrossRef]
- Pan, J.; Li, C.; Liu, X.; He, L.; Zhang, M.; Huang, S.; Huang, S.; Liu, Y.; Zhang, Y.; Jin, G. A Multivariate Insight into the Organoleptic Properties of Porcine Muscle by Ultrasound-Assisted Brining: Protein Oxidation, Water State and Microstructure. LWT 2022, 159, 113136. [Google Scholar] [CrossRef]
- Kang, D.; Wang, A.; Zhou, G.; Zhang, W.; Xu, S.; Guo, G. Power Ultrasonic on Mass Transport of Beef: Effects of Ultrasound Intensity and NaCl Concentration. Innovative Food Science & Emerging Technologies 2016, 35, 36–44. [Google Scholar] [CrossRef]
- Inguglia, E.S.; Zhang, Z.; Burgess, C.; Kerry, J.P.; Tiwari, B.K. Influence of Extrinsic Operational Parameters on Salt Diffusion during Ultrasound Assisted Meat Curing. Ultrasonics 2018, 83, 164–170. [Google Scholar] [CrossRef]
- Bai, H.; Li, L.; Wu, Y.; Chen, S.; Zhao, Y.; Cai, Q.; Wang, Y. Ultrasound Improves the Low-Sodium Salt Curing of Sea Bass: Insights into the Effects of Ultrasound on Texture, Microstructure, and Flavor Characteristics. Ultrasonics Sonochemistry 2023, 100, 106597. [Google Scholar] [CrossRef]
- Wang, X.; Xia, C.; Li, H.; Sun, Y.; Pan, D.; He, J. Ultrasound-Assisted Salt Penetration in Sauced Duck: Insights from LF-NMR and MRI Combined Analysis. Foods 2025, 14, 3553. [Google Scholar] [CrossRef]
- Kang, D.; Jiang, Y.; Xing, L.; Zhou, G.; Zhang, W. Inactivation of Escherichia Coli O157:H7 and Bacillus Cereus by Power Ultrasound during the Curing Processing in Brining Liquid and Beef. Food Research International 2017, 102, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Kutsanedzie, F.; Zhao, J.; Chen, Q. Quantifying Total Viable Count in Pork Meat Using Combined Hyperspectral Imaging and Artificial Olfaction Techniques. Food Anal. Methods 2016, 9, 3015–3024. [Google Scholar] [CrossRef]
- Sobhy, M.; Ali, S.S.; Khalil, M.A.; Chen, X.; Cui, H.; Lin, L.; El-Sapagh, S. Exploring the Potential of Zinc Oxide Nanoparticles against Pathogenic Multi-Drug Resistance Staphylococcus Aureus from Ready-to-Eat Meat and Its Proposed Mechanism. Food Control 2024, 156, 110117. [Google Scholar] [CrossRef]
- Lin, L.; Liao, X.; Li, C.; Abdel-Samie, M.A.; Siva, S.; Cui, H. Cold Nitrogen Plasma Modified Cuminaldehyde/β-Cyclodextrin Inclusion Complex and Its Application in Vegetable Juices Preservation. Food Research International 2021, 141, 110132. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Y.; Bassey, A.P.; Singh, M.; Zhu, Y.; Corradini, M.G.; Cui, X.; Zhang, X.; Liu, X. Efficacy and Mechanisms of Pseudomonas Aeruginosa PAO1 Biofilm Inactivation Using High-Power Pulsed Microwave. LWT 2024, 198, 115982. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Wu, H.; Fan, L.; Wang, Y.; Liu, X.; Zhang, H. Sterilising Effect of High Power Pulse Microwave on Listeria Monocytogenes. IFRJ 2022, 29, 1168–1178. [Google Scholar] [CrossRef]
- Cui, H.; Lu, J.; Li, C.; Rashed, M.M.A.; Lin, L. Antibacterial and Physical Effects of Cationic Starch Nanofibers Containing Carvacrol@casein Nanoparticles against Bacillus Cereus in Soy Products. International Journal of Food Microbiology 2022, 364, 109530. [Google Scholar] [CrossRef]
- Bai, M.; Dai, J.; Li, C.; Cui, H.; Lin, L. Antibacterial and Antibiofilm Performance of Low-Frequency Ultrasound against Escherichia Coli O157:H7 and Its Application in Fresh Produce. International Journal of Food Microbiology 2023, 400, 110266. [Google Scholar] [CrossRef]
- Mustapha, A.T.; Zhou, C.; Sun, Y.; Wahia, H.; Sarpong, F.; Owusu-Ansah, P.; Osae, R.; Otu, P.; Ma, H. Simultaneous Multifrequency: A Possible Alternative to Improve the Efficacy of Ultrasound Treatment on Cherry Tomato during Storage. J Food Process Preserv 2019, 43. [Google Scholar] [CrossRef]
- Zhao, C.; Dai, J.; Chen, F.; Zhao, Z.; Zhao, X. The Effect of Different Sterilization Methods on the Shelf Life and Physicochemical Indicators of Fermented Pork Jerky. Front. Nutr. 2023, 10, 1240749. [Google Scholar] [CrossRef]
- Piñon, M.I.; Alarcon-Rojo, A.D.; Renteria, A.L.; Carrillo-Lopez, L.M. Microbiological Properties of Poultry Breast Meat Treated with High-Intensity Ultrasound. Ultrasonics 2020, 102, 105680. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, X.; Chen, X.; Zheng, K.; Li, J.; Li, X. Effect of Slightly Acidic Electrolyzed water(SAEW) Combined with Ultrasound Sterilization on Quality of Bigeye Tuna (Thunnus Obesus) during Cryogenic Storage. Journal of Food Composition and Analysis 2023, 115, 104999. [Google Scholar] [CrossRef]
- Owusu-Ansah, P.; Yu, X.; Osae, R.; Mustapha, A.T.; Zhang, R.; Zhou, C. Inactivation of Bacillus Cereus from Pork by Thermal, Non-Thermal and Single-Frequency/Multi-Frequency Thermosonication: Modelling and Effects on Physicochemical Properties. LWT 2020, 133, 109939. [Google Scholar] [CrossRef]
- Valenzuela, C.; Garcia-Galicia, I.A.; Paniwnyk, L.; Alarcon-Rojo, A.D. Physicochemical Characteristics and Shelf Life of Beef Treated with High-intensity Ultrasound. J. Food Process. Preserv. 2021, 45. [Google Scholar] [CrossRef]
- Ashraf, N.; Arshad, Z.; Sami, R.; Almehmadi, A.M.; Alsanei, W.A.; Bedaiwi, R.I.; Kadi, R.H.; Abu-Zaid, A.A.; Al-Dhumri, S.A.; Abushal, S.A.; et al. Effect of Peppermint Essential Oil and Ultrasonication on Microbiology Evaluation and Quality Parameters of Stored Chicken Meat. Front. Sustain. Food Syst. 2025, 9, 1552570. [Google Scholar] [CrossRef]
- Piñon, M.; Alarcon-Rojo, A.; Paniwnyk, L.; Mason, T.; Luna, L.; Renteria, A. Ultrasound for Improving the Preservation of Chicken Meat. Food Sci. Technol 2019, 39, 129–135. [Google Scholar] [CrossRef]
- Chen, X.; Wang, J.; Han, J.; Yang, X.; Zhu, L.; Luo, X.; Zhang, Y.; Sun, D.; Zhang, X.; Liang, R. Effects of Ultrasound-Assisted Sodium Hypochlorite on the Decontamination and Storage Quality of Chilled Chicken Leg-Quarters. Food Control 2025, 168, 110895. [Google Scholar] [CrossRef]
- Krasnikova, E.S.; Morgunova, N.L.; Krasnikov, A.V.; Ivanova, Z.I.; Shorina, A.V.; Sukharev, M.I. Development of Technology for Cooked Smoked Lamb Products Using Ultrasonic Treatment. IOP Conf. Ser.: Earth Environ. Sci. 2021, 845, 012085. [Google Scholar] [CrossRef]
- Alenyorege, E.A.; Ma, H.; Ayim, I.; Zhou, C. Ultrasound Decontamination of Pesticides and Microorganisms in Fruits and Vegetables: A Review. J. Food Saf. Food Qual. 2018, 69. [Google Scholar] [CrossRef]
- Mustapha, A.T.; Zhou, C.; Amanor-Atiemoh, R.; Ali, T.A.A.; Wahia, H.; Ma, H.; Sun, Y. Efficacy of Dual-Frequency Ultrasound and Sanitizers Washing Treatments on Quality Retention of Cherry Tomato. Innovative Food Science & Emerging Technologies 2020, 62, 102348. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, D.; Sun, J.; Sun, Z.; Liu, F.; Du, L.; Wang, D. Synergistic Antibiofilm Effects of Ultrasound and Phenyllactic Acid against Staphylococcus Aureus and Salmonella Enteritidis. Foods 2021, 10, 2171. [Google Scholar] [CrossRef] [PubMed]
- Alenyorege, E.A.; Ma, H.; Ayim, I.; Aheto, J.H.; Hong, C.; Zhou, C. Reduction of Listeria Innocua in Fresh-Cut Chinese Cabbage by a Combined Washing Treatment of Sweeping Frequency Ultrasound and Sodium Hypochlorite. LWT 2019, 101, 410–418. [Google Scholar] [CrossRef]
- Song, L.; Yang, H.; Cheng, S.; Zhang, Z.; Zhang, L.; Su, R.; Li, Y.; Zhan, X.; Yang, B.; Lin, L.; et al. Combination Effects of Ultrasound and Citral Nanoemulsion against Shigella Flexneri and the Preservation Effect on Fresh-Cut Carrots. Food Control 2024, 155, 110069. [Google Scholar] [CrossRef]
- Duan, L.; Jiang, T.; Zhou, Y.; Bai, X.; Wang, Y.; Lü, X.; Xia, X.; Lin, L.; Shi, C. The Inactivation of Shigella Flexneri by Synergistic Effect of Ultrasound Combined with Basil Essential Oil Nanoemulsion and Application in Cabbage Cleaning. Food Control 2024, 156, 110142. [Google Scholar] [CrossRef]
- Li, P.; Sun, L.; Wang, J.; Wang, Y.; Zou, Y.; Yan, Z.; Zhang, M.; Wang, D.; Xu, W. Effects of Combined Ultrasound and Low-Temperature Short-Time Heating Pretreatment on Proteases Inactivation and Textural Quality of Meat of Yellow-Feathered Chickens. Food Chemistry 2021, 355, 129645. [Google Scholar] [CrossRef]
- Qian, J.; Zhao, F.; Zhang, Z.; Huo, S.; Ma, H. Effects of Ultrasound Combined with Mild Heat and Potassium Sorbate on Pichia Membranaefaciens and the Quality of Soy Sauce. Flavour & Fragrance J 2023, 38, 368–377. [Google Scholar] [CrossRef]
- Alenyorege, E.A.; Ma, H.; Ayim, I. Inactivation Kinetics of Inoculated Escherichia Coli and Listeria Innocua in Fresh-cut Chinese Cabbage Using Sweeping Frequency Ultrasound. Journal of Food Safety 2019, 39, e12696. [Google Scholar] [CrossRef]
- Mustapha, A.T.; Wahia, H.; Ji, Q.; Fakayode, O.A.; Zhang, L.; Zhou, C. Multiple-frequency Ultrasound for the Inactivation of Microorganisms on Food: A Review. J Food Process Engineering 2024, 47, e14587. [Google Scholar] [CrossRef]
- Dai, J.; Bai, M.; Li, C.; Cui, H.; Lin, L. Advances in the Mechanism of Different Antibacterial Strategies Based on Ultrasound Technique for Controlling Bacterial Contamination in Food Industry. Trends in Food Science & Technology 2020, 105, 211–222. [Google Scholar] [CrossRef]
- Dai, J.; Bai, M.; Li, C.; Cui, H.; Lin, L. Advances in the Mechanism of Different Antibacterial Strategies Based on Ultrasound Technique for Controlling Bacterial Contamination in Food Industry. Trends in Food Science & Technology 2020, 105, 211–222. [Google Scholar] [CrossRef]
- Miernik, A. Effect of Ultrasound Stimulation on the Growth of Gram Positive and Gram Negative Bacteria. ELECTROTECHNICAL REVIEW 2025, 1, 82–85. [Google Scholar] [CrossRef]
- Alenyorege, E.A.; Ma, H.; Ayim, I.; Zhou, C.; Wu, P.; Hong, C.; Osae, R. Effect of Multi-frequency Ultrasound Surface Washing Treatments on Escherichia Coli Inactivation and Some Quality Characteristics of Non-heading Chinese Cabbage. J Food Process Preserv 2018, 42, e13747. [Google Scholar] [CrossRef]
- Al-Hilphy, A.R.; Al-Asadi, M.H.; AlLami, M.K.; Tiwari, B.K.; Gavahian, M. In-Pack Sonication of Chicken Breast: Effects of Ultrasound Conditions on Physicochemical and Microbiological Properties, and Shelf-Life of Optimally Processed Meat during Frozen Storage. Food Measure 2024, 18, 8903–8917. [Google Scholar] [CrossRef]
- Sun, L.; Liu, L.; Yang, L.; Wang, Y.; Dabbour, M.; Mintah, B.K.; He, R.; Ma, H. Effects of Low-intensity Ultrasound on the Biomass and Metabolite of Ganoderma Lucidum in Liquid Fermentation. J Food Process Engineering 2021, 44, e13601. [Google Scholar] [CrossRef]
- Pitt, W.G.; Ross, S.A. Ultrasound Increases the Rate of Bacterial Cell Growth. Biotechnol. Prog. 2003, 19, 1038–1044. [Google Scholar] [CrossRef]
- Yang, Y.; Xiang, J.; Zhang, Z.; Umego, E.C.; Huang, G.; He, R.; Ma, H. Stimulation of in Situ Low Intensity Ultrasound on Batch Fermentation of Saccharomyces cerevisiae to Enhance the GSH Yield. J Food Process Engineering 2020, 43, e13489. [Google Scholar] [CrossRef]
- Shokri, S.; Terefe, N.S.; Shekarforoush, S.S.; Hosseinzadeh, S. Ultrasound-Assisted Fermentation for Enhancing Metabolic and Probiotic Activities of LactoBacillus Brevis. Chemical Engineering and Processing - Process Intensification 2021, 166, 108470. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, K.; Lu, F.; Wang, Y.; Ouyang, N.; Ma, H. Application of Ultrasound Technology in the Field of Solid-state Fermentation: Increasing Peptide Yield through Ultrasound-treated Bacterial Strain. J Sci Food Agric 2021, 101, 5348–5358. [Google Scholar] [CrossRef]
- De Lima Alves, L.; Stefanello Da Silva, M.; Martins Flores, D.R.; Rodrigues Athayde, D.; Roggia Ruviaro, A.; Da Silva Brum, D.; Fagundes Batista, V.S.; De Oliveira Mello, R.; Ragagnin De Menezes, C.; Bastianello Campagnol, P.C.; et al. Effect of Ultrasound on the Physicochemical and Microbiological Characteristics of Italian Salami. Food Research International 2018, 106, 363–373. [Google Scholar] [CrossRef]
- Wójciak, K.M.; Stasiak, D.M.; Ferysiuk, K.; Solska, E. The Influence of Sonication on the Oxidative Stability and Nutritional Value of Organic Dry-Fermented Beef. Meat Science 2019, 148, 113–119. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Zhou, L.; Zhang, R. Study on the Influences of Ultrasound on the Flavor Profile of Unsmoked Bacon and Its Underlying Metabolic Mechanism by Using HS-GC-IMS. Ultrasonics Sonochemistry 2021, 80, 105807. [Google Scholar] [CrossRef]
- Yu, H.; Liu, Y.; Yang, F.; Xie, Y.; Guo, Y.; Cheng, Y.; Yao, W. Synergistic Efficacy of High-Intensity Ultrasound and Chlorine Dioxide Combination for Staphylococcus Aureus Biofilm Control. Food Control 2021, 122, 107822. [Google Scholar] [CrossRef]
- Xu, L.; Zhou, C.; Pan, D.; Wang, Z.; Yang, D.; Cao, J.; Sant’ana, A.S.; Zhang, H.; Chen, J.; Wang, Y.; et al. Effects of Sonication and Yeast Fermentation on the Physicochemical and Sensory Features of Duck Liver Paste. International Journal of Food Properties 2025, 28, 2486342. [Google Scholar] [CrossRef]
- Wang, X.; Lan, Y.; Yang, B.; Yang, J.; Ma, J.; Cheng, M.; Xia, X.; Xu, W.; Wang, D.; Zou, Y. Effect of Low-Frequency Ultrasound Pretreatment on Taste Substances in Chicken Liver by Fermentation. LWT 2025, 218, 117458. [Google Scholar] [CrossRef]
- Hu, Y.; Dong, Z.; Wen, R.; Kong, B.; Yu, W.; Wang, J.; Liu, H.; Chen, Q. Combination of Ultrasound Treatment and Starter Culture for Improving the Quality of Beef Jerky. Meat Science 2023, 204, 109240. [Google Scholar] [CrossRef]
- De Lima Alves, L.; Donadel, J.Z.; Athayde, D.R.; Da Silva, M.S.; Klein, B.; Fagundes, M.B.; De Menezes, C.R.; Barin, J.S.; Campagnol, P.C.B.; Wagner, R.; et al. Effect of Ultrasound on Proteolysis and the Formation of Volatile Compounds in Dry Fermented Sausages. Ultrasonics Sonochemistry 2020, 67, 105161. [Google Scholar] [CrossRef]
- Bao, G.; Niu, J.; Li, S.; Zhang, L.; Luo, Y. Effects of Ultrasound Pretreatment on the Quality, Nutrients and Volatile Compounds of Dry-Cured Yak Meat. Ultrasonics Sonochemistry 2022, 82, 105864. [Google Scholar] [CrossRef]
- Hao, J.; Xu, H.; Yan, P.; Yang, M.; Mintah, B.K.; Dai, C.; Zhang, R.; Ma, H.; He, R. Application of Fixed-frequency Ultrasound in the Cultivation of Saccharomyces Cerevisiae for Rice Wine Fermentation. J Sci Food Agric 2024, 104, 6417–6430. [Google Scholar] [CrossRef]
- Yu, Z.; Su, Y.; Zhang, Y.; Zhu, P.; Mei, Z.; Zhou, X.; Yu, H. Potential Use of Ultrasound to Promote Fermentation, Maturation, and Properties of Fermented Foods: A Review. Food Chemistry 2021, 357, 129805. [Google Scholar] [CrossRef]
- Zhang, Y.; Magro, A.; Puolanne, E.; Zotte, A.D.; Ertbjerg, P. Myofibrillar Protein Characteristics of Fast or Slow Frozen Pork during Subsequent Storage at −3 °C. Meat Science 2021, 176, 108468. [Google Scholar] [CrossRef]
- Yu, H.; Mei, J.; Xie, J. New Ultrasonic Assisted Technology of Freezing, Cooling and Thawing in Solid Food Processing: A Review. Ultrasonics Sonochemistry 2022, 90, 106185. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Sun, F.; Xia, X.; Xu, H.; Kong, B. The Comparison of Ultrasound-Assisted Immersion Freezing, Air Freezing and Immersion Freezing on the Muscle Quality and Physicochemical Properties of Common Carp (Cyprinus Carpio) during Freezing Storage. Ultrasonics Sonochemistry 2019, 51, 281–291. [Google Scholar] [CrossRef]
- Astráin-Redín, L.; Abad, J.; Rieder, A.; Kirkhus, B.; Raso, J.; Cebrián, G.; Álvarez, I. Direct Contact Ultrasound Assisted Freezing of Chicken Breast Samples. Ultrasonics Sonochemistry 2021, 70, 105319. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Cui, F.; Wang, J.; Zhu, W.; Xu, Y.; Yi, S.; Li, X.; Li, J. Effects of Ultrasound-Assisted Immersion Freezing on the Muscle Quality and Myofibrillar Protein Oxidation and Denaturation in Sciaenops Ocellatus. Food Chemistry 2022, 377, 131949. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Bao, Y.; Gou, H.; Xu, B.; Hong, H.; Gao, R. Mitigation of Mechanical Damage and Protein Deterioration in Giant River Prawn (Macrobrachium Rosenbergii) by Multi-Frequency Ultrasound-Assisted Immersion Freezing. Food Chemistry 2024, 458, 140324. [Google Scholar] [CrossRef]
- Sardari, H.; Alimardani, R.; Soltani Firouz, M.; Hosseinpour, S. Probe-Type Ultrasound-Assisted Immersion Freezing of Meat, Study of Selected Quality Factors and Taguchi-Based Process Optimization. Applied Food Research 2025, 5, 101115. [Google Scholar] [CrossRef]
- Chen, X.; Liu, H.; Li, X.; Wei, Y.; Li, J. Effect of Ultrasonic-Assisted Immersion Freezing and Quick-Freezing on Quality of Sea Bass during Frozen Storage. LWT 2022, 154, 112737. [Google Scholar] [CrossRef]
- Zhang, M.; Haili, N.; Chen, Q.; Xia, X.; Kong, B. Influence of Ultrasound-Assisted Immersion Freezing on the Freezing Rate and Quality of Porcine Longissimus Muscles. Meat Science 2018, 136, 1–8. [Google Scholar] [CrossRef]
- Chen, X.; Liu, H.; Li, X.; Wei, Y.; Li, J. Effect of Ultrasonic-Assisted Immersion Freezing and Quick-Freezing on Quality of Sea Bass during Frozen Storage. LWT 2022, 154, 112737. [Google Scholar] [CrossRef]
- Xu, B.; Zhang, M.; Bhandari, B.; Sun, J.; Gao, Z. Infusion of CO2 in a Solid Food: A Novel Method to Enhance the Low-Frequency Ultrasound Effect on Immersion Freezing Process. Innovative Food Science & Emerging Technologies 2016, 35, 194–203. [Google Scholar] [CrossRef]
- Zhang, P.; Zhu, Z.; Sun, D.-W. Using Power Ultrasound to Accelerate Food Freezing Processes: Effects on Freezing Efficiency and Food Microstructure. Critical Reviews in Food Science and Nutrition 2018, 58, 2842–2853. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, Y.; Xia, X.; Liu, Q.; Chen, Q.; Kong, B. Changes in Muscle Quality and Physicochemical Characteristics of Chicken Breast Subjected to Ultrasound-Assisted Immersion Freezing during Long-Term Frozen Storage. International Journal of Refrigeration 2022, 142, 10–18. [Google Scholar] [CrossRef]
- Carrillo-Lopez, L.M.; Robledo, D.; Martínez, V.; Huerta-Jimenez, M.; Titulaer, M.; Alarcon-Rojo, A.D.; Chavez-Martinez, A.; Luna-Rodriguez, L.; Garcia-Flores, L.R. Post-Mortem Ultrasound and Freezing of Rabbit Meat: Effects on the Physicochemical Quality and Weight Loss. Ultrasonics Sonochemistry 2021, 79, 105766. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Ertbjerg, P.; Estévez, M.; Yuan, L.; Gao, R. Freezing of Meat and Aquatic Food: Underlying Mechanisms and Implications on Protein Oxidation. Comp Rev Food Sci Food Safe 2021, 20, 5548–5569. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Sun, Q.; Xuan, L.; Lin, Z.; Yang, Z.; Huang, X.; Li, Z.; Gao, W.; Ren, J.; Shi, J.; et al. Ultrasonic Technology for Predicting Beef Thawing Degree and Endpoint. Journal of Food Engineering 2024, 383, 112236. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, Y.; Zhao, J.; Sun, J. Ultrasound Thawing for Improving the Eating Quality and Off-Flavor of Frozen Duck Meat and Its Possible Mechanisms. LWT 2023, 187, 115314. [Google Scholar] [CrossRef]
- Shi, W.; Wang, Y.; Chen, X.; Hu, H.; Chen, X.; Mintah, B.K.; Dabbour, M.; Meng, X.; Zhang, Z.; He, R.; et al. Thawing of Goose Meat by Simultaneous Dual Frequency Ultrasound for Process Efficiency: Thawing Rate, Quality Attributes, Structural Characteristics. Innovative Food Science & Emerging Technologies 2024, 92, 103588. [Google Scholar] [CrossRef]
- Zhang, C.; Sun, Q.; Chen, Q.; Liu, Q.; Kong, B. Effectiveness of Ultrasound-assisted Immersion Thawing on the Thawing Rate and Physicochemical Properties of Chicken Breast Muscle. Journal of Food Science 2021, 86, 1692–1703. [Google Scholar] [CrossRef]
- Guo, Z.; Ge, X.; Yang, L.; Ma, G.; Ma, J.; Yu, Q.; Han, L. Ultrasound-Assisted Thawing of Frozen White Yak Meat: Effects on Thawing Rate, Meat Quality, Nutrients, and Microstructure. Ultrasonics Sonochemistry 2021, 70, 105345. [Google Scholar] [CrossRef]
- Xu, C.; Zang, M.; Qiao, X.; Wang, S.; Zhao, B.; Shi, Y.; Bai, J.; Wu, J. Effects of Ultrasound-Assisted Thawing on Lamb Meat Quality and Oxidative Stability during Refrigerated Storage Using Non-Targeted Metabolomics. Ultrasonics Sonochemistry 2022, 90, 106211. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, Y.; Guo, L.; Yolandani; Ouyang, N.; Wang, B. Application of Multi-Frequency Ultrasonic Thawing on Pork: Thawing Rate, Quality Properties and Microstructure. Food Physics 2024, 1, 100002. [Google Scholar] [CrossRef]
- Cai, L.; Zhang, W.; Cao, A.; Cao, M.; Li, J. Effects of Ultrasonics Combined with Far Infrared or Microwave Thawing on Protein Denaturation and Moisture Migration of Sciaenops Ocellatus (Red Drum). Ultrasonics Sonochemistry 2019, 55, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Chen, J.; Yuan, J.; Azam, S.R.; Zhang, M. Effect of Different Thawing Methods on the Efficiency and Quality Attributes of Frozen Red Radish. J Sci Food Agric 2021, 101, 3237–3245. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Wang, H.; Zhou, F.; Wu, Y.; Ma, H.; Zhao, R.; He, J.; Gu, Z. Effect of Ultrasonic Thawing Temperature on the Quality of Quick-Frozen Small Yellow Croaker (Larimichthys Polyactis) and Its Possible Mechanisms. LWT 2023, 179, 114620. [Google Scholar] [CrossRef]
- Qiu, L.; Zhang, M.; Chitrakar, B.; Bhandari, B. Application of Power Ultrasound in Freezing and Thawing Processes: Effect on Process Efficiency and Product Quality. Ultrasonics Sonochemistry 2020, 68, 105230. [Google Scholar] [CrossRef]
- Wu, B.; Qiu, C.; Guo, Y.; Zhang, C.; Guo, X.; Bouhile, Y.; Ma, H. Ultrasonic-Assisted Flowing Water Thawing of Frozen Beef with Different Frequency Modes: Effects on Thawing Efficiency, Quality Characteristics and Microstructure. Food Research International 2022, 157, 111484. [Google Scholar] [CrossRef]
- Wang, B.; Bai, X.; Du, X.; Pan, N.; Shi, S.; Xia, X. Comparison of Effects from Ultrasound Thawing, Vacuum Thawing and Microwave Thawing on the Quality Properties and Oxidation of Porcine Longissimus Lumborum. Foods 2022, 11, 1368. [Google Scholar] [CrossRef]
- Wang, X.; Dong, Y.; Wu, R.; Liu, D.; Hu, F.; Wang, C.; Wu, D. A Method to Improve Water-holding Capacity of Beef during Freezing-thawing Process Using Ultrasound Treatment. J Food Process Preserv 2021, 45. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Yan, J.-K.; Ding, Y.; Rashid, M.T.; Ma, H. Effect of Sweep Frequency Ultrasound and Fixed Frequency Ultrasound Thawing on Gelling Properties of Myofibrillar Protein from Quick-Frozen Small Yellow Croaker and Its Possible Mechanisms. LWT 2021, 150, 111922. [Google Scholar] [CrossRef]
- Zhang, L.H.; Zhu, J.L.; Wang, S.J.; Chen, L.; Song, Z.H.; Zhang, L.; Ma, H.L. Effect of radio frequency energy combined with natamycin on Aspergillus niger survival and quality of dried shiitake mushroom with different moisture content. Food Control 2025, 170, 111053. [Google Scholar] [CrossRef]
- Osae, R.; Zhou, C.; Xu, B.; Tchabo, W.; Tahir, H.E.; Mustapha, A.T.; Ma, H. Effects of Ultrasound, Osmotic Dehydration, and Osmosonication Pretreatments on Bioactive Compounds, Chemical Characterization, Enzyme Inactivation, Color, and Antioxidant Activity of Dried Ginger Slices. J Food Biochem 2019, 43, e12832. [Google Scholar] [CrossRef]
- Feng, M.; Xu, B.; Nahidul Islam, Md.; Zhou, C.; Wei, B.; Wang, B.; Ma, H.; Chang, L. Individual and Synergistic Effect of Multi-Frequency Ultrasound and Electro-Infrared Pretreatments on Polyphenol Accumulation and Drying Characteristics of Edible Roses. Food Research International 2023, 163, 112120. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wu, B.; Lu, D.; Pan, Z.; Ma, H. Tri-Frequency Ultrasound as Pretreatment to Infrared Drying of Carrots: Impact on Enzyme Inactivation, Color Changes, Nutrition Quality Parameters and Microstructures. International Journal of Food Engineering 2021, 17, 275–284. [Google Scholar] [CrossRef]
- Rashid, M.T.; Ma, H.; Jatoi, M.A.; Hashim, M.M.; Wali, A.; Safdar, B. Influence of Ultrasonic Pretreatment with Hot Air Drying on Nutritional Quality and Structural Related Changes in Dried Sweet Potatoes. International Journal of Food Engineering 2019, 15, 20180409. [Google Scholar] [CrossRef]
- Oladejo, A.O.; Ma, H.; Qu, W.; Zhou, C.; Wu, B. Effects of Ultrasound on Mass Transfer Kinetics, Structure, Carotenoid and Vitamin C Content of Osmodehydrated Sweet Potato (Ipomea Batatas). Food Bioprocess Technol 2017, 10, 1162–1172. [Google Scholar] [CrossRef]
- Oladejo, A.O.; Ma, H. Optimisation of Ultrasound-assisted Osmotic Dehydration of Sweet Potato ( Ipomea Batatas ) Using Response Surface Methodology. J Sci Food Agric 2016, 96, 3688–3693. [Google Scholar] [CrossRef]
- Xu, B.; Sylvain Tiliwa, E.; Yan, W.; Roknul Azam, S.M.; Wei, B.; Zhou, C.; Ma, H.; Bhandari, B. Recent Development in High Quality Drying of Fruits and Vegetables Assisted by Ultrasound: A Review. Food Research International 2022, 152, 110744. [Google Scholar] [CrossRef]
- Başlar, M.; Kılıçlı, M.; Toker, O.S.; Sağdıç, O.; Arici, M. Ultrasonic Vacuum Drying Technique as a Novel Process for Shortening the Drying Period for Beef and Chicken Meats. Innovative Food Science & Emerging Technologies 2014, 26, 182–190. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, L.; Yagoub, A.E.A.; Yu, X.; Ma, H.; Zhou, C. Effects of Ultrasound, Freeze-Thaw Pretreatments and Drying Methods on Structure and Functional Properties of Pectin during the Processing of Okra. Food Hydrocolloids 2021, 120, 106965. [Google Scholar] [CrossRef]
- Aksoy, A.; Karasu, S.; Akcicek, A.; Kayacan, S. Effects of Different Drying Methods on Drying Kinetics, Microstructure, Color, and the Rehydration Ratio of Minced Meat. Foods 2019, 8, 216. [Google Scholar] [CrossRef]
- Jiang, M.; Sun, J.; Obadi, M.; Bai, X.; Zhu, W. Effects of Ultrasonic Vacuum Drying on the Drying Kinetics, Dynamic Moisture Distribution, and Microstructure of Honey Drying Process. Food sci. technol. int. 2021, 27, 426–440. [Google Scholar] [CrossRef]
- Rashid, M.T.; Ma, H.; Jatoi, M.A.; Wali, A.; El-Mesery, H.S.; Ali, Z.; Sarpong, F. Effect of Infrared Drying with Multifrequency Ultrasound Pretreatments on the Stability of Phytochemical Properties, Antioxidant Potential, and Textural Quality of Dried Sweet Potatoes. J Food Biochem 2019, 43, e12809. [Google Scholar] [CrossRef]
- Guo, Y.; Wu, B.; Guo, X.; Ding, F.; Pan, Z.; Ma, H. Effects of Power Ultrasound Enhancement on Infrared Drying of Carrot Slices: Moisture Migration and Quality Characterizations. LWT 2020, 126, 109312. [Google Scholar] [CrossRef]
- Gu, C.; Ma, H.; Tuly, J.A.; Guo, L.; Zhang, X.; Liu, D.; Ouyang, N.; Luo, X.; Shan, Y. Effects of Catalytic Infrared Drying in Combination with Hot Air Drying and Freeze Drying on the Drying Characteristics and Product Quality of Chives. LWT 2022, 161, 113363. [Google Scholar] [CrossRef]
- Su, D.; Sun, W.; Li, B.; Yang, Y.; Wang, Y.; Lv, W.; Li, D.; Wang, L. Influence of Ultrasonic Pretreatments on Microwave Hot-air Flow Rolling Drying Mechanism, Thermal Characteristics and Rehydration Dynamics of Pleurotus Eryngii. J Sci Food Agric 2022, 102, 2100–2109. [Google Scholar] [CrossRef] [PubMed]
- Rashid, M.T.; Ma, H.; Jatoi, M.A.; Safdar, B.; El-Mesery, H.S.; Sarpong, F.; Ali, Z.; Wali, A. Multi-frequency Ultrasound and Sequential Infrared Drying on Drying Kinetics, Thermodynamic Properties, and Quality Assessment of Sweet Potatoes. J Food Process Engineering 2019, 42, e13127. [Google Scholar] [CrossRef]
- Amanor-Atiemoh, R.; Zhou, C.; Abdullaleef Taiye, M.; Sarpong, F.; Wahia, H.; Amoa-Owusu, A.; Ma, H.; Chen, L. Effect of Ultrasound-ethanol Pretreatment on Drying Kinetics, Quality Parameters, Functional Group, and Amino Acid Profile of Apple Slices Using Pulsed Vacuum Drying. J Food Process Engineering 2020, 43, e13347. [Google Scholar] [CrossRef]
- Zhou, C.; Wang, Z.; Wang, X.; Yagoub, A.E.; Ma, H.; Sun, Y.; Yu, X. Effects of Tri-frequency Ultrasound-ethanol Pretreatment Combined with Infrared Convection Drying on the Quality Properties and Drying Characteristics of Scallion Stalk. J Sci Food Agric 2021, 101, 2809–2817. [Google Scholar] [CrossRef]
- Gao, J.; Cheng, S.; Sun, X.; Bai, Y.; Yu, X.; Zeng, X.; Hu, S.; Zhang, M.; Yue, J.; Xu, X.; et al. Combination of Contact Ultrasound and Infrared Radiation for Improving the Quality and Flavor of Air-Dried Beef during Hot Air Drying. Ultrasonics Sonochemistry 2024, 110, 107047. [Google Scholar] [CrossRef]
- Mtetwa, M.D.; Qian, L.; Zhu, H.; Cui, F.; Zan, X.; Sun, W.; Wu, D.; Yang, Y. Ultrasound-Assisted Extraction and Antioxidant Activity of Polysaccharides from Acanthus Ilicifolius. Food Measure 2020, 14, 1223–1235. [Google Scholar] [CrossRef]
- Qu, W.; Fan, W.; Feng, Y.; Li, Y.; Ma, H.; Pan, Z. Preparation of Heat-Sensitivity Proteins from Walnut Meal by Sweep Frequency Ultrasound-Assisted Alkali Extraction. Journal of Food Quality 2021, 2021, 1–12. [Google Scholar] [CrossRef]
- Feng, Y.; Juliet, I.C.; Wen, C.; Duan, Y.; Zhou, J.; He, Y.; Zhang, H.; Ma, H. Effects of Multi-Mode Divergent Ultrasound Pretreatment on the Physicochemical and Functional Properties of Polysaccharides from Sagittaria Sagittifolia L. Food Bioscience 2021, 42, 101145. [Google Scholar] [CrossRef]
- Muatasim, R.; Ma, H.; Yang, X. Effect of Multimode Ultrasound Assisted Extraction on the Yield of Crude Polysaccharides from Lycium Barbarum (Goji). Food Sci. Technol 2018, 38, 160–166. [Google Scholar] [CrossRef]
- Geng, N.; Song, J.; Luo, S.; Li, Y.; Wu, G.; Liu, C.; Wu, C. Ultrasound-Assisted Enzymatic Extraction of Soluble Dietary Fiber from Fresh Corn Bract and Its Physio-Chemical and Structural Properties. Qual. Assur. Saf. Crops Foods 2022, 14, 119–130. [Google Scholar] [CrossRef]
- Yang, F.; Chen, W.; Dabbour, M.; Kumah Mintah, B.; Xu, H.; Pan, J.; Dai, C.; Ma, H.; He, R. Preparation of Housefly (Musca Domestica) Larvae Protein Hydrolysates: Influence of Dual-Sweeping-Frequency Ultrasound-Assisted Enzymatic Hydrolysis on Yield, Antioxidative Activity, Functional and Structural Attributes. Food Chemistry 2024, 440, 138253. [Google Scholar] [CrossRef]
- Chikari, F.; Han, J.; Wang, Y.; Luo, P.; He, X.; Kwaw, E.; Otu, P. Dual-frequency Ultrasound-assisted Alcohol/Salt Aqueous Two-phase Extraction and Purification of Astragalus Polysaccharides. J Food Process Engineering 2020, 43, e13366. [Google Scholar] [CrossRef]
- Cheng, Y.; Shi, X.; Yeboah, G.B.; Chen, L.; Wu, J. Effect of Multi-Mode Divergent Ultrasound Pretreatment on Hardness, Microstructure and Digestion of Acid-Induced Whey Protein Gels. Foods 2024, 13, 1926. [Google Scholar] [CrossRef]
- Qian, J.; Chen, D.; Zhang, Y.; Gao, X.; Xu, L.; Guan, G.; Wang, F. Ultrasound-Assisted Enzymatic Protein Hydrolysis in Food Processing: Mechanism and Parameters. Foods 2023, 12, 4027. [Google Scholar] [CrossRef]
- Ran, X.; Wang, L. Use of Ultrasonic and Pepsin Treatment in Tandem for Collagen Extraction from Meat Industry By-products. J Sci Food Agric 2014, 94, 585–590. [Google Scholar] [CrossRef]
- Schmidt, M.M.; Vidal, A.R.; Mello, R.O.; Mazutti, M.A.; Cansian, R.L.; Dornelles, R.C.P.; Demiate, I.M.; Kubota, E.H. Ultrasound as an Alternative Method to Increase the Extraction Yield from Chicken Mecanically Separated Meatresidue Collagen. J Food Sci Technol 2021, 58, 2487–2496. [Google Scholar] [CrossRef]
- Yu, X.; Tu, S.; Li, Y.; Yagoub, A.E.A.; Ma, H.; Zhou, C. Effects of Single- and Tri-Frequency Ultrasound on Self-Assembly and Characterizations of Bionic Dynamic Rat Stomach Digestion of Pepsin-Soluble Collagen from Chicken Leg Skin. Food Research International 2020, 137, 109710. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Feng, M.; Tiliwa, E.S.; Yan, W.; Wei, B.; Zhou, C.; Ma, H.; Wang, B.; Chang, L. Multi-Frequency Power Ultrasound Green Extraction of Polyphenols from Pingyin Rose: Optimization Using the Response Surface Methodology and Exploration of the Underlying Mechanism. LWT 2022, 156, 113037. [Google Scholar] [CrossRef]
- Gulzar, S.; Benjakul, S. Ultrasound Waves Increase the Yield and Carotenoid Content of Lipid Extracted From Cephalothorax of Pacific White Shrimp ( Litopenaeus Vannamei ). Euro J Lipid Sci & Tech 2018, 120, 1700495. [Google Scholar] [CrossRef]
- Sinthusamran, S.; Benjakul, S.; Kijroongrojana, K.; Prodpran, T.; Agustini, T.W. Yield and Chemical Composition of Lipids Extracted from Solid Residues of Protein Hydrolysis of Pacific White Shrimp Cephalothorax Using Ultrasound-Assisted Extraction. Food Bioscience 2018, 26, 169–176. [Google Scholar] [CrossRef]
- Ojha, K.S.; Kerry, J.P.; Tiwari, B.K. Investigating the Influence of Ultrasound Pre-Treatment on Drying Kinetics and Moisture Migration Measurement in Lactobacillus Sakei Cultured and Uncultured Beef Jerky. LWT - Food Science and Technology 2017, 81, 42–49. [Google Scholar] [CrossRef]
- Hao, G.; Lin, S.; Jiang, Y.; Cao, W.; Liu, Y.; Chen, Z. Enhancing Processed Quality of Roasted Eel with Ultrasound Treatment: Effect on Texture, Taste, and Flavor. Food Processing Preservation 2022, 46. [Google Scholar] [CrossRef]
- Shi, Z.; Zhong, S.; Yan, W.; Liu, M.; Yang, Z.; Qiao, X. The Effects of Ultrasonic Treatment on the Freezing Rate, Physicochemical Quality, and Microstructure of the Back Muscle of Grass Carp (Ctenopharyngodon Idella). LWT 2019, 111, 301–308. [Google Scholar] [CrossRef]
- Zhou, C.; Hu, J.; Yu, X.; Yagoub, A.E.A.; Zhang, Y.; Ma, H.; Gao, X.; Otu, P.N.Y. Heat and/or Ultrasound Pretreatments Motivated Enzymolysis of Corn Gluten Meal: Hydrolysis Kinetics and Protein Structure. LWT 2017, 77, 488–496. [Google Scholar] [CrossRef]
- Cheng, Y.; Donkor, P.O.; Ren, X.; Wu, J.; Agyemang, K.; Ayim, I.; Ma, H. Effect of Ultrasound Pretreatment with Mono-Frequency and Simultaneous Dual Frequency on the Mechanical Properties and Microstructure of Whey Protein Emulsion Gels. Food Hydrocolloids 2019, 89, 434–442. [Google Scholar] [CrossRef]
- Fu, Y.; Jiang, G.; Sun, X.; Yang, S.; Yu, J.; Liu, X.; Wang, L.; Zhu, S. Ultrasound-Assisted Enhancement of Gel Properties in Hypomesus Olidus Surimi. Foods 2025, 14, 2363. [Google Scholar] [CrossRef]

| Material | Treatment methods | Optimal parameters | Main results | References |
|---|---|---|---|---|
| Chicken gizzards | US | 500 W, 30 kHz, 3 s/3 s, 30 min | Decreasing the shear force and muscle fiber diameter by 27.1% and 26.2%, increasing the myofibril fragmentation index by 238.1%, and lowering hydroxylysine pyridinoline and lysine pyridinoline by 23.1% and 40.5%, respectively. | [49] |
| Yak meat |
US+Lactic acid+ Papain | 350 W, 45 kHz, 30 min; 0.03% lactic acid, and 200 U/g Papain | Exhibiting a 62.16% reduction in cutting force, a 31.25% decrease in cooking loss, and a 4.3-fold increase in the myofibrillar rupture index. | [53] |
| Old chicken breast meat | US+Potassium alginate (PA) (UPA) | 300 W, 20 kHz, 15.6 W/cm2, 5 min; (PA; 0.2%–1.0%, 25 min) | UPA-0.4% group exhibiting the lowest moisture loss (drip loss: 1.29±0.09 g/kg and cooking loss: 16.53±0.20 g/kg), and shear force (12.67±0.52 N), respectively. | [56] |
| Beef | US | 37 kHz, 90 W/cm2 25 min (bath); or 24 kHz, 400 W, 50 min (probe) | Decreasing the water holding capacity and shear force by 3.1%–5% and 0.59–0.72 kgf, favoring the muscle tenderization after storage, and significantly increasing the muscle lightness, respectively. | [57] |
| 45 kHz, 11 W/cm2, 20 min, 4.7–6 °C | Increasing the luminosity and yellowness (b*), and decreasing the shear force of meat, showing a tenderizing effect. | [58] | ||
| Spent-hen breast meat | US+Papain | 300 W, 40 kHz, 30 °C, < 20 min | Significantly reducing shear force, and increasing water holding capacity (p < 0.05). | [59] |
| Material | Treatment methods | US parameters | Main results | References |
|---|---|---|---|---|
| Fermented pork jerky | US | 480 W, 30 kHz, 30 min | No coliform bacteria, Salmonella, or Shigella were detected during the storage. S. aureus was first observed on day 18. | [112] |
| Chicken breasts | US | 40 kHz, 9.6 W/cm2, 50 min | The number of S. aureus decreased significantly after 7 d of refrigeration. | [113] |
| Cooked smoked lamb products | US | 35 kHz, 2 min; 26 kHz, 1 min | C. albicans, E. coli, B. subbillis, and S. aureus was reduced by 33.3%, 43.8%, 46.8%, and 80.6% by US (35 kHz, 2 min). US (26 kHz, 1 min) reduced them by 50%, 64.6%, 89.1%, and 86.8%, respectively. | [120] |
| Semitendinosus beef muscle | US | 40 kHz, 11 W/cm2 | Significantly reducing the counts of mesophilic bacteria, psychrophilic bacteria, Staphylococcus spp., and coliform bacteria. | [116] |
| Pork meat | US+heating | 20/40/60 kHz, 70°C heating, 5 min | Reduction in B.cereus spores at 4.16 log CFU/mL | [115] |
| Tuna fish | US + slightly acidic electrolyzed water (SAEW) |
55 mg/mL SAEW + 280 W US | Demonstrating the strong antibacterial effect during storage, with a total viable count (TVC) of 3.11 ± 0.01 log CFU/g. | [114] |
| Chicken meat | US+Peppermint essential oil | 37 kHz, 600 W, 2/4/6 min | During refrigeration (12 d, 4 °C), the treated group exhibited the total plate count, Salmonella, coliform, and E. coli of 1.53–3.76, 1.21–1.99 , 1.08–1.48, and 1.95–2.99 CFU/g, significantly lower than untreated group of 2.4–7.71, 3.56–5.61, 1.87–4.41, and 4.47–7.23 CFU/g, respectively. | [117] |
| US | 20 kHz, 27.6 W/cm2; 40 kHz, 10.3 W/cm2; 850 kHz, 24.1 W/cm2 | Microbial counts in samples treated at 20 kHz (3.7 ± 0.4 and 8 ± 0.6 log CFU/g) were significantly lower than the control and the groups treated at 40 kHz and 850 kHz. | [118] | |
| US+sodium hypochlorite (SH) |
25 kHz, 6 kw, 25 min | US+SH and 2×(US+SH) reduced TVC, Enterobacteriaceae, and psychrophilic bacteria on the surface at day 0, while preserving sensory quality during 5 d of chilled storage. 2×(US+SH) achieved reduction in TVC (4.72 log CFU/g), Enterobacteriaceae (2.83 log CFU/g), and psychrophilic bacteria (4.89 log CFU/g), inhibiting the growth of Acinetobacter, Aeromonas, Shewanella, and Pseudomonas, extending shelf life by more than 2 d. | [119] |
| Material | Treatment methods | US parameters | Main results | References |
|---|---|---|---|---|
| Chicken breast | US in an air-forced cooling tunnel | 37% net sonication time; 40 kHz; 50 W, -13 to -22 ℃ | Reducing freezing time by 11%. No significant differences between US-assisted frozen samples and the control in WHC and cooking loss. | [155] |
| Ultrasound-assisted immersion freezing (UIF) | 165 W | UIF produced smaller ice crystals than immersion freezing (IF) and air freezing (AF). It significantly reduced thawing and cooking losses, color deterioration, and lipid oxidation levels during frozen storage (p < 0.05). UF minimized water migration throughout storage (p < 0.05). | [152] | |
| Sciaenops ocellatus | UIF | 150, 200, and 250W, 25 kHz | UIF (200 W) increased freezing rate by 712.81% and shortened freezing time by > 83% compared with AF. UIF (200 W) group had higher protein stability after 90 days of storage. | [156] |
| Macrobrachium rosenbergii | Multi-frequency ultrasound-assisted immersion freezing (MUIF) | MUIF (20+40 kHz), (20+60 kHz), (40+60 kHz), and (20+40+60 kHz), 180 W | The average diameter of ice crystals in IF is 28 micrometers, whereas in MUIF-20 + 40 + 60 it is only 8 micrometers. MUIF mitigates oxidative degradation of lipids and proteins. | [157] |
| Beef | UIF | 0-400 W, 24 kHz | UIF (125 W, 50% duty cycle) significantly reduced freezing time, thawing and cooking losses, and improved color quality of the samples. | [158] |
| Sea bass | UIF | 320W, 45 kHz, -20 ± 0.5°C and -40 ± 0.5°C | UIF (at -40°C) lowered ice crystals, maintained stable proteins during frozen storage, and reduced thawing and cooking losses. | [159] |
| Material | Treatment methods | US parameters | Main results | References |
|---|---|---|---|---|
| White yak meat | US-assisted thawing (UAT) | 0, 200, 400, and 600 W, 20 kHz | Shortening thawing time by 30.95%–64.28%; reducing thawing loss, cooking loss, L* and b* values, and pH (p < 0.05), whereas increasing a* value and cutting force, free amino acids, minerals, water-soluble vitamins, and volatile compounds (p < 0.05). | [171] |
| Lamb | UAT | 350W, 40 kHz | Improving the water retention capacity, preserving color, and effectively inhibiting protein oxidation (p < 0.05). | [172] |
| Duck meat | UAT | 200, 400, and 600 W, 40 kHz | Shortening thawing time by 30.96%–55.05% with power (200 to 600W), reducing thawing loss, pH, L*, b*, shear force, and pressure water loss, and increasing a*, color, tenderness, and WHC (p < 0.05) by UAT (400 W), and reducing the off-flavor. | [168] |
| Chicken breast | UAT | 200, 300, 400, and 500 W, 15 ± 0.5 ℃ | UAT (300 W) decreasing thawing time by57% compared to air thawing, and reducing the damage to myofibrillar protein structure. | [170] |
| Goose meat | Simultaneous dual-frequency US (SDU) | Combining 20, 25, 28, 40, 50 kHz in pairs, 25±1°C, 50 W/L | Shortening thawing time by 17.76–36.06% over running water thawing; achieving the lowest thawing loss (13.36%) and surface hydrophobicity (67.76 μg) by SDU (28+50 kHz) . | [169] |
| Pork | US (mono-, dual- or tri-frequency sequential and simultaneous mode) | 20, 35, 50 kHz; 20/35, 20/50, 35/50 kHz; 20/35/50, 20/50/35 kHz; 20+35, 20+50, 35+50, and 20+35+50 kHz, 40 W/L | Reducing thawing time by 26.72%-64.99%, inhibiting lipid oxidation; UAT (20/50/35 kHz) achieving superior water retention (lower thawing and cooking losses) and physicochemical quality (lowest hardness, highest tenderness, and reduced TBARS values), withmicrostructure most resembling fresh meat. | [173] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




