Submitted:
22 March 2026
Posted:
23 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- -
- explicit communication of AI’s role in clinical reasoning, documentation, and research;
- -
- clear statements regarding clinician oversight and ultimate decision-making responsibility;
- -
- differentiation between consent for AI research and consent for AI-assisted clinical care; and
- -
- the potential utility of dynamic or tiered consent models reflecting varying degrees of AI involvement.
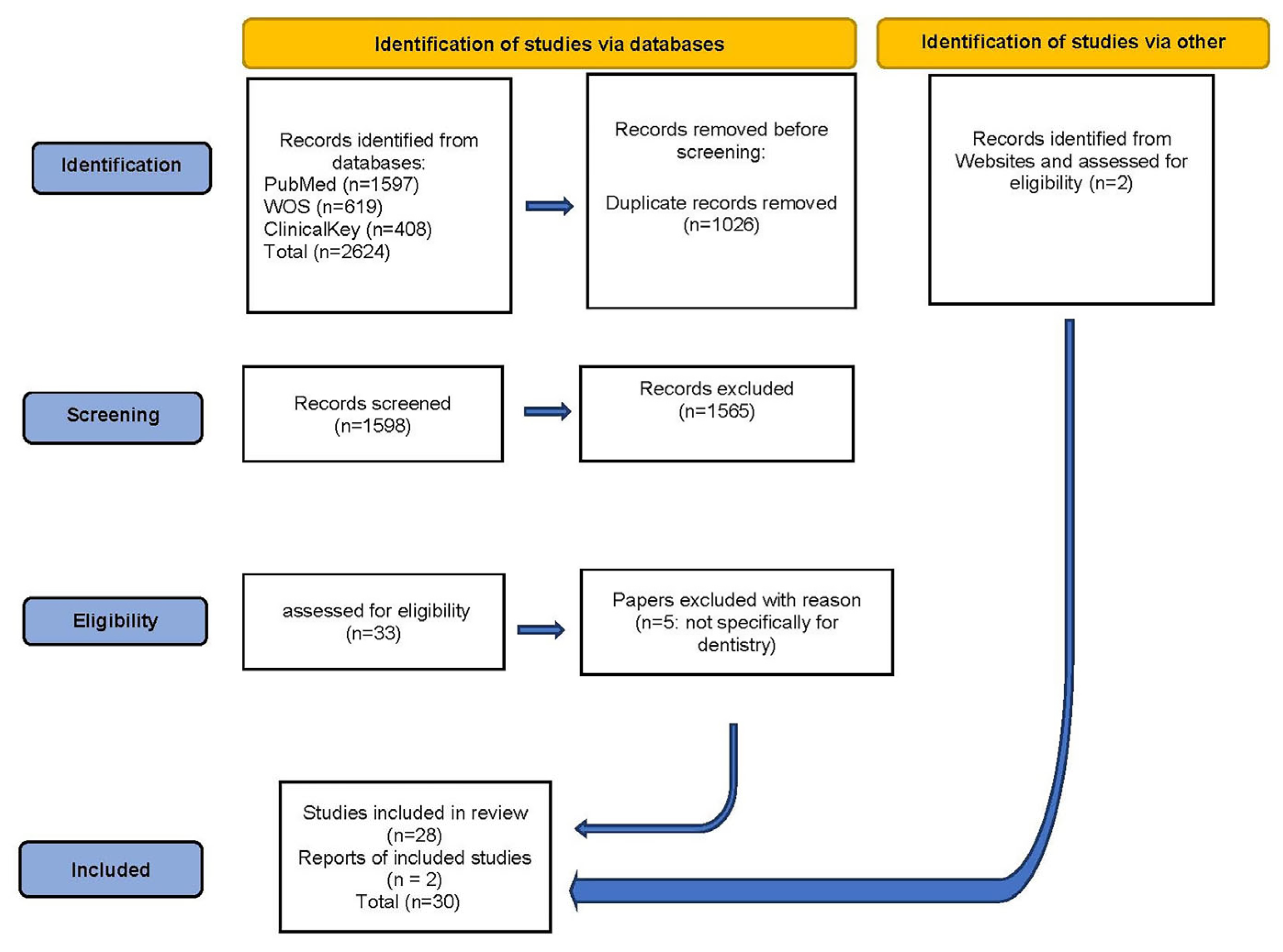
2. Materials and Methods
2.1. Eligibility Criteria
2.1.1. Inclusion Criteria
2.1.2. Exclusion Criteria
3. Results
4. Discussion
4.1. Informed Consent Beyond Disclosure: From Ethical Principles to Clinical Practice
4.2. Clinical Care Versus Research: A Persistent Ethical Fault Line
4.3. Emerging Consent Models: Promise Without Validation
4.4. Patient Understanding, Trust, and the Dentist–Patient–AI Relationship
4.5. AI-Generated Consent Documents: A Recursive Ethical Challenge
4.6. Implications for Dental Practice and Policy
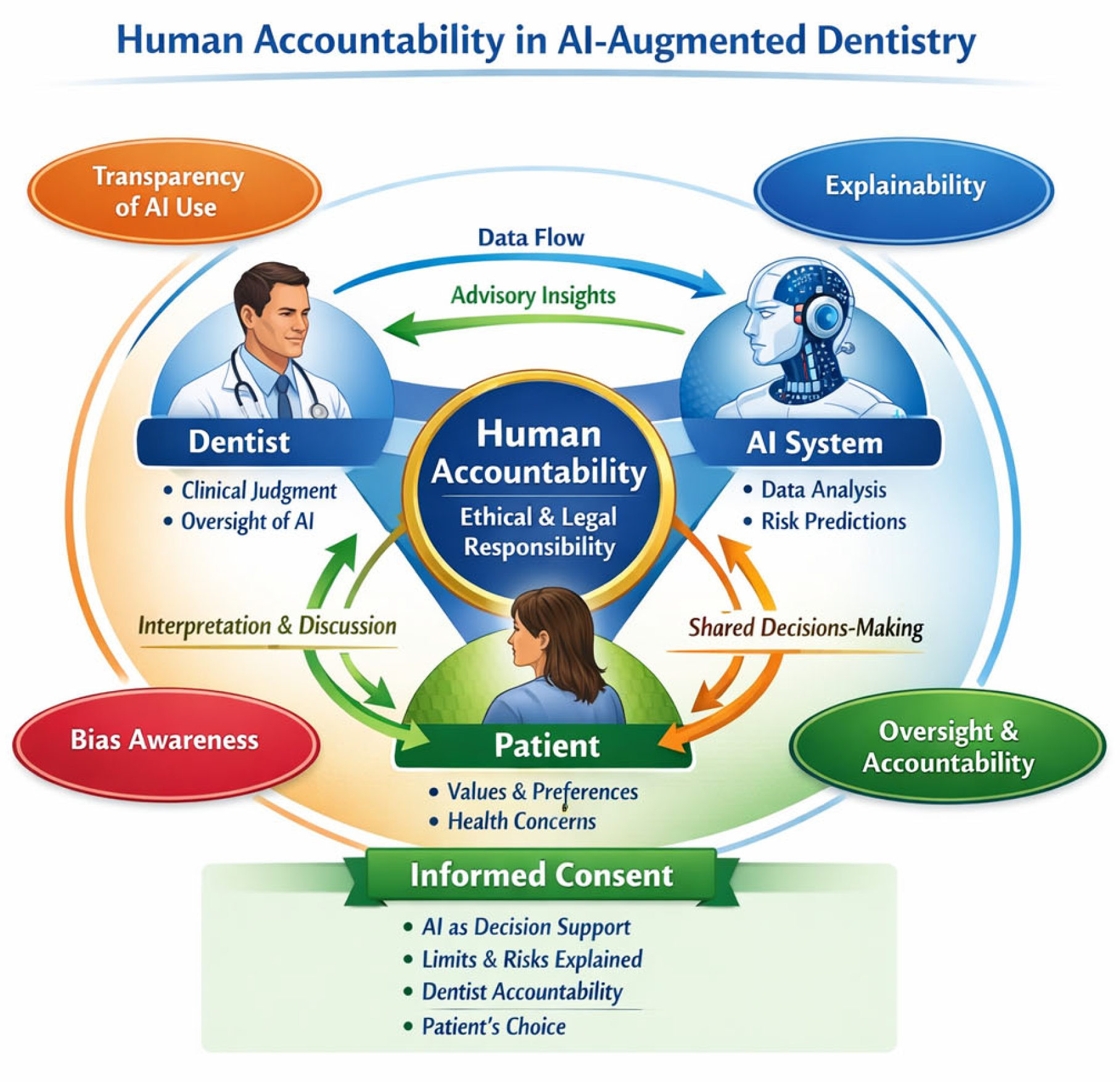
4.7. Human Accountability in AI-Augmented Dental Care: Implications for Informed Consent
4.8. Review Limitations
4.9. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AI | Artificial intelligence |
| CDS | Clinical decision support |
| NLP | Natural language processing |
| CBCT | Cone Beam Computed Tomography |
| LLM | Large language models |
| EHR | Electronic health record |
| mHealth | mobile health |
| OSF | Open Science Framework |
| PCC | Population–Concept–Context |
| GDPR | General Data Protection Regulation |
| EU | European Union |
| FDI | World Dental Federation |
| WHO | World Health Organization |
| IRB | Institutional review board |
References
- La Rosa, S.; Quinzi, V.; Palazzo, G.; Ronsivalle, V.; Lo Giudice, A. The Implications of Artificial Intelligence in Pedodontics: A Scoping Review of Evidence-Based Literature. Healthcare (Switzerland) 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Khattak, O.; Hashem, A.S.; Alqarni, M.S.; Almufarrij, R.A.S.; Siddiqui, A.Y.; Anis, R.; Ahmad, S.; Fareed, M.A.; Alothmani, O.S.; Alkhershawy, L.H.S.; et al. Deep Learning Applications in Dental Image-Based Diagnostics: A Systematic Review. Healthcare 2025, Vol. 13, Page 1466 2025, 13, 1466. [CrossRef]
- Schwendicke, F.; Rossi, J.G.; Göstemeyer, G.; Elhennawy, K.; Cantu, A.G.; Gaudin, R.; Chaurasia, A.; Gehrung, S.; Krois, J. Cost-Effectiveness of Artificial Intelligence for Proximal Caries Detection. Journal of dental research 2021, 100, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Blatz, M.; Uribe, S.; Cheung, W.; Verma, M.; Linton, J.; Kim, I. Artificial Intelligence for Dentistry - White Paper | FDI; 2023. [Google Scholar]
- Rokhshad, R.; Zhang, P.; Mohammad-Rahimi, H.; Shobeiri, P.; Schwendicke, F. Current Applications of Artificial Intelligence for Pediatric Dentistry: A Systematic Review and Meta-Analysis. Pediatric dentistry 2024, 46, 27–35. [Google Scholar] [PubMed]
- Elgarba, B.M.; Fontenele, R.C.; Tarce, M.; Jacobs, R. Artificial Intelligence Serving Pre-Surgical Digital Implant Planning: A Scoping Review. Journal of dentistry 2024, 143. [Google Scholar] [CrossRef]
- Macrì, M.; D’Albis, V.; D’Albis, G.; Forte, M.; Capodiferro, S.; Favia, G.; Alrashadah, A.O.; García, V.D.F.; Festa, F. The Role and Applications of Artificial Intelligence in Dental Implant Planning: A Systematic Review. Bioengineering 2024, Vol. 11 11, 778. [Google Scholar] [CrossRef]
- Revilla-León, M.; Gómez-Polo, M.; Vyas, S.; Barmak, B.A.; Galluci, G.O.; Att, W.; Krishnamurthy, V.R. Artificial Intelligence Applications in Implant Dentistry: A Systematic Review. The Journal of prosthetic dentistry 2023, 129, 293–300. [Google Scholar] [CrossRef]
- Lee, C.; Britto, S.; Diwan, K. Evaluating the Impact of Artificial Intelligence (AI) on Clinical Documentation Efficiency and Accuracy Across Clinical Settings: A Scoping Review. Cureus 2024, 16, e73994. [Google Scholar] [CrossRef]
- Lakhotia, S.; Godrej, H.; Kaur, A.; Nutakki, C.S.; Mun, M.; Eber, P.; Celi, L.A. Machine Learning in Dentistry: A Scoping Review. PLOS Digital Health 2025, 4, e0000940. [Google Scholar] [CrossRef]
- 11. Martinengo, L.; Lin, X.; Jabir, A.I.; Kowatsch, T.; Atun, R.; Car, J.; Car, L.T. Conversational Agents in Health Care: Expert Interviews to Inform the Definition, Classification, and Conceptual Framework. J Med Internet Res 2023;25:e50767 https://www.jmir.org/2023/1/e50767 2023, 25, e50767. [CrossRef]
- Liu, T.Y.; Lee, K.H.; Mukundan, A.; Karmakar, R.; Dhiman, H.; Wang, H.C. AI in Dentistry: Innovations, Ethical Considerations, and Integration Barriers. Bioengineering 2025, 12, 928. [Google Scholar] [CrossRef] [PubMed]
- Sciarra, F.M.; Caivano, G.; Cacioppo, A.; Messina, P.; Cumbo, E.M.; Di Vita, E.; Scardina, G.A. Dentistry in the Era of Artificial Intelligence: Medical Behavior and Clinical Responsibility. Prosthesis 2025, 7, 95. [Google Scholar] [CrossRef]
- Tay, F.R.; Loveless, R.; Ravenel, T.D. The Role of Artificial Intelligence in Shaping Dentistry through Advancement in Data Acquisition, Clinical Practice, Education, and Research. Dental Research 2026, 1, 100005. [Google Scholar] [CrossRef]
- Beauchamp, T.L.; Childress, J.F. Principles of Biomedical Ethics; Oxford University Press: USA, 2001; ISBN 0195143310. [Google Scholar]
- WHO Ethics and Governance of Artificial Intelligence for Health: Guidance on Large Multi-Modal Models; 2024.
- Federation, F.W.D. Artificial Intelligence in Dentistry. International Dental Journal 2025, 75, 3. [Google Scholar] [CrossRef]
- Yu, S.; Lee, S.-S.; Hwang, H. The Ethics of Using Artificial Intelligence in Medical Research. Kosin Medical Journal 2024, 39, 229–237. [Google Scholar] [CrossRef]
- Roganović J. Developing a Consent Checklist for AI in Dentistry: Thematic Analysis and Pilot Survey Validation. Digital Health 2025, 11, doi:10.1177/20552076251393227/ASSET/2DCE4F62-6DD3-4163-A6C0-66A91ECC75E7/ASSETS/IMAGES/LARGE/10.1177_20552076251393227-FIG2.JPGDeveloping a Consent Checklist for AI in Dentistry: Thematic Analysis and Pilot Survey Validation. Digital Health, 2025; 11. [CrossRef]
- Price, W.N.; Cohen, I.G. Privacy in the Age of Medical Big Data. Nature Medicine 2019 25:1 2019, 25, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Ong, J.C.L.; Chang, S.Y.H.; William, W.; Butte, A.J.; Shah, N.H.; Chew, L.S.T.; Liu, N.; Doshi-Velez, F.; Lu, W.; Savulescu, J.; et al. Ethical and Regulatory Challenges of Large Language Models in Medicine. The Lancet Digital Health 2024, 6, e428–e432. [Google Scholar] [CrossRef]
- Palaniappan, K.; Lin, E.Y.T.; Vogel, S.; Lim, J.C.W. Gaps in the Global Regulatory Frameworks for the Use of Artificial Intelligence (AI) in the Healthcare Services Sector and Key Recommendations. Healthcare 2024, 12, 1730. [Google Scholar] [CrossRef]
- Monteith, S.; Glenn, T.; Geddes, J.R.; Whybrow, P.C.; Achtyes, E.D.; Bauer, R.; Bauer, M. Artificial Intelligence and Deskilling in Medicine. The British Journal of Psychiatry 2026, 1–3. [Google Scholar] [CrossRef]
- Natali, C.; Marconi, L.; Dias Duran, L.D.; Cabitza, F. AI-Induced Deskilling in Medicine: A Mixed-Method Review and Research Agenda for Healthcare and Beyond. Artificial Intelligence Review 2025, 58, 356. [Google Scholar] [CrossRef]
- Wong, K.K.L.; Han, Y.; Cai, Y.; Ouyang, W.; Du, H.; Liu, C. From Trust in Automation to Trust in AI in Healthcare: A 30-Year Longitudinal Review and an Interdisciplinary Framework. Bioengineering 2025, 12, 1070. [Google Scholar] [CrossRef]
- Vaira, L.A.; Lechien, J.R.; Maniaci, A.; Tanda, G.; Abbate, V.; Allevi, F.; Arena, A.; Beltramini, G.A.; Bergonzani, M.; Bolzoni, A.R.; et al. Evaluating AI-Generated Informed Consent Documents in Oral Surgery: A Comparative Study of ChatGPT-4, Bard Gemini Advanced, and Human-Written Consents. Journal of Cranio-Maxillofacial Surgery 2025, 53, 18–23. [Google Scholar] [CrossRef]
- Petrou, E.; Ormond, K.E.; Stammbach, D.; Ash, E.; Buchholz, O.; Vayena, E. Evaluating GPT-4’s Ability to Generate Informed Consent Material for Genetic Testing. npj Artificial Intelligence 2025 1:1 2025, 1, 32. [Google Scholar] [CrossRef]
- Gaessler, J.; Remschmidt, B.; Jopp, A.K.; Arefnia, B.; Franke, A.; Rieder, M. Quality of Conventional versus Artificial Intelligence Oral Surgery Consent Forms: Comparative Analysis. J Med Internet Res 2026;28:e59851 https://www.jmir.org/2026/1/e59851 2026, 28, e59851. [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Annals of Internal Medicine 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Roganović, J. Consent for Artificial Intelligence in Dentistry. Journal of the American Dental Association 2025, 156, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Rokhshad, R.; Ducret, M.; Chaurasia, A.; Karteva, T.; Radenkovic, M.; Roganovic, J.; Hamdan, M.; Mohammad-Rahimi, H.; Krois, J.; Lahoud, P.; et al. Ethical Considerations on Artificial Intelligence in Dentistry: A Framework and Checklist. Journal of dentistry 2023, 135. [Google Scholar] [CrossRef] [PubMed]
- Rokhshad, R.; Karteva, T.; Chaurasia, A.; Richert, R.; Mörch, C.M.; Tamimi, F.; Ducret, M. Artificial Intelligence and Smile Design: An e-Delphi Consensus Statement of Ethical Challenges. Journal of Prosthodontics 2024, 33, 730–735. [Google Scholar] [CrossRef]
- Ducret, M.; Wahal, E.; Gruson, D.; Amrani, S.; Richert, R.; Mouncif-Moungache, M.; Schwendicke, F. Trustworthy Artificial Intelligence in Dentistry: Learnings from the EU AI Act. Journal of Dental Research 2024, 103, 1051–1056. [Google Scholar] [CrossRef]
- Weerakoon, A.T.; Girdis, T.; Peters, O. Artificial Intelligence in Australian Dental and General Healthcare: A Scoping Review. Australian dental journal 2025, 70, 209–256. [Google Scholar] [CrossRef]
- Shah, S.M.M. HARNESSING ELECTRONIC PATIENT RECORDS FOR AI INNOVATION: BALANCING DATA PRIVACY AND DIAGNOSTIC ADVANCEMENT. JOURNAL OF KHYBER COLLEGE OF DENTISTRY 2024, 14, 1–1. [Google Scholar] [CrossRef]
- Brinz, J.; Eslamiamirabadi, N.; Salamati, A.; Tresp, V.; Schwendicke, F.; Tichy, A. Data Sharing for Responsible Artificial Intelligence in Dentistry: A Narrative Review of Legal Frameworks and Privacy-Preserving Techniques. Journal of Dentistry 2025, 163, 106130. [Google Scholar] [CrossRef] [PubMed]
- Pethani, F. Promises and Perils of Artificial Intelligence in Dentistry. Australian dental journal 2021, 66, 124–135. [Google Scholar] [CrossRef]
- Roganović, J.; Radenković, M.; Miličić, B. Responsible Use of Artificial Intelligence in Dentistry: Survey on Dentists’ and Final-Year Undergraduates’ Perspectives. Healthcare (Switzerland) 2023, 11, 1480. [Google Scholar] [CrossRef]
- Navdeep Kaur, N.K.; Jacob, G.; Singh, A.; Khan, S.; Dhir, P.; Kakarla, G. Artificial Intelligence in Dentistry: Balancing Innovation with Ethical Responsibility. Bioinformation 2025, 21, 489. [Google Scholar] [CrossRef] [PubMed]
- Roganović, J.; Radenković, M.; Roganović, J.; Radenković, M. Ethical Use of Artificial Intelligence in Dentistry. In Ethics - Scientific Research, Ethical Issues, Artificial Intelligence and Education [Working Title]; 2023. [Google Scholar] [CrossRef]
- Darmadi, E.Y.; Fauziah, Y.A.; Alvin, J.D.; Mayfrila, A.A.; Cyntia, W. ETHICAL AND LEGAL ASPECTS OF ARTIFICIAL INTELLIGENCE IN ORAL HEALTH. HEARTY 2025, 13, 1101–1107. [Google Scholar] [CrossRef]
- Fahim, Y.A.; Hasani, I.W.; Kabba, S.; Ragab, W.M. Artificial Intelligence in Healthcare and Medicine: Clinical Applications, Therapeutic Advances, and Future Perspectives. European Journal of Medical Research 2025, 30, 848. [Google Scholar] [CrossRef]
- Ducret, M.; Wahal, E.; Gruson, D.; Amrani, S.; Richert, R.; Mouncif-Moungache, M.; Schwendicke, F. Trustworthy Artificial Intelligence in Dentistry: Learnings from the EU AI Act. J. Dent. Res. 2024, 103, 1051–1056, doi:10.1177/00220345241271160/ASSET/3BED355D-1C9A-46E3-8AC4-69F8E603EFE0/ASSETS/IMAGES/LARGE/10.1177_00220345241271160-FIG1.JPG.
- Budin-Ljøsne, I.; Teare, H.J.A.; Kaye, J.; Beck, S.; Bentzen, H.B.; Caenazzo, L.; Collett, C.; D’Abramo, F.; Felzmann, H.; Finlay, T.; et al. Dynamic Consent: A Potential Solution to Some of the Challenges of Modern Biomedical Research. BMC Medical Ethics 2017, 18, 4. [Google Scholar] [CrossRef]
- Allen, J.W.; Earp, B.D.; Koplin, J.; Wilkinson, D. Consent-GPT: Is It Ethical to Delegate Procedural Consent to Conversational AI? Journal of medical ethics 2024, 50, 77–83. [Google Scholar] [CrossRef] [PubMed]
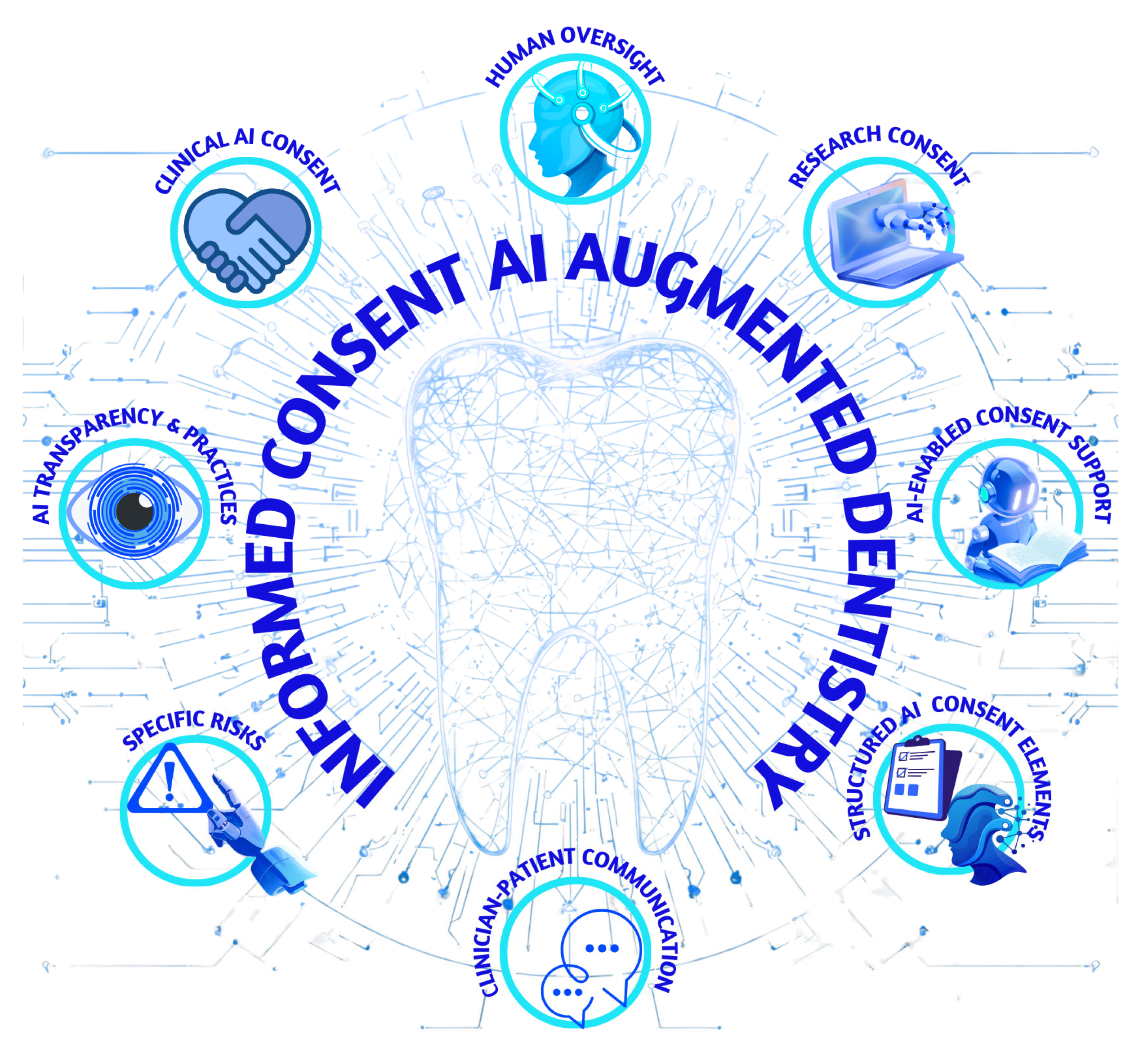
| Domain | Key Consent Element | What Must be Communicated to the Patient (Dentist / Oral Surgeon Perspective) | Clinical & Ethical Relevance | Key References |
|---|---|---|---|---|
| AI Disclosure Practices | Disclosure of AI involvement | Explicitly inform the patient when AI contributes to diagnosis, treatment planning, imaging interpretation, documentation, or decision support | Prevents hidden automation and supports informed decision-making | Roganović (2024) [19,30]; Rokhshad et al. (2023) [31]; Rokhshad et al. (2024) [32] |
| Nature of AI output | Explain whether AI outputs are deterministic or probabilistic, and whether the system is validated or experimental | Sets realistic expectations and mitigates overreliance | Roganović (2024) [30]; Rokhshad et al. (2023) [31] | |
| Documentation of disclosure | Record AI disclosure in the clinical file, including AI role and clinician review | Supports medico-legal traceability | Roganović (JADA 2024) | |
| Clinician Accountability & Oversight | Final responsibility | Clearly state that the dentist retains ultimate clinical responsibility for decisions | Reinforces professional accountability and legal clarity | Rokhshad et al. (2024) [32]; Ducret et al. (2024) [33] |
| Human oversight | Confirm that AI outputs are reviewed and may be overridden by the clinician | Prevents automation bias and unsafe delegation | Weerakoon et al. (2025) [34]; Ducret et al. (2024) [33] | |
| Clinician competence | Ensure the clinician understands AI system limits and performance | Ethical obligation to avoid misuse of AI | Rokhshad et al. (2024) [32] | |
| Clinical Care vs. Research Consent | Separate consent pathways | Distinguish consent for AI-assisted clinical care from consent for AI research | Prevents ethical conflation of care and research | Shah (2024) [35]; Brinz et al. (2025) [36]; Roganović (2024) [30] |
| Secondary data use | Explicit opt-in required for reuse of clinical data in AI training or validation | GDPR and research ethics compliance | Shah (2024) [35]; Brinz et al. (2025) [36] | |
| Data protection | Inform patients about anonymization, de-identification, and privacy safeguards | Addresses data governance concerns | Brinz et al. (2025) [36] | |
| AI-Specific Risks | Algorithmic bias | Explain that AI may perform differently across populations or clinical contexts | Supports fairness and risk awareness | Rokhshad et al. (2023) [31]; Ducret et al. ( 2024) [33] |
| Diagnostic errors | Disclose risks of false positives/negatives and model limitations | Aligns AI risks with conventional clinical risk disclosure | Pethani (2021) [37]; Rokhshad et al. (2023) [31] | |
| Explainability limits | Inform patients when AI decisions are not fully interpretable | Ethical transparency requirement | Ducret et al. (2024) [33] | |
| Right to refuse AI | Offer non-AI alternatives where feasible | Preserves patient autonomy | Rokhshad et al. (2023) [31] | |
| Consent Formats | Structured AI consent elements | Include AI role, benefits, risks, clinician oversight, and data use | Standardizes AI disclosure across dental practice | Rokhshad et al. (2023) [31]; Roganović (2024) [30] |
| Tiered / layered consent | Adapt depth of explanation to level of AI involvement and risk | Improves comprehension without overburdening patients | Rokhshad et al. (2024) [32]; Roganović (2024) [30] | |
| AI-specific acknowledgment | Use a separate checkbox or signature line for AI use | Makes AI consent explicit and auditable | Rokhshad et al. (2023) [31] | |
| Patient Understanding & Trust | Communication quality | Clinician explanation strongly influences patient trust in AI | Maintains therapeutic alliance | Roganović et al. (2023) [38]; Weerakoon et al. (2025) [34] |
| Clinician confidence | Dentist uncertainty about AI undermines patient understanding | Highlights need for professional training | Rokhshad et al. (2023) [31] | |
| Monitoring understanding | Assess patient comprehension during early implementation | Moves beyond formalistic consent | Weerakoon et al. (2025) [34] | |
| AI-Generated Consent Documents | Use of AI to draft consent | AI-generated consent may improve readability but lacks validation | Prevents uncritical reliance on AI-generated text | Shah (2024) [35]; Brinz et al. (2025) [36] |
| Mandatory human review | AI-drafted consent must be reviewed and approved by a clinician | Ensures ethical and legal accuracy | Brinz et al. (2025) [36] | |
| Data provenance | Protect patient data used in generating consent text | Prevents secondary misuse of sensitive data | Shah (2024) [35] |
| AI Context | AI Role | Consent Focus | Consent Requirements | Key References |
|---|---|---|---|---|
| Routine clinical care (low-risk AI) | Administrative support, image enhancement, scheduling | Transparency | General disclosure of AI use; no separate written consent required | Rokhshad et al. (2023) [31]; Ducret et al. (2024) [33] |
| Clinical decision support (moderate risk) | Diagnostic suggestions, treatment planning assistance | Autonomy & oversight | Explicit disclosure of AI role, limitations, and clinician responsibility; inclusion in written consent | Roganović (2024) [30]; Rokhshad et al. 2023 |
| High-impact clinical AI | AI significantly influences diagnosis or treatment decisions | Risk & accountability | Explicit, documented consent; explanation of AI uncertainty, bias, and alternatives; right to refuse AI | Roganović (2024) [30]; Ducret et al. (2024) [33] |
| Hybrid care–research AI | Deployed AI still undergoing validation or learning | Dual-purpose transparency | Disclosure of developmental status; separate explanation of care vs research functions | Roganović (2024) [30]; Brinz et al. (2025) [36] |
| AI-based research | Model training, validation, algorithm development | Research ethics | Separate research consent; purpose, data use, withdrawal rights, data sharing | Shah (2024) [35]; Brinz et al. (2025) [36] |
| Secondary data use | Retrospective data reuse for AI improvement | Data governance | Explicit opt-in consent; explanation of anonymization and sharing | Brinz et al. (2025) [36]; Roganović (2024) [30] |
| Dynamic / learning AI systems | Continuous model updating | Ongoing autonomy | Tiered or dynamic consent; possibility to modify preferences over time | Roganović (2024) [30]; Rokhshad et al. (2024) [32] |
| AI-generated consent tools | AI assists consent drafting or explanation | Meta-consent | Mandatory human review; disclosure of AI-generated content | Shah (2024) [35]; Brinz et al. (2025) [36] |
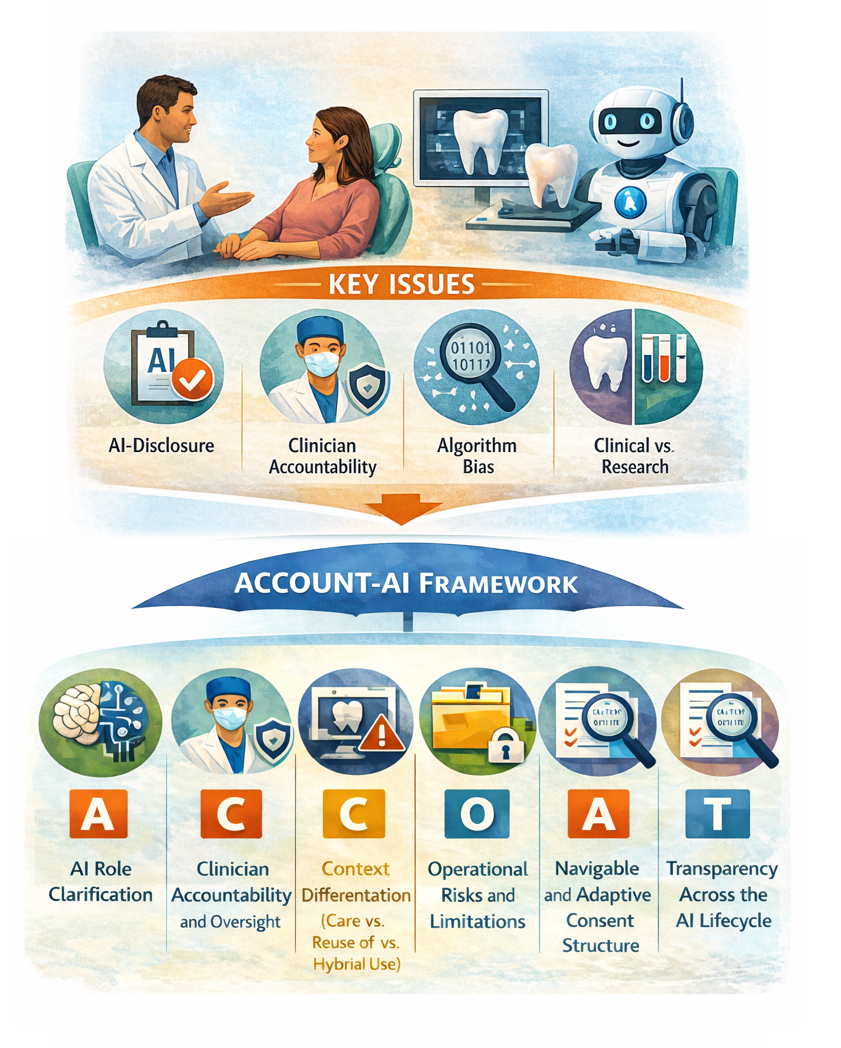
| ACCOUNT-AI Framework | Framework Domain | Required Patient Disclosure and Consent Elements | Purpose and Ethical Justification |
|---|---|---|---|
| A | AI Role Clarification (Functional Transparency) |
Patients must be clearly informed:
|
This domain addresses hidden automation and prevents algorithmic opacity in clinical reasoning. |
| C | Clinician Accountability and Oversight | Consent must explicitly state that:
|
This reinforces that AI functions as a decision-support instrument, not an autonomous agent. |
| C | Context Differentiation (Care vs. Research vs. Hybrid Use) | Consent must clearly distinguish between:
|
Separate or tiered consent pathways are recommended to avoid conflating treatment with experimentation. |
| O | Operational Risks and Limitations | Patients should be informed of AI-specific risks, including:
|
This aligns AI disclosure with traditional risk–benefit communication in dentistry. |
| U | Use and Reuse of Data (Secondary Data Governance) | Consent should clarify:
|
Reuse of high-quality clinical data contributes to improving AI accuracy, robustness, bias mitigation, and clinical safety by enabling model validation, recalibration, and population representativeness. |
| N | Navigable and Adaptive Consent Structure | Consent should be structured using:
|
This ensures proportionality between AI impact and disclosure burden. |
| T | Transparency Across the AI Lifecycle | Structurally integrate:
|
This domain consolidates the triadic dentist–patient–AI model by ensuring that AI systems operate within defined accountability boundaries. Transparency across the lifecycle transforms data reuse into an ethically governed feedback loop that enhances system reliability while preserving patient autonomy and trust. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




