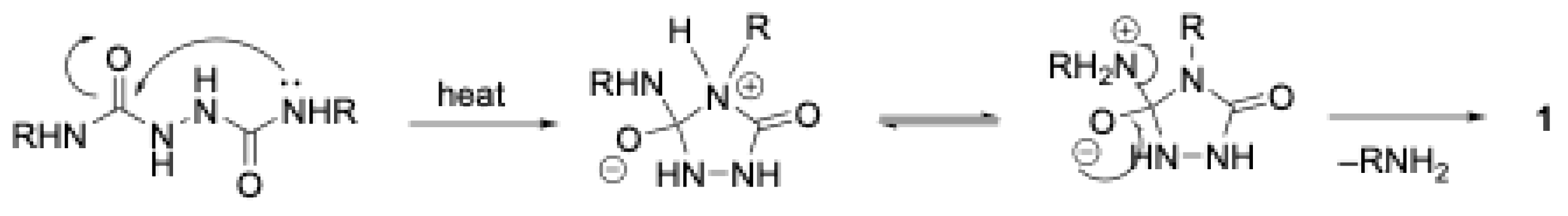
2.2. Experimental Procedures
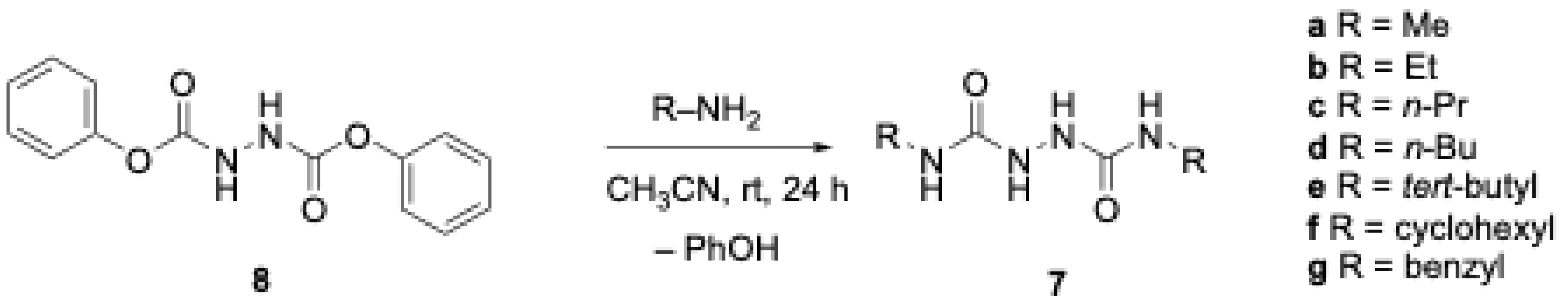
2.2.1 N,N-Diphenyl-1,2-hydrazinedicarboxamide (8). To a stirring mixture of 1.05 g (12.6 mmol) of hydrazine hydrate (60% by weight) and 2.76 g (2 equiv) of sodium carbonate in 125 mL of THF at 0 °C was added 4 g (25.5 mmol) of phenyl chloroformate dropwise via Pasteur pipette. The mixture was allowed to warm to room temperature and stirred overnight after which a white precipitate appeared. 50 mL of 0.5 M aq. HCl was added to the mixture and the THF removed via rotary evaporation to leave the white solid suspended in the aqueous layer. The mixture was filtered and the separated solid rinsed well with 100 mL of water. After air drying, the product was dried in the oven at 90 °C for at least two hours after which 84.7 g (96%) of
8 was isolated as a fluffy white solid, m.p. 153-154 °C:
1H NMR (DMSO-d
6) δ 9.44-10.10 (multiple s, 2H,
NH), 7.42 (t,
J = 7.8 Hz, 4H), 7.25 (t,
J = 7.8 Hz, 2H), 7.13 (d,
J = 7.8 Hz, 4H);
13C NMR (DMSO-d
6) δ 154.8, 150.5, 129.6, 125.6, 121.6. The spectra were consistent with those reported in the literature [
11].
2.2.3 1,6-Dimethylbisurea (7a). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 15 mL of CH3CN was added 5.50 mL (6 eq) of a 2 M solution of methyl amine in THF via syringe. A precipitate began to form some minutes later and stirring was continued overnight. The resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.25 g (94% yield) of 7a as a white powder, m.p. 243-244 °C: IR (ATR) cm-1 3311.6, 1662.0, 1561.1, 1413.9, 1325.6,; 1H NMR (CF3CO2D) δ 11.82 (br s, 4H, NH), 2.99 (s, 6H); 13C NMR (CF3CO2D) δ 164.2, 28.1; HRMS (ESI) m/z [M+H]+ Calcd for C4H11N4O2 147.08765; Found 147.08695.
2.2.4 1,6-Diethylbisurea (7b). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 15 mL of CH3CN was added 5.50 mL (6 eq) of a 2 M solution of ethyl amine in THF via syringe. A precipitate began to form some minutes later and stirring was continued overnight. The resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.28 g (88% yield) of 7b as a white powder, m.p. 238-239 °C: IR (ATR) cm-1 3297.7, 1665.0, 1554.2, 1328.6,; 1H NMR (CF3CO2D) δ 11.73 (br s, 4H, NH), 3.47 (q, J = 7.7 Hz, 4H), 1.28 (t, J = 7.7 Hz, 6H); 13C NMR (CF3CO2D) δ 160.3, 34.9, 11.7; HRMS (ESI) m/z [M+H]+ Calcd for C6H15N4O2 175.11895; Found 175.11874.
2.2.5 1,6-Di-n-propylbisurea (7c). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 20 mL of CH3CN was added 0.64 g (6 eq) of n-propyl amine dropwise via Pasteur pipette. The solution became cloudy after approximately 10 mins, and stirring was continued overnight. The resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.32 g (87% yield) of 7c as a white powder, m.p. 243-244 °C: IR (ATR) cm-1 3290.0, 2961.8, 1658.5, 1560.2, 1335.3; 1H NMR (CF3CO2D) δ 11.72 (br s, 4H, NH), 3.39 (t, J = 7.3 Hz, 4H), 1.68 (h, J = 7.3 Hz, 4H), 0.99 (t, J = 7.3 Hz, 6H); 13C NMR (CF3CO2D) δ 164.1, 44.8, 24.1, 11.5; HRMS (ESI) m/z [M+H]+ Calcd for C8H19N4O2 203.15025; Found 203.15006.
2.2.6 1,6-Di-n-butylbisurea (7d). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 20 mL of CH3CN was added 0.80 g (6 eq) of n-butyl amine dropwise via Pasteur pipette. The solution became cloudy after approximately 10 mins, and stirring was continued overnight. The resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.38 g (90% yield) of 7d as a white powder, m.p. 246-247 °C: IR (ATR) cm-1 3295.0, 2956.6, 1659.6, 1555.3, 1377.0,; 1H NMR (CF3CO2D) δ 11.82 (br s, 4H, NH), 3.43 (t, J = 7.3 Hz, 4H), 1.63 (p, J = 7.3 Hz, 4H), 1.42 (h, J = 7.3 Hz, 4H), 0.98 (t, J = 7.3 Hz, 6H); 13C NMR (CF3CO2D) δ 163.5, 42.8, 32.9, 21.3, 14.0; HRMS (ESI) m/z [M+H]+ Calcd for C10H23N4O2 231.18155; Found 231.18157.
2.2.7 1,6-Di-tert-butylbisurea (7e). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 20 mL of CH3CN was added 0.80 g (6 eq) of tert-butyl amine dropwise via Pasteur pipette. After stirring overnight, the solution was concentrated via rotary evaporation to afford a pale orange solid. The solid was taken up in 20 mL of CH2Cl2 and washed 2 x 20 mL 0.5 M aq. NaOH. The organic layer was then dried over Na2SO4, filtered, and concentrated to afford 0.24 g (57% yield) of 7e as a white solid, m.p. 189-190 °C: IR (ATR) cm-1 3314.7, 2968.1, 1654.9, 1558.4, 1363.3, 1217.6; 1H NMR (CDCl3) δ 6.88 (br s, 2H, NH), 5.62 (br s, 2H, NH), 1.34 (s, 9H); 13C NMR (CDCl3) δ 158.1, 50.6, 29.1; HRMS (ESI) m/z [M+H]+ Calcd for C10H23N4O2 231.18155; Found 231.18111.
2.2.8 1,6-Dicyclohexylbisurea (7g). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 20 mL of CH3CN was added 1.09 g (6 eq) of cyclohexyl amine dropwise via Pasteur pipette. After stirring overnight, the resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.45 g (77% yield) of 7g as a white powder, m.p. 203-204 °C: IR (ATR) cm-1 3675.5, 2972.0, 1646.4, 1531.5, 1393.9, 1066.0; 1H NMR (CF3CO2D) δ 11.69 (br s, 4H, NH), 3.77 (m, 2H), 1.97 (br s, 4H), 1.87 (br s, 4H), 1.72 (br d, J = 12.4 Hz, 2H), 1.35-1.50 (m, 8H), 1.26 (m, 2H); 13C NMR (CF3CO2D) δ 162.6, 53.6, 34.6, 26.6, 26.5; HRMS (ESI) m/z [M+H]+ Calcd for C14H27N4O2 283.21285; Found 283.21103.
2.2.9 1,6-Dibenzylbisurea (7g). To a stirring solution of 0.5 g (1.83 mmol) of compound 8 in 20 mL of CH3CN was added 1.17 g (6 eq) of benzyl amine dropwise via Pasteur pipette. After stirring overnight, the resulting white precipitate was isolated via vacuum filtration and washed with 10 mL of water. Air drying afforded 0.42 g (76% yield) of 7g as a white powder, m.p. 245-246 °C: IR (ATR) cm-1 3292.2, 1660.3, 1552.1, 1298.6, 1219.0; 1H NMR (CF3CO2D) δ 11.68 (br s, 4H, NH), 7.26-7.34 (m, 10H), 4.53 (s, 4H); 13C NMR (CF3CO2D) δ 163.6, 138.1, 130.89, 130.3, 129.5, 46.6; HRMS (ESI) m/z [M+H]+ Calcd for C16H19N4O2 299.15025; Found 299.15007.
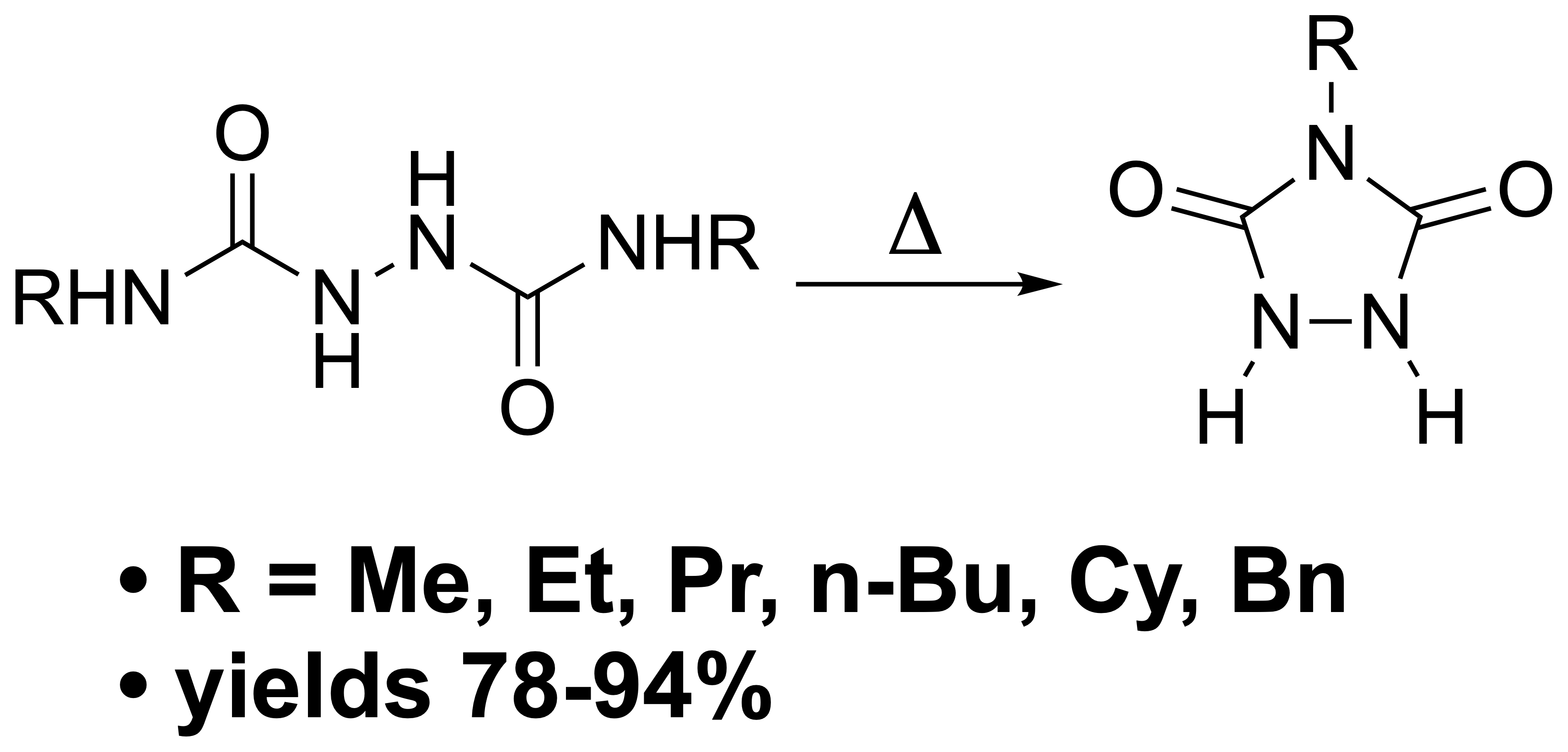
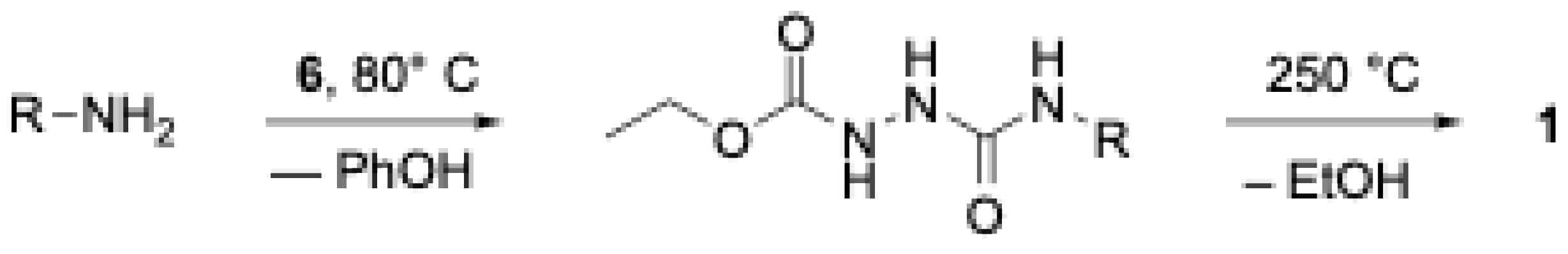
2.2.10 N-Methylurazole (1a). 100 mg (0.68 mmol) of finely powdered
7a was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 240 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated methyl amine to escape. After 10 mins, fuming had ceased, and the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 62 mg of
1a as a white crystalline product (80% yield), m.p. 237-238 °C (lit. 232-233 °C [
12]):
1H NMR (DMSO-
d6) δ 9.59 (br s, 2H,
NH), 2.84 (s, 3H);
13C NMR (DMSO-
d6) δ 155.3, 24.3. The spectra were consistent with those reported in the literature [
13].
Repeating this process on a larger scale starting with 0.50 g of 7a afforded 0.39 g (85% yield) of 1a.
2.2.11 N-Ethylurazole (1b). 100 mg (0.68 mmol) of finely powdered
7b was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 240 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated ethyl amine to escape. After 10 mins, fuming had ceased, and the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 68 mg of
1b as a white crystalline product (80% yield), m.p. 193-194 °C (lit. 195-196 °C [
12]):
1H NMR (DMSO-
d6) δ 9.90 (br s, 2H,
NH), 3.37 (q,
J = 7.3 ppm, 2H), 1.09 (t,
J = 7.3 ppm, 3H);
13C NMR (DMSO-
d6) δ 154.9, 32.9, 13.3. The spectra were consistent with those reported in the literature [
14].
2.2.12 N-Propylurazole (1c). 100 mg (0.68 mmol) of finely powdered
7c was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 240 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated propyl amine to escape. After 10 mins, fuming had ceased, and the temperature was dropped to 190 °C where it was held for 25 min, again with occasional swirling. After the heating period, the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 62 mg of
1c as a white crystalline product (89% yield), m.p. 166-167 °C (lit. 168-169 °C [
12]):
1H NMR (DMSO-
d6) δ 9.90 (br s, 2H,
NH), 3.30 (t,
J = 7.4 ppm, 2H), 1.52 (h,
J = 7.4 ppm, 2H), 0.81 (t,
J = 7.5 ppm, 3H);
13C NMR (DMSO-
d6) δ 155.2, 39.51, 21.0, 11.0. The spectra were consistent with those reported in the literature [
15].
2.2.13 N-Butylurazole (1d). 100 mg (0.68 mmol) of finely powdered
7d was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 240 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated butyl amine to escape. After 15 mins, fuming had ceased, and the temperature was dropped to 190 °C where it was held for 25 min, again with occasional swirling. After the heating period, the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 53 mg of
1d as a white crystalline product (78% yield), m.p. 170-171 °C (lit. 167-168 °C [
12]):
1H NMR (DMSO-
d6) δ 10.00 (br s, 2H,
NH), 3.34 (t,
J = 7.4 ppm, 2H), 1.50 (p,
J = 7.4 ppm, 2H), 1.04 (h,
J = 7.4 ppm, 2H ), 0.87 (t,
J = 7.5 ppm, 3H);
13C NMR (DMSO-
d6) δ 155.2, 37.6, 29.6, 19.4, 13.5. The spectra were consistent with those reported in the literature [
13].
Repeating this process on a larger scale starting with 0.95 g of 7d afforded 0.49 g (75% yield) of 1d.
2.2.14 N-Cyclohexylurazole (1f). 100 mg (0.68 mmol) of finely powdered
7f was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 250 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated butyl amine to escape. After 30 min, fuming had ceased, and the temperature was dropped to 190 °C where it was held for 25 min where it solidified. After the heating period, the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 55 mg of
1f as a white crystalline product (85% yield), m.p. 242-243 °C (lit. 239-241 °C [
16]):
1H NMR (DMSO-
d6) δ 9.95 (br s, 2H,
NH), 3.68 (dt,
J = 12.4, 3.9 Hz, 1H), 2.02 (dg,
J = 12.4, 3.9 Hz, 1H), 1.76 (br d, 2H), 1.55-1.65 (m, 3H), 1.25 (qt,
J = 12.4, 3.3 Hz, 1H), 1.12 (dt,
J = 12.4, 3.0 Hz, 1H);
13C NMR (DMSO-
d6) δ 155.3, 50.6, 29.5, 25.8, 25.3. The spectra were consistent with those reported in the literature [
16].
2.2.15 N-Benzylurazole (1g). 100 mg (0.68 mmol) of finely powdered
7g was heated in a vial to 260 °C over approximately a 10 min period. Visible fuming began to appear at a temperature of ~ 250 °C. As the sample was held at 260 °C it melted to provide a colorless oil. The melt was held at this temperature with occasional swirling to allow liberated butyl amine to escape. After 1 hr, fuming had ceased, and the temperature dropped to 190 °C where it was held for 25 min, again with occasional swirling. After the heating period, the vial was removed from the heat with swirling to aid in crystallization of the urazole product with cooling. The product was scraped from the vial to afford 60 mg of
1g as a white crystalline product (94% yield), m.p. 181-182 °C (lit. 182-183 °C [
12]):
1H NMR (DMSO-
d6) δ 10.21 (br s, 2H,
NH), 7.26-7.35 (m, 5H), 4.54 (s, 2H);
13C NMR (DMSO-
d6) δ 154.8, 136.8, 128.6, 127.6, 127.5, 41.3. The spectra were consistent with those reported in the literature [
13].