Submitted:
19 March 2026
Posted:
20 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
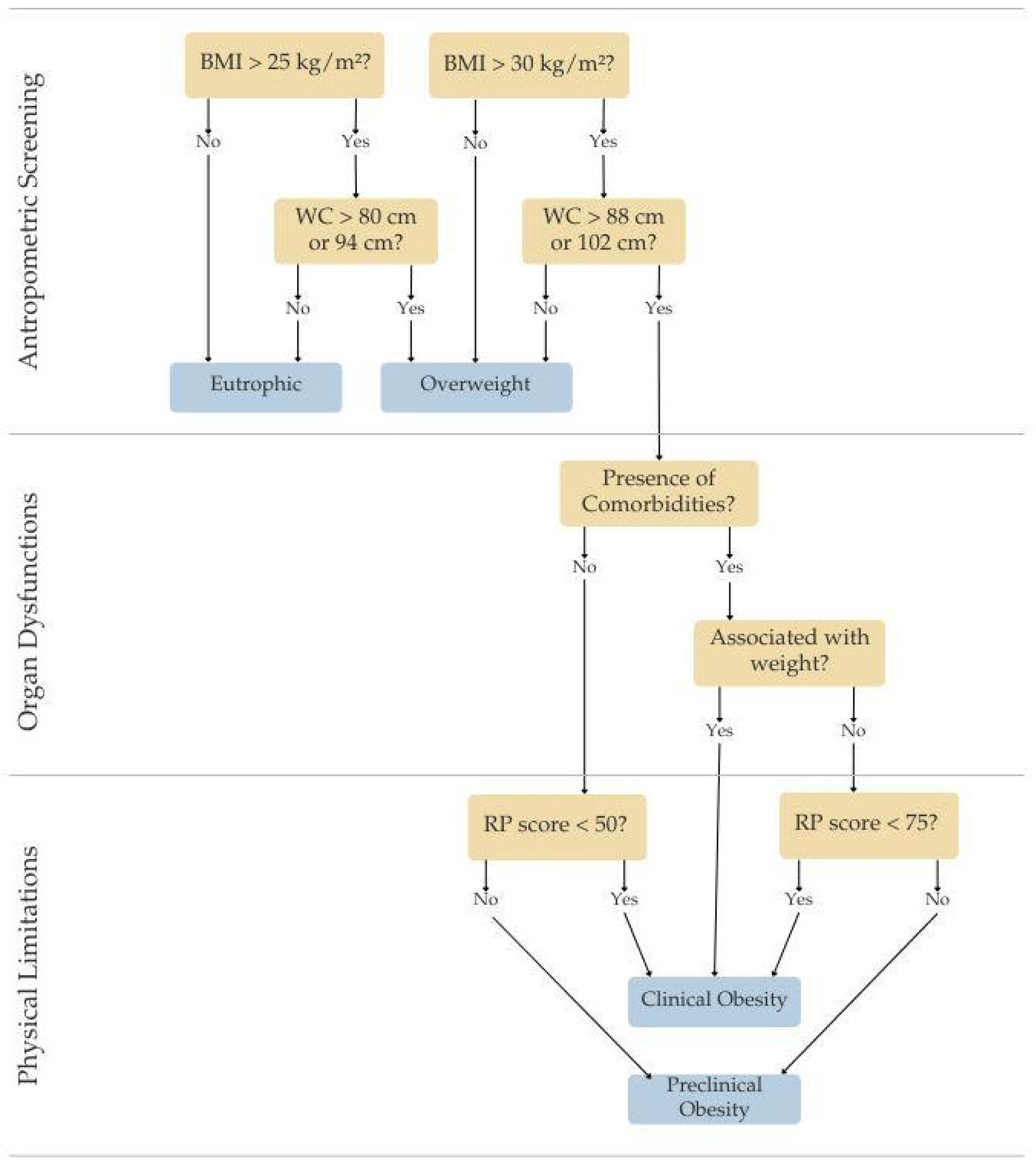
2. Patients, Materials and Methods
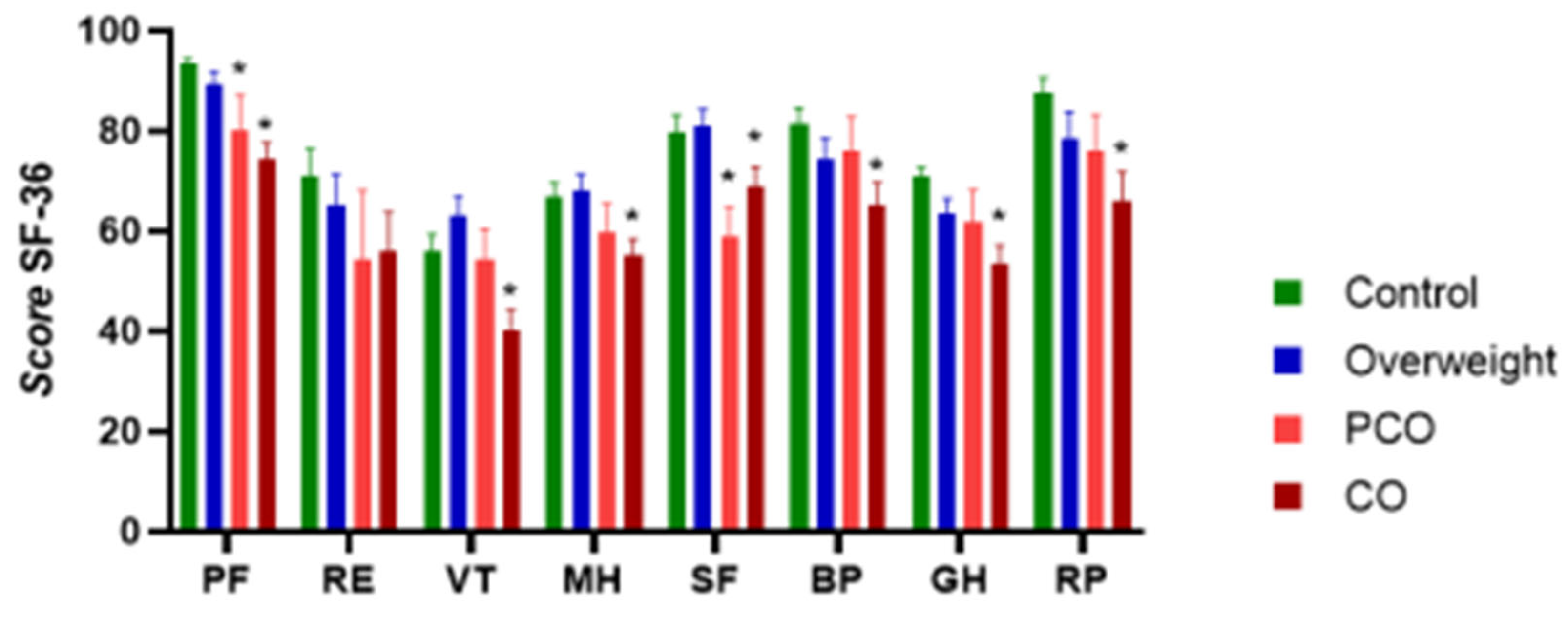
3. Results and Discussion
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Consultation on Obesity (1999: Geneva S.; Organization W.H. Obesity :preventing and managing the global epidemic : report of a WHO consultation; World Health Organization, 2000; ISBN 978-92-4-120894-9.
- Sweatt, K.; Garvey, W.T.; Martins, C. Correction: Strengths and Limitations of BMI in the Diagnosis of Obesity: What Is the Path Forward? Curr Obes Rep 2024, 13, 831. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.; Cummings, D.E.; Eckel, R.H.; Cohen, R.V.; Wilding, J.P.H.; Brown, W.A.; Stanford, F.C.; Batterham, R.L.; Farooqi, I.S.; Farpour-Lambert, N.J.; et al. Definition Diagnostic Criteria of Clinical Obesity. The Lancet Diabetes & Endocrinology 2025, 13, 221–262. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E.; Sherbourne, C.D. The MOS 36-Item Short-Form Health Survey (SF-36). Conceptual Framework and Item Selection. Med Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Lins, L.; Carvalho, F.M. SF-36 Total Score as a Single Measure of Health-Related Quality of Life: Scoping Review. SAGE Open Med 2016, 4, 2050312116671725. [Google Scholar] [CrossRef] [PubMed]
- Ciconelli, R.M.; Ferraz, M.B.; Santos, W.; Meinão, I.; Quaresma, M.R. Tradução para a língua portuguesa e validação do questionário genérico de avaliação de qualidade de vida SF-36 (Brasil SF-36). Rev. bras. reumatol 1999, 143–150. [Google Scholar]
- Laguardia, J.; Campos, M.R.; Travassos, C.; Najar, A.L.; dos Anjos, L.A.; Vasconcellos, M.M. Brazilian Normative Data for the Short Form 36 Questionnaire, Version 2. Rev. bras. epidemiol. 2013, 16, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Tamura, L.S.; Cazzo, E.; Chaim, E.A.; Piedade, S.R. Influence of Morbid Obesity on Physical Capacity, Knee-Related Symptoms and Overall Quality of Life: A Cross-Sectional Study. Rev Assoc Med Bras (1992) 2017, 63, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, T.-I.; Tveitå, E.K.; Natvig, G.K.; Tonstad, S.; Hjelmesæth, J. Validity of the SF-36 in Patients with Morbid Obesity. Obes Facts 2011, 4, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Kolotkin, R.L.; Meter, K.; Williams, G.R. Quality of Life and Obesity. Obes Rev. 2001, 2, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, K.R.; Barofsky, I. Obesity and Health-Related Quality of Life. Obes Rev 2001, 2, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Sarwer, D.B.; Polonsky, H.M. The Psychosocial Burden of Obesity. Endocrinol Metab Clin North Am 2016, 45, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Adorno, M.L.G.R.; Brasil-Neto, J.P. Avaliação da qualidade de vida com o instrumento SF-36 em lombalgia crônica. Acta ortop. bras. 2013, 21, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Tylutka, A.; Walas, Ł.; Zembron-Lacny, A. Level of IL-6, TNF, and IL-1β and Age-Related Diseases: A Systematic Review and Meta-Analysis. Front. Immunol. 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Ion, R.-M.; Sibianu, M.; Hutanu, A.; Beresescu, F.G.; Sala, D.T.; Flavius, M.; Rosca, A.; Constantin, C.; Scurtu, A.; Moriczi, R.; et al. A Comprehensive Summary of the Current Understanding of the Relationship between Severe Obesity, Metabolic Syndrome, and Inflammatory Status. Journal of Clinical Medicine 2023. [Google Scholar] [CrossRef] [PubMed]
- Polak-Szczybyło, E.; Tabarkiewicz, J.; Polak-Szczybyło, E.; Tabarkiewicz, J. IL-17A, IL-17E and IL-17F as Potential Biomarkers for the Intensity of Low-Grade Inflammation and the Risk of Cardiovascular Diseases in Obese People. Nutrients 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. - References - Scientific Research Publishing. 2024. Available online: https://www.scirp.org/reference/referencespapers?referenceid=3887298 (accessed on December 2025).
- Venables, B.; Ripley, B. Modern Applied Statistics With S; Springer, 2002. [Google Scholar]
- Başhan, İ.; Bakman, M. The Comparison of Body Mass Index and Waist Circumference to Assess Health-Related Quality of Life. TJFMPC 2021, 15, 230–235. [Google Scholar] [CrossRef]
- Seo, D.-C.; Choe, S.; Torabi, M.R. Is Waist Circumference ≥ 102/88 Cm Better than Body Mass Index ≥ 30 to Predict Hypertension and Diabetes Development Regardless of Gender, Age Group, and Race/Ethnicity? Meta-Analysis. Preventive Medicine 2017, 97, 100–108. [Google Scholar] [CrossRef] [PubMed]
- The Performance of Anthropometric Tools to Determine Obesity: A Systematic Review and Meta-Analysis | Scientific Reports. Available online: https://www.nature.com/articles/s41598-020-69498-7 (accessed on 10 November 2025).
- Kern, L.; Mittenbühler, M.J.; Vesting, A.J.; Ostermann, A.L.; Wunderlich, C.M.; Wunderlich, F.T.; Kern, L.; Mittenbühler, M.J.; Vesting, A.J.; Ostermann, A.L.; et al. Obesity-Induced TNFα and IL-6 Signaling: The Missing Link between Obesity and Inflammation—Driven Liver and Colorectal Cancers. Cancers 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.H.; Park, H.S.; Kim, K.-S.; Choi, W.H.; Ahn, C.W.; Kim, B.T.; Kim, S.M.; Lee, S.Y.; Ahn, S.M.; Kim, Y.K.; et al. Effect of Weight Loss on Some Serum Cytokines in Human Obesity: Increase in IL-10 after Weight Loss. J Nutr Biochem 2008, 19, 371–375. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Question | Limit | Variation |
| Physical Functioning | 3 | 10 | 20 |
| Role-Physical | 4 | 4 | 4 |
| Role-Emotional | 5 | 3 | 3 |
| Vitality | 09 (items a, e, g, i) | 4 | 8 |
| Mental Health | 09 (items b, c, d, f, h) | 5 | 25 |
| Social Functioning | 06 and 10 | 2 | 8 |
| Bodily Pain | 07 and 08 | 2 | 10 |
| General Health | 01 and 11 | 5 | 20 |
|
Control (n=45) |
Overweight (n=27) |
PCO (n=11) |
CO (n=25) |
Overall (n=108) |
|
| Ethnicity | |||||
| African descent | 13 (28.9%) | 15 (55.6%) | 6 (54.5%) | 11 (44.0%) | 45 (41.7%) |
| Caucasian | 32 (71.1%) | 12 (44.4%) | 5 (45.5%) | 14 (56.0%) | 63 (58.3%) |
| Age - range (years) | |||||
| 19 to 29 | 32 (71.1%) | 9 (33.3%) | 7 (63.6%) | 7 (28.0%) | 55 (50.9%) |
| 30 to 39 | 6 (13.3%) | 10 (37.0%) | 0 (0%) | 9 (36.0%) | 25 (23.1%) |
| 40 to 49 | 4 (8.9%) | 2 (7.4%) | 1 (9.1%) | 4 (16.0%) | 11 (10.2%) |
| 50 to 59 | 2 (4.4%) | 5 (18.5%) | 3 (27.3%) | 5 (20.0%) | 15 (13.9%) |
| 60 to 65 | 1 (2.2%) | 1 (3.7%) | 0 (0%) | 0 (0%) | 2 (1.9%) |
| Variables | Simple Model | ||
| Beta | 95% CI | P | |
| RP | |||
| Overweight vs Control | -7.65 | -19.84, 4.55 | 0.22 |
| PCO vs Control | -11.67 | -28.52, 5.18 | 0.17 |
| CO vs Control | -19.83 | -32.32, -7.33 | <0.001 |
| PF | |||
| Overweight vs Control | -2.80 | -8.61, 3.01 | 0.34 |
| PCO vs Control | -8.54 | 16.57, -0.51 | 0.04 |
| CO vs Control | -17.13 | -23.09, -11.18 | <0.001 |
| RE | |||
| Overweight vs Control | -8.18 | -26.75, 10.39 | 0.39 |
| PCO vs Control | -18.55 | -44.21, 7.11 | 0.16 |
| CO vs Control | -17.12 | -36.15, 1.91 | 0.08 |
| VT | |||
| Overweight vs Control | 5.69 | -4.72, 16.09 | 0.29 |
| PCO vs Control | -1.08 | -15.45, 13.30 | 0.88 |
| CO vs Control | -17.34 | -28.00, -6.68 | <0.001 |
| MH | |||
| Overweight vs Control | 0.62 | -8.54, 9.78 | 0.89 |
| PCO vs Control | -5.30 | -17.96, 7.36 | 0.42 |
| CO vs Control | -12.20 | -21.59, -2.81 | 0.01 |
| SF | |||
| Overweight vs Control | -1.39 | -9.65, 6.86 | 0.74 |
| PCO vs Control | -22.20 | -33.60, -10.80 | <0.001 |
| CO vs Control | -12.80 | -21.26, -4.35 | 0.00 |
| BP | |||
| Overweight vs Control | -7.18 | -16.88, 2.52 | 0.15 |
| PCO vs Control | -4.57 | -17.98, 8.83 | 0.51 |
| CO vs Control | -16.07 | -26.01, -6.13 | 0.00 |
| GH | |||
| Overweight vs Control | -6.59 | -14.08, 0.90 | 0.09 |
| PCO vs Control | -8.63 | -18.98, 1.72 | 0.12 |
| CO vs Control | -18.07 | -25.75, -10.40 | <0.001 |
| Variables | Simple Model | ||
| Beta | 95% CI | P | |
| TNF | |||
| Overweight vs Control | -0.02 | -0.04, 0.00 | 0.08 |
| PCO vs Control | -0.02 | -0.05, 0.01 | 0.24 |
| CO vs Control | 0.00 | -0.02, 0.02 | 0.75 |
| IL-6 | |||
| Overweight vs Control | 0.74 | -0.74, 2.23 | 0.29 |
| PCO vs Control | 0.63 | -1.53, 2.80 | 0.54 |
| CO vs Control | 18.59 | 17.04, 20.14 | <0.001 |
| IL-17 | |||
| Overweight vs Control | -0.00 | -0.00, 0.00 | 0.72 |
| PCO vs Control | 0.00 | 0.00, 0.00 | 0.01 |
| CO vs Control | 2.58 | 2.58, 2.58 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




