Submitted:
19 March 2026
Posted:
20 March 2026
You are already at the latest version
Abstract
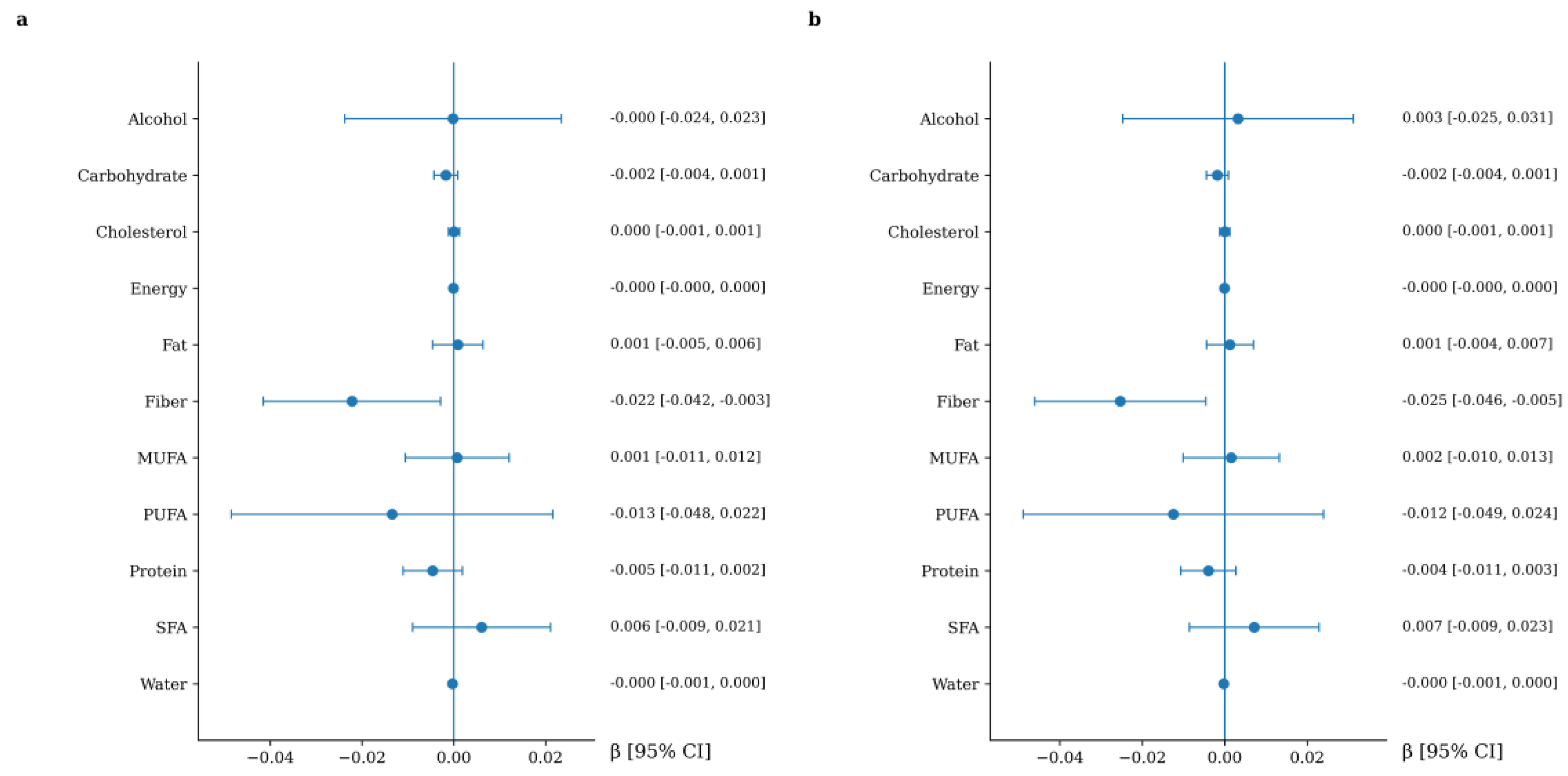
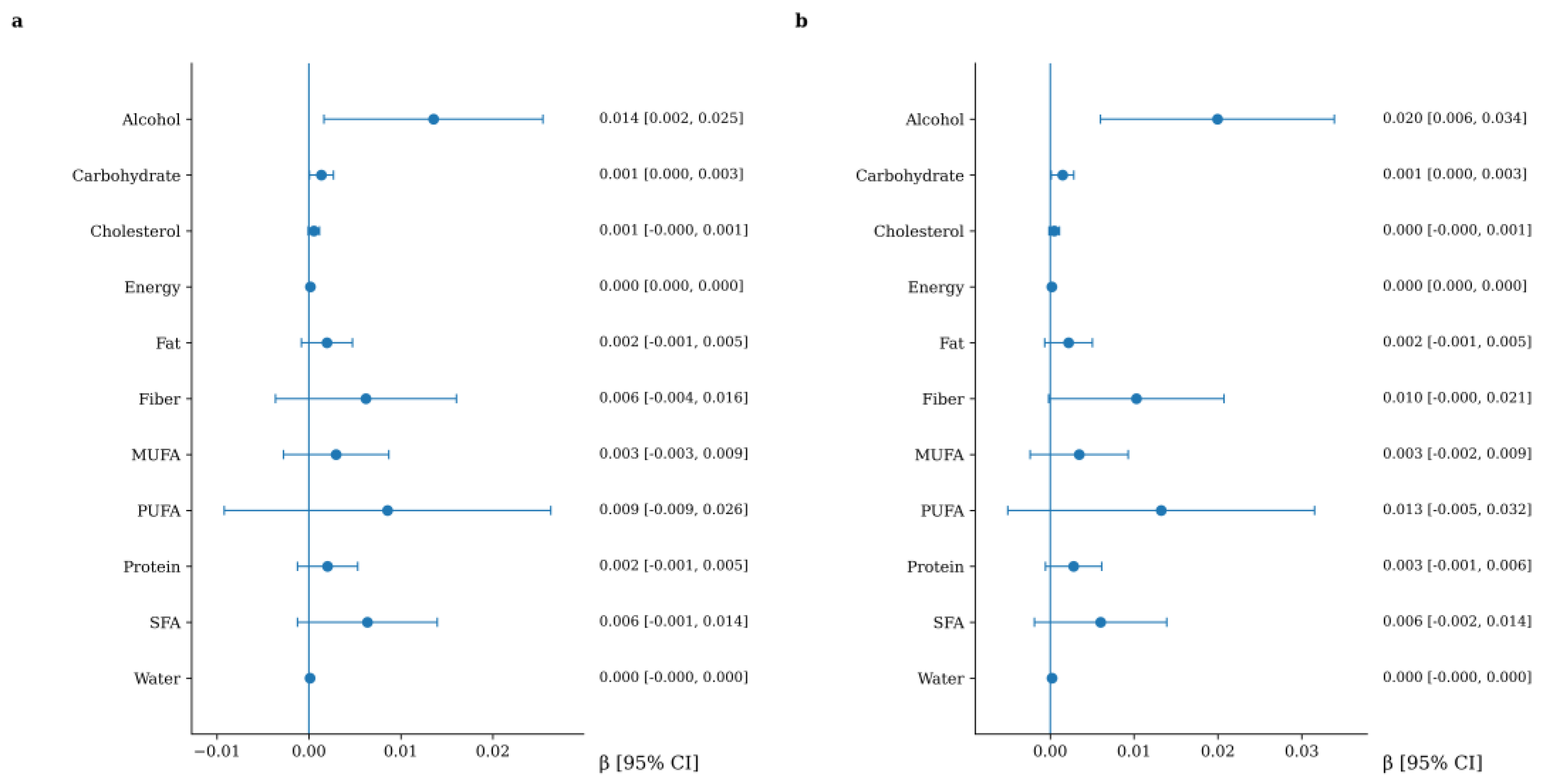
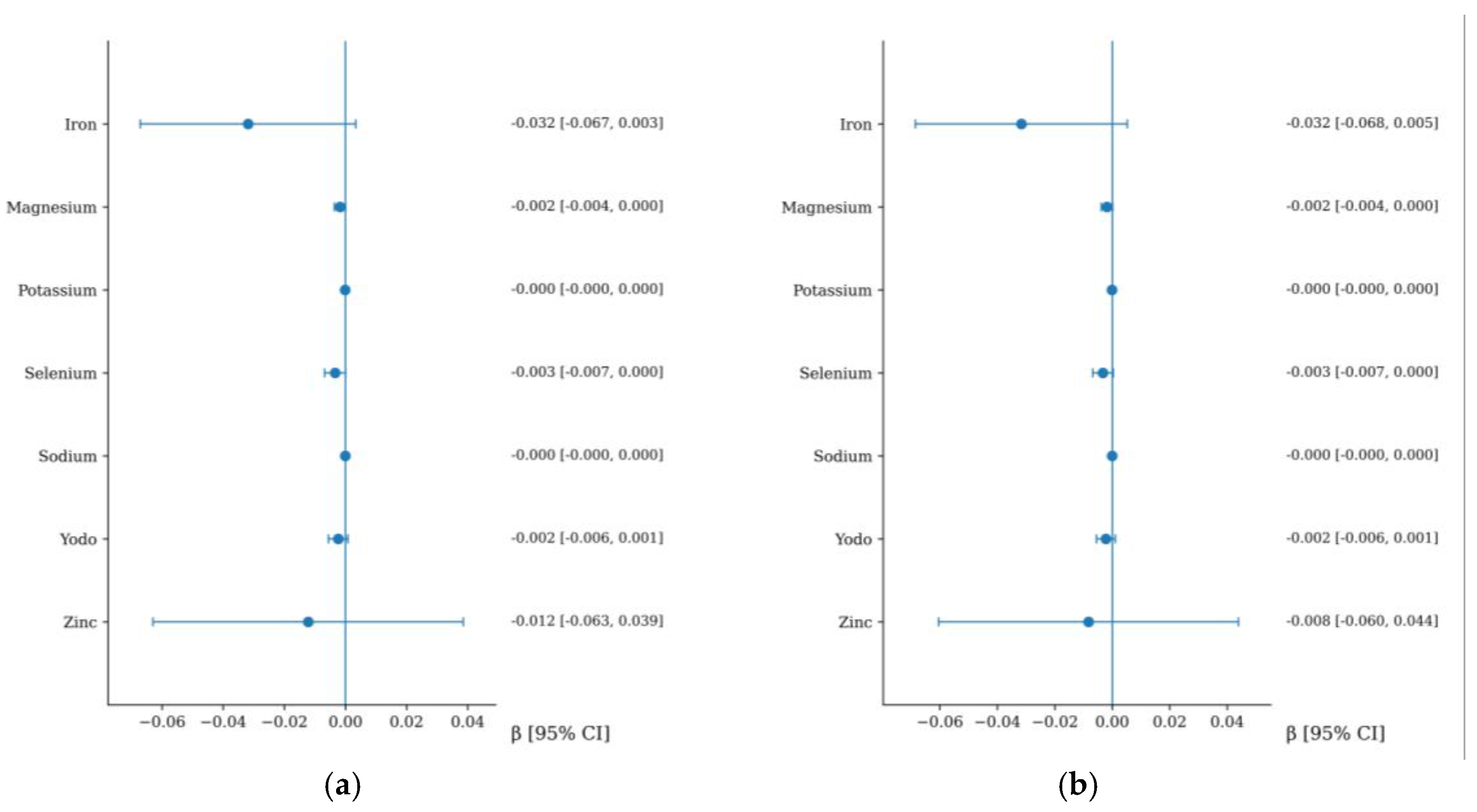
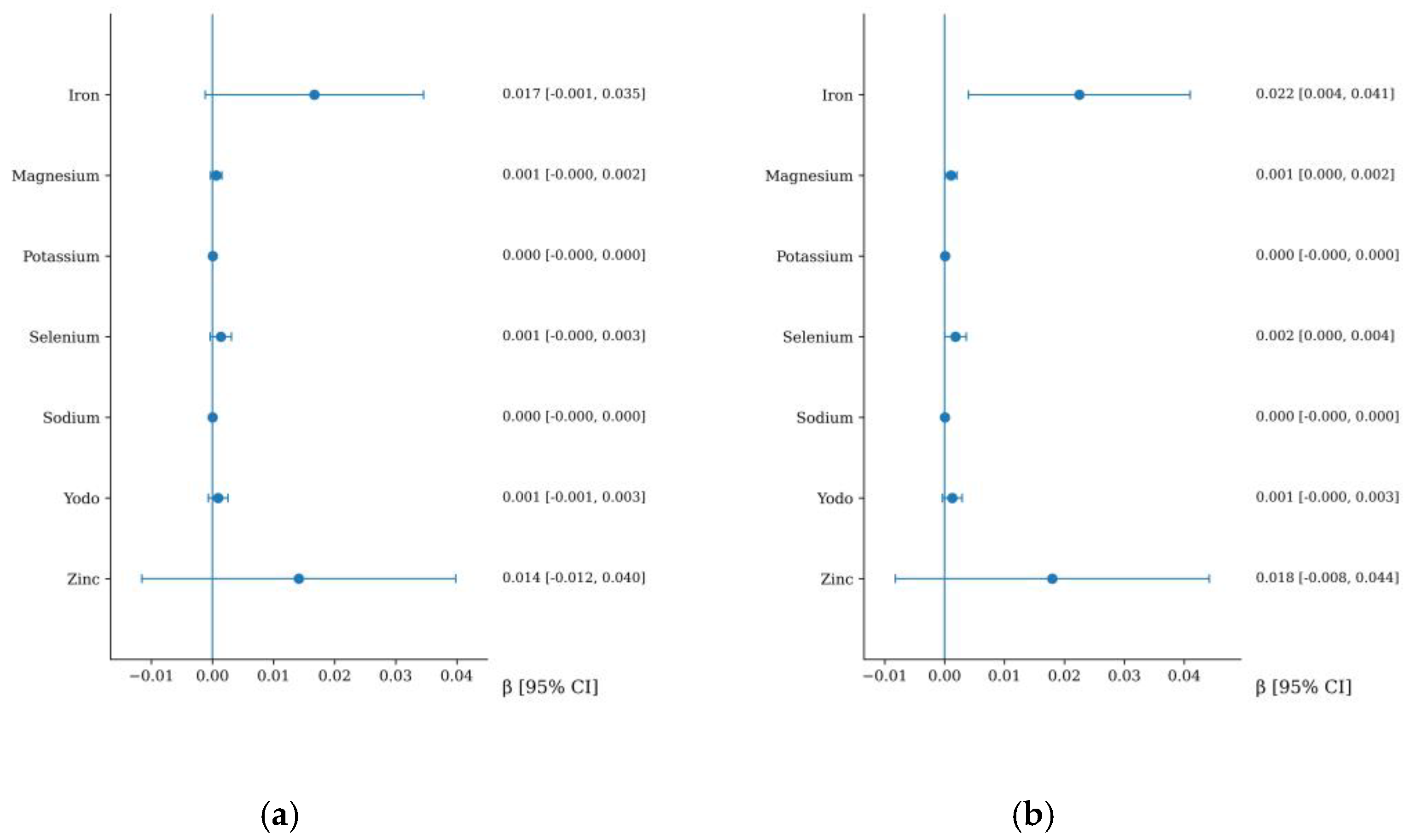
Background: Arterial stiffness is a key marker of vascular aging and an independent predictor of cardiovascular risk. Although diet has been proposed as an important modifiable factor influencing vascular health, the independent associations between specific macro- and micronutrients and the progression of arterial stiffness remain insufficiently characterized. Objective: To evaluate the association between dietary macronutrient and micronutrient intake and changes in arterial stiffness over a five-year follow-up in adults without previous cardiovascular disease. Methods: This longitudinal study included 466 participants from the EVA study who were evaluated at baseline and after a five-year follow-up (mean age 55.96 ± 14.15 years; 51.1% women). Arterial stiffnes was assessed using carotid–femoral pulse wave velocity (cfPWV) and the cardio-ankle vascular index (CAVI). Dietary intake of macronutrients and micronutrients was estimated using the EVIDENT smartphone application. Multivariable linear regression models were used to examine the association between nutrient intake and arterial stiffness progression. Model 1 was adjusted for age and sex, and Model 2 was additionally adjusted for lifestyle variables and cardiovascular risk factors. Results: Higher dietary fiber intake was independently associated with a lower increment in cfPWV after full adjustment (β = −0.025; 95% CI: −0.046 to −0.005). Alcohol intake showed a positive association with CAVI increment in the fully adjusted model (β = 0.020; 95% CI: 0.006 to 0.034). Iron intake was also independently associated with increased CAVI (β = 0.022; 95% CI: 0.004 to 0.041). Carbohydrate intake showed a small positive association with CAVI, whereas no consistent independent associations were observed for other macro- or micronutrients. Conclusions: In this adult population without previous cardiovascular disease, higher dietary fiber intake was associated with lower progression of central arterial stiffness, whereas alcohol and iron intake showed positive associations with peripheral arterial stiffness. Overall, most nutrients were not independently related to arterial stiffness after comprehensive adjustment. These findings suggest that selected dietary components may contribute modestly to vascular aging.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design, Participants, and Sample Size
2.2. Ethical Considerations
2.3. Dependent Variables
2.4. Independent Variables
2.5. Confounding Variables
2.6. Statistical Analysis
3. Results
3.1. Clinical and Vascular Characteristics
3.2. Macronutrient Intake
3.3. Micronutrient Intake
3.4. Association between Macronutrient Intake and cfPWV Increment

3.5. Association between Macronutrient Intake and CAVI Increment

3.6. Association between Micronutrient Intake and cfPWV Increment
3.7. Association between Micronutrient Intake and CAVI Increment
4. Discussion
4.1. Main Findings
4.2. Dietary Fiber and Arterial Stiffness
4.3. Alcohol and Arterial Stiffness
4.4. Iron, Oxidative Stress, and Arterial Stiffness
4.5. Carbohydrates and Arterial Stiffness
4.6. Lack of Associations for Other Nutrients
4.7. Clinical Implications
4.8. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| APISAL | Primary Care Research Unit of Salamanca |
| AS | Arterial stiffness |
| BMI | Body mass index |
| cfPWV | Carotid–femoral pulse wave velocity |
| CI | Confidence interval |
| CVR | Cardiovascular risk |
| CVRFs | Cardiovascular risk factors |
| CVD | Cardiovascular disease |
| ECG | Electrocardiogram |
| IBSAL | Institute of Biomedical Research of Salamanca |
| IPAQ-SF | International Physical Activity Questionnaire–Short Form |
| IU | International units |
| MEDAS | Mediterranean Diet Adherence Screener |
| MET | Metabolic equivalent of task |
| PWV | Pulse wave velocity |
| REDIAPP | Primary Care Research Group of Castilla y León |
| RICAPPS | Research Network on Chronicity, Primary Care and Health Promotion |
| SDoH | Social determinants of health |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| WHO | World Health Organization |
References
- Zhong, Q.; Hu, M.-J.; Cui, Y.-J.; Liang, L.; Zhou, M.-M.; Yang, Y.-W.; Huang, F. Carotid–Femoral Pulse Wave Velocity in the Prediction of Cardiovascular Events and Mortality: An Updated Systematic Review and Meta-Analysis. Angiology 2018, 69, 617–629. [Google Scholar] [CrossRef]
- Sequí-Domínguez, I.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Pozuelo-Carrascosa, D.P.; Nuñez de Arenas-Arroyo, S.; Martínez-Vizcaíno, V. Accuracy of Pulse Wave Velocity Predicting Cardiovascular and All-Cause Mortality. A Systematic Review and Meta-Analysis. J Clin Med 2020, 9, 2080. [Google Scholar] [CrossRef]
- Segers, P.; Rietzschel, E.R.; Chirinos, J.A. How to Measure Arterial Stiffness in Humans. Arterioscler Thromb Vasc Biol 2020, 40, 1034–1043. [Google Scholar] [CrossRef]
- Wilkinson, I.B.; Mäki-Petäjä, K.M.; Mitchell, G.F. Uses of Arterial Stiffness in Clinical Practice. Arterioscler Thromb Vasc Biol 2020, 40, 1063–1067. [Google Scholar] [CrossRef]
- Townsend, R.R.; Wilkinson, I.B.; Schiffrin, E.L.; Avolio, A.P.; Chirinos, J.A.; Cockcroft, J.R.; Heffernan, K.S.; Lakatta, E.G.; McEniery, C.M.; Mitchell, G.F.; et al. Recommendations for Improving and Standardizing Vascular Research on Arterial Stiffness. Hypertension 2015, 66, 698–722. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-K. Cardio-Ankle Vascular Index (CAVI) as an Indicator of Arterial Stiffness. Integr Blood Press Control 2013, 27. [Google Scholar] [CrossRef]
- Shirai, K.; Hiruta, N.; Song, M.; Kurosu, T.; Suzuki, J.; Tomaru, T.; Miyashita, Y.; Saiki, A.; Takahashi, M.; Suzuki, K.; et al. Cardio-Ankle Vascular Index (CAVI) as a Novel Indicator of Arterial Stiffness: Theory, Evidence and Perspectives. J Atheroscler Thromb 2011, 18, 924–938. [Google Scholar] [CrossRef]
- Miyoshi, T.; Ito, H.; Shirai, K.; Horinaka, S.; Higaki, J.; Yamamura, S.; Saiki, A.; Takahashi, M.; Masaki, M.; Okura, T.; et al. Predictive Value of the Cardio-Ankle Vascular Index for Cardiovascular Events in Patients at Cardiovascular Risk. J Am Heart Assoc 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Pase, M.P.; Grima, N.A.; Sarris, J. The Effects of Dietary and Nutrient Interventions on Arterial Stiffness: A Systematic Review. Am J Clin Nutr 2011, 93, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Ravera, A.; Carubelli, V.; Sciatti, E.; Bonadei, I.; Gorga, E.; Cani, D.; Vizzardi, E.; Metra, M.; Lombardi, C. Nutrition and Cardiovascular Disease: Finding the Perfect Recipe for Cardiovascular Health. Nutrients 2016, 8. [Google Scholar] [CrossRef]
- Stanek, A.; Grygiel-Górniak, B.; Brożyna-Tkaczyk, K.; Myśliński, W.; Cholewka, A.; Zolghadri, S. The Influence of Dietary Interventions on Arterial Stiffness in Overweight and Obese Subjects. Nutrients 2023, 15, 1440. [Google Scholar] [CrossRef]
- Gomez-Sanchez, M.; Gomez-Sanchez, L.; Patino-Alonso, M.C.; Cunha, P.G.; Recio-Rodriguez, J.I.; Alonso-Dominguez, R.; Sanchez-Aguadero, N.; Rodriguez-Sanchez, E.; Maderuelo-Fernandez, J.A.; Garcia-Ortiz, L.; et al. Vascular Aging and Its Relationship with Lifestyles and Other Risk Factors in the General Spanish Population: Early Vascular Ageing Study. J Hypertens 2020, 38, 1110–1122. [Google Scholar] [CrossRef]
- Saz-Lara, A.; Cavero-Redondo, I.; Pascual-Morena, C.; Martínez-García, I.; Rodríguez-Gutiérrez, E.; Lucerón-Lucas-Torres, M.; Bizzozero-Peroni, B.; Moreno-Herráiz, N.; Martínez-Rodrigo, A. Early Vascular Aging as an Index of Cardiovascular Risk in Healthy Adults: Confirmatory Factor Analysis from the EVasCu Study. Cardiovasc Diabetol 2023, 22, 209. [Google Scholar] [CrossRef] [PubMed]
- Tisdel, D.M.; Gadberry, J.J.; Burke, S.L.; Carlini, N.A.; Fleenor, B.S.; Campbell, M.S. Dietary Fat and Alcohol in the Prediction of Indices of Vascular Health among Young Adults. Nutrition 2021, 84, 111120. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.-T.; Chan, Y.-H.; Yiu, K.H.; Li, S.-W.; Tam, S.; Lau, C.-P.; Tse, H.-F. Worsened Arterial Stiffness in High-Risk Cardiovascular Patients with High Habitual Carbohydrate Intake: A Cross-Sectional Vascular Function Study. BMC Cardiovasc Disord 2014, 14, 24. [Google Scholar] [CrossRef]
- Arnberg, K.; Larnkjær, L.; Michaelsen, K.F.; Mølgaard, C. Central Adiposity and Protein Intake Are Associated with Arterial Stiffness in Overweight Children. J Nutr 2012, 142, 878–885. [Google Scholar] [CrossRef]
- van de Laar, R.J.; da Stehouwer, C.; van Bussel, B.C.; te Velde, S.J.; Prins, M.H.; Twisk, J.W.; Ferreira, I. Lower Lifetime Dietary Fiber Intake Is Associated with Carotid Artery Stiffness: The Amsterdam Growth and Health Longitudinal Study. Am J Clin Nutr 2012, 96, 14–23. [Google Scholar] [CrossRef]
- Campbell, M.S.; Fleenor, B.S. Whole Grain Consumption Is Negatively Correlated with Obesity-Associated Aortic Stiffness: A Hypothesis. Nutrition 2018, 45, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health Benefits of Dietary Fiber. Nutr Rev 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Veronese, N.; Solmi, M.; Caruso, M.G.; Giannelli, G.; Osella, A.R.; Evangelou, E.; Maggi, S.; Fontana, L.; Stubbs, B.; Tzoulaki, I. Dietary Fiber and Health Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Am J Clin Nutr 2018, 107, 436–444. [Google Scholar] [CrossRef]
- Ronksley, P.E.; Brien, S.E.; Turner, B.J.; Mukamal, K.J.; Ghali, W.A. Association of Alcohol Consumption with Selected Cardiovascular Disease Outcomes: A Systematic Review and Meta-Analysis. BMJ 2011, 342, d671–d671. [Google Scholar] [CrossRef]
- Roerecke, M.; Tobe, S.W.; Kaczorowski, J.; Bacon, S.L.; Vafaei, A.; Hasan, O.S.M.; Krishnan, R.J.; Raifu, A.O.; Rehm, J. Sex-Specific Associations Between Alcohol Consumption and Incidence of Hypertension: A Systematic Review and Meta-Analysis of Cohort Studies. J Am Heart Assoc 2018, 7. [Google Scholar] [CrossRef]
- Piano, M.R. Alcohol’s Effects on the Cardiovascular System. Alcohol Res 2017, 38, 219–241. [Google Scholar] [CrossRef]
- del Giorno, R.; Maddalena, A.; Bassetti, S.; Gabutti, L. Association between Alcohol Intake and Arterial Stiffness in Healthy Adults: A Systematic Review. Nutrients 2022, 14, 1207. [Google Scholar] [CrossRef]
- Wood, A.M.; Kaptoge, S.; Butterworth, A.S.; Willeit, P.; Warnakula, S.; Bolton, T.; Paige, E.; Paul, D.S.; Sweeting, M.; Burgess, S.; et al. Risk Thresholds for Alcohol Consumption: Combined Analysis of Individual-Participant Data for 599 912 Current Drinkers in 83 Prospective Studies. Lancet 2018, 391, 1513–1523. [Google Scholar] [CrossRef]
- Ha, J.Y.; Kim, M.K.; Kang, S.; Nam, J.S.; Ahn, C.W.; Kim, K.R.; Park, J.S. Serum Ferritin Levels Are Associated with Arterial Stiffness in Healthy Korean Adults. Vascular Medicine 2016, 21, 325–330. [Google Scholar] [CrossRef]
- Kiechl, S.; Willeit, J.; Egger, G.; Poewe, W.; Oberhollenzer, F. Body Iron Stores and the Risk of Carotid Atherosclerosis: Prospective Results from the Bruneck Study. Circulation 1997, 96, 3300–3307. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Li, K.; Xing, W.; Dong, M.; Yi, M.; Zhang, H. Role of Iron-Related Oxidative Stress and Mitochondrial Dysfunction in Cardiovascular Diseases. Oxid Med Cell Longev 2022, 2022. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, K.T.; de Jesus, A.; Ardehali, H. Iron Metabolism in Cardiovascular Disease: Physiology, Mechanisms, and Therapeutic Targets. Circ Res 2023, 132, 379–396. [Google Scholar] [CrossRef] [PubMed]
- Salonen, J.T.; Nyyssönen, K.; Korpela, H.; Tuomilehto, J.; Seppänen, R.; Salonen, R. High Stored Iron Levels Are Associated with Excess Risk of Myocardial Infarction in Eastern Finnish Men. Circulation 1992, 86, 803–811. [Google Scholar] [CrossRef]
- García-Ortiz, L.; Recio-Rodríguez, J.I.; Rodríguez-Sánchez, E.; Patino-Alonso, M.C.; Agudo-Conde, C.; Rodríguez-Martín, C.; Castaño-Sánchez, C.; Runkle, I.; Gómez-Marcos, M.A. Sodium and Potassium Intake Present a J-Shaped Relationship with Arterial Stiffness and Carotid Intima-Media Thickness. Atherosclerosis 2012, 225, 497–503. [Google Scholar] [CrossRef]
- D’Elia, L.; Galletti, F.; la Fata, E.; Sabino, P.; Strazzullo, P. Effect of Dietary Sodium Restriction on Arterial Stiffness. J Hypertens 2018, 36, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Ishida, A.; Isotani, A.; Fujisawa, M.; del Saz, E.G.; Okumiya, K.; Kimura, Y.; Manuaba, I.I.B.; Rantetampang, A.L.; Ohya, Y.; Matsubayashi, K. Effects of a Low-Salt and High-Potassium Diet on Arterial Stiffness and Left Ventricular Function in Indigenous Papuans. J Am Heart Assoc 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Marcos, MA; Martinez-Salgado, C; Gonzalez-Sarmiento, R; Hernandez-Rivas, JM; Sanchez-Fernandez, PL; Recio-Rodriguez, JI; Rodriguez-Sanchez, E; Garcia-Ortiz, L. Association between different risk factors and vascular accelerated ageing (EVA study): study protocol for a cross-sectional, descriptive observational study. BMJ Open. 2016, 6, e011031. [Google Scholar] [CrossRef]
- Vandenbroucke, JP; von Elm, E; Altman, DG; Gøtzsche, PC; Mulrow, CD; Pocock, SJ; Poole, C; Schlesselman, JJ; Egger, M. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. Int J Surg. 2014, 12, 1500–1524. [Google Scholar] [CrossRef]
- World Medical Association. Declaration of Helsinki: ethical principles for medical research involving human participants. JAMA 2025, 333, 71–74. [Google Scholar] [CrossRef]
- Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: 'establishing normal and reference values'. Eur Heart J. 2010, 31, 2338–2350. [CrossRef]
- Shirai, K.; Utino, J.; Otsuka, K.; Takata, M. A Novel Blood Pressure-Independent Arterial Wall Stiffness Parameter; Cardio-Ankle Vascular Index (CAVI). J Atheroscler Thromb 2006, 13, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Recio-Rodriguez, JI; Rodriguez-Martin, C; Gonzalez-Sanchez, J; Rodriguez-Sanchez, E; Martin-Borras, C; Martínez-Vizcaino, V; Arietaleanizbeaskoa, MS; Magdalena-Gonzalez, O; Fernandez-Alonso, C; Maderuelo-Fernandez, JA; et al. EVIDENT smartphone app, a new method for the dietary record: comparison with a food frequency questionnaire. JMIR Mhealth Uhealth 2019, 7, e11463. [Google Scholar] [CrossRef]
- Schröder, H; Fitó, M; Estruch, R; Martínez-González, MA; Corella, D; Salas-Salvadó, J; Lamuela-Raventós, R; Ros, E; Salaverría, I; Fiol, M; et al. A short screener is valid for assessing Mediterranean diet adherence among older Spanish men and women. J Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Marshall, AL; Miller, YD; Burton, NW; Brown, WJ. Measuring total and domain-specific sitting: a study of reliability and validity. Med Sci Sports Exerc. 2010, 42, 1094–1102. [Google Scholar] [CrossRef]
- Chu, AHY; Ng, SHX; Koh, D; Müller-Riemenschneider, F. Domain-specific adult sedentary behaviour questionnaire (ASBQ) and the GPAQ single-item question: a reliability and validity study in an Asian population. Int J Environ Res Public Health 2018, 15. [Google Scholar] [CrossRef]
- Lee, PH; Macfarlane, DJ; Lam, TH; Stewart, SM. Validity of the international physical activity questionnaire short form (IPAQ-SF): a systematic review. Int J Behav Nutr Phys Act. 2011, 8, 115. [Google Scholar] [CrossRef]
- Meh, K; Jurak, G; Sorić, M; Rocha, P; Sember, V. Validity and reliability of IPAQ-SF and GPAQ for assessing sedentary behaviour in adults in the European Union: a systematic review and meta-analysis. Int J Environ Res Public Health 2021, 18. [Google Scholar] [CrossRef]
- Williams, B; Mancia, G; Spiering, W; Agabiti Rosei, E; Azizi, M; Burnier, M; Clement, DL; Coca, A; de Simone, G; Dominiczak, A; et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Threapleton, D.E.; Greenwood, D.C.; Evans, C.E.L.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.; Cade, J.E.; Gale, C.P.; Burley, V.J. Dietary Fibre Intake and Risk of Cardiovascular Disease: Systematic Review and Meta-Analysis. BMJ 2013, 347, f6879–f6879. [Google Scholar] [CrossRef]
- Kim, Y.; Je, Y. Dietary Fibre Intake and Mortality from Cardiovascular Disease and All Cancers: A Meta-Analysis of Prospective Cohort Studies. Arch Cardiovasc Dis 2016, 109, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Demirci, B.G.; Tutal, E.; Eminsoy, I.O.; Kulah, E.; Sezer, S. Dietary Fiber Intake: Its Relation With Glycation End Products and Arterial Stiffness in End-Stage Renal Disease Patients. J Ren Nutr 2019, 29, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; te Morenga, L. Carbohydrate Quality and Human Health: A Series of Systematic Reviews and Meta-Analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Pal, S.; Khossousi, A.; Binns, C.; Dhaliwal, S.; Radavelli-Bagatini, S. The Effects of 12-Week Psyllium Fibre Supplementation or Healthy Diet on Blood Pressure and Arterial Stiffness in Overweight and Obese Individuals. British Journal of Nutrition 2012, 107, 725–734. [Google Scholar] [CrossRef]
- Briasoulis, A.; Agarwal, V.; Messerli, F.H. Alcohol Consumption and the Risk of Hypertension in Men and Women: A Systematic Review and Meta-Analysis. J Clin Hypertens (Greenwich) 2012, 14, 792–798. [Google Scholar] [CrossRef]
- O’Neill, D.; Britton, A.; Brunner, E.J.; Bell, S. Twenty-Five-Year Alcohol Consumption Trajectories and Their Association with Arterial Aging: A Prospective Cohort Study. J Am Heart Assoc 2017, 6. [Google Scholar] [CrossRef]
- Vinchi, F.; Muckenthaler, M.U.; da Silva, M.C.; Balla, G.; Balla, J.; Jeney, V. Atherogenesis and Iron: From Epidemiology to Cellular Level. Front Pharmacol 2014, 5, 94. [Google Scholar] [CrossRef] [PubMed]
- Bhupathiraju, S.N.; Tobias, D.K.; Malik, V.S.; Pan, A.; Hruby, A.; Manson, J.E.; Willett, W.C.; Hu, F.B. Glycemic Index, Glycemic Load, and Risk of Type 2 Diabetes: Results from 3 Large US Cohorts and an Updated Meta-Analysis. Am J Clin Nutr 2014, 100, 218–232. [Google Scholar] [CrossRef] [PubMed]
- Seidelmann, S.B.; Claggett, B.; Cheng, S.; Henglin, M.; Shah, A.; Steffen, L.M.; Folsom, A.R.; Rimm, E.B.; Willett, W.C.; Solomon, S.D. Dietary Carbohydrate Intake and Mortality: A Prospective Cohort Study and Meta-Analysis. Lancet Public Health 2018, 3, e419–e428. [Google Scholar] [CrossRef] [PubMed]
| Variable | Total (n = 466) | Men (n = 226) | Women (n = 240) | p-Value |
| Age (years) | 55.96 ± 14.15 | 55.94 ± 14.19 | 55.98 ± 14.13 | 0.973 |
| Alcohol (g/week) | 0.00 (0.00–65.00) | 30.00 (0.00–105.00) | 0.00 (0.00–20.00) | <0.001 |
| Mediterranean diet score | 7.17 ± 2.07 | 6.73 ± 1.98 | 7.58 ± 2.08 | <0.001 |
| Total physical activity (MET-min/week) | 1537.50 (742.12–2772.00) | 2106.00 (1386.00–4134.00) | 1263.75 (693.00–2079.00) | <0.001 |
| Sitting time (h/week) | 42.14 ± 17.81 | 47.97 ± 16.54 | 36.66 ± 17.23 | <0.001 |
| Systolic BP (mmHg) | 119.70 ± 17.76 | 125.73 ± 16.49 | 114.03 ± 17.06 | <0.001 |
| Diastolic BP (mmHg) | 75.64 ± 10.00 | 77.68 ± 9.16 | 73.73 ± 10.39 | <0.001 |
| Pulse pressure (mmHg) | 44.06 ± 12.32 | 48.05 ± 12.03 | 40.30 ± 11.40 | <0.001 |
| Heart rate (bpm) | 68.72 ± 9.52 | 67.65 ± 9.99 | 69.73 ± 8.96 | 0.018 |
| Total cholesterol (mg/dL) | 194.97 ± 32.84 | 192.61 ± 33.09 | 197.20 ± 32.52 | 0.132 |
| LDL cholesterol (mg/dL) | 115.58 ± 29.51 | 117.60 ± 30.57 | 113.68 ± 28.42 | 0.153 |
| Glucose (mg/dL) | 85.00 (79.00–93.00) | 87.00 (80.00–95.00) | 84.00 (78.00–90.00) | 0.001 |
| Weight (kg) | 72.48 ± 13.85 | 79.73 ± 11.85 | 65.66 ± 12.02 | <0.001 |
| Height (cm) | 165.06 ± 9.69 | 171.81 ± 7.26 | 158.70 ± 7.03 | <0.001 |
| Body mass index (kg/m²) | 26.55 ± 4.22 | 26.99 ± 3.41 | 26.13 ± 4.82 | 0.027 |
| Pulse wave velocity (m/s) | 7.60 (6.50–9.10) | 7.90 (6.60–10.10) | 7.30 (6.40–8.70) | <0.001 |
| ΔcfPWV (m/s) | 1.14 ± 1.76 | 1.33 ± 1.75 | 0.96 ± 1.75 | 0.023 |
| Mean CAVI | 8.01 ± 1.46 | 8.16 ± 1.50 | 7.87 ± 1.40 | 0.030 |
| ΔCAVI | 0.18 ± 0.89 | 0.24 ± 0.86 | 0.12 ± 0.92 | 0.129 |
| Variable | Total (n = 466) | Men (n = 226) | Women (n = 240) | p-Value |
| Energy intake (kcal/day) | 2061 (1770–24766) | 2177 (1812–2557) | 1958 (1714–2324) | <0.001 |
| Protein (g/day) | 96 (80–111) | 99 (85–115) | 91 (76–108) | <0.001 |
| Carbohydrates (g/day) | 193 (159–236) | 203 (162–242) | 188 (156–233) | 0.059 |
| Dietary fiber (g/day) | 25 (19–30) | 25 (19–30) | 25 (19–31) | 0.970 |
| Total fat (g/day) | 92(77–114) | 98 (81–117) | 88 (75–109) | <0.001 |
| Saturated fatty acids (g/day) | 30 (24–37) | 32 (26–38) | 28 (22–35) | 0.002 |
| Monounsaturated fatty acids (g/day) | 43 (35–54) | 47 (38–56) | 40 (34–51) | <0.001 |
| Polyunsaturated fatty acids (g/day) | 16 (9–15) | 12 (9–15) | 11 (9–14) | 0.093 |
| Dietary cholesterol (mg/day) | 358 (287–439) | 394 (307–470) | 328 (268–402) | <0.001 |
| Alcohol (g/day) | 0.00 (0.00–4.62) | 0.37 (0.00–7.55) | 0.00 (0.00–2.31) | <0.001 |
| Water (mL/day) | 1358 (1150–1606) | 1357 (1148–1666) | 1358 (1150–1562) | 0.217 |
| Variable | Total (n = 466) | Men (n = 226) | Women (n = 240) | p-Value |
| Iron (mg/day) | 156 (13–19) | 160 (130–20) | 15 (12–18) | 0.017 |
| Iodine (µg/day) | 103 (80–135) | 109 (82–141) | 100 (79–128) | 0.024 |
| Magnesium (mg/day) | 319 (261–375) | 321 (270–376) | 317 (257–373) | 0.422 |
| Zinc (mg/day) | 11 (9–13) | 11 (9–13) | 10 (9–12) | 0.006 |
| Selenium (µg/day) | 118 (94–151) | 122 (97–163) | 116 (90–142) | 0.008 |
| Sodium (mg/day) | 3384 (2509–4335) | 3669 (2843–4673) | 3137 (2355–4108) | <0.001 |
| Potassium (mg/day) | 3555 (2934–4261) | 3607 (2979–4345) | 3523 (2914–4170) | 0.146 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




