Submitted:
19 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
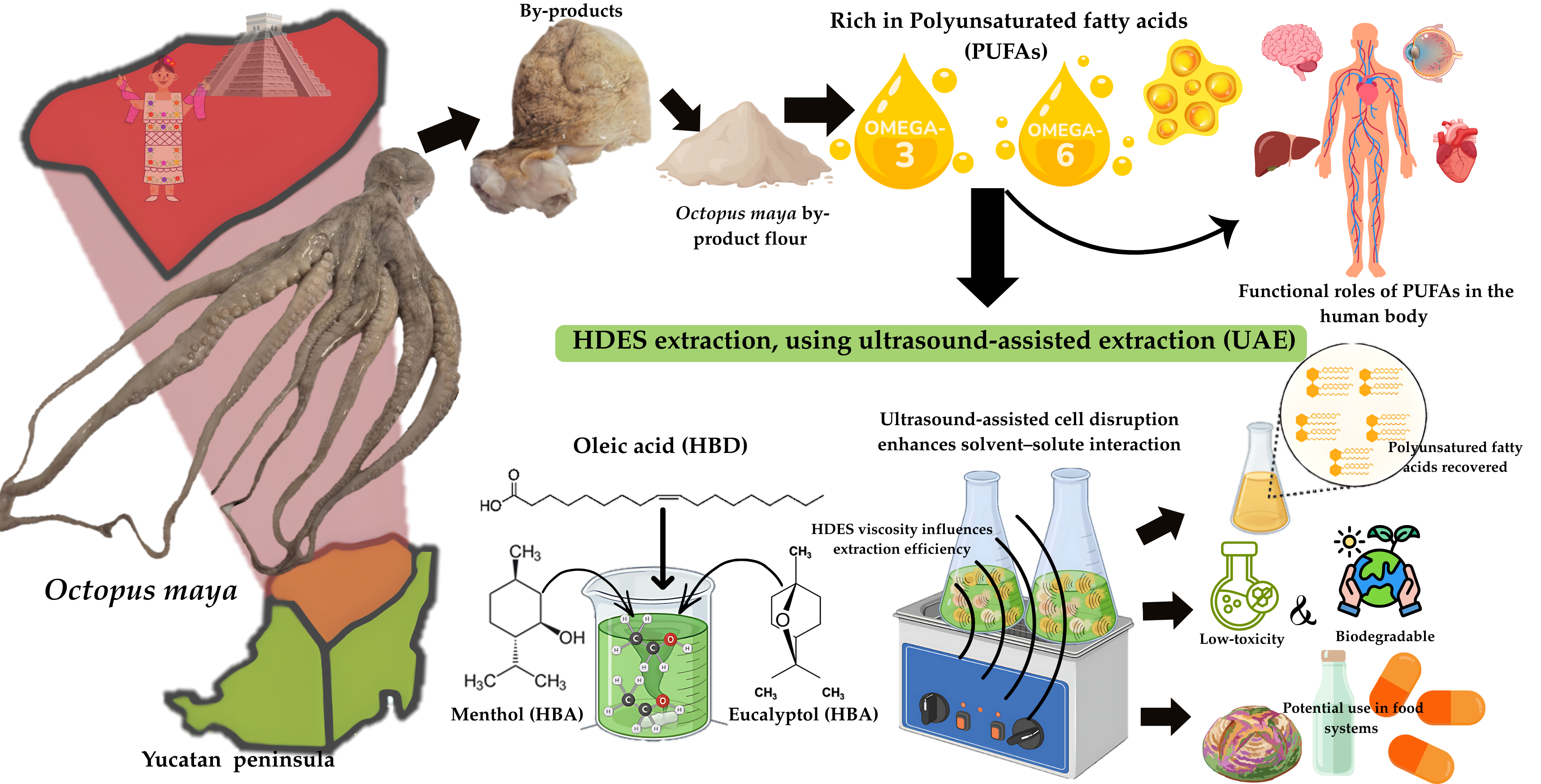
Keywords:
1. Introduction
2. Materials and Methods
2.1. Solvents and Reagents
2.2. Octopus maya Sample Collection
2.3. Processing of Octopus maya By-Product Samples
2.4. Conventional Oil Extraction (COE)
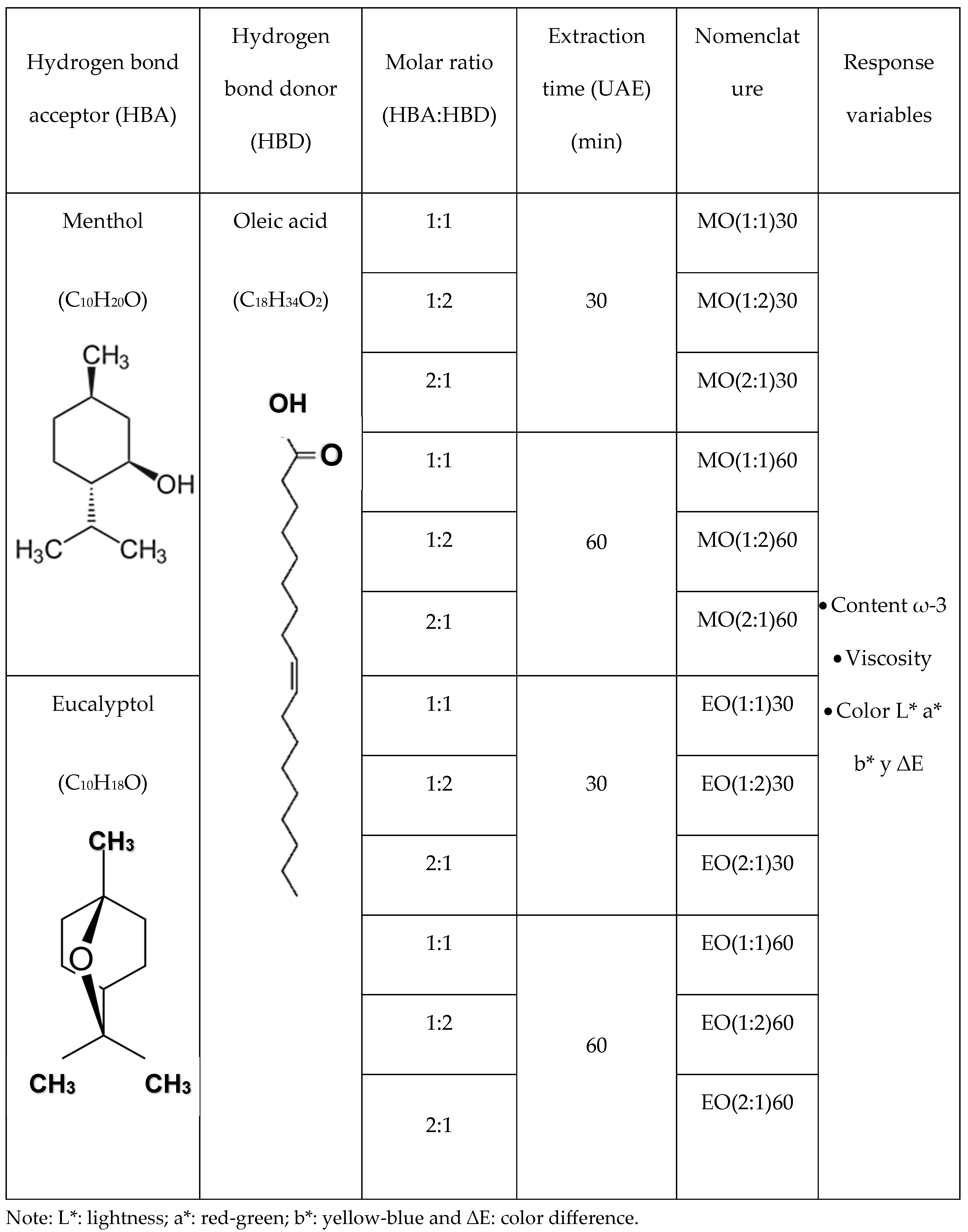
2.5. Preparation of HDES
2.6. Fatty acid Extraction Using HDES Under UAE
2.7. Viscosity of HDES Extracts
2.8. Color of HDES Extracts
2.9. Determination of ω-3 by UV–Vis Spectrophotometry
2.10. Fatty Acid Profiling by Gas Chromatography (GC)
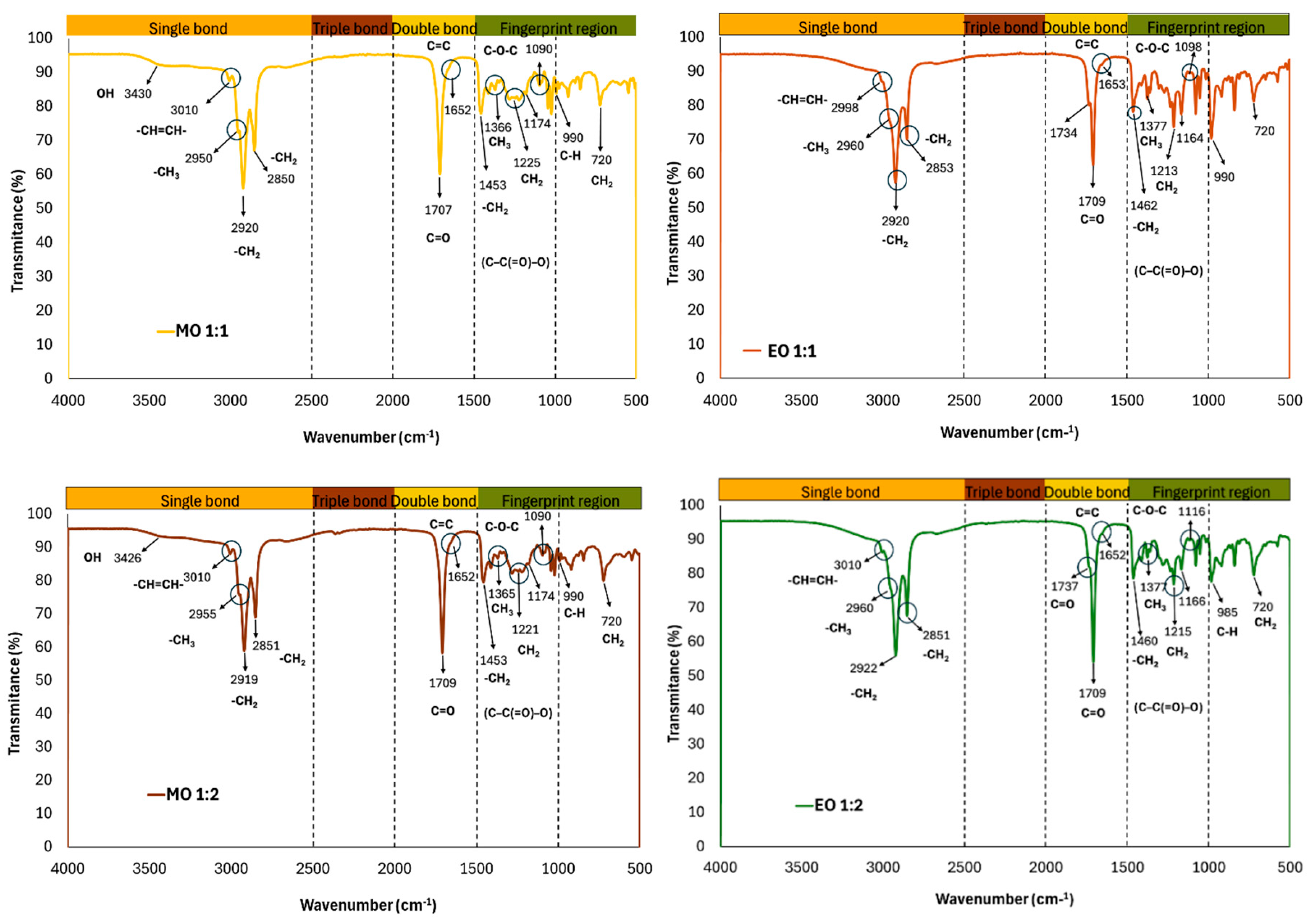
2.11. Fourier Transform Infrared Spectroscopy (FTIR)
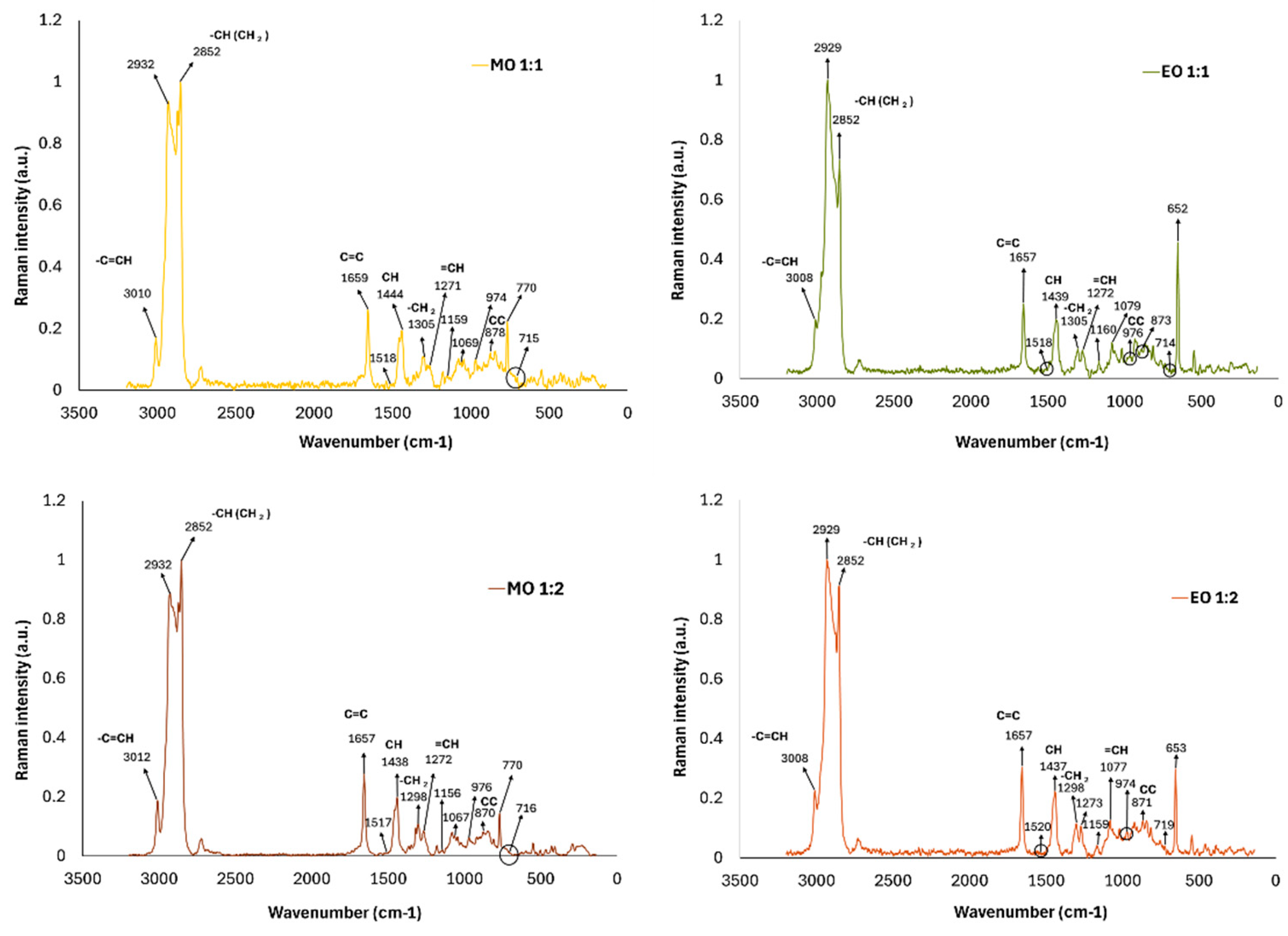
2.12. RAMAN Spectroscopy
2.13. Statistical Analysis
3. Results and Discussion
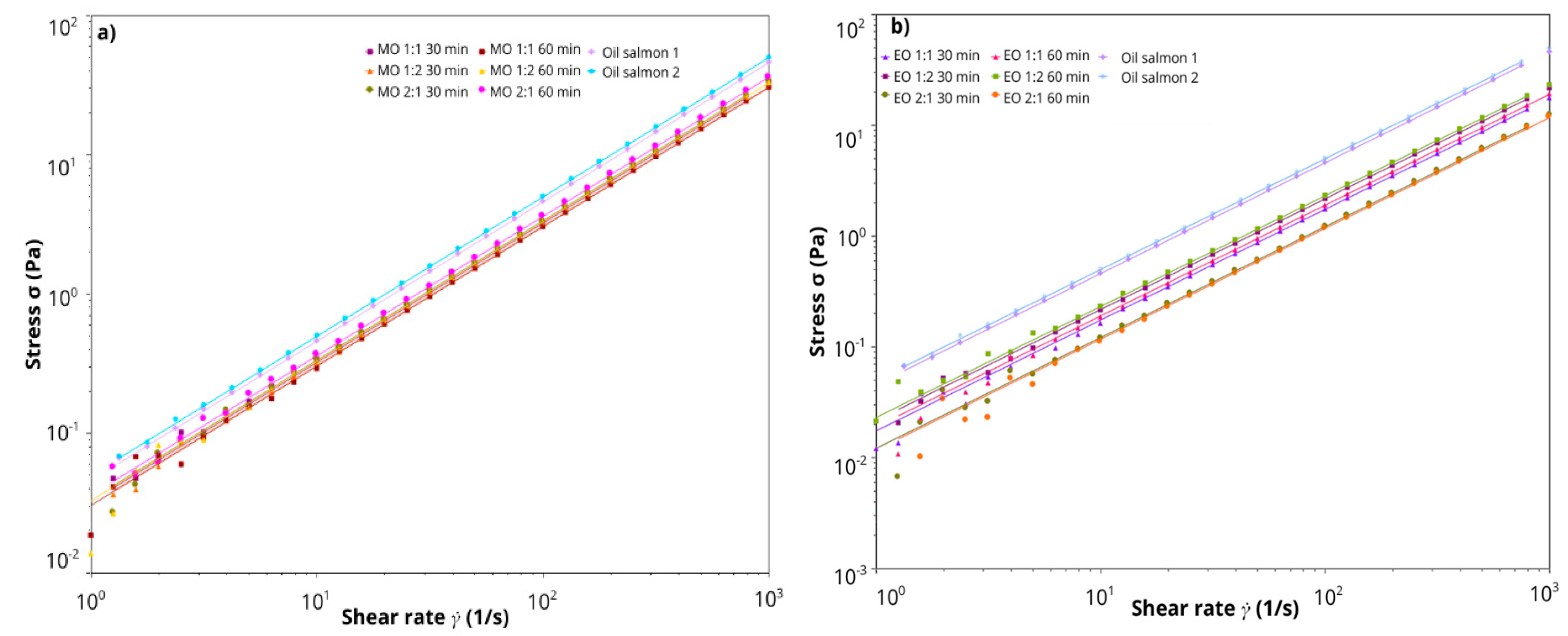
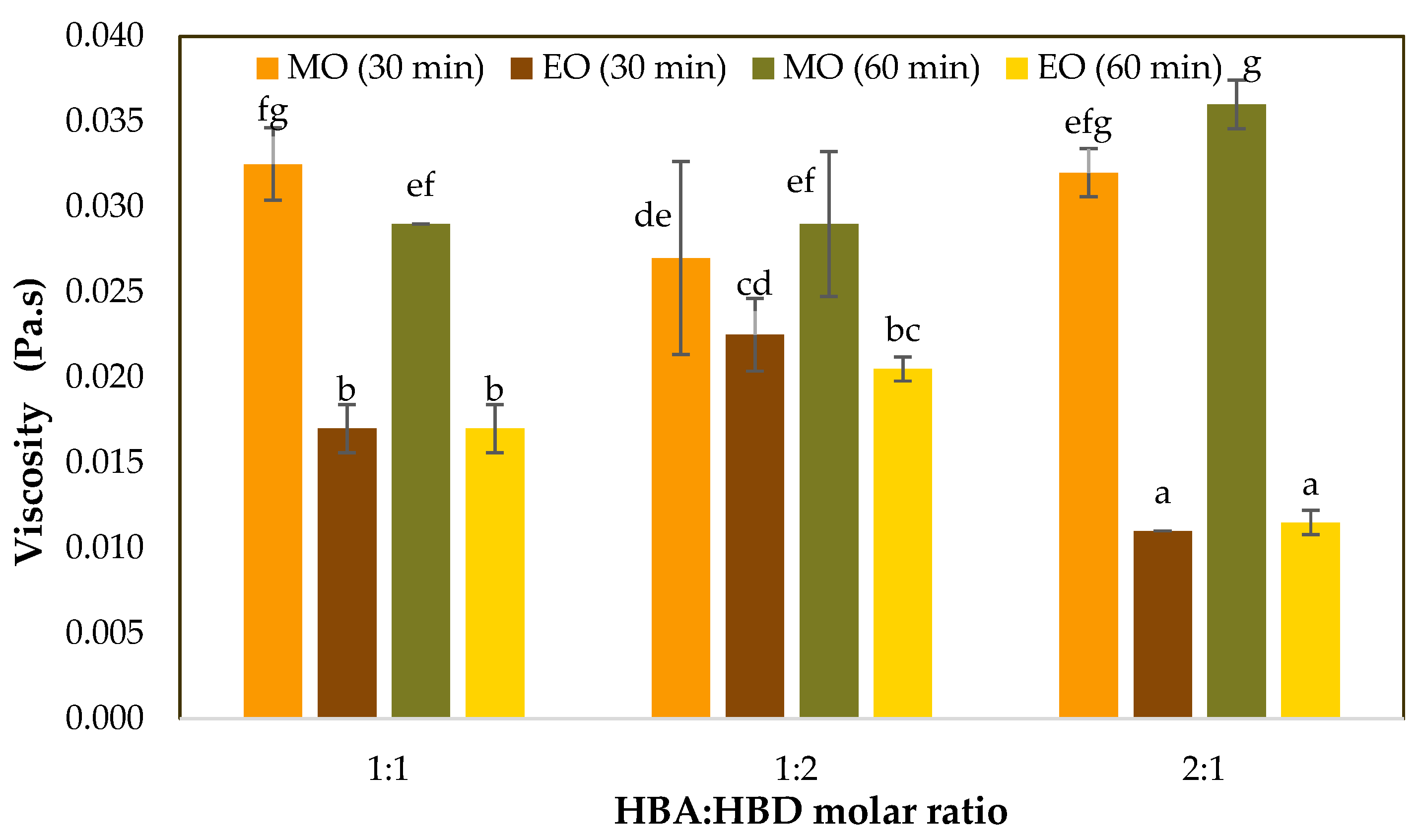
3.1. Viscosity of HDES Extracts
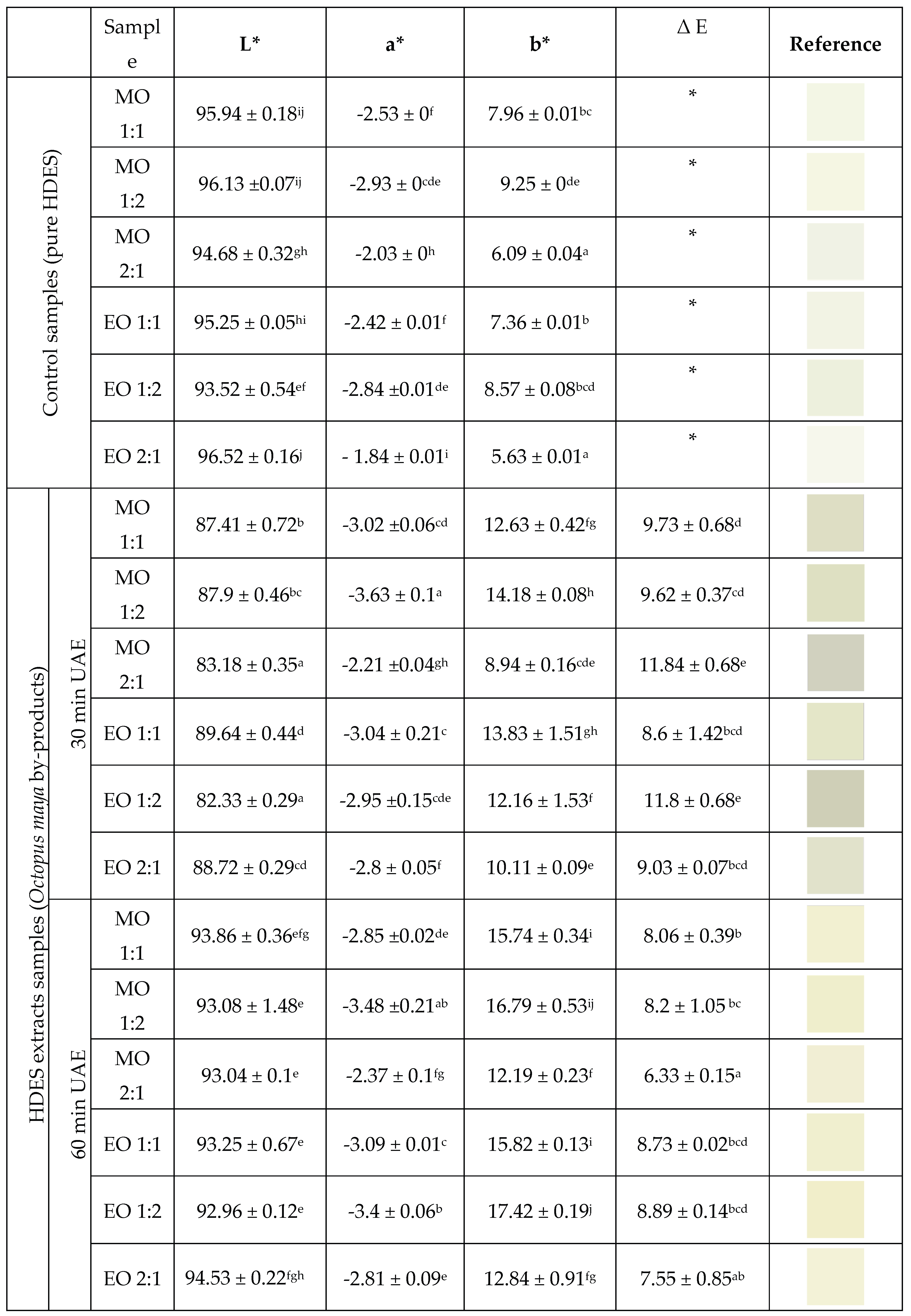
3.2. Color of HDES Extracts
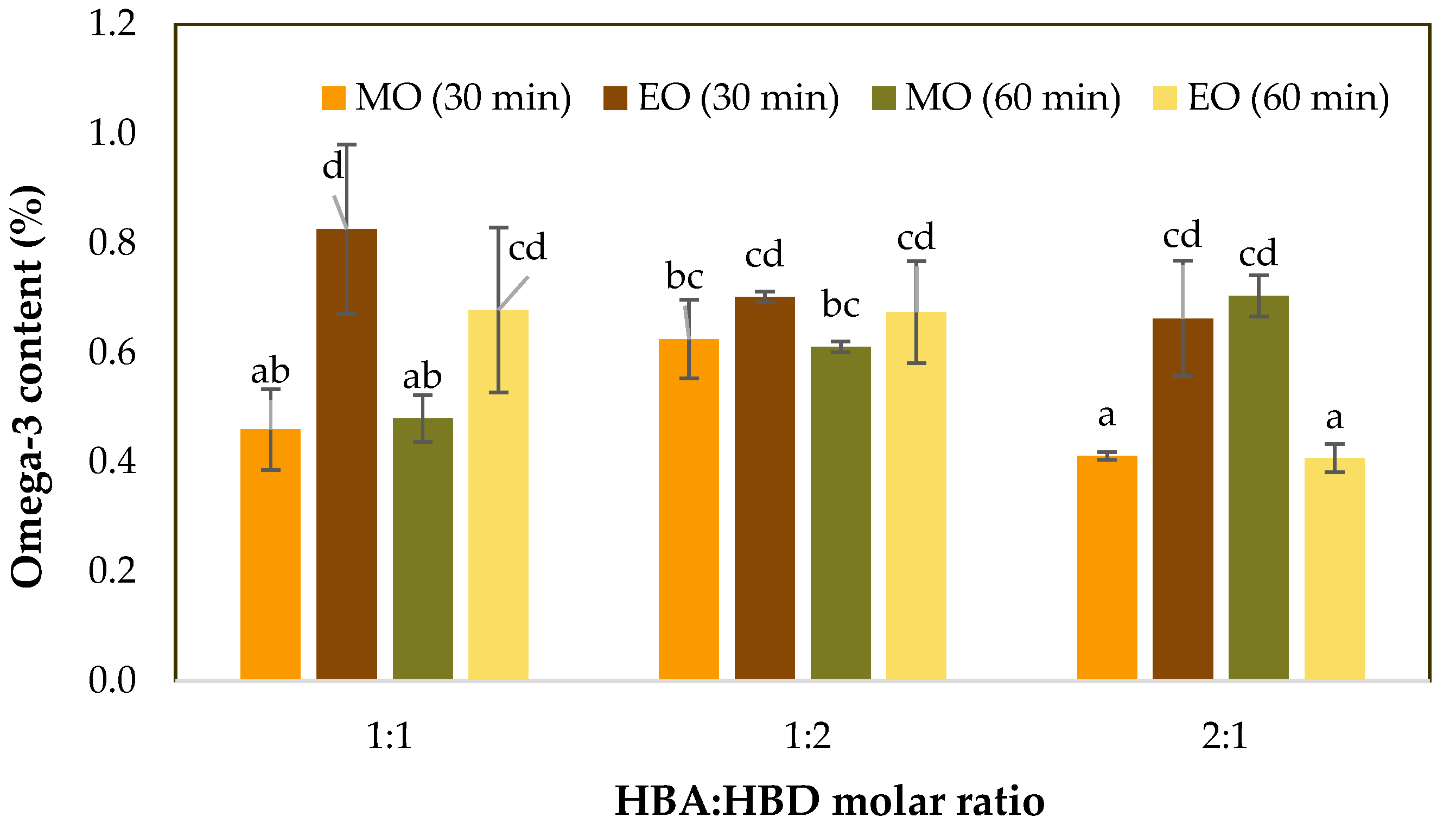
3.3. ω-3 Concentration in HDES Extracts (UV–Vis)
| Response variables | P values | ||||||
| Individual factors | Double interactions | Triple interactions | |||||
| HBA | MR (mol/mol) | ET (min) | HBA x MR | HBA x ET | MR x ET | HBA x MR x ET | |
| ω-3 (%) | 0.007 | 0.067 | 0.524 | 0.009 | 0.003 | 0.617 | 0.019 |
| Viscosity (Pa.s) | <0.001 | 0.240 | 0.867 | <0.001 | 0.506 | 0.280 | 0.252 |
| L* | 0.061 | <0.001 | <0.001 | <0.001 | 0.331 | <0.001 | <0.001 |
| a* | 0.387 | <0.001 | 0.041 | <0.001 | 0.004 | 0.060 | 0.112 |
| b* | 0.351 | <0.001 | <0.001 | 0.095 | 0.574 | 0.184 | 0.047 |
| ∆E | 0.641 | 0.033 | <0.001 | 0.018 | 0.023 | 0.006 | 0.005 |
3.4. FTIR and Raman Characterization of HDES Extracts from Octopus maya By-Products
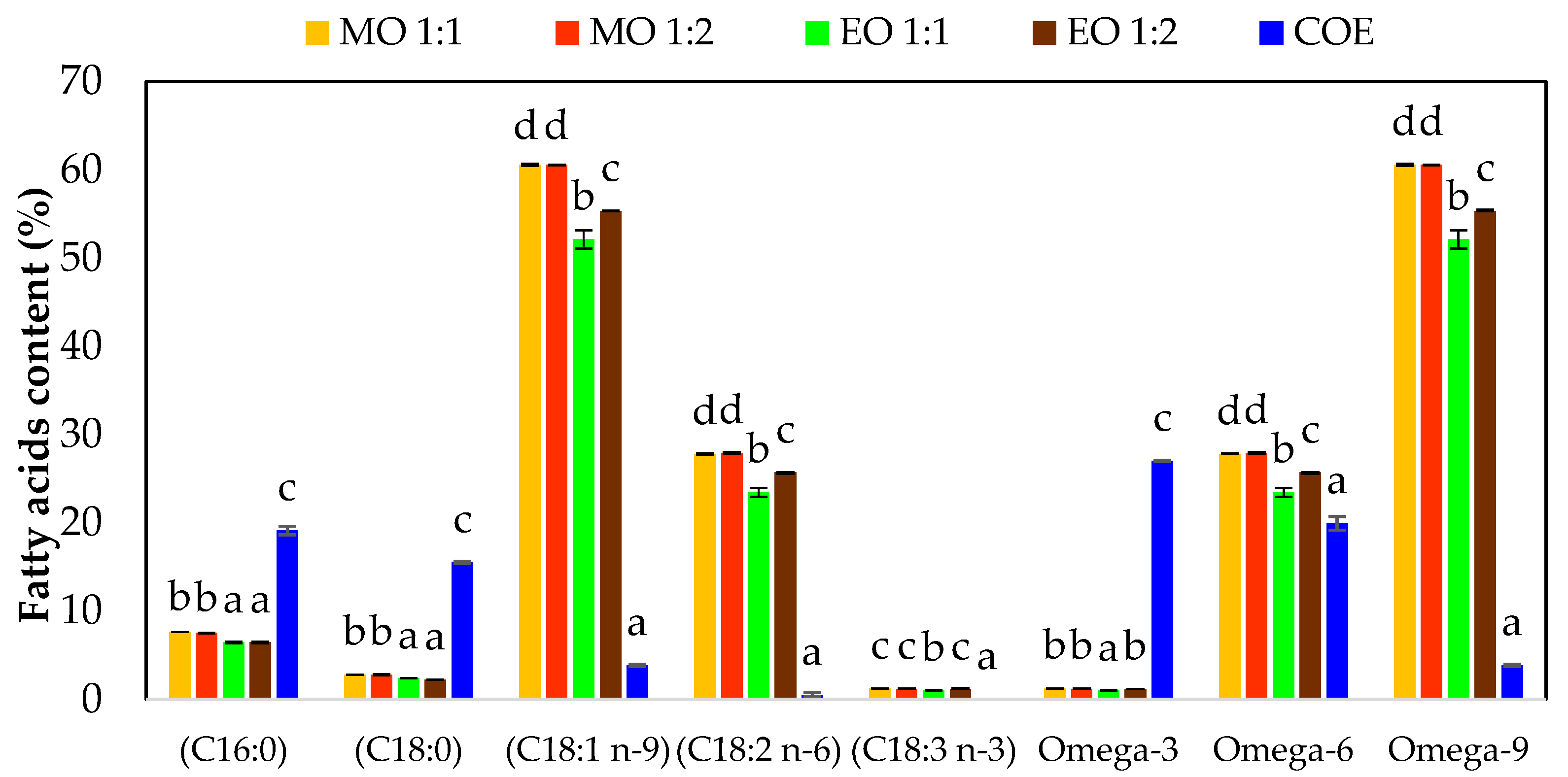
3.5. Fatty Acid Profile of Octopus maya By-Products
4. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Gobierno de México; Secretaría de Agricultura y Desarrollo Rural. El pulpo: la pesquería más importante de Yucatán inició este 1° de agosto . 2024. Available online: https://www.gob.mx/agricultura/yucatan/articulos/el-pulpo-la-pesqueria-mas-importante-de-yucatan-inicia-este-1o-de-agosto?idiom=es.
- Pineda-Suazo, D.; Escobedo-Hinojosa, W.; Fabian-Canseco, L. E.; Gallardo, P.; Moguel-Ojeda, C.; Caamal-Monsreal, C.; Sánchez-Arteaga, A.; Rosas, C. Evaluation of Octopus maya enzyme activity of the digestive gland and gastric juice . Biology Open 2024, 13(9), bio060429. [Google Scholar] [CrossRef] [PubMed]
- Rosas, C.; Markaida, U.; López Rocha, J. Octopus maya, the Mayan Octopus . In Octopus Biology and Ecology; Rosa, R., Gleadall, I., Pierce, G., Villanueva, R., Eds.; Elsevier, 2024; pp. 97–119. [Google Scholar] [CrossRef]
- Gobierno del Estado de Yucatán. Costa yucateca cierra exitosa temporada de pulpo . Comunicado de prensa. 2025. Available online: https://www.yucatan.gob.mx/saladeprensa/ver_nota.php?id=10213.
- Rasmiya Begum, S. L.; Himaya, S. M. M. S.; Imthiyas, M. S. M.; Afreen, S. M. M. S. Fish Waste: Understanding the pollution potential and sustainable mitigation strategies. In Fish Waste to Valuable Products; Springer Nature Singapore: Singapore, 2024; pp. 427–440. [Google Scholar] [CrossRef]
- Torrinha, Á.; Cruz, R.; Gomes, F.; Mendes, E.; Casal, S.; Morais, S. Octopus lipid and vitamin E composition: Interspecies, interorigin, and nutritional variability . Journal of Agricultural and Food Chemistry 2014, 62, 8508–8517. Available online: https://pubs.acs.org/doi/10.1021/jf502502b. [CrossRef]
- Calder, P. C.; Cawood, A. L.; James, C.; Page, F.; Putnam, S.; Minihane, A. M. An overview of national and international long chain omega-3 polyunsaturated fatty acid intake recommendations for healthy populations. Nutrition Research Reviews 2025, 1–33. [Google Scholar] [CrossRef]
- Banaszak, M.; Dobrzyńska, M.; Kawka, A.; Górna, I.; Woźniak, D.; Przysławski, J.; Drzymała-Czyż, S. Role of Omega-3 fatty acids eicosapentaenoic (EPA) and docosahexaenoic (DHA) as modulatory and anti-inflammatory agents in noncommunicable diet-related diseases–Reports from the last 10 years. Clinical Nutrition ESPEN 2024, 63, 240–258. [Google Scholar] [CrossRef]
- Gutierres, D.; Pacheco, R.; Reis, C. P. The Role of Omega-3 and Omega-6 Polyunsaturated Fatty Acid Supplementation in Human Health. Foods 2025, 14(19), 3299. [Google Scholar] [CrossRef]
- Shalfoh, E.; Ahmad, M. I.; Binhweel, F.; Shaah, M. A.; Senusi, W.; Hossain, M. S.; Alsaadi, S. Fish waste oil extraction using supercritical CO2 extraction for biodiesel production: Mathematical, and kinetic modeling. Renewable Energy 2024, 220, 119659. [Google Scholar] [CrossRef]
- De Jesus, S. S.; Ferreira, G. F.; Moreira, L. S.; Wolf Maciel, M. R.; Maciel Filho, R. Comparison of several methods for effective lipid extraction from wet microalgae using green solvents . Renewable Energy 143 2019, 130–141. [Google Scholar] [CrossRef]
- Rahman, N.; Hashem, S.; Akther, S.; Jothi, J. S. Impact of various extraction methods on fatty acid profile, physicochemical properties, and nutritional quality index of Pangus fish oil. Food Science & Nutrition 2023, 11(8), 4688–4699. [Google Scholar] [CrossRef]
- David, E.; Niculescu, V. C. Volatile organic compounds (VOCs) as environmental pollutants: occurrence and mitigation using nanomaterials. International journal of environmental research and public health 2021, 18(24), 13147. [Google Scholar] [CrossRef]
- Martins, R.; Barbosa, A.; Advinha, B.; Sales, H.; Pontes, R.; Nunes, J. Green extraction techniques of bioactive compounds: a state-of-the-art review. Processes 2023, 11(8), 2255. [Google Scholar] [CrossRef]
- Méndez, L.; Rodríguez, A.; Aubourg, S. P.; Medina, I. Low-toxicity solvents for the extraction of valuable lipid compounds from octopus (Octopus vulgaris) waste. Foods 2023, 12(19), 3631. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, L.; Ricardo, F.; Melo, T.; Domingues, P.; Domingues, M. R.; Calado, R.; Rey, F. Lipidomics of common octopus'(Octopus vulgaris) arm muscle using untargeted high-resolution liquid chromatography-mass spectrometry. Journal of Food Composition and Analysis 2023, 115, 104871. [Google Scholar] [CrossRef]
- Saini, R. K.; Prasad, P.; Shang, X.; Keum, Y. S. Advances in lipid extraction methods—a review. International Journal of Molecular Sciences 2021, 22(24), 13643. [Google Scholar] [CrossRef]
- Ivanković, A.; Dronjić, A.; Martinović Bevanda, A.; Talić, S. Review of 12 Principles of Green Chemistry in Practice . International Journal of Sustainable and Green Energy 2017, 6(3), 39–48. [Google Scholar] [CrossRef]
- Devi, M.; Moral, R.; Thakuria, S.; Mitra, A.; Paul, S. Hydrophobic deep eutectic solvents as greener substitutes for conventional extraction media: examples and techniques. ACS omega 2023, 8(11), 9702–9728. [Google Scholar] [CrossRef]
- Martel-Martín, S.; Di Pietro, M. E.; Gutiérrez, A.; Aguilar, N.; Bol-Arreba, A.; Aparicio, S.; Mele, A. A paradigm for natural eutectic solvents based on fatty acids: Molecular interactions and toxicological considerations. Journal of Molecular Liquids 2024, 414, 126148. [Google Scholar] [CrossRef]
- Ristivojević, P.; Krstić Ristivojević, M.; Stanković, D.; Cvijetić, I. Advances in extracting bioactive compounds from food and agricultural waste and by-products using natural deep eutectic solvents: A circular economy perspective. Molecules 2024, 29(19), 4717. [Google Scholar] [CrossRef]
- Trusheva, B.; Petkov, H.; Chimshirova, R.; Popova, M.; Dimitrova, L.; Zaharieva, M. M.; Bankova, V. Insight into the influence of natural deep eutectic solvents on the extraction of phenolic compounds from poplar type propolis: Composition and in vitro biological activity. Heliyon 2024, 10(7). [Google Scholar] [CrossRef]
- Makoś, P.; Słupek, E.; Gębicki, J. Hydrophobic deep eutectic solvents in microextraction techniques–A review. Microchemical journal 2020, 152, 104384. [Google Scholar] [CrossRef]
- Trenzado, J. L.; Benito, C.; Atilhan, M.; Aparicio, S. Hydrophobic Deep eutectic Solvents based on cineole and organic acids. Journal of Molecular Liquids 2023, 377, 121322. [Google Scholar] [CrossRef]
- Aguilar, N.; Benito, C.; Martel-Martin, S.; Gutiérrez, A.; Rozas, S.; Marcos, P. A.; Aparicio, S. Insights into Carvone: Fatty Acid Hydrophobic NADES for Alkane Solubilization. Energy & Fuels 2024, 38(24), 23633–23653. [Google Scholar] [CrossRef]
- Rozas, S.; Zamora, L.; Benito, C.; Atilhan, M.; Aparicio, S. A study on monoterpenoid-based natural deep eutectic solvents. Green Chemical Engineering 2023, 4(1), 99–114. [Google Scholar] [CrossRef]
- Anstiss, L.; Weber, C. C.; Baroutian, S.; Shahbaz, K. Menthol-based deep eutectic solvents as green extractants for the isolation of omega-3 polyunsaturated fatty acids from Perna canaliculus. Journal of Chemical Technology & Biotechnology 2023, 98(7), 1791–1802. [Google Scholar] [CrossRef]
- Topal, T.; Card, A.; Mackenzie, A. D.; Lagutin, K.; Marshall, S. N.; Cumming, A. H.; Killeen, D. P. Hydrophobic natural deep eutectic solvents for marine lipid extraction. Journal of the American Oil Chemists' Society 2024, 101(3), 361–367. [Google Scholar] [CrossRef]
- Strieder, M. M.; Bragagnolo, F. S.; Mendiola, J. A.; Rostagno, M. A.; Ibáñez, E. Screening and characterization of 1, 8-cineole-based solvents as an alternative to hexane for obtaining nonpolar compounds from plant-based milk coproducts. ACS Sustainable Chemistry & Engineering 2024, 12(43), 16052–16063. [Google Scholar] [CrossRef]
- Kuo, C. H.; Liao, H. Z.; Wang, Y. H.; Wang, H. M. D.; Shieh, C. J.; Tseng, C. Y. Highly efficient extraction of EPA/DHA-enriched oil from cobia liver using homogenization plus sonication. European Journal of Lipid Science and Technology 2017, 119(10), 1600466. [Google Scholar] [CrossRef]
- Ramírez-Sucre, M. O.; Avilés-Betanzos, K. A.; López-Martínez, A.; Rodríguez-Buenfil, I. M. Evaluation of polyphenol profile from citrus peel obtained by natural deep eutectic solvent/ultrasound extraction. Processes 2024, 12(10), 2072. [Google Scholar] [CrossRef]
- Ramírez-Sucre, M. O.; Baigts-Allende, D. K. Efecto del tratamiento térmico en el comportamiento reológico de salsas de chile habanero (Capsicum chinense) adicionadas con gomas guar y xantana. Agrociencia 2016, 50(7), 837–847. Available online: https://www.scielo.org.mx/scielo.php?pid=S1405-31952016000700837&script=sci_arttext.
- Abo-Elwafa, G. A.; Hashim, A. F.; Afifi, S. M.; Ahmed, M.; Youssef, A. M. Simplified visual colorimetric method for edible oil oxidation detection using Agarose-Polyvinyl alcohol (AGR/PVA) colorimetric biofilms. Journal of Food Measurement and Characterization 2025, 1–17. [Google Scholar] [CrossRef]
- Oliveira, C.; Santos, J.; Sousa, R. G. UV Spectrophotometry applied to the quantification of omega-3, -6 and -9 in fresh tissues of wild and farmed tambaqui. International Journal for Innovation Education and Research 2020, 8(12), 183–195. [Google Scholar] [CrossRef]
- Avilés-Betanzos, K. A.; Cauich-Rodríguez, J. V.; Ramírez-Sucre, M. O.; Rodríguez-Buenfil, I. M. Optimization of Spray Drying Conditions for a Capsicum chinense Leaf Extract Rich in Polyphenols Obtained by Ultrasonic Probe/NADES. ChemEngineering 2024, 8(6). [Google Scholar] [CrossRef]
- Pech-Pisté, R.; Pérez-Aranda, C.; Balam, A.; Vargas-Coronado, R.; Cauich-Rodríguez, J. V.; Avilés, F. Piezoimpedance of carbon nanotube yarns coupled with Raman spectroscopy and its implementation for sensing polymerization kinetics. Carbon 2023, 213, 118246. [Google Scholar] [CrossRef]
- Adeoye, D. O.; Gano, Z. S.; Ahmed, O. U.; Shuwa, S. M.; Atta, A. Y.; Iwarere, S. A.; Daramola, M. O. Synthesis and characterisation of menthol-based hydrophobic deep eutectic solvents. Chemistry Proceedings 2023, 14(1), 98. [Google Scholar] [CrossRef]
- Dabbagh Hosseini Pour, M.; Jahanbin Sardroodi, J.; Hadidi, N.; Pazuki, G. Molecular dynamics and COSMO-RS model of menthol–fatty acid deep eutectic solvents: thermodynamic, structural, and dynamics insights. Scientific Reports 2025, 15(1), 41849. [Google Scholar] [CrossRef]
- Guruk, M.; Fickers, P.; Selli, S.; Erten, H. Investigation of the antioxidant effect of two thiols,γ-glutamyl cysteine and glutathione, in sunflower oil under accelerated storage. Authorea Preprints 2023. [Google Scholar] [CrossRef]
- Rojas, M. L.; Kubo, M. T.; Caetano-Silva, M. E.; Augusto, P. E. Ultrasound processing of fruits and vegetables, structural modification and impact on nutrient and bioactive compounds: a review. International Journal of Food Science and Technology 2021, 56(9), 4376–4395. [Google Scholar] [CrossRef]
- Hoch, C. C.; Petry, J.; Griesbaum, L.; Weiser, T.; Werner, K.; Ploch, M.; Wollenberg, B. 1, 8-cineole (eucalyptol): A versatile phytochemical with therapeutic applications across multiple diseases. Biomedicine & Pharmacotherapy 2023, 167, 115467. [Google Scholar] [CrossRef]
- Cherniakova, M.; Varchenko, V.; Belikov, K. Menthol-based (deep) eutectic solvents: a review on properties and application in extraction. The Chemical Record 2024, 24(2), e202300267. [Google Scholar] [CrossRef]
- Bagović Kolić, M.; Železnjak, M.; Markov, K.; Gaurina Srček, V.; Cvjetko Bubalo, M.; Radošević, K.; Radojčić Redovniković, I. Physicochemical and Biological Properties of Menthol and Thymol-Based Natural Deep Eutectic Solvents. Molecules 2025, 30(8), 1713. [Google Scholar] [CrossRef]
- Nandiyanto, A. B. D.; Oktiani, R.; Ragadhita, R. How to read and interpret FTIR spectroscope of organic material. Indonesian journal of science and technology 2019, 4(1), 97–118. [Google Scholar] [CrossRef]
- Vongsvivut, J.; Miller, M. R.; McNaughton, D.; Heraud, P.; Barrow, C. J. Rapid discrimination and determination of polyunsaturated fatty acid composition in marine oils by FTIR spectroscopy and multivariate data analysis. Food and bioprocess technology 2014, 7(8), 2410–2422. [Google Scholar] [CrossRef]
- Ahmmed, F.; Killeen, D. P.; Gordon, K. C.; Fraser-Miller, S. J. Rapid quantitation of adulterants in premium marine oils by Raman and IR spectroscopy: A data fusion approach. Molecules 2022, 27(14), 4534. [Google Scholar] [CrossRef] [PubMed]
- Ahmmed, F.; Gordon, K. C.; Killeen, D. P.; Fraser-Miller, S. J. Detection and quantification of adulteration in krill oil with Raman and infrared spectroscopic methods. Molecules 2023, 28(9), 3695. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, G. D. Saturated Fats: Time to Assess Their Beneficial Role in a Healthful Diet. Dietetics 2024, 3(4), 452–462. [Google Scholar] [CrossRef]
- Gullian-Klanian, M.; Terrats-Preciat, M.; Pech-Jiménez, E. C.; Cutz De Ocampo, J. Effect of frozen storage on protein denaturation and fatty acids profile of the red octopus (Octopus maya). Journal of Food Processing and Preservation 2017, 41(4), e13072. [Google Scholar] [CrossRef]
- Swetha, N.; Mathanghi, S. K. Towards sustainable omega-3 fatty acids production–A comprehensive review on extraction methods, oxidative stability and bio-availability enhancement. Food Chemistry Advances 2024, 4, 100603. [Google Scholar] [CrossRef]
- Rohman, A.; Irnawati; Windarsih, A.; Riswanto, F. D. O.; Indrayanto, G.; Fadzillah, N. A.; Bakar, N. K. A. Application of chromatographic and spectroscopic-based methods for analysis of Omega-3 (Ω-3 Fas) and Omega-6 (Ω-6 Fas) fatty acids in marine natural products. Molecules 2023, 28(14), 5524. [Google Scholar] [CrossRef]
- García-García, P.; Ospina, M.; Señoráns, F. J. Tisochrysis lutea as a source of omega-3 polar lipids and fucoxanthin: Extraction and characterization using green solvents and advanced techniques. Journal of Applied Phycology 2024, 36(4), 1697–1708. [Google Scholar] [CrossRef]
 |
 |
| Response Variables | P value | ||
| Individual factors | Double interactions | ||
| HBA | MR | HBA x MR | |
| C16:0 | <0.001 | 0.444 | 0.444 |
| C18:0 | <0.001 | 0.034 | 0.092 |
| C18:1 n-9 | <0.001 | 0.012 | 0.012 |
| C18:2 n-6 | <0.001 | 0.003 | 0.005 |
| C18:3 n-3 | 0.005 | 0.050 | 0.028 |
| ω – 3 | 0.005 | 0.050 | 0.028 |
| ω – 6 | <0.001 | 0.003 | 0.005 |
| ω – 9 | <0.001 | 0.012 | 0.012 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




