Submitted:
19 March 2026
Posted:
20 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
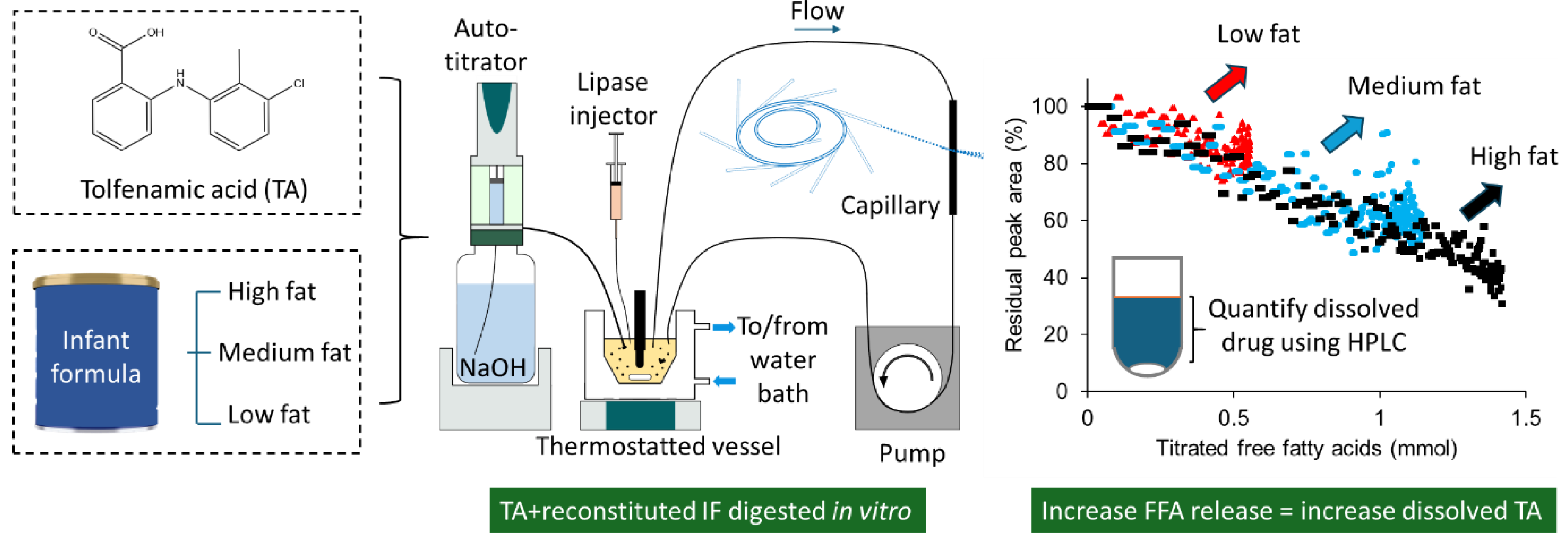
2.2. In Vitro Digestion of Reconstituted Infant Formula Containing Tolfenamic acid
2.3. Quantifying Dissolved Tolfenamic Acid in Samples Retrieved During the Digestion of Reconstituted Infant Formula Using HPLC
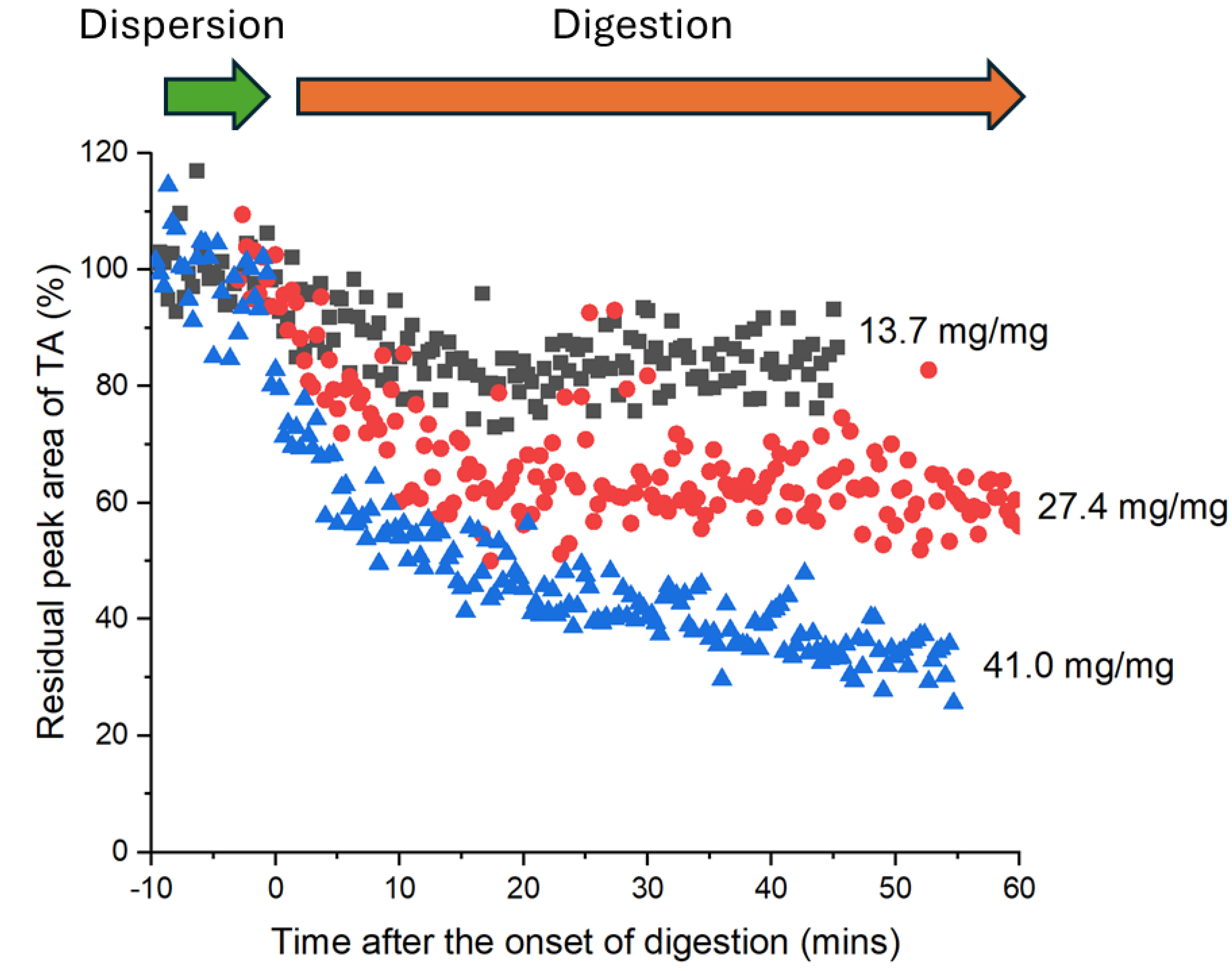
2.4. Tracking the Dissolution of Tolfenamic Acid During the Digestion of Reconstituted Infant Formula Using Small-Angle X-Ray Scattering
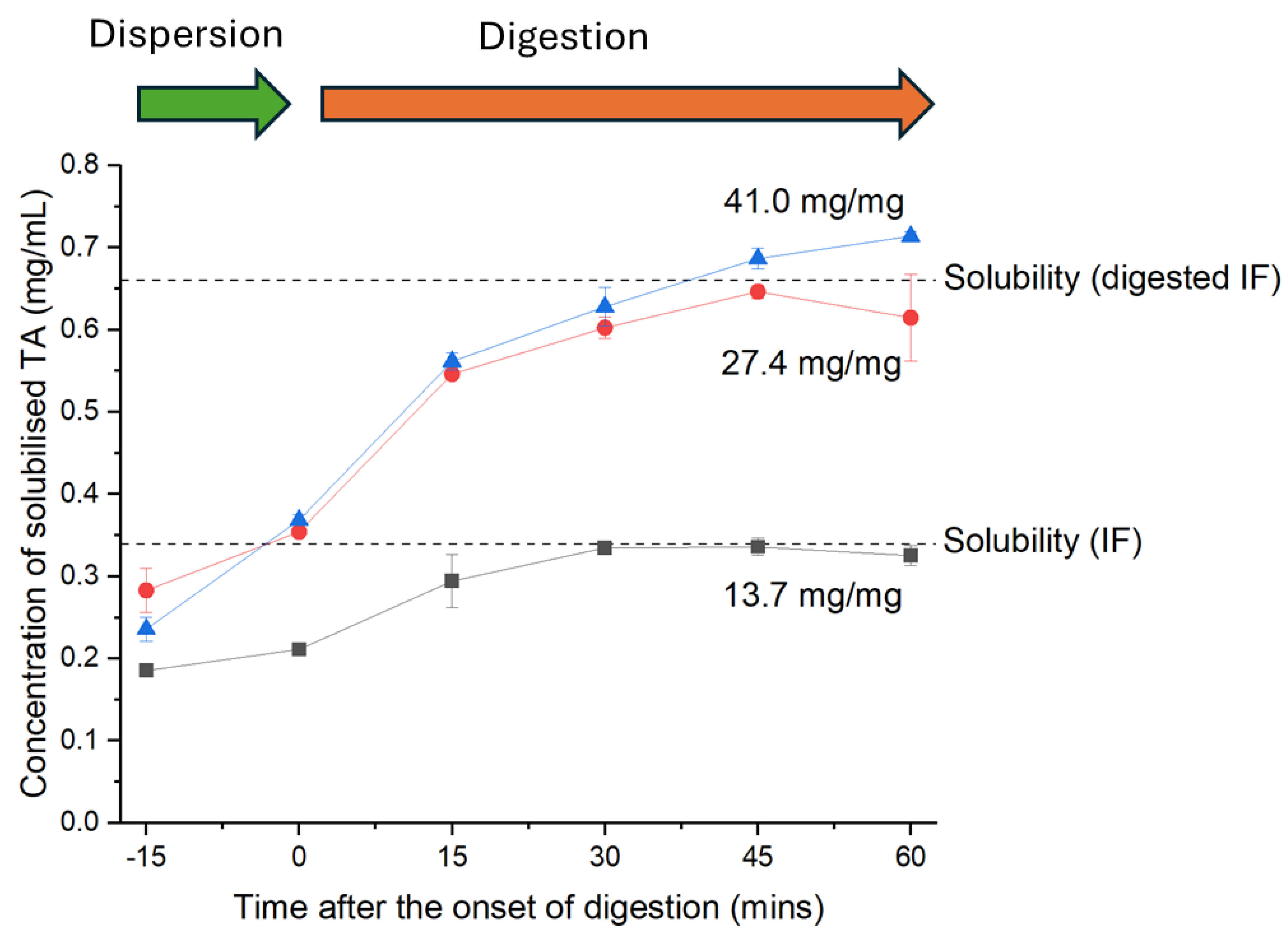
2.5. Measuring the Equilibrium Solubility of Tolfenamic Acid in Reconstituted Infant Formula
3. Results
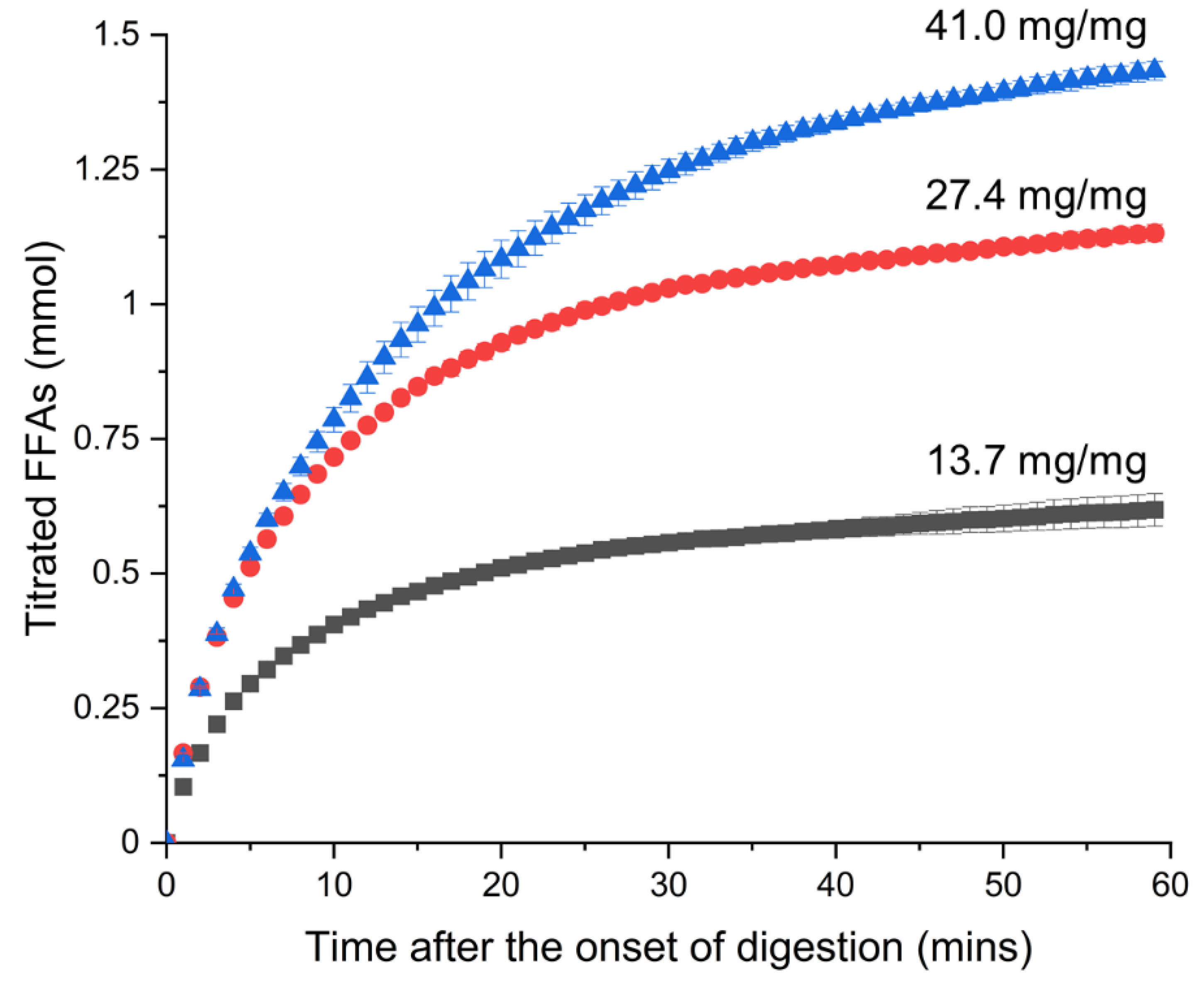
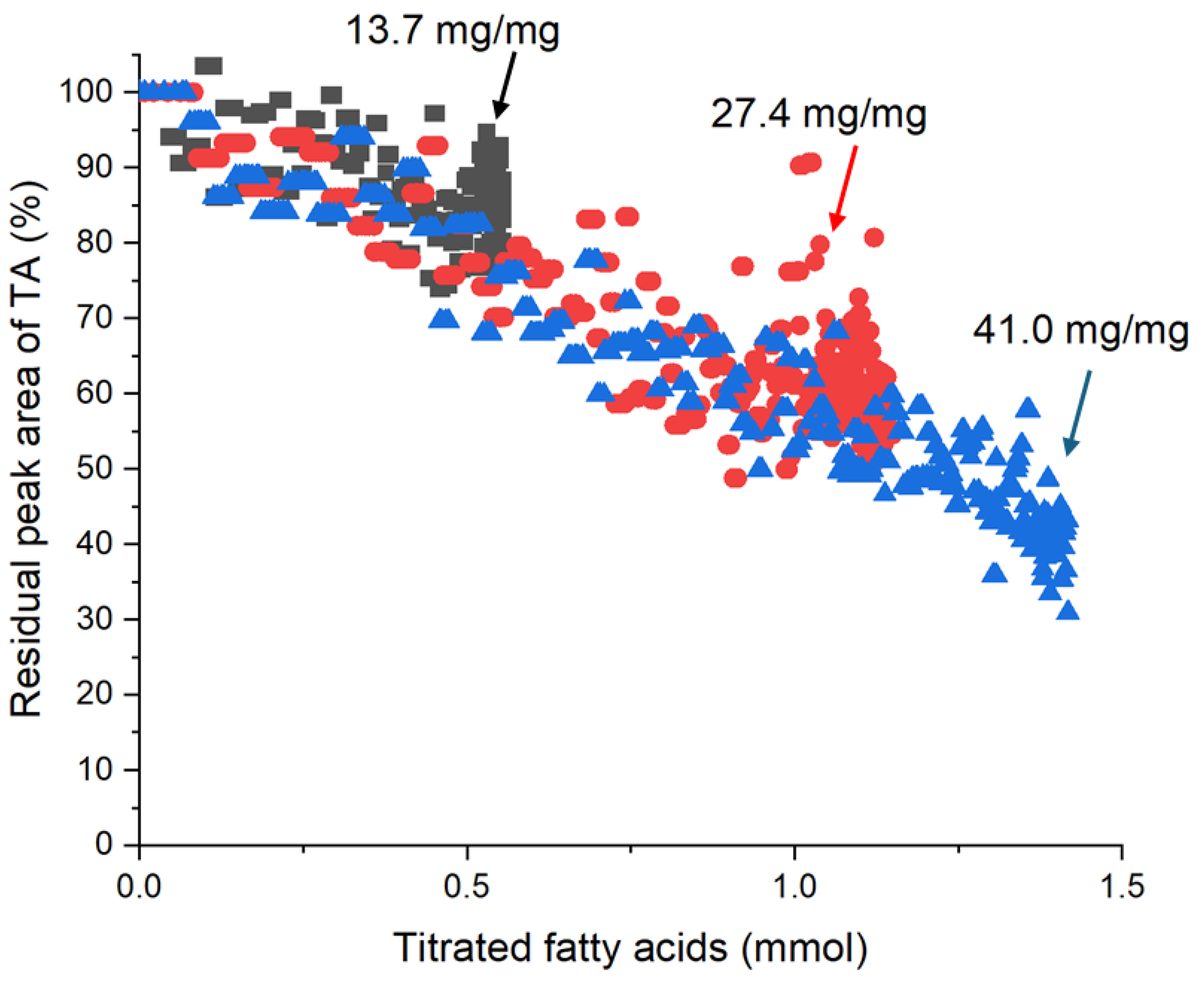
3.1. Kinetics of Lipid Digestion in Reconstituted Infant Formula
3.2. Solubility and Dissolution of Tolfenamic Acid in Digested and Undigested Reconstituted Infant Formula
4. Discussion
5. Conclusions
Supplementary Materials
Data Availability Statement
Acknowledgments
References
- Porter, C. J. H.; Trevaskis, N. L.; Charman, W. N. Nat. Rev. Drug Discov. 2007, 6, 231–248. [CrossRef]
- Pham, A. C.; Clulow, A. J.; Boyd, B. J. Front. Cell Dev. Biol. 2021, 9, 657886. [CrossRef]
- Salim, M.; MacGibbon, A. K. H.; Nowell, C. J.; Clulow, A. J.; Boyd, B. J. Colloids Interfaces 2023, 7, 56. [CrossRef]
- Clulow, A. J.; Salim, M.; Hawley, A.; Boyd, B. J. Chem. Phys. Lipids 2018, 211, 107–116. [CrossRef]
- Iurian, S.; Bogdan, C.; Suciu, Ș.; Muntean, D.-M.; Rus, L.; Berindeie, M.; Bodi, S.; Ambrus, R.; Tomuță, I. Pharmaceutics 2022, 14, 1342. [CrossRef] [PubMed]
- Coldani, M. E.; Palugan, L.; Foppoli, A.; Cerea, M.; Pinto, J. F. Int. J. Pharm. 2024, 666, 124836. [CrossRef] [PubMed]
- Salim, M.; Eason, T.; Boyd, B. J. Adv. Drug Deliv. Rev. 2022, 183, 114139. [CrossRef]
- Boyd, B. J.; Salim, M.; Clulow, A. J.; Ramirez, G.; Pham, A. C.; Hawley, A.J. Control. Release 2018, 292, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Larsen, A. T.; Holm, R.; Müllertz, A. Eur. J. Pharm. Biopharm. 2017, 117, 308–314. [CrossRef]
- Salim, M.; Fraser-Miller, S. J.; Be̅rziņš, K. r.; Sutton, J. J.; Ramirez, G.; Clulow, A. J.; Hawley, A.; Beilles, S.; Gordon, K. C.; Boyd, B. J. Mol. Pharm. 2020, 17, 885–899. [CrossRef]
- Salim, M.; Ramirez, G.; Clulow, A. J.; Zhang, Y.; Ristroph, K. D.; Feng, J.; McManus, S. A.; Hawley, A.; Prud’homme, R. K.; Boyd, B. J. Mol. Pharm. 2019, 16, 2755–2765. [CrossRef]
- Eason, T.; Ponsonby-Thomas, E.; Pham, A. C.; Huang, S.; Offersen, S. M.; Thymann, T.; Zann, V.; Salim, M.; Boyd, B. J. Eur. J. Pharm. Biopharm. 2026, 220, 114979. [CrossRef]
- Salim, M.; Khan, J.; Ramirez, G.; Murshed, M.; Clulow, A. J.; Hawley, A.; Ramachandruni, H.; Beilles, S.; Boyd, B. J. Mol. Pharm. 2019, 16, 1658–1668. [CrossRef]
- Alskär, L. C.; Keemink, J.; Johannesson, J.; Porter, C. J. H.; Bergström, C. A. S. Mol. Pharm. 2018, 15, 4733–4744. [CrossRef]
- Sacchi, P.; Reutzel-Edens, S. M.; Cruz-Cabeza, A. J. CrystEngComm. 2021, 23, 3636–3647. [CrossRef]
- Williams, H. D.; Sassene, P.; Kleberg, K.; Calderone, M.; Igonin, A.; Jule, E.; Vertommen, J.; Blundell, R.; Benameur, H.; Müllertz, A.; Porter, C. J. H.; Pouton, C. W. J. Pharm. Sci. 2014, 103, 2441–2455. [CrossRef]
- Khan, J.; Rades, T.; Boyd, B. J. J. Pharm. Sci. 2018, 107, 2420–2427. [CrossRef]
- Eason, T.; Ramirez, G.; Clulow, A. J.; Salim, M.; Boyd, B. J. Pharmaceutics 2022, 14, 2228. [CrossRef]
- Kirby, N. M.; Mudie, S. T.; Hawley, A. M.; Cookson, D. J.; Mertens, H. D. T.; Cowieson, N.; Samardzic-Boban, V. J. Appl. Cryst. 2013, 46, 1670–1680. [CrossRef]
- Pouton, C. W.; Porter, C. J. Adv. Drug Deliv. Rev. 2008, 60, 625–637. [CrossRef]
- Cuiné, J. F.; Charman, W. N.; Pouton, C. W.; Edwards, G. A.; Porter, C. J. H. Pharm. Res. 2007, 24, 748–757. [CrossRef]
- Ejskjær, L.; Kimpe, K.; Bohets, H.; Bevernage, J.; Griffin, B. T.; O’Dwyer, P. J. J. Pharm. Sci. 2025, 114, 103911. [CrossRef]
- Suys, E. J. A.; Chalmers, D. K.; Pouton, C. W.; Porter, C. J. H. Mol. Pharm. 2018, 15, 2355–2371. [CrossRef]
- Khan, J.; Rades, T.; Boyd, B. J. Mol. Pharm. 2016, 13, 3783–3793. [CrossRef]
- Khan, J.; Rades, T.; Boyd, B. J. J. Pharm. Sci. 2018, 107, 2420–2427. [CrossRef]
- Siqueira Jørgensen, S.; Rades, T.; Mu, H.; Graeser, K.; Müllertz, A. Acta Pharm Sin B 2019, 9, 194–201. [CrossRef]
- Huang, S.; Pham, A. C.; Salim, M.; Eason, T.; Ramirez, G.; Boyd, B. J. Int. J. Pharm. 2024, 660, 124257. [CrossRef]
- Salim, M.; Ramirez, G.; Peng, K.-Y.; Clulow, A. J.; Hawley, A.; Ramachandruni, H.; Beilles, S.; Boyd, B. J. Mol. Pharm. 2020, 17, 2749–2759. [CrossRef]
- Koehl, N. J.; Holm, R.; Kuentz, M.; Griffin, B. T. Pharm. Res. 2020, 37, 124. [CrossRef]
- Alskär, L. C.; Porter, C. J. H.; Bergström, C. A. S. Mol. Pharm. 2016, 13, 251–261. [CrossRef]
- Fagerberg, J. H.; Karlsson, E.; Ulander, J.; Hanisch, G.; Bergström, C. A. Pharm. Res. 2015, 32, 578–589. [CrossRef]
- Salim, 29. M.; Fraser-Miller, S. J.; Sutton, J. J.; Be̅rziņš, K. r.; Hawley, A.; Clulow, A. J.; Beilles, S.; Gordon, K. C.; Boyd, B. J. J. Phys. Chem. Lett. 2019, 10, 2258–2263. [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



