Submitted:
18 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
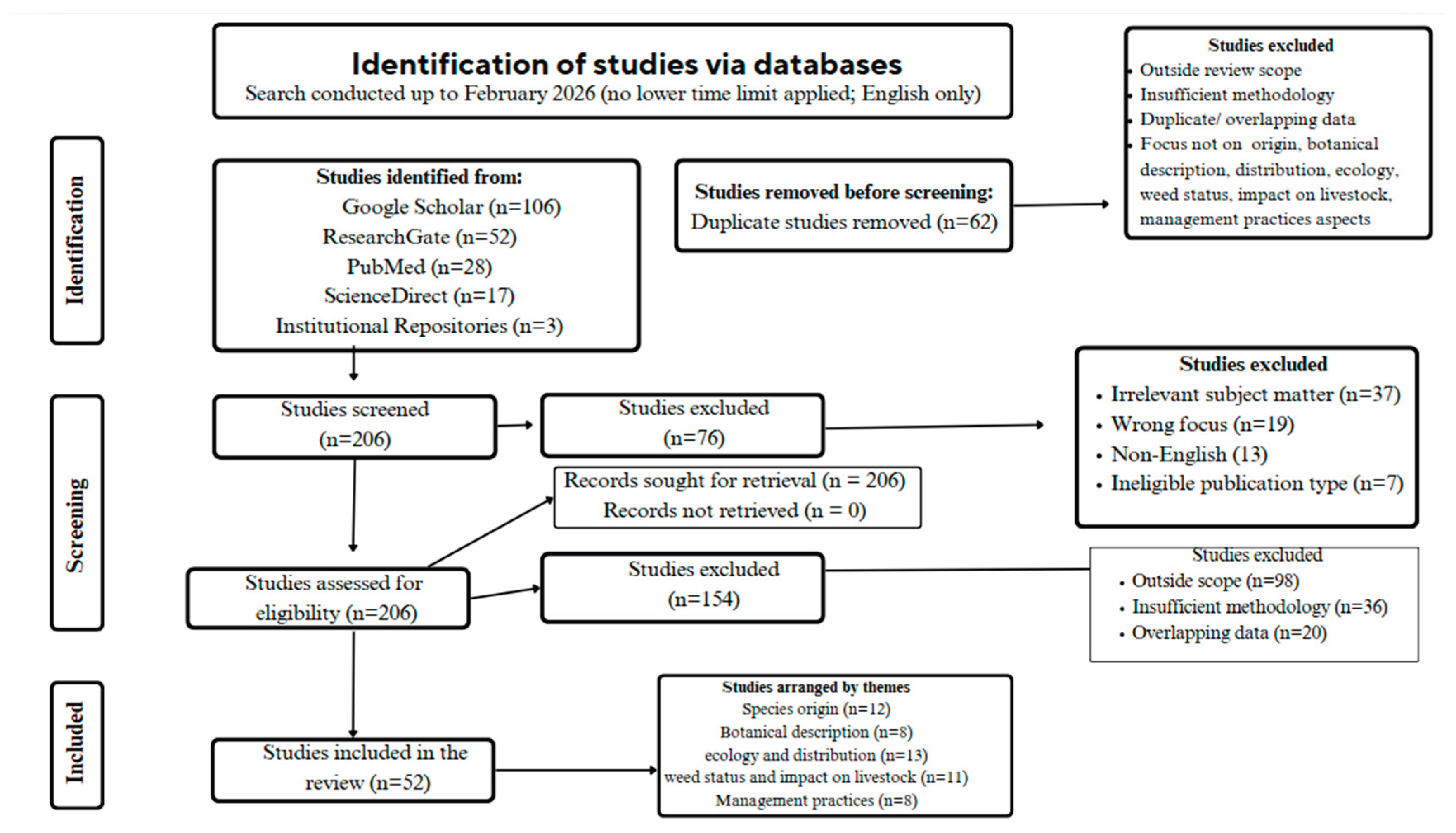
2. Materials and Methods
3. History and Origin of Johnsongrass
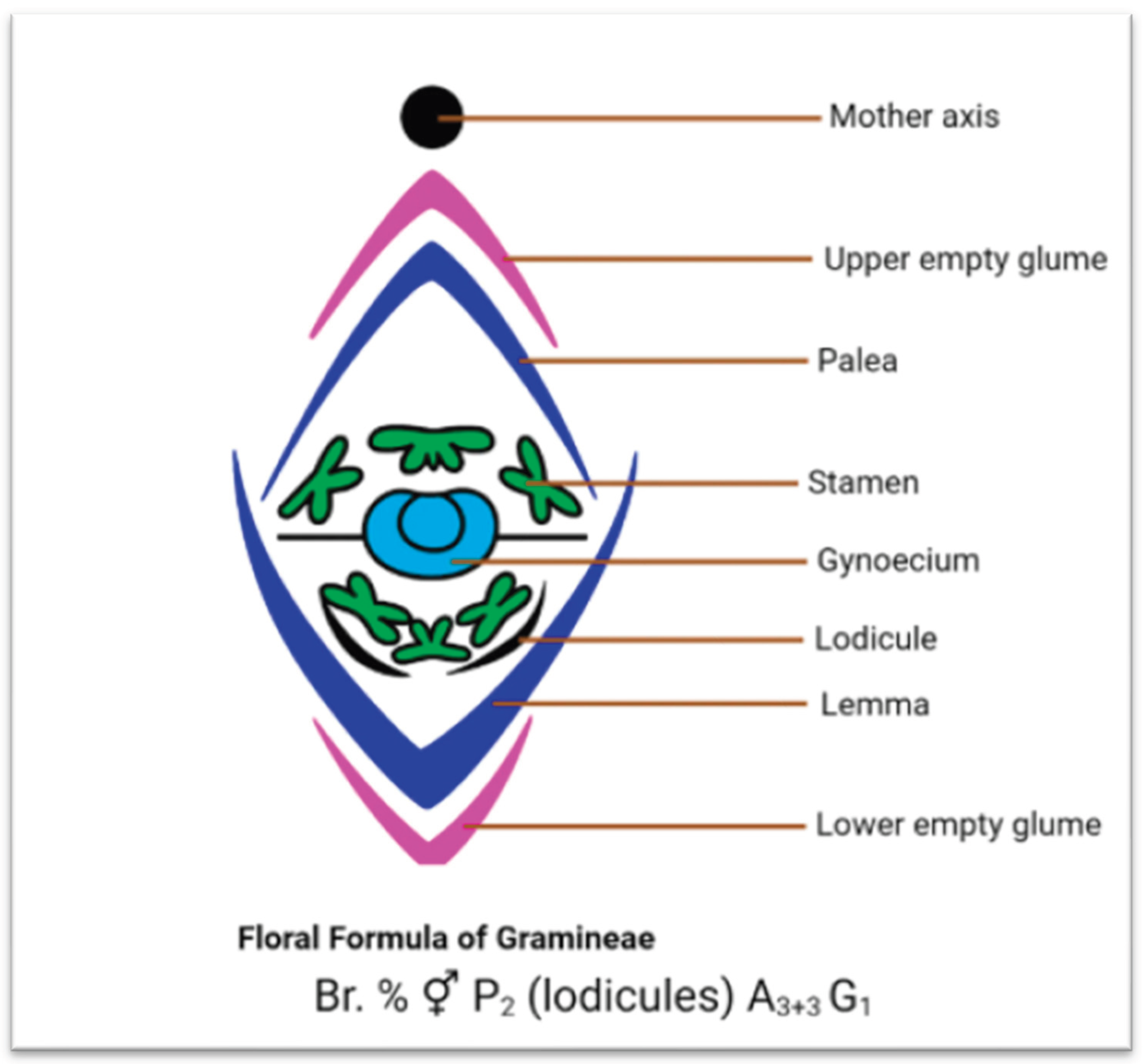
4. Botanical Classification and Description of Johnsongrass
5. Distribution and Spread of Johnson Grass (S. halepense)
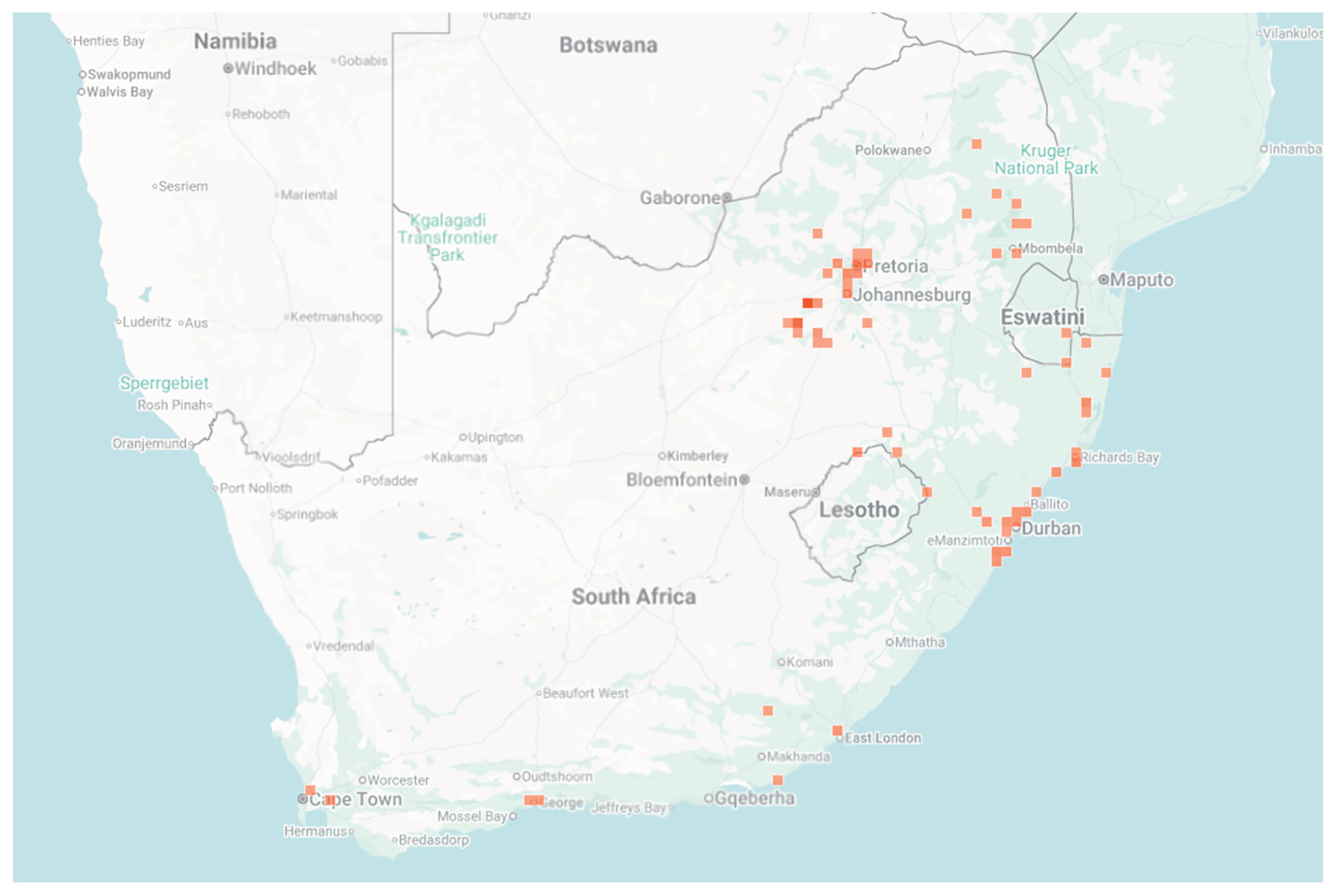
6. Ecological Invasion and Impact of Johnsongrass (S. halepense) on Rangeland Biodiversity
7. Impact of Johnsongrass (S. halepense) on Livestock
8. Management and Control of Johnson Grass
8.1. Mechanical Control Method
8.2. Integrated Methods (Ploughing and Burning)
8.3. Chemical Control Method
9. Conclusion
Acknowledgments
Conflicts of Interest
References
- Eberlein, C.V.; Miller, T.L.; Luryey, E.L. Seasonal emergence and growth of Sorghum almum. Weed Tech 1988, 2, 275–281. [Google Scholar] [CrossRef]
- Schantz, M.C., 2025. Sorghum halepense (Johnsongrass): a review of the invasion, management, and spread in the changing climate of the Southern Great Plains. Weed Science, pp.1-22. pp. 1–22.
- McWhorter, C.G. Introduction and spread of Johnsongrass in the United States. Weed Science 1971, 19, 496–501. [Google Scholar] [CrossRef]
- Masood, P.A. 2017. Eco-biology, impact, and management of Sorghum helepense (L) Pers. Biological invasions.
- Ryder, N. Transcriptome assembly and annotation of Johnsongrass (Sorghum helepense) rhizomes identify candidate rhizome-specific genes. Plant Direct 2018, 2, e00065. [Google Scholar] [CrossRef]
- Overpeck, J.K.; Bartlein, P.J.; Webb, T. Potential magnitude of future vegetation change in eastern north America: Comparisons with the past. Science 1991, 254, 692–705. [Google Scholar] [CrossRef]
- Kelly, S.; Fletcher, R.A.; Barney, J.N. Intraspecific, ecotypic and home climate variation in photosynthetic traits of the widespread invasive grass Johngrass. AoB plants 2020, 12–3. [Google Scholar]
- Yim, K.O.; Bayer, D.E. Rhizome expression in a selected cross in the Sorghum genus. Euphytica 1997, 94, 6. [Google Scholar] [CrossRef]
- Coiner, H; A. Hayhoe, K.; Ziska, L.H.; Van Dorn, J.; Sage, R.F. Tolerance of subzero winter cold in kudzu (Pueraria var. lobata). Oecolgia 2018, 187, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Hariprasanna, K.; Patil, J.V. Sorghum: origin, classification, biology, and improvement. In Sorghum molecular breeding; Springer India: New Delhi, 2015; pp. 3–20. [Google Scholar]
- Bednarova, M.; Karafiatova, M.; Hribova, E.; Bartos, J. B chromosomes in genus Sorghum (Poaceae). Plants 2021, 10(3), 505. [Google Scholar] [CrossRef] [PubMed]
- Garber, E.D. Cytotaxanomic studies in the genus sorghum. University California Publication Botany 1950, 23, 283–362. [Google Scholar]
- Lazarides, M.; Hacker, J.B.; Andrew, M.H. Taxonomy, cytology, and ecology of indigenous Australian sorghum (Sorghum Moench: Andropogoneae: Poaceae). Australian Systematic Botany 1991, 4, 591–635. [Google Scholar] [CrossRef]
- Gamashe, S.S.; Tayade, N.; Ganapathy, K.N. Sorghum. In Plant Genebank Utilization for Trait Discovery in Milletps: Volume IV; Springer Nature Singapore: Singapore, 2025; pp. 43–76. [Google Scholar]
- Sias, C. Understanding Interspecific Hybridization Between Sorghum bicolor and Its Weedy Congener, S. halepense. Doctoral dissertation, 2020. [Google Scholar]
- Gui, H.; Jiao, Y.; Tan, X.; Wang, X.; Huang, X.; Paterson, A.H. Duplication and genetic innovation in cereal genomes. Genome Res 2019, 29, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Peterson, A.H.; Kong, W.; Johnston, R.M.; Nabukalu, P.; Wu, G.; Poehlman Sin, B.; Kadıoğlu, İ.; Yıldırım, C. Determination of some biological properties of the seeds of the Johnson grass (Sorghum halepense (L.) Pers.). Journal of Agricultural Biotechnology 2024, 5(2), 71–78. [Google Scholar] [CrossRef]
- Khan, M.N.; Ali, S.; Yaseen, T.; Ullah, S.; Zaman, A.; Iqbal, M.; Shah, S. Eco-taxonomic study of family Poaceae (Gramineae). RADS Journal of Biological Research and Applied Sciences 2019, 10(2), 63–75. [Google Scholar] [CrossRef]
- Xu, Z. and Zhou, G., 2017. Gramineae. In Identification and Control of Common Weeds: Volume 1 (pp. 3-364). Dordrecht: Springer Netherlands.
- Liang, Z.; Shi, S.; Xue, B.; Li, D.; Liu, Y.; Liu, C. Comparative analysis of TALE gene family in Gramineae. Agronomy 2025, 15(6), 14–60. [Google Scholar] [CrossRef]
- Klein, P.; Smith, C.M. Invasive Johnsongrass, a threat to native grasslands and agriculture. Biologia 2021, 76(2), 413–420. [Google Scholar] [CrossRef]
- Warwick, S.I.; Black, L.D. The biology of Canadian weeds.: 61. Sorghum halepense (L.) Pers. Canadian Journal of Plant Science 1983, 63(4), 997–1014. [Google Scholar] [CrossRef]
- Nestorović Živković, J.; Simonović, M.; Mišić, D.; Nešić, M.; Jovanović, V.; Gašić, U.; Bjedov, I.; Dmitrović, S. Bioherbicidal Evaluation of Methanol Extract of Sorghum halepense L. Rhizome and Its Bioactive Components Against Selected Weed Species. Molecules 2025, 15, 30–60. [Google Scholar] [CrossRef]
- Kigel, J.; Rubin, B. Sorghum halepense. In Handbook of Flowering; CRC Press, 2019; pp. 376–379. [Google Scholar]
- Rout, M.E.; Chrzanowski, T.H.; Smith, W.K.; Gough, L. Ecological impacts of the invasive grass Sorghum halepense on native tallgrass prairie. Biological invasions 2013, 15(2), 327–339. [Google Scholar] [CrossRef]
- Giantin, S.; Franzin, A.; Brusa, F.; Montemurro, V.; Bozzetta, E.; Caprai, E.; Fedrizzi, G.; Girolami, F.; Nebbia, C. Overview of cyanide poisoning in cattle from Sorghum halepense and S. bicolor cultivars in Northwest Italy. Animals 2024, 14(5), 743. [Google Scholar] [CrossRef] [PubMed]
- iNaturalist. 2026. https://www.inaturalist.org/observations?taxon_id=58387.
- Taylor, J.R., 2019. Sorghum and millets: Taxonomy, history, distribution, and production. In Sorghum and millets (pp. 1-21). AACC International Press.
- edayetullah, M. and Zaman, P., 2018. Johnson Grass (aleppo Grass) Md. Hedayetullah And Parveen Zaman. In Forage Crops of the World, Volume I: Major Forage Crops (pp. 99-110). Apple Academic Press.
- Linder, H.P.; Lehmann, C.E.; Archibald, S.; Osborne, C.P.; Richardson, D.M. Global grass (Poaceae) success underpinned by traits facilitating colonization, persistence, and habitat transformation. Biological Reviews 2018, 93(2), 1125–1144. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, L.; Wangdi, K. Country pasture/forage resource profiles; FAO: Rome, Italy, 2006. [Google Scholar]
- Wood, A.R.; Den Breeÿen, A. Plant pathogens and biological control of invasive alien plants in South Africa: a review of projects and progress (2011–2020). African Entomology 2021, 29(3), 983–1004. [Google Scholar] [CrossRef]
- O’Connor, T.G.; van Wilgen, B.W. The impact of invasive alien plants on rangelands in South Africa. In Biological Invasions in South Africa; Springer International Publishing: Cham, 2020; pp. 459–487. [Google Scholar]
- Friedman, J. The evolution of annual and perennial plant life histories: ecological correlates and genetic mechanisms. Annual Review of Ecology, Evolution, and Systematics 2020, 51(1), 461–481. [Google Scholar] [CrossRef]
- Mordecai, E.A.; Molinari, N.A.; Stahlheber, K.A.; Gross, K.; D'Antonio, C. Controls over native perennial grass exclusion and persistence in California grasslands invaded by annuals. Ecology 2015, 96(10), 2643–2652. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, T.E.; Parveen, S.G.; Aruna, C.; Hariprasanna, K.; Singh, S.P.; Singh, A.K.; Joshi, D.C.; Joshi, P.; Tomar, S.M.S.; Talukdar, A.; Kumar, S. Millets and pseudocereals: A treasure for climate resilient agriculture ensuring food and nutrition security. Indian Journal of Genetics and Plant Breeding (The) 2024, 84(1), 1–37. [Google Scholar] [CrossRef]
- Yazlik, A.; Üremiş, İ. Impact of Sorghum halepense (L.) Pers. on the species richness in native range. Phytoparasitica 2022, 50(5), 1107–1122. [Google Scholar] [CrossRef]
- Bennett H (1973) Johnsongrass, dallisgrass, and other grasses for the humid south. Pages 333–343 in Heath ME, Metcalfe DS, Barnes RF, eds. Forages: The Science of Grassland Agriculture. 3rd edition. Ames: Iowa State University press.
- Hawkins,G.E., Kelley,W., and Smith, L. 1958. Comparison of starr Millet, Sweet, Sudangrass, Johngrass as Dairy Forages. Auburn: Alabama Agricultural Experiment Station Leaflet Circular 60.4 p.
- Recatali, A., and Manuchehri. 2017. Johmgrass in Pastures: Weed or Forage? PSS-2598.
- Watson, V.H.; Coats, R.E.; Kimbrough, L.E. Johnsongrass as a forage in Mississippi. Mississippi State University Bulletin 1980, 886, 1–6. [Google Scholar]
- Andrae, J. Grazing impacts on pasture composition. UGA Cooperative Extension Bulletin 2009, 1243, 1–6. [Google Scholar]
- Stichler, C.; Reagor, J.C. Nitrate and prussic acid poisoning. 2024. [Google Scholar]
- Gensa, U., 2019. Review on cyanide poisoning in ruminants. Journal of Biology and Agricultural Health, pp, 9-6.
- Amjadian, O.A.; Arji, I.; Changizi, M.; Khaghani, S.; Salehi, H.R. Determination of cyanogenic glycosides in endemic species of wild almond seeds in the Zagros Mountains. Brazilian Journal of Botany 2020, 43(4), 697–704. [Google Scholar] [CrossRef]
- Baeza, M.; Luis, D.; Raventos, J.; Escarre, A. Factors influencing fire behaviour in shrublands of different stand ages and the implications for using prescribed burning to reduce wildfire risk. Journal of Environmental Management 2002, 65, 199–208. [Google Scholar] [CrossRef]
- Ceseki, A., Al-Khatib, K., Dahlberg, J.A. 2017. Biology and management of Johnson grass (Sorghum halepense). ANNR Publication 8569. Davis: University of California Agriculture and Natural Resources. 11p.
- Cox, S.; Nabukalu, P.; Paterson, A.H.; Kong, W.; Nakasagga, S. Development of Perennial Grain Sorghum. Sustainability 2018, 10, 172–300. [Google Scholar] [CrossRef]
- Warwick, S.; Black, L. The biology of Canadian weeds: 61. Sorghum Halepense (L.) Pers. Canadian Journal of Plant Science 1983, 63, 997–1014. [Google Scholar] [CrossRef]
- Johnson, W.G.; Wait, J.D. Johnsongrass control, total nonstructural carboydrates in rhizomes, and regrowth after application of hervbicide-resistant corn (Zea mays). Weed Technology 2003, 17, 36–41. [Google Scholar] [CrossRef]
- McCulough, P.; Shilling, D. Johnsongrass control in pastures, roadsides, and noncropland areas. UGA Cooperative Extension bulletin 2022, 15(13), 1–4. [Google Scholar]
- Emendack, Y.; Sanchez, J.; Laza, H. Dhurrin: a potential endogenous nitrogen turnover source for early seedling growth in sorghum. Frontiers in Plant Science 2025, 16, 155–871. [Google Scholar] [CrossRef]
- Vinall, H. A study of the literature concerning poisoning of cattle by the prussic acid in sorghum, Sudan grass and Johnson grass. Journal of American Society of Agronomy 1921, 13, 267–280. [Google Scholar] [CrossRef]
- Morell, P.L.; Williams-Coplin, T.D.; Lattu, A.L.; Browsers, J.E.; Chandler, J.M.; Paterson, A.H. Crop to weed introgression has impacted allelic composition of Johnsongrass populations with and without recent exposure to caltivated sorghum. Molecular Ecology 2005, 14(7), 2143–2154. [Google Scholar] [CrossRef] [PubMed]
- Sankaran, S., Rajasekaran, M.P., Govindaraj, V., Sowmiya, P., Shiny-Rebekka, S. and Kaleeswaran, B., 2020, July. Acute Cyanide Poisoning: Identification of Prussic Acid in by Analyzing of Various Parameters in Cattle. In 2020 International Conference on Communication and Signal Processing. pp. 561-568.
- Provin, T.; Pitt, J.L. Nitrates and Prussic Acid in Forages: Sampling, Testing and Management Strategies; 2024. [Google Scholar]
- Rashid, S., Ashraf, R., Jamil, F., Sanawar, S., Zubair, Z. and Iftikhar, H.F., 2023. Use and abuse of sorghum and jequirity plants in cattle. One Health Triad, Unique Scientific Publishers, Faisalabad, Pakistan, 3, 194-201.
- Kohler, B.; Gigon, A.; Edwards, P.J.; Krusi, B.; Langenauer, R.; Luscher, A. Changes in the species composition and conservation value of limestone grassland in Northern Switzerland after 22 years of contrasting managements. Perspectives in Plant Ecology, Evolution and Systematics 2005, 7, 51–67. [Google Scholar] [CrossRef]
- Deen, A.U.; Kumari, V.; Sharma, A.N.; Mondal, G.; Singh, G.P. Understanding cyanogenic glycoside toxicity in livestock: A review. International Chemistry Studies 2018, 6, 1559–1561. [Google Scholar]
- Harris, B., and Shearer, J. 2003. Nitrate, Prussic Acid and Grass Tetany Problems in cattle feeding. DS6. Gainesville: Animal Science Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. 5p.
- Dweikat, I. A diploid, interspecific, fertile hybried from caltivated Sorghum, Sorghum bicolor, and the common Johngrass weed Sorghum helepense. Molecular Breeding 2005, 16, 93–101. [Google Scholar] [CrossRef]
- Venkateswaran, K., Elangovan, M., Sivaraj.N. 2019. Origin, Domestication, and diffusion of sorghum bicolor. Breeding sorghum for Diverse End Uses, pp 15-31.
- Van Saun, R.J. Trace mineral nutrition of sheep. Veterinary Clinics: Food Animal Practice 2023, 39(3), 517–533. [Google Scholar] [CrossRef] [PubMed]
- T’oth, V.; Lehoczky. Investigations on the germination depth of Johnson grass (Sorghum helepense) pers. Communications in agricultural and Applied Biological Sciences 2006, 71, 803–808. [Google Scholar]
- Sinha, R.K.; Kumari, B.I.B.H.A.; Kumar, A.N.I.L.; Azad, C.S. Sorghum poisoning in cattle and its therapeutic management. Journal of Agri Search 2019, 6, 108–109. [Google Scholar]
- Selk, G. 1988. Nitrate and Prussic Poising in Cattle. CR-3272. Stillwater: Oklahoma Cooperative Extension Service. 6p.

| Plant family | Scientific name of the plant | Type of Cyanogenic glycoside | Concentration level |
References |
|---|---|---|---|---|
| Gramineae | Sorghum helepense & bicolor species | Dhurin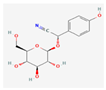
|
High | Emendack et al. (2025). Deen et al, (2018). |
| Fabaceae | Phaseolus vulgaris | Linamarin 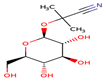
|
High | Alcázar-Valle et al. (2020). |
| Rosaceae | Prunus amygdalus | Prunacin 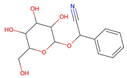
|
High | Amjadian et al. (2020). |
| Hydrocyanic acid (ppm) | Effect on livestock |
|---|---|
| Less than 500 | Considered safe |
| 500 -750 | Slightly toxic, should not be the only source of feed to livestock |
| >750 | Toxic and will cause animal death. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




