Submitted:
18 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Thermal and Non-Thermal Pretreatments
2.3. Drying Methods
2.3.1. Hot-Air Drying (HAD)
2.3.2. Freeze-Drying (FD)
2.3.3. Hybrid Freeze-Drying and Hot-Air Drying (FD–HAD)
2.4. Analytical Methods
2.4.1. Drying Parameters
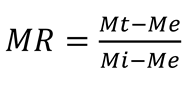
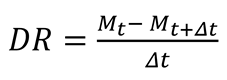
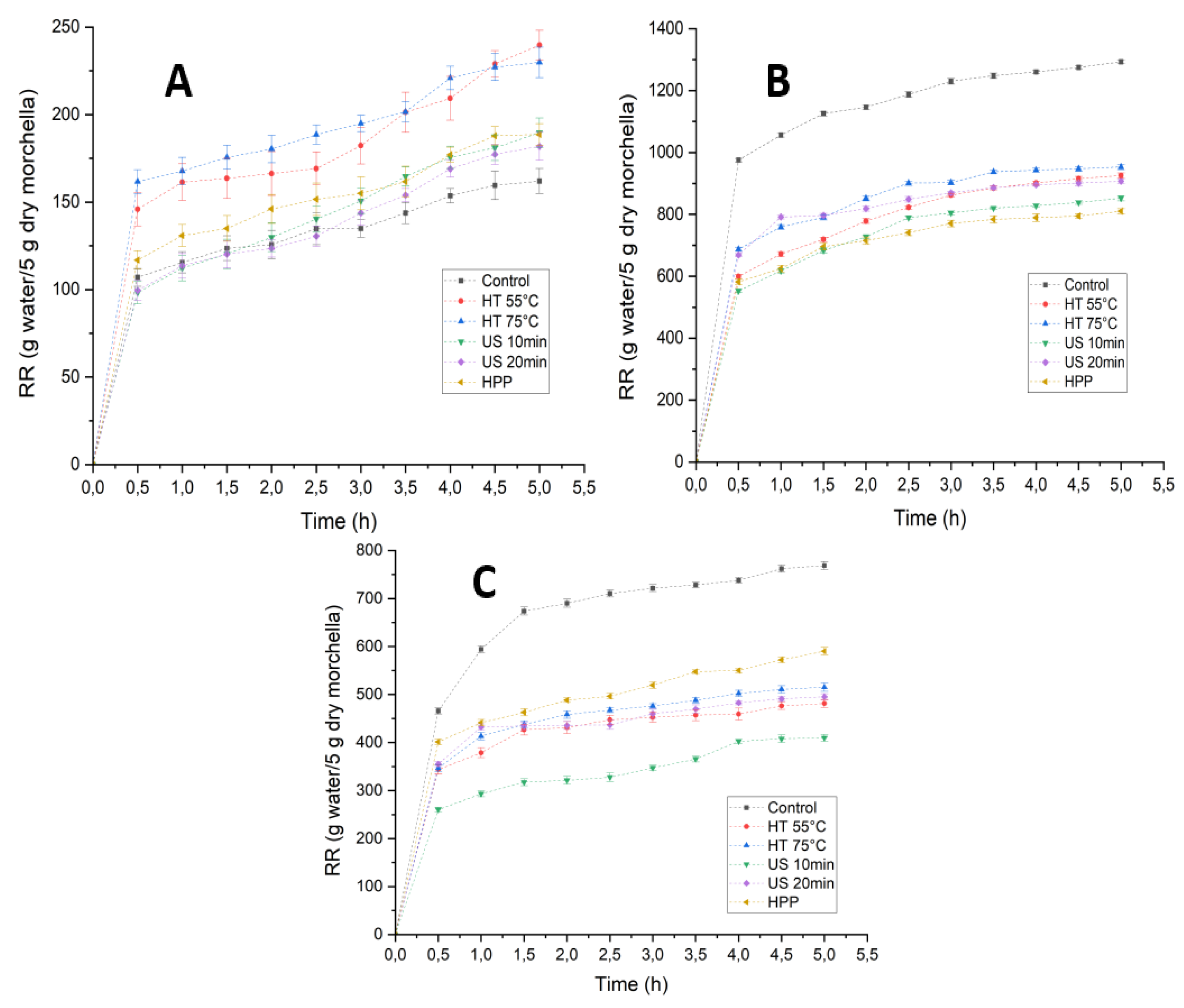
2.4.2. Rehydration Ratio (RR)
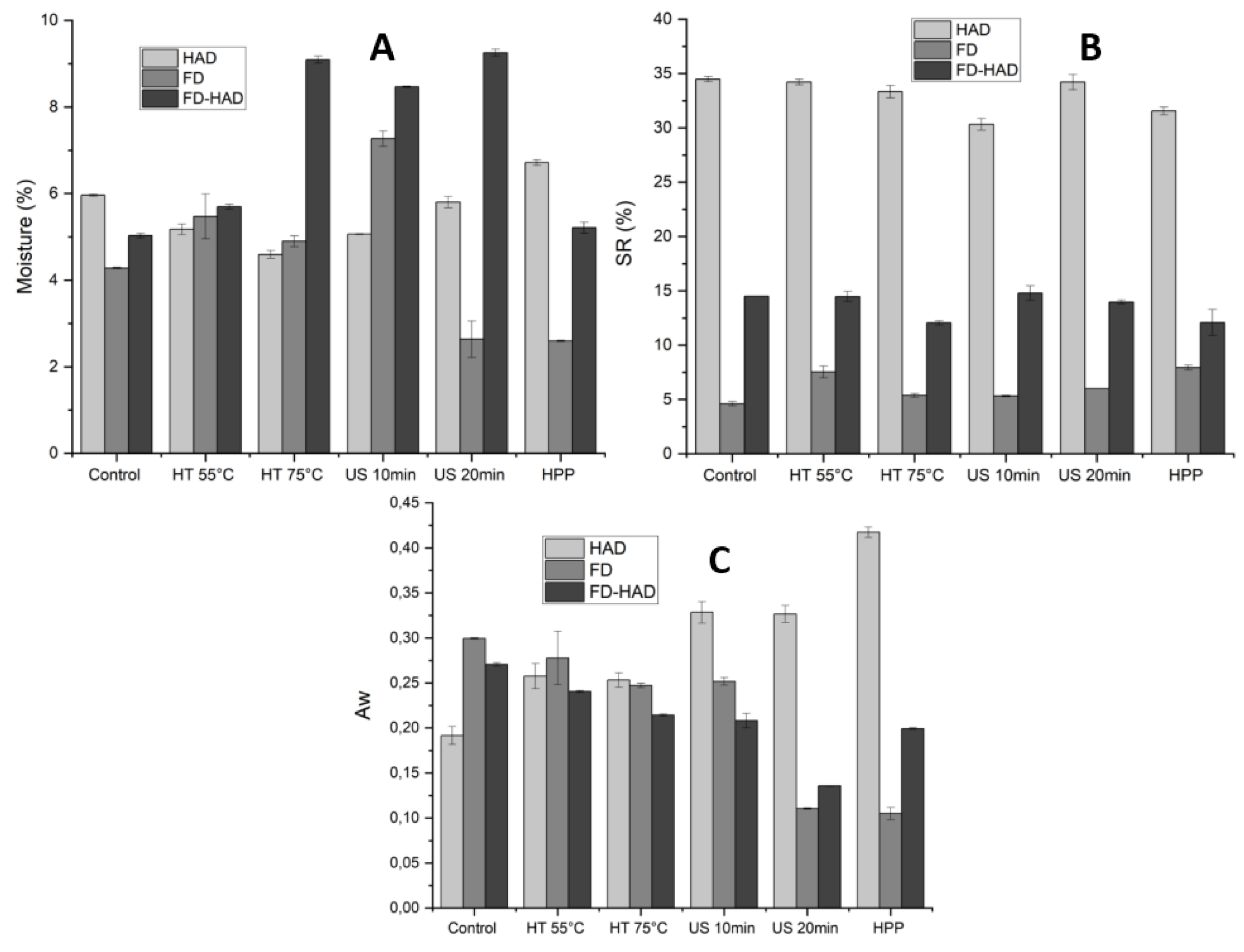
2.4.3. Physical Parameters
2.4.4. Proximate Composition Analysis
2.4.5. Total Phenolic Content and Antioxidant Activity
2.5. Statistical Analysis
3. Results and Discussion
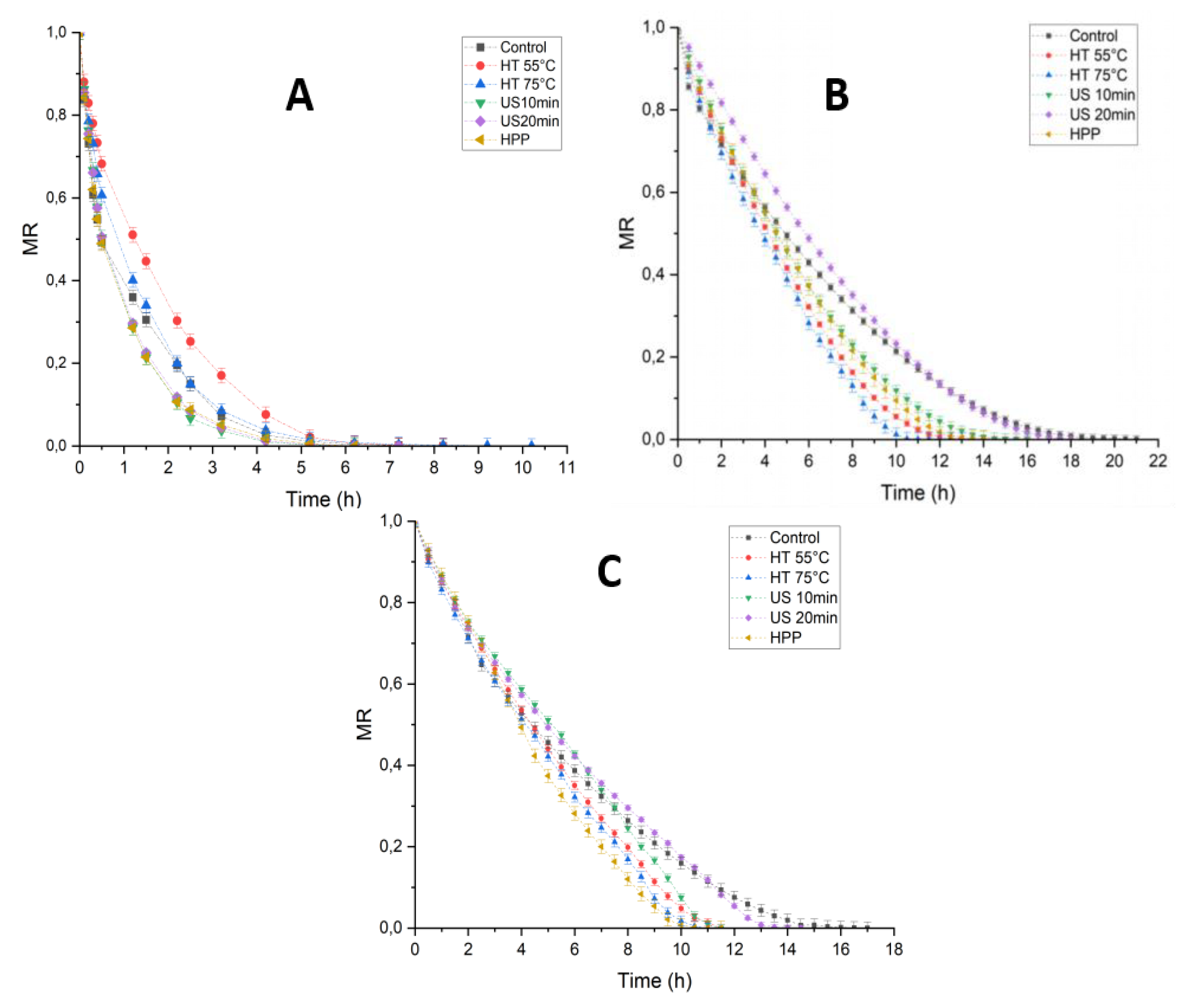
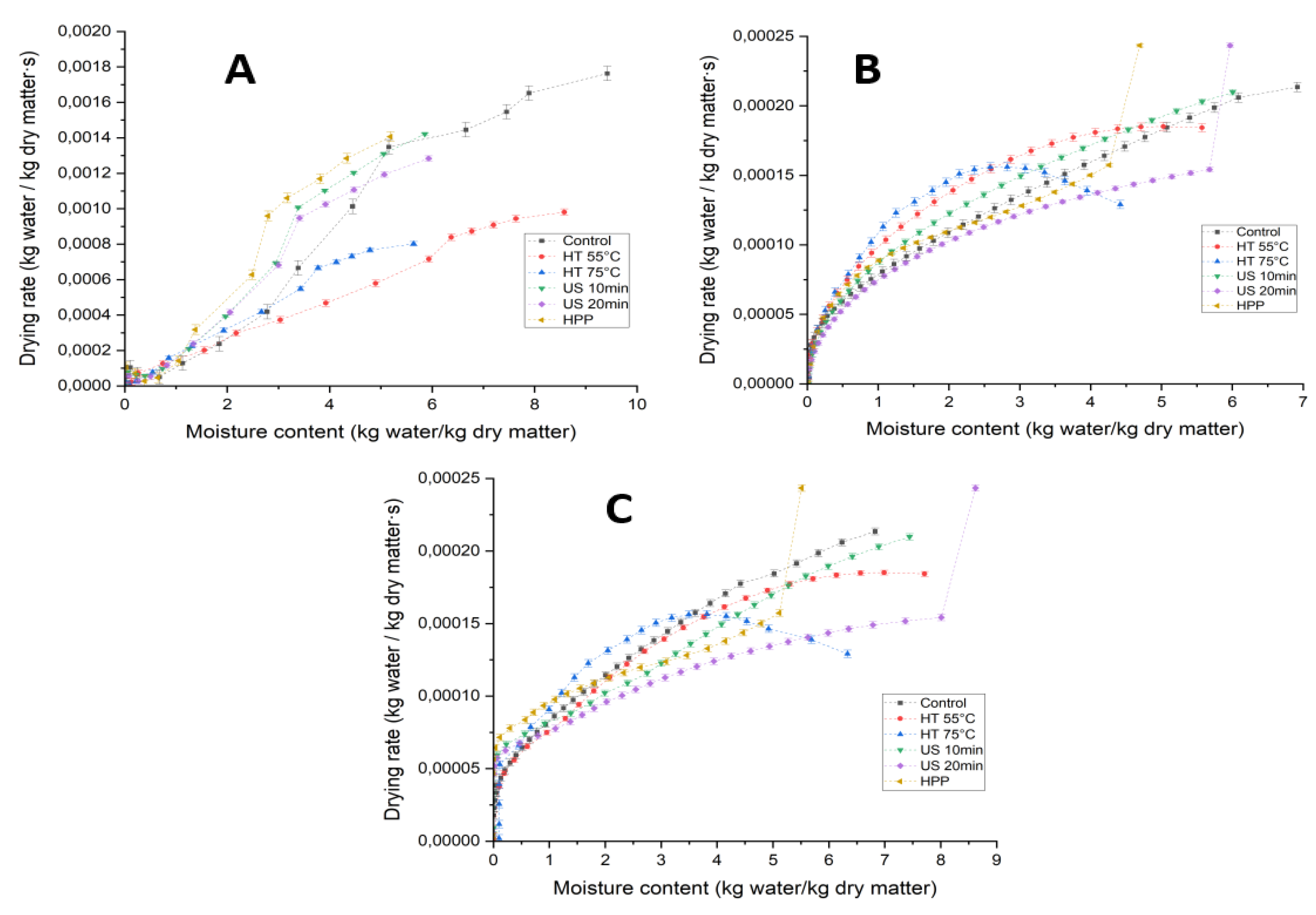
3.1. Drying Kinetics and Weibull Model of Morchella conica Under Different Pretreatments and Drying Methods
3.2. Influence of Different Pretreatments and Drying Methods on the Physical Properties of Morchella conica
3.3. Influence of Pretreatments and Drying Methods on the Rehydration Properties of Morchella conica
3.4. Influence of Pretreatments and Drying Methods on the Proximate Composition of Morchella conica
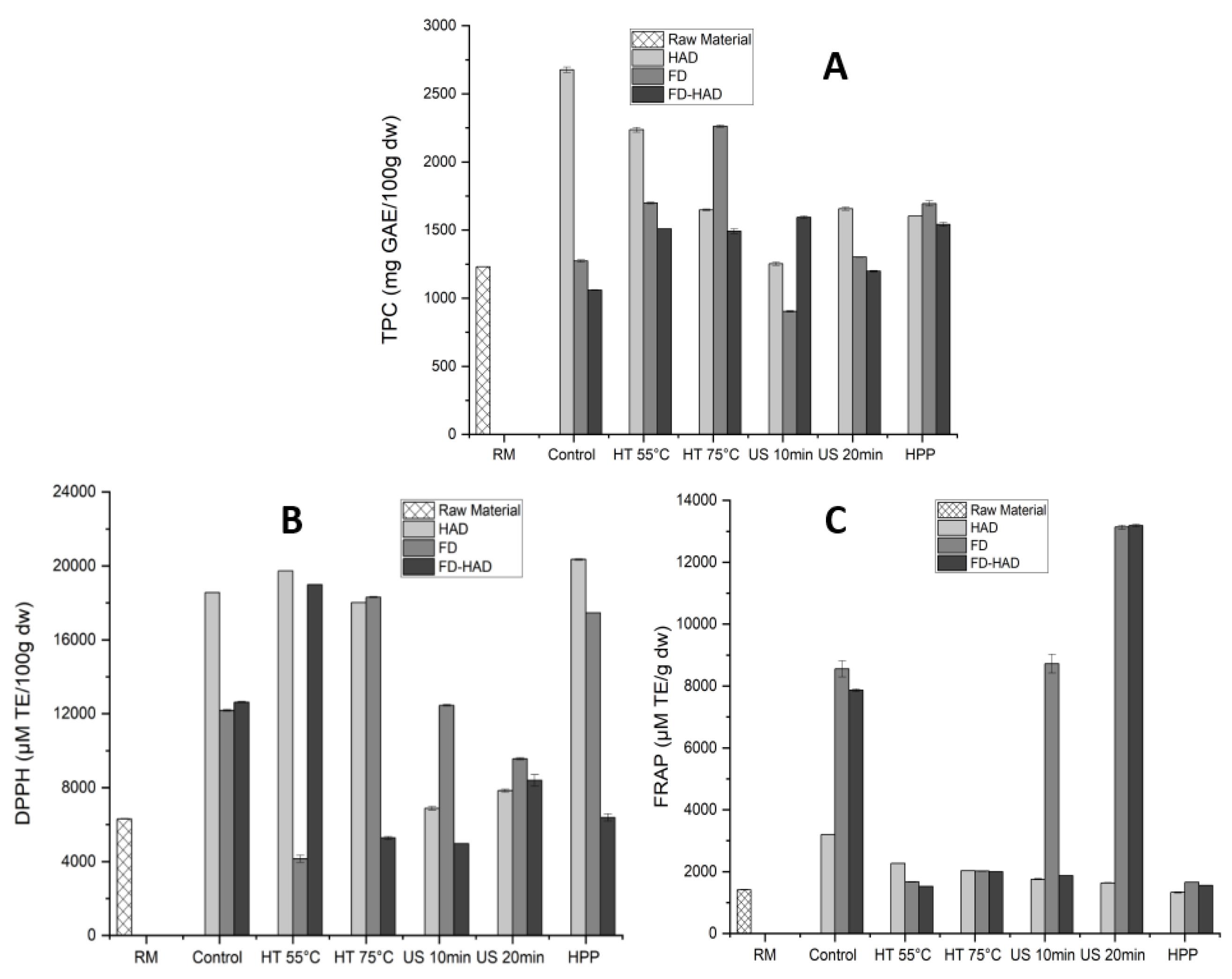
3.5. Influence of Pretreatments and Drying Methods on Bioactive Compounds of Morchella conica
3.5.1. Total Phenolic Content (TPC)
3.5.2. Antioxidant Activity (DPPH)
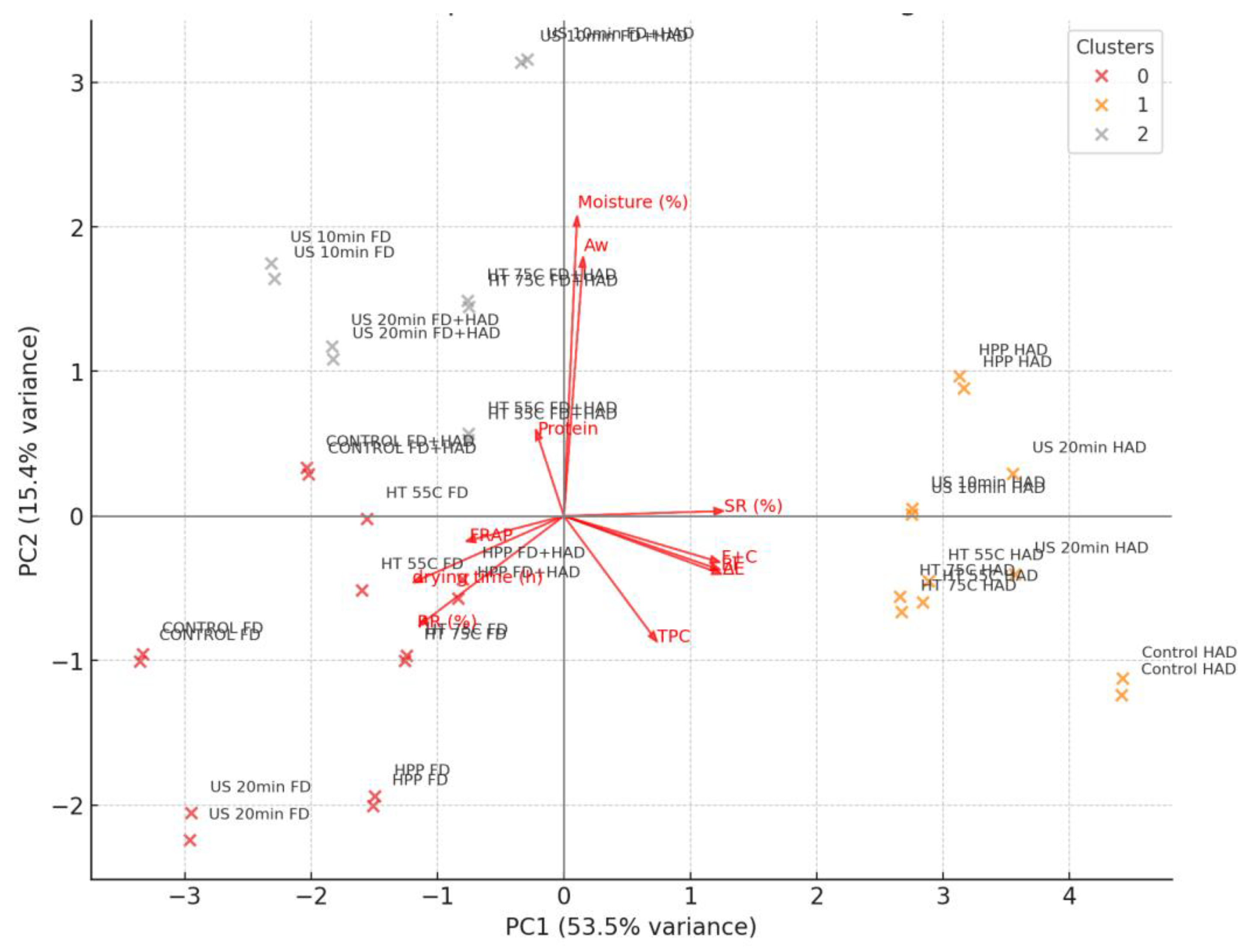
3.6. Principal Component Analysis (PCA)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taşkın, H.; Süfer, Ö.; Attar, ŞH.; Bozok, F.; Baktemur, G.; Büyükalaca, S.; Kafkas, NE. Total phenolics, antioxidant activities and fatty acid profiles of six Morchella species. J. Food Sci. Technol. 2021, 58, 692–700. [Google Scholar] [CrossRef]
- Deng, G.; Li, J.; Liu, H.; Wang, Y. Volatile compounds and aroma characteristics of mushrooms: A review. Crit. Rev. Food Sci. Nutr. 2024, 64, 13175–13192. [Google Scholar] [CrossRef]
- Tietel, Z.; Masaphy, S. True morels (Morchella)—nutritional and phytochemical composition, health benefits and flavor: a review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1888–901. [Google Scholar] [CrossRef]
- Acar, İ.; Blando, F.; Gul, B.; Greco, A.; Mukemre, M.; Uzun, Y.; Dalar, Y. The phenolic profile and biological activities of the wild-edible mushrooms Helvella leucopus and Morchella pulchella. Food Meas. 2021, 15, 555–566. [Google Scholar] [CrossRef]
- Jaime-Moya, I.; Gómez, C. Morchella (Morchella spp.): El hongo de los bosques norpatagónicos de la Región de Aysén; Instituto Forestal: Coyhaique, Chile, 2018; Volume Documento de Divulgación No. 45, p. p. 25. ISBN 978-956-318-147-0. [Google Scholar]
- Leal, P.; Carrasco, A. Antecedentes para la producción de Morchella en Chile. PortalFrutícola. 2018. Available online: https://www.portalfruticola.com/noticias/2018/05/23/antecedentes-para-la-produccion-de-morchella (accessed on 5 March 2026).
- Poblete Hernández, P.; Catelicán Vera, L. Productos Forestales No Madereros; Instituto Forestal: Chile, 2025; Volume Boletín No. 44, p. 30. ISBN 978-956-318-147-0. [Google Scholar]
- Castellanos-Reyes, K.; Villalobos-Carvajal, R.; Beldarrain-Iznaga, T. Fresh Mushroom Preservation Techniques. Foods 2021, 10, 2126. [Google Scholar] [CrossRef]
- Wang, T.; Ma, J.; Chen, H.; Li, Y. Drying kinetics and quality evaluation of Oudemansiella raphanipes treated with ultrasound-assisted hot air drying. J. Food Process. Preserv. 2023, 47, e4648. [Google Scholar] [CrossRef]
- Popa, M.; Tausan, I.; Draghici, O.; Soare, A.; Oancea, S. Influence of Convective and Vacuum-Type Drying on Quality, Microstructural, Antioxidant and Thermal Properties of Pretreated Boletus edulis Mushrooms. Molecules 2022, 27, 4063. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xu, H.; Wang, Y.; Liu, Y. Drying methods impact on color, texture and bioactives in mushrooms: A review. Foods 2023, 12, 1220. [Google Scholar] [CrossRef]
- Zhang, L.; Qiao, Y.; Wang, C.; Liao, L.; Liu, L.; Shi, D.; An, K.; Hu, J.; Xu, Q. Effects of Freeze Vacuum Drying Combined with Hot Air Drying on the Sensory Quality, Active Components, Moisture Mobility, Odors, and Microstructure of Kiwifruits. J. Food Qual. 2019, 1–11. [Google Scholar] [CrossRef]
- Hou, H.; Lu, X.; Du, H.; Chen, X.; Fang, D.; Fan, X.; Hu, Q.; Zhao, L. Effects of pre-cutting treatments and combination drying with different orders on drying characteristics and physicochemical properties of Lentinula edodes. J. Sci. Food Agric. 2021, 101, 2063–2071. [Google Scholar] [CrossRef]
- Hu, L.; Bi, J.; Jin, X.; Qiu, Y.; van der Sman, R.G.M. Study on the rehydration quality improvement of shiitake mushroom by combined drying methods. Foods 2021, 10, 769. [Google Scholar] [CrossRef]
- Moutia, I.; Lakatos, E.; József Kovács, A. Impact of Dehydration Techniques on the Nutritional and Microbial Profiles of Dried Mushrooms. Foods 2024, 13, 3245. [Google Scholar] [CrossRef]
- Daud, N.F.S.; Said, F.M.; Mohyiddin, I.M.; Mohamad, S.F.S.; Zin, N.H. M.; Zakil, F.A.; Muhamad, S.S. Investigating the impact of blanching and salt treatment on the drying kinetics of oyster mushrooms. Bioresour. Technol. Rep. 2024, 27, 101943. [Google Scholar] [CrossRef]
- Maikon da Rocha, E.; Avila, S.; de Lima, J.J.; Scheffer de Andrade Silva, R.; Miranda Bacila, D.; Mathias, A.L.; Jorge, R.M.M. Avaliação da eficácia do branqueamento e condições ideais de temperatura e tempo para minimizar o escurecimento de cogumelos comerciais. Scientia Plena 2024, 20, 6. [Google Scholar] [CrossRef]
- Sun, M.; Xu, Y.; Ding, Y.; Gu, Y.; Zhuang, Y.; Fan, X. Effect of Ultrasound Pretreatment on the Moisture Migration and Quality of Cantharellus cibarius Following Hot Air Drying. Foods 2023, 12, 2705. [Google Scholar] [CrossRef]
- Coşkun, N.; Sümeyye, S.; Yassine, J.; Bordiga, N.; Karav, S. The Impact of Freeze Drying on Bioactivity and Physical Properties of Food Products. Appl. Sci. 2024, 14, 9183. [Google Scholar] [CrossRef]
- Yuan-Yuan, Z.; Jian-Yong, Y.; Jin-Feng, B.; Qin-Qin, C.; Mo, Z.; Biao, Z. Improving of texture and rehydration properties by ultrasound pretreatment for infrared-dried shiitake mushroom slices. Dry. Technol. 2019, 37, 352–362. [Google Scholar] [CrossRef]
- Dziki, D. Recent trends in pretreatment of food before freeze-drying. Processes 2020, 8, 1661. [Google Scholar] [CrossRef]
- Yuan, L.; Lao, F.; Shi, X.; Zhang, D.; Wu, J. Effects of cold plasma, high hydrostatic pressure, ultrasound, and high-pressure carbon dioxide pretreatments on the quality characteristics of vacuum freeze-dried jujube slices. Ultrason. Sonochem. 2022, 90, 106219. [Google Scholar] [CrossRef] [PubMed]
- Llavata, B.; García-Pérez, J.; Simal, S.; Cárcel, J. Innovative pre-treatments to enhance food drying: a current review. Curr. Opin. Food Sci. 2020, 35, 20–26. [Google Scholar] [CrossRef]
- Chiozzi, V.; Agriopoulou, S.; Varzakas, T. Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing. Appl. Sci. 2022, 12, 2202. [Google Scholar] [CrossRef]
- Santos, N.C.; Almeida, R.L.J.; da Silva, G.M.; Fonseca, J.; Alcântara, M.; Tavares, M.; França, A.; André, A.; Almeida, M.; Brito, A. Impact of high hydrostatic pressure (HHP) pre-treatment drying cashew (Anacardium occidentale L.): drying behavior and kinetic of ultrasound-assisted extraction of total phenolics compounds. Food Meas. 2023, 17, 1033–1045. [Google Scholar] [CrossRef]
- Navarro-Baez, J.E.; Martínez, L.M.; Welti-Chanes, J.; Buitimea-Cantúa, G.V.; Escobedo-Avellaneda, Z. High Hydrostatic Pressure to Increase the Biosynthesis and Extraction of Phenolic Compounds in Food: A Review. Molecules. 2022, 27, 1502. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xie, Y.; Liu, D.; Liu, R.; Han, J. Effects of Drying Process and High Hydrostatic Pressure on Extraction of Antioxidant Ergothioneine from Pleurotus citrinopileatus Singer. Foods 2024, 13, 878. [Google Scholar] [CrossRef]
- Xu, L.; Fang, X.; Wu, W.; Chen, H.; Mu, H.; Gao, H. Effects of high-temperature pre-drying on the quality of air-dried shiitake mushrooms (Lentinula edodes). Food Chem. 2019, 285, 406–413. [Google Scholar] [CrossRef]
- Li, X.; Li, J.; Wang, R.; Rahaman, A.; Zeng, X.A.; Brennan, C.S. Combined effects of pulsed electric field and ultrasound pretreatments on mass transfer and quality of mushrooms. LWT 2021, 150, 112008. [Google Scholar] [CrossRef]
- Villalobos-Pezos, M.; Muñoz-Fariña, O.; Ah-Hen, K.S.; Garrido Figueroa, M.-F.; García-Figueroa, O.; González-Esparza, A.; González-Pérez de Medina, L.; Bastías-Montes, J.M. Effects of Drying Treatments on the Physicochemical Characteristics and Antioxidant Properties of the Edible Wild Mushroom Cyttaria espinosae Lloyd (Digüeñe Mushroom). J. Fungi. 2025, 11, 13. [Google Scholar] [CrossRef]
- Tamarit-Pino, Y.; Bastías-Montes, J.M.; Segura-Ponce, L.A.; Díaz-Álvarez, R.E.; Guzmán-Meza, M.F.; Quevedo-León, R.A. Effect of electrohydrodynamic pretreatment on drying rate and rehydration properties of Chilean sea cucumber (Athyonidium chilensis). Food Bioprod. Process. 2020, 123, 284–295. [Google Scholar] [CrossRef]
- Quevedo, R.; Díaz, O.; Valencia, E.; Pedreschi, F.; Bastías, J.M.; Siche, R. Differences between the order model and the weibull model in the modeling of the enzymatic browning. Food Bioprocess Technol. 2016, 9, 1961–1967. [Google Scholar] [CrossRef]
- Zhang, C.; Quek, S.Y.; Fu, N.; Su, Y.; Kilmartin, P.A.; Chen, X. D. Storage stability and in vitro digestion of microencapsulated powder containing fermented noni juice and probiotics. Food Biosci. 2020, 37, 100740. [Google Scholar] [CrossRef]
- Wang, Q.; Li, S.; Han, X.; Ni, Y.; Zhao, D.; Hao, J. Quality evaluation and drying kinetics of shiitake mushrooms dried by hot air, infrared and intermittent microwave–assisted drying methods. LWT 2019, 107, 236–242. [Google Scholar] [CrossRef]
- Nasiri, M.; Barzegar, M.; Sahari, M.A.; Niakousari, M. Application of Tragacanth gum impregnated with Satureja khuzistanica essential oil as a natural coating for enhancement of postharvest quality and shelf life of button mushroom (Agaricus bisporus). Int. J. Biol. Macromol. 2018, 106, 218–226. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 21st ed.; AOAC International: Rockville, MD, USA, 2019. [Google Scholar]
- Leiva-Portilla, D.J.; Rodríguez-Núñez, K.E.; Rodríguez-Ramos, F.J.; Delgadillo Acevedo, Á.; Uribe, E. Impact on Physicochemical Composition and Antioxidant Activity of the Wild Edible Mushroom Cyttaria espinosae Subjected to Drying. Chem. Biodivers. 2020, 17, e2000642. [Google Scholar] [CrossRef]
- Pirce, F.; Vieira, T.M.F.S.; Augusto-Obara, T.R.; Alencar, S.M.; Romero, F.; Scheuermann, E. Effects of convective drying assisted by ultrasound and osmotic solution on polyphenol, antioxidant and microstructure of murtilla (Ugni molinae Turcz) fruit. J. Food Sci. Technol. 2021, 58, 138–146. [Google Scholar] [CrossRef]
- Abbaspour-Gilandeh, Y.; Kaveh, M.; Fatemi, H.; Aziz, M. Combined Hot Air, Microwave, and Infrared Drying of Hawthorn Fruit: Effects of Ultrasonic Pretreatment on Drying Time, Energy, Qualitative, and Bioactive Compounds’ Properties. Foods 2021, 10, 1006. [Google Scholar] [CrossRef]
- Tianhai, L.; Xiang, W.; Weiwei, L.; Yingying, X.; Yang, Y.; Haixia, W. The Effects of Different Postharvest Drying Temperatures on the Volatile Flavor Components and Non-Volatile Metabolites of Morchella sextelata. Horticulturae 2024, 10, 812. [Google Scholar] [CrossRef]
- Tao, Y.; Wang, P.; Wang, Y.; Kadam, S.U.; Han, Y.; Wang, J.; Zhou, J. Power ultrasound as a pretreatment to convective drying of mulberry (Morus alba L.) leaves: Impact on drying kinetics and selected quality properties. Ultrason. Sonochem. 2016, 31, 310–318. [Google Scholar] [CrossRef]
- Oliveira, F.I.; Gallão, M.I.; Rodrigues, S.; Fernandes, F.A.N. Dehydration of Malay apple (Syzygium malaccense L.) using ultrasound as pre-treatment. Food Bioprocess Technol. 2011, 4, 610–615. [Google Scholar] [CrossRef]
- George, J.M.; Sowbhagya, H.B.; Rastogi, N. K. Effect of high pressure pretreatment on drying kinetics and oleoresin extraction from ginger. Dry. Technol. 2018, 36, 1107–1116. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, Z.; Liu, C.; Li, D.; Jiang, N.; Liu, C. Effects of ultrasound pretreatment on drying kinetics and quality parameters of button mushroom slices. Dry. Technol. 2016, 34, 1791–1800. [Google Scholar] [CrossRef]
- Marçal, S.; Sousa, A.S.; Taofiq, O.; Antunes, F.; Morais, A.M.; Freitas, A.C.; Barros, L.; Ferreira, I.C.; Pintado, M. Impact of postharvest preservation methods on nutritional value and bioactive properties of mushrooms. Trends Food Sci. Technol. 2021, 110, 418–431. [Google Scholar] [CrossRef]
- Ju, H.Y.; Zhao, S.H.; Mujumdar, A.S.; Fang, X.M.; Gao, Z.J.; Zheng, Z.A.; Xiao, H.W. Energy efficient improvements in hot air drying by controlling relative humidity based on Weibull and Bi-Di models. Food Bioprod. Process. 2018, 111, 20–29. [Google Scholar] [CrossRef]
- Issis, Q.F.; Antonio, V.G.; Elsa, U.; Valeria, V.; Nicole, C.; Jacqueline, P. Vacuum drying application to maqui (Aristotelia chilensis [Mol] Stuntz) berry: Weibull distribution for process modelling and quality parameters. J. Food Sci. Technol. 2019, 56, 1899–1908. [Google Scholar] [CrossRef]
- Dai, J.W.; Xiao, H.W.; Zhang, L.H.; Chu, M.Y.; Qin, W.; Wu, Z.J.; Han, D.D.; Li, Y.L.; Liu, Y.W.; Yin, P.F. Drying characteristics and modeling of apple slices during microwave intermittent drying. J. Food Process Eng. 2019, 42, e13212. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Y.; Pandiselvam, R.; Su, D.; Xu, H. Comparative analysis of drying methods on Pleurotus eryngii: Impact on drying efficiency, nutritional quality, and flavor profile. Food Bioprocess Technol. 2024, 17, 4598–4616. [Google Scholar] [CrossRef]
- Carrión, C.; Mulet, A.; García-Pérez, J.V.; Cárcel, J.A. Ultrasonically assisted atmospheric freeze-drying of button mushroom. Drying kinetics and product quality. Dry. Technol. 2018, 36, 1814–1823. [Google Scholar] [CrossRef]
- Swami Hulle, N.R.; Rao, P.S. Effect of High Pressure Pretreatments on Structural and Dehydration Characteristics of Aloe Vera (Aloe barbadensis Miller) Cubes. Dry. Technol. 2016, 34, 105–118. [Google Scholar] [CrossRef]
- Zhang, Y.; Xie, Y.; Wang, F.; Ming, C.; Li, Y.; Xingyi, L. Effects of different drying methods on the quality characteristics of Morchella. Trans. Chin. Soc. Agric. Eng. 2024, 40, 111–119. [Google Scholar] [CrossRef]
- Liao, Y.; Xin, M.; Dong, H.; Liu, Y.; Li, L.; Guo, X.; Cheng, S.; Chen, G. Comparison of different drying processes of Morchella sextelata: Changes in volatile and non-volatile components, color and texture. Food Chem. X 2025, 25, 102220. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhang, L.; Feng, Y.; Zhou, C.; Yagoub, A.E.G.A.; Wahia, H.; Ma, H.; Zhang, J.; Sun, Y. Ultrasound freeze-thawing style pretreatment to improve the efficiency of the vacuum freeze-drying of okra (Abelmoschus esculentus (L.) Moench) and the quality characteristics of the dried product. Ultrason. Sonochem. 2021, 70, 105300. [Google Scholar] [CrossRef]
- Pimenta Inada, K.O.; Nunes, S.; Martínez-Blázquez, J.A.; Tomás-Barberán, F.A.; Perrone, D.; Monteiro, M. Effect of high hydrostatic pressure and drying methods on phenolic compounds profile of jabuticaba (Myrciaria jaboticaba) peel and seed. Food Chem. 2020, 309, 125794. [Google Scholar] [CrossRef]
- Jiajia, M.A.; Minghui, D.; Xinyao, Q.; Qianshuo, S.; Meijuan, J.; Yuning, W. Effects of different drying methods on the quality characteristics and rehydration performance of Morchella esculenta. Trans. Chin. Soc. Agric. Eng. 2024, 40, 288–297. [Google Scholar] [CrossRef]
- Alfiya, P.V.; Rajesh, G.K.; Murali, S.; Delfiya, D.S.A. Experimental evaluation of hot air-assisted continuous microwave drying of oyster mushroom (Pleurotus ostreatus). Heat Transfer 2025, 54, 4207–4222. [Google Scholar] [CrossRef]
- Chen, R.; Xu, L.; Zhang, S.; Duan, Y.; Zhu, C.; Zhao, P.; Liu, C.; Jia, F.; Zhang, X.; Liu, J.; Han, Y. Optimizing slicing to boost quality in hot air-dried shiitake mushroom. LWT 2025, 228, 118026. [Google Scholar] [CrossRef]
- Liu, Z.; Luo, J.; Zhang, J.; Liu, W.; Li, H.; Hu, L.; Wang, D.; Hengchao, E.; Sun, Z.; Xia, Y.; Weng, R. Advantages of Multiphase Microwave Drying (MMD) Technology in Shiitake Mushroom Processing: Dual Optimization of Efficiency and Quality. Food Bioprocess Technol. 2025, 18, 8751–8770. [Google Scholar] [CrossRef]
- Maoloni, A.; Cardinali, F.; Milanović, V.; Reale, A.; Boscaino, F.; Di Renzo, T.; Ferrocino, I.; Rampanti, G.; Garofalo, C.; Osimani, A.; Aquilanti, L. Impact of Different Drying Methods on the Microbiota, Volatilome, Color, and Sensory Traits of Sea Fennel (Crithmum maritimum L.) Leaves. Molecules 2023, 28, 7207. [Google Scholar] [CrossRef]
- Labuza, T.P.; Altunakar, B. Water activity prediction and moisture sorption isotherms. In Water Activity in Foods: Fundamentals and Applications; Barbosa-Cánovas, G.V., Fontana, A.J., Schmidt, S.J., Labuza, T.P., Eds.; Blackwell Publishing: Ames, IA, USA, 2007; pp. 109–154. [Google Scholar]
- Tamarit-Pino, Y.; Batías-Montes, J.M.; Segura-Ponce, L.A.; Guzmán-Meza, M.F.; Quevedo-León, R.A. Shelf-life prediction and quality changes in dried Chilean sea cucumber (Athyonidium chilensis) during accelerated storage. J. Food Process. Preserv. 2020, 44, e14644. [Google Scholar] [CrossRef]
- Deng, L.Z.; Mujumdar, A.S.; Zhang, Q.; Yang, X.H.; Wang, J.; Zheng, Z.A.; Xiao, H.W. Chemical and physical pretreatments of fruits and vegetables: Effects on drying characteristics and quality attributes—a comprehensive review. Crit. Rev. Food Sci. Nutr. 2017, 59, 1408–1432. [Google Scholar] [CrossRef]
- Belmiro, R.H.; Tribst, A.A.L.; Cristianini, M. Impact of high pressure processing in hydration and drying curves of common beans (Phaseolus vulgaris L.) Innov. Food Sci. Emerg. Technol. 2018, 47, 279–285. [Google Scholar] [CrossRef]
- Zhang, K.; Pu, Y.Y.; Sun, D.W. Recent advances in quality preservation of postharvest mushrooms (Agaricus bisporus): A review. Trends Food Sci. Technol. 2018, 78, 52–61. [Google Scholar] [CrossRef]
- Villalobos-Pezos, M.; Muñoz-Fariña, O.; Ah-Hen, K.S.; Garrido-Figueroa, M.F.; García-Figueroa, O.; González-Esparza, A.; González-Pérez de Medina, L.; Bastías-Montes, J.M. Optimization of Phenolic Content Extraction and Effects of Drying Treatments on Physicochemical Characteristics and Antioxidant Properties of Edible Mushroom Pleurotus ostreatus (Jacq.) P. Kumm (Oyster Mushroom). Antioxidants. 2024, 13, 1581. [Google Scholar] [CrossRef]
- Rorato, A.C.; Longhi, D.A.; Biz, A.P.; Muniz, V.R.G.F. Enhancing quality of dried Agaricus bisporus: Advanced techniques in vacuum impregnation and freeze-drying. Dry. Technol. 2025, 43, 653–667. [Google Scholar] [CrossRef]
- Kibar, B. Influence of different drying methods and cold storage treatments on the postharvest quality and nutritional properties of P. ostreatus mushroom. Turk. J. Agric. For. 2021, 45, 565–579. [Google Scholar] [CrossRef]
- Uçar, T.M.; Karadağ, A. The effects of vacuum and freeze-drying on the physicochemical properties and in vitro digestibility of phenolics in oyster mushroom (Pleurotus ostreatus). LWT 2021, 148, 111764. [Google Scholar] [CrossRef]
- Kalač, P. A review of chemical composition and nutritional value of wild-growing and cultivated mushrooms. J. Sci. Food Agric. 2013, 93, 209–218. [Google Scholar] [CrossRef]
- Heleno, S.A.; Barros, L.; Martins, A.; Ferreira, I.C.F.R. Nutritional value, bioactive compounds and antioxidant properties of edible mushrooms. Food Chem. 2015, 160, 27–34. [Google Scholar] [CrossRef]
- Gąsecka, M.; Siwulski, M.; Mleczek, M.; Budka, A.; Niedzielski, P. The effect of drying temperature on bioactive compounds and antioxidant activity of Leccinum scabrum (Bull.) Gray and Hericium erinaceus (Bull.) Pers. LWT 2018, 96, 420–425. [Google Scholar] [CrossRef]
- Pérez-Lamela, C.; Franco, I.; Falqué, E. Impact of High-Pressure Processing on Antioxidant Activity during Storage of Fruits and Fruit Products: A Review. Molecules. 2021, 26, 5265. [Google Scholar] [CrossRef]
- Tepsongkroh, B.; Thaihuttakij, C.; Supawong, S.; Jangchud, K. Impact of high pressure pre-treatment and hot water extraction on chemical properties of crude polysaccharide extract obtained from mushroom (Volvariella volvacea). Food Chem. X 2023, 19, 100864. [Google Scholar] [CrossRef]
- Izham, I.; Avin, F.; Raseetha, S. Systematic review: heat treatments on phenolic content, antioxidant activity, and sensory quality of Malaysian mushroom: Oyster (Pleurotus spp.) and Black Jelly (Auricularia spp.) Front. Sustain. Food Syst. 2022, 6, 882939. [Google Scholar] [CrossRef]
| Treatment | k | n | R2 | MSE |
|---|---|---|---|---|
| Control HAD | 1.028±0.05a | 0.692 | 0.996 | 0.0009 |
| HT 55 °C HAD | 0.643±0.05b | 0.866 | 0.997 | 0.0008 |
| HT 75 °C HAD | 0.857±0.05c | 0.822 | 0.999 | 0.0003 |
| US 10 min HAD | 1.146±0.05d | 0.856 | 0.999 | 0.0001 |
| US 20 min HAD | 1.120±0.05e | 0.823 | 0.999 | 0.0002 |
| HPP HAD | 1.168±0.05f | 0.789 | 0.999 | 0.0002 |
| Control FD | 0.127±0.05g | 1.074 | 0.993 | 0.0011 |
| HT 55 °C FD | 0.103±0.05h | 1.383 | 0.997 | 0.0008 |
| HT 75 °C FD | 0.122±0.05i | 1.351 | 0.994 | 0.0014 |
| US 10 min FD | 0.096±0.05jo | 1.338 | 0.998 | 0.0005 |
| US 20 min FD | 0.061±0.05k | 1.400 | 0.998 | 0.0005 |
| HPP FD | 0.094±0.05hjo | 1.364 | 0.995 | 0.0010 |
| Control FD-HAD | 0.125±0.05gi | 1.178 | 0.996 | 0.0008 |
| HT 55 °CFD-HAD | 0.098±0.05hjmno | 1.374 | 0.993 | 0.0013 |
| HT 75 °C FD-HAD | 0.114±0.05ghjl | 1.333 | 0.991 | 0.0018 |
| US 10 min FD-HAD | 0.078±0.05jkmo | 1.418 | 0.987 | 0.0024 |
| US 20 min FD-HAD | 0.099±0.05hjno | 1.262 | 0.991 | 0.0016 |
| HPP FD-HAD | 0.089±0.05hjmno | 1.518 | 0.997 | 0.0007 |
| Treatment | L* | a* | b* | ΔE | BI |
|---|---|---|---|---|---|
| Fresh | 23.75±0.07a | 1.51±0.01a | 8.81±0.01a | - | 49.40±0.29a |
| Control HAD | 18.05±0.01b | 2.23±0.02b | 10.73±0.01b | 6.10±0.01a | 93.51±0.09b |
| HT 55 °C HAD | 18.80±0.01c | 2.76±0.01c | 8.27±0.08c | 5.18±0.01b | 66.66±0.74c |
| HT 75 °C HAD | 18.93±0.01d | 2.56±0.02d | 9.43±0.02d | 5.02±0.01c | 75.88±0.19d |
| US 10 min HAD | 18.81±0.01c | 2.51±0.01e | 9.01±0.14e | 5.04±0.07c | 72.17±1.27e |
| US 20 min HAD | 18.60±0.08e | 2.82±0.01f | 9.91±0.02f | 5.41±0.00d | 83.31±0.28f |
| HPP HAD | 18.90±0.01d | 3.01±0.10g | 9.16±0.03g | 5.08±0.04c | 75.24±0.62d |
| Control FD | 24.16±0.01f | 0.17±0.01h | 7.94±0.01h | 1.64±0.02efg | 38.86±0.03gh |
| HT 55 °C FD | 24.03±0.01ghi | 0.19±0.01hi | 7.98±0.01h | 1.60±0.01efgh | 39.39±0.09hi |
| HT 75 °C FD | 24.08±0.01fhi | 0.21±0.01hij | 7.99±0.01h | 1.57±0.04ehi | 39.45±0.12hi |
| US 10 min FD | 23.98±0.01g | 0.22±0.01hij | 8.03±0.04h | 1.54±0.02hi | 39.88±0.26i |
| US 20 min FD | 23.77±0.04aj | 0.26±0.01j | 7.73±0.04i | 1.66±0.03fg | 38.64±0.31gh |
| HPP FD | 24.01±0.02gh | 0.23±0.01ij | 7.60±0.14jk | 1.79±0.09j | 37.38±0.83j |
| Control FD-HAD | 23.95±0.07g | 0.39±0.01k | 7.77±0.01i | 1.54±0.01hi | 38.98±0.07ghi |
| HT 55 °CFD-HAD | 24.25±0.07k | 0.45±0.01l | 7.68±0.01hil | 1.63±0.05efg | 38.04±0.15gj |
| HT 75 °C FD-HAD | 24.12±0.01if | 0.46±0.00l | 7.56±0.01j | 1.67±0.02g | 37.65±0.05j |
| US 10 min FD-HAD | 23.75±0.07a | 0.42±0.01kl | 7.66±0.01hjk | 1.58±0.00efhi | 38.79±0.20gh |
| US 20 min FD-HAD | 23.73±0.01a | 0.44±0.01kl | 7.65±0.01hjk | 1.57±0.00ehi | 38.87±0.09gh |
| HPP FD-HAD | 23.85±0.07j | 0.46±0.01l | 7.73±0.04hj | 1.51±0.00i | 39.14±0.32hi |
| Treatment | Protein (g/100 g) | Lipids (g/100 g) | Ash (g/100 g) | Fiber (g/100 g) | Carbohydrates (g/100 g) | F + C (g/100 g) |
|---|---|---|---|---|---|---|
| Fresh | 30.83 ± 0.29ᵃ | 2.99 ± 0.20ᵃ | 12.20 ± 0.82ᵃ | 15.18 ± 1.47ᵃ | 38.81 ± 0.16ᵃ | 53.99 ± 1.31ᵃ |
| Control HAD | 32.71 ± 0.71ᵇ | 3.81 ± 0.29ᵇ | 15.67 ± 0.08ᵇ | 21.63 ± 0.11ᶜ | 13.96 ± 0.40ᶠ | 35.59 ± 0.50ᶠ |
| HT 55 °C HAD | 33.27 ± 0.25ᵇ | 2.17 ± 0.20ᶜ | 12.16 ± 0.45ᵃ | 21.85 ± 0.10ᶜ | 20.46 ± 0.10ᵉ | 42.32 ± 0.00ᵉ |
| HT 75 °C HAD | 32.63 ± 0.12ᵇ | 3.79 ± 0.06ᵇ | 12.48 ± 0.08ᵃ | 20.14 ± 0.05ᶜ | 21.97 ± 0.18ᵉ | 42.12 ± 0.14ᵉ |
| US 10 min HAD | 28.77 ± 1.15ᶜ | 3.06 ± 0.04ᵃᵇ | 7.35 ± 0.49ᵈ | 25.48 ± 0.28ᵇ | 25.46 ± 1.96ᵈ | 50.95 ± 1.67ᵇ |
| US 20 min HAD | 29.87 ± 0.57ᶜ | 3.65 ± 0.11ᵇ | 6.70 ± 0.16ᵈ | 26.89 ± 0.11ᵇ | 21.61 ± 0.73ᵉ | 48.51 ± 0.84ᶜ |
| HPP HAD | 31.19 ± 0.11ᵃᵇ | 3.58 ± 0.07ᵇ | 11.09 ± 0.11ᵃ | 31.69 ± 0.27ᵃ | 9.48 ± 0.13ᵍ | 41.16 ± 0.15ᵉ |
| Control FD | 31.84 ± 0.45ᵃᵇ | 2.52 ± 0.51ᵃ | 11.60 ± 0.60ᵃ | 18.04 ± 0.51ᶜ | 27.57 ± 1.04ᶜ | 45.60 ± 1.56ᵈ |
| HT 55 °C FD | 31.93 ± 0.43ᵃᵇ | 3.16 ± 0.39ᵃᵇ | 11.06 ± 0.18ᵃ | 17.84 ± 0.01ᶜ | 25.36 ± 0.21ᵈ | 43.20 ± 0.22ᵉ |
| HT 75 °C FD | 31.45 ± 0.08ᵃᵇ | 3.05 ± 0.12ᵃᵇ | 14.55 ± 0.34ᵇ | 16.56 ± 0.25ᵈ | 24.84 ± 0.79ᵈ | 41.40 ± 0.54ᵉ |
| US 10 min FD | 31.30 ± 0.08ᵃᵇ | 3.27 ± 0.09ᵃᵇ | 7.67 ± 0.44ᵈ | 26.63 ± 0.19ᵇ | 17.35 ± 0.80ᶠ | 43.99 ± 0.61ᵉ |
| US 20 min FD | 30.54 ± 0.34ᵃ | 4.72 ± 0.14ᶜ | 6.63 ± 0.49ᵈ | 21.39 ± 0.53ᶜ | 31.53 ± 0.24ᵇ | 52.92 ± 0.29ᵇ |
| HPP FD | 31.25 ± 0.20ᵃᵇ | 5.39 ± 0.49ᶜ | 8.60 ± 0.16ᵈ | 22.38 ± 0.41ᶜ | 27.22 ± 0.12ᶜ | 49.59 ± 0.53ᶜ |
| Control FD-HAD | 35.87 ± 0.52ᵇ | 1.48 ± 0.30ᵈ | 14.70 ± 0.41ᵇ | 23.57 ± 0.18ᶜ | 22.44 ± 0.07ᵉ | 46.01 ± 0.11ᵈ |
| HT 55 °C FD-HAD | 35.87 ± 0.52ᵇ | 2.52 ± 0.51ᵃ | 11.60 ± 0.60ᵃ | 18.04 ± 0.51ᶜ | 20.90 ± 0.07ᵉ | 38.93 ± 0.58ᶠ |
| HT 75 °C FD-HAD | 33.21 ± 0.03ᵇ | 3.74 ± 0.00ᵇ | 15.00 ± 0.22ᵇ | 26.63 ± 0.19ᵇ | 17.35 ± 0.80ᶠ | 43.99 ± 0.61ᵉ |
| US 10 min FD-HAD | 32.08 ± 0.33ᵇ | 1.73 ± 0.06ᵈ | 6.31 ± 0.09ᵈ | 24.28 ± 0.10ᶜ | 19.39 ± 0.46ᵉ | 43.67 ± 0.36ᵉ |
| US 20 min FD-HAD | 33.43 ± 0.34ᵇ | 4.05 ± 0.09ᵇ | 6.31 ± 0.02ᵈ | 25.51 ± 0.62ᵇ | 13.15 ± 0.39ᶠ | 38.67 ± 0.23ᶠ |
| HPP FD-HAD | 28.51 ± 0.06ᶜ | 5.51 ± 0.13ᶜ | 10.30 ± 0.29ᵃ | 28.53 ± 0.06ᵃᵇ | 13.21 ± 0.53ᶠ | 41.74 ± 0.47ᵉ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




