Submitted:
18 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Phytoremediation Experiment
2.1.1. Experimental Design
2.2. Measurement of Metal Content
2.3. Steam Distillation Experiment
2.4. Statistical Analysis
2.5. Biochar Synthesis
3. Results and Discussion
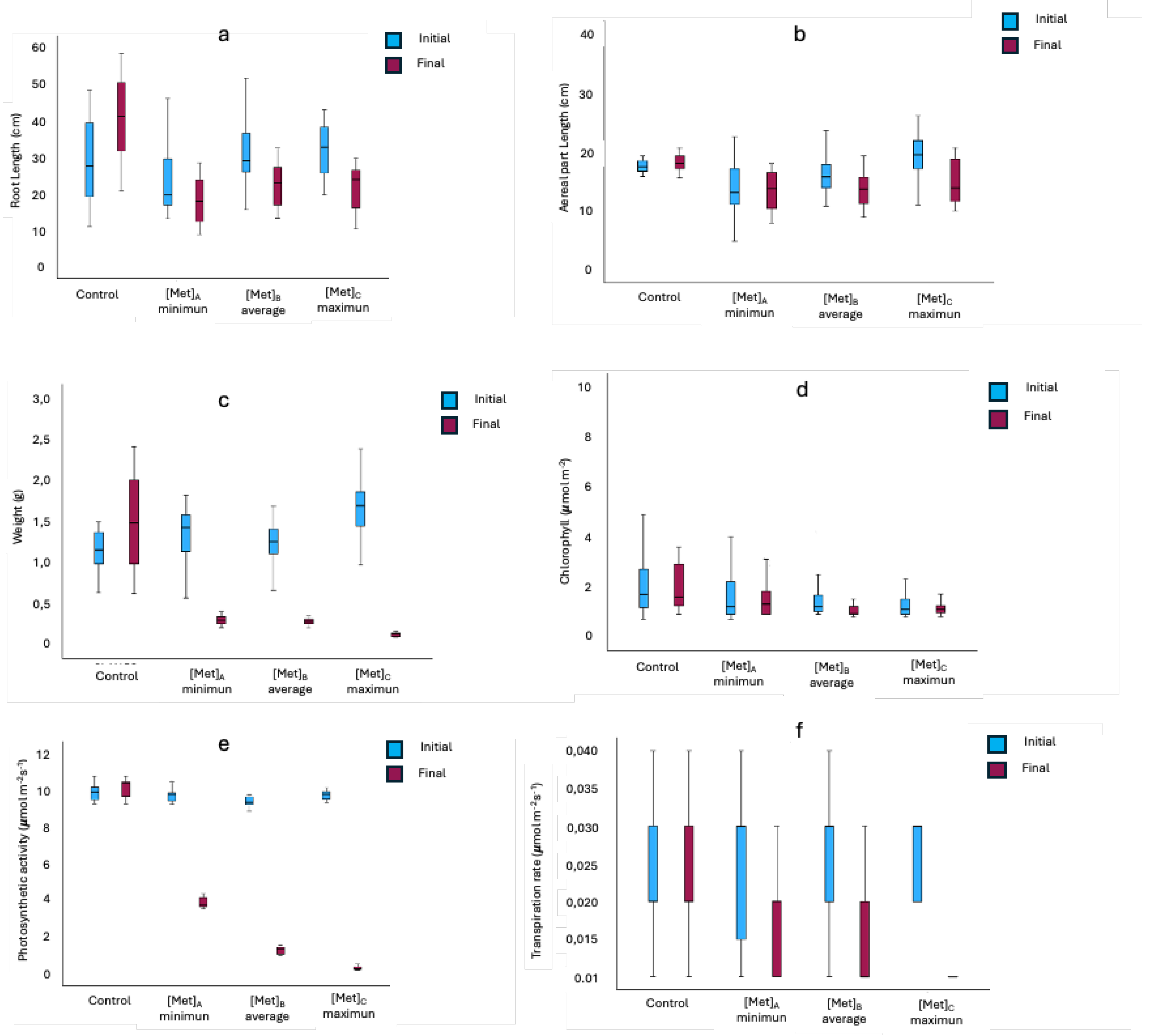
3.1. Morphometry
3.2. Study of Heavy Metal(loid)s Content
| Metal | BAF | FT | Final soil | Root | Aerial | |
| (ppm) | ||||||
| A | Pb | 0.38±0.08 | 0.19±0.06 | 32.93±4.25 | 12.71±1.25 | 2.40±0.32 |
| Zn | 0.18±0.02 | 0.64±0.10 | 216.08±25.30 | 38.60±4.12 | 24.76±4.17 | |
| Tl | 0.20±0.03 | 0.94±0.20 | 0.7±0.06 | 0.14±0.01 | 0.16±0.01 | |
| B | Pb | 0.42±0.12 | 0.15±0.05 | 40.73±15.23 | 16.94±1.23 | 2.5±0.02 |
| Zn | 0.18±0.06 | 0.41±0.15 | 278.72±29.28 | 51.01±8.84 | 21.04±1.96 | |
| Tl | 0.33±0.08 | 0.55±0.15 | 0.82±0.06 | 0.27±0.01 | 0.15±0.01 | |
| C | Pb | 0.25±0.02 | 0.88±0.20 | 1022.21±133.87 | 253.4±33.66 | 223.22±18.84 |
| Zn | 0.11±0.01 | 0.39±0.10 | 607.14±45.23 | 68.93±9.47 | 27.26±3.42 | |
| Tl | 0.25±0.02 | 0.63±0.17 | 493.01±39.39 | 123.41±9.87 | 77.42±9.63 | |
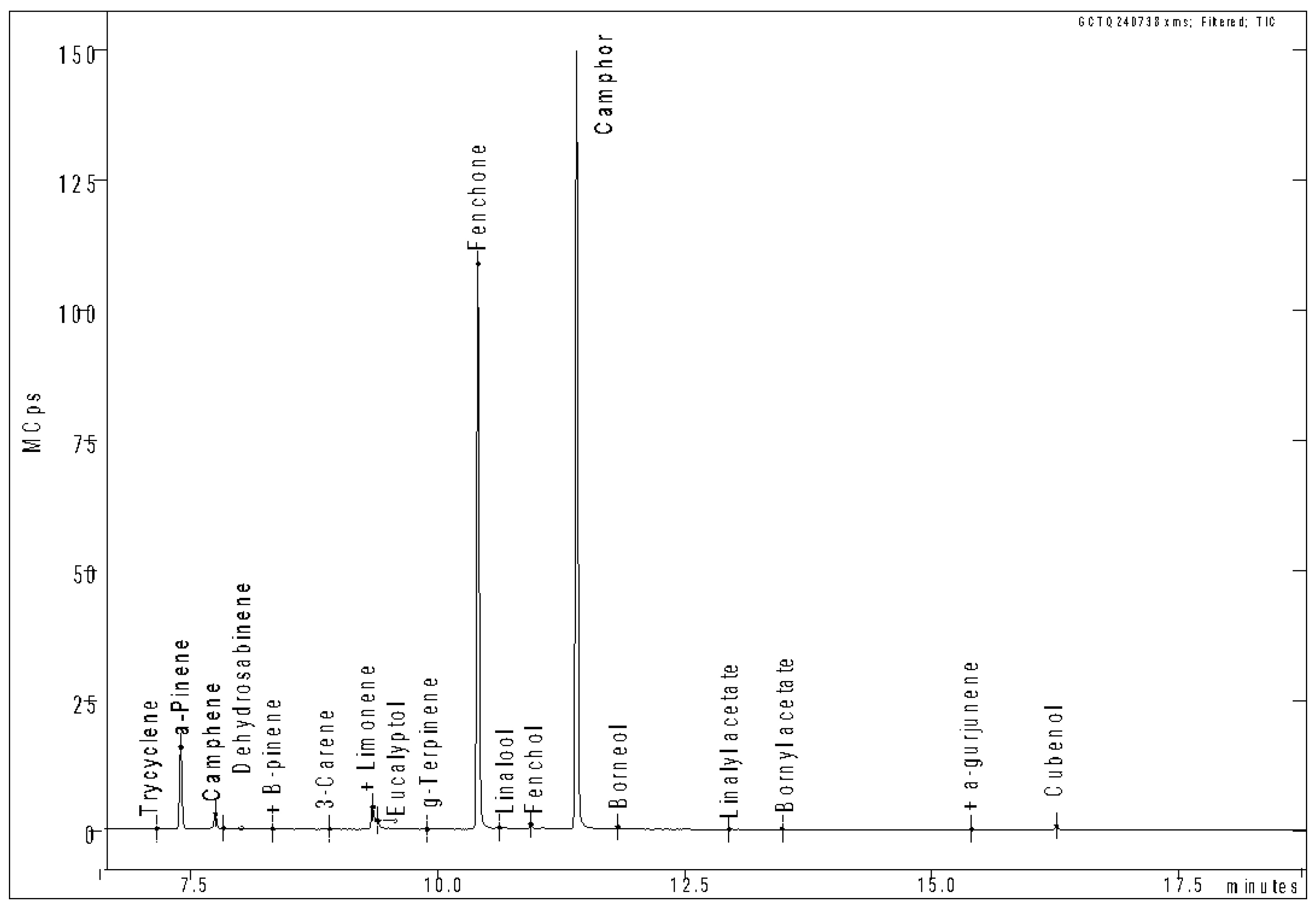
3.3. Essential Oil Extraction
| Extraction | Biomass (g) | Essential oil (mL) | Yield (%) |
| 1 | 589.88 | 3.00 | 0.47 |
| 2 | 434.75 | 1.96 | 0.42 |
| 3 | 489.98 | 2.00 | 0.38 |
3.4. Biochar Characterization
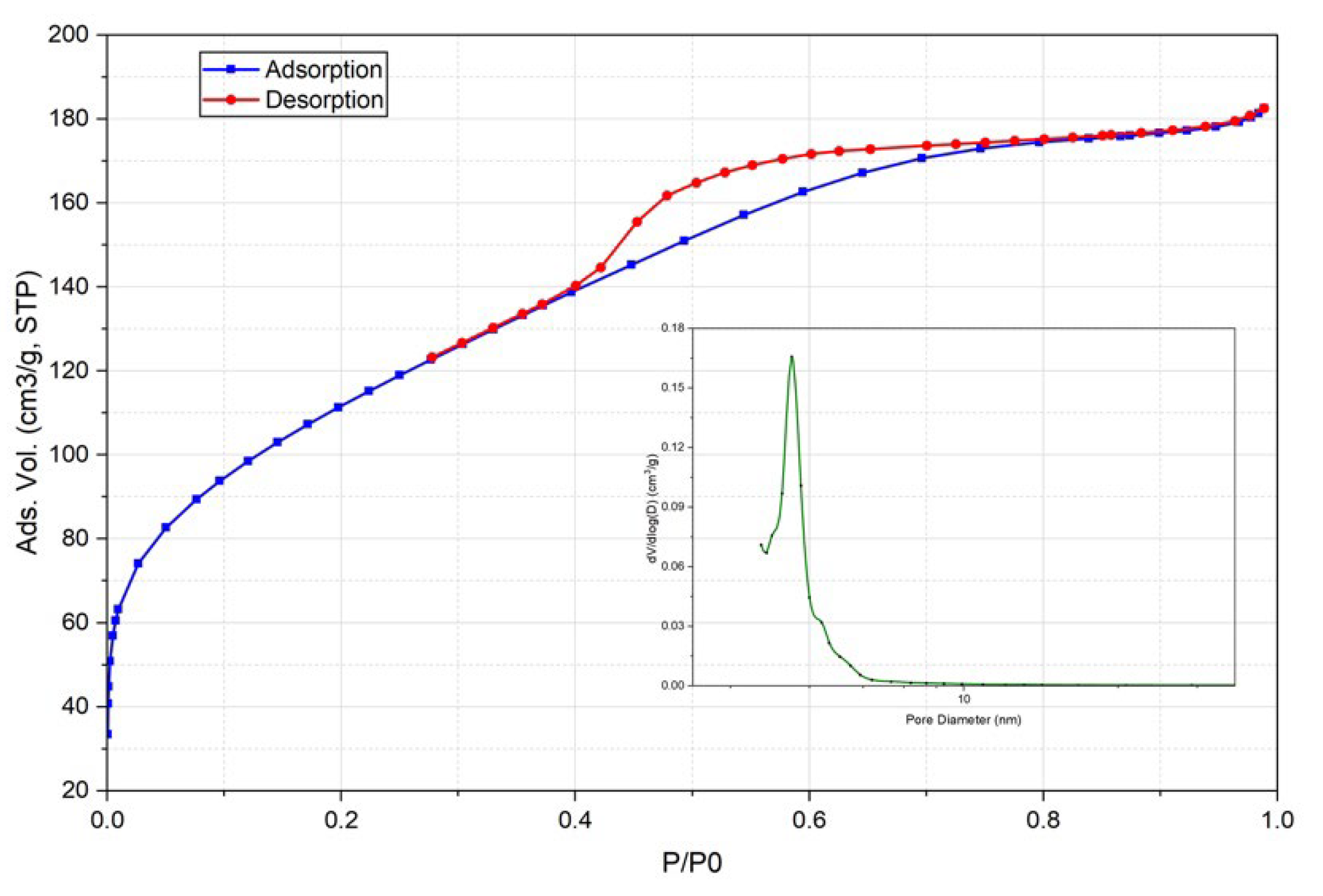
3.4.1. Textural Characterization of Biochar (N2 Adsorption)
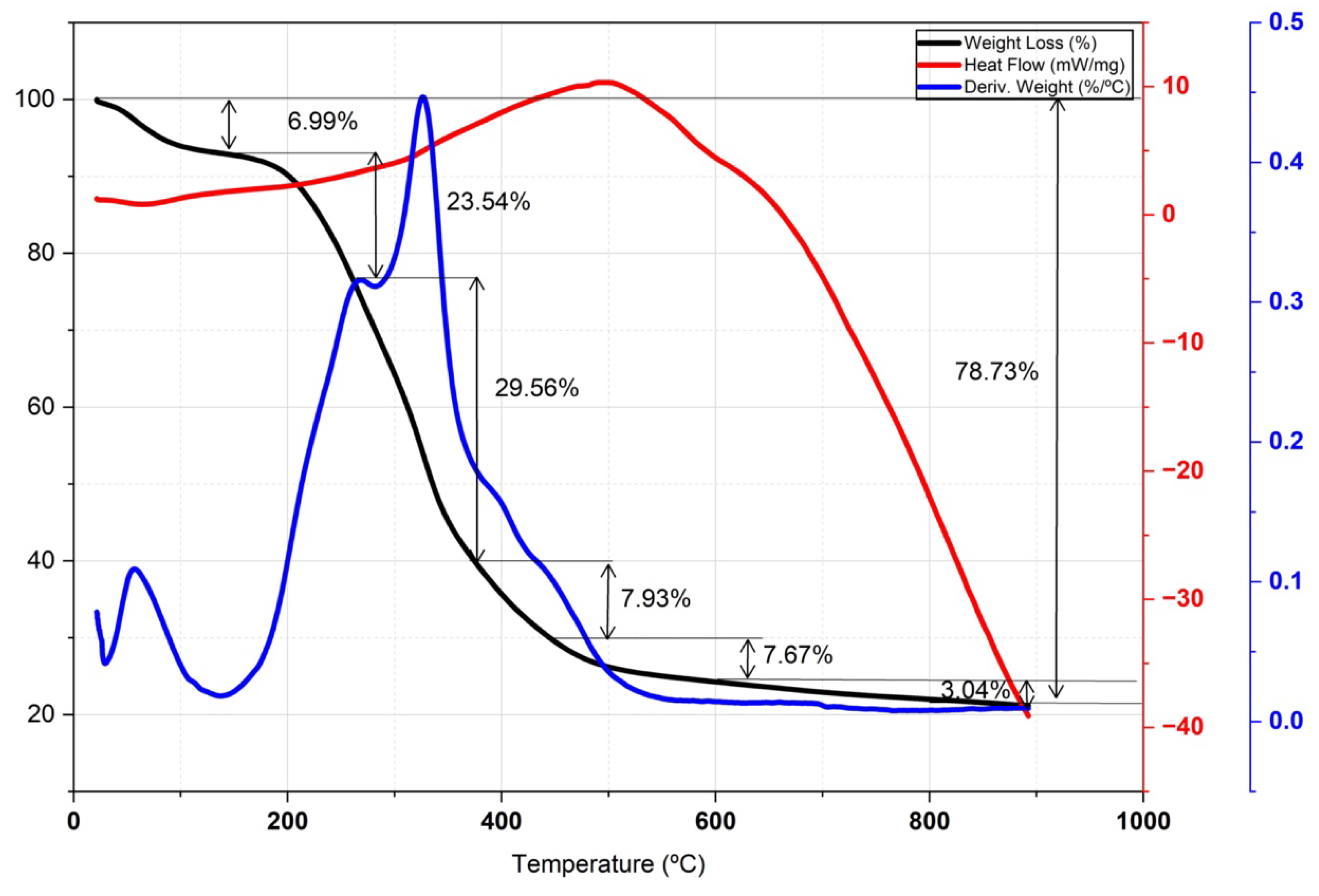
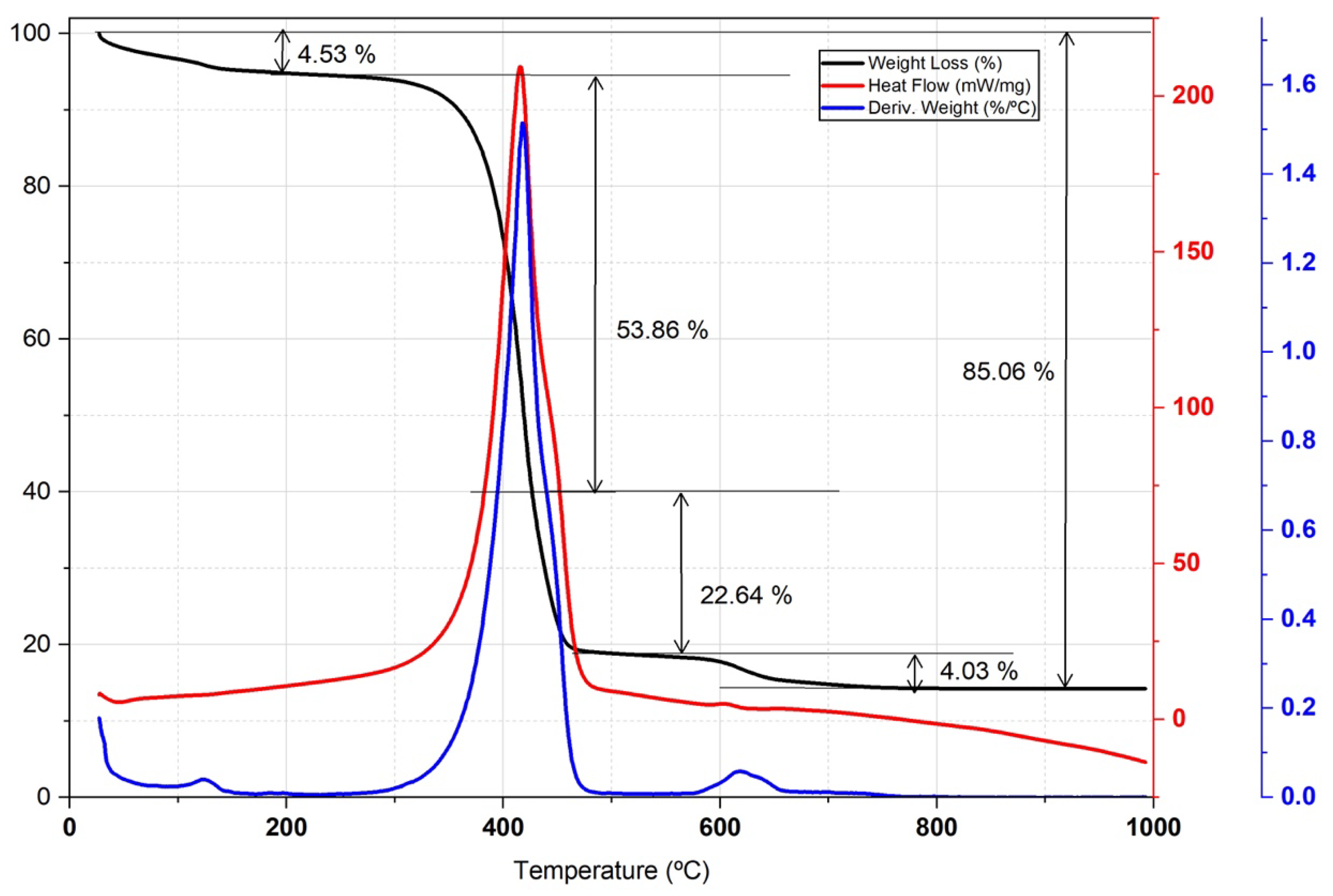
3.4.2. Thermal Characterization (Atd-Tg)
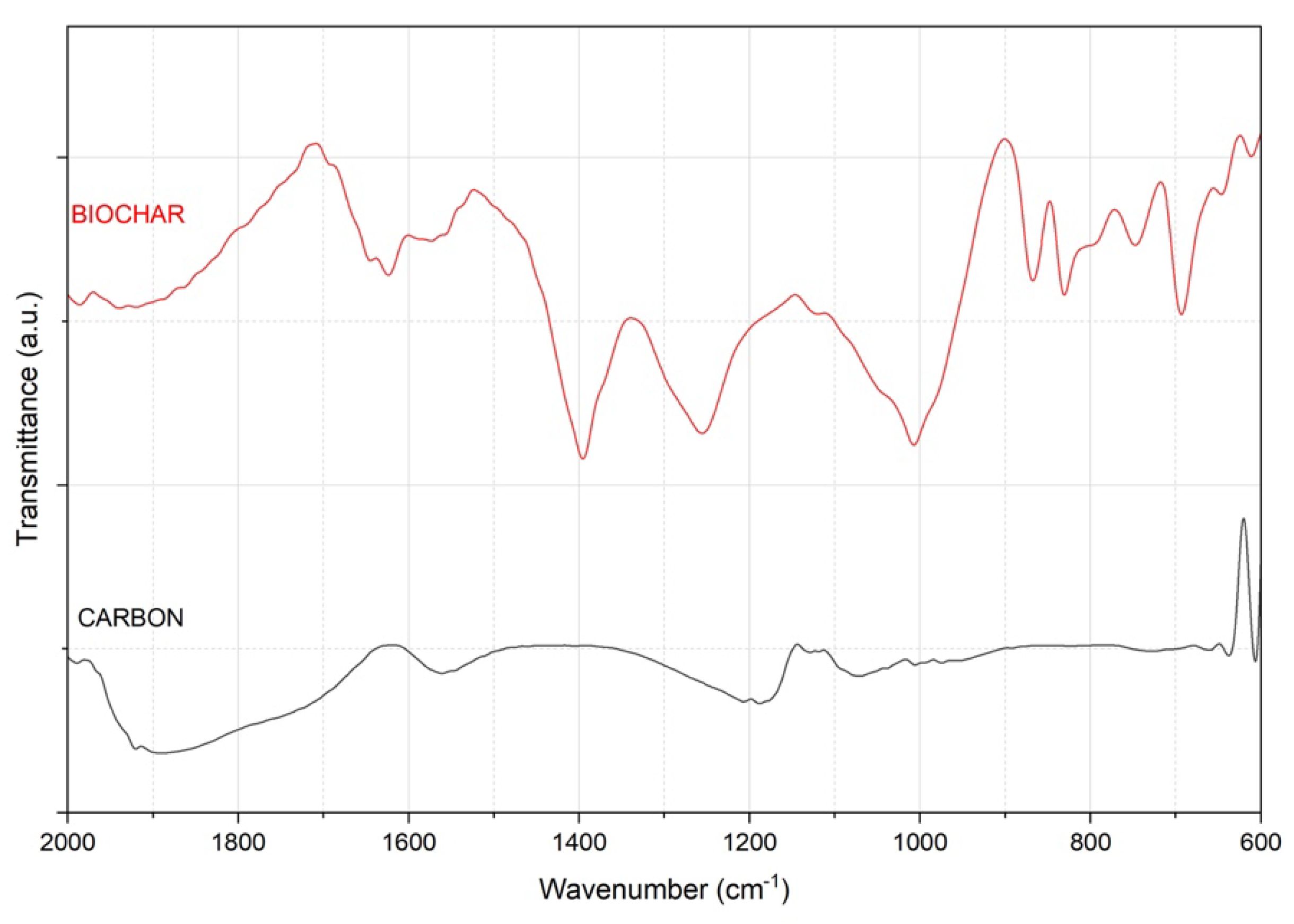
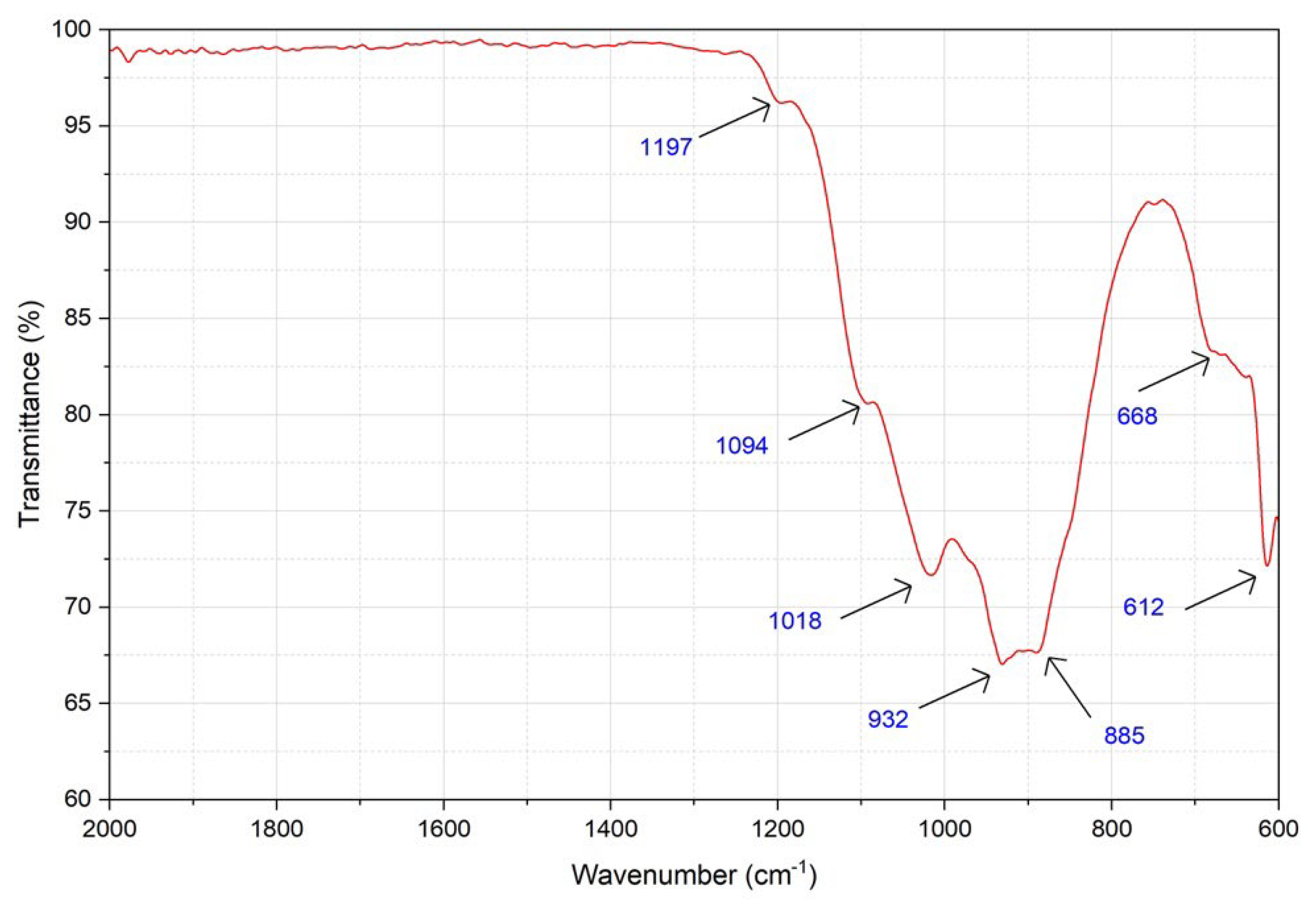
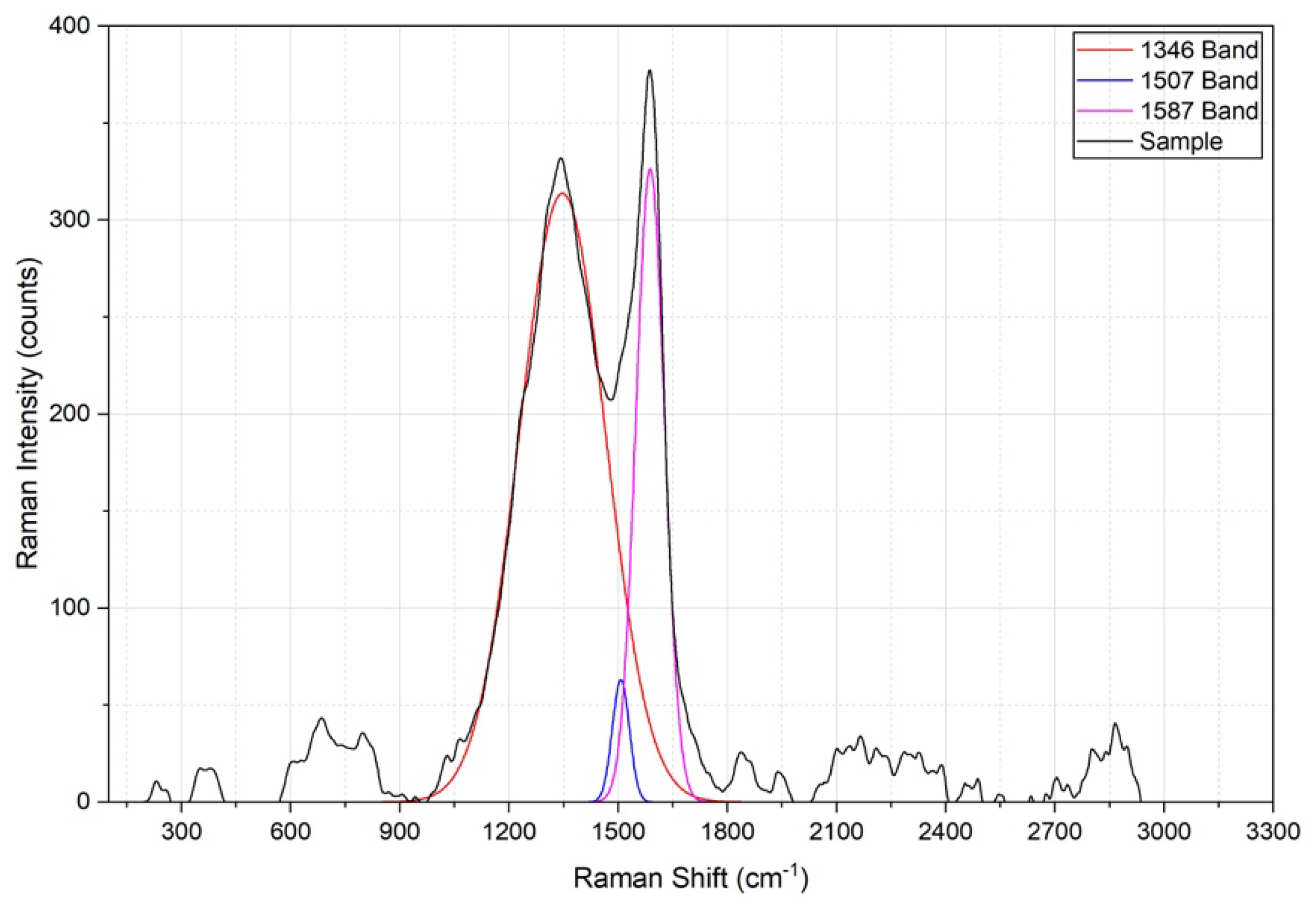
3.4.3. Structural Characterization
3.5. Application of Biochar in Electrodes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, R.A.H.; Bradshaw, A.D. The Use of Metal Tolerant Plant Populations for the Reclamation of Metalliferous Wastes. Journal of Applied Ecology 1979, 16, 595–612. [CrossRef]
- Sun, Z.; Hu, Y.; Cheng, H. Public Health Risk of Toxic Metal(Loid) Pollution to the Population Living near an Abandoned Small-Scale Polymetallic Mine. Science of The Total Environment 2020, 718, 137434. [CrossRef]
- Eurostat Objetivos de Desarrollo Sostenible Available online: https://ec.europa.eu/commission/presscorner/detail/es/ip_23_2887 (accessed on 8 July 2025).
- González-Morales, M.; Rodríguez-González, M.Á.; Fernández-Pozo, L. Status of Ecosystem Services in Abandoned Mining Areas in the Iberian Peninsula: Management Proposal. Toxics 2023, 11, 275. [CrossRef]
- González-Morales, M.; Fernández-Pozo, L.; Rodríguez-González, M.Á. Threats of Metal Mining on Ecosystem Services. Conservation Proposals. Environmental Research 2022, 214, 114036. [CrossRef]
- Oropesa, A.-L.; Gala, J.-A.; Fernandez-Pozo, L.; Cabezas, J.; Soler, F. Lead Content in Soils and Native Plants near an Abandoned Mine in a Protected Area of South-Western Spain: An Approach to Determining the Environmental Risk to Wildlife and Livestock. Environ Sci Pollut Res 2019, 26, 30386–30398. [CrossRef]
- Palma, P.; López-Orozco, R.; Mourinha, C.; Oropesa, A.L.; Novais, M.H.; Alvarenga, P. Assessment of the Environmental Impact of an Abandoned Mine Using an Integrative Approach: A Case-Study of the “Las Musas” Mine (Extremadura, Spain). Science of The Total Environment 2019, 659, 84–94. [CrossRef]
- Madouh, T.A. Eco-Physiological Responses of Native Desert Plant Species to Drought and Nutritional Levels: Case of Kuwait. Front. Environ. Sci. 2022, 10. [CrossRef]
- Díaz, O.G.E.; Fiegehen, L.E.G.; Guajardo, D.C.; Aguayo, S.N. Condicionantes de la retención estudiantil en “escuelas de segunda oportunidad” en Chile. Revista Latinoamericana de Ciencias Sociales, Niñez y Juventud 2019, 17, 1–27. [CrossRef]
- Kumar Rai, P.; Singh, J.S. Invasive Alien Plant Species: Their Impact on Environment, Ecosystem Services and Human Health. Ecological Indicators 2020, 111, 106020. [CrossRef]
- Mishra, B.; Chandra, M. Evaluation of Phytoremediation Potential of Aromatic Plants: A Systematic Review. Journal of Applied Research on Medicinal and Aromatic Plants 2022, 31, 100405. [CrossRef]
- Ez zoubi, Y.; Bousta, D.; Farah, A. A Phytopharmacological Review of a Mediterranean Plant: Lavandula Stoechas L. Clinical Phytoscience 2020, 6, 9. [CrossRef]
- Carrasco, A.; Ortiz-Ruiz, V.; Martinez-Gutierrez, R.; Tomas, V.; Tudela, J. Lavandula Stoechas Essential Oil from Spain: Aromatic Profile Determined by Gas Chromatography–Mass Spectrometry, Antioxidant and Lipoxygenase Inhibitory Bioactivities. Industrial Crops and Products 2015, 73, 16–27. [CrossRef]
- Domingues, J.; Delgado, F.; Gonçalves, J.C.; Zuzarte, M.; Duarte, A.P. Mediterranean Lavenders from Section Stoechas: An Undervalued Source of Secondary Metabolites with Pharmacological Potential. Metabolites 2023, 13, 337. [CrossRef]
- Dragićević, A.; Pavlović, D.; Tasić-Kostov, M. Hydrolates as Water Phase in Cosmetic Creams: Physicochemical and in Vivo Assessment. Maced. Pharm. Bull. 2023, 69, 125–126. [CrossRef]
- Neves, J.M.; Matos, C.; Moutinho, C.; Queiroz, G.; Gomes, L.R. Ethnopharmacological Notes about Ancient Uses of Medicinal Plants in Trás-Os-Montes (Northern of Portugal). Journal of Ethnopharmacology 2009, 124, 270–283. [CrossRef]
- Livia, W.P.; Plata, Y.; Diaz, W.S.G. Disminución de pasivos ambientales mineros PAM mediante la obtención de espuma de vidrio. Revista Colombiana de Materiales 2021, 9–9. [CrossRef]
- Lv, X.; Shen, W.; Wang, L.; Dong, Y.; Zhang, J.; Xie, Z. A Comparative Study on the Practical Utilization of Iron Tailings as a Complete Replacement of Normal Aggregates in Dam Concrete with Different Gradation. Journal of Cleaner Production 2019, 211, 704–715. [CrossRef]
- Geng, Y.; Cui, D.; Yang, L.; Xiong, Z.; Pavlostathis, S.G.; Shao, P.; Zhang, Y.; Luo, X.; Luo, S. Resourceful Treatment of Harsh High-Nitrogen Rare Earth Element Tailings (REEs) Wastewater by Carbonate Activated Chlorococcum Sp. Microalgae. Journal of Hazardous Materials 2022, 423, 127000. [CrossRef]
- Zhang, T.; Wang, L.; Zhang, Y. Ekoloji. 2018, pp. 1615–1624.
- Zamarreño, R.; Díaz, F. Recuperación de metales económicamente importantes desde relaves mineros abandonados, usando biolixiviación en columnas de fase inversa, de bajo costo y ambientalmente sostenible. Avances en Ciencias e Ingeniería 2021, 12, 31–42.
- Álvarez-Ayuso, E.; Murciego, A.; Rodríguez, M.A.; Mosser-Ruck, R. Cement Encapsulation Processes to Mitigate the Risks Posed by Different Types of Antimony-Bearing Mine Waste. Journal of Cleaner Production 2022, 372, 133671. [CrossRef]
- Kinnunen, P.; Karhu, M.; Yli-Rantala, E.; Kivikytö-Reponen, P.; Mäkinen, J. A Review of Circular Economy Strategies for Mine Tailings. Cleaner Engineering and Technology 2022, 8, 100499. [CrossRef]
- Kuppens, T.; Van Dael, M.; Vanreppelen, K.; Thewys, T.; Yperman, J.; Carleer, R.; Schreurs, S.; Van Passel, S. Techno-Economic Assessment of Fast Pyrolysis for the Valorization of Short Rotation Coppice Cultivated for Phytoextraction. Journal of Cleaner Production 2015, 88, 336–344. [CrossRef]
- Qian, K.; Kumar, A.; Zhang, H.; Bellmer, D.; Huhnke, R. Recent Advances in Utilization of Biochar. Renewable and Sustainable Energy Reviews 2015, 42, 1055–1064. [CrossRef]
- Kang, Z.; Xu, D.; Zhao, L.; Liu, D. Boosting Supercapacitor Performance with High-Specific Surface Area Porous Carbon Derived from Sugarcane Bagasse. Journal of Energy Storage 2024, 104, 114718. [CrossRef]
- Almada, I. Electrodos Para Supercondensadores Obtenidos Por Electrofdeposición, Universidad Autonoma de Madrid, 2015.
- Norouzi, O.; Maria, F.D.; Dutta, A. Biochar-Based Composites as Electrode Active Materials in Hybrid Supercapacitors with Particular Focus on Surface Topography and Morphology. Journal of Energy Storage 2020, 29, 101291. [CrossRef]
- Rawat, S.; Boobalan, T.; Krishna, B.B.; Sathish, M.; Hotha, S.; Bhaskar, T. Biochar for Supercapacitor Application: A Comparative Study. Chemistry – An Asian Journal 2022, 17, e202200982. [CrossRef]
- Andrade, M. das G.; da Boa Morte, E.S.; Batista dos Santos, D.C.M.; Castro, J.T.; Barbosa, J.T.P.; Teixeira, A.P.; Fernandes, A.P.; Welz, B.; dos Santos, W.P.C.; Nunes dos Santos, E.B.G.; et al. Sample Preparation for the Determination of Metals in Food Samples Using Spectroanalytical Methods—A Review. Applied Spectroscopy Reviews 2008, 43, 67–92. [CrossRef]
- Palacios, O.F.; Zúñiga, L.E. Extracción del aceite esencial de Lavanda (lavándula angustifolia), mediante la metodología de arrastre de vapor. Trabajo Fin de Estudios (Ecuador). Trabajo Fin de Estudios. Universidad Ecuador 2022.
- Nazem, V.; Sabzalian, M.R.; Saeidi, G.; Rahimmalek, M. Essential Oil Yield and Composition and Secondary Metabolites in Self- and Open-Pollinated Populations of Mint (Mentha Spp.). Industrial Crops and Products 2019, 130, 332–340. [CrossRef]
- Hossain, M.A.; Piyatida, P.; da Silva, J.A.T.; Fujita, M. Molecular Mechanism of Heavy Metal Toxicity and Tolerance in Plants: Central Role of Glutathione in Detoxification of Reactive Oxygen Species and Methylglyoxal and in Heavy Metal Chelation. Journal of Botany 2012, 2012, 872875. [CrossRef]
- Srivastava, R.K.; Pandey, P.; Rajpoot, R.; Rani, A.; Dubey, R.S. Cadmium and Lead Interactive Effects on Oxidative Stress and Antioxidative Responses in Rice Seedlings. Protoplasma 2014, 251, 1047–1065. [CrossRef]
- Garrido, I.; Ortega, A.; Hernández, M.; Fernández-Pozo, L.; Cabezas, J.; Espinosa, F. Effect of Antimony in Soils of an Sb Mine on the Photosynthetic Pigments and Antioxidant System of Dittrichia Viscosa Leaves. Environ Geochem Health 2021, 43, 1367–1383. [CrossRef]
- Sierra, M.J.; Millán, R.; Esteban, E. Mercury Uptake and Distribution in Lavandula Stoechas Plants Grown in Soil from Almadén Mining District (Spain). Food and Chemical Toxicology 2009, 47, 2761–2767. [CrossRef]
- Arenas-Lago, D.; Carvalho, L.C.; Santos, E.S.; Abreu, M.M. Influence of Seed Source and Soil Contamination on Ecophysiological Responses of Lavandula Pedunculata in Rehabilitation of Mining Areas. Plants 2022, 11, 105. [CrossRef]
- Çolak, S.; Yilmaz, Ş.B.A.; Öztekin, E. Bioaccumulation Factors of Heavy Metal(Loid)s in Some Medicinal and Aromatic Plants Species: Example of Zonguldak/Türkiye. Water Air Soil Pollut 2023, 234, 522. [CrossRef]
- Angelova, V.R.; Grekov, D.F.; Kisyov, V.K.; Ivanov, K.I. Potential of Lavender (Lavandula Vera L.) for Phytoremediation of Soils Contaminated with Heavy Metals. International J. of Agricultural and Biosystems Engineering 2015, 9.
- Bella, S.L.; Tuttolomondo, T.; Dugo, G.; Ruberto, G.; Leto, C.; Napoli, E.M.; Potortì, A.G.; Fede, M.R.; Virga, G.; Leone, R.; et al. Composition and Variability of the Essential Oil of the Flowers of Lavandula Stoechas from Various Geographical Sources. Natural Product Communications 2015, 10, 1934578X1501001150. [CrossRef]
- Şahinler, S.Ş.; Yılmaz, B.S.; Sarıkürkcü, C.; Tepe, B. The Importance of Lavandula Stoechas L. in Pharmacognosy and Phytotherapy. Int. J. Sec. Metabolite 2022, 9, 360–376. [CrossRef]
- Rufino, A.T.; Ribeiro, M.; Sousa, C.; Judas, F.; Salgueiro, L.; Cavaleiro, C.; Mendes, A.F. Evaluation of the Anti-Inflammatory, Anti-Catabolic and pro-Anabolic Effects of E-Caryophyllene, Myrcene and Limonene in a Cell Model of Osteoarthritis. European Journal of Pharmacology 2015, 750, 141–150. [CrossRef]
- Sharma, M.; Grewal, K.; Jandrotia, R.; Batish, D.R.; Singh, H.P.; Kohli, R.K. Essential Oils as Anticancer Agents: Potential Role in Malignancies, Drug Delivery Mechanisms, and Immune System Enhancement. Biomedicine & Pharmacotherapy 2022, 146, 112514. [CrossRef]
- Reyes-Jurado, F.; Navarro-Cruz, A.R.; Ochoa-Velasco, C.E.; Palou, E.; López-Malo, A.; Ávila-Sosa, R. Essential Oils in Vapor Phase as Alternative Antimicrobials: A Review. Crit Rev Food Sci Nutr 2020, 60, 1641–1650. [CrossRef]
- Blažeković, B.; Vladimir-Knežević, S.; Brantner, A.; Štefan, M.B. Evaluation of Antioxidant Potential of Lavandula x Intermedia Emeric Ex Loisel. “Budrovka”: A Comparative Study with L. Angustifolia Mill. Molecules 2010, 15, 5971–5987. [CrossRef]
- Di Sotto, A.; Mazzanti, G.; Carbone, F.; Hrelia, P.; Maffei, F. Genotoxicity of Lavender Oil, Linalyl Acetate, and Linalool on Human Lymphocytes in Vitro. Environmental and Molecular Mutagenesis 2011, 52, 69–71. [CrossRef]
- Zheljazkov, V.D.; Craker, L.E.; Xing, B. Effects of Cd, Pb, and Cu on Growth and Essential Oil Contents in Dill, Peppermint, and Basil. Environmental and Experimental Botany 2006, 58, 9–16. [CrossRef]
- Gautam, M.; Agrawal, M. Influence of Metals on Essential Oil Content and Composition of Lemongrass (Cymbopogon Citratus (D.C.) Stapf.) Grown under Different Levels of Red Mud in Sewage Sludge Amended Soil. Chemosphere 2017, 175, 315–322. [CrossRef]
- Gonzalez-Rivera, J.; Campanella, B.; Pulidori, E.; Bramanti, E.; Tiné, M.R.; Bernazzani, L.; Onor, M.; Bàrberi, P.; Duce, C.; Ferrari, C. From Volatiles to Solid Wastes: Towards the Full Valorization of Lavender and Rosemary by Simultaneous in Situ Microwaves and Ultrasounds Irradiation Extraction. Industrial Crops and Products 2023, 194, 116362. [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure and Applied Chemistry 2015, 87, 1051–1069. [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Journal of the American Chemical Society. 1938,.
- Barret, E.P.; Joyner, L.G.; Halenda, P.P. Journal of the American Chemical Society. 1951,.
- Li, S.; Chen, G. Thermogravimetric, Thermochemical, and Infrared Spectral Characterization of Feedstocks and Biochar Derived at Different Pyrolysis Temperatures. Waste Management 2018, 78, 198–207. [CrossRef]
- Kawakami, M.; Kanba, H.; Sato, K.; Takenaka, T.; Gupta, S.; Chandratilleke, R.; Sahajwalla, V. Characterization of Thermal Annealing Effects on the Evolution of Coke Carbon Structure Using Raman Spectroscopy and X-Ray Diffraction. ISIJ Int. 2006, 46, 1165–1170. [CrossRef]
- Chu, H.; Lu, Z.; Man, M.; Song, S.; Zhang, H.; Cheng, J.; Zhao, X.; Duan, J.; Chen, X.; Zhu, Y. Hard Carbon-Based Electrode Boosts the Performance of a Solid-State Symmetric Supercapacitor. Journal of Energy Storage 2024, 76, 109660. [CrossRef]
- Barroso Bogeat, A. Understanding and Tuning the Electrical Conductivity of Activated Carbon: A State-of-the-Art Review. Critical Reviews in Solid State and Materials Sciences 2021, 46, 1–37. [CrossRef]
- Moliner, R. Bol Grupo Español del Carbón. 2016,.
- Eliad, L.; Pollak, E.; Levy, N.; Salitra, G.; Soffer, A.; Aurbach, D. Appl. Phys. A. 2006,.
- Jo, K.; Ha, J.; Ryu, J.; Lee, E.; Lee, H. DC 4-Point Measurement for Total Electrical Conductivity of SOFC Cathode Material. Applied Sciences 2021, 11, 4963. [CrossRef]
| Month | Ja | Feb | March | Apr | May | Jun | Jul | Aug | Sep | Oct | Nov | Dec |
| mL | 187 | 224 | 268 | 268 | 121 | 41 | 13 | 11 | 121 | 227 | 293 | 211 |
| Metal |
Initial soil dosage (ppm) |
Final soil (%) |
Root (%) |
Aerial (%) |
Leachate (%) |
|
|
Control |
Pb | 0.01 | 100 | 0 | 0 | 0 |
| Zn | 1.60 | 25.62 | 28.12 | 38.12 | 8.12 | |
| Tl | 0.04 | 25 | 0 | 75 | 0.00 | |
| A | Pb | 48.18 | 68.34 | 26.38 | 4.98 | 0.29 |
| Zn | 281.34 | 76.80 | 13.72 | 8.80 | 0.67 | |
| Tl | 1 | 70 | 14 | 16 | 0 | |
| B | Pb | 60.26 | 67.59 | 28.11 | 4.14 | 0.15 |
| Zn | 352.79 | 79 | 14.45 | 0.06 | 0.57 | |
| Tl | 1.25 | 65.60 | 21.60 | 12.00 | 0.80 | |
| C | Pb | 1500.43 | 68.13 | 16.89 | 14.88 | 0.11 |
| Zn | 706.61 | 85.92 | 9.75 | 3.86 | 0.46 | |
| Tl | 700.62 | 70.37 | 17.61 | 11.05 | 0.97 |
| Pb (ppm) | Zn (ppm) | Tl (ppm) | |
| Essential oil | n.d. | n.d. | n.d. |
| Hydrolat | n.d. | n.d. | n.d. |
| Biomass after distillation | 173±14 | 307±11 | 195±15 |
| Residual water after distillation | 0.02±0.01 | 0.96 ±0.04 | 0.04±0.02 |
| Mine soil | 254.6±183.6 | 367.4±212.4 | 314.9±95.4 |
| L. stoechas mine (leaves and flower) | 185.3±92.8 | 339.8±141.7 | 216.0±85.1 |
| Al2O3 | SiO2 | Fe2O3 | CaO | K2O | ZnO | SrO | Na2O | P2O5 | MgO | TiO2 | Pb2O3 | MnO | SO3 | |
| LMA % | 6.5 | 25.8 | 2.30 | 31.1 | 10.3 | 0.30 | 0.33 | 2.41 | 5.64 | 10.2 | 036 | 0,003 | 0.31 | 4.26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




