Submitted:
18 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Physical integration between endosymbiotic algae and host mitochondria is a recurring feature across photosynthetic symbioses, yet the structural nature of this association has remained unresolved. In the ciliate Paramecium bursaria, each endosymbiotic Chlorella cell is enclosed by a perialgal vacuole (PV) membrane consistently surrounded by host mitochondria, suggesting a conserved architecture for metabolic interaction. Although transmission electron microscopy has shown close membrane apposition, it has remained unclear whether this reflects incidental proximity or a reinforced adhesion. Here, we provide direct evidence that the PV membrane and host mitochondrial membrane form a stable physical association. Using discontinuous Percoll density-gradient centrifugation, we isolated intact units in which Chlorella and mitochondria co-sedimented, indicating that their association withstands mechanical disruption. By fluorescently labeling the PV and mitochondrial membranes with BODIPY FL C5-ceramide complexed to BSA, together with a mitochondria-specific monoclonal antibody and DAPI, we visualized the PV membrane under light microscopy and demonstrated that the mitochondrial–PV membrane complex persists after homogenization and centrifugation. As expected from the membrane-insertion behavior of BC5C, this fluorescent labeling revealed that the PV–mitochondrial membrane association is structurally reinforced rather than incidental, providing a mechanistic framework for understanding how Chlorella cells are stably positioned beneath the host cortex.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Strains and Cultures
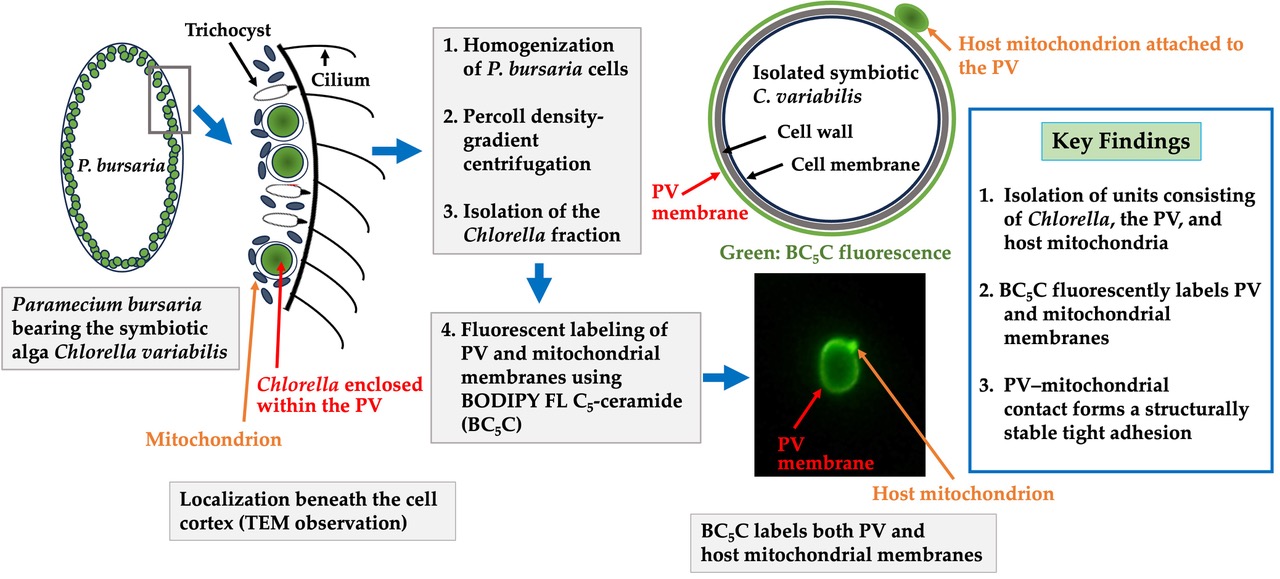
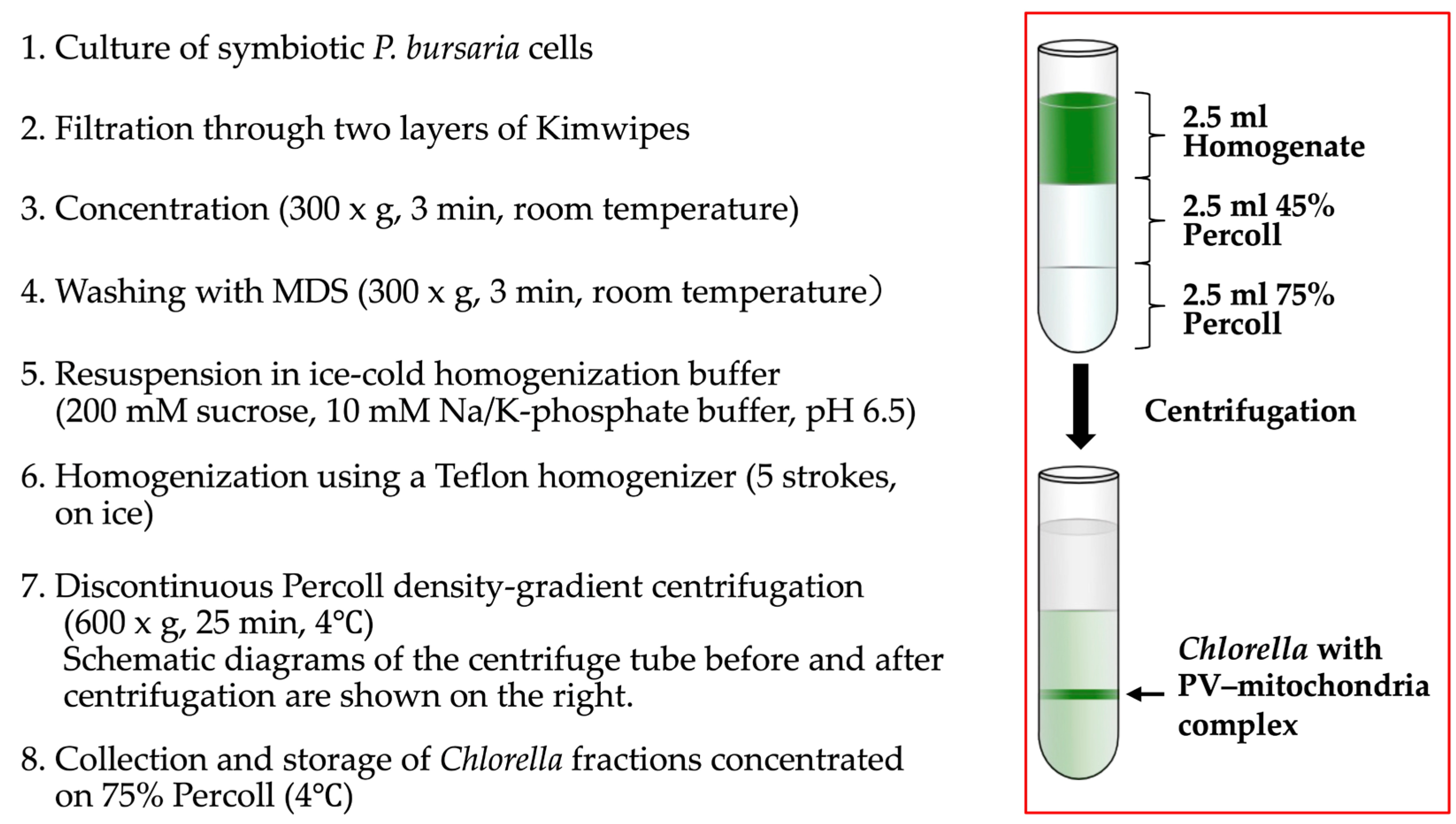
2.2. Isolation of Symbiotic Algae Possessing PV Membranes and Mitochondria by Discontinuous Percoll Density-Gradient Centrifugation
2.3. Routine Isolation of Symbiotic Chlorella
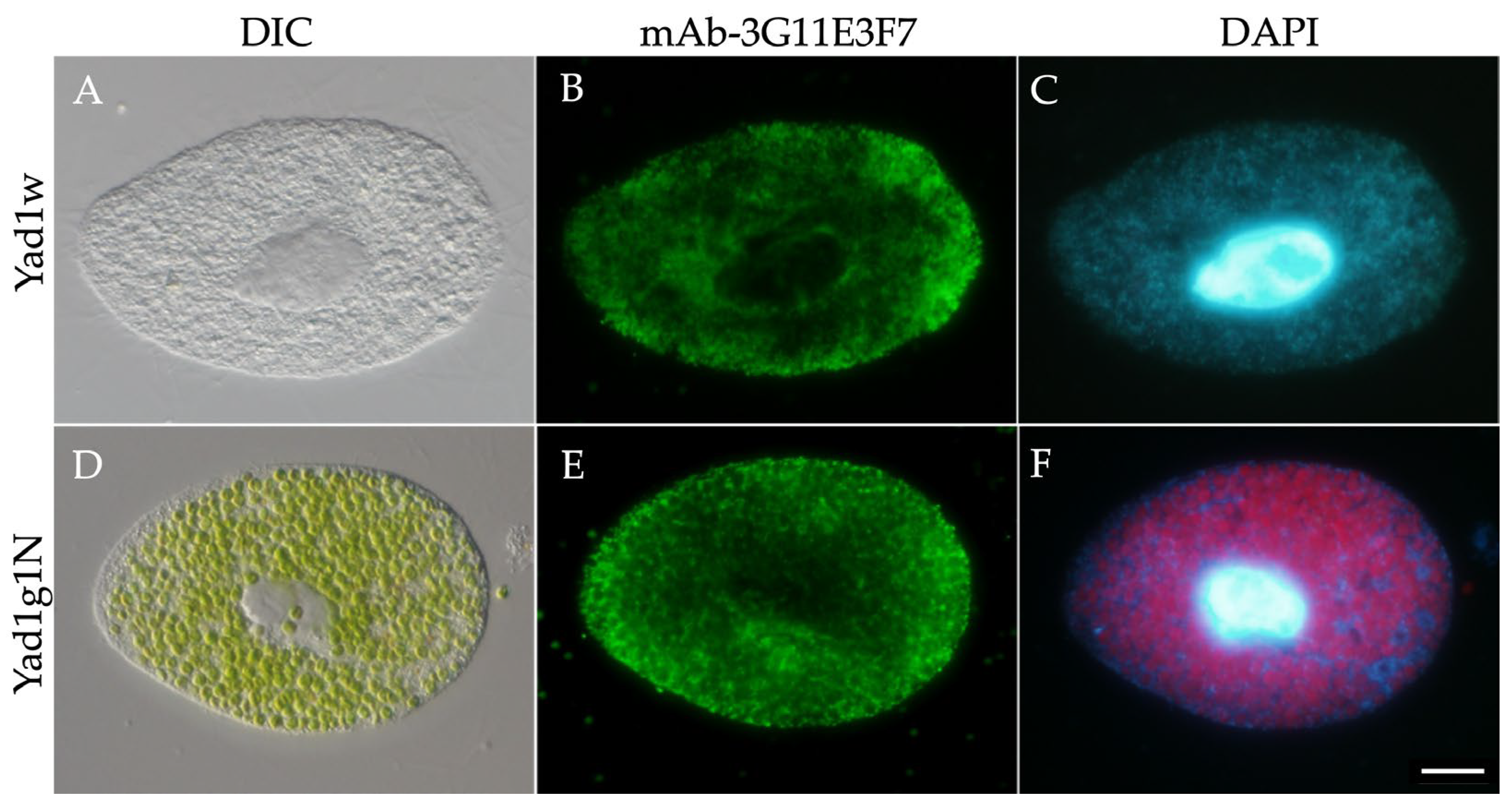
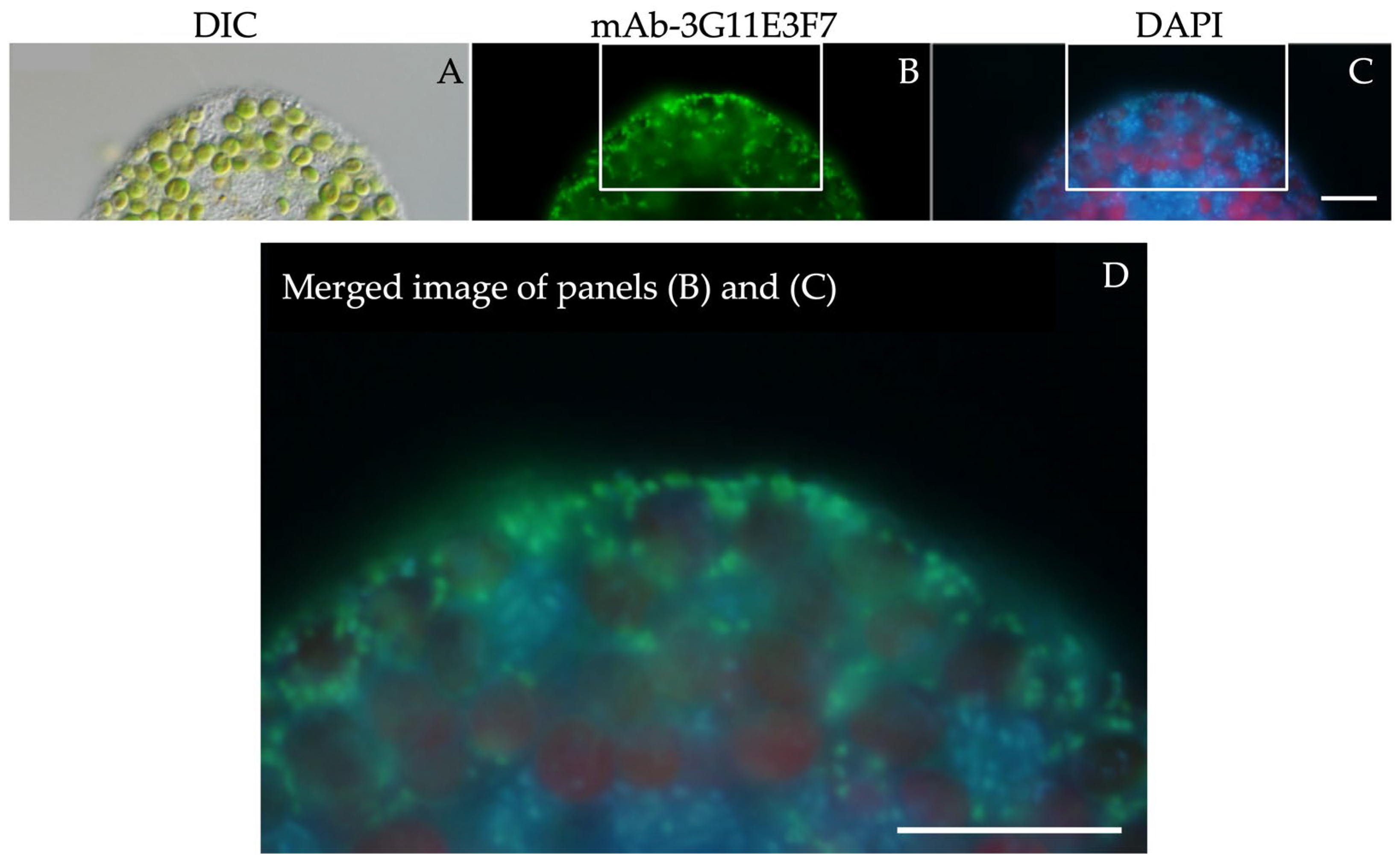
2.4. Production of Monoclonal Antibodies Against P. bursaria Mitochondria
2.5. Indirect Immunofluorescence Microscopy
2.6. Fluorescent Staining of the PV Membrane and Mitochondrial Membrane
3. Results
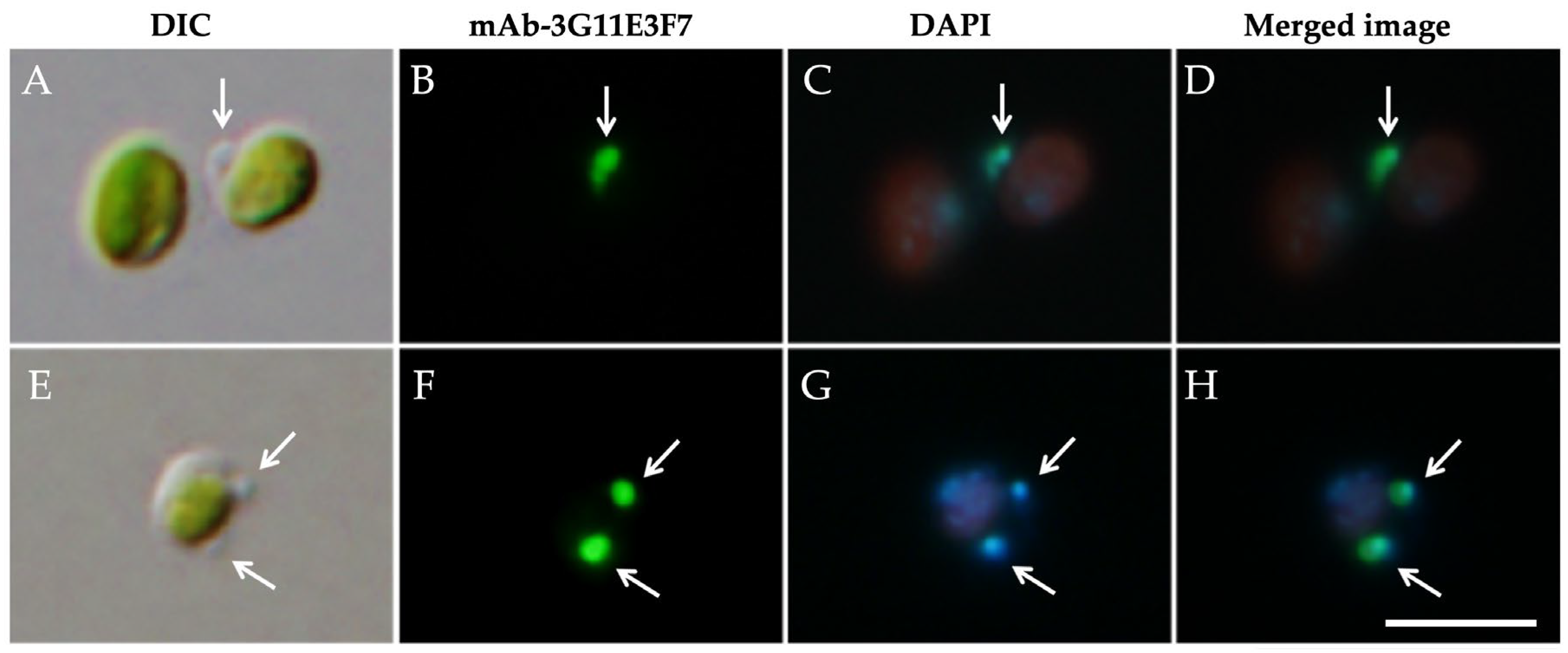
3.1. Intracellular Distribution of Mitochondria
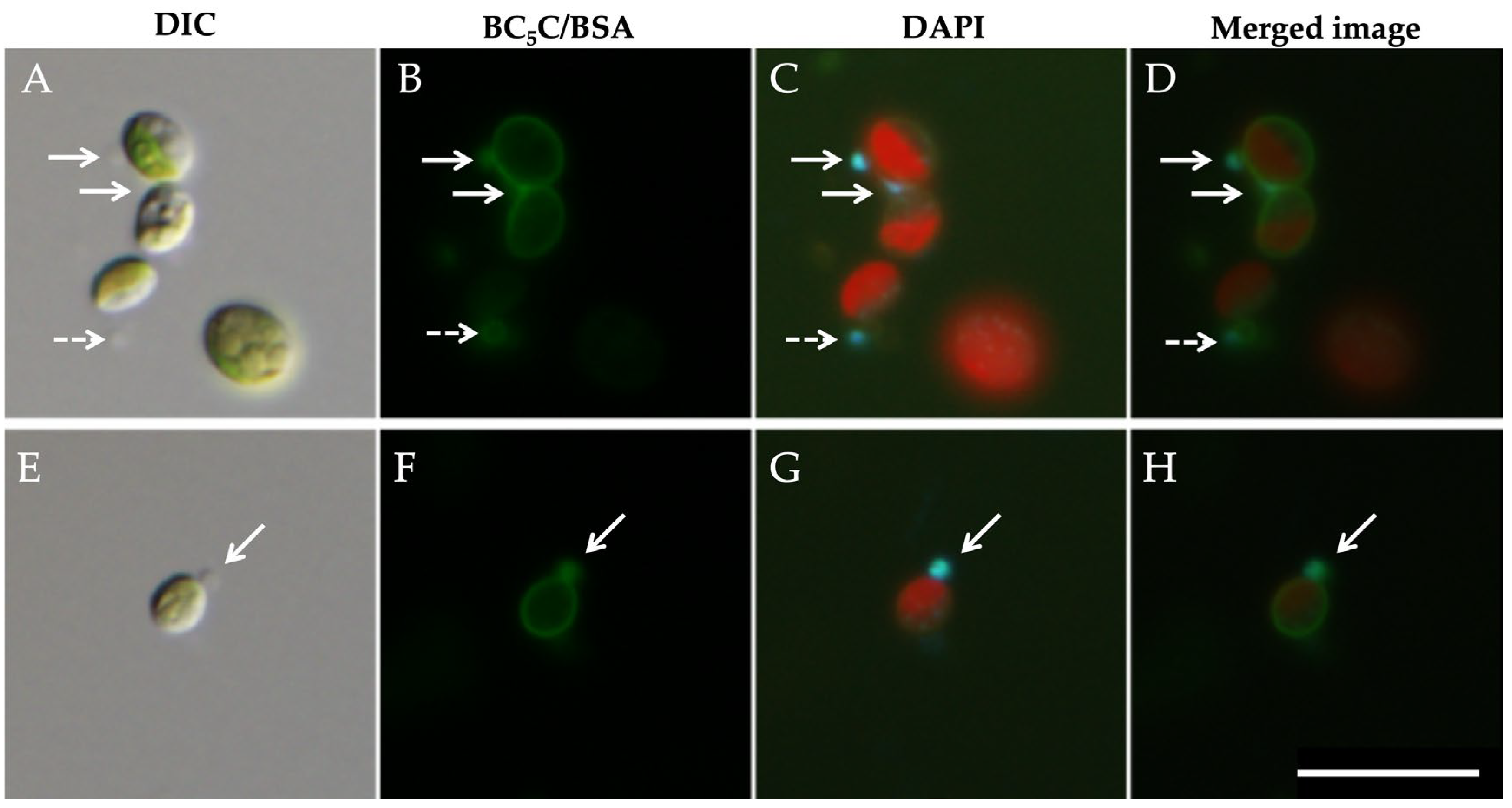
3.2. Isolation of Symbiotic Algae Possessing PV Membranes and Mitochondria from Homogenates of Symbiotic P. bursaria
3.3. The Attachment Between Endosymbiotic Chlorella and Host Mitochondria Is Mediated by the PV Membrane
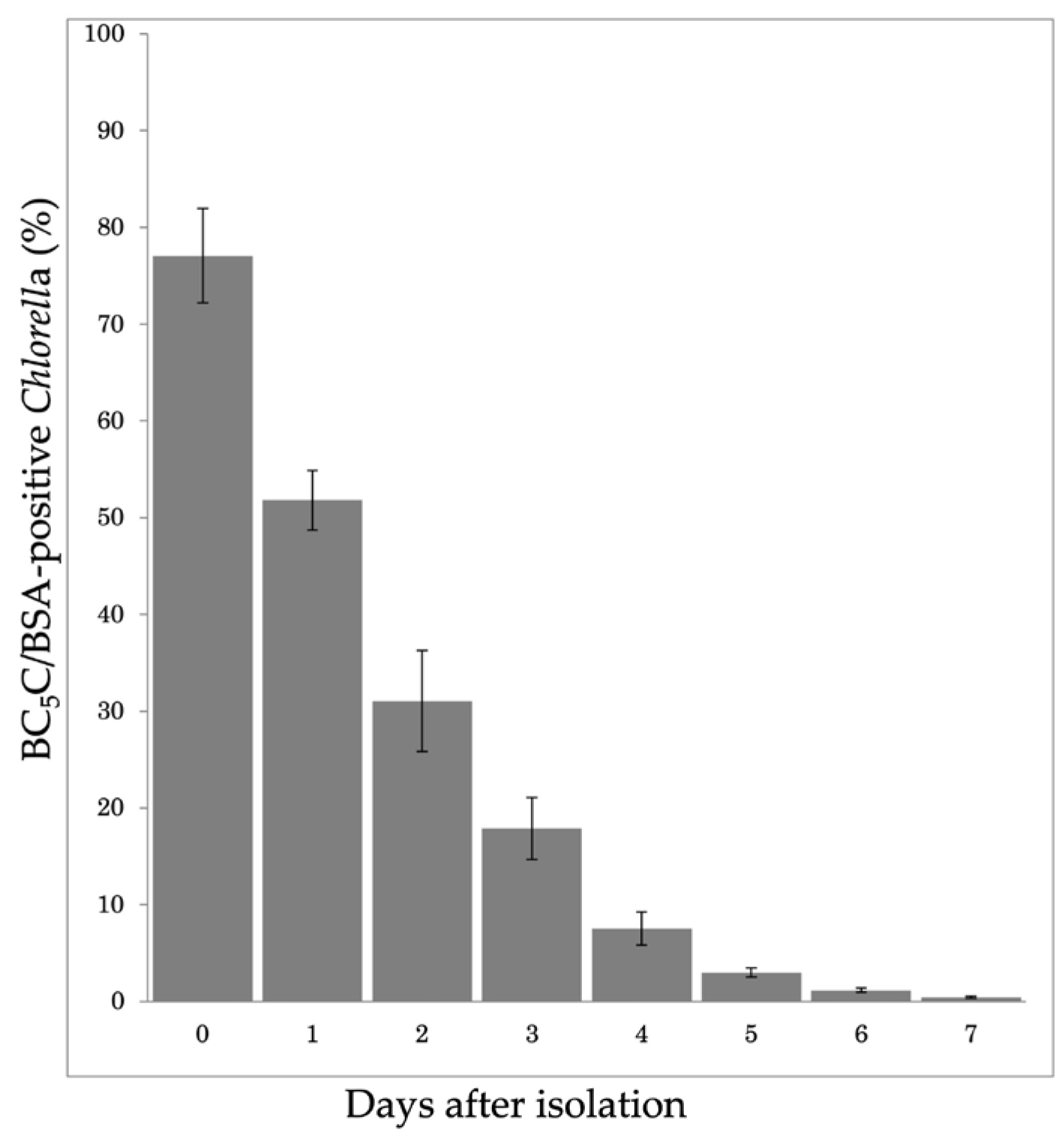
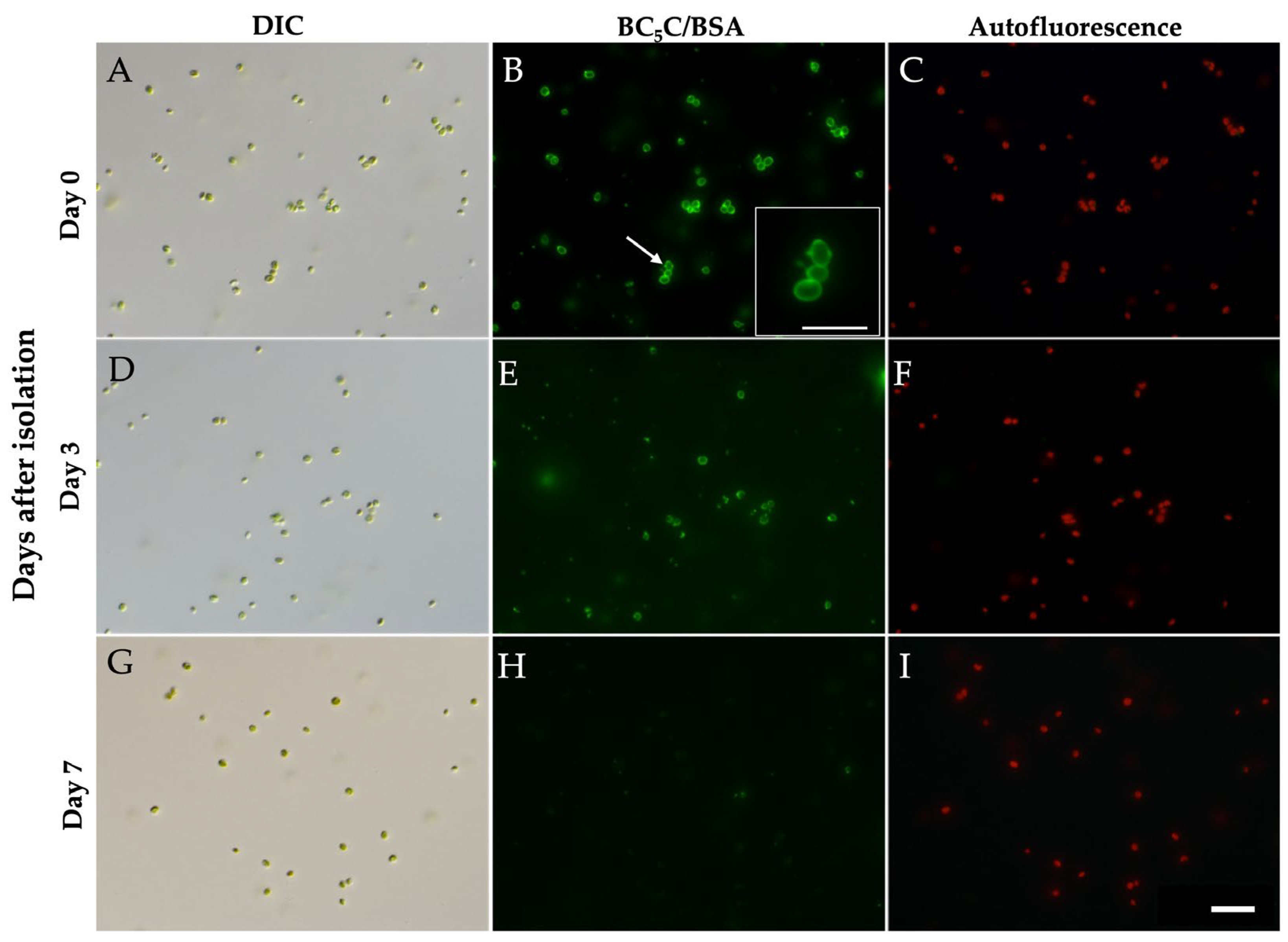
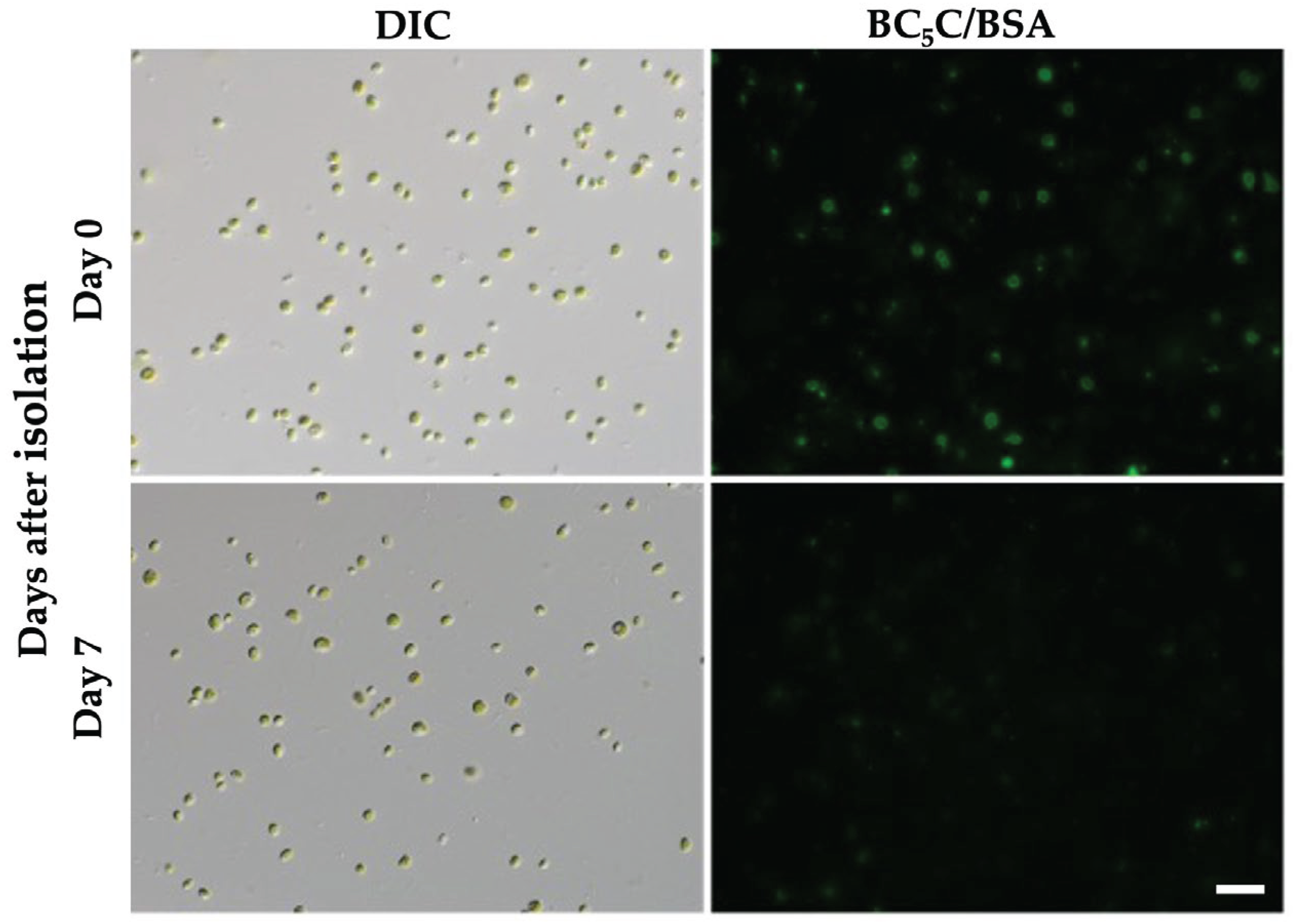
3.4. Stability of PV Membranes After Isolation by Discontinuous Percoll Density-Gradient Centrifugation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PV | Perialgal Vacuole |
| TEM | Transmission Electron Microscope |
| DV | Digestive Vacuole |
| AcPase | Acid Phosphatase Activity |
| ER | Endoplasmic Reticulum |
| MDS | Modified Dryl’s Solution |
| NBRP | National BioResource Project |
| SIP | Stock Isotonic Percoll |
| PBS | Phosphate-Buffered Saline |
| PBST | PBS-Containing 0.05% (v/v) Tween 20 |
| DAPI | 4’,6-DiAmidino-2-PhenolIndole |
| DIC | Differential Interference Contrast |
| BC5C/BSA | BODIPY FL C5-ceramide complexed to BSA |
| BSA | Bovine Serum Albumin |
References
- Kodama, Y.; Fujishima, M. Cycloheximide induces synchronous swelling of perialgal vacuoles enclosing symbiotic chlorella vulgaris and digestion of the algae in the ciliate Paramecium bursaria. Protist 2008, 159, 483–494. [Google Scholar] [CrossRef]
- Iwai, S.; Fujita, K.; Takanishi, Y.; Fukushi, K. Photosynthetic endosymbionts benefit from host’s phagotrophy, including predation on potential competitors. Curr. Biol. 2019, 29, 3114–3119. [Google Scholar] [CrossRef]
- Kodama, K.; Miyazaki, S. Autolysis of Chlorella variabilis in Starving Paramecium bursaria Help the Host Cell Survive Against Starvation Stress. Curr. Microbiol. 2021, 78, 558–565. [Google Scholar] [CrossRef]
- Okada, K.; Fujiwara, T.; Hirooka, S.; Kobayashi, Y.; Onuma, R.; Miyagishima, S. -y. The closed nutrient recycling system in the Paramecium-Chlorella photosymbiosis contributes to survival under oligotrophic conditions. Sci. Adv. 2025, 11, 44. [Google Scholar] [CrossRef]
- Kodama, Y.; Suzuki, H.; Dohra, H.; Sugii, M.; Kitazume, T.; Yamagishi, K.; Shigenobu, S.; Fujishima, M. Comparison of gene expression of Paramecium bursaria with and without Chlorella variabilis symbionts. BMC Genomics Mar 10. 2014, 15(1), 183. [Google Scholar] [CrossRef]
- Reisser, W. The metabolic interactions between Paramecium bursaria Ehrbg. and Chlorella spec. in the Paramecium bursaria-symbiosis. II. Symbiosis-specific properties of the physiology and the cytology of the symbiotic unit and their regulation (author’s transl). Arch. Microbiol. 1976, 111, 161–170. [Google Scholar] [CrossRef]
- Kessler, E. Evidence of de novo synthesis of maltose excreted by the endosymbiotic Chlorella from Paramecium bursaria. Planta 1982, 153, 481–485. [Google Scholar]
- Tanaka, M.; Miwa, I. Significance of photosynthetic products of symbiotic Chlorella to establish the Paramecium–Chlorella symbiosis. Zool. Sci. 1996, 13, 685–692. [Google Scholar] [CrossRef]
- Shibata, A.; Takahashi, F.; Kasahara, M.; Imamura, N. Induction of maltose release by light in the endosymbiotic Chlorella of Paramecium bursaria. Protist 2016, 167, 468–478. [Google Scholar] [CrossRef] [PubMed]
- Kodama, Y.; Fujishima, M. Symbiotic Chlorella sp. of the ciliate Paramecium bursaria do not prevent acidification and lysosomal fusion of host digestive vacuoles during infection. Protoplasma 2005, 225, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Kodama, Y.; Fujishima, M. Infection of Paramecium bursaria by symbiotic Chlorella species. In Endosymbionts in Paramecium; Fujishima, M., Ed.; Springer: Berlin Heidelberg, Germany, 2009; pp. 31–55. [Google Scholar]
- Kodama, Y.; Fujishima, M. Timing of perialgal vacuole membrane differentiation from digestive vacuole membrane in infection of symbiotic alga Chlorella vulgaris of the ciliate Paramecium bursaria. Protist 2009, 160, 65–74. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Four important cytological events needed to establish endosymbiosis of symbiotic Chlorella sp. To the alga-free Paramecium bursaria. Jpn J. Protozool 2011, 44, 1–20. [Google Scholar]
- Tonooka, Y.; Watanabe, G. Genetics of the relationship between the ciliate Paramecium bursaria and its symbiotic algae. Invertebrate Biol. 2007, 126, 287–294. [Google Scholar] [CrossRef]
- Summerer, M.; Sonntag, B.; Hörtnagl, P.; Sommaruga, R. Symbiotic ciliates receive protection against UV damage from their algae: A test with Paramecium bursaria and Chlorella. Protist 2009, 160, 232–243. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Infectivity of Chlorella species for the ciliate Paramecium bursaria is not based on sugar residues of their cell wall components, but on their ability to localize beneath the host cell membrane after escaping from the host digestive vacuole in the early infection process. Protoplasma 2007, 231, 55–63. [Google Scholar] [CrossRef]
- Reisser, W. Endosymbiotic associations of freshwater protozoa and algae. In Progress in Protistology; Corliss, J.O., Patterson, D.J., Eds.; Biopress Ltd.: Bristol, England, 1986; pp. 195–214. [Google Scholar]
- Wakefield, T.S.; Farmer, M.A.; Kempf, S.C. Revised description of the fine structure of in situ “zooxanthellae” in the sea anemone Aiptasia pallida. Biol. Bull. 2000, 199, 76–84. [Google Scholar] [CrossRef]
- Wakefield, T.S.; Kempf, S.C. Development of host- and symbiont-specific monoclonal antibodies and confirmation of the origin of the symbiosome membrane in a cnidarian-dinoflagellate symbiosis. Biol. Bull. 2001, 200, 127–143. [Google Scholar] [CrossRef]
- Kopp, C.; Domart-Coulon, I.; Barthelemy, D.; Meibom, A. Nutritional input from dinoflagellate symbionts in reef-building corals is minimal during planula larval life stage. Sci. Adv. 2016, 2, e1500681. [Google Scholar] [CrossRef]
- Norton, J.H.; Shepherd, M.A.; Long, H.M.; Fitt, W.K. The zooxanthellal tubular system in the giant clam. Biol. Bull. 1992, 183, 503–506. [Google Scholar] [CrossRef]
- Rumpho, M.E.; Pelletreau, K.; Moustafa, A.; Bhattacharya, D. The making of a photosynthetic animal. J. Exp. Biol. 2011, 214, 303–311. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Chapter 2—Secondary symbiosis between Paramecium and Chlorella cells. In International Review of Cell and Molecular Biology; Academic Press: Cambridge, MA, USA, 2010; Volume 279, pp. 33–77. [Google Scholar] [CrossRef]
- Kodama, Y.; Inoue, I.; Fujishima, M. Symbiotic Chlorella vulgaris of the ciliate Paramecium bursaria plays an important role in maintaining perialgal vacuole membrane functions. Protist 2011, 162, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Kodama, Y.; Fujishima, M. Endosymbiosis of Chlorella species to the ciliate Paramecium bursaria alters the distribution of the host’s trichocysts beneath the host cell cortex. Protoplasma 2011, 248, 325–337. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Endosymbiotic Chlorella variabilis reduces mitochondrial number in the ciliate Paramecium bursaria. Sci. Rep. 2022, 12, 8216. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Role of host ciliate Paramecium bursaria mitochondria and trichocysts for symbiotic Chlorella variabilis attachment beneath the host cell cortex. FEMS Microbiol. Lett. 2023, 370. [Google Scholar] [CrossRef]
- Kodama, Y.; Fujishima, M. Effects of the symbiotic Chlorella variabilis on the host cliate Paramecium bursaria phenotypes. Microorganisms 2024, 12, 2537. [Google Scholar] [CrossRef]
- Song, C.; Murata, K.; Suzaki, T. Intracellular symbiosis of algae with possible involvement of mitochondrial dynamics. Sci. Rep. 2017, 7, 1221. [Google Scholar] [CrossRef]
- Reisser, W. The endosymbiotic unit of Stenter polymorphus and Chlorella sp. Morphological and physiological studies. Protoplasma 1981, 105, 273–284. [Google Scholar] [CrossRef]
- Kodama, Y.; Nakahara, M.; Fujishima, M. Symbiotic alga Chlorella vulgaris of the ciliate Paramecium bursaria shows temporary resistance to host lysosomal enzymes during the early infection process. Protoplasma 2007, 230, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Dryl, S. Antigenic transformation in Paramecium aurelia after homologous antiserum treatment during autogamy and conjugation. J. Protozool. 1959, 6, 25. https://cir.nii.ac.jp/crid/1571698599030519168.
- Hiwatashi, K. Determination and inheritance of mating type in Paramecium caudatum. Genetics 1968, 58, 373–386. Available online: https://cir.nii.ac.jp/crid/1572543025382930304. [CrossRef] [PubMed]
- Fujishima, M.; Inoue, Y.; Sawada, T.; Fukumoto, T. Developmental expression of macronuclear specific antigen in Parameciuim caudatum. Dev. Genet. 1992, 13, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, N.G.; Pagano, R.E. A vital stain for the Golgi apparatus. Science 1985, 228, 745–747. [Google Scholar] [CrossRef] [PubMed]
- Pagano, R.E.; Martin, O.C.; Kang, H.C.; Haugland, R.P. A novel fluorescent ceramide analogue for studying membrane traffic in animal cells: Accumulation at the Golgi apparatus results in altered spectral properties of the sphingolipid precursor. J. Cell Biol. 1991, 113, 1267–1279. [Google Scholar] [CrossRef]
- Peters, T. All About Albumin: Biochemistry, Genetics, and Medical Applications; Academic Press: San Diego, CA, USA, 1995. [Google Scholar]
- Carpita, N.; Gibeaut, D.M. Structural models of primary cell walls in flowering plants: Consistency of molecular structure with the physical properties of the walls during growth. Plant J. 1993, 3, 1–30. [Google Scholar] [CrossRef]
- Finkelstein, A. Water and nonelectrolyte permeability of lipid bilayer membranes. J. Gen. Physiol. 1976, 68, 127–135. [Google Scholar] [CrossRef]
- Hehl, A. B.; Marti, M.; Köhler, P. Stage-specific expression and targeting of cyst wall protein-green fluorescent protein chimeras in Giardia. Mol. Biol. Cell 2000, 11, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M.; Allen, R. D. Uptake and rapid transfer of fluorescent ceramide analogues to acidosomes (late endosomes) in Paramecium, J. Histochem. Cytochem. 2004, 52, 557–565. [Google Scholar] [CrossRef]
- Sinai, A. P.; Webster, P.; Joiner, K. A. Association of host cell endoplasmic reticulum and mitochondria with the Toxoplasma gondii parasitophorous vacuole membrane: a high affinity interaction. J. Cell Sci. 1997, 110, 2117–2128. [Google Scholar] [CrossRef] [PubMed]
- Sinai, A. P.; Joiner, K. A. The Toxoplasma gondii protein ROP2 mediates host organelle association with the parasitophorous vacuole membrane. J. Cell Biol. 2001, 154, 95–108. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



