Submitted:
18 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Patients’ Samples
2.2. Clinical, Anthropometric and Metabolic Assessments
2.3. microRNAs and RNA Isolation
2.4. Protein Biomarkers by Enzyme-Linked Immunosorbent Assay (ELISA)
2.5. Oxidative Stress Markers - Malondialdehyde (MDA)
2.6. Oxidative Stress Markers - Total Antioxidant Capacity (TAC)
2.7. Gene Expression Profile Analysis
2.8. Statistical Analysis of Biomarkers
3. Results
4. Discussion
Authors contribution
Declaration of funding
Ethics Statement
Consent
Disclosure statement
Data Availability Statement
Acknowledgments
References
- Piché, ME; Tchernof, A; Després, JP. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ Res. 2020, 126(11), 1477–500. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z; Wang, H; Yang, Z; Lu, Y; Zou, C. Causal associations between type 1 diabetes mellitus and cardiovascular diseases: a Mendelian randomization study. Cardiovasc Diabetol. 2023, 22, 236. [Google Scholar] [CrossRef] [PubMed]
- Yun, JS; Ko, SH. Current trends in epidemiology of cardiovascular disease and cardiovascular risk management in type 2 diabetes. Metabolism 2021, 123, 154838. [Google Scholar] [CrossRef]
- Ma, CX; Ma, XN; Guan, CH; Li, YD; Mauricio, D; Fu, SB. Cardiovascular disease in type 2 diabetes mellitus: progress toward personalized management. Cardiovasc Diabetol 2022, 21, 74. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M; Santamarina, J; Pavesi, A; Musso, C; Umpierrez, GE. Glycemic variability and cardiovascular disease in patients with type 2 diabetes. BMJ Open Diabetes Res Care 2021, 9, e002032. [Google Scholar] [CrossRef]
- Li, WJ; Wang, Y; Liu, R; Kasinski, AL; Shen, H; Slack, FJ; Tang, DG. MicroRNA-34a: potent tumor suppressor, cancer stem cell inhibitor, and potential anticancer therapeutic. Front Cell Dev Biol. 2021, 9, 640587. [Google Scholar] [CrossRef]
- Thounaojam, MC; Bartoli, M. MicroRNA-34a and vascular senescence in diabetes. Aging (Albany NY) 2019, 11(24), 11799–800. [Google Scholar] [CrossRef]
- Hua, CC; Liu, XM; Liang, LR; Wang, LF; Zhong, JC. Targeting the microRNA-34a as a novel therapeutic strategy for cardiovascular diseases. Front Cardiovasc Med. 2022, 8, 784044. [Google Scholar] [CrossRef]
- Chen, Q; Shen, Z; Mao, Y; Li, Q; Liu, Y; Mei, M; et al. Inhibition of microRNA-34a mediates protection of thymosin beta 4 in endothelial progenitor cells against advanced glycation end-products by targeting B-cell lymphoma 2. Can J Physiol Pharmacol. 2019, 97(10), 945–51. [Google Scholar] [CrossRef]
- Zhang, F; Cui, J; Liu, X; Lv, B; Liu, X; Xie, Z; Yu, B. Roles of microRNA-34a targeting SIRT1 in mesenchymal stem cells. Stem Cell Res Ther. 2015, 6, 195. [Google Scholar] [CrossRef]
- Okonkwo, UA; DiPietro, LA. Diabetes and wound angiogenesis. Int J Mol Sci. 2017, 18(7), 1419. [Google Scholar] [CrossRef] [PubMed]
- Li, Q; Kim, YR; Vikram, A; Kumar, S; Kassan, M; Gabani, M; et al. P66Shc-induced microRNA-34a causes diabetic endothelial dysfunction by downregulating sirtuin 1. Arterioscler Thromb Vasc Biol. 2016, 36(12), 2394–403. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C; Parikh, NS. Framingham General Cardiovascular Risk Score and cognitive impairment: the power of foresight. J Am Coll Cardiol. 2020, 75(20), 2535–7. [Google Scholar] [CrossRef] [PubMed]
- Matsudo, S; Araújo, T; Matsudo, V; Andrade, D; Andrade, E; de Oliveira, L; Braggion, G. International Physical Activity Questionnaire (IPAQ): study of validity and reliability in Brazil. Rev Bras Ativ Fís Saúde 2001, 6(2), 5–18. [Google Scholar] [CrossRef]
- Hitsumoto, T. Clinical significance of cardio-ankle vascular index as a cardiovascular risk factor in elderly patients with type 2 diabetes mellitus. J Clin Med Res. 2018, 10(4), 330–6. [Google Scholar] [CrossRef]
- Viigimaa, M; Sachinidis, A; Toumpourleka, M; Koutsampasopoulos, K; Alliksoo, S; Titma, T. Macrovascular complications of type 2 diabetes mellitus. Curr Vasc Pharmacol. 2020, 18(2), 110–6. [Google Scholar] [CrossRef]
- Priya, HK; Jha, KP; Kumar, N; Singh, S. Reactive oxygen species and mitochondrial calcium’s roles in the development of atherosclerosis. Curr Pharm Des. 2024, 30(23), 1812–21. [Google Scholar] [CrossRef]
- Batty, M; Bennett, MR; Yu, E. The role of oxidative stress in atherosclerosis. Cells 2022, 11(23), 3843. [Google Scholar] [CrossRef]
- Chen, P; Chen, F; Lei, J; Li, Q; Zhou, B. Activation of the miR-34a-mediated SIRT1/mTOR signaling pathway by urolithin A attenuates D-galactose-induced brain aging in mice. Neurotherapeutics 2019, 16(4), 1269–82. [Google Scholar] [CrossRef]
- Jianbing, H; Xiaotian, L; Jie, T; Xueying, C; Honge, J; Bo, Z; et al. The effect of allograft inflammatory factor-1 on inflammation, oxidative stress, and autophagy via miR-34a/ATG4B pathway in diabetic kidney disease. Oxid Med Cell Longev. 2022, 2022, 1668000. [Google Scholar] [CrossRef]
- Zhu, H; Lin, Y; Liu, Y. miR-34a increases inflammation and oxidative stress levels in patients with necrotizing enterocolitis by downregulating SIRT1 expression. Mol Med Rep. 2021, 24(3), 664. [Google Scholar] [CrossRef]
- Liang, M; Shao, A; Tang, X; Feng, M; Wang, J; Qiu, Y. MiR-34a affects dexmedetomidine-inhibited chronic inflammatory visceral pain by targeting HDAC2. BMC Anesthesiol. 2019, 19, 131. [Google Scholar] [CrossRef]
- Wu, J; Liang, W; Tian, Y; Ma, F; Huang, W; Jia, Y; et al. Inhibition of p53/miR-34a improves diabetic endothelial dysfunction via activation of SIRT1. J Cell Mol Med. 2019, 23(5), 3538–48. [Google Scholar] [CrossRef]
- Kaur, P; Kotru, S; Singh, S; Munshi, A. Role of miRNAs in diabetic neuropathy: mechanisms and possible interventions. Mol Neurobiol. 2022, 59(3), 1836–49. [Google Scholar] [CrossRef]
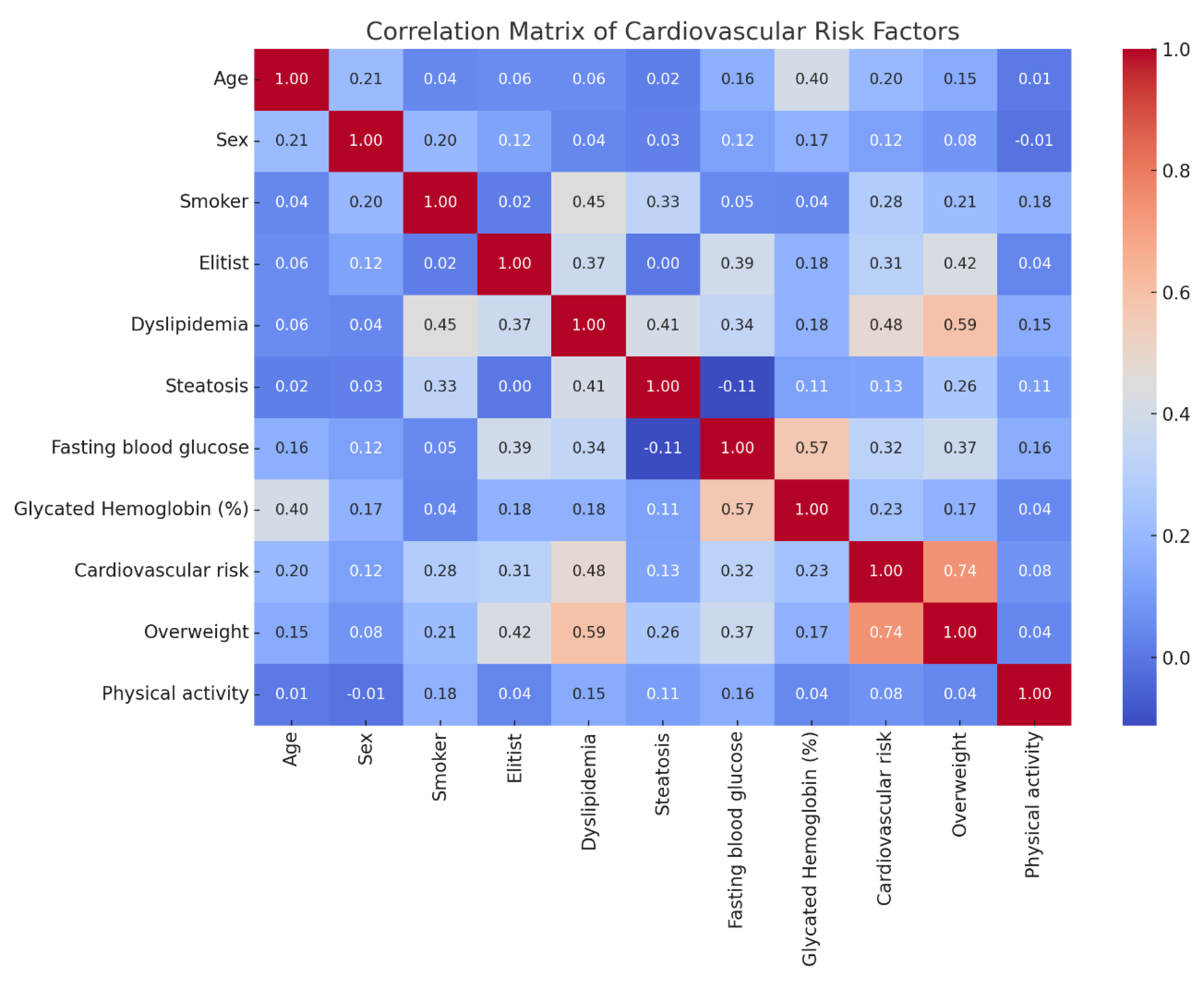
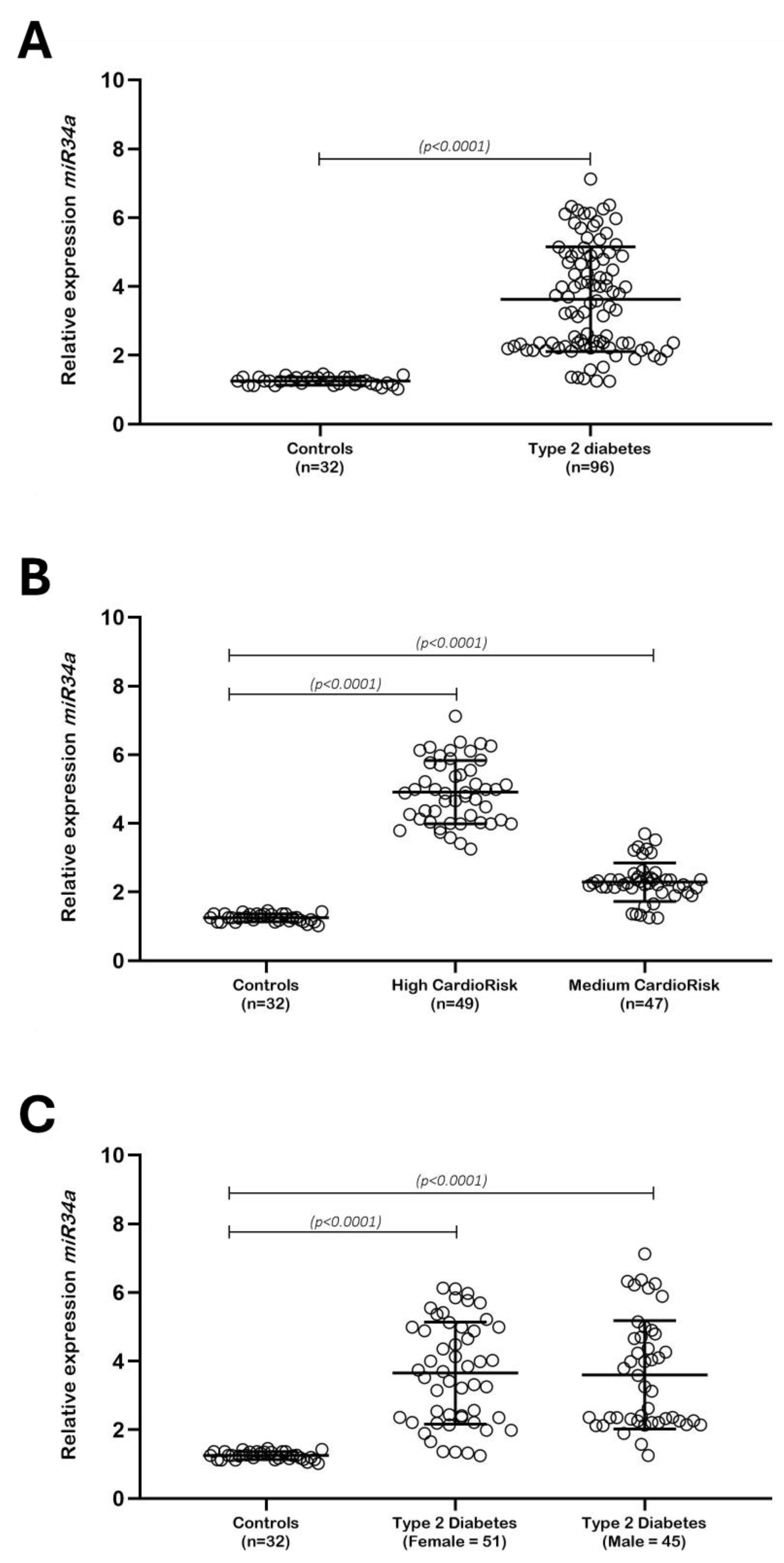
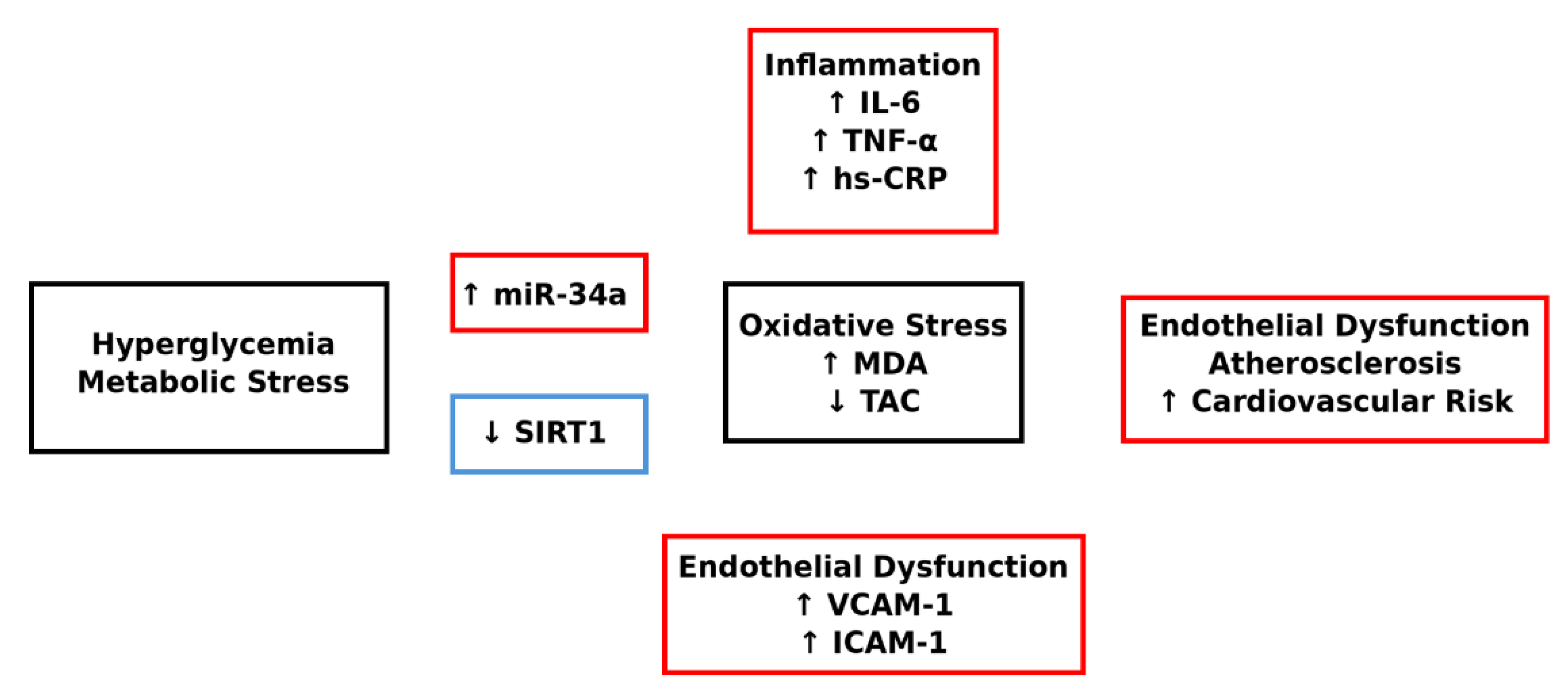
| Variables | n (%) |
| Age (years) | |
| < 59 | 57 (59.3) |
| ≥ 60 | 39 (40.7) |
| Sex | |
| Female | 51 (53.1) |
| Male | 45 (46.9) |
| Smoker | |
| Yes | 33 (34.3) |
| No | 51 (53.1) |
| Not informed | 12 (12.5) |
| Elitist | |
| Yes | 30 (31.2) |
| No | 51 (53.1) |
| Not informed | 15 (15.6) |
| Dyslipidemia | |
| Yes | 52 (54.1) |
| No | 44 (45.9) |
| Steatosis | |
| Yes | 46 (47.9) |
| No | 37 (38.5) |
| Not informed | 13 (13.5) |
| Fasting blood glucose (mg/dL) | |
| < 126 | 13 (13.5) |
| ≥ 126 | 83 (86.5) |
| Glycated Hemoglobin (%) | |
| < 6.5 | 24 (25.0) |
| ≥ 6.5 | 72 (75.0) |
| Cardiovascular risk | |
| High | 49 (51.0) |
| Medium | 47 (49.0) |
| Overweight | |
| Yes | 51 (53.1) |
| No | 45 (46.9) |
| Physical activity | |
| Yes | 46 (47.9) |
| No | 50 (52.1) |
| Inflammatory Markers Results | |||
| Subjects | Interleukin-6 (IL-6) – pg/mL |
Tumor Necrosis Factor-α (TNF-α) – pg/mL |
High-Sensitivity C-Reactive Protein (hs-CRP) – mg/L |
| Controls (n = 32) | 1.82 ± 0.54 | 2.95 ± 0.72 | 1.21 ± 0.63 |
| T2DM – Medium Risk (n = 47) | 3.45 ± 0.78 | 4.88 ± 0.94 | 3.64 ± 1.08 |
| T2DM – High Risk (n = 49) | 5.12 ± 1.05 | 6.73 ± 1.12 | 6.42 ± 1.75 |
| Oxidative Stress Markers Results | ||
| Malondialdehyde (MDA) – nmol/mL plasma | Total Antioxidant Capacity (TAC) – mmol Trolox equivalents/L | |
| Controls (n = 32) | 2.10 ± 0.42 | 1.78 ± 0.28 |
| T2DM – Medium Risk (n = 47) | 3.48 ± 0.63 | 1.32 ± 0.24 |
| T2DM – High Risk (n = 49) | 4.92 ± 0.81 | 0.94 ± 0.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




