Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Clinical Evaluation
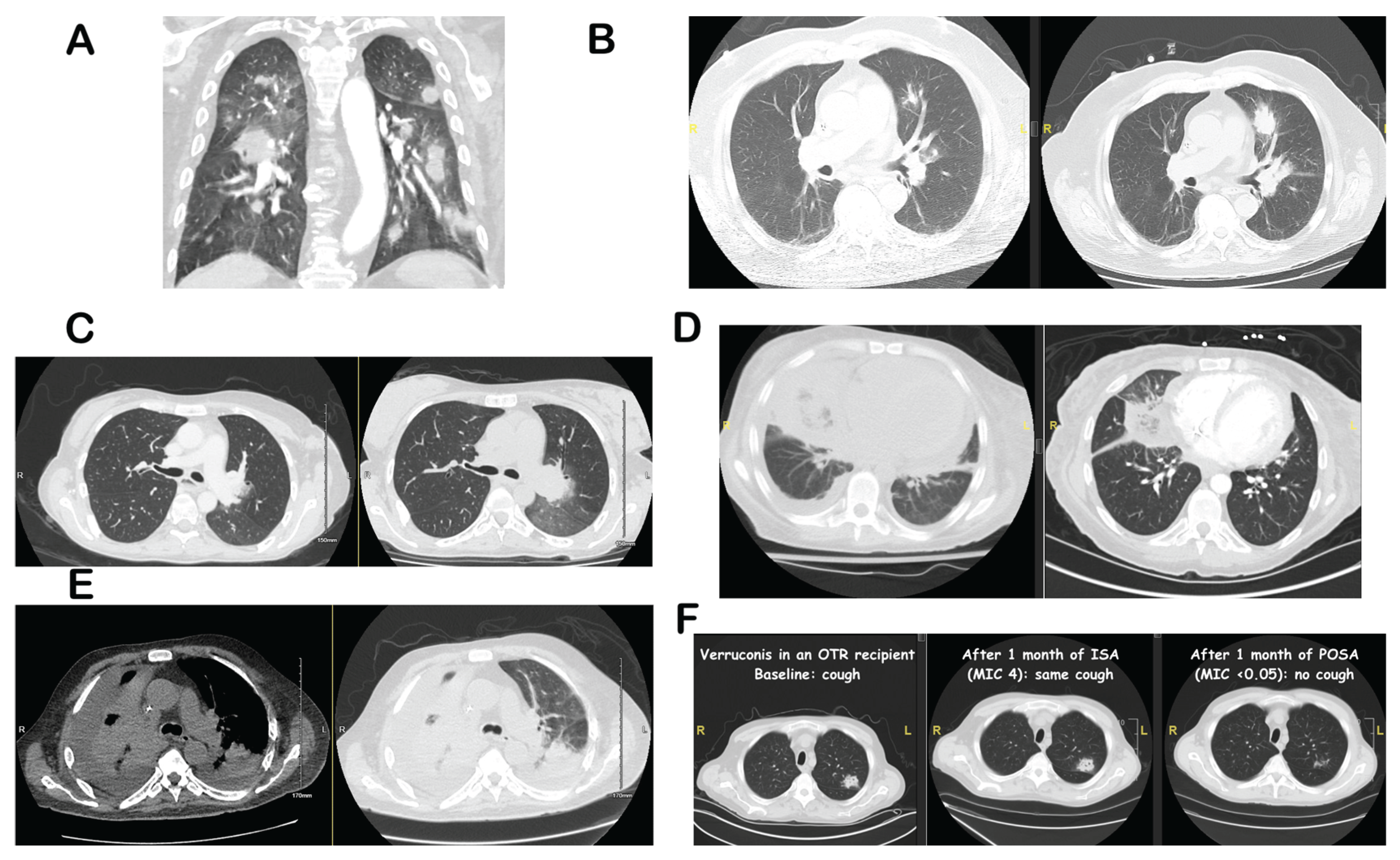
3. Radiographic Findings
4. Invasive Diagnostics
5. Non-Invasive Diagnostics
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Azar MM, Turbett S, Gaston D, Gitman M, Razonable R, Koo S; et al. A consensus conference to define the utility of advanced infectious disease diagnostics in solid organ transplant recipients. Am J Transplant. 2022;22:3150-69. [CrossRef]
- Murali S, Marks A, Heeger A, Dako F, Febbo J. Pneumonia in the immunocompromised host. Semin Roentgenol. 2022;57:90-104. [CrossRef]
- Tong X, Liu T, Jiang K, Wang D, Liu S, Wang Y; et al. Clinical characteristics and prognostic risk factors of patients with proven invasive pulmonary aspergillosis: A single-institution retrospective study. Front Med. 2021;8:756237. [CrossRef]
- Jung J, Kim MY, Lee HJ, Park YS, Lee SO, Choi SH; et al. Comparison of computed tomographic findings in pulmonary mucormycosis and invasive pulmonary aspergillosis. Clin Microbiol Infect. 2015;21:684.e11-8. [CrossRef]
- Scherer E, Iriart X, Bellanger AP, Dupont D, Guitard J, Gabriel F; et al. Quantitative PCR (qPCR) detection of Mucorales DNA in bronchoalveolar lavage fluid to diagnose pulmonary mucormycosis. J Clin Microbiol. 2018;56:e00289-18. [CrossRef]
- Azoulay E, Russell L, Van de Louw A, Metaxa V, Bauer P, Povoa P; et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensive Care Med. 2020;46:298-314. [CrossRef]
- Fernández-Cruz A, Magira E, Heo ST, Evans S, Tarrand J, Kontoyiannis DP. Bronchoalveolar lavage fluid cytology in culture-documented invasive pulmonary aspergillosis in patients with hematologic diseases: Analysis of 67 episodes. J Clin Microbiol. 2018;56:e00962-18. [CrossRef]
- Georgiadou SP, Sipsas NV, Marom EM, Kontoyiannis DP. The diagnostic value of halo and reversed halo signs for invasive mold infections in compromised hosts. Clin Infect Dis. 2011;52:1144-55. [CrossRef]
- Azar MM. A diagnostic approach to fungal pneumonia: An infectious diseases perspective. Chest. 2024;165:559-72. [CrossRef]
- Gali V, Al-Ghanamah R, Finnigan K, Kalchiem-Dekel O, Kamboj M, Hohl TM; et al. Evaluating the clinical utility of Aspergillus, Mucorales, and Nocardia bronchoalveolar PCRs for the diagnosis of invasive pulmonary infections in patients with hematological malignancies. J Clin Microbiol. 2025;63:e01355-24. [CrossRef]
- Szvalb AD, Malek AE, Jiang Y, Bhatti MM, Wurster S, Kontoyiannis DP. Serum (1,3)-beta-D-glucan has suboptimal performance for the diagnosis of Pneumocystis jirovecii pneumonia in cancer patients and correlates poorly with respiratory burden as measured by quantitative PCR. J Infect. 2020;81:443-51. [CrossRef]
- Morjaria S, Frame J, Franco-Garcia A, Geyer A, Kamboj M, Babady NE. Clinical performance of (1,3) beta-D glucan for the diagnosis of Pneumocystis pneumonia (PCP) in cancer patients tested with PCP polymerase chain reaction. Clin Infect Dis. 2019;69:1303-9. [CrossRef]
- Bergin SP, Chemaly RF, Dadwal SS, Hill JA, Lee YJ, Haidar G; et al. Plasma microbial cell-free DNA sequencing in immunocompromised patients with pneumonia: A prospective observational study. Clin Infect Dis. 2024;78:775-84. [CrossRef]
- Arvanitis M, Ziakas PD, Zacharioudakis IM, Zervou FN, Caliendo AM, Mylonakis E. PCR in diagnosis of invasive aspergillosis: A meta-analysis of diagnostic performance. J Clin Microbiol. 2014;52:3731-42. [CrossRef]
- Pechacek J, Lionakis MS. Invasive fungal infections as a complication of new therapies. Annu Rev Med. 2025. [CrossRef]
- Farmakiotis D, Ciurea AM, Cahuayme-Zuniga L, Kontoyiannis DP. The diagnostic yield of skin biopsy in patients with leukemia and suspected infection. J Infect. 2013;67:265-72. [CrossRef]
- Farmakiotis D, Kontoyiannis DP. Mucormycoses. Infect Dis Clin North Am. 2016;30:143-63. [CrossRef]
- Chamilos G, Marom EM, Lewis RE, Lionakis MS, Kontoyiannis DP. Predictors of pulmonary zygomycosis versus invasive pulmonary aspergillosis in patients with cancer. Clin Infect Dis. 2005;41:60-6. [CrossRef]
- Jang HM, Kim MY, Lim SY, Chang EJ, Bae S, Jung J; et al. CT findings for differentiating pulmonary mucormycosis from invasive pulmonary aspergillosis prior to invasive procedure such as a biopsy or surgery: A 22-year single-center experience. Mycoses. 2025;68:e70115. [CrossRef]
- Stanzani M, Sassi C, Lewis RE, Tolomelli G, Bazzocchi A, Cavo M; et al. High resolution computed tomography angiography improves the radiographic diagnosis of invasive mold disease in patients with hematological malignancies. Clin Infect Dis. 2015;60:1603-10. [CrossRef]
- Stanzani M, Sassi C, Lewis R, Sartor C, Rasetto G, Cavo M; et al. Early low-dose computed tomography with pulmonary angiography to improve the early diagnosis of invasive mould disease in patients with haematological malignancies: A pilot study. J Infect. 2021;83:371-80. [CrossRef]
- Lewis RE, Stanzani M, Morana G, Sassi C. Radiology-based diagnosis of fungal pulmonary infections in high-risk hematology patients: Are we making progress? Curr Opin Infect Dis. 2023;36:250-6. [CrossRef]
- Donnelly JP, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE; et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367-76. [CrossRef]
- Smith CB, Shi X, Liesman RM, Thomas LA, Bahr NC, Brownback KR. Evaluation of the diagnostic accuracy and clinical utility of fungal profile plus polymerase chain reaction assay in pulmonary infections. Open Forum Infect Dis. 2022;9:ofac646. [CrossRef]
- Pandey M, Xess I, Singh G, Kumar R, Mahapatra M, Jyotsna VP; et al. Conventional PCR as a reliable method for diagnosing invasive mucormycosis in resource-limited settings. J Med Microbiol. 2021;70. [CrossRef]
- Kitmiridou D, Aung SN, Farmakiotis D. Disseminated mucormycosis with positive Aspergillus galactomannan. Case Rep Infect Dis. 2018;2018:4294013. [CrossRef]
- Knipe RS, Alba GA, Harvey Barnes JM, Hariri LP. Case 15-2022: A 57-year-old man with persistent cough and pulmonary opacities. N Engl J Med. 2022;386:1933-44. [CrossRef]
- Rose SR, Vallabhajosyula S, Velez MG, Fedorko DP, VanRaden MJ, Gea-Banacloche JC; et al. The utility of bronchoalveolar lavage beta-D-glucan testing for the diagnosis of invasive fungal infections. J Infect. 2014;69:278-83. [CrossRef]
- Kubiak J, Morgan A, Kirmaier A, Arnaout R, Riedel S. Universal PCR for bacteria, mycobacteria, and fungi: A 10-year retrospective review of clinical indications and patient outcomes. J Clin Microbiol. 2023;61:e00952-23. [CrossRef]
- Kirn TJ, Weinstein MP. Update on blood cultures: How to obtain, process, report, and interpret. Clin Microbiol Infect. 2013;19:513-20. [CrossRef]
- Amaya S, Giovannini-Sanguineti G, Lopez C, Alonso CD, Riedel S. Disseminated Lomentospora prolificans infection in a neutropenic patient with acute monocytic leukemia: A clinical and diagnostic challenge. ASM Case Rep. 2025;1:e00093-25. [CrossRef]
- Vergidis P, Razonable RR, Wheat LJ, Estes L, Caliendo AM, Baden LR; et al. Reduction in false-positive Aspergillus serum galactomannan enzyme immunoassay results associated with use of piperacillin-tazobactam in the United States. J Clin Microbiol. 2014;52:2199-201. [CrossRef]
- Arvanitis M, Anagnostou T, Mylonakis E. Galactomannan and polymerase chain reaction-based screening for invasive aspergillosis among high-risk hematology patients: A diagnostic meta-analysis. Clin Infect Dis. 2015;61:1263-72. [CrossRef]
- Millon L, Caillot D, Berceanu A, Bretagne S, Lanternier F, Morio F; et al. Evaluation of serum Mucorales polymerase chain reaction (PCR) for the diagnosis of mucormycoses: The MODIMUCOR prospective trial. Clin Infect Dis. 2022;75:777-85. [CrossRef]
- Allos H, John TM, Stewart AG. Microbial cell-free DNA for diagnosis of bacterial and fungal infection in the immunocompromised host — what do we know? Curr Opin Infect Dis. 2025. [CrossRef]
- Huygens S, Schauwvlieghe A, Wlazlo N, Moors I, Boelens J, Reynders M; et al. Diagnostic value of microbial cell-free DNA sequencing for suspected invasive fungal infections: A retrospective multicenter cohort study. Open Forum Infect Dis. 2024;11:ofae252. [CrossRef]
- Sim BZ, Mah JK, Heldman MR, Stanly KL, Hanson KE, Caliendo AM; et al. Plasma microbial cell-free DNA metagenomic sequencing for diagnosis of invasive fungal diseases among high-risk outpatient and inpatient immunocompromised hosts. Clin Infect Dis. 2025;81:1008-14. [CrossRef]
- Koo S, Thomas HR, Daniels SD, Lynch RC, Fortier SM, Shea MM; et al. A breath fungal secondary metabolite signature to diagnose invasive aspergillosis. Clin Infect Dis. 2014;59:1733-40. [CrossRef]
- Ghosh C, Leon A, Koshy S, Aloum O, Al-Jabawi Y, Ismail N; et al. Breath-based diagnosis of infectious diseases: A review of the current landscape. Clin Lab Med. 2021;41:185-202. [CrossRef]
- Chambers ST, Syhre M, Murdoch DR, McCartin F, Epton MJ. Detection of 2-pentylfuran in the breath of patients with Aspergillus fumigatus. Med Mycol. 2009;47:468-76. [CrossRef]
- Li ZT, Zeng PY, Chen ZM, Guan WJ, Wang T, Lin Y; et al. Exhaled volatile organic compounds for identifying patients with chronic pulmonary aspergillosis. Front Med. 2021;8:720119. [CrossRef]
- Acharige MJT, Koshy S, Ismail N, Aloum O, Jazaerly M, Astudillo CL; et al. Breath-based diagnosis of fungal infections. J Breath Res. 2018;12:027108. [CrossRef]
- Lewis RE, Cahyame-Zuniga L, Leventakos K, Chamilos G, Ben-Ami R, Tamboli P; et al. Epidemiology and sites of involvement of invasive fungal infections in patients with haematological malignancies: A 20-year autopsy study. Mycoses. 2013;56:638-45. [CrossRef]
- Tsikala-Vafea M, Cao W, Olszewski AJ, Donahue JE, Farmakiotis D. Fatal mucormycosis and aspergillosis in an atypical host: What do we know about mixed invasive mold infections? Case Rep Infect Dis. 2020;2020:8812528. [CrossRef]
| Category | Test / Modality | Specimen | Typical TAT | Sensitivity (%) | Specificity (%) | Approximate Cost (USD, 2024–2025)* |
|---|---|---|---|---|---|---|
| Microscopy & Histopathology | Direct stains (GMS, PAS, calcofluor) | BAL, sputum, tissue | Minutes–hours | <50 | High when tissue invasion present | ~$10–30 per specimen |
| Microscopy & Histopathology | Histopathology (FFPE or fresh tissue) | Biopsy | Hours–1 day | Variable (sampling dependent) | High for tissue invasion; demonstrates angioinvasion | ~$100–200 per biopsy (pathology technical fee) |
| Culture-Based Diagnostics | Fungal culture | BAL, sputum, tissue, sterile fluids | 1–7 days (up to 21 days for slow growers) | ~50 overall; <30 in respiratory IPA | ~100 (species-level identification) | ~$30–60 per culture set |
| Culture-Based Diagnostics | Blood culture for molds | Blood | 1–7 days | Low overall; higher for Fusarium/Scedosporium | High; usually indicates disseminated disease | ~$60–120 per set (aerobic/anaerobic bottles) |
| Culture-Based Diagnostics | MALDI-TOF MS (from culture) | Culture isolate | Minutes once colony available | Same as culture yield | ~100 (species-level identification) | Incremental ~$5–15 per isolate (reagent cost) |
| Targeted Molecular / Tissue-Based Tests | PCR + sequencing (species-level) | FFPE or fresh tissue | 1–3 days | High when fungal elements seen on histology | High; useful for non-distinct morphology or culture-negative disease | ~$250–400 per assay |
| Non–Culture Biomarkers | Serum galactomannan (GM) | Serum, plasma | 1–3 days | 20–90 (host- and specimen-dependent) | ~80–90 | ~$100–200 per test |
| Non–Culture Biomarkers | BAL galactomannan | BAL fluid | 1–3 days | Up to ~90 | ~90 | ~$120–220 per test |
| Non–Culture Biomarkers | (1→3)-β-D-glucan (BDG) | Serum | Same day–1 day | ~60–80 (highest in hematologic/HSCT) | ~70–90; reduced in ICU due to false positives | ~$400–450 per test |
| Fungal PCR (Pathogen-Specific) | Aspergillus PCR | Serum, plasma, whole blood, BAL | 1–2 days | 70–100 | 85–95 | ~$200–350 per assay |
| Fungal PCR (Pathogen-Specific) | Mucorales PCR | Serum, BAL, tissue | 1–2 days | ~85 | ~89–90 | ~$200–350 per assay |
| Broad Molecular & Advanced Diagnostics | Broad-range fungal PCR | Tissue, sterile fluids | 1–3 days | 50–70 | High; adjunct when microscopy positive but cultures/PCR negative | ~$250–450 per assay |
| Broad Molecular & Advanced Diagnostics | Plasma microbial cell-free DNA sequencing / mNGS | Plasma (± BAL, tissue) | 24–48 hours | 40–70 | 80–90 | ~$1,800–2,500 per test |
| Emerging Noninvasive Technologies | Exhaled VOCs by GC–MS | Exhaled breath | Minutes–hours | 77–96 (IPA and CPA) | 78–97 | Research only; estimated reagent cost ~$200–400 per run |
| Imaging | High-resolution CT (HRCT) chest | Imaging | Immediate | High for typical angioinvasive patterns | Limited; findings often nonspecific in non-neutropenic hosts | ~$130–200 (Medicare technical payment) to >$500 list price |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




