Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Because of the lack of lung toxicity data for non-reducing disaccharides, D-sucrose (C12H22O11), an inexpensive intermediate size sugar (mw 342.3 Da), which presents a very low oral toxicity, is widely used in both food and pharmaceutical industries, [30] and, was tested. D-sucrose possesses 14 potential H-bonds per molecule [30] and an intermediate Tg (63°C).
2. Materials and Methods
2.1. Material
2.2. Methods
2.2.1. pH Stability Evaluation of pAb
2.2.2. Spray-Dried Dry Powder for Inhalation Formulations
2.2.3. Physicochemical Properties of DPIs
- Morphology – Scanning Electron Microscopy (SEM)
- Residual moisture – Thermogravimetric Analyses
- Glassy transition temperature - Modulated Differential Scanning Calorimetry (MDSC)
- Crystalline properties – X-Ray Powder Diffraction (XRPD)
2.2.4. Physical Degradation – pAb Aggregate Determination in DPIs
- Sample preparation
- Determination of high-order aggregates percentage and soluble protein recovery
- Determination of monomer content – semi-quantitative analysis
- Determination of low-order aggregates percentage
2.2.5. Calculation
- Theoretical calculation of the number of hydrogen bonds
2.2.6. Mixture Design of Experiments (DoE)
2.2.7. Aerosolization and dispersion of DPIs through dry powder inhaler
2.2.8. Statistical Analysis
3. Results
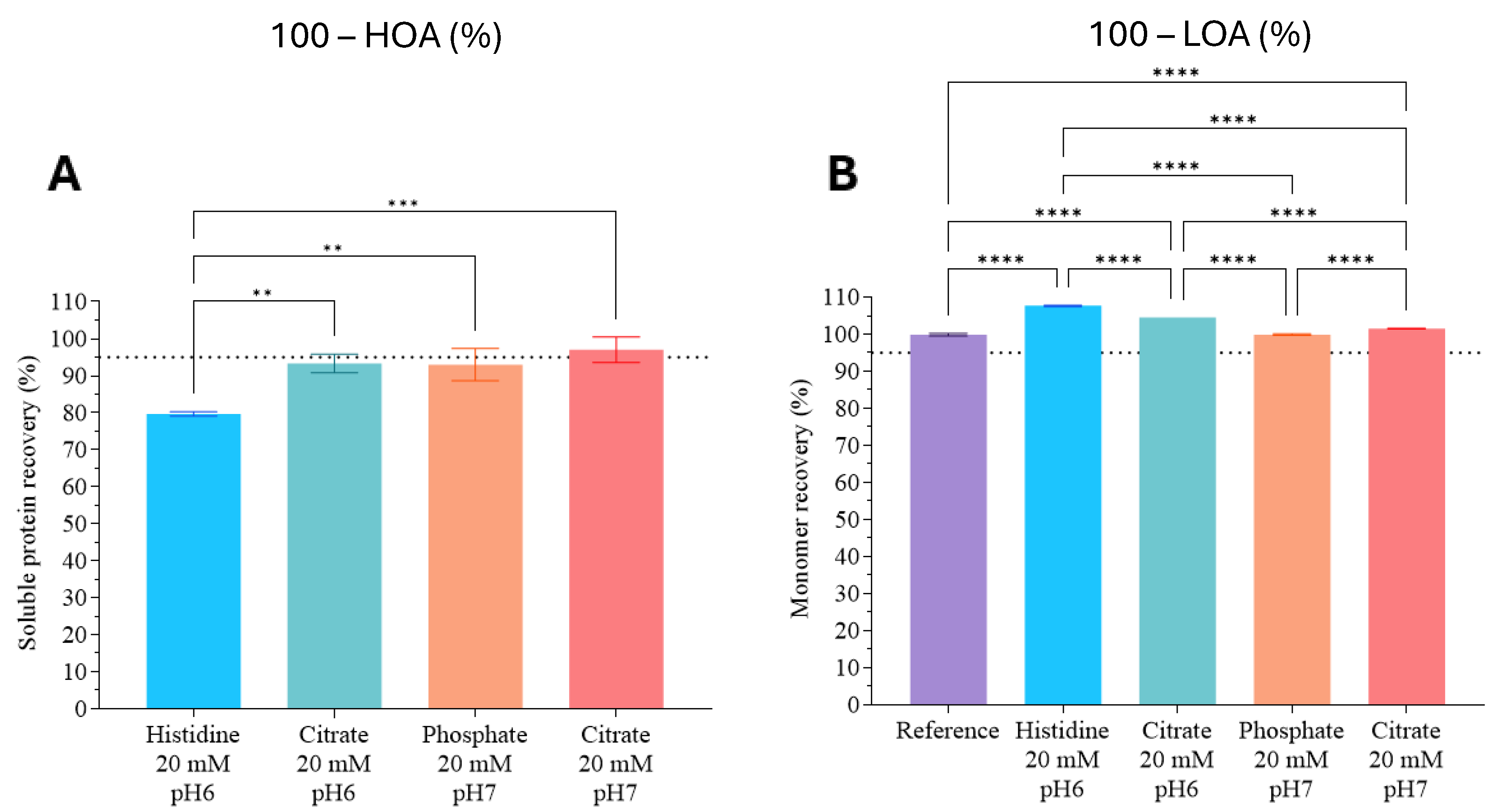
3.1. Stable-Buffer Determination for pAb
3.2. Dry Powders for Inhalation Produced by Spray-Drying
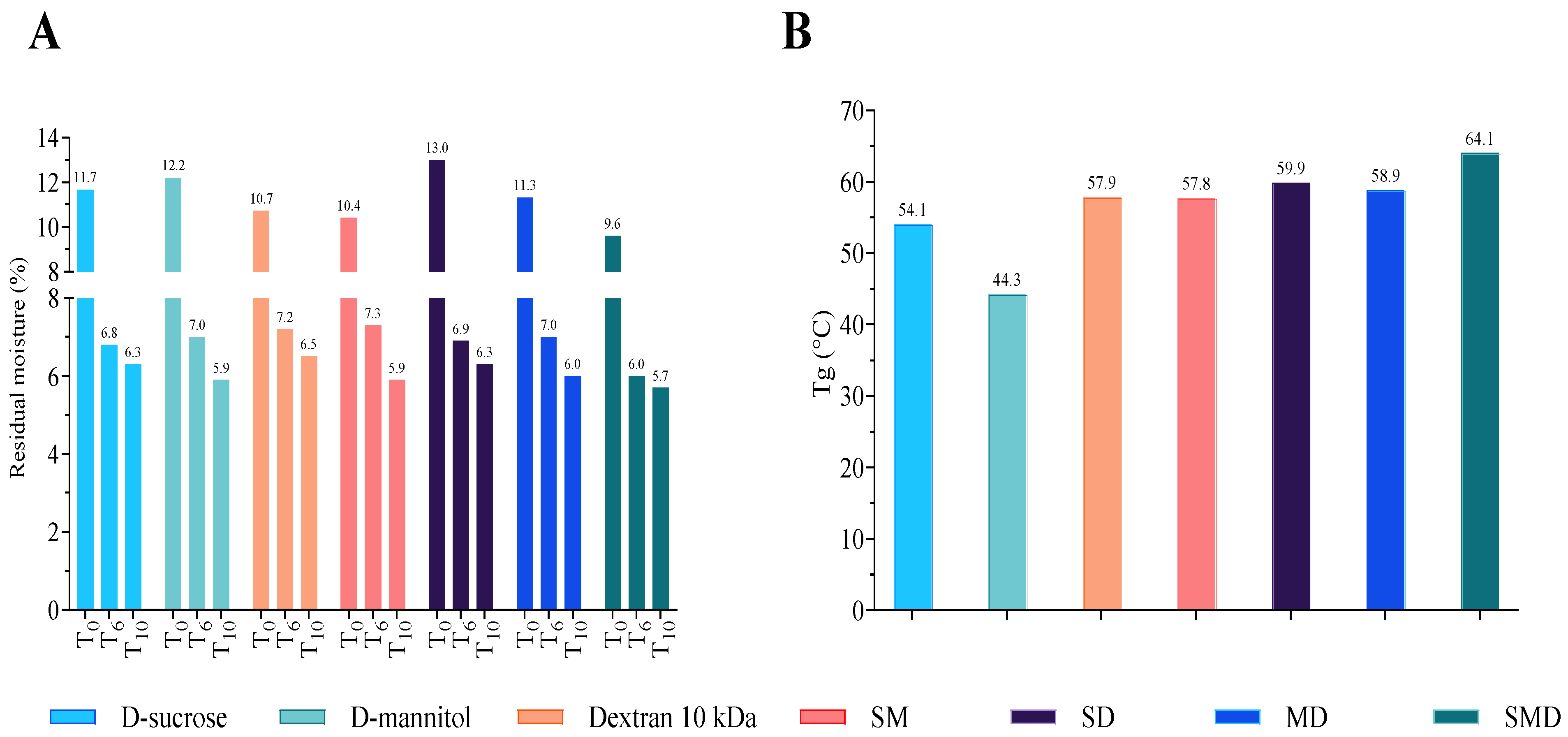
3.2.1. Physicochemical Properties – Morphology, Residual Moisture and Glass Transition Temperature of DPIs
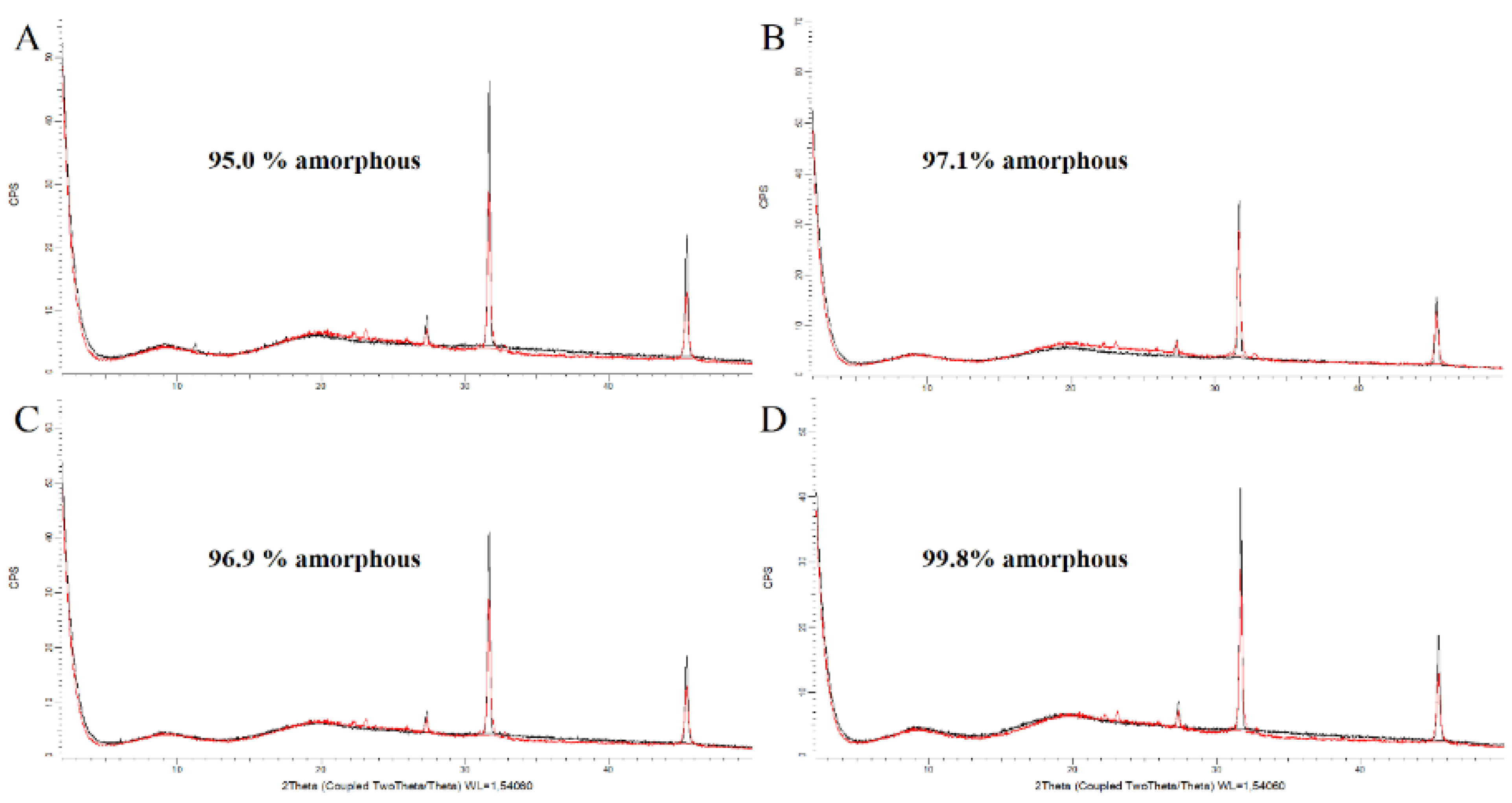
3.2.2. XRPD on DPIs
3.2.3. pAb Aggregates Determination in DPIs
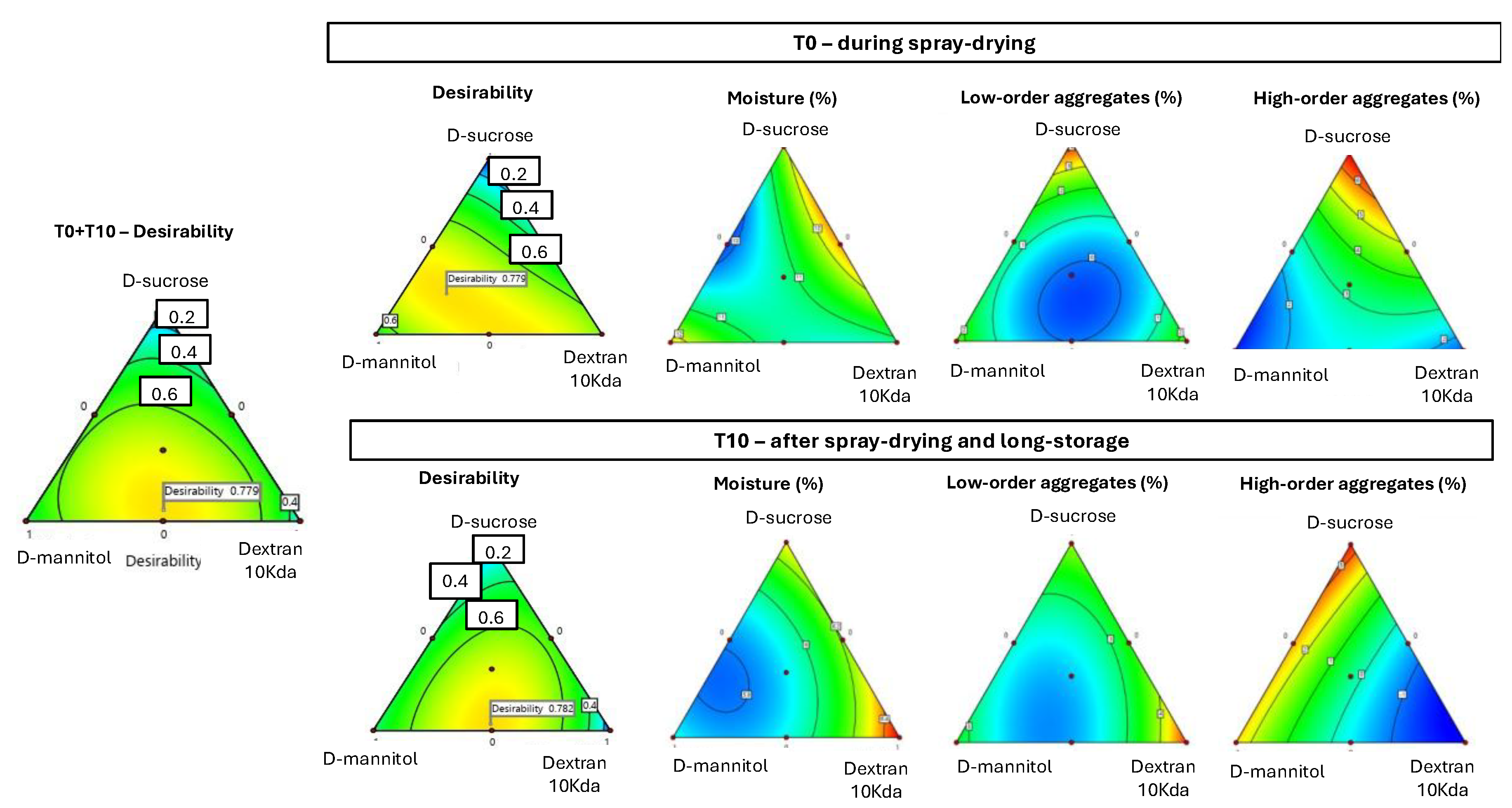
3.2.4. Design of Experiments Analysis
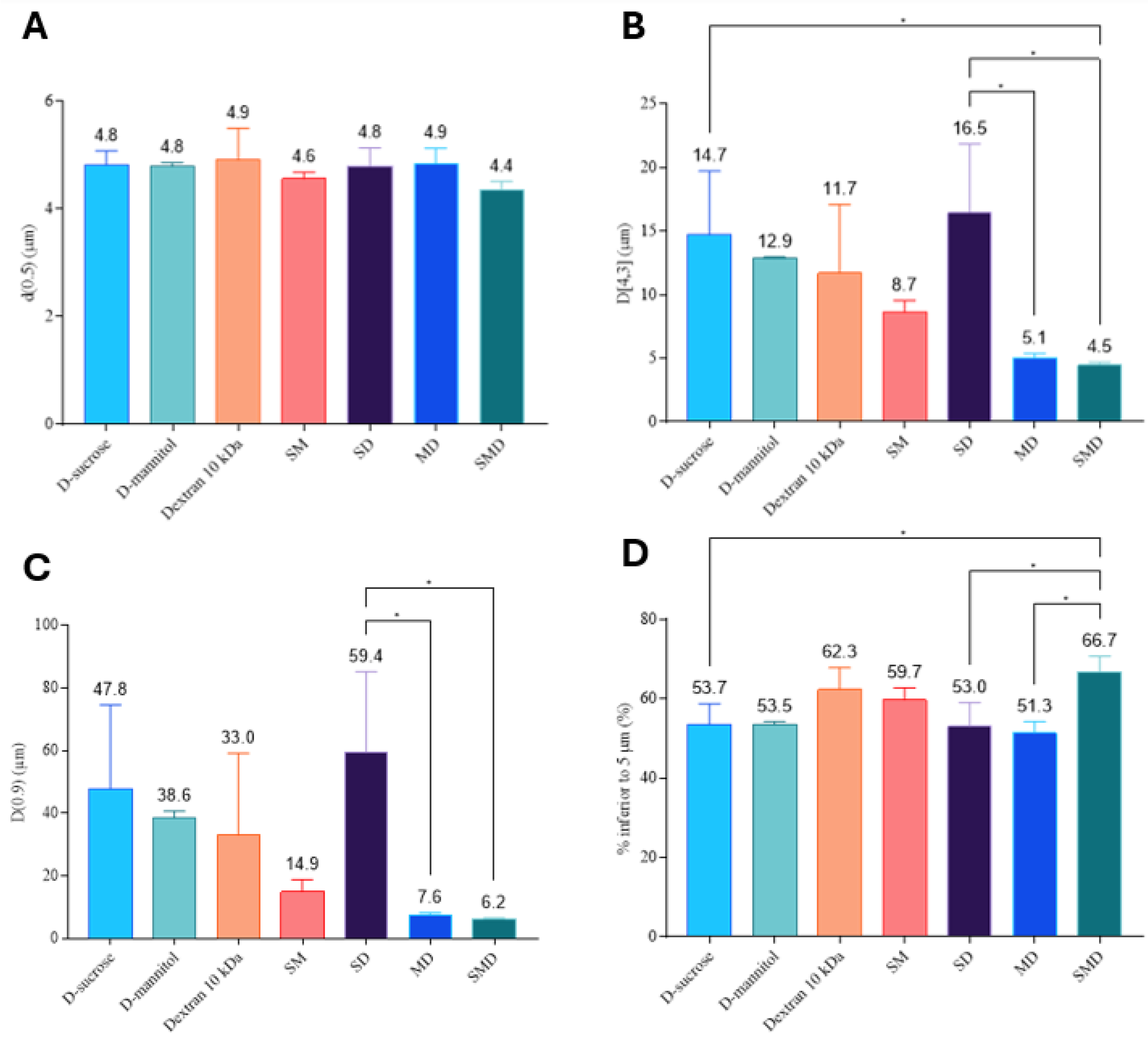
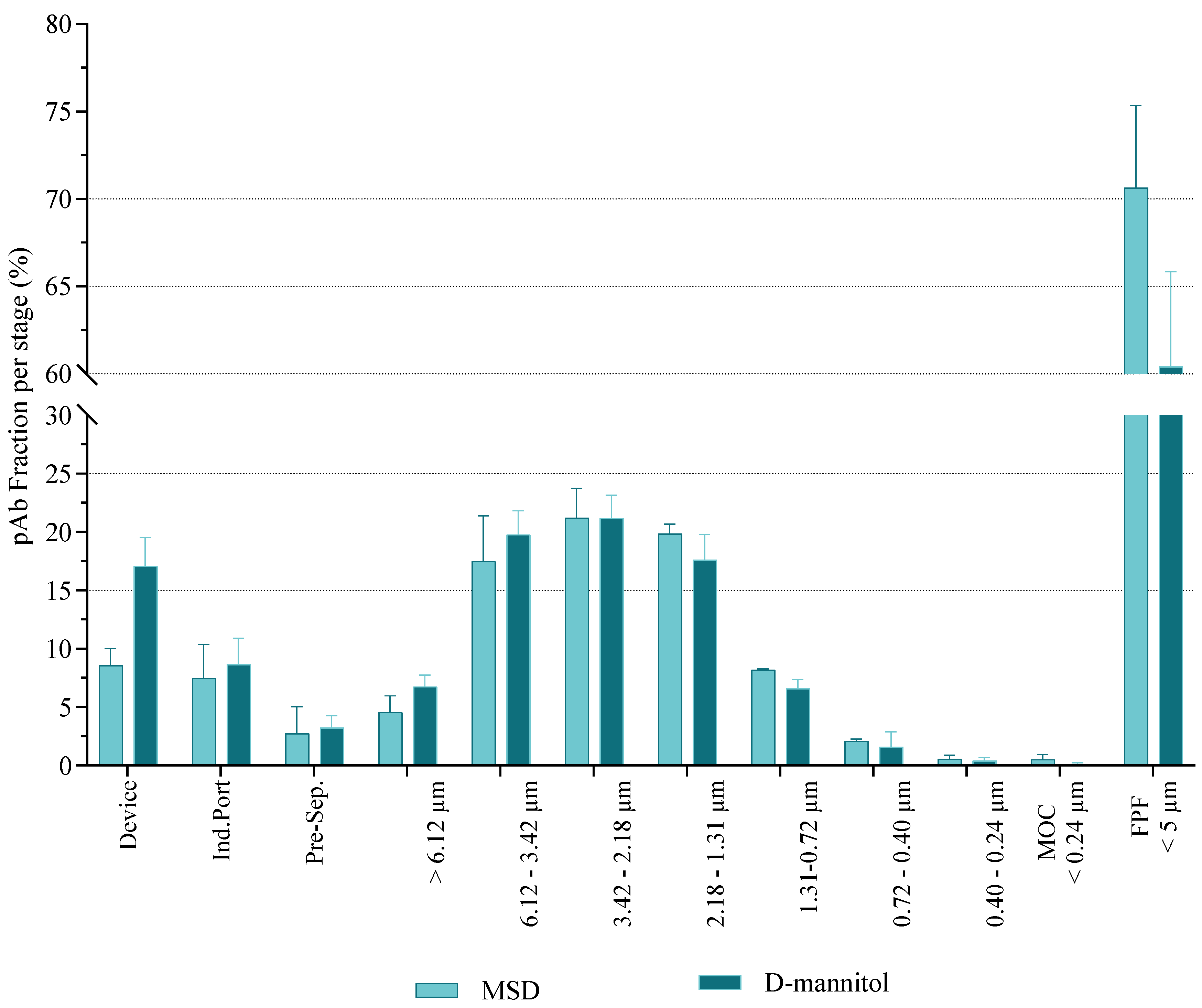
3.2.5. Aerodynamic Behavior of DPIs
- Aerosol particle size distribution - Laser diffraction technique
- Aerosol particle size distribution - Next generation impactor-based assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
| ANOVA | Analysis of variance |
| AUC | Area under the curve |
| BCA | Bicinchoninic acid |
| BEH | Bridged Ethylene Hybrid |
| BGG | Bovine gamma globulin |
| D | Dextran 10 kDa |
| Dgeo | Geometric diameter |
| DoE | Design of experiments |
| DPI | Dry powder for inhalation |
| DPI-MD | DPI including D-mannitol and dextran 10 kDa |
| DPI-SD | DPI including D-sucrose and dextran 10 kDa |
| DPI-MS | DPI including D-mannitol and D-sucrose |
| DPI-MSD | DPI including D-mannitol, D-sucrose, and dextran 10 kDa |
| FDA | Food and Drug Administration |
| FPFn | Fine particle fraction related to the nominal dose |
| GSD | Geometric standard deviation |
| H-bond | Hydrogen bond |
| HOA | High-order aggregates |
| LOA | Low-order aggregates |
| M | D-mannitol |
| mAbs | Monoclonal antibodies |
| MDSC | Modulated differential scanning calorimetry |
| MMAD | Median mass aerodynamic diameter |
| mw | Molecular weight |
| NGI | Next generation impactor |
| pAb | polyclonal IgG |
| PBS | Phosphate buffered saline |
| RM | Residual moisture |
| RT | Room temperature |
| S | D-sucrose |
| SEC | Size exclusion chromatography |
| SEM | Scanning electron microscopy |
| T0 | Time of analysis right after spray-drying |
| T6 | Time of analysis after a 6-month storage |
| T10 | Time of analysis after a 10-month storage |
| Tg | glassy transition temperature |
| XRPD | X-ray powder diffraction |
References
- Kohler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256(5517), 495–497. [Google Scholar] [CrossRef]
- Rajewsky, K. Clonal selection and learning in the antibody system. Nature 1996, 381(6585), 751–758. [Google Scholar] [CrossRef]
- Bourel, D.; Teillaud, J. L. Monoclonal antibodies: technology around the clock for new therapeutic hopes. C R Biol 2006, 329(4), 217–227. [Google Scholar] [CrossRef]
- Bogard, W. C., Jr.; Dean, R. T.; Deo, Y.; Fuchs, R.; Mattis, J. A.; McLean, A. A.; Berger, H. J. Practical considerations in the production, purification, and formulation of monoclonal antibodies for immunoscintigraphy and immunotherapy. Semin Nucl Med 1989, 19(3), 202–220. [Google Scholar] [CrossRef]
- Le Basle, Y.; Chennell, P.; Tokhadze, N.; Astier, A.; Sautou, V. Physicochemical Stability of Monoclonal Antibodies: A Review. J Pharm Sci 2020, 109(1), 169–190. [Google Scholar] [CrossRef] [PubMed]
- Elgundi, Z.; Reslan, M.; Cruz, E.; Sifniotis, V.; Kayser, V. The state-of-play and future of antibody therapeutics. Adv Drug Deliv Rev 2017, 122, 2–19. [Google Scholar] [CrossRef]
- Carpenter, J. F.; Randolph, T. W.; Jiskoot, W.; Crommelin, D. J.; Middaugh, C. R.; Winter, G.; Fan, Y. X.; Kirshner, S.; Verthelyi, D.; Kozlowski, S.; et al. Overlooking subvisible particles in therapeutic protein products: gaps that may compromise product quality. J Pharm Sci 2009, 98(4), 1201–1205. [Google Scholar] [CrossRef]
- Sreenivasan, S.; Schoneich, C.; Rathore, A. S. Aggregation of therapeutic monoclonal antibodies due to thermal and air/liquid interfacial agitation stress: Occurrence, stability assessment strategies, aggregation mechanism, influencing factors, and ways to enhance stability. Int J Pharm 2024, 666, 124735. [Google Scholar] [CrossRef] [PubMed]
- Emami, F.; Keihan Shokooh, M.; Mostafavi Yazdi, S. J. Recent progress in drying technologies for improving the stability and delivery efficiency of biopharmaceuticals. J Pharm Investig 2023, 53(1), 35–57. [Google Scholar] [CrossRef] [PubMed]
- S.J., S. Stability of monoclonal antibodies (mAbs); Elsevier, 2015. [Google Scholar] [CrossRef]
- Maurício, A. C.; Domingos, J.; Santos, D.; Sencadas, V.; Gomes, P.; Fernandes, M. H. Spray Drying: An Overview. In Biomaterials - Physics and Chemistry - New Edition; Pignatello, R., Musumeci, T., Eds.; IntechOpen, 2017. [Google Scholar]
- Depreter, F.; Pilcer, G.; Amighi, K. Inhaled proteins: challenges and perspectives. Int J Pharm 2013, 447(1-2), 251–280. [Google Scholar] [CrossRef]
- Levy, M. L.; Carroll, W.; Izquierdo Alonso, J. L.; Keller, C.; Lavorini, F.; Lehtimaki, L. Understanding Dry Powder Inhalers: Key Technical and Patient Preference Attributes. Adv Ther 2019, 36(10), 2547–2557. [Google Scholar] [CrossRef]
- Pilcer, G.; Amighi, K. Formulation strategy and use of excipients in pulmonary drug delivery. Int J Pharm 2010, 392(1-2), 1–19. [Google Scholar] [CrossRef]
- Arakawa, T.; Prestrelski, S. J.; Kenney, W. C.; Carpenter, J. F. Factors affecting short-term and long-term stabilities of proteins. Adv Drug Deliv Rev 2001, 46(1-3), 307–326. [Google Scholar] [CrossRef] [PubMed]
- FDA. Drug approval package for Exubera . 1 27 2006. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2006/021868_exubera_toc.cfm (accessed on 21 October 2025).
- FDA. Drug Approval Package Trelstar . 03 10 2010. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022437_trelstar_toc.cfm (accessed on 21 October 2025).
- FDA. Drug Approval Package of Somatuline Depot . 08 30 2007. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2007/022074s000TOC.cfm (accessed on 21 October 2025).
- FDA. Raplixa . 2015. Available online: https://www.fda.gov/vaccines-blood-biologics/approved-blood-products/raplixa (accessed on 21 October 2025).
- Donthi, M. R.; Butreddy, A.; Saha, R. N.; Kesharwani, P.; Dubey, S. K. Leveraging spray drying technique for advancing biologic product development–A mini review. Health Sciences Review 2024, 10, 100142. [Google Scholar] [CrossRef]
- Pinto, J. T.; Faulhammer, E.; Dieplinger, J.; Dekner, M.; Makert, C.; Nieder, M.; Paudel, A. Progress in spray-drying of protein pharmaceuticals: Literature analysis of trends in formulation and process attributes. Drying Technology 2021, 39(11), 1415–1446. [Google Scholar] [CrossRef]
- Chang, L. L.; Pikal, M. J. Mechanisms of protein stabilization in the solid state. J Pharm Sci 2009, 98(9), 2886–2908. [Google Scholar] [CrossRef]
- Carpenter, J. F.; Crowe, J. H. An infrared spectroscopic study of the interactions of carbohydrates with dried proteins. Biochemistry 1989, 28(9), 3916–3922. [Google Scholar] [CrossRef]
- Karunnanithy, V.; Abdul Rahman, N. H. B.; Abdullah, N. A. H.; Fauzi, M. B.; Lokanathan, Y.; Min Hwei, A. N.; Maarof, M. Effectiveness of Lyoprotectants in Protein Stabilization During Lyophilization. Pharmaceutics 2024, 16(10). [Google Scholar] [CrossRef] [PubMed]
- Information, N. C. f. B. PubChem Compound Summary for CID 6251, Mannitol . 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/6251 (accessed on 21 October 2025).
- Kim, A. I.; Akers, M. J.; Nail, S. L. The physical state of mannitol after freeze-drying: effects of mannitol concentration, freezing rate, and a noncrystallizing cosolute. J Pharm Sci 1998, 87(8), 931–935. [Google Scholar] [CrossRef]
- FDA. US Package Insert NDA 21-868 Exubera . Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2006/021868lbl.pdf (accessed on 21 October 2025).
- Larsen, B. S.; Skytte, J.; Svagan, A. J.; Meng-Lund, H.; Grohganz, H.; Lobmann, K. Using dextran of different molecular weights to achieve faster freeze-drying and improved storage stability of lactate dehydrogenase. Pharm Dev Technol 2019, 24(3), 323–328. [Google Scholar] [CrossRef]
- Vodak, D.; Dobry, D.E.; Friesen, D.; et al. Dextran-based materials as excipients in engineered particle formulations: Tailoring Physical Properties to optimize performance, manufacturability, and safety. RDD Europe, 2011. [Google Scholar]
- Information, N. C. f. B. PubChem Compound Summary for CID 5988, Sucrose . 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/5988#section=Non-Human-Toxicity-Excerpts (accessed on 21 October 2025).
- Pilcer, G.; Vanderbist, F.; Amighi, K. Correlations between cascade impactor analysis and laser diffraction techniques for the determination of the particle size of aerosolised powder formulations. Int J Pharm 2008, 358(1-2), 75–81. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, S.; Kennedy, R. O.; Tully, E. IMMUNOASSAYS | Production of Antibodies; 2005. [Google Scholar]
- Cicerone, M. T.; Douglas, J. F. β-Relaxation governs protein stability in sugar-glass matrices. Soft Matter 2012, 8(10), 2983–2991. [Google Scholar] [CrossRef]
- Wang, S.; Liu, A. P.; Yan, Y.; Daly, T. J.; Li, N. Characterization of product-related low molecular weight impurities in therapeutic monoclonal antibodies using hydrophilic interaction chromatography coupled with mass spectrometry. J Pharm Biomed Anal 2018, 154, 468–475. [Google Scholar] [CrossRef]
- Information, N. C. f. B. PubChem Compound Summary for CID 6274, L-Histidine . 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Histidine (accessed on 21 October 2025).
- Rowe, R. C.; Sheskey, P. J.; Owen, S. C.; Association, A. P. Handbook of Pharmaceutical Excipients; Pharmaceutical Press, 2009. [Google Scholar]
- Stadtman, E. R. Metal ion-catalyzed oxidation of proteins: biochemical mechanism and biological consequences. Free Radic Biol Med 1990, 9(4), 315–325. [Google Scholar] [CrossRef]
- Stadtman, E. R.; Levine, R. L. Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 2003, 25(3-4), 207–218. [Google Scholar] [CrossRef]
- Shetty, N.; Cipolla, D.; Park, H.; Zhou, Q. T. Physical stability of dry powder inhaler formulations. Expert Opin Drug Deliv 2020, 17(1), 77–96. [Google Scholar] [CrossRef]
- Mensink, M. A.; Frijlink, H. W.; van der Voort Maarschalk, K.; Hinrichs, W. L. How sugars protect proteins in the solid state and during drying (review): Mechanisms of stabilization in relation to stress conditions. Eur J Pharm Biopharm 2017, 114, 288–295. [Google Scholar] [CrossRef]
- Duerkop, M.; Berger, E.; Durauer, A.; Jungbauer, A. Impact of Cavitation, High Shear Stress and Air/Liquid Interfaces on Protein Aggregation. Biotechnol J 2018, 13(7), e1800062. [Google Scholar] [CrossRef] [PubMed]
- Cicerone, M. T.; Pikal, M. J.; Qian, K. K. Stabilization of proteins in solid form. Adv Drug Deliv Rev 2015, 93, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Moorthy, B. S.; Iyer, L. K.; Topp, E. M. Characterizing Protein Structure, Dynamics and Conformation in Lyophilized Solids. Curr Pharm Des 2015, 21(40), 5845–5853. [Google Scholar] [CrossRef]
- Cicerone, M. T.; Soles, C. L. Fast dynamics and stabilization of proteins: binary glasses of trehalose and glycerol. Biophys J 2004, 86(6), 3836–3845. [Google Scholar] [CrossRef] [PubMed]
- Rupley, J. A.; Careri, G. Protein hydration and function. Adv Protein Chem 1991, 41, 37–172. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, A.; Park, W. W.; Kwon, O. H. Hydrogen-Bond Dynamics and Energetics of Biological Water. Chempluschem 2020, 85(12), 2657–2665. [Google Scholar] [CrossRef] [PubMed]

| DPI type | Polyclonal IgG (%) | Buffer (%) | Mannitol (%) | D-sucrose (%) | Dextran 10kDa (%) |
DPI Yield (%) |
| M | 60.7 (90%)a | 32.5 | 6.8 (10%) a 100%b |
- | - | 71.6 |
| S | 60.8 (90%)a | 32.3 | - | 6.8 (10%) a 100% b |
- | 79.1 |
| D | 61.0 (90%) a | 32.2 | - | - | 6.8 (10%) a 100% b |
75.6 |
| MS | 60.7 (90%) a | 32.4 | 3.4 (5%) a 50% b |
3.4 (5%) a 50% b |
- | 76.1 |
| MD | 60.8 (90%) a | 32.4 | 3.4 (5%) a 50% b |
- | 3.4 (5%) a 50% b |
76.6 |
| SD | 60.7 (90%) a | 32.3 | - | 3.4 (5%) a 50% b |
3.4 (5%) a 50% b |
79.6 |
| MSD | 60.9 (90%) a | 32.3 | 2.3 (3.3%) a 33% b |
2.3 (3.3%) a 33% b |
2.3 (3.3%) a 33% b |
75.3 |
| DPI | HOA (%) | LOA (%) | ||
| T0 | T10 | T0 | T10 | |
| S | 7 ± 2 | 3 ± 3 | 4.0 ± 2.0 | 4.07 ± 0.06 |
| M | 1 ± 5 | 2 ± 5 | 2.3 ± 0.9 | 3.50 ± 0.60 |
| D | 1 ± 1 | -2 ± 3 | 2.3 ± 0.4 | 5.20 ± 0.20 |
| MS | 3 ± 0 | 2 ± 3 | 1.6 ± 0.6 | 2.00 ± 0.30 |
| SD | 5 ± 2 | -1 ± 2 | 0.8 ± 0.3 | 2.80 ± 0.30 |
| MD | 3 ± 3 | -1 ± 2 | 0.2 ± 0.4 | 1.50 ± 0.30 |
| MSD | 2 ± 0 | 1 ± 1 | -0.6 ± 0.6 | 3.60 ± 1.20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




