Submitted:
18 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
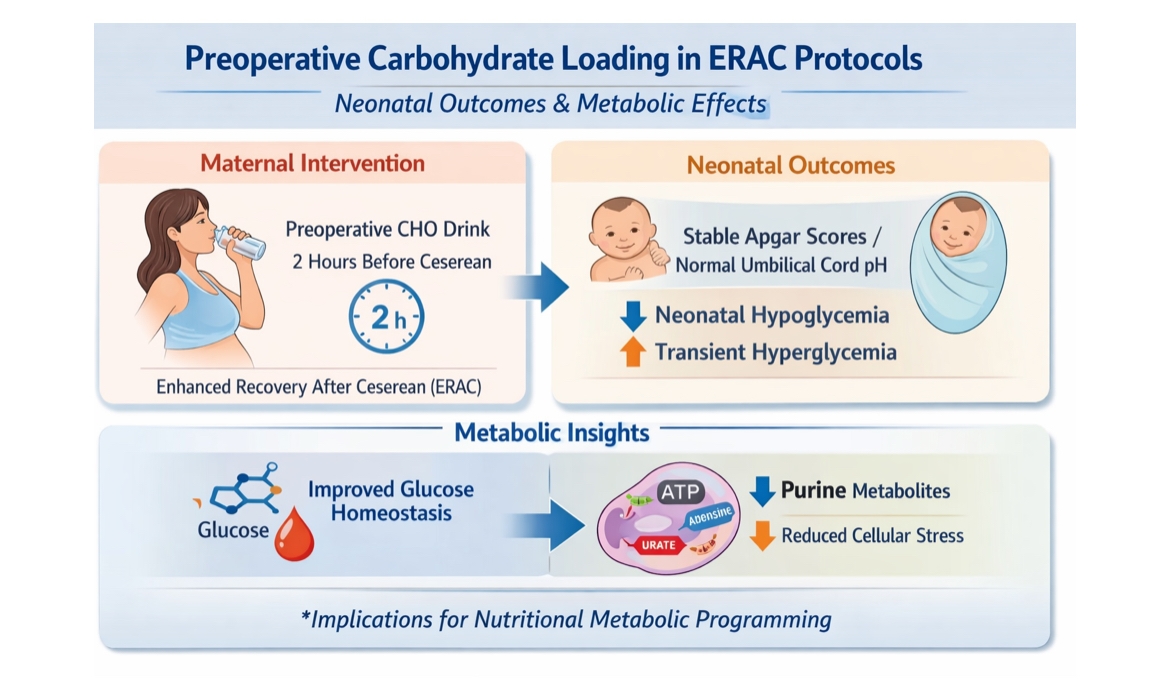
Keywords:
1. Introduction
2. The Rationale for Carbohydrate Loading: More Than Just a Drink [5]
| Domain | Effect of CHO loading | Strength of evidence | Clinical implication |
|---|---|---|---|
| Apgar score | No significant change | High | Confirms immediate neonatal safety |
| Umbilical cord arterial pH | Minimal differences within physiological range | Moderate | No risk of clinically relevant acidosis |
| Neonatal hypoglycemia | Significant reduction | High | Main clinical benefit |
| Neonatal hyperglycemia | Increased transient episodes | Moderate | Benign, self-limiting |
| Cellular metabolic stress | Reduced purinergic metabolites | Emerging | Suggests improved neonatal energy balance |
| Maternal comfort | Improved vs fasting | High | Better perioperative experience |
| Comparison with liberal fluids (STS) | Limited additional comfort benefit | Moderate | Organizational decision depends on metabolic priorities |
| Study (Year) | Design | Population | Intervention | Main neonatal outcomes | Key findings |
|---|---|---|---|---|---|
| Kotfis et al. (2023) | Randomized controlled trial | Elective cesarean delivery | CHO drink 2 h pre-op | Apgar, cord pH, neonatal glucose | No differences in Apgar or pH; reduced hypoglycemia |
| Shi et al. (2024) | Meta-analysis | Elective CD | CHO vs fasting/placebo | Apgar, maternal comfort | No adverse neonatal effects |
| Ding et al. (2022) | Randomized controlled trial | Women undergoing CD | Carbohydrate-rich beverage | Neonatal hypoglycemia | Significant reduction in hypoglycemia (RR 0.45) |
| Zhou et al. (2024) | Prospective study | Gestational diabetes | ERAC with CHO loading | Neonatal glucose homeostasis | Marked reduction in hypoglycemia |
| Sadiq et al. (2025) | Randomized controlled trial | Elective CD | CHO vs Sip-til-Send | Maternal comfort, nausea | Metabolic benefits but higher intraoperative nausea |
3. Maternal Benefits (Concise Overview)
Improved Patient Comfort
4. Maternal Comfort: CHO Loading vs. Fasting
Safety Profile
Key Takeaways
5. Analysis of Apgar Scores
6. Deep Dive into Umbilical Cord Arterial pH
6.1. The Nuance of “Significance”
6.2. Exploring Confounding Factors
6.3. Overall Conclusion on pH
7. Key Takeaways
8. Beyond Apgar and pH: The Broader Metabolic Picture for the Neonate
9. Neonatal Glucose Homeostasis: The Key Trade-Off
9.1. Reducing Neonatal Hypoglycemia: A Major Clinical Benefit
9.2. The Risk of Transient Hyperglycemia
10. Deeper Metabolic Insights: The Purinergic Pathway
11. Key Takeaways
12. The Evolving Landscape: Controversies, Gaps, and Future Directions
13. Research Limitations and Future Needs
14. Conclusion: A Balanced Verdict on Carbohydrate Loading for Cesarean Delivery
15. Final Clinical Takeaway
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patel K, Zakowski M. Enhanced recovery after cesarean: Current and emerging trends. Curr Anesthesiol Rep 2021;11:136–44. [CrossRef]
- Brown JK, Singh K, Dumitru R, Chan E, Kim MP. The benefits of enhanced recovery after surgery programs and their application in cardiothoracic surgery. Methodist Debakey Cardiovasc J 2018;14:77–88. [CrossRef]
- Betran AP, Ye J, Moller AB, Souza JP, Zhang J. Trends and projections of caesarean section rates: global and regional estimates. BMJ Glob Health 2021;6:e005671. [CrossRef]
- Chien P. Global rising rates of caesarean sections. BJOG 2021;128:781–2. [CrossRef]
- Echeverria-Villalobos M, Stoicea N, Todeschini AB, Fiorda-Diaz J, Uribe AA, Weaver T, et al. Enhanced Recovery After Surgery (ERAS): postoperative pain management and opioid crisis. Clin J Pain 2020;36:219–26.
- De Simone B, Sartelli M, Coccolini F, Ball CG, Brambillasca P, Chiarugi M, et al. Intraoperative surgical site infection control. World J Emerg Surg 2020;15:10.
- Peahl AF, Smith R, Johnson TRB, Morgan DM, Pearlman MD. Why obstetricians must implement enhanced recovery after cesarean. Am J Obstet Gynecol 2019;221:117.e1–7. [CrossRef]
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. PRISMA 2020 statement. BMJ 2021;372:n71.
- Umar TP, Samudra MG, Nashor KMN, Agustini D, Syakurah RA. Volunteering during COVID-19: systematic review. Front Med 2022;9:797153.
- Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan app for systematic reviews. Syst Rev 2016;5:210.
- Liana P, Liberty IA, Murti K, Hafy Z, Salim EM, Zulkarnain M, et al. NETs in COVID-19: systematic review. Immunol Res 2022;70:449–60.
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2 tool. BMJ 2019;366:l4898.
- Sterne JAC, Sutton AJ, Ioannidis JPA, Terrin N, Jones DR, Lau J, et al. Funnel plot asymmetry. BMJ 2011;343:d4002. [CrossRef]
- Wan X, Wang W, Liu J, Tong T. Estimating mean and SD. BMC Med Res Methodol 2014;14:135.
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Inconsistency in meta-analyses. BMJ 2003;327:557–60. [CrossRef]
- Birchall CL, Maines JL, Kunselman AR, Stetter CM, Pauli JM. Enhanced recovery for cesarean delivery. J Matern Neonatal Med 2022;35:10253–61. [CrossRef]
- Felder L, Cao CD, Konys C, Weerasooriya N, Mercier R, Berghella V, et al. ERAS and disparities in pain management. Am J Perinatol 2022;39:1375–82.
- Mangala JK, Remadevi C, Loganathan P, R S, Gopukrishnan, Vasudevan A. ERAS in COVID era. J Obstet Gynaecol India 2021;71(Suppl 1):12–7.
- McCoy JA, Gutman S, Hamm RF, Srinivas SK. ERAC pathway and opioid use. Am J Perinatol 2021;38:1341–7.
- Mullman L, Hilden P, Goral J, Gwacham N, Tauro C, Spinola K, et al. Improved outcomes with ERAC. Obstet Gynecol 2020;136:685–91.
- Shinnick JK, Ruhotina M, Has P, Kelly BJ, Brousseau EC, O’Brien J, et al. ERAS and LOS reduction. Am J Perinatol 2021;38(S01):e215–23.
- Sordia Pineyro MO, Villegas-Cruz C, Hernandez-Bazaldua M, Pineyro-Cantu A, Gaston-Locsin T, Sordia-Hernandez LH. ERAS elective cesarean. Ginekol Pol 2023;94:141–5.
- Tamang T, Wangchuk T, Zangmo C, Wangmo T, Tshomo K. ERAS in Bhutan. BMC Pregnancy Childbirth 2021;21:637.
- Tanner LD, Chen HY, Chauhan SP, Sibai BM, Ghebremichael SJ. ERAS pre-post study. J Matern Fetal Neonatal Med 2022;35:9170–7.
- Tepper JL, Harris OM, Triebwasser JE, Ewing SH, Mehta AD, Delaney EJ, et al. ERAS and opioid reduction. Am J Perinatol 2023;40:945–52.
- Uyanıklar ÖÖ, Türk P, Aslan K, Aslan EK, Özden O, Gürlüer J, et al. HERMES study. Int J Gynaecol Obstet 2023;161:168–74. [CrossRef]
- Chiao SS, Razzaq KK, Sheeran JS, Forkin KT, Spangler SN, Knio ZO, et al. ERAS and neonatal outcomes. J Perinatol 2022;42:1283–7.
- Combs CA, Robinson T, Mekis C, Cooper M, Adie E, Ladwig-Scott E, et al. ERAC impact on opioid use. Am J Obstet Gynecol 2021;224:237–9.
- Grasch JL, Rojas JC, Sharifi M, McLaughlin MM, Bhamidipalli SS, Haas DM. ERAS and postoperative pain. AJOG Glob Rep 2023;3:100169.
- Gupta S, Gupta A, Baghel AS, Sharma K, Choudhary S, Choudhary V. ERAS vs traditional protocol. J Obstet Anaesth Crit Care 2022;12:28–33.
- Hedderson M, Lee D, Hunt E, Lee K, Xu F, Mustille A, et al. ERAS quality improvement. Obstet Gynecol 2019;134:511–9.
- Kleiman AM, Chisholm CA, Dixon AJ, Sariosek BM, Thiele RH, Hedrick TL, et al. Maternal outcomes after ERAS. Int J Obstet Anesth 2020;43:39–46.
- Lester SA, Kim B, Tubinis MD, Morgan CJ, Powell MF. ERAS and opioid use. Int J Obstet Anesth 2020;43:47–55.
- MacGregor CA, Neerhof M, Sperling MJ, Alspach D, Plunkett BA, Choi A, et al. Post-cesarean opioid use. Am J Perinatol 2021;38:637–42. [CrossRef]
- Baluku M, Bajunirwe F, Ngonzi J, Kiwanuka J, Ttendo S. ERAS vs standard care. Anesth Analg 2020;130:769–76.
- Klangprapan N, Narkwichean A, Luanpholcharoenchai J, Laosooksathit W. ERAS RCT. Thai J Obstet Gynaecol 2022;30:393–402.
- Pan J, Hei Z, Li L, Zhu D, Hou H, Wu H, et al. ERAS and acute pain. Ther Clin Risk Manag 2020;16:369–78.
- Teigen NC, Sahasrabudhe N, Doulaveris G, Xie X, Negassa A, Bernstein J, et al. ERAS and LOS RCT. Am J Obstet Gynecol 2020;222:372.e1–10.
- Macones GA, Caughey AB, Wood SL, Wrench IJ, Huang J, Norman M, et al. ERAS Society recommendations. Am J Obstet Gynecol 2019;221:247.e1–9.
- Chandra S, Nugroho AM, Agus H, Susilo AP. Low-dose bupivacaine trial. Anesthesiol Pain Med 2019;9:e91275.
- Liu ZQ, Du WJ, Yao SL. ERAS challenges for anesthesiologists. Chin Med J 2020;133:590–6.
- Ituk U, Habib AS. Enhanced recovery after cesarean delivery. F1000Research 2018;7:513. [CrossRef]
- Zangouei A, Zahraei SAH, Sabertanha A, Nademi A, Golafshan Z, Zangoue M. Ketamine and post-spinal headache. Anesthesiol Pain Med 2019;9:e97249.
- Meng X, Chen K, Yang C, Li H, Wang X. ERAS systematic review and meta-analysis. Front Med 2021;8:694385.
- Fay EE, Hitti JE, Delgado CM, Savitsky LM, Mills EB, Slater JL, et al. ERAS reduces stay and cost. Am J Obstet Gynecol 2019;221:349.e1–9.
- Supattanarungsi W, Chullapram T. Early vs delayed oral feeding. Thai J Obstet Gynaecol 2021;29:92–9.
- Ogbadua AO, Agida TE, Akaba GO, Akitoye OA, Ekele BA. Early oral feeding RCT. Niger J Surg 2018;24:6–11.
- Saad AF, Saoud F, Diken ZM, Hegde S, Kuhlmann MJ, Wen TS, et al. Early vs late feeding. Am J Perinatol 2016;33:415–9.
- Chiu AS, Jean RA, Hoag JR, Freedman-Weiss M, Healy JM, Pei KY. Opioid prescribing defaults. JAMA Surg 2018;153:1012–9.
- Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC opioid guideline. MMWR Recomm Rep 2022;71:1–95.
- Sultan P, Sharawi N, Blake L, Habib AS, Brookfield KF, Carvalho B. ERAC meta-analysis. Anaesth Crit Care Pain Med 2021;40:100935.
- Ting Ding1†, Chun-Mei Deng1†, Xiao-Feng Shen2, Yao-Wu Bai3, Xiao-Lan Zhang4, Ji-Ping Liu5, Li-Juan Yang6, Hai-Tao Yu7, Lei Xie8, Hong Chen9, Dong-Liang Mu1, Yuan Qu1, Hui-Xia Yang10, Ai-Rong Bao10, Sai-Nan Zhu11 and Dong-Xin Wang1,12. Effect of a carbohydrate-rich beverage on rate of cesarean delivery in primigravidae with epidural labor analgesia: a multicenter randomized trial.BMC Pregnancy and Childbirth (2022) 22:339. [CrossRef]
- Jin Zhou, Peizhen Zhang, Zhangmin Tan, Chuo Li, Lin Yao, Tiantian He, Hongyin Hou and Yuzhu YinEnhanced recovery after surgery in elective cesarean section patients with gestational diabetes mellitus does notlead to glucose-related maternal and neonatal complications.Front. Endocrinol. 15:1403754. [CrossRef]
- Katarzyna Kotfis, Arleta Wojciechowska, Małgorzata Zimny, Dominika Jamioł-Milc, Aleksandra Szylin ́ska, Sebastian Kwiatkowski, Karolina Kaim, Barbara Dołe ̨gowska, Ewa Stachowska, Maciej Zukowski, Maria Pankowiak, Andrzej Torbé and Paul Wischmeyer Preoperative Oral Carbohydrate (CHO) Supplementation Is Beneficial for Clinical and Biochemical Outcomes in Patients Undergoing Elective Cesarean Delivery under Spinal Anaesthesia—A Randomized Controlled TrialJ. Clin. Med. 2023, 12, 4978. [CrossRef]
- Rana Shoaib Sadiq, Cathriona Murphy and Thomas Drew, A carbohydrate loading fasting protocol versus ‘Sip til Send’: a randomised trial of two different fasting protocols at elective caesarean delivery. BMC Anesthesiology (2025) 25:495. [CrossRef]
- Katarzyna Kotfis, Dominika Jamioł-Milc, Arleta Wojciechowska, Aleksandra Szylińska, Arleta Drozd, Małgorzata Zimny, Barbara Dołęgowska, Sebastian Kwiatkowski, Violetta Dziedziejko and Krzysztof Safranow. Maternal and umbilical cord plasma purine concentrations after oral carbohydrate loading prior to elective Cesarean delivery under spinal anesthesia: a randomized controlled trial. BMC Pregnancy and Childbirth (2025) 25:556 . [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




