Submitted:
17 March 2026
Posted:
19 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Electrode Selection
2.2. Substrate
2.3. Research Apparatus
2.4. Research Еquipment and Measurement Methodology
2.4.1. Roughness and Thickness of the Coatings
2.4.2. Mechanical Properties of the Coatings
2.4.3. Corrosion Behaviour of the Coatings
2.4.4. Microstructure and Elemental Composition of the Layers
3. Results and Discussion
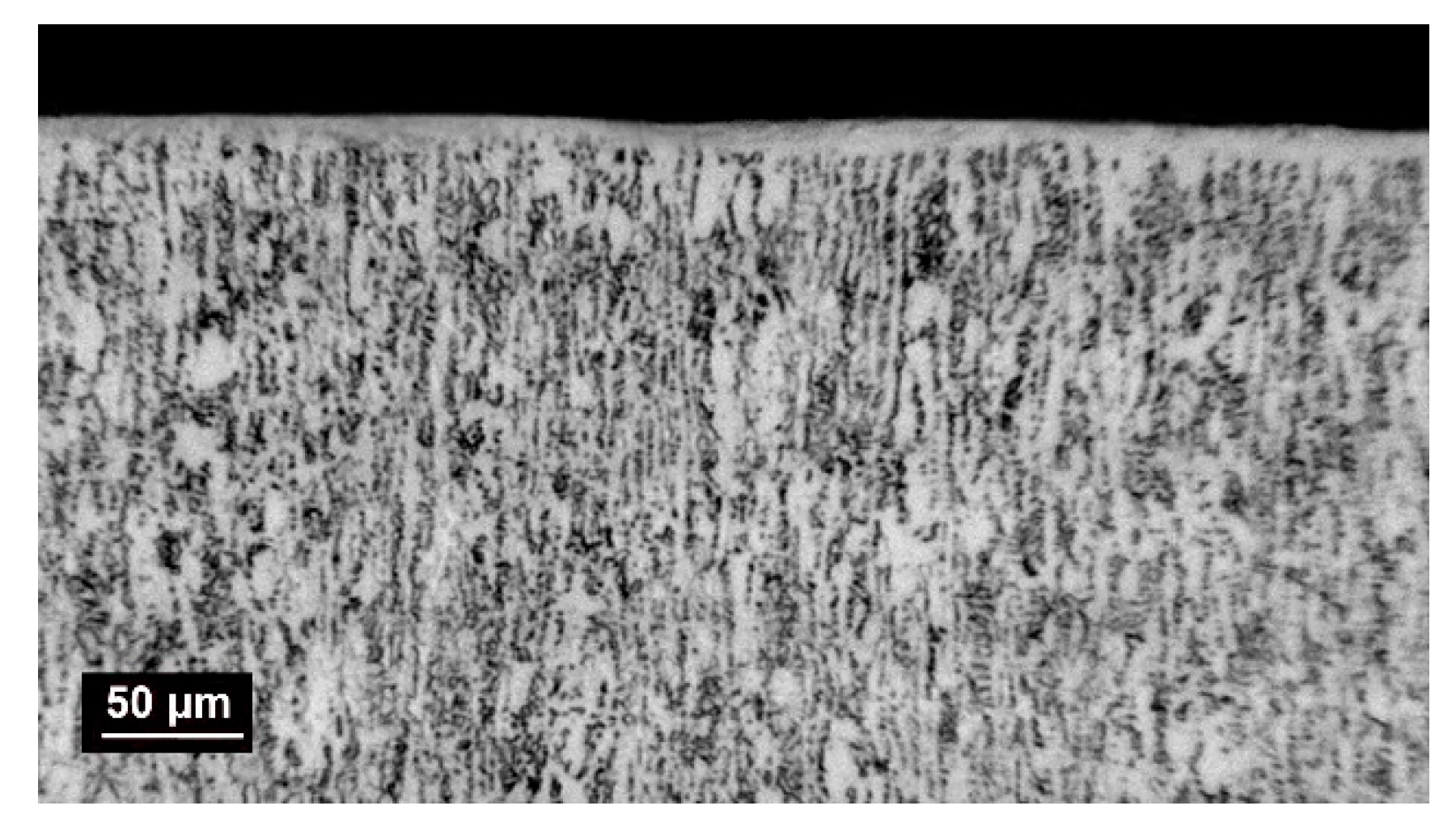
3.1. Coating Characterization –Roughness Ra and Thickness, Structure and Micro-Hardness of Coatings
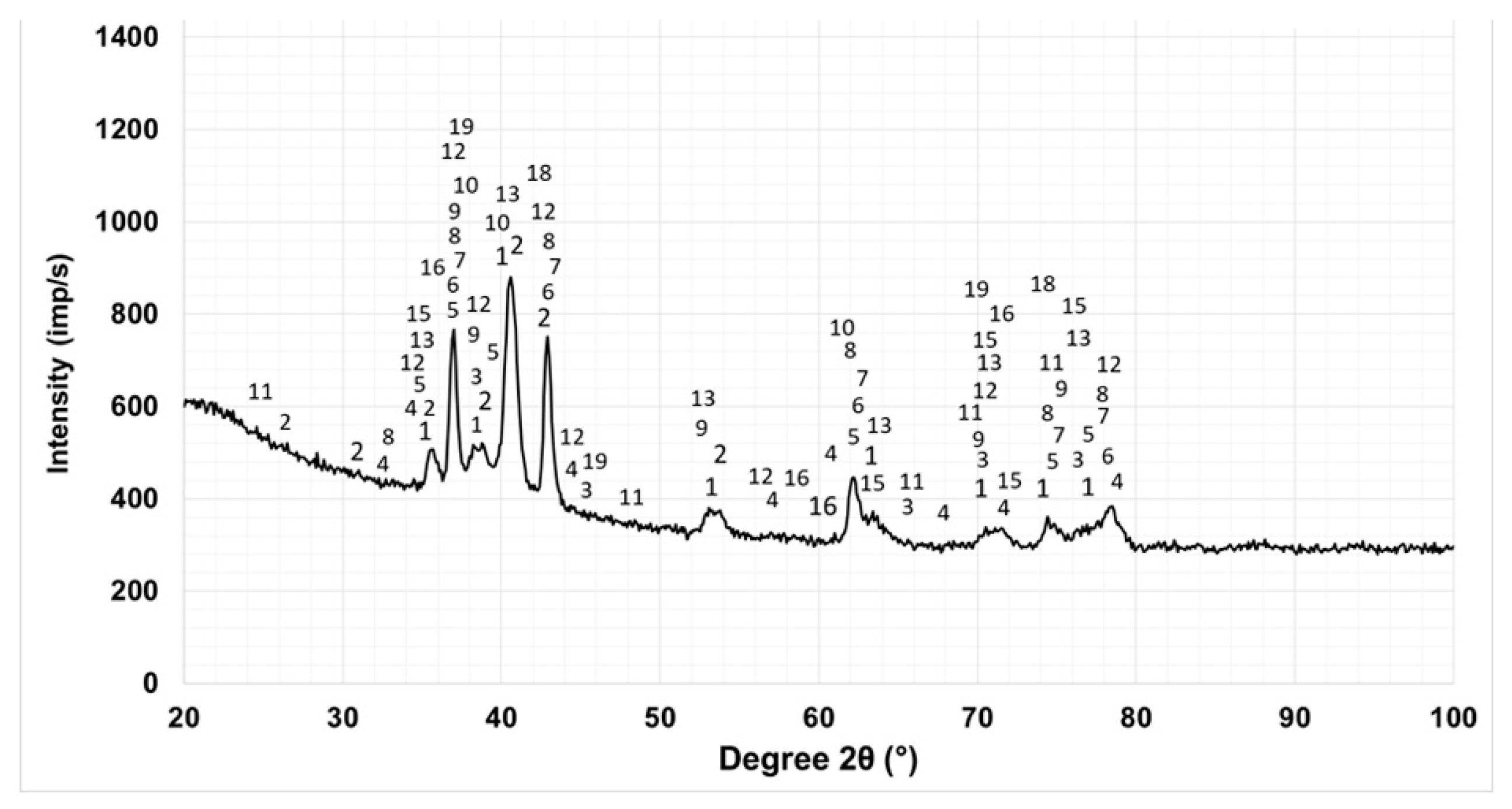
3.2. Phase Composition
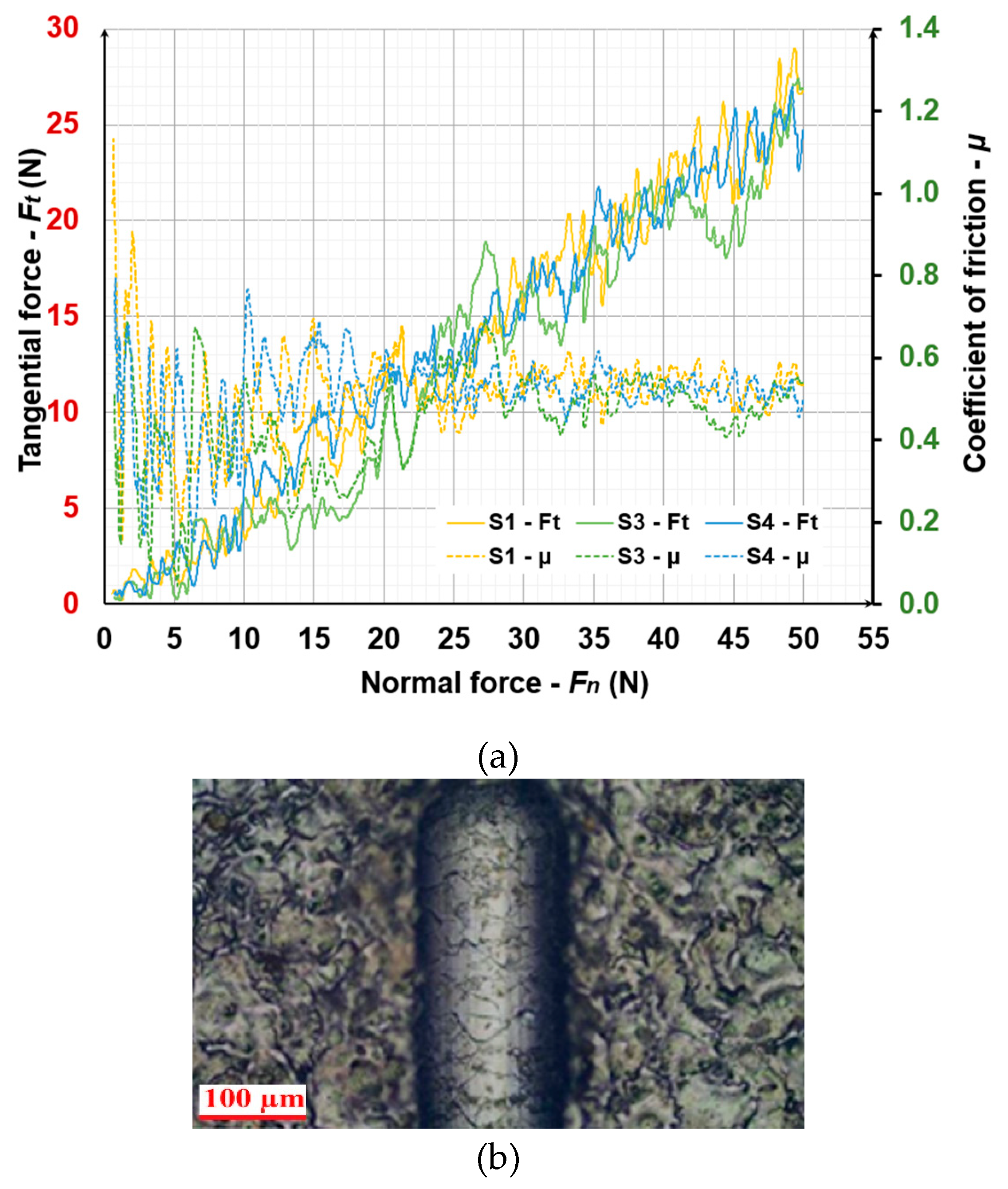
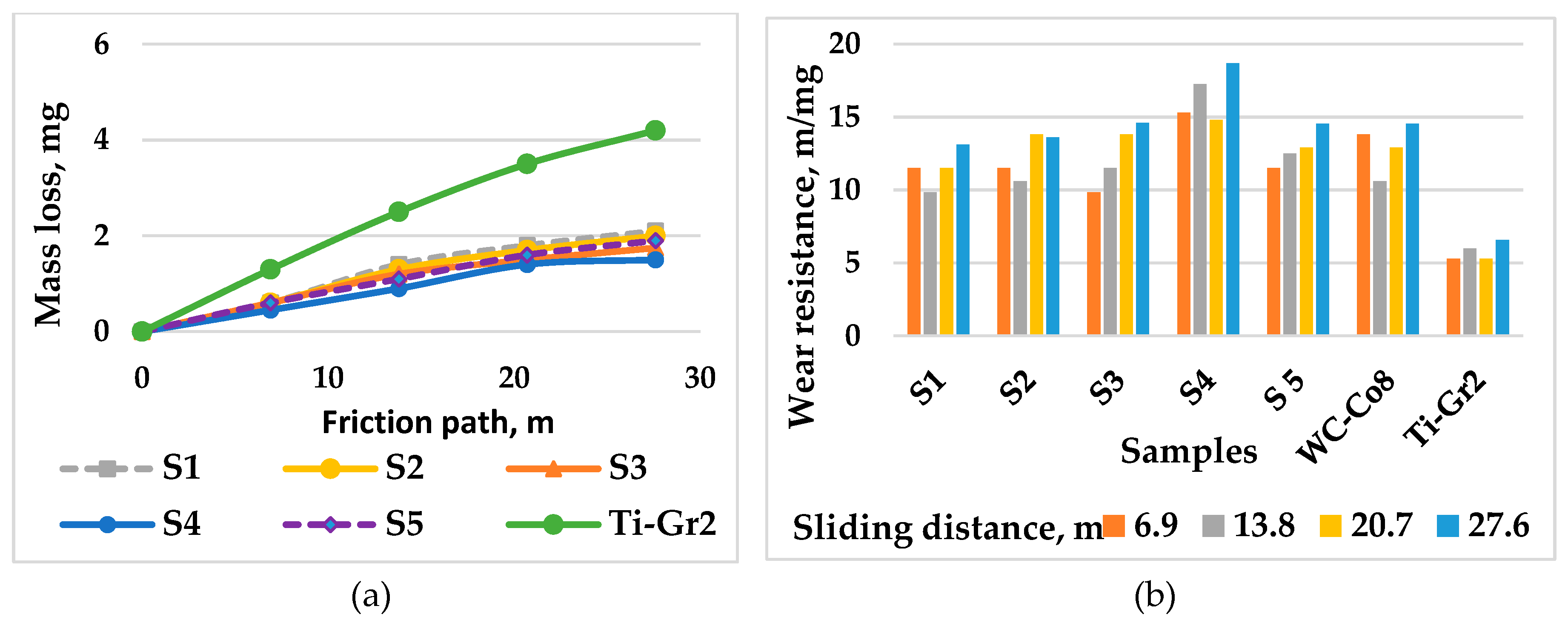
3.3. Tribological Tests
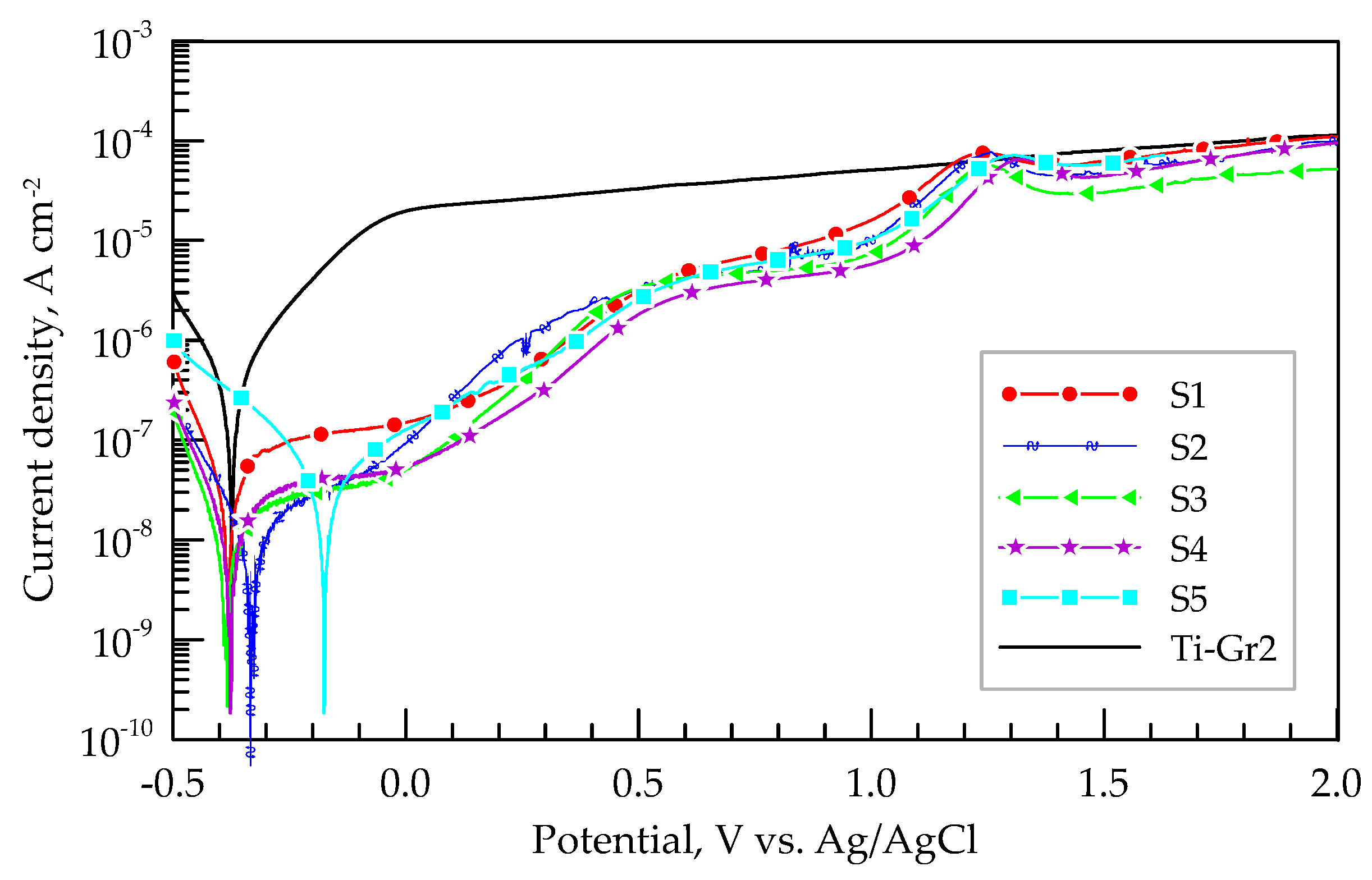
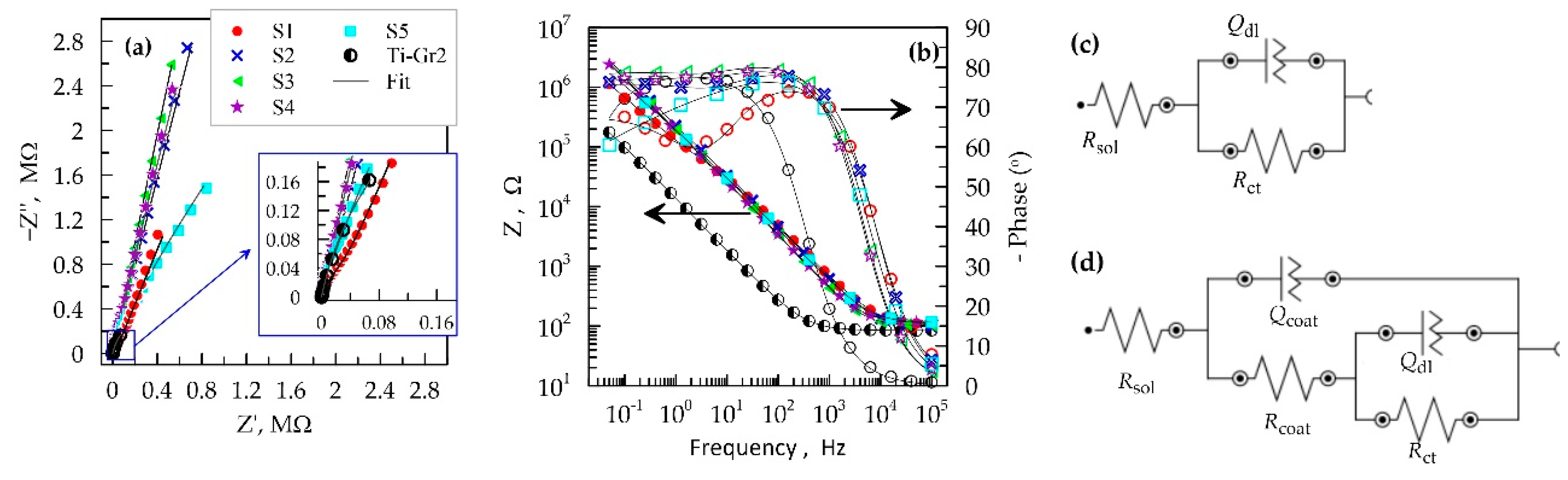
3.4. Corrosion Tests

4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petrică Vizureanu; Manuela-Cristina Perju; Dragoş-Cristian Achiţei; Carmen Nejneru. Advanced Electro- Spark Deposition Process on Metallic Alloys. Chapter 3 in Advanced Surface Engineering Research, 2018. [CrossRef]
- Zhang Zhengchuan; Liu Guanjun; I. Konoplianchenko; V. Tarelnyk; Ge Zhiqin; Du Xin. A Review of the Electro-Spark Deposition Technology. Bull Sumy Nat. Agr. Univ. Ser.: Mechanization and Automation of Production Processes, 2021, 2(44). [CrossRef]
- Wang, J.; Zhang, M.; Dai, S.; Zhu, L. Research Progress in Electrospark Deposition Coatings on Titanium Alloy Surfaces: A Short Review. Coating. 2023, 13, 1473. [Google Scholar] [CrossRef]
- Burkov, A.A. The effect of discharge pulse energy in electrospark deposition of amorphous coatings. Prot. Met. Phys. Chem. Surf. 2022, 58, 1018–1027. [Google Scholar] [CrossRef]
- Mulin, Y.I.; Verkhoturov, A.D.; Vlasenko, V.D. Electrospark alloying of surfaces of titanium alloys. Perspect. Mater. 2006, 1, 79–85. [Google Scholar]
- Mikhailov, V.V.; Gitlevich, A.; Verkhoturov, A.; Mikhailyuk, A.; Belyakov, A.; Konevtsov, L. Electrospark alloying of titanium and its alloys, physical and technological aspects and the possibility of practical use. Short review. Surf. Eng. Appl. Electrochem. 2013, 49(5), 373–395. [Google Scholar] [CrossRef]
- Podchernyaeva, I.; Juga, A.I.; Berezanskaya, V.I. Properties of electrospark coatings on titanium alloy. Key Eng. Mater. 1997, 132, 1507–1510. [Google Scholar] [CrossRef]
- Cadney, S.; Goodall, G.; Kim, G.; Moran, A.; Brochu, M. The transformation of an Al based crystalline electrode material to an amorphous deposit via the electrospark welding process. J. Alloys Compd. 2009, 476, 147–151. [Google Scholar] [CrossRef]
- Milligan, J.; Heard, D.W.; Brochu, M. Formation of nanostructured weldments in the Al-Si system using electrospark welding. Appl. Surf. Sci. 2010, 256, 4009–4016. [Google Scholar] [CrossRef]
- Burkov, A.A.; Chigrin, P.G. Synthesis of Ti–Al intermetallic coatings via electro spark deposition in a mixture of Ti and Al granules technique. Surf. Coat. Technol. 2020, 387, 125550. [Google Scholar] [CrossRef]
- Tarelnyk, V.B.; Gaponova, O.P.; Konoplyanchenko, I.V.; Yevtushenko, N.S.; Herasymenko, V.O. The Analysis of a Structural State of Surface Layer after Electroerosive Alloying. II. Features of Formation of Electroerosive Coatings on Special Steels and Alloys by Hard Wear-Resistant and Soft Antifriction Materials. Metallofiz. NoveishieTekhnol 2018, 40, 795–815, in Russian. [Google Scholar] [CrossRef]
- Burkov, A.A.; Konevtsov, L.A.; Khe, V.K. Formation of electric spark WC-Co coatings with modifying Cr2O3 additives. Letters on Materials 2022, 12(3), 237–242. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, D.; Deng, C.; Huo, L.; Wang, L.; Fang, R. Novel method to fabricate Ti–Al intermetallic compound coatings onTi–6Al–4V alloy by combined ultrasonic impact treatment and electrospark deposition. Journal of Alloys and Compounds 2015, 628, 208–212. [Google Scholar] [CrossRef]
- Riquelme, A.; Torres, B.; Rams, J.; Rodrigo, P. Process–microstructure–property relationships in Ti–Al alloys fabricated by laser directed energy deposition of Ti6Al4V and al powders. Prog Addit Manuf. 2026, 11, 1823–1842. [Google Scholar] [CrossRef]
- Kornienko, L.P.; Chernova, G.P.; Mihailov, V.V.; Gitlevich, A.E. Use of the electrospark alloying method to increase the corrosion resistance of a titanium surface. Surf. Engin. Appl. Electrochem. 2011, 47, 9–17. [Google Scholar] [CrossRef]
- Penyashki, T.G.; Kamburov, V.V.; Kostadinov, G.D.; Kandeva, M.K.; Dimitrova, R.B.; Nikolov, A.A. Possibilities and prospects for improving the tribological properties of titanium and its alloys by electrospark deposition Surf. Engin. Appl. Electrochem. 2022, 58(2), 135–146. [Google Scholar] [CrossRef]
- Hong, X.; Feng, K.; Tan, Y.F.; Wang, X.; Tan, H. Effects of process parameters on microstructure and wear resistance of TiN coatings deposited on TC11 titanium alloy by electro spark deposition. Trans. Nonferrous Met. Soc. China 2017, 27, 1767–1776. [Google Scholar] [CrossRef]
- Levashov, E.A.; Zamulaeva, A.E.; Kudryashov, A.E.; Vakaev, P.V.; Petrzhik, M.I.; Sanz, A. Materials science and technological aspects of electrospark deposition of nanostructured WC-Co coatings onto titanium substrates. Plasma Processes and Polymers 2007, 4(3), 293–300. [Google Scholar] [CrossRef]
- Teplenko, M.A.; Podchernyaeva, I.A.; Panasyuk, A.D. Structure and wear resistance of coatings on titanium alloy and steels obtained by electro spark alloying with AlN–ZrB2 material. Powder Metall. Met. Ceram. 2002, 41, 154–161. [Google Scholar] [CrossRef]
- Zamulaeva, E.I.; Kuptsov, K.A.; Petrzhik, M.I.; Mukanov, S.K.; Loginov, P.A.; Levashov, E.A. Electrospark Deposition of Hard, Wear-Resistant and Anti-Friction Coatings on γ-TiAl Substrates in a Controlled Gas Environment. Surf. Engin. Appl. Electrochem. 2024, 60, 728–741. [Google Scholar] [CrossRef]
- Manakova, O.S.; Kudryashov, A.E.; Levashov, E.A. On the application of dispersion–hardened SHS electrode materials based on (Ti, Zr)C carbide using electrospark deposition. Surf. Eng. Appl. Electrochem. 2015, 51, 413–421. [Google Scholar] [CrossRef]
- Kudryashov, A.E.; Levashov, E.A. Application of SHS-electrode materials in pulsed electrospark deposition technology, XV. In ternational Symposium on Self-Propagating High-Temperature Synthesis: Book of Abstracts; IPCP RAS: Chernogolovka, Russia, 2019; pp. 199–201. Available online: https://www.ism.ac.ru/events/SHS2019/doc/abstract_shs2019.pdf?utm_source=chatgpt.com (accessed on 24 December 2025).
- Levashov, E.A.; Vakaev, P.V.; Zamulaeva, E.I.; Kudryashov, A.E.; Kurbatkina, V.V.; Shtansky, D.V.; Voevodin, A.A.; Sanz, A. Disperse-strengthening by nanoparticles advanced tribological coatings and electrode materials for their deposition. Surf. Coat. Technol. 2007, 201, 6176–6181. [Google Scholar] [CrossRef]
- Levashov, E.A.; Malochkin, O.Y.; Kudryashov, A.E.; Glukhov, S.A.; Sviridova, T.A.; Gammel, F.; Zuhentrung, R. Influence of nano-sized powders on combustion processes and formation of composition, structure, and properties of alloys of the system Ti-Al-B. J. Non-Ferr. Met. 2003, 1, 54–59. [Google Scholar]
- Kudryashov, A.E.; Levashov, E.A.; Aksenov, L.B.; Petrov, V.M. Use of electric spark alloying technology and promising nanostructured electrode materials for improving the life of punching equipment. Metallurgist 2010, 54, 514–522. [Google Scholar] [CrossRef]
- Kudryashov, A.E.; Mukanov, S.K.; Sheveyko, A.N.; Rupasov, S.I.; Levashov, E.A. Peculiarities of formation of structure and properties of electrospark coatings on 20X13 steel using SHS-electrodes. Metallurgist 2025, 69, 871–881. [Google Scholar] [CrossRef]
- Kuptsov, K.A.; Sheveyko, A.N.; Mukanov, S.K.; Bazlov, A.I.; Zaitsev, A.A.; Shtansky, D.V. Effect of boron content and heat treatment on microstructural evolution and tribocorrosion behavior of HEA FeCrNiCoMo-Bx coatings. Materials Today Communications 2025, 48, 113342. [Google Scholar] [CrossRef]
- Lv, X.; Yin, Z.; Yang, Z.; Chen, J.; Zhang, S.; Song, S.; Yu, G. Review on the Development of Titanium Diboride Ceramics. Recent Progress in Materials. 2024, 6(2), 1–48. [Google Scholar] [CrossRef]
- Anuj Bansal , Vikrant Singh , Anil Kumar Singla , Jasdeep Singh , Jonny Singla , Rashad A. Al-Hammadi. Effect of TiB2-B4C reinforcement and laser remelting on the microstructural, mechanical, and tribological performance of HVOF sprayed WC-10Co-4Cr coating, Colloids and Surfaces A: Physicochemical and Engineering Aspects. 2026, 734, 139486. [CrossRef]
- Zhang, X.; Zhang, S.; Peng, Y.; Geng, L.; Kang, C.; Feng, Z.; Fan, W.; Tan, H.; Lin, X. Process Optimization, Microstructure and Mechanical Properties of SiC + TiB2/AlSi10Mg Composites Fabricated by Laser-Directed Energy Deposition. J. Manuf. Mater. Process. 2025, 9, 404. [Google Scholar] [CrossRef]
- Syrovatka, V. Obtaining composite powder materials and coatings based on titanium diboride. International Science Journal of Engineering & Agriculture 2023, 2(3), 10–17. [Google Scholar] [CrossRef]
- Kazamer, N.; Valean, P.C.; Pascal, D.; Serban, V.A.; Muntean, R.; Margineal, G. Microstructure and phase composition of NiCrBSi–TiB2 vacuum furnace fused flame-sprayed coatings. Published in IOP Conference Series Materials Science and Engineering Materials Science, Engineering, October 2018; 416, p. 139816788. [Google Scholar] [CrossRef]
- Burkov, A.A.; Kulik, M.A.; Bytsura, A.Y.; Krutikova, V.O. Electrospark deposition of boron carbide powder on titanium alloy Ti-6Al-4V. Letters on Materials 2025, 15(3), 169–175. [Google Scholar] [CrossRef]
- Storozhenko, M.; Umanskyi, O.; Tarelnyk, V.; Koval, O.; Gubin, Yu.; Mikulina, M.; Martsenyuk, I.; Kostenko, O.; Kurinna, T. Structure and Wear Resistance of FeNiCrBSiC–MeB2 Electrospark Coatings. Powder Metall Met Ceram. 2020, 59, 330–341. [Google Scholar] [CrossRef]
- Mesut Gökçe; Yusuf Kayali; Şükrü Talaş. The Ceramic Composite Coating (TiC+TiB2) by ESD on Ti6AL4V Alloy and Its Characterization, Ceramic Sciences and Engineering. 2020, 3(1). [CrossRef]
- Shafyei Hassan, H.; Salehi, M.; Bahrami, A. Fabrication, microstructural characterization and mechanical properties evaluation of Ti/TiB/TiB2 composite coatings deposited on Ti6Al4V alloy by electro–spark deposition method. Ceram. Int. 2020, 46(10), Part A, 15276– 15284. [CrossRef]
- Oksana Gaponova; Viacheslav Tarelnyk; Nataliia Tarelnyk; Gennadii Laponog. Investigation of Aluminum Electrospark Alloyed Coatings on Steels, Metallurgical and Materials Transactions A. 2025, 56, 4204-4229. [CrossRef]
- Kostadinov, G.; Danailov, P.; Dimitrova, R.; Kandeva, M.; Penyashki, T.; Kamburov, V.; Nikolov, A.; Elenov, Bl. Surface topography and roughness parameters of electrospark coatings on titanium and nickel alloys. Applied Engineering Letters 2021, 6(3), 89–98. [Google Scholar] [CrossRef]
- Penyashki, T.G.; Kostadinov, G.D.; Dimitrova, R.B.; Kamburov, V.V.; Kandeva, M.K.; Valkanov, S.T.; Nikolov, A.A.; Elenov, B.P. Improving Surface Properties of Titanium Alloys by Electrospark Deposition with Low Pulse Energy. Surface Engineering and Applied Electrochemistry 2022, 6, 580. [Google Scholar] [CrossRef]
- Penyashki, T.; Kostadinov, G.; Kandeva, M.; Nikolov, A.; Dimitrova, R.; Kamburov, V.; Danailov, P.; Bozhkov, S. Study of the Influence of Coating Roughness on the Properties and Wear Resistance of Electrospark Deposited Ti6Al4V Titanium Alloy. Tribology in Industry 2024, 46(1), 13–28. [Google Scholar] [CrossRef]
- Kostadinov, G.; Nikolov, А.; Sofronov, Y.; Penyashki, T.; Mishev, V.; Tzaneva, B.; Dimitrova, R.; Petrov, K.; Miltchev, R.; Gavrilov, T. Characteristics and Microstructure of Coatings of Ultradisperse TiB2-TiAl Electrodes with Nanosized Additives Deposited on Ti-Gr2 by Non-Contact Electrospark Deposition. Materials 2026, 19(3), 572. [Google Scholar] [CrossRef]
- Antonov, B. Device for Local Electric-Spark Layering of Metals and Alloys by Means of Rotating Electrode. U.S. Patent No. 3,832,514, 27 August 1974. [Google Scholar]
- Jönsson, B.; Hogmark, S. Hardness measurements of thin films. Thin Solid Film. 1984, 114, 257–269. [Google Scholar] [CrossRef]
- Felix, L.M.; Kwan, C.C.; Zhou, N.Y. The Effect of Pulse Energy on the Defects and Microstructure of Electro-Spark- Deposited Inconel 718. Metall. Mater. Trans. A 2019, 50(9), 4223–4231. [Google Scholar] [CrossRef]
- Hong, X.; Tan Y.; Zhou C.;H Xu, T., Z. Zhang, Microstructure and tribological properties of Zr–based amorphous nanocrystalline coatings deposited on the surface of titanium alloys by electrospark deposition. Appl. Surf. Sci., 2015, 356, 1244–1251. [CrossRef]
- Trotsky, M.; Petrovich, S.; Andreeva, V.; Popovich, A.; Zamozdra, M. Synthesis of Ceramics Titanium Compounds by Mechanical Alloying of TiB2-Tin Systems. Proceedings 30th Anniversary International Conference on Metallurgy and Materials, Brno, Czech Republic, May 26 - 28, 2021; pp. 824–828. [Google Scholar] [CrossRef]
- Koga, G.Y.; Bolfarini, C.; Kiminami, C.S.; Jorge, A.M.; Botta, W.J. An Overview of Thermally Sprayed Fe-Cr-Nb-B Metallic Glass Coatings: From the Alloy Development to the Coating’s Performance Against Corrosion and Wear. J Therm Spray Tech. 2022, 31, 923–955. [Google Scholar] [CrossRef]
- Brochu, M.; Portillo, J.G.; Milligan, J.; Heard, D.W. Development of Metastable Solidification Structures Using the Electrospark Deposition Process. The Open Surface Science Journal 2011, 3, 105–114. [Google Scholar] [CrossRef]
- Nianwei Dai, Lai-Chang Zhang, Junxi Zhang, Qimeng Chen, Maoliang Wu. Corrosion behavior of selective laser melted Ti-6Al-4V alloy in NaCl solution, Corrosion Science, 2016, 102, 484-489. [CrossRef]
- Norbert Radek, Izabela Pliszka, Aneta Gądek-Moszczak, Jarosław Rolek, Jana Petru, Properties of Carbide Electro-Spark Coatings After Laser Processing, AIP Conference Proceedings, 2018, 020024. [CrossRef]
- Socorro-Perdomo, P.P.; Florido-Suárez, N.R.; Mirza-Rosca, J.C.; Saceleanu, M.V. EIS Characterization of Ti Alloys in Relation to Alloying Additions of Ta. Materials, 2022, 15, 476. [CrossRef] [PubMed]

| Designation of samples | Current, I, A |
Capacitance C, μF |
Pulse duration Ti, μs |
Frequency f, kHz |
Pulse energy E, J |
|---|---|---|---|---|---|
| S1 | 16.0 | 4.4 | 12 | 8.33 | 0.025 |
| S2 | 11.2 | 0.5 | 12 | 8.33 | 0.013 |
| S3 | 12.8 | 0.5 | 8 | 12.5 | 0.013 |
| S4 | 16.0 | 2.0 | 12 | 8.33 | 0.020 |
| S5 | 12.8 | 4.4 | 8 | 12.5 | 0.020 |
| Sample | Ra, µm | Rz, µm | Rt, µm | δ, µm | HV, GPa |
|---|---|---|---|---|---|
| S1 | 3.36 | 10.94 | 15.93 | 19 | 13.5 |
| S2 | 2.07 | 7.53 | 9.05 | 11 | 11.9 |
| S3 | 1.63 | 6.39 | 8.65 | 9 | 11.2 |
| S4 | 2.45 | 9.05 | 11.66 | 16 | 13.2 |
| S 5 | 2.57 | 9.89 | 13.86 | 13 | 12.3 |
| WC-Co8 | 2.68 | 10.14 | 11.33 | 12 | 11.8 |
| Ti-Gr2 | 2.07 | 5.62 | 5.67 | 3.4 |
| N | Main phases | N | Phases in small amounts | N | Traces of phases |
|---|---|---|---|---|---|
| 1 | Ti ≈ 32.9 % | 7 | TiN ≈ 4.4 % | 14 | Al ≈ 0.8 % |
| 2 | AlTi3 ≈ 2.6 % | 8 | Ti3.2B1.6N2.4 ≈ 15 % | 15 | TiC0.7N0.3 ≈ 0.8 % |
| 3 | AlTi ≈ 1.8 % | 9 | Ti6O ≈ 3.3 % | 16 | AlN ≈ 2.1 % |
| 4 | TiB2 ≈ 4.5 % | 10 | Ti3O ≈ 1.7 % | 17 | AlB ≈ 0.9 % |
| 5 | TiN0.3 ≈ 5.3 % | 11 | Al3Ti ≈ 2.6 % | 18 | BN ≈ 1.1 % |
| 6 | TiB ≈ 8.8 %, | 12 | Al2O3 ≈ 2.5 % | 19 | Al2.86O3.45N0.55 ≈ 2.4% |
| 13 | Ti2O ≈3%, | 20 | TiC0.3N0.7 ≈ 3.5% |
| Phases | Ti | TiN0.3 | TiN | TiC1-x | TiCхNy | TiB | TiB2 | TiAl3 | Ti2O | Al2O3 | Ti₃.₂B₁.₆N₂.₄ | TiAl | Ti2Al |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Size, nm | 39 | 36 | 29 | 26 | 24 | 35 | 68 | 41 | 39 | 51 | 42 | 42 | 32 |
| Coating | α-Тi, | TiN0.3 | AlTi3 | TiB2 | Ti3.2B1.6N2.4 | Ti2O | Al2O3 |
|---|---|---|---|---|---|---|---|
| TiB2-TiAl-Phases,2θ | 35.7;38.8;40.7;53.5;63;71;75.4;76.8;78.6 | 35;37.5;39.5;52.2;62.5;69.275.5;77 | 26.33;31.12;35.65;38.9;39.4;40.8;43.05;53.8 | 27.7;33.38.8;34.2;44.6;56.9;61.1;68.5;72;78.6 | 33.36;37;42.95;62.25;74.5;78.5 | 33.6;35.65;38.45;40.7;53.2;63.75;70.5;76.6;77.15;78.5 | 19.5;32.1;35.65;37.6;39.2;43.05;44.5;45.6;50;56.7;60.5;66.8;71.4;75.3;78.55 |
|
S1, Lattice, Å crystallite size, nm |
a= 2.936 c= 4.652 32 |
а=2.969, с=4.783 45 |
a= 5.768 c=4.6424 46 |
а= 3.027 с=3.211 21 |
a= 4.23 Å 42 |
a= 2.9194 c= 4.7130 |
a= 4.6490 Å c= 12.6870 a= 7.955 76 |
|
S2, Lattice, Å crystallite size, nm |
а=2.949 с=4.684 27 |
а=2.956 с=4.77 36 |
a= 5.764 c= 4.664 36 |
а= 3.032 с=3.219 45 |
a= 4.2350 Å |
a= 2.9194 Å c= 4.7130 Å |
Gamma cubic a= 7.9320 30 a=7.947 54 |
|
S3, Lattice, Å crystallite size, nm |
а= 2.956, с=4.692 26 |
a=2.968, с=4.78 43 |
а= 2.96 Å, с=3.31 15 |
a= 4.2430 Å |
a=7.9581 a=7.954 58 a= 4.7154 c= 12.8510 35 |
||
|
S4, Lattice, Å crystallite size, nm |
а= 2.945, с=4.683 27 |
а=2.963, с=4.78 31 |
Hexagonal a= 5.768 c= 4.642 33 |
а= 2.96, с=3.32 21 |
a= 4.220 Å |
a= 2.9194 Å c= 4.7130 Å 39 |
Trigonal (hexagonal axes) a= 4.7540 Å c= 12.9820 Å 47 |
| Samples | Sliding distance, m | |||
|---|---|---|---|---|
| 6.9 | 13.8 | 20.7 | 27.6 | |
| Wear intensity, mg/m.10-2 | ||||
| S1 | 8.7 | 10 | 8.6 | 7.5 |
| S2 | 8.71 | 9.3 | 8 | 6.1 |
| S3 | 8.6 | 8.5 | 7.1 | 6.3 |
| S4 | 6.5 | 6.4 | 6.7 | 5.2 |
| S5 | 6.8 | 8.2 | 7.8 | 7.2 |
| Ti-Gr2 | 18.8 | 17.8 | 16.9 | 15.6 |
| WC-Co8 | 7.2 | 9.4 | 7.7 | 6.9 |
| Friction path, m / The durability enhancement factor; Wear, mg |
Uncoated Ti-Gr2 |
LESD with TiB2-TiAl | LESD with WC-Co8 |
||||
|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | S5 | |||
| 1.5 | 7.2/1 | 17/2.36 | 17.6/2.43 | 21/2.92 | 28/3.89 | 18.2/2.57 | 18.1/2.51 |
| 1 | 5.5/1 | 11/2 | 11.5/2.1 | 12/2.2 | 19/3.45 | 13/2.36 | 12/2.2 |
| 0.5 | 2.2/1 | 5/2.3 | 5.2/2.4 | 5.5/2.5 | 10/4.54 | 5.9/2.7 | 5.85/2.66 |
| Samples | OCP (600s), V | Ecorr, V | Jcorr, A/cm2 | jpass, A/cm2 | Rp, Ohm | CR, mm/year |
|---|---|---|---|---|---|---|
| S1 | -0.076 | -0.382 | 1.11 × 10-7 | - | 8.58 x 107 | 12.9 × 10-5 |
| S2 | -0.194 | -0.336 | 1.35 × 10-8 | 4.76 x 10-6 | 3.08 × 107 | 1.57 × 10-5 |
| S3 | -0.143 | -0.388 | 9.25 × 10-9 | 5.02 x 10-6 | 2.83 × 107 | 1.08 × 10-5 |
| S4 | -0.158 | -0.378 | 1.68 × 10-8 | 4.15 x 10-6 | 2.06 × 107 | 1.95 × 10-5 |
| S5 | -0.147 | -0.170 | 7.84 × 10-8 | 4.24 x 10-6 | 3.45 × 106 | 9.11 × 10-5 |
| Ti-Gr2 | -0.380 | -0.375 | 3.97 × 10-7 | - | 8.50 × 105 | 4.62 × 10-4 |
| Samples | Rsol, Ω | Rct, Ω | Qdl, µS sn | ndl | Rcoat, kΩ | Qcoat, µS sn | ncoat |
|---|---|---|---|---|---|---|---|
| S1 | 104 | 73.2 × 107 | 1.400 | 0.714 | 85.3 | 0.791 | 0.864 |
| S2 | 94.8 | 63.0 × 109 | 0.252 | 0.740 | 249 | 0.697 | 0.890 |
| S3 | 102 | 93.0 × 109 | 0.177 | 0.740 | 384 | 0.877 | 0.907 |
| S4 | 113 | 43.0 × 109 | 0.221 | 0.705 | 273 | 0.894 | 0.899 |
| S5 | 114 | 20.9 × 106 | 0.543 | 0.560 | 153 | 0.837 | 0.876 |
| Ti-Gr2 | 84.3 | 8.92 × 105 | 14.70 | 0.872 | – | – | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




